Abstract
Infection in an immunocompromised host can be a life-threatening nursing emergency. Collection of blood cultures is an important method for detecting organisms when infection is suspected. One aspect of proper collection of blood cultures is obtaining an accurate volume of the blood sample. Underfilling of blood culture specimens can lead to decreased sensitivity of the culture and delay in choice of appropriate antibiotic selection. At our institution, the Department of Laboratory Medicine (DLM) routinely monitors volumes of blood culture bottles. For blood culture specimens that nursing collects, overfill/underfill events are sent to nursing and reviewed. Over several years, nursing and DLM noted that events of overfill and underfill of blood culture specimens were increasing. One of the Clinical Nurse Specialists and three staff nurses in the Oncology/Critical Care Services area partnered with DLM to determine why overfill/underfill was occurring. On investigation of nursing practice, several issues, such as misunderstanding on technique in blood culture collection and correct volume, were observed. In response to these observations, a plan was developed where the nursing standard of practice and the DLM webpage was updated, and nursing education was performed. As a result, overfill/underfill events decreased by 71% in a little over a year.
Keywords: blood cultures, collection, volumes, nursing procedure
I. Introduction
Sepsis is one of the leading causes of death globally (Rudd, et. al., 2020). In the immunocompromised oncology population, rapid identification and treatment of infection before the onset of sepsis saves lives. The cancer population has a ten-fold risk of developing sepsis and a two to three times increase in the risk of death from sepsis compared to the general population (Sepsis Alliance, 2020). Immediate nursing intervention, which includes the proper collection of blood culture specimens, impacts overall survival from infection.
Blood cultures are important for identifying the correct treatment for an infection. Obtaining the correct volume in blood culture bottles is one aspect of the blood culture collection procedure. The detection of organisms increases by 1% to 3% for each milliliter of blood collected thus increasing sensitivity of the culture and allowing for appropriate antibiotic selection (Neves, et. al., 2015). Reviews from institutions around the world have acknowledged that blood culture volume is the most variable part of the collection procedure (Henning, et. al., 2019; Sacco, Peterson, & Libertin, 2018) and a major detriment to blood culture sensitivity (Lamy, et. al., 2016). The reasons behind this variability are due to technique of the phlebotomist, patient characteristics, and gaps in understanding the importance of obtaining the correct volume.
Manufacturers of blood culture bottles specify the ideal volume for their products that will increase rapid detection of organisms. Under filling of the culture bottle can lead to false negative results; in the oncology population, this can increase the risk of death from sepsis. The College of American Pathologists (CAP) (2018) addressed the under filling of blood culture bottles by mandating that laboratories should have a process in place for monitoring blood culture volumes to increase the accuracy in diagnosis. The Department of Laboratory Medicine (DLM) at our institution meets this requirement by regularly tracking fill volumes. Overfill and under fill of bottles collected by nursing staff is regularly reported to the nursing department by DLM. This article describes our institution’s Department of Nursing quality improvement initiative undertaken in partnership with DLM to match and improve blood culture collection volumes as recommended by the manufacturer.
II. Background
The DLM at our institution routinely tracks blood culture fill volumes as part of their quality assurance program and CAP mandates. The reports of fill volumes collected by nurses are regularly reported back to the nursing department. Nursing and the Office of Patient Safety and Quality track the reports to identify patient unit trends of overfill and underfill of the bottles.
This institution utilizes the BD Bac-Tec® blood culture bottles. The optimal fill volume of these bottles for adult patients per BD ranges from 8–10 milliliters (mls). DLM weighs all unfilled bottles with a dedicated scale before blood culture bottles are distributed to the units. The weights of these bottles are kept on a spreadsheet. Once the bottle is used to collect the blood culture, the bottle is re-weighed when it arrives at the lab. The difference in weight from baseline (not filled) to filled is calculated with 1ml equal to 1 gram (gm). So, a correct fill volume would weigh 8–10 grams over the baseline weight. Although under filled bottles are not rejected by the lab, the sensitivity of an accurate diagnosis is decreased. In pediatric patients, blood culture volumes are based on the weight of the child.
In 2018, DLM reached out to the nursing department to collaborate with us to decrease overfill and underfill events. Ongoing education and tracking occurred from 2018 to 2019 however, the prevalence of overfill and underfill in the hospital continued to concern DLM. In contrast, the nursing department questioned DLM’s method of weighing blood culture bottles in batches rather than as single bottles as an accurate way to measure volumes. Within the Oncology/Critical Care Nursing Department, 62 incidences of overfill or underfill were reported from December 2019 through February 2020. From the ongoing reports provided by DLM coupled with questions from nursing regarding how blood culture volumes are tracked, an effort was underway to determine the causes of underfill, overfill, and correct the problem.
