Abstract
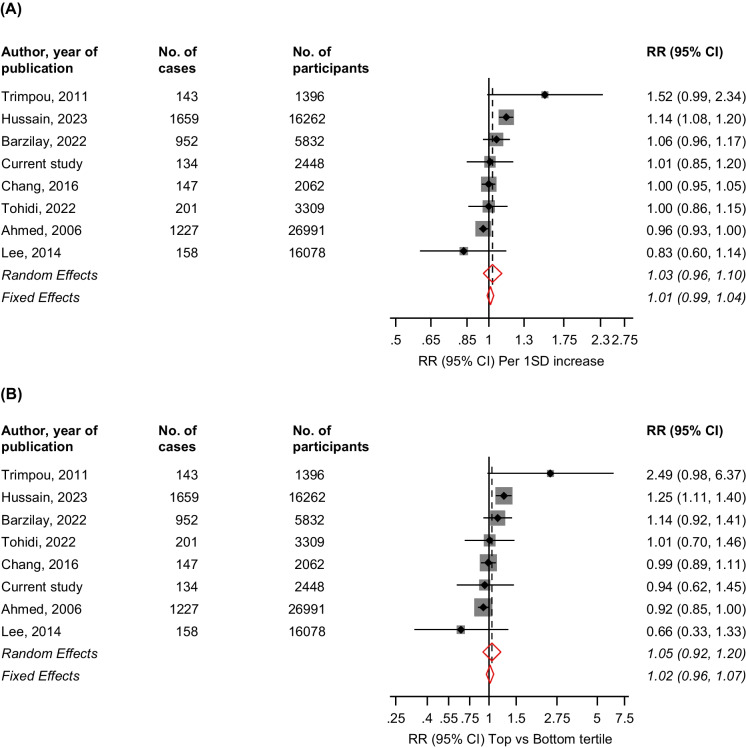
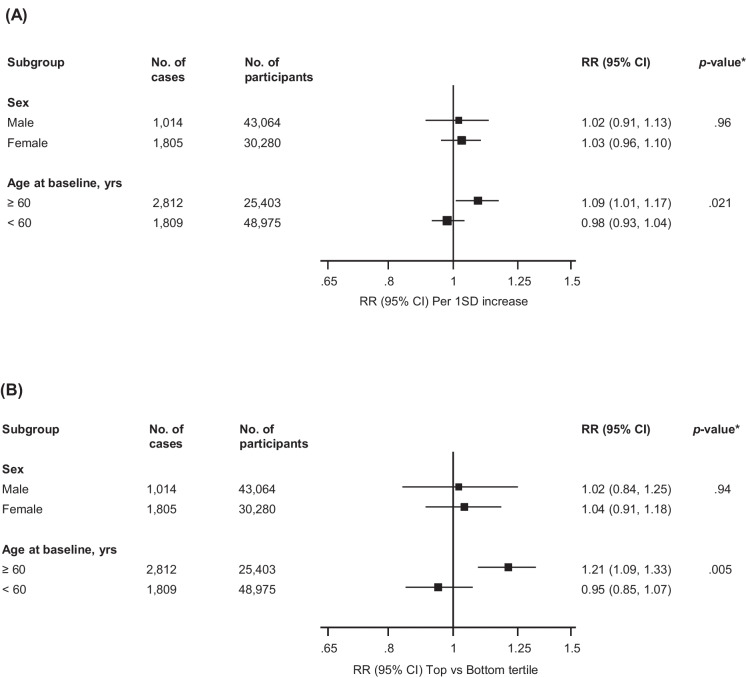
Previous findings on the association between high-density lipoprotein cholesterol (HDL-C) and fracture have yielded inconsistent results and it is unclear if the association varies with age and sex. We sought to evaluate the prospective association between HDL-C levels and fracture risk and assess if the association is modified by age and sex. Circulating HDL-C levels were measured at baseline in a population-based sample of 2,448 men aged 42–61 years. Cox regression was used to estimate hazard ratios (HRs) and 95% CIs. Incident fractures (n = 134) occurred during a median follow-up of 25.7 years. In analysis adjusted for several risk factors, the HR (95% CI) for fractures was 1.00 (0.85–1.20) per 1 standard deviation (SD) increase in HDL-C levels. Comparing the extreme tertiles of HDL-C levels, the corresponding adjusted HR (95% CI) was 0.94 (0.62–1.45). In a meta-analysis of eight cohort studies (including the current study) comprising 74,378 participants and 4,621 fracture cases, the fully-adjusted risk estimate (95% CI) for fracture was 1.03 (0.96–1.10) per 1SD increase in HDL-C levels and 1.05 (0.92–1.20) comparing extreme tertiles of HDL-C. The pooled risk estimate (95% CIs) for fracture per 1SD increase were 1.09 (1.01–1.17) and 0.98 (0.93–1.04) for age groups ≥ 60 and < 60 years, respectively, and the corresponding risks comparing the extreme tertiles of HDL-C levels were 1.21 (1.09–1.33) and 0.95 (0.85–1.07) (p-value for interaction < 0.05). Age may modify the association between HDL-C levels and fracture risk – an increased fracture risk associated with increased HDL-C levels is only evident in older age (≥ 60 years).
