Abstract
Background
Many media are commercially available for culturing pre‐implantation human embryos in assisted reproductive technology (ART) cycles. It is unknown which culture medium leads to the best success rates after ART.
Objectives
To evaluate the safety and effectiveness of different human pre‐implantation embryo culture media in used for in vitro fertilisation (IVF) and intracytoplasmic sperm injection (ICSI) cycles.
Search methods
We searched the Cochrane Menstrual Disorders and Subfertility Group's Trials Register, Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, EMBASE, the National Research Register, the Medical Research Council's Clinical Trials Register and the NHS Center for Reviews and Dissemination databases from January 1985 to March 2015. We also examined the reference lists of all known primary studies, review articles, citation lists of relevant publications and abstracts of major scientific meetings.
Selection criteria
We included all randomised controlled trials which randomised women, oocytes or embryos and compared any two commercially available culture media for human pre‐implantation embryos in an IVF or ICSI programme.
Data collection and analysis
Two review authors independently selected the studies, assessed their risk of bias and extracted data. We sought additional information from the authors if necessary. We assessed the quality of the evidence using Grades of Recommendation, Assessment, Development and Evaluation (GRADE) methods. The primary review outcome was live birth or ongoing pregnancy.
Main results
We included 32 studies in this review. Seventeen studies randomised women (total 3666), three randomised cycles (total 1018) and twelve randomised oocytes (over 15,230). It was not possible to pool any of the data because each study compared different culture media.
Only seven studies reported live birth or ongoing pregnancy. Four of these studies found no evidence of a difference between the media compared, for either day three or day five embryo transfer. The data from the fifth study did not appear reliable.
Six studies reported clinical pregnancy rate. One of these found a difference between the media compared, suggesting that for cleavage‐stage embryo transfer, Quinn's Advantage was associated with higher clinical pregnancy rates than G5 (odds ratio (OR) 1.56; 95% confidence interval (CI) 1.12 to 2.16; 692 women). This study was available only as an abstract and the quality of the evidence was low.
With regards to adverse effects, three studies reported multiple pregnancies and six studies reported miscarriage. None of them found any evidence of a difference between the culture media used. None of the studies reported on the health of offspring.
Most studies (22/32) failed to report their source of funding and none described their methodology in adequate detail. The overall quality of the evidence was rated as very low for nearly all comparisons, the main limitations being imprecision and poor reporting of study methods.
Authors' conclusions
An optimal embryo culture medium is important for embryonic development and subsequently the success of IVF or ICSI treatment. There has been much controversy about the most appropriate embryo culture medium. Numerous studies have been performed, but no two studies compared the same culture media and none of them found any evidence of a difference between the culture media used. We conclude that there is insufficient evidence to support or refute the use of any specific culture medium. Properly designed and executed randomised trials are necessary.
Keywords: Female; Humans; Pregnancy; Culture Media; Culture Media/adverse effects; Embryo, Mammalian; Fertilization in Vitro; Oocytes; Sperm Injections, Intracytoplasmic; Abortion, Spontaneous; Embryo Transfer; Live Birth; Pregnancy Rate; Pregnancy, Multiple; Randomized Controlled Trials as Topic
Plain language summary
Culture media for human pre‐implantation embryos for in assisted reproductive technology cycles
Review question:
What is the impact of different human embryo culture media in women undergoing in vitro fertilisation (IVF) or intracytoplasmic sperm injection (ICSI) cycles, with regard to live birth or ongoing pregnancy and adverse events?
Background:
Embryo culture refers to the culture of human gametes and embryos during in vitro fertilisation treatments. The procedure is initiated when eggs and sperm are combined in a culture dish and it ends with the transfer of the resulting embryo to the uterus. The culture period varies between one and six days, and the embryo culture process is vital to the success of any IVF or ICSI procedure. The type of culture medium used may affect embryo quality and hence the success rate of the treatment. Despite its importance, it is unknown which culture medium is most effective and safest.
Study characteristics:
The evidence is current to March 2015. We included 32 randomised controlled trials of a wide variety of different commercially available culture media in women undergoing IVF or ICSI. Sixteen studies randomised women (total 3666), three randomised cycles (total 1018) and twelve randomised oocytes (over 15,230). Most studies (22/32) failed to report their source of funding.
Key results:
No two studies compared the same culture media. Only seven studies reported our primary outcome of live birth or ongoing pregnancy, and they found no good evidence of a difference between the media compared. A single study found low‐quality evidence that for day three embryo transfer, Quinn's Advantage may be associated with higher rates than G5, but this study was available only as an abstract and the methods used were not clearly reported. With regard to adverse effects, three studies reported multiple pregnancies and six studies reported miscarriage. None of them found any evidence of a difference between the culture media used. None of the studies reported on the health of offspring. We conclude that there is insufficient evidence to support or refute the use of any specific culture medium. Properly designed and executed randomised trials are necessary.
Quality of the evidence:
There was very low‐quality evidence for nearly all comparisons, the main limitations being imprecision and poor reporting of study methods.
Summary of findings
Summary of findings for the main comparison. Live birth or ongoing pregnancy per woman.
| Live birth or ongoing pregnancy in women receiving human pre‐implantation embryos using different culture media | ||||||
| Population: Women in assisted reproductive technology cycles (ART) receiving human pre‐implantation embryos in either early (cleavage stage transfer) or late (blastocyst stage transfer) stages Intervention: specific embryo culture media Comparison: other embryo culture media | ||||||
| Comparisons** | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of Participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Other embryo culture media | Specific embryo culture media | |||||
| Early embryo transfer ‐ GM501 (specific) versus ISMI (other) | 264 per 1000 | 270 per 1000 (160 to 422) | OR 1.03 (0.53 to 2.03) | 172 (1 study) | ⊕⊝⊝⊝ very low1,2 | No evidence of a difference between the two media in live birth rate |
| Early embryo transfer ‐ GM501 (specific) versus Sydney IVF (other) | 188 per 1000 | 167 per 1000 (81 to 311) | OR 0.87 (0.38 to 1.96) | 158 (1 study) | ⊕⊝⊝⊝ very low1,2 | No evidence of a difference between the two media in live birth rate |
| Early embryo transfer ‐ G2 (specific) versus Universal IVF (other) | 194 per 1000 | 269 per 1000 (139 to 457) | OR 1.53 (0.67 to 3.51) | 129 (1 study) | ⊕⊝⊝⊝ very low1,2 | No evidence of a difference between the two media in live birth rate |
| Early embryo transfer: Cook (K‐SIFM or K‐SICM) (specific) versus Vitrolife (IVF or G3) (other) | 220 per 1000 |
216 per 1000 (151 to 302) |
OR 0.98 (0.63 to 1.54) |
449 (1 study) |
⊕⊝⊝⊝ low3,4 | No evidence of a difference between the two media in live birth rate |
| Early embryo transfer ‐ G2 (specific) versus Ham's F10 (other) | 579 per 1000 |
676 per 1000 (444 to 846) |
OR 1.52 (0.58 to 3.99) |
72 (1 study) |
⊕⊝⊝⊝ very low2,3 | No evidence of a difference between the two media in ongoing pregnancy rate |
| Late embryo transfer ‐ GM501 (specific) versus Sydney IVF (other) | 200 per 1000 | 143 per 1000 (7 to 778) | OR 0.67 (0.03 to 14.03) | 12 (1 study) | ⊕⊝⊝⊝ very low1,2 | No evidence of a difference between the two media in live birth rate |
| Late embryo transfer ‐ G2 (specific) versus Universal IVF (other) | 148 per 1000 | 250 per 1000 (71 to 591) | OR 1.92 (0.44 to 8.31) | 47 (1 study) | ⊕⊝⊝⊝ very low1,2 | No evidence of a difference between the two media in live birth rate |
| Late embryo transfer ‐ ECM/Multiblast (specific) versus Global (other) | 700 per 1000 |
538 per 1000 (318 to 746) |
OR 0.50 (0.20 to 1.26) |
79 (1 study) |
⊕⊝⊝⊝ very low1,2 | No evidence of a difference between the two media in ongoing pregnancy rate |
| *The assumed risk is the risk in the study control group. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; OR: Odds ratio | ||||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | ||||||
** In addition to the studies in Table 2, one study compared HTF/CCM versus ISM‐1/ISM‐2 and reported that blastocyst transfer HTF/CCM was associated with higher birth rates. The data were not entered in data tables as they did not appear reliable; we have been unable to obtain clarification from the study authors.
1 Serious risk of bias. Poor reporting of study methods. 2 Very serious imprecision. Only one small study. 95% confidence interval around the estimate of effect includes both no effect and appreciable benefit or appreciable harm
3Serious risk of bias. Method of allocation concealment not reported.
4Serious imprecision: 95% confidence interval around the estimate of effect includes both no effect and appreciable benefit or appreciable harm
Background
Description of the condition
Human pre‐implantation embryos are exposed to culture media for several days during their development in in vitro fertilisation (IVF) or intracytoplasmic sperm injection (ICSI) treatment. A wide variety of culture media is available on the market with variable formulations, ranging from simple salt solutions to complex tissue culture media with synthetically derived amino acids and growth factors. Despite the well recognized need for an optimal in vitro environment for human pre‐implantation embryos, there are few data on the success rates of specific culture media.
Description of the intervention
Embryo culture media used in early human IVF had successfully been used in animal embryo culture. In 1985, an embryo culture medium called Human Tubal Fluid (HTF) was designed, based on the analysis of human tubal fluids (Quinn 1985). Since the development of HTF, many modifications have been made in the composition of human embryo culture media, with the addition of different components such as amino acids, vitamins, and nucleic acid precursors (Gardner 2002 a; Menezo 2004).
Culture media can be stratified into two types according to the changing metabolic needs of the rapidly growing and changing embryo. Cleavage media are formulated specifically to support the growth requirements of the early cleavage‐stage embryo until day three of pre‐implantation development. Embryos that are to be cultured until day five or six are placed into another medium, referred to as blastocyst medium, on day three. Blastocyst medium has different and additional components that are thought to be required by the embryo for its transition from the cleavage stage to the blastocyst stage. For the blastocyst culture, two media can be used sequentially (with cleavage‐stage specific medium replaced by blastocyst‐specific medium at day three of development), or one medium can be used continuously (possibly replacing the medium with the same but fresh medium at day three of development).