At our institution, the nursing department follows the “Nursing Procedure on Blood Culture Collection”. Peripheral cultures were measured by marking the blood culture bottles for 10mls above the level of the broth. Cultures through a central venous access device are collected using a four-way stopcock (see Figure 1). An empty 10 cc syringe is attached to one port of the stopcock, allowing for accurate volume measurement when blood is pulled back into the syringe. A vacutainer, attached to a second port on the stopcock, is used to transfer the blood into the culture bottle. when the stopcock is turned off to the patient and saline syringe. In the oncology/critical care areas, most of the patients have central venous access devices and the nurses use the four-way stopcock to collect the cultures. How the blood culture volumes for these patients could be inaccurate caused confusion within the nursing department. DLM took the opportunity to observe the collection of a blood culture by a nurse using the four-way stopcock and was also puzzled as to why the fill volumes were inaccurate.
Figure 1.
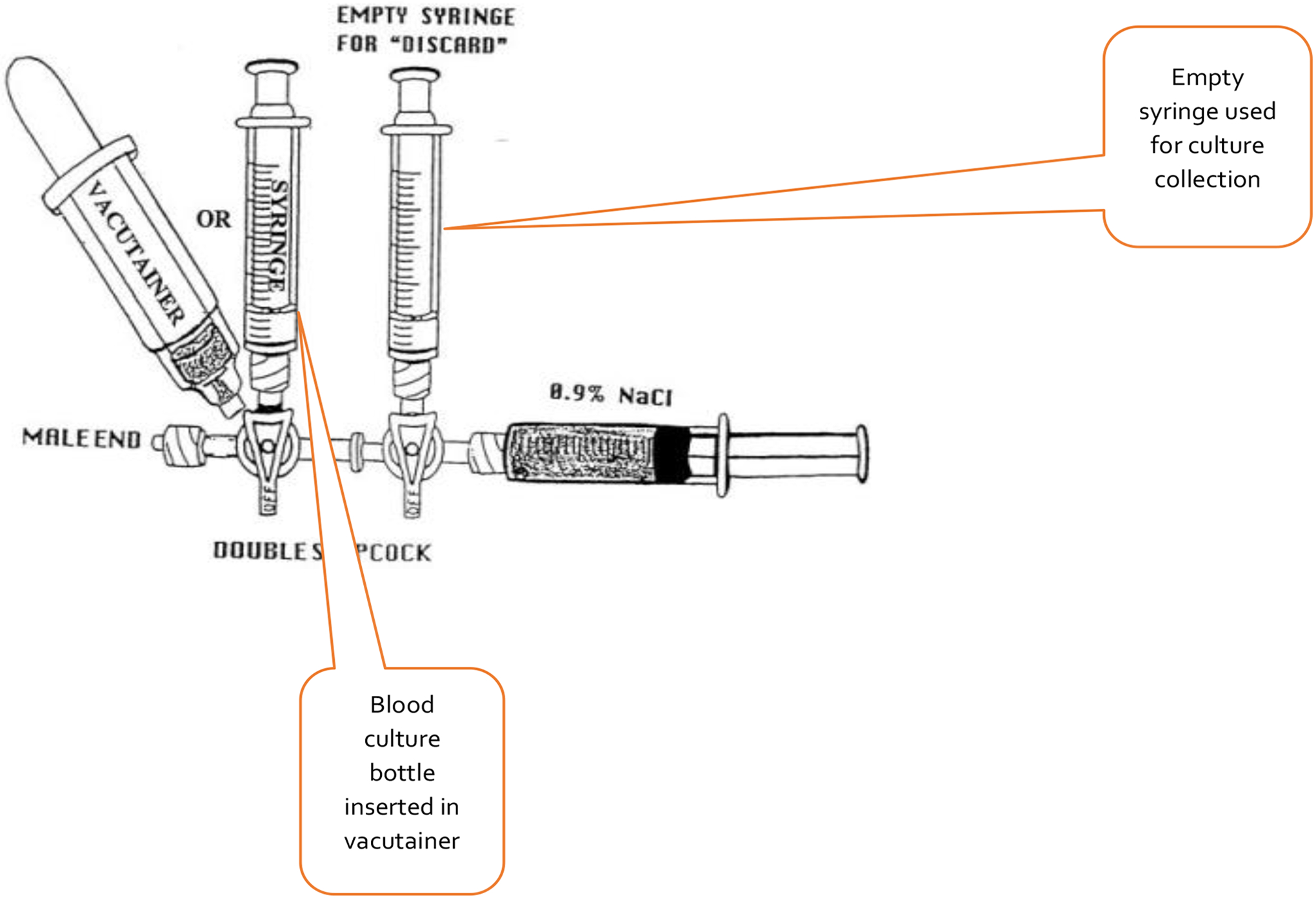
4-way stopcock
It was easy to understand how marking the bottles for peripheral cultures could result in an estimate of volume rather than an accurate measurement. Cultures collected through the four-way stopcock were more of a mystery. In order to understand this discrepancy, the Clinical Nurse Specialist (CNS) for the oncology areas partnered with three nurses from the inpatient oncology and intensive care units to design a project to find answers.