Supplementary Information
The online version contains supplementary material available at 10.1007/s11357-023-00801-w.
Keywords: High-density lipoprotein cholesterol, Age, Fracture, Cohort study, Systematic review, Meta-analysis
Introduction
Fractures (particularly the non-traumatic types) constitute a global public health burden – they are associated with morbidity, disability, poor quality of life, high economic costs and mortality [1]. Though major risk factors such as sociodemographic characteristics, comorbidities, and bone mineral density (BMD) contribute to fracture risk [2], it appears some of the these factors do not explain a large proportion of the risk of fractures. Other factors may contribute to the residual risk of fractures. Unfavourable lipid profiles are major contributors of atherosclerotic cardiovascular diseases (ASCVDs), but they have also been linked to other disease conditions including fractures [3]. High-density lipoprotein cholesterol (HDL-C) is a major lipid. Given preclinical evidence showing that HDL-C reduces bone mineral density [4], it has been hypothesized that high HDL-C levels may be associated with an increased risk of fractures. However, previous evaluations of the associations have yielded inconsistent results. Indeed, some studies have reported positive associations [5], whereas others have reported null results [6]. Given the inconsistent nature of the evidence, there is a need to re-evaluate the evidence. To investigate the detailed nature, magnitude and specificity of the prospective association between HDL-C levels and fracture risk, we (1) utilized a new population-based prospective cohort of men with no previous history of fractures from eastern Finland and (2) conducted a pooled analysis of previous studies (plus the current study) that have evaluated the prospective associations between baseline HDL-C levels and fracture risk. This enabled us to (i) overcome sample size limitations of single cohort studies; (ii) increase precision; and (iii) minimise biases.
Materials and methods
Study design and population
The primary cohort study was conducted in accordance with STROBE (STrengthening the Reporting of OBservational studies in Epidemiology) guidelines for reporting observational studies in epidemiology (Electronic Supplementary Material 1). Study participants for the primary cohort analysis were part of the Finnish Kuopio Ischemic Heart Disease (KIHD) population-based prospective cohort study. The study design and recruitment methods have been described in several previous reports [7, 8]. To summarise briefly, a representative sample of 3,433 men aged 42–61 years who were inhabitants of Kuopio city and its surrounding areas in eastern Finland were invited for screening which was carried out between March 1984 and December 1989. Of the 3,433 men, 3,235 were found to be potentially eligible and of this number, 553 did not respond to the invitation or declined to participate and 2,682 men provided consent to participate in the study. For the current analysis, 2448 men had complete information on HDL-C, relevant covariates, and fracture outcomes (Electronic Supplementary Material 2).
Ethics
The Research Ethics Committee of the University of Kuopio and Kuopio University Hospital, Kuopio, Finland approved the study protocol (License number 143/97). All study procedures were conducted in accordance with the Declaration of Helsinki and written informed consent was obtained from all participants.
Assessment of HDL-C and relevant risk markers
For measurements of blood biomarkers including lipoproteins, study participants provided blood specimens between 8:00 and 10:00 a.m. after having abstained from alcohol consumption for 3 days, from smoking for 12 h, and after an overnight fast. After a study participant had rested in the supine position for 30 min, blood was drawn with Terumo Venoject VT-100PZ vacuum tubes (Terumo Corp., Tokyo, Japan). No tourniquet was used. The main serum lipoprotein fractions consisting of HDL, low-density lipoprotein (LDL) and very-low-density lipoprotein (VLDL), were separated within three days of blood sampling by a combination of ultracentrifugation and precipitation. The cholesterol content (mmol/L) of all lipoprotein fractions were determined with enzymatic methods (cholesterol CHOD-pap method, Boehringer Mannheim, Mannheim, FRG) [9]. The measurement of pH-corrected serum active calcium concentrations was made using ion selective electrodes (Microlyte 6, Kone, Finland; CV 1.6%) [10]. A random-zero sphygmomanometer was used to measure resting blood pressure; following a supine rest of 5-min, blood pressure was measured three times in supine position, once in a standing position, and twice in a sitting position with 5-min intervals, and the arithmetic mean of all available measurements was taken [11, 12]. Self-administered lifestyle and health questionnaires were used to assess lifestyle characteristics such as smoking, alcohol consumption, physical activity and socioeconomic status (SES), prevalent medical conditions and use of medications [13]. The assessment of SES involved the creation of a summary index based on indicators such as income, education, occupational prestige, material standard of living and housing conditions [14-16]. The composite SES index ranged from 0 to 25, with higher values indicating lower SES. A history of coronary heart disease (CHD) was defined as previous myocardial infarction, angina pectoris, the use of nitroglycerin for chest pain ≥ once a week or chest pain. Energy expenditure of physical activity was assessed using the validated KIHD 12-month leisure-time physical activity questionnaire [17, 18], which was modified from the Minnesota Leisure-Time physical activity Questionnaire [19].