How the intervention might work
The accumulated knowledge of mammalian embryo physiology and biochemistry in culture media has led to the production of theoretically more stable and optimised environments to which human embryos are exposed (Gardner 2002 b). However, differences between media and their effect on embryonic, prenatal and postnatal development, blastulation rates, embryo implantation rates, live birth rates, and birth weight of the offspring have been reported (Dumoulin 2010; Mantikou 2013). Earlier studies have shown that the environment of gametes and pre‐implantation embryos has an effect on the expression and imprinting of a number of key genes that affect the development of the embryos (Blake 2007; Doherty 2000; Fauque 2007; Khosla 2001; Zander 2006). The differences in success rates of IVF treatments could be explained by differences in the composition and balance of the elements present in the culture medium. Currently, there is limited information concerning the composition of the culture media, which restricts the understanding of the mechanism of action.
Why it is important to do this review
Culture media are generally considered to play an important role in the success rates of an assisted reproductive technology (ART) programme (Mantikou 2013). They could have an effect on the health of the newborns (Dumoulin 2010). However, it is not known which specific culture medium leads to the best outcomes in terms of safety and effectiveness. We performed this systematic review to try to answer this question.
Objectives
To evaluate the safety and effectiveness of different human pre‐implantation embryo culture media used in IVF or ICSI cycles.
Methods
Criteria for considering studies for this review
Types of studies
Included studies:
We included all randomised controlled trials (RCT) that randomised women, oocytes, or embryos, and that compared different culture media used in IVF or ICSI cycles.
Excluded studies:
We excluded quasi‐randomised trials.
Types of participants
Women undergoing IVF or ICSI cycles.
Types of interventions
We included studies that compared any commercially available culture media for pre‐implantation embryo in an IVF or ICSI programme. The comparison groups were:
Studies comparing different culture media by randomising women for day two or three embryo transfer.
Studies comparing different culture media by randomising women for day five or six embryo transfer.
Studies which compared different culture media by randomising oocytes or embryos for day two or three embryo transfer.
Studies which compared different culture media by randomising oocytes or embryos for day five or six embryo transfer.
We excluded studies involving the addition of supplements (such as globulin or human serum albumin) to commercially available media.
Types of outcome measures
Primary outcomes
Live birth or ongoing pregnancy.
Live birth was defined as the number of live births after twenty completed weeks of gestational age per randomised woman.
Ongoing pregnancy was defined as the number of viable gestations with a positive fetal heart beat at 12 weeks per randomised woman.
Where studies reported both live births and ongoing pregnancy, we used the data on live births.
Secondary outcomes
Health of babies born: determined by birth weight and presence of congenital anomalies per baby
Clinical pregnancy rate: defined as the number of clinical pregnancies (demonstrated by the presence of a gestational sac on ultrasound scan at six weeks of pregnancy) per randomised woman
Multiple pregnancy rate: defined as the number of multiple pregnancies per randomised woman (adverse events)
Miscarriage rate: defined as the number of miscarriages per randomised woman (adverse events)
Implantation rate: defined as the number of women with one or more fetal sacs detected ultrasonographically at six weeks of gestation per total number of embryos transferred
Cryopreservation rate: defined as the number of embryos cryopreserved per total number of embryos
Embryo quality: defined as the number of good quality embryos per total oocytes or embryos randomised on day three. Assessment of embryo quality was based on the heterogeneous definitions used by the original investigators.
Fertilisation rate: defined as the number of fertilised oocytes per total number of oocytes injected or inseminated
Studies that compared different commercially available media but did not report any of the above outcomes were included for completeness, but none of their data were used in the analysis. We excluded pregnancy outcomes from studies that randomised oocytes or embryos.
Search methods for identification of studies
We searched for all published and unpublished RCTs that examined the use of different embryo culture media, using a search strategy designed in consultation with the Cochrane Menstrual Disorders and Subfertility Group's (MDSG) Trials Search Co‐ordinator, without language restriction. See Appendix 1; Appendix 2; Appendix 3; Appendix 4; Appendix 5.
Electronic searches
The following electronic databases, trials registers and websites were searched up to March 2015:
Cochrane MDSG Trials Register Appendix 1
Cochrane Central Register of Controlled Trials (CENTRAL) Appendix 2
MEDLINE Appendix 3
EMBASE Appendix 4
PsycINFO Appendix 5
Trials registers for ongoing and registered trials: Current Controlled Trials; the US National Institutes of Health: Clinical Trials Gov; The World Health Organisation International Trials Registry Platform search portal: WHO
DARE (Database of Abstracts of Reviews of Effects), on the Cochrane Library (DARE); reference lists from relevant non‐Cochrane reviews
LILACS database, providing a source of trials from the Portuguese‐ and Spanish‐speaking world (LILACS)
PubMed (PubMed)
Searching other resources
In addition, we searched:
The reference lists of all known primary studies, review articles and citation lists of relevant publications
Abstracts of major scientific meetings (e.g. the European Society of Human Reproduction and Embryology (ESHRE), the American Society for Reproductive Medicine (ASRM)) in the ISI Web of Knowledge (ISI)
Known experts and personal contacts were contacted regarding any unpublished materials
Data collection and analysis
Selection of studies
Two review authors (MY, EM) independently selected the trials to be included, using forms designed according to our inclusion criteria and Cochrane guidelines. We sought additional information on trial methodology from the authors of trials which appeared to meet the eligibility criteria but had unclear methodology. Differences of opinion were resolved after discussion with the other authors (MvW, FvdV, SR, SM, and HG). Subsequently, a detailed 'Characteristics of excluded studies' table was constructed for trials that did not satisfy the inclusion criteria. A 'Characteristics of included studies' table was constructed for those considered suitable.
The search and selection process was documented using a PRISMA flow diagram (Figure 1).
1.

Study flow diagram.
Data extraction and management
A standardised data extraction form was developed and piloted for consistency and completeness. Data extraction was performed independently by two review authors (MY, EM). The two sets of extracted data were compared and discrepancies were resolved by discussion with the other authors. Where studies had multiple publications, the main trial was used as a reference, and additional information was derived from the secondary papers.
We sought additional information from the authors of trials that reported data in a form unsuitable for meta‐analysis. Studies that included such data (e.g. means, percentages) were still included for completeness.
Assessment of risk of bias in included studies
Two review authors (MY, EM) independently assessed the risk of bias in the included studies, using the Cochrane risk of bias assessment tool to assess randomisation, allocation concealment, blinding of participants and personnel, blinding of outcome assessor, completeness of outcome data, selective outcome reporting, and other biases. Each trial characteristic was judged and given a risk of bias rating as low, unclear, or high. We entered the information into 'Risk of bias' tables.
Random sequence generation: risk of bias was considered low if an adequate method of sequence generation was described e.g. reference to a random number table or a computer random number generator.
Allocation concealment: risk of bias was considered low if a third‐party system, serially‐numbered sealed opaque envelopes, or a similar system was described. Concealment was stated as 'uncertain' if sealed envelopes were mentioned without further description or if no information was available pertaining to allocation concealment.
Blinding: examined with regard to who was blinded in the trials. All levels were sought and categorized as follows: single or double blinding; no blinding (doctors, embryologist and participant knew the allocated treatment); unclear.
Completeness of outcome data: risk of bias was considered low if there were no missing outcome data or the missing outcome data balanced in numbers across intervention groups, with similar reasons for missing data across groups.
Selective outcome reporting: risk of bias was considered low if the study protocol was available and all of the study’s pre‐specified (primary and secondary) outcomes that were of interest in the review had been reported as pre‐specified.
Measures of treatment effect
For dichotomous data (e.g. live birth rates), we used the number of events in the control and intervention groups of each study to calculate the odds ratio (OR) with 95% confidence interval (CI) using the Mantel‐Haenszel method with a fixed‐effect model.
If continuous data had been reported (e.g. birth weight), we had planned to calculate mean differences between the groups, with 95% CIs.
Unit of analysis issues
Our analysis was based on an intention‐to‐treat protocol; all clinical outcomes were analysed per randomised woman, even if this definition was not used by the original investigators. For studies that randomised cycles, we only included studies that had one cycle per woman in the analysis. For studies that randomised women, we developed additional tables for all outcomes reported per oocyte or embryo (implantation rate, embryo quality, fertilisation rate). We also developed additional tables for all studies that randomised oocytes or embryos.
We counted multiple live births as one live birth event.
When the studies did not use the correct unit of analysis, we attempted to contact authors to obtain data per randomised woman.
Dealing with missing data
We analysed the data on an intention‐to‐treat basis as far as possible; losses to follow‐up were included as failure for pregnancy and live birth. We attempted to obtain missing data from the original investigators. We calculated miscarriage rates when possible, based on differences between clinical and ongoing pregnancies.
Assessment of heterogeneity
We had planned to assess heterogeneity by visual inspection of the forest plots (Higgins 2011). We had planned to calculate the I² statistic when appropriate, and to quantify any apparent inconsistency. We defined I2 of 50% or more as indicating substantial heterogeneity.
Assessment of reporting biases
In view of the difficulty of detecting and correcting for publication bias and other reporting biases, the review authors minimised their potential impact by performing a comprehensive search for eligible studies, and by being alert for duplication of data. If there were ten or more studies in an analysis, we had planned to use a funnel plot to explore the possibility of small study effects (a tendency for estimates of the intervention effect to be more beneficial in smaller studies).
Data synthesis
If the studies were sufficiently similar, we had planned to combine the data using a fixed‐effect model to calculate pooled odds ratios or mean differences, with 95% CIs.
We conducted separate analyses for the following groups:
studies that compared culture media for days two or three embryo transfer (cleavage‐stage transfer)
studies that compared culture media for days four to six embryo transfer (blastocyst‐stage transfer)
studies that randomised oocytes or embryos instead of women for the following outcomes: fertilisation rate and embryo quality
We displayed the results of the included studies graphically in analysis tables.
Subgroup analysis and investigation of heterogeneity
We planned to conduct subgroup analyses if there was obvious clinical heterogeneity, to examine whether the effects varied by clinical setting (e.g. in different populations). If substantial statistical heterogeneity had been detected, we planned to explore whether effects varied by differences in the methodological characteristics of the studies (e.g. with differing risks of bias.)
Sensitivity analysis
We planned to conduct sensitivity analyses for the primary outcome to determine whether the conclusions were robust for the decisions we made regarding study eligibility and analyses. These analyses would have included consideration of whether the review conclusions would have differed if:
Analysis had been restricted to studies without high risk of bias;
A random‐effects model had been adopted;
Alternative imputation strategies had been implemented;
The summary effect measure had been relative risk rather than odds ratio;
The primary outcome had been live birth only.
Overall quality of the body of evidence: Summary of findings table
We generated a 'Summary of findings' table using GRADEpro software. This table evaluated the overall quality of the body of evidence for the primary review outcome, using Grades of Recommendation, Assessment, Development and Evaluation (GRADE) criteria (study limitations (i.e. risk of bias), consistency of effect, imprecision, indirectness and publication bias). We justified, documented, and incorporated our judgements about the quality of the evidence (high, moderate, low or very low) into the reporting of results for each outcome. Due to the large number of comparisons in the review, we chose to focus on our primary outcome and report findings for all relevant comparisons in a single table.