III. Methods
This was a quality improvement (QI) study that sought to decrease the incidence of overfill and underfill of blood cultures that were obtained by nurses and patient care technicians. The Oncology/Critical Care Nursing Service draws the majority of blood cultures within this hospital. This project took place across the four inpatient units that make up this service during February 2020 which included the intensive care unit (ICU), surgical oncology/immunotherapy unit, stem cell transplant/hematology unit, and the medical oncology lymphoma unit. The project consisted of two phases. Phase One was the background investigative phase and Phase Two consisted of the education/revalidation phase.
At our institution, one blood culture set consists of one aerobic and one anaerobic bottle. Each bottle must contain between 8–10 milliliters of blood for the microbiology lab to accurately determine a blood stream infection. Patient care technicians are allowed to draw blood cultures peripherally but only registered nurses can draw from a central line. Although our institution has a phlebotomy department that draws blood cultures peripherally, phlebotomy draws were excluded from this project as the phlebotomy department has its own monitoring processes for blood culture volumes.
Phase One
The Department of Laboratory Medicine weighs blood culture bottles before and after collection to determine the volume of blood in the bottle. Per DLM, one gram of weight was equal to one milliliter of fluid. Each blood culture bottle at “dry weight”, meaning without the blood culture sample, weighs 60 grams per DLM. DLM weighs multiple bottles at the same time and then stocks them on the units. Each bottle is cataloged with its unique lot number and when it returns to the lab with the blood culture sample, the bottle is reweighed. In this way, DLM tracks events of overfill and underfill. The information is kept on a spreadsheet and nursing is notified of each overfill or underfill event. For example, if the bottle is assumed to weigh 60 grams at dry weight, the bottle with the correct volume should weigh between 68 to 70 grams.
The DLM method for determining overfill and underfill was replicated by our nursing team to determine if the weighing of blood culture bottles was an accurate method for determining overfill and underfill. Our nursing team obtained a small bedside scale that had been calibrated by the Biomedical Department from the ICU; this scale would be the sole scale that nursing used for weighing all blood culture bottles. Our team of four nurses validated the method of weighing blood culture bottles with each other before beginning the project so that our technique would be consistent between each of us: The scale was zeroed prior to weighing the bottle(s) and the bottles were weighed before blood culture collection and after collection. To ensure reliability of the measurements, Phase One investigations were limited to our team of four nurses.
The study included the three oncology units and the ICU to observe the nurses drawing blood cultures and to determine if the weighing of blood culture bottles is a precise way to determine accurate volumes. The original sample size was to be 60 patients (15 patients on each unit). This was a random number selected as it was thought to be enough patients and samples to allow us to answer our questions. Only samples drawn from central lines were included. The nurses on each unit were asked to notify the nursing team when blood cultures were ordered. Once contacted, one of the nursing team went to the bedside, zeroed the scale, weighed each bottle that the nurse was using prior to culture collection, observed the procedure, and then re-weighed the bottles after the sample was obtained. A spreadsheet was utilized for data collection and uploaded into an electronic Excel spreadsheet that was password protected on the CNS’s computer to protect patient privacy. Incidence reports filed by DLM for overfill/underfill events, if any, were also tracked for each patient.
IV. Findings
Data was collected from four inpatient units within the Oncology and Critical Care Service over the month of February 2020. Although the original intent was to observe 60 blood culture collections, after 22 collections/observations, we were able to answer our original questions and note variations in practice that helped us understand why overfill/underfill events were occurring.