Ascertainment of incident fractures
The outcome assessed was any non-traumatic fracture (defined as hip, humeral, or wrist fractures) that occurred from study entry to 2014 [8]. Data on incident fractures was collected from the National Hospital Discharge Register data (maintained by the Finnish Institute for Health and Welfare) by computer linkage using Finnish personal identification codes and a comprehensive review of hospital records, discharge diagnoses, and inpatient physician claims. Fracture outcomes were coded according to the International Classification of Diseases Tenth Revision diagnostic codes for fractures by site.
Statistical analyses
Prospective cohort analysis
Baseline characteristics were presented as means (standard deviation, SD) or medians (interquartile range, IQR) for continuous variables and counts (percentages) for categorical variables using descriptive analyses. Hazard ratios (HRs) with 95% confidence intervals (CIs) for fractures were calculated using Cox proportional hazard models after confirmation of no major departure from the proportionality of hazards assumptions using Schoenfeld residuals. High-density lipoprotein cholesterol was modeled as continuous [per 1SD increase] and categorical (tertiles) exposures. Hazard ratios were adjusted for in two models: (model 1) age and (model 2) model 1 plus systolic blood pressure (SBP), history of hypertension, CHD and type 2 diabetes (T2D), smoking status, SES, physical activity, alcohol consumption and circulating calcium. These confounders were selected based on the following criteria: (i) their established roles as risk factors for fractures, (ii) published associations with fracture in the KIHD study [8, 20, 21] or (iii) their potential as confounders based on known associations with fracture outcomes and observed associations with the exposure using the available data [22].
Systematic review and meta-analysis
The systematic review protocol was registered in the PROSPERO prospective register of systematic reviews (CRD42023393414) and was conducted in accordance with PRISMA and MOOSE guidelines (Electronic Supplementary Materials 3–4). We searched MEDLINE and Embase from inception to 24 January 2023 for published observational population-based cohort studies that had examined the relation of circulating HDL-C with the risk of fracture events in general adult populations and had at least 1 year of follow-up. Details of the search strategy are reported in Electronic Supplementary Material 5. The risk of bias within individual observational studies was assessed using the Cochrane Risk of Bias in Non-randomised Studies – of Interventions (ROBINS-I) tool [23]. To enable a consistent approach to the meta-analysis and enhance comparison, reported study-specific risk estimates were all transformed to per 1 SD increase and extreme tertiles of HDL-C using standard statistical methods, which have been described previously [7, 8, 24-26]. Summary relative risks (RRs) with 95% confidence intervals (CIs) were pooled using random effects models to minimize the effect of between-study heterogeneity. Fixed effects models were used in parallel analyses. In-between study statistical heterogeneity was quantified using standard chi-square tests and the I2 statistic [27]. Given that the risk of fractures varies with age and sex [2], we also assessed if the association between HDL-C and fracture risk is modified by age and sex using stratified analysis and random effects meta-regression [28]. STATA release MP 16 (StataCorp LP, College Station, TX, USA) was used for all statistical analyses.
Results
Table 1 summarizes the baseline characteristics of the 2,448 participants in the primary cohort study. The mean (SD) of circulating HDL-C was 49.9 (11.7) mg/dl. The follow-up duration ranged from 0.02 to 30.8 years with a median (IQR) follow-up duration of 25.7 (17.5, 27.7) years. During the follow-up period, 134 fractures (annual rate 2.49/1,000 person-years at risk; 95% CI: 2.10–2.94) occurred. In analysis adjusted for age, SBP, history of hypertension, CHD and T2D, smoking status, SES, physical activity, alcohol consumption and circulating calcium, the HR (95% CI) for fractures was 1.00 (0.85–1.20) per 1SD increase in HDL-C levels. Alternatively, comparing the top versus bottom tertiles of HDL-C levels, the corresponding adjusted HR (95% CI) was 0.94 (0.62–1.45).
Table 1.