Results
Description of studies
The diversity of media used and outcome measures assessed can be seen in the Characteristics of included studies table. Each of the trials studied the effects of different culture media on different outcome measures, precluding a meta‐analysis.
Results of the search
We identified 1242 potentially relevant abstracts by our search strategy. After reading the abstracts, we discarded 1088 because they compared cleavage‐ stage versus blastocyst‐stage embryo transfer, media supplementation, or because they studied home‐made media or media for non‐human embryos. We retrieved the full papers of the remaining 154 abstracts that appeared to meet the inclusion criteria.Thirty‐two studies (38 articles) met our inclusion criteria (Arenas 2007; Artini 2004; Barak 1998; Baum 2004; Bird 2012; Bungum 2003; Campo 2010; Cano 2001; Carrasco 2013; Chatziioannou 2010; Chen 2009; Fechtali 2004; Findikli 2004; Gimeno 2006; Hazlett 2003; Jamieson 1997; Khan 2004; Kyono 2000; Lambert 2005; Mauri 2001; Mayer 2003; Mendoza 2003; Paternot 2010; Rubino 2004; Sepulveda 2009; Shih 2014; Sifer 2009; Stevens 2000; Tedesco 1990; Von During 2004; Yamamoto 2006; Zollner 2004), We excluded 100 studies (112 articles), and four studies are ongoing (See Figure 1). See Characteristics of excluded studies table for full exclusion details.
Included studies
Thirty two studies met the inclusion criteria (See Characteristics of included studies table).
Study design and setting
All included studies were randomised controlled trials. All were performed at a single private, university‐based or medical centre with the exception of three studies, where this was unclear (Arenas 2007; Baum 2004; Bird 2012).
Participants
Seventeen studies randomised 3666 women (Arenas 2007; Bungum 2003; Campo 2010; Carrasco 2013; Chatziioannou 2010; Gimeno 2006; Hazlett 2003; Jamieson 1997; Khan 2004; Mauri 2001; Mendoza 2003; Paternot 2010; Rubino 2004; Sepulveda 2009; Shih 2014Stevens 2000; Zollner 2004); three studies randomised 1018 cycles (Cano 2001; Barak 1998; Von During 2004); and twelve studies randomised 15,230 oocytes or embryos (Artini 2004; Baum 2004; Bird 2012; Chen 2009; Fechtali 2004; Findikli 2004; Kyono 2000; Lambert 2005; Mayer 2003; Sifer 2009; Tedesco 1990; Yamamoto 2006). Three of the studies that randomised oocytes did not report their sample size (Baum 2004; Lambert 2005; Yamamoto 2006).
Sampling methods varied: two studies randomised all women who visited their centre (Bungum 2003; Von During 2004); fourteen studies had restrictive inclusion criteria (Artini 2004; Bird 2012; Campo 2010; Carrasco 2013; Chatziioannou 2010; Chen 2009; Findikli 2004; Gimeno 2006; Lambert 2005; Mayer 2003; Paternot 2010; Sepulveda 2009; Sifer 2009; Zollner 2004); and it was unclear in the rest of the studies.
Maternal age was reported in seventeen of the studies (Artini 2004; Barak 1998; Bird 2012; Campo 2010; Carrasco 2013; Findikli 2004; Gimeno 2006; Hazlett 2003; Lambert 2005; Mauri 2001; Mendoza 2003; Paternot 2010; Rubino 2004; Sepulveda 2009; Sifer 2009; Stevens 2000; Zollner 2004). The mean age for women across these studies was 33.7 years, with a range from 25.6 to 39.3 years. The mean paternal age was 34.9 years, and was reported in two studies (Zollner 2004; Findikli 2004). The mean number of oocytes retrieved was 11.2, reported in eleven studies (Arenas 2007; Artini 2004; Campo 2010; Findikli 2004; Hazlett 2003; Lambert 2005; Mauri 2001; Mendoza 2003; Paternot 2010; Rubino 2004; Zollner 2004). The mean duration of subfertility was five years, with a range of 3.8 to 8.3 years, and was reported in five studies (Mendoza 2003; Zollner 2004; Findikli 2004; Artini 2004; Sifer 2009).
Interventions
Intracytoplasmic sperm injection (ICSI) was performed in five studies (Arenas 2007; Findikli 2004; Khan 2004; Kyono 2000; Mauri 2001); IVF was performed in five studies (Chen 2009; Hazlett 2003; Mayer 2003; Stevens 2000; Tedesco 1990); five studies did not report the fertilisation method used (Bird 2012; Bungum 2003; Cano 2001; Jamieson 1997; Yamamoto 2006); and both IVF and ICSI were performed in the rest of the studies.
Two studies reported on donor oocytes only (Gimeno 2006; Sepulveda 2009); one study used both non‐donor and donor oocytes (Cano 2001); and two studies used only non‐donor oocytes (Campo 2010; Paternot 2010). For the remaining studies, the use of donor oocytes was unclear. None of the studies commented on frozen‐thawed cycles.
Fifteen studies performed the transfer during the cleavage‐stage embryo development (day two or three; Artini 2004; Barak 1998; Baum 2004; Campo 2010; Carrasco 2013; Chatziioannou 2010; Chen 2009; Hazlett 2003; Jamieson 1997; Khan 2004; Mauri 2001; Rubino 2004; Sifer 2009; Stevens 2000; Von During 2004); five studies performed the transfer during the blastocyst‐stage embryo development (day four to six; Bird 2012; Findikli 2004; Kyono 2000; Lambert 2005; Sepulveda 2009); six studies performed transfers during both the cleavage and blastocyst stage (Gimeno 2006; Mayer 2003; Mendoza 2003; Paternot 2010; Yamamoto 2006; Zollner 2004); while the day of transfer was unknown in five studies (Arenas 2007; Bungum 2003; Cano 2001; Fechtali 2004; Tedesco 1990). The data from these studies have been analysed separately, according to the timing of the intervention. All included studies transferred fresh embryos. All studies transferred multiple embryos per treatment cycle.
For late embryo transfer, one study used continuous interrupted (renewed) single medium versus sequential media (Sepulveda 2009); three studies used continuous uninterrupted single medium versus sequential media (Bird 2012; Findikli 2004, Paternot 2010); and seven studies used sequential media (Gimeno 2006; Kyono 2000; Lambert 2005; Mayer 2003; Mendoza 2003; Yamamoto 2006; Zollner 2004).
Culture media from twelve commercial companies were represented in the review. The companies represented and their included media were: MediCult ‐ ISM, BlastAssist, IVF (Barak 1998; Campo 2010; Cano 2001; Fechtali 2004; Findikli 2004; Jamieson 1997; Lambert 2005; Mendoza 2003; Sifer 2009; Von During 2004; Yamamoto 2006; Zollner 2004); Irvine Scientific ‐ P1, HTF, ECM, MultiBlast, SSM (Artini 2004; Barak 1998; Baum 2004; Chen 2009; Hazlett 2003; Khan 2004; Mauri 2001; Mayer 2003; Sepulveda 2009; Yamamoto 2006); Vitrolife ‐ IVF, CCM,G2, G3, G5 (Arenas 2007; Bungum 2003; Carrasco 2013; Chatziioannou 2010; Findikli 2004; Lambert 2005; Mayer 2003; Mendoza 2003; Shih 2014; Sifer 2009; Stevens 2000; Zollner 2004); Gynemed ‐ GM501 (Campo 2010; Paternot 2010); Cook ‐ Sydney IVF (Baum 2004; Mendoza 2003; Paternot 2010; Von During 2004); Scandinivian IVF ‐ IVF (Cano 2001; Jamieson 1997; Mauri 2001; Rubino 2004); IVF Online ‐ Global (Bird 2012; Sepulveda 2009); In Vitro Care ‐ IVC (Arenas 2007; Hazlett 2003); Sage ‐ Quinn's advantage (Bird 2012; Chatziioannou 2010; Mayer 2003; Rubino 2004; Shih 2014); Gibco ‐ Ham's F10 (Stevens 2000); FertiPro ‐ Ferticult (Fechtali 2004); and Api‐System (Tedesco 1990). SeeTable 2
1. Types of culture media used in the included studies.
| Culture media | Manufacturer | Type of medium | Availability |
| BlastAssist | Medicult | S | Yes |
| ECMS | Irvine scientific | S | Yes |
| Ferticult | Fertipro | S | Yes |
| G2 | Vitrolife | S | No |
| G3 | Vitrolife | S | No |
| G5 | Vitrolife | S | Yes |
| GM501 | Gynemed | G | Yes |
| Global | IVF Online | G | Yes |
| Ham's F‐10 | Gibco | G | Yes |
| HTF | Irvine Scientific | G | Yes |
| IVC | In Vitro Care | S | Yes |
| IVF‐50 | Scandinavian IVF | S | Yes |
| ISM | Medicult | S | Yes |
| IVF medium | Vitrolife | S | Yes |
| IVF | Scandinavian IVF | S? | Yes |
| K‐SICM | Cook | S | Yes |
| K‐SIBM | Cook | S | Yes |
| K‐SIFM | Cook | S | Yes |
| M3 | Medicult | S | Yes |
| Menezo‐B | Api‐System | G? | Yes |
| Multiblast | Irvine Scientific | S | Yes |
| P1 | Irvine Scientific | S | Yes |
| Quinn's Advantage | SAGE | S | Yes |
| Sydney IVF | Cook | S? | Yes |
| Universal IVF | Medicult | G | Yes |
Outcomes
Primary clinical outcomes
Seven of the thirty two studies reported our primary outcomes. Five reported live birth rate (Campo 2010; Carrasco 2013; Gimeno 2006; Paternot 2010; Zollner 2004), and four reported ongoing pregnancy rate (Campo 2010; Paternot 2010; Sepulveda 2009; Stevens 2000).
One of these studies reported data that did not appear reliable, as reported live birth rates were higher than clinical pregnancy rates (Gimeno 2006). Therefore, these data were not entered into data tables. Attempts have been made to contact the study authors for clarification.
Secondary clinical outcomes
One study reported on congenital abnormalities or birth weight of babies born. However, the data could not be used as a different culture medium (IVF or G3) from the same commercial company (Vitrolife) was used for IVF and ICSI, but the results were presented together (Carrasco 2013).