The first finding from this project was that not all blood culture bottles weigh 60 grams. With the ideal blood volume of 8–10 grams in the culture bottle, the weight of each bottle expected in DLM was 68–70 grams based on the dry weight of the bottle at 60 grams. The range in dry weight of the bottles without a culture sample varied from 59 to 62 grams. Thus, if the bottle weighed 61 grams and the nurse drew exactly 10ml of blood into that bottle, the weight of the bottle would be measured as 71 grams. This would create an overfill event. When DLM investigated this issue, they agreed that weighing the bottles in batches was not a precise measurement for calculating overfill/underfill. To account for the weight variation and to resolve this issue, DLM allowed for a weight of +/− 3 grams with the blood sample. In addition, at the time of this QI project, the Nursing Standard of Practice (SOP) and the DLM webpage allowed a range of 3ml-10ml as the correct volume of blood for an adult blood culture. As stated above, the optimal volume for a blood culture is 8–10ml per the manufacturer in order to increase sensitivity in detection of organisms. The nursing SOP and the DLM webpage were corrected for the correct volume of a blood culture and messaging went out to the nursing staff about the update.
The second finding involved the use of the 4-way stopcock when drawing blood cultures. The use of the stopcock allows the nurse to draw blood into a syringe, thus obtaining accurate volume of blood before the blood flows into the culture bottle. It was assumed that nurses were using the 4-way stopcock with the same method when drawing the cultures through the central line. We found that some nurses were using the 4-way stopcock but not pulling the blood into the syringe first but connecting the bottle directly into the vacutainer. This only allowed for an estimate of the volume in the bottle. Since the blood culture bottles can allow up to 30ml of blood, an overfill event can easily occur. Some nurses were marking the bottles at 10ml over the top of the bottle’s broth. This too caused inaccurate measurement as the bottle would need to be on a flat surface and upright during collection -a difficult feat to achieve when drawing blood from the central line. On speaking to the nursing staff, some nurses admitted they had not been trained on how to use the 4-way stopcock. This would explain the variation in practice with the use of the stopcock and led to the development of Phase Two of this QI project.
Phase Two
Based on the above findings regarding confusion on how to use the 4-way stopcock, we implemented one on one education with the staff in the oncology and intensive care units beginning March 2020. The study locations were expanded to included the outpatient oncology clinics, oncology day hospital, and radiation oncology. With the assistance of the clinical educators on each unit, the nurses were asked to demonstrate the procedure for utilizing the 4-way stopcock to obtain samples. Validation of competency was achieved specifically addressing the technique for blood culture collection through a central vascular access device. The Nursing Procedure on Blood Culture Collection was also reviewed with each nurse as part of the education with emphasis on the correct volume required for each bottle.
Although peripheral blood cultures were not included as part of the initial investigation into overfill/underfill events, these events were tracked and analyzed. The practice for obtaining peripheral blood cultures involves marking the bottles at 10ml above the broth. Knowing that this practice can be inaccurate unless the bottle is placed on a flat surface and upright, it was decided that the 2-way stopcock similar to the method used with central access collection would be used to ensure accurate volumes. Nurses were taught how to do this with peripheral culture collection as part of the education.
Patient care technicians (PCTs) draw the majority of peripheral blood cultures in the oncology areas. The PCTs also received one on one education similar to the nursing staff for peripheral blood culture collection, including use of the stopcock for accurate peripheral collection. The feedback from the PCTs was that the use of a three-way stopcock with a syringe at the end of a butterfly needle was less cumbersome than using the four-way stopcock. This feedback was incorporated into the education for both the nurses and the PCTs and was updated in the nursing procedure.
V. Results
The outcome of this project resulted in a trend of decreasing overfill/underfill events starting May 2020 through December 2021. In 2020, overfill/underfill events were 263. By the end of December 2021, a total of 76 events were recorded (see Figure 2). This was a decrease of 71%. Furthermore, because of the decrease in events related to under and over fill, in October 2021, DLM stopped weighing every bottle and now weighs only 20% of the samples. This project was successful in discovering the reason behind overfill/underfill events with blood cultures, developing interventions to resolve the problem, and increasing sensitivity of blood cultures through accurate filling volumes.
Figure 2.