Baseline characteristics of primary cohort participants (N = 2448)
| Mean (SD) or median (IQR) | |
|---|---|
| Exposure | |
| HDL-C (mg/dl) | 49.9 (11.7) |
| Questionnaire/Prevalent conditions | |
| Age at survey (years) | 53 (5) |
| Alcohol consumption (g/week) | 32.0 (6.3–93.4) |
| Socioeconomic status | 8.45 (4.24) |
| History of type 2 diabetes, n (%) | 97 (4.0) |
| Current smokers, n (%) | 786 (32.1) |
| History of CHD, n (%) | 620 (25.3) |
| History of hypertension, n (%) | 736 (30.1) |
| Physical measurements | |
| BMI (kg/m2) | 26.9 (3.6) |
| SBP (mmHg) | 134 (17) |
| DBP (mmHg) | 89 (10) |
| Total physical activity (kj/day) | 1204 (632–2015) |
| Blood-based markers | |
| Total cholesterol (mmol/l) | 5.90 (1.07) |
| Fasting plasma glucose (mmol/l) | 5.35 (1.25) |
| Serum ionized calcium (mmol/l) | 1.18 (0.05) |
BMI body mass index; CHD coronary heart disease; CI confidence interval; DBP diastolic blood pressure; HDL-C high-density lipoprotein cholesterol; IQR interquartile range; SD standard deviation; SBP systolic blood pressure
In the systematic meta-analysis, we identified seven general population-based prospective cohort studies reporting on the associations between HDL-C levels and incident fracture risk [3, 5, 6, 29-32] (Electronic Supplementary Material 6 and Table 2). Including the current cohort study, the pooled analysis comprised eight studies involving 74,378 participants and 4,621 fracture cases. The average age and HDL-C levels at baseline ranged from approximately 45–75 years and 42.2–61.0 mg/dl, respectively, and the follow-up duration ranged from 2.0–26.0 years. Though there was variation in the degree of covariate adjustment, all eight studies adjusted for established risk factors (Table 2). All eight were at moderate risk of bias using the Cochrane Risk of Bias in Non-randomised Studies – of Interventions (ROBINS-I) tool (Electronic Supplementary Material 7). The pooled multivariable-adjusted random and fixed effects RRs (95% CIs) for fracture per 1SD HDL-C increase were 1.03 (0.96–1.10) and 1.01 (0.99–1.04), respectively; (I2 = 79.2%, 95% CI: 0.0 to 92.8%; p-value < 0.001). The corresponding risks comparing the top versus bottom tertiles of HDL-C levels were 1.05 (0.92–1.20) and 1.02 (0.96–1.07), respectively; (I2 = 71.4%, 95% CI: 0.0 to 89.7%; p-value = 0.001) (Fig. 1). The pooled random effects RRs (95% CIs) for fracture per 1SD increase were 1.09 (1.01–1.17) and 0.98 (0.93–1.04) for age groups ≥ 60 and < 60 years, respectively, and the corresponding risks comparing the top versus bottom tertiles of HDL-C levels were 1.21 (1.09–1.33) and 0.95 (0.85–1.07), respectively (p-value for meta-regression for all < 0.05) (Fig. 2). There was no evidence of effect modification by sex. Using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) tool, the certainty of the evidence was very low (Electronic Supplementary Material 8).
Table 2.
Baseline characteristics of eligible studies
| Author, year of publication | Study name | Country | Baseline year | Mean/median age, yrs | Male, % | Follow-up, yrs | Exposure | Assay | Fasting samples | No. of cases | No. of participants | Fracture outcomes | Adjustment factors |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ahmed, 2006 | Tromso | Norway | 1994–1995 | 47.0 | 47.3 | 6.0 | Serum HDL-C | NR | Non-fasting | 1227 | 26991 | Nonvertebral fractures | Age, DM, smoking and physical activity |
| Trimpou, 2011 | Gothenburg WHO MONICA Project | Sweden | 1985 | 44.6 | 47.0 | 20.0 | Plasma HDL-C | Enzymatic methods | Overnight fast | 143 | 1396 | Upper arm, wrist, ankle, leg, hip, pelvis, rib, vertebrae, and foot | Age, sex, time since baseline, BMI and cholesterol |
| Lee, 2014 | South Korean National Claim Registry | Korea | 2007–2011 | 57.1 | 100.0 | 3.0 | Serum HDL-C | Enzymatic colorimetric | 12 h fast | 158 | 16078 | Fractures at osteoporosis-related sites | Age, smoking and drinking habits, physical exercise, dairy product consumption, history of stroke, use of drugs that can affect bone metabolism, including bisphosphonates, vitamin D, thyroid hormone, or glucocorticoids, during study periods, and the presence of baseline radiological vertebral fractures |
| Chang, 2016 | SWAN | USA | 1995–1997 | 46.0 | 0.0 | 2.0 | Plasma HDL-C | Enzymatic methods | 12 h fast | 147 | 2062 | Foot, ankle, wrist, ribs, and legs | Age, race/ethnicity, study site, menopausal stage, smoking, alcohol use, physical activity, DM, BMI, and lumbar spine BMD |
| Barzilay, 2022 | CHS | USA | 1989–1990 | 73.0 | 42.5 | 13.5 | Plasma HDL-C | Enzymatic methods | NR | 952 | 5832 | Hip | Age, gender, race, smoking, alcohol, HTN, estimated GFR, DM, energy expended per week, frailty, CRP, ADLs, iADLs, weight, height, and prevalent CVD |
| Tohidi, 2022 | TLGS | Iran | 1999–2001/2002–2005 | 60.3 | 48.3 | 18.0 | Plasma HDL-C | Enzymatic colorimetric method | 12–14 h | 201 | 3309 | Any fracture | Age, BMI, smoking, T2D, hypertension, and lipid-lowering medication |
| Hussain, 2023 | ASPREE | Australia and USA | 2010–2014 | 75.0 | 45.0 | 4.0 | Plasma HDL-C | NR | NR | 1659 | 16262 | Vertebral, hip or nonhip, nonvertebral | Age, sex, physical activity, alcohol use, prefrailty/frailty status, education, body mass index, smoking status, aspirin use, diabetes, chronic kidney disease, use of lipid-lowering medication, and use of antiosteoporosis medications |
| Current study | KIHD | Finland | 1984–1989 | 53.0 | 100.0 | 25.7 | Plasma HDL-C | Enzymatic methods | Overnight fast | 134 | 2448 | Hip, humeral, or wrist fractures | Age, SBP, history of hypertension, history of CHD, smoking, history of DM, SES, physical activity, alcohol consumption and circulating calcium |
ADL activities of daily living; BMD bone mineral density; BMI body mass index; CHD coronary heart disease; CRP C-reactive protein; CVD cardiovascular disease; DM diabetes mellitus; GFR glomerular filtration rate; HDL-C high-density lipoprotein cholesterol; HTN hypertension; NR not reported; SBP systolic blood pressure; SES socioeconomic status; T2D type 2 diabetes
Study abbreviations: ASPREE Aspirin in Reducing Events in the Elderly’; CHS Cardiovascular Health Study; KIHD Kuopio Ischemic Heart Disease; SWAN Study of Women’s Health Across the Nation; TLGS Tehran Lipid and Glucose Study
Fig. 1.