Thirteen studies reported clinical pregnancy (Campo 2010; Carrasco 2013; Chatziioannou 2010; Gimeno 2006; Hazlett 2003; Mayer 2003; Paternot 2010; Rubino 2004; Sepulveda 2009; Sifer 2009; Von During 2004; Yamamoto 2006; Zollner 2004), but only seven were included in the analysis. Three studies were excluded since the exact numbers of participants were not reported (Hazlett 2003; Rubino 2004; Von During 2004); three more were excluded because the unit of analysis (women or cycles) was different from the unit of randomisation (oocytes; Mayer 2003; Sifer 2009; Yamamoto 2006).
Seven studies reported pregnancy rates without providing a definition, so it was not possible to assess whether the pregnancy was biochemical, clinical, or ongoing (Arenas 2007; Artini 2004; Barak 1998; Cano 2001; Jamieson 1997; Khan 2004; Mauri 2001).
Four studies reported multiple pregnancies (Zollner 2004, Paternot 2010; Sifer 2009;Carrasco 2013 ); three were included in the analysis. One study was not included as the unit of analysis (women or cycles) was different than the unit of randomisation (oocytes; Sifer 2009).
Seven studies reported miscarriage (Campo 2010; Cano 2001; Carrasco 2013; Mauri 2001; Paternot 2010; Sepulveda 2009; Zollner 2004). One study was not included in the analysis as the time of transfer was not known, precluding subgroup analysis (Cano 2001).
Fifteen studies reported implantation, but none were included in the analysis (Arenas 2007; Artini 2004; Campo 2010; Cano 2001; Gimeno 2006; Hazlett 2003; Mauri 2001; Mendoza 2003; Paternot 2010; Rubino 2004; Sepulveda 2009; Sifer 2009; Stevens 2000; Von During 2004; Zollner 2004). Nine of the studies did not report the numbers in sufficient detail for inclusion in this outcome (Arenas 2007; Cano 2001; Gimeno 2006; Hazlett 2003; Mendoza 2003; Rubino 2004; Sifer 2009; Stevens 2000; Von During 2004); in six studies, the units of analysis (oocytes or embryos) were different from the units of randomisation (embryos, women or cycles; Artini 2004; Campo 2010; Mauri 2001; Paternot 2010; Sepulveda 2009; Zollner 2004).
Three studies reported cryopreservation rate, but none were included in the analysis. One study was excluded since the exact numbers were not reported (Khan 2004); in two, the unit of analysis (embryos) was different from the unit of randomisation (women or cycles; Campo 2010; Von During 2004).
The number of good quality embryos obtained was reported in eighteen studies, but none were included in the analysis (Arenas 2007; Artini 2004; Baum 2004; Bird 2012; Cano 2001; Chatziioannou 2010; Chen 2009; Fechtali 2004; Findikli 2004; Jamieson 1997; Mendoza 2003; Paternot 2010; Rubino 2004; Sepulveda 2009; Sifer 2009; Von During 2004; Yamamoto 2006; Zollner 2004). Twelve studies were excluded because the exact numbers were not reported (Arenas 2007; Baum 2004; Bird 2012; Cano 2001; Chatziioannou 2010; Fechtali 2004; Findikli 2004; Jamieson 1997; Rubino 2004; Sepulveda 2009; Sifer 2009; Zollner 2004); in two, the unit of analysis (embryos) was different from the unit of randomisation (oocytes, women or cycles; Paternot 2010; Von During 2004).
Fertilisation rate was reported in twenty‐two studies; none were included in the analysis (Arenas 2007; Artini 2004; Barak 1998; Baum 2004; Campo 2010; Chatziioannou 2010; Fechtali 2004; Findikli 2004; Gimeno 2006; Hazlett 2003; Jamieson 1997; Khan 2004; Lambert 2005; Mauri 2001; Mayer 2003; Mendoza 2003; Paternot 2010; Rubino 2004; Sepulveda 2009; Sifer 2009; Tedesco 1990; Zollner 2004). One study was excluded since embryos were cultured in the same medium until assessment of fertilisation (Sepulveda 2009), ten studies were excluded because the exact numbers of embryos or oocytes were not reported (Arenas 2007; Baum 2004; Chatziioannou 2010; Fechtali 2004; Hazlett 2003; Jamieson 1997; Khan 2004; Lambert 2005; Rubino 2004; Sifer 2009), and in six more, the unit of analysis (embryos or oocytes) was different from the unit of randomisation (women or cycles; Barak 1998; Campo 2010; Gimeno 2006; Mauri 2001; Paternot 2010; Zollner 2004).
One study compared culture media and aimed to determine the correlation between levels of reactive oxygen species in different media. It was included for completeness, although it did not report any outcomes of interest to this review (Shih 2014).
Excluded studies
One hundred studies (112 articles) were excluded from the review for the following reasons:
16 were not relevant to the topic under study
15 were retrospective studies
3 were reviews
53 were quasi randomised studies
12 were not comparative studies
1 was conducted in mice
See Characteristics of excluded studies table for details.
Risk of bias in included studies
The risk of bias was unclear for the majority of the studies due to poor reporting in the available articles. We received some missing data from the authors of included studies, but in many cases, the risk of bias still remained unclear (Figure 2; Figure 3).
2.
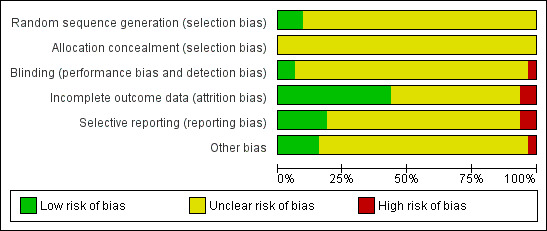
Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
3.
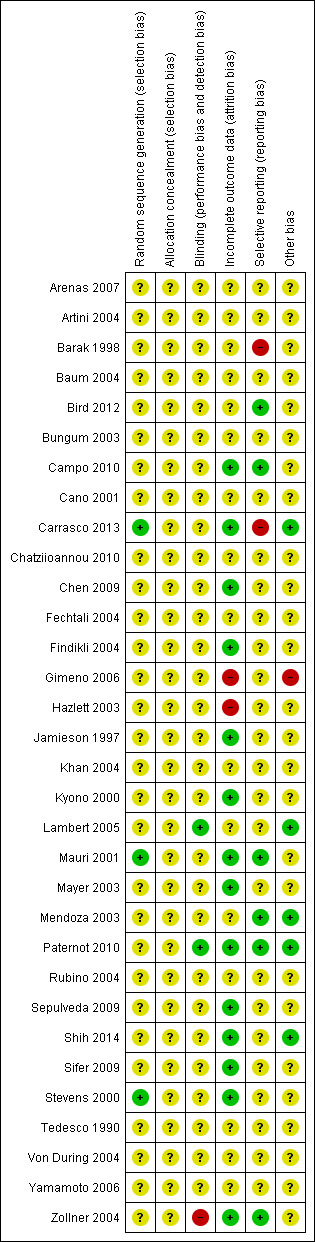
Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
Generation of random sequence
Three studies described satisfactory methods of sequence generation and were rated at low risk of bias in this domain (Carrasco 2013; Mauri 2001; Stevens 2000). Twenty‐nine studies did not describe sequence generation and were deemed at an unclear risk of this bias.
Allocation concealment
All studies were rated at an unclear risk of this bias, as they either failed to mention allocation concealment, or did not report sufficient details about the method used.
Blinding
Two studies reported use of blinding of either study personnel (Paternot 2010), or outcome assessors (Lambert 2005), and were rated at low risk of bias related to blinding. One study did not perform blinding and was rated at a high risk of bias related to blinding (Zollner 2004). The other studies were rated as unclear risks. Blinding is unlikely to influence our primary review outcome (live birth), but may be relevant for subjective outcomes such as assessment of embryo quality.
Incomplete outcome data
Fourteen studies reported complete or almost complete outcome data, with few or no losses to follow‐up (Campo 2010; Chen 2009; Findikli 2004; Jamieson 1997; Kyono 2000; Mauri 2001; Mayer 2003; Paternot 2010; Sepulveda 2009; Shih 2014; Sifer 2009; Stevens 2000; Tedesco 1990; Zollner 2004). Two studies were rated at high risk of attrition bias (Carrasco 2013; Gimeno 2006). The other sixteen studies had an attrition rate of up to 20%, or (more commonly) did not clearly report the denominator for their reported results. These studies were rated as unclear risks of attrition bias.
Selective reporting
Six studies reported all expected outcomes and were rated at low risk of this bias (Bird 2012; Campo 2010; Mauri 2001; Mendoza 2003; Paternot 2010; Zollner 2004). Two studies were rated at high risk of bias in this domain because results were not reported for all prespecified outcomes (Barak 1998; Carrasco 2013) . All other studies were rated as unclear risks of bias because too few details were reported to assess the risk of selective reporting bias.
Other potential sources of bias
Five studies were rated at low risk of other potential sources of bias (Carrasco 2013; Lambert 2005; Mendoza 2003; Paternot 2010; Shih 2014). One was rated at high risk of other potential bias as reported ongoing pregnancy rates were lower than live birth rates Gimeno 2006.
Twenty nine studies were rated as unclear risks of bias, due to poor or insufficient reporting of methods. Nineteen of these studies were published only as abstracts.
Effects of interventions
See: Table 1
Different culture media and various outcome measures were assessed in each study. A meta‐analysis was not possible but a forest plot is provided as a visual summary of the different comparisons and their effects.
Primary outcome
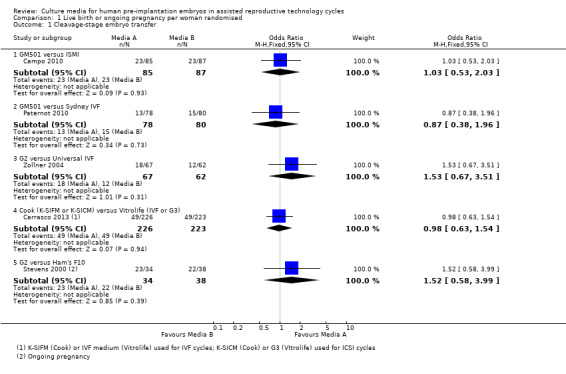
1 Live birth or ongoing pregnancy per woman randomised
Seven studies reported live birth or ongoing pregnancy.
Five reported live birth rate (Campo 2010; Carrasco 2013; Gimeno 2006; Paternot 2010; Zollner 2004) and two reported ongoing pregnancy (Sepulveda 2009; Stevens 2000); all compared different media.
None of the studies included in our analysis found a difference between the groups (Figure 4; Figure 5)
4.

Forest plot of comparison: 1 Live birth or ongoing pregnancy per woman randomised, outcome: 1.1 Cleavage‐stage embryo transfer.
5.