Overfill/underfill events 2020–2021
Courtesy Susan Smith, CNS
VI. Discussion
This QI project was initiated at our institution in an effort to decrease blood culture overfill and underfill events. Accurate volumes of blood cultures are needed in order to identify infectious organisms to correctly treat the patient with the correct antibiotic and avoid overprescribing of antibiotics. Additionally, decreasing events of overfill/underfill is of interest to the microbiology laboratory in an effort to meet certification requirements with the College of American Pathologists. This project revealed opportunities to validate and clarify blood drawing techniques and practice among the staff. In collaboration with DLM, the exact volumes needed were defined in both the nursing standard of practice and the DLM resource webpage. Modifcations were made in DLM’s calculations of volumes based on varying weights of the bottles as weighing batches of blood culture bottles on the same scale was recognized as not a precise method for determing volumes of blood culture bottles. In response, DLM made allowance for a +/− 3 gm weight difference from the theoretical 60gm baseline weight of the unfilled bottle.
Moving forward, the nursing department continues to receive reports from DLM of overfill/underfill events quarterly. If the incidence of overfill/underfill is noted to be trending up on a particular unit, the CNS meets with the leadership of the unit to develop a plan for re-education. In addition, all new staff, including nurses and PCTs, are provided education on hire of the importance of the correct method for obtaining blood cultures. This includes emphasis on correct volumes and opportunity for hands on skill session of blood culture collection during the Nursing Department’s IV and Blood Workshop class. This class is required on orientation. Ongoing tracking of blood culture volumes has ensured that we continue to remain consistently below CAP thresholds for overfill/underfill events as well as assist with antibiotic stewardship.
VII. Conclusion
The nursing staff and CNS recognized the need for education on blood culture collection and correct use of the 4-way stopcock after determining that formal education had historically not been provided. Not only have blood culture events decreased but nurses are more confident in the use of the 4-way stopcock. Including the PCTs in the educational initiative provided them with the opportunity to improve their skills and give valuable insight into how to make peripheral collection easier. The nursing department continues to monitor events of overfill and underfill from DLM and re-validation of proper blood culture collection is performed as needed. New staff also receive this education on hire.
Realizing different manufacturers of blood culture bottles have different requirements when it comes to collecting blood cultures, this project served to increase awareness in the proper collection of blood cultures in case this institution switches blood culture products. The CNS and nurses have a heightened awareness in tracking events of overfill/underfill and are ready to intervene should the number of events start to increase.
Contributor Information
Leslie Smith, National Institutes of Health Clinical Center, 10 Center Dr, Bethesda, MD 20892.
Ruth Ali-Napo, National Institutes of Health Clinical Center, 10 Center Dr, Bethesda, MD 20892.
Deborah Ben-Zeev, National Institutes of Health Clinical Center, 10 Center Dr, Bethesda, MD 20892.
Justina Olawuyi, National Institutes of Health Clinical Center, 10 Center Dr, Bethesda, MD 20892.
References
- College of American Pathologists (2018). Microbiology checklist. Available at http://www.cap.org/apps/docs/laboratory_accreditation/checklists/microbiology_sep07.pdf. Accessed November 20, 2021.
- Henning C, Aygül N, Dinnétz P, Wallgren K, & Özenci V (2019). Detailed Analysis of the Characteristics of Sample Volume in Blood Culture Bottles. Journal of clinical microbiology, 57(8), e00268–19. 10.1128/JCM.00268-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamy B, Dargere S, Arendrup MC, Parienti JJ, Tattevin P. (2016). How to optimize the use of blood cultures for the diagnosis of bloodstream infections? A state-of-the art. Frontiers in Microbiology (7),697. 10.3389/fmicb.2016.00697 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neves L, Marra AR, Camargo TZS, dos Santos MC, Zulin F, da Silva PC, de Moura NA, Victor Eda S, Pasternak J, dos Santos OFP, Edmond MB, Martino MDV (2015). Correlation between mass and volume of collected blood with positivity of blood cultures. BMC Research Notes (8), 383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudd KE, Johnson SC, Agesa KM, Shackelford KA, Tsoi D, Kievlan DR, Colombara DV, Ikuta KS, Kissoon N, Finfer S, Fleischmann-Struzek C, Machado FR, Reinhart KK, Rowan K, Seymour CW, Watson RS, West TE, Marinho F, Hay SI, Lozano R, … Naghavi M (2020). Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the Global Burden of Disease Study. Lancet (London, England), 395(10219), 200–211. 10.1016/S0140-6736(19)32989-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sacco KA, Peterson JH, Libertin CR. (2018). Education and coaching to optimise blood culture volumes: continuous quality improvement in microbiology. BMJ Open Quality (7), 228. doi: 10.1136/bmjoq-2017-000228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sepsis Alliance (2020). Sepsis and oncology. Retrieved November 1, 2021 from https://www.sepsisinstitute.org/content/sepsis-and-oncology