Observational Cohort Studies of HDL-C and Risk of Fractures. (A) Per 1 standard deviation increase in HDL-C levels (B). Top versus bottom tertiles of HDL-C levels. The summary random and fixed effects estimates presented are based on fully adjusted estimates; CI, confidence interval (bars); HDL-C, high-density lipoprotein cholesterol; RR, relative risk; SD, standard deviation
Fig. 2.
Association Between HDL-C and Fracture Risk According to Age and Sex. (A) Per 1 SD increase (B) Top vs Bottom tertile of HDL-C. CI, confidence interval; HDL-C, high-density lipoprotein cholesterol; RR, relative risk; SD, standard deviation
Discussion
Our primary analysis of a population-based prospective study of middle-aged and older Finnish men demonstrated no evidence of an association between HDL-C levels and fracture risk. Pooled analysis of seven previous studies plus the current study using random or fixed effects models showed no evidence of an association. However, stratified analysis by age showed significant evidence of an increased risk of fracture with increased HDL-C levels in participants ≥ 60 years, with no evidence of an association in the younger age group (< 60 years).
In the most recent evaluation of the relationship between HDL-C and fracture risk, Hussain and colleagues conducted a posthoc analysis of the Aspirin in Reducing Events in the Elderly (ASPREE) trial comprising 16,262 participants aged ≥ 65 years and their results suggested that HDL-C might be an independent risk marker for fracture risk [5]. Their results showed a linear relationship between HDL-C and risk of fractures for both male and female participants [5]. The current findings add to the controversy about the nature of the epidemiological associations between HDL-C and outcomes. High-density lipoprotein cholesterol is considered an established risk factor for ASCVD and has a consistent, independent, strong, graded and inverse relationship with adverse cardiovascular outcomes [33]. High-density lipoprotein cholesterol has always been regarded as the “good cholesterol” with high levels conferring a protective effect and vice versa. However, emerging evidence suggest that higher levels of HDL-C are also associated with increased risk of adverse cardiovascular outcomes, with the association being consistent with a U-shaped relationship [34]. Similar relationships have been demonstrated for other outcomes such as age-related macular degeneration [35]. Barzilay and colleagues in their evaluation of the associations of conventional lipid and lipoprotein levels with incident fracture risk, demonstrated a non-linear U-shaped relationship between HDL-C and hip fracture risk [6]. There have been efforts to explain these conflicting results, but these have mainly been speculative. This is complicated by the fact that HDL and its functional biology and metabolism are complex. Hussain and colleagues suggested the most likely pathophysiological explanation for their findings could be the link between HDL-C levels and BMD [5]; HDL-C has been reported in preclinical studies to reduce BMD via reduction in osteoblast production [36]. A recent study has also suggested that HDL-C might be causally related to fracture risk [37]. The lack of significant evidence of an association between HDL-C levels and fracture risk in the primary cohort could be due to the fact the cohort was based on male participants, who have a lower predisposition to osteoporotic fractures than women [38]. Furthermore, this may be due to inadequate statistical power given the low event rate. Conversely, the null association demonstrated in the primary cohort may be a true one, reflecting the fact that increased levels of HDL-C might only increase the risk of fractures in older people. The mechanisms by which increased HDL-C levels might increase fracture risk in older age are unclear; however, it may be related to the fact that older age is a major risk factor for increased risk of fractures. Furthermore, aging is also known to alter HDL composition resulting in structural and functional impairment [39]; for example, it has been shown that HDL isolated from older people have a reduced capacity to inhibit LDL oxidation and promote cholesterol efflux from macrophages [40, 41]. It has been reported that the structural and functional changes in HDL in older people may be linked to a loss in its potential anti-atherogenic properties, contributing to the high burden of ASCVD in older people [39].
Based on a pooled analysis of all studies conducted on the topic, our findings do suggest that HDL-C may potentially be used as a biomarker to estimate the increased risk of fractures in older participants. It is known that cardiovascular risk estimation varies with different HDL levels [42] and HDL-C levels are also known to decrease with aging [43], hence, the prognostic ability of HDL-C for CVD is expected to change with age. This goes to suggest that the potential prognostic relevance of HDL-C for fracture risk may also vary with age. Nevertheless, these findings and their potential implications are still early and need to be considered in light of the fact that HDL is a complex molecule and its epidemiology may vary with age, gender, alcohol consumption, metabolic status, presence and type of comorbidities, ethnicity, follow-up durations and endpoints. With regards to the current findings, further research is needed to clarify whether the associations of HDL particles and its functional measures with fracture risk vary with age.