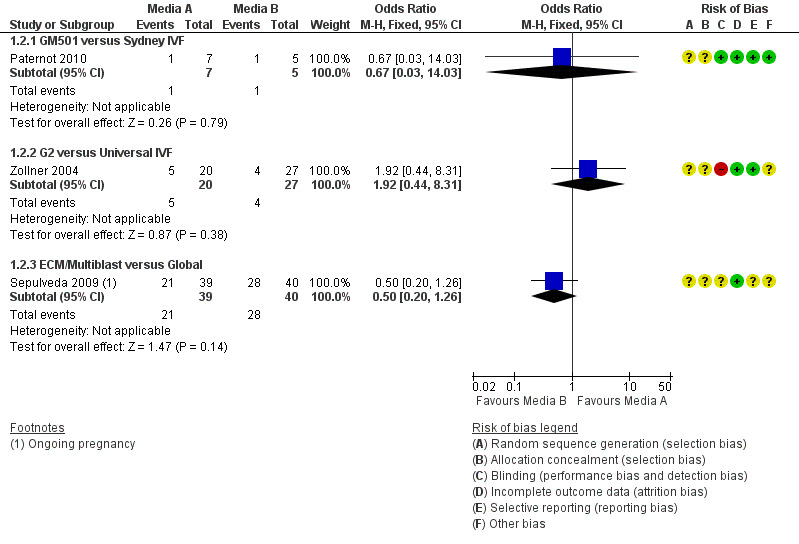
Forest plot of comparison: 1 Live birth or ongoing pregnancy per woman randomised, outcome: 1.2 Blastocyst‐stage embryo transfer.
One study reported that there was no difference between the live birth rate per woman when HTF was compared with ISM‐1 in cleavage‐stage embryo transfers (27/44 versus 21/47), but that CCM was associated with a higher live birth rate than ISM‐2 in blastocyst‐stage transfers (11/12 versus 6/15 ). These data were not included in our analysis because they did not appear reliable, as clinical pregnancy rates reported in this study were lower than the live birth rates (Gimeno 2006). We have sought clarification from the study authors.
Secondary outcomes
1. Health of babies born
No studies reported this outcome.
2. Clinical pregnancy rate (CPR) per randomised woman
Six studies were included in the analysis for clinical pregnancy (Campo 2010; Chatziioannou 2010; Gimeno 2006; Paternot 2010; Sepulveda 2009; Zollner 2004). One reported more clinical pregnancies in the group whose embryos were cultured in Quinn's advantage than in those whose embryos were cultured in G5 (Chatziioannou 2010). The rest of the studies found no evidence of a difference between the compared media for either cleavage‐ or for blastocyst‐embryo transfer (Figure 6; Figure 7).
6.
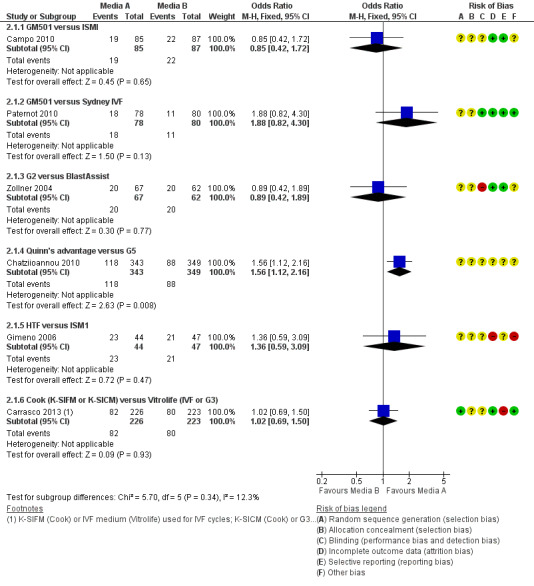
Forest plot of comparison: 2 Clinical pregnancy rate per woman randomised, outcome: 2.1 Cleavage‐stage embryo transfer.
7.
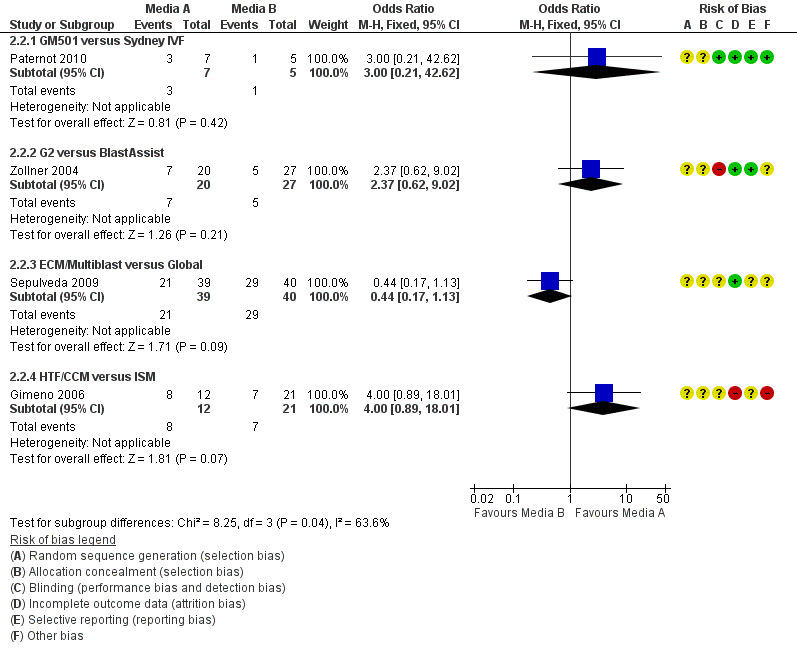
Forest plot of comparison: 2 Clinical pregnancy rate per woman randomised, outcome: 2.2 Blastocyst‐stage embryo transfer.
3. Multiple pregnancy rate per woman randomised
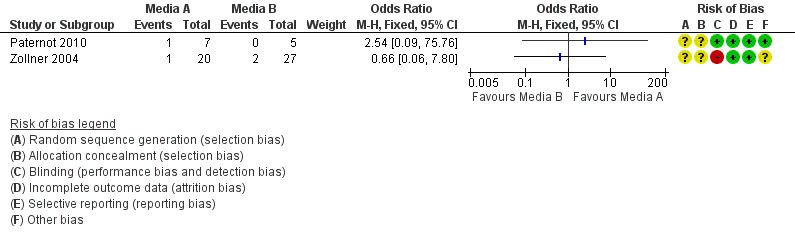
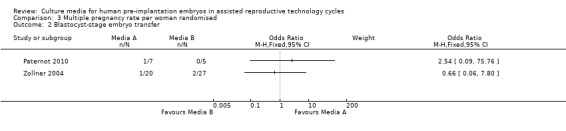
Three studies were included in the analysis for multiple pregnancies (Paternot 2010; Zollner 2004;Carrasco 2013 ), none of which found evidence of a difference between the compared media for either day three or day five embryo transfers (Figure 8; Figure 9).
8.
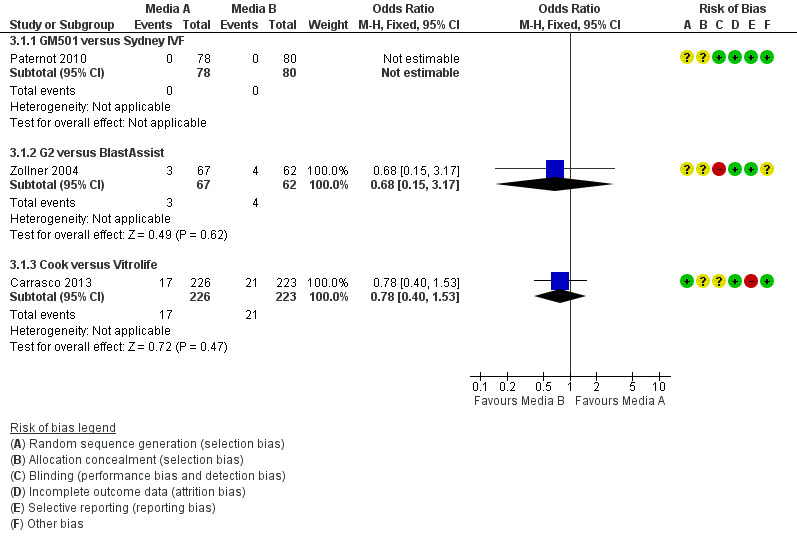
Forest plot of comparison: 3 Multiple pregnancy rate per woman randomised, outcome: 3.1 Cleavage‐stage embryo transfer.
9.
Forest plot of comparison: 3 Multiple pregnancy rate per woman randomised, outcome: 3.2 Blastocyst‐stage embryo transfer.
4. Miscarriage rate per woman randomised
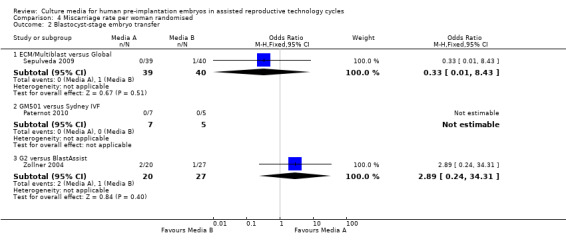
Five studies were included in the analysis for miscarriage (Campo 2010; Mauri 2001; Paternot 2010; Sepulveda 2009; Zollner 2004). None found evidence of a difference between the compared media for either day three or day five embryo transfers (Figure 10; Figure 11).
10.

Forest plot of comparison: 4 Miscarriage rate per woman randomised, outcome: 4.1 Cleavage‐stage embryo transfer.
11.

Forest plot of comparison: 4 Miscarriage rate per woman randomised, outcome: 4.2 Blastocyst‐stage embryo transfer.
5. Implantation rate per embryo transferred
One study that randomised oocytes reported implantation rate per embryo transferred (Artini 2004; Table 3). We reported the results for studies that randomised women in Table 4 and Table 5 (Campo 2010; Mauri 2001; Paternot 2010; Sepulveda 2009; Zollner 2004). One study reported that Global was more effective than ECM/MultiBlast (Sepulveda 2009); the rest of the studies found no difference between the groups.