Strengths and limitations
Strengths of this evaluation included the use of a new prospective study and systematic meta-analysis of all available studies to re-evaluate the relationship, enhanced power and the ability to harmonise the data into consistent comparisons to enhance pooling. Most of the limitations were inherent and included the substantial variation in study designs and population and the potential for biases such as residual confounding, reverse causation bias and regression dilution.
Conclusions
New evidence based on a systematic meta-analysis of available observational prospective cohort studies plus a new population-based cohort study suggest age may modify the association between HDL-C levels and fracture risk – an increased risk of fracture associated with increased HDL-C levels is only evident in older groups (≥ 60 years).
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
We thank the staff of the Kuopio Research Institute of Exercise Medicine and the Research Institute of Public Health and University of Eastern Finland, Kuopio, Finland for the data collection in the study.
Authors’ contributions
Study design SKK and JAL. Study conduct: SKK and JAL. Data collection: SKK and JAL. Data analyses: SKK. Data interpretation: SKK. Drafting manuscript: SKK. Revising manuscript content: SKK and JAL. Approving final version of manuscript. SKK and JAL. SKK takes responsibility for the integrity of the data analysis.
Funding
SKK is supported by the National Institute for Health and Care Research (NIHR) Leicester Biomedical Research Centre (BRC). The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care. JAL is supported by the Finnish Foundation for Cardiovascular Research, Helsinki, Finland. The funder had no role in the design and conduct of this study; collection, management, analysis, and interpretation of the data; and decision to submit the manuscript for publication but was given the opportunity to review and comment on the manuscript.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declarations
Ethical approval
The study was conducted in accordance with the ethical standards set forth in the Helsinki Declaration (1983). Participants in the study were included after obtaining their written and informed consent for the study. The institutional review board of the University of Kuopio and Kuopio University Hospital, Kuopio, Finland (License number 143/97) approved the study protocol.
Conflict of interest
The authors declare that they have no conflicts of interest.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Griffin XL, Parsons N, Achten J, Fernandez M, Costa ML. Recovery of health-related quality of life in a United Kingdom hip fracture population. The Warwick Hip Trauma Evaluation–a prospective cohort study. Bone Joint J. 2015;97-B(3):372–82. doi: 10.1302/0301-620X.97B3.35738. [DOI] [PubMed] [Google Scholar]
- 2.International Osteoporosis Foundation. Epidemiology of osteoporosis and fragility fractures. https://www.osteoporosis.foundation/facts-statistics/epidemiology-of-osteoporosis-and-fragility-fractures. Assessed 31 Mar 2023.
- 3.Trimpou P, Oden A, Simonsson T, Wilhelmsen L, Landin-Wilhelmsen K. High serum total cholesterol is a long-term cause of osteoporotic fracture. Osteoporos Int. 2011;22(5):1615–1620. doi: 10.1007/s00198-010-1367-2. [DOI] [PubMed] [Google Scholar]
- 4.Papachristou NI, Blair HC, Kypreos KE, Papachristou DJ. High-density lipoprotein (HDL) metabolism and bone mass. J Endocrinol. 2017;233(2):R95–R107. doi: 10.1530/JOE-16-0657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hussain SM, Ebeling PR, Barker AL, Beilin LJ, Tonkin AM, McNeil JJ. Association of plasma high-density lipoprotein cholesterol level with risk of fractures in healthy older adults. JAMA Cardiol. 2023;8(3):268–72. [DOI] [PMC free article] [PubMed]
- 6.Barzilay JI, Buzkova P, Kuller LH, Cauley JA, Fink HA, Sheets K, et al. The association of lipids and lipoproteins with hip fracture risk: The cardiovascular health study. Am J Med. 2022;135(9):1101–8 e1. doi: 10.1016/j.amjmed.2022.05.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kunutsor SK, Isiozor NM, Myers J, Seidu S, Khunti K, Laukkanen JA. Baseline and usual cardiorespiratory fitness and the risk of chronic kidney disease: A prospective study and meta-analysis of published observational cohort studies. Geroscience. 2023. [DOI] [PMC free article] [PubMed]
- 8.Kunutsor SK, Seidu S, Voutilainen A, Blom AW, Laukkanen JA. Handgrip strength-a risk indicator for future fractures in the general population: findings from a prospective study and meta-analysis of 19 prospective cohort studies. Geroscience. 2021;43(2):869–880. doi: 10.1007/s11357-020-00251-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Salonen JT, Salonen R, Seppanen K, Rinta-Kiikka S, Kuukka M, Korpela H, et al. Effects of antioxidant supplementation on platelet function: a randomized pair-matched, placebo-controlled, double-blind trial in men with low antioxidant status. Am J Clin Nutr. 1991;53(5):1222–1229. doi: 10.1093/ajcn/53.5.1222. [DOI] [PubMed] [Google Scholar]
- 10.Kunutsor SK, Laukkanen JA. Circulating active serum calcium reduces the risk of hypertension. Eur J Prev Cardiol. 2017;24(3):239–243. doi: 10.1177/2047487316681174. [DOI] [PubMed] [Google Scholar]
- 11.Kunutsor SK, Jae SY, Kurl S, Laukkanen JA. The interplay between systolic blood pressure, cardiorespiratory fitness, and mortality risk: a prospective cohort study. J Cardiopulm Rehabil Prev. 2023. [DOI] [PubMed]
- 12.Laukkanen JA, Jae SY, Kurl S, Kunutsor SK. High fitness levels attenuate the increased risk of cardiovascular deaths in individuals with high systolic blood pressure: a prospective cohort study. Eur J Prev Cardiol. 2023:zwad034. [DOI] [PubMed]
- 13.Salonen JT, Nyyssonen K, Korpela H, Tuomilehto J, Seppanen R, Salonen R. High stored iron levels are associated with excess risk of myocardial infarction in eastern Finnish men. Circulation. [Research Support, Non-U.S. Gov't] 1992;86(3):803–11. doi: 10.1161/01.cir.86.3.803. [DOI] [PubMed] [Google Scholar]
- 14.Kunutsor SK, Jae SY, Makikallio TH, Kurl S, Laukkanen JA. High fitness levels offset the increased risk of chronic obstructive pulmonary disease due to low socioeconomic status: A cohort study. Respir Med. 2021;11(189):106647. doi: 10.1016/j.rmed.2021.106647. [DOI] [PubMed] [Google Scholar]
- 15.Kunutsor SK, Jae SY, Makikallio TH, Laukkanen JA. High fitness levels attenuate the increased risk of heart failure due to low socioeconomic status: A cohort study. Eur J Clin Invest. 2022;14:e13744. doi: 10.1111/eci.13744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kunutsor SK, Jae SY, Makikallio TH, Laukkanen JA. High fitness levels attenuate the increased risk of hypertension due to low socioeconomic status in middle-aged men: a cohort study. J Cardiopulm Rehabil Prev. 2022;42(2):134–136. doi: 10.1097/HCR.0000000000000673. [DOI] [PubMed] [Google Scholar]
- 17.Laukkanen JA, Laaksonen D, Lakka TA, Savonen K, Rauramaa R, Makikallio T, et al. Determinants of cardiorespiratory fitness in men aged 42 to 60 years with and without cardiovascular disease. Am J Cardiol. 2009;103(11):1598–1604. doi: 10.1016/j.amjcard.2009.01.371. [DOI] [PubMed] [Google Scholar]
- 18.Kunutsor SK, Khan H, Laukkanen JA. Serum albumin concentration and incident type 2 diabetes risk: new findings from a population-based cohort study. Diabetologia. 2015;58(5):961–967. doi: 10.1007/s00125-015-3520-0. [DOI] [PubMed] [Google Scholar]
- 19.Taylor HL, Jacobs DR, Jr, Schucker B, Knudsen J, Leon AS, Debacker G. A questionnaire for the assessment of leisure time physical activities. J Chronic Dis. 1978;31(12):741–755. doi: 10.1016/0021-9681(78)90058-9. [DOI] [PubMed] [Google Scholar]
- 20.Kunutsor SK, Blom AW, Whitehouse MR, Kehoe PG, Laukkanen JA. Renin-angiotensin system inhibitors and risk of fractures: a prospective cohort study and meta-analysis of published observational cohort studies. Eur J Epidemiol. 2017;32(11):947–959. doi: 10.1007/s10654-017-0285-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kunutsor SK, Whitehouse MR, Blom AW, Laukkanen JA. Low serum magnesium levels are associated with increased risk of fractures: a long-term prospective cohort study. Eur J Epidemiol. 2017;32(7):593–603. doi: 10.1007/s10654-017-0242-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Groenwold RH, Klungel OH, Grobbee DE, Hoes AW. Selection of confounding variables should not be based on observed associations with exposure. Eur J Epidemiol. 2011;26(8):589–593. doi: 10.1007/s10654-011-9606-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sterne JA, Hernan MA, Reeves BC, Savovic J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;12(355):i4919. doi: 10.1136/bmj.i4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kunutsor SK, Apekey TA, Khan H. Liver enzymes and risk of cardiovascular disease in the general population: A meta-analysis of prospective cohort studies. Atherosclerosis. 2014;236(1):7–17. doi: 10.1016/j.atherosclerosis.2014.06.006. [DOI] [PubMed] [Google Scholar]
- 25.Kunutsor SK, Apekey TA, Cheung BM. Gamma-glutamyltransferase and risk of hypertension: a systematic review and dose-response meta-analysis of prospective evidence. J Hypertens. 2015;33(12):2373–2381. doi: 10.1097/HJH.0000000000000763. [DOI] [PubMed] [Google Scholar]