2. Implantation rate per embryo transferred in study that randomised oocytes.
| Comparison | Study | Events Media A | Events Media B | OR (95% CI) |
| HTF versus PI | Artini 2004 | 10/153 | 19/159 | 0.52 (0.23 to 1.15) |
3. Implantation rate per embryos transferred in studies randomising women after cleavage stage embryo transfer.
| Comparison | Study | Events Media A | Events Media B | OR (95% CI) |
| GM501 versus ISM1 | Campo 2010 | 26/130 | 22/124 | 1.16 (0.62 to 2.18) |
| P1 versus IVF | Mauri 2001 | 44/252 | 38/240 | 1.12 (0.70 to 1.81) |
| GM501 versus Sydney IVF | Paternot 2010 | 18/69 | 13/66 | 1.44 (0.64 to 3.24) |
| G2 versus BlastAssist | Zollner 2004 | 31/235 | 31/239 | 1.02 (0.60 to 1.74) |
4. Implantation rate per embryos transferred in studies randomising women after blastocyst stage embryo transfer.
| Comparison | Study | Events Media A | Events Media B | OR (95% CI) |
| GM501 versus Sydney IVF | Paternot 2010 | 3/7 | 1/5 | 3.00 (0.21 to 42.62) |
| Global versus ECM/MultiBlast | Sepulveda 2009 | 46/80 | 28/76 | 2.32 (1.21 to 4.41) |
6. Cryopreservation rate per total number of embryos
None of the studies randomising oocytes or embryos reported cryopreservation rate. We reported results for the two studies that randomised women in Table 6. Von During 2004 reported a difference between the media compared, with Sydney IVF leading to more cryopreserved embryos than Universal IVF; Campo 2010 found no evidence of a difference between the compared media.
5. Cryopreservation rate per number of embryos in studies randomising women.
| Comparison | Study | Events Media A | Events Media B | OR (95% CI) |
| GM501 versus ISM1 | Campo 2010 | 181/595 | 177/652 | 1.17 (0.95 to 1.50) |
| Universal IVF versus Sydney IVF | Von During 2004 | 240/1004 | 445/1062 | 0.44 (0.36 to 0.53) |
7. Embryo quality per total number of embryos
Three studies that randomised oocytes reported embryo quality per total number of embryos (Artini 2004; Chen 2009; Yamamoto 2006; Table 7). We reported the results of studies that randomised women in Table 8 (Paternot 2010; Von During 2004). Four studies reported evidence that ECM was superior to PI (Chen 2009), Multiblast was superior to BlastAssist (Yamamoto 2006), Sydney IVF was superior to GM501 (Paternot 2010), and Sydney IVF was superior to Universal IVF (Von During 2004). There was no evidence of a difference between the media for the other comparisons (PI versus IVF).
6. Embryo quality per total number of embryos in studies randomising oocytes.
| Comparison | Study | Events Media A | Events Media B | OR (95% CI) |
| BlastAssist versus MultiBlast | Yamamoto 2006 | 236/320 | 225/277 | 0.65 (0.44 to 0.96) |
| P1 versus HTF | Artini 2004 | 194/320 | 194/294 | 0.79 (0.57 to 1.10) |
| P1 versus ECM | Chen 2009 | 139/655 | 168/627 | 0.74 (0.57 to 0.95) |
7. Embryo quality per total number of embryos in studies randomising women.
| Comparison | Study | Events Media A | Events Media B | OR (95% CI) |
| GM501 versus Sydney IVF | Paternot 2010 | 177/583 | 169/419 | 0.64 (0.50 to 0.84) |
| Universal IVF versus Sydney IVF | Von During 2004 | 527/1004 | 711/1062 | 0.55 (0.46 to 0.65) |
8. Fertilisation rate per total number of oocytes retrieved
We reported fertilisation rate per total number of oocytes retrieved from four studies that randomised oocytes in Table 9 (Artini 2004; Findikli 2004; Mayer 2003; Tedesco 1990). We reported the results for studies that randomised women in Table 10 (Barak 1998; Campo 2010; Gimeno 2006; Mauri 2001; Paternot 2010; Zollner 2004). Seven studies reported a difference between the media compared: GM501 was superior to Sydney IVF (Paternot 2010); G2 was superior to BlastAssist (Zollner 2004); PI was superior to HTF and M3 (Barak 1998); and Universal IVF was superior to P1, HTF and M3 (Barak 1998). There was no evidence of a difference between the media in the other ten comparisons.
8. Fertilization rate per total number of oocytes retrieved in studies randomising oocytes.
| Comparison | Study | Events Media A | Events Media B | OR (95% CI) |
| HTF versus Ham's F10 | Tedesco 1990 | 17/25 | 18/26 | 0.94 (0.29 to 3.08) |
| P1 versus HTF | Artini 2004 | 320/512 | 294/502 | 1.18 (0.92 to 1.52) |
| Menzo B versus Ham's F10 | Tedesco 1990 | 22/40 | 28/51 | 1.00 (0.44 to 2.31) |
| P1 versus G2 | Mayer 2003 | 864/1182 | 821/1146 | 1.08 (0.90 to 1.29) |
| P1 versus Quninn's Advantage | Mayer 2003 | 321/453 | 324/430 | 0.80 (0.59 to 1.07) |
| ISM versus G2 | Findikli 2004 | 524/717 | 520/717 | 1.03 (0.82 to 1.03) |
9. Fertilization rate per total number of oocytes retrieved in studies randomising women.
| Comparison | Study | Events Media A | Events Media B | OR (95% CI) |
| P1 versus HTF | Barak 1998 | 222/315 | 138/225 | 1.50 (1.05 to 2.16) |
| P1 versus M3 | Barak 1998 | 222/315 | 250/447 | 1.88 (1.39 to 2.55) |
| P1 versus Universal IVF | Barak 1998 | 222/315 | 452/578 | 0.67 (0.49 to 0.91) |
| HTF versus M3 | Barak 1998 | 138/225 | 250/447 | 1.25 (0.70 to 1.93) |
| HTF versus Universal IVF | Barak 1998 | 138/225 | 452/578 | 0.44 (0.32 to 0.62) |
| M3 versus Universal IVF | Barak 1998 | 250/447 | 452/578 | 0.35 (0.27 to 0.46) |
| GM501 versus ISM1 | Campo 2010 | 433/595 | 446/652 | 1.23 (0.97 to 1.58) |
| P1 versus IVF | Mauri 2001 | 725/965 | 751/1010 | 1.04 (0.85 to 1.28) |
| Sydney IVF versus GM501 | Paternot 2010 | 419/669 | 583/788 | 0.59 (0.47 to 0.74) |
| G2 versus BlastAssist | Zollner 2004 | 914/1044 | 712/890 | 1.76 (1.37 to 2.25) |
| HTF versus ISM1 | Gimeno 2006 | 316/455 | 411/567 | 0.88 (0.66 to 1.13) |
Sensitivity analyses
None of our planned subgroup or sensitivity analyses were possible since each study compared different media.
Discussion
Summary of main results
We included thirty‐two studies in this review. It was not possible to pool the results of any of the included studies since every study compared different media.
Overall we found no clear evidence of any difference between the media used, with respect to clinical outcomes including live birth rate, ongoing pregnancy rate, clinical pregnancy rate, multiple pregnancy rate or miscarriage rate. This is more likely to be due to a lack of good quality studies reporting on these outcomes, rather than a true absence of any difference. One large study (N = 692) reported that Quinn's Advantage was associated with more clinical pregnancies than G3 Chatziioannou 2010, However, this study was available only as an abstract, did not report live births or ongoing pregnancy, and was assessed to have an unclear risk of bias in all domains.
Overall completeness and applicability of evidence
Although live birth is considered to be the most important outcome for in vitro fertilisation (IVF) or intracytoplasmic sperm injection (ICSI) treatment cycles, it was only reported in five of the thirty‐two included studies. Ongoing pregnancy was reported in four studies. There were also limited data on clinical pregnancy and adverse events. Because of the different comparison groups and the lack of data for important clinical outcomes, a firm conclusion regarding the objectives of this review was impossible.
None of the studies commented on the use of frozen‐thawed embryos. This could lead to bias in the interpretation of the results if one medium were to culture more top quality embryos. The same pregnancy rates might be obtained if only fresh transfers were considered, but better results might be obtained when cumulative rates, including frozen‐thawed cycles, were considered.
Recent controlled studies have indicated an effect of culture media on the birth weight of the newborn (Dumoulin 2010; Kleijkers 2014; Nelissen 2012), but to date, no RCT has reported on these outcomes.
Quality of the evidence
The included studies contained methodological limitations such as an unclear randomisation protocol, randomisation of oocytes and embryos rather than women, and small sample sizes. None of the studies provided adequate description of their methodology (e.g. allocation concealment, blinding, attrition rate). Most studies (22/32) failed to report their source of funding. The overall quality of the evidence for the primary review outcome was assessed as low or very low for all comparisons, using Grades of Recommendation, Assessment, Development and Evaluation (GRADE) criteria: See Table 1.
The baseline characteristics of randomised women and the composition of the media used were not reported in most of the studies. Not all studies reported on all outcomes, and outcome reporting was poor in many studies,. Several studies reported percentages or means without providing the raw data. Quality scoring of the embryos lacked detail and clarity, resulting in the exclusion of many potentially informative data.
We excluded the results from pregnancy outcomes from studies that randomised oocytes or embryos from our review. When oocytes and embryos are the units of randomisation, oocytes and embryos from one participant are randomised to different media, and only the best morphological embryos are selected for transfer. This design introduces bias since the morphology of the embryos that are transferred might depend on the medium used, but the analysis of the clinical outcomes is based on the number of women. It is impossible to calculate the live birth and pregnancy rates per woman from trials that analysed outcomes per oocyte or embryo.
Other factors during embryo culture could also influence IVF and ICSI success rates. Such factors include the number of embryos per drop, and culture dish, patient selection and characteristics, stimulation protocols, oxygen concentration during culture, temperature or pH monitoring (Hoelker 2010).
Potential biases in the review process
Our analyses were done on an intention‐to‐treat basis as far as possible; original investigators were contacted for missing data.
Embryo data from trials that randomised women were extracted and are provided in additional tables. We acknowledge the potential confounding effect of multiple oocytes or embryos from one woman or cycle, but this only concerned the secondary outcomes of our review. However, had we excluded these trials, very limited data would have been available for review.
Agreements and disagreements with other studies or reviews
We have previously published a review on the same topic, but in that review we applied less stringent inclusion criteria (Mantikou 2013). We included quasi randomised trials, while they were excluded in this review, and we conducted an unconventional hypothesis‐generating meta‐analysis by combining data from studies that shared the same medium in one of the treatment arms. There was also a stronger focus on embryo parameters. The current review focuses more on clinical outcomes and follows stricter methodological quality criteria, including only properly randomised controlled trials. The conclusions of Mantikou 2013 were consistent with those of the current review.
Authors' conclusions
Implications for practice.
No two studies compared the same culture media and none of them found any evidence of a difference between the culture media used. We conclude that there is insufficient evidence to support or refute the use of any specific culture medium.
Implications for research.