- 26.Kunutsor SK, Seidu S, Laukkanen JA. Physical activity reduces the risk of pneumonia: systematic review and meta-analysis of 10 prospective studies involving 1,044,492 participants. Geroscience. 2022;44(1):519–532. doi: 10.1007/s11357-021-00491-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Thompson SG, Sharp SJ. Explaining heterogeneity in meta-analysis: a comparison of methods. Stat Med. 1999;18(20):2693–2708. doi: 10.1002/(SICI)1097-0258(19991030)18:20<2693::AID-SIM235>3.0.CO;2-V. [DOI] [PubMed] [Google Scholar]
- 29.Chang PY, Gold EB, Cauley JA, Johnson WO, Karvonen-Gutierrez C, Jackson EA, et al. Triglyceride levels and fracture risk in midlife women: Study of Women's Health Across the Nation (SWAN) J Clin Endocrinol Metab. 2016;101(9):3297–3305. doi: 10.1210/jc.2016-1366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lee SH, Baek S, Ahn SH, Kim SH, Jo MW, Bae SJ, et al. Association between metabolic syndrome and incident fractures in Korean men: a 3-year follow-up observational study using national health insurance claims data. J Clin Endocrinol Metab. 2014;99(5):1615–1622. doi: 10.1210/jc.2013-3608. [DOI] [PubMed] [Google Scholar]
- 31.Ahmed LA, Schirmer H, Berntsen GK, Fonnebo V, Joakimsen RM. Features of the metabolic syndrome and the risk of non-vertebral fractures: the Tromso study. Osteoporos Int. 2006;17(3):426–432. doi: 10.1007/s00198-005-0003-z. [DOI] [PubMed] [Google Scholar]
- 32.Tohidi M, Barzegar N, Hasheminia M, Azizi F, Hadaegh F. Association of different lipid measures with incident bone fractures: Tehran lipid and glucose study. Postgrad Med. 2022;134(3):326–332. doi: 10.1080/00325481.2022.2050980. [DOI] [PubMed] [Google Scholar]
- 33.Goff DC, Jr, Lloyd-Jones DM, Bennett G, Coady S, D'Agostino RB, Sr, Gibbons R, et al. 2013 ACC/AHA guideline on the assessment of cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;63(25 Pt B):2935–59. doi: 10.1016/j.jacc.2013.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kunutsor SK, Laukkanen JA. Further proof of a paradoxical relationship between high-density lipoprotein levels and adverse cardiovascular outcomes - Are there implications for cardiovascular disease prevention? Eur J Prev Cardiol. 2022;30(4):290–92 [DOI] [PubMed]
- 35.Nordestgaard LT, Christoffersen M, Lauridsen BK, Afzal S, Nordestgaard BG, Frikke-Schmidt R, et al. Long-term Benefits and Harms Associated With Genetic Cholesteryl Ester Transfer Protein Deficiency in the General Population. JAMA Cardiol. 2022;7(1):55–64. doi: 10.1001/jamacardio.2021.3728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Blair HC, Kalyvioti E, Papachristou NI, Tourkova IL, Syggelos SA, Deligianni D, et al. Apolipoprotein A-1 regulates osteoblast and lipoblast precursor cells in mice. Lab Invest. 2016;96(7):763–772. doi: 10.1038/labinvest.2016.51. [DOI] [PubMed] [Google Scholar]
- 37.Chen H, Shao Z, Gao Y, Yu X, Huang S, Zeng P. Are blood lipids risk factors for fracture? Integrative evidence from instrumental variable causal inference and mediation analysis using genetic data. Bone. 2020;131:115174. doi: 10.1016/j.bone.2019.115174. [DOI] [PubMed] [Google Scholar]
- 38.Alswat KA. Gender disparities in osteoporosis. J Clin Med Res. 2017;9(5):382–387. doi: 10.14740/jocmr2970w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Holzer M, Trieb M, Konya V, Wadsack C, Heinemann A, Marsche G. Aging affects high-density lipoprotein composition and function. Biochim Biophys Acta. 2013;1831(9):1442–1448. doi: 10.1016/j.bbalip.2013.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Berrougui H, Isabelle M, Cloutier M, Grenier G, Khalil A. Age-related impairment of HDL-mediated cholesterol efflux. J Lipid Res. 2007;48(2):328–336. doi: 10.1194/jlr.M600167-JLR200. [DOI] [PubMed] [Google Scholar]
- 41.Jaouad L, de Guise C, Berrougui H, Cloutier M, Isabelle M, Fulop T, et al. Age-related decrease in high-density lipoproteins antioxidant activity is due to an alteration in the PON1's free sulfhydryl groups. Atherosclerosis. 2006;185(1):191–200. doi: 10.1016/j.atherosclerosis.2005.06.012. [DOI] [PubMed] [Google Scholar]
- 42.Gulec S, Erol C. High-density lipoprotein cholesterol and risk of cardiovascular disease. e-Journal Cardiol Pract. 2020;19(3).
- 43.Ferrara A, Barrett-Connor E, Shan J. Total, LDL, and HDL cholesterol decrease with age in older men and women The Rancho Bernardo Study 1984–1994. Circulation J. 1997;96(1):37–43. doi: 10.1161/01.CIR.96.1.37. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.