More good quality studies are needed to clarify whether culture media have an effect on in vitro fertilisation (IVF) or intracytoplasmic sperm injection (ICSI) success rates and which culture medium has the best rates. Changing the scope from embryological to clinical outcomes, including neonatal outcomes, performing and clearly reporting proper methods of randomisation and allocation concealment, and reporting clear data by providing exact numbers, together with percentages, means and variances will lead to better quality data being available for meta‐analysis. All studies should clearly disclose their source of funding. Authors are also encouraged to publish a full report of their studies, rather than only publishing abbreviated abstracts that do not allow the data to be included in meta‐analyses.
The lack of good quality studies is surprising. The potential effect of culture media on the success rate of an IVF programme and the increasing indication of a potential effect of culture media on the health of newborns, highlight the need for reliable studies on these topics. Another issue is the lack of information on the composition of the available culture media, as this information is often not disclosed by the manufacturers. Apart from the need to know what is in the media, there is a need for research on the effect of the different components on the safety and effectiveness of IVF or ICSI treatment.
Given the number of available culture media and the available comparisons, a network meta‐analysis, a method used to assess the comparative effectiveness of experimental treatment among similar patient populations that have not been compared directly in a randomised controlled trial, could be a useful research project.
History
Protocol first published: Issue 3, 2009 Review first published: Issue 11, 2015
| Date | Event | Description |
|---|---|---|
| 19 November 2008 | Amended | Title edited to 'Culture media for human preimplantation embryos for in assisted reproductive technology cycles' from 'Media for culturing human embryos for artificial reproductive technology cycles' |
| 24 September 2007 | New citation required and major changes | Substantive amendment |
Acknowledgements
The authors gratefully acknowledge: Dr. Ben‐Yosef, Dr. Bisioli, Dr. Grainger, Dr. Hashimoto, Dr. Meintjes, Dr. Mortimer, Dr. Parinaud, Dr. Reed, Dr. Sifer, Dr. Sieren, Dr. Summers‐Chase, Dr. Ubaldi, Dr. Van den Bergh, Dr. Van Langendonckt, Dr. Vansteenbruge, Dr. Dumoulin, Dr. Paternot, Dr. Binda, Dr Yakin, Dr. Khoury, Dr. Battaglia, Dr. Spiessens, Dr. Torres and Dr. Stavreus‐Evers, all authors of included or excluded studies for supplying additional information upon our request. Finally, special thanks to the highly supportive team at the Cochrane office in Auckland: Review Group Coordinators Helen Nagels (present) and Jane Clarke (previous), Trials Search Co‐ordinator Marian Showell, Editor Jane Marjoribanks, and Editor Anne Lethaby, who translated Gimeno 2006 for us.
Appendices
Appendix 1. MDSG
Menstrual Disorders and Subfertility (MDSG) database search strategy for MH1610 28.08.13
Keywords CONTAINS "IVF" or "in vitro fertilization" or "in‐vitro fertilisation" or "ICSI" or"intracytoplasmic sperm injection" or "Embryo" or "ET" or "*Embryo Transfer" or "in‐vitro fertilization" or "assisted reproduction"or "Blastocyst"or "oocyte " or Title CONTAINS"IVF" or "in vitro fertilization" or "in‐vitro fertilisation" or "ICSI" or"intracytoplasmic sperm injection" or "Embryo" or "ET" or "*Embryo Transfer" or "in‐vitro fertilization" or "assisted reproduction"or "Blastocyst"or "oocyte "
AND
Keywords CONTAINS "embryo culture" or "embryo culture media" or "Culture‐Media" or "culture" or "culture incubator"or"cumulus coculture"or"blastocyst culture technique"or"blastocyst media"or"media"or"G1"or"G1.2"or"G2"or"G2.2 sequential"or"Medicult"or"Medicult Sequential Medium"or"Vitrolife"or"sequential culture" or"sequential media"or "fetal bovine serum"or"fetal cord serum"or"P1"or "P1 culture medium"or"human tubal fluid"or"SMART"or"G3 culture media"or"BM1 medium"or"human tubal fluid"or"sydney ivf medium"or"Sage culture medium"or"Quinns Advantage sequential culture media"or"single media"or"hams f10"or "oocyte culture"or Title CONTAINS "embryo culture"or"embryo culture media"or "Culture‐Media"or"culture"or"culture incubator"or"cumulus coculture"or"blastocyst culture technique"or"blastocyst media"or"media"or"G1"or"G1.2"or"G2"or"G2.2 sequential"or"Medicult"or"Medicult Sequential Medium"or"Vitrolife"or"sequential culture"or"sequential media"or "fetal bovine serum"or"fetal cord serum"or"P1"
Appendix 2. Cochrane Central Register of Controlled Trials
Database: EBM Reviews ‐ Cochrane Central Register of Controlled Trials <January 2014> Search Strategy: ‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐ 1 exp embryo transfer/ or exp fertilization in vitro/ or exp sperm injections, intracytoplasmic/ (1655) 2 embryo$.tw. (2271) 3 (vitro fertilisation or vitro fertilization).tw. (1437) 4 (ivf or icsi).tw. (2423) 5 intracytoplasmic sperm injection$.tw. (466) 6 assisted reproducti$.tw. (445) 7 blastocyst$.tw. (274) 8 oocyte$.tw. (1600) 9 or/1‐8 (4513) 10 exp Culture Media/ (250) 11 Culture Media$.tw. (121) 12 exp Embryo Culture Techniques/ (40) 13 (Embryo$ adj5 Culture$).tw. (249) 14 (Embryo$ adj5 medi$).tw. (149) 15 fet$ umbilic$ cord$.tw. (9) 16 (oocyte$ adj5 Culture$).tw. (65) 17 (oocyte$ adj5 medi$).tw. (70) 18 bovine serum$.tw. (62) 19 P1 medi$.tw. (7) 20 BM1 medium.tw. (3) 21 HTF.tw. (29) 22 ISM1.tw. (3) 23 Human tubal fluid.tw. (20) 24 medicult.tw. (11) 25 Menezo B2.tw. (6) 26 vitrolife.tw. (9) 27 monoculture$.tw. (5) 28 (blastocyst$ adj5 culture$).tw. (74) 29 (blastocyst$ adj5 medi$).tw. (35) 30 homemade medi$.tw. (0) 31 (commercial$ adj2 media).tw. (11) 32 Universal IVF Medi$.tw. (1) 33 FCS.tw. (34) 34 (g1 2 or g2 2 or g3 or g5).tw. (373) 35 SMART2.tw. (2) 36 (Sydney IVF adj5 med$).tw. (4) 37 Earle`s balanced salt solution.tw. (7) 38 monoculture.tw. (3) 39 sage.tw. (123) 40 GIII Series$.tw. (4) 41 Quinn$ Advantage$.tw. (1) 42 (sequential adj2 medi$).tw. (74) 43 universal ivf medi$.tw. (1) 44 blastAssist system.tw. (0) 45 SCANDINAVIAN.tw. (594) 46 single medi$.tw. (61) 47 fetal cord serum.tw. (7) 48 (Ham?F10 or Ham$ F10).tw. (4) 49 Cook ivf.tw. (3) 50 or/10‐49 (1973) 51 randomised controlled trial.pt. (333965) 52 controlled clinical trial.pt. (83200) 53 randomized.ab. (187732) 54 placebo.tw. (128418) 55 clinical trials as topic.sh. (33027) 56 randomly.ab. (96217) 57 trial.ti. (115436) 58 (crossover or cross‐over or cross over).tw. (44677) 59 or/51‐58 (527665) 60 exp animals/ not humans.sh. (4) 61 59 not 60 (527665) 62 9 and 50 and 61 (300) 63 limit 62 to yr="2013 ‐Current" (7)
Appendix 3. Ovid MEDLINE
Database: Ovid MEDLINE(R) In‐Process & Other Non‐Indexed Citations, Ovid MEDLINE(R) Daily and Ovid MEDLINE(R) <1946 to Present> Search Strategy: ‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐ 1 exp embryo transfer/ or exp fertilization in vitro/ or exp sperm injections, intracytoplasmic/ (32136) 2 embryo$.tw. (270450) 3 (vitro fertilisation or vitro fertilization).tw. (16709) 4 (ivf or icsi).tw. (18757) 5 intracytoplasmic sperm injection$.tw. (4891) 6 assisted reproducti$.tw. (8922) 7 blastocyst$.tw. (16061) 8 oocyte$.tw. (54704) 9 or/1‐8 (329600) 10 exp Culture Media/ (131482) 11 Culture Media$.tw. (14236) 12 exp Embryo Culture Techniques/ (2158) 13 (Embryo$ adj5 Culture$).tw. (20167) 14 (Embryo$ adj5 medi$).tw. (5652) 15 fet$ umbilic$ cord$.tw. (99) 16 (oocyte$ adj5 Culture$).tw. (2604) 17 (oocyte$ adj5 medi$).tw. (2007) 18 bovine serum$.tw. (29620) 19 P1 medi$.tw. (135) 20 BM1 medium.tw. (3) 21 HTF.tw. (510) 22 ISM1.tw. (11) 23 Human tubal fluid.tw. (166) 24 medicult.tw. (17) 25 Menezo B2.tw. (25) 26 vitrolife.tw. (35) 27 monoculture$.tw. (1697) 28 (blastocyst$ adj5 culture$).tw. (1883) 29 (blastocyst$ adj5 medi$).tw. (663) 30 homemade medi$.tw. (7) 31 (commercial$ adj2 media).tw. (296) 32 Universal IVF Medi$.tw. (8) 33 FCS.tw. (4649) 34 (g1 2 or g2 2 or g3 or g5).tw. (7809) 35 SMART2.tw. (4) 36 (Sydney IVF adj5 med$).tw. (7) 37 Earle`s balanced salt solution.tw. (114) 38 monoculture.tw. (971) 39 sage.tw. (2485) 40 GIII Series$.tw. (2) 41 Quinn$ Advantage$.tw. (7) 42 (sequential adj2 medi$).tw. (548) 43 universal ivf medi$.tw. (8) 44 blastAssist system.tw. (2) 45 SCANDINAVIAN.tw. (6143) 46 single medi$.tw. (1097) 47 fetal cord serum.tw. (80) 48 (Ham?F10 or Ham$ F10).tw. (179) 49 Cook ivf.tw. (1) 50 or/10‐49 (216308) 51 randomised controlled trial.pt. (368102) 52 controlled clinical trial.pt. (87922) 53 randomized.ab. (288115) 54 placebo.tw. (156052) 55 clinical trials as topic.sh. (168783) 56 randomly.ab. (208984) 57 trial.ti. (123679) 58 (crossover or cross‐over or cross over).tw. (59911) 59 or/51‐58 (909667) 60 exp animals/ not humans.sh. (3905327) 61 59 not 60 (838111) 62 9 and 50 and 61 (487) 63 (201308$ or 201309$ or 201310$ or 201311$ or 201312$).ed. (394653) 64 2014$.ed. (215664) 65 (2013$ or 2014$).dp. (1197783) 66 63 or 64 or 65 (1291748) 67 62 and 66 (52)
Appendix 4. EMBASE
Database: Embase <1980 to 2014 Week 12> Search Strategy: ‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐ 1 exp embryo transfer/ or exp fertilization in vitro/ or exp sperm injections, intracytoplasmic/ (51614) 2 embryo$.tw. (286135) 3 (in vitro fertilisation or in vitro fertilization).tw. (20184) 4 (ivf or icsi).tw. (28361) 5 intracytoplasmic sperm injection$.tw. (6178) 6 assisted reproducti$.tw. (12809) 7 blastocyst$.tw. (18504) 8 oocyte$.tw. (61723) 9 or/1‐8 (357220) 10 exp Culture Media/ (114329) 11 Culture Media$.tw. (16031) 12 (Embryo$ adj5 Culture$).tw. (20223) 13 (Embryo$ adj5 medi$).tw. (6383) 14 (oocyte$ adj5 Culture$).tw. (2983) 15 (oocyte$ adj5 medi$).tw. (2328) 16 fet$ umbilic$ cord$.tw. (117) 17 bovine serum$.tw. (30868) 18 P1 medi$.tw. (115) 19 BM1 medium.tw. (3) 20 HTF.tw. (712) 21 Human tubal fluid.tw. (211) 22 medicult.tw. (77) 23 vitrolife.tw. (195) 24 monoculture$.tw. (1906) 25 blastocyst$ culture$.tw. (444) 26 blastocyst$ medi$.tw. (82) 27 homemade medi$.tw. (10) 28 (commercial$ adj2 medi$).tw. (1886) 29 FCS.tw. (5442) 30 (g1 2 or g2 2 or g3 or g5).tw. (11785) 31 SMART2.tw. (10) 32 (sequential adj2 medi$).tw. (730) 33 (Sydney IVF adj5 med$).tw. (10) 34 Quinn$ Advantage$.tw. (41) 35 Earle`s balanced salt solution.tw. (122) 36 monoculture.tw. (1089) 37 sage.tw. (18467) 38 universal ivf medi$.tw. (11) 39 blastAssist system.tw. (4) 40 GIII Series$.tw. (3) 41 SCANDINAVIAN.tw. (8493) 42 (sequential adj2 medi$).tw. (730) 43 single medi$.tw. (1416) 44 fetal cord serum.tw. (90) 45 (Ham?F10 or Ham$ F10).tw. (196) 46 Cook ivf.tw. (2) 47 embryo culture/ (5923) 48 or/10‐47 (225365) 49 9 and 48 (36054) 50 Clinical Trial/ (829332) 51 Randomized Controlled Trial/ (337936) 52 exp randomization/ (61409) 53 Single Blind Procedure/ (17980) 54 Double Blind Procedure/ (112169) 55 Crossover Procedure/ (38233) 56 Placebo/ (235705) 57 Randomi?ed controlled trial$.tw. (95389) 58 Rct.tw. (13307) 59 random allocation.tw. (1287) 60 randomly allocated.tw. (19716) 61 allocated randomly.tw. (1893) 62 (allocated adj2 random).tw. (707) 63 Single blind$.tw. (13886) 64 Double blind$.tw. (137780) 65 ((treble or triple) adj blind$).tw. (347) 66 placebo$.tw. (193066) 67 prospective study/ (243812) 68 or/50‐67 (1336346) 69 case study/ (24866) 70 case report.tw. (252953) 71 abstract report/ or letter/ (882275) 72 or/69‐71 (1154651) 73 68 not 72 (1299194) 74 49 and 73 (894) 75 (201308$ or 201309$ or 201310$ or 201311$ or 201312$).em. (109485) 76 2014$.em. (458708) 77 (2013$ or 2014$).dp. (226104) 78 75 or 76 or 77 (719826) 79 74 and 78 (39)
Appendix 5. PsycINFO
Database: PsycINFO <1806 to March Week 3 2014> Search Strategy: ‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐ 1 exp Reproductive Technology/ or exp Embryo/ (2597) 2 embryo$.tw. (7194) 3 (vitro fertilisation or vitro fertilization).tw. (529) 4 (ivf or icsi).tw. (390) 5 intracytoplasmic sperm injection$.tw. (36) 6 assisted reproducti$.tw. (523) 7 blastocyst$.tw. (47) 8 oocyte$.tw. (536) 9 or/1‐8 (9036) 10 Culture Medi$.tw. (444) 11 (Embryo$ adj2 Culture$).tw. (150) 12 (Embryo$ adj2 medi$).tw. (24) 13 fet$ umbilic$ cord$.tw. (0) 14 bovine serum$.tw. (133) 15 P1 medi$.tw. (2) 16 HTF.tw. (7) 17 Human tubal fluid.tw. (1) 18 medicult.tw. (0) 19 vitrolife.tw. (0) 20 monoculture$.tw. (63) 21 blastocyst$ culture$.tw. (0) 22 blastocyst$ medi$.tw. (0) 23 homemade medi$.tw. (0) 24 (commercial$ adj2 media).tw. (118) 25 FCS.tw. (190) 26 (g1 2 or g2 2 or g3 or g5).tw. (181) 27 (sequential adj2 medi$).tw. (52) 28 universal ivf medi$.tw. (0) 29 blastAssist system.tw. (0) 30 SCANDINAVIAN.tw. (1104) 31 or/10‐30 (2456) 32 9 and 31 (214) 33 random.tw. (40004) 34 control.tw. (310797) 35 double‐blind.tw. (17711) 36 clinical trials/ (7380) 37 placebo/ (3724) 38 exp Treatment/ (572587) 39 or/33‐38 (874097) 40 32 and 39 (50) 41 limit 40 to yr="2013 ‐Current" (7)
Data and analyses
Comparison 1. Live birth or ongoing pregnancy per woman randomised.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Cleavage‐stage embryo transfer | 5 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 GM501 versus ISMI | 1 | 172 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.53, 2.03] |
| 1.2 GM501 versus Sydney IVF | 1 | 158 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.38, 1.96] |
| 1.3 G2 versus Universal IVF | 1 | 129 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.53 [0.67, 3.51] |
| 1.4 Cook (K‐SIFM or K‐SICM) versus Vitrolife (IVF or G3) | 1 | 449 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.98 [0.63, 1.54] |
| 1.5 G2 versus Ham's F10 | 1 | 72 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.52 [0.58, 3.99] |
| 2 Blastocyst‐stage embryo transfer | 3 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 2.1 GM501 versus Sydney IVF | 1 | 12 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.67 [0.03, 14.03] |
| 2.2 G2 versus Universal IVF | 1 | 47 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.92 [0.44, 8.31] |
| 2.3 ECM/Multiblast versus Global | 1 | 79 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.5 [0.20, 1.26] |
1.1. Analysis.
Comparison 1 Live birth or ongoing pregnancy per woman randomised, Outcome 1 Cleavage‐stage embryo transfer.
1.2. Analysis.
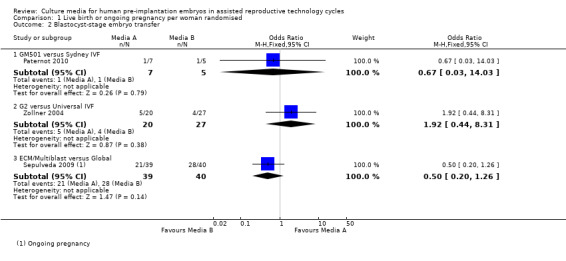
Comparison 1 Live birth or ongoing pregnancy per woman randomised, Outcome 2 Blastocyst‐stage embryo transfer.
Comparison 2. Clinical pregnancy rate per woman randomised.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Cleavage‐stage embryo transfer | 6 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 GM501 versus ISMI | 1 | 172 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.85 [0.42, 1.72] |
| 1.2 GM501 versus Sydney IVF | 1 | 158 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.88 [0.82, 4.30] |
| 1.3 G2 versus BlastAssist | 1 | 129 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.42, 1.89] |
| 1.4 Quinn's advantage versus G5 | 1 | 692 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.56 [1.12, 2.16] |
| 1.5 HTF versus ISM1 | 1 | 91 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.36 [0.59, 3.09] |
| 1.6 Cook (K‐SIFM or K‐SICM) versus Vitrolife (IVF or G3) | 1 | 449 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.69, 1.50] |
| 2 Blastocyst‐stage embryo transfer | 4 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 2.1 GM501 versus Sydney IVF | 1 | 12 | Odds Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.21, 42.62] |
| 2.2 G2 versus BlastAssist | 1 | 47 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.37 [0.62, 9.02] |
| 2.3 ECM/Multiblast versus Global | 1 | 79 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.44 [0.17, 1.13] |
| 2.4 HTF/CCM versus ISM | 1 | 33 | Odds Ratio (M‐H, Fixed, 95% CI) | 4.0 [0.89, 18.01] |
2.1. Analysis.
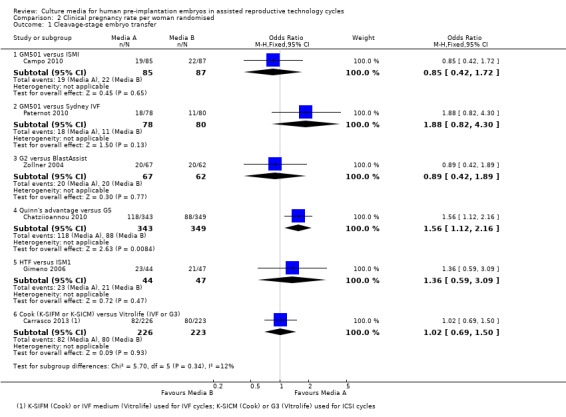
Comparison 2 Clinical pregnancy rate per woman randomised, Outcome 1 Cleavage‐stage embryo transfer.
2.2. Analysis.
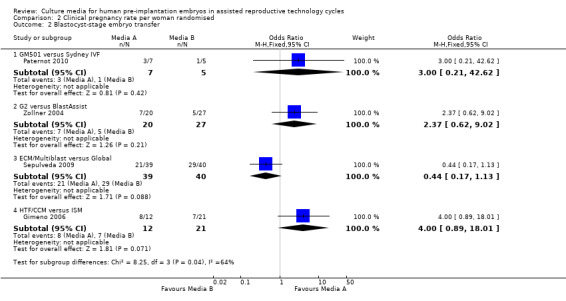
Comparison 2 Clinical pregnancy rate per woman randomised, Outcome 2 Blastocyst‐stage embryo transfer.
Comparison 3. Multiple pregnancy rate per woman randomised.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Cleavage‐stage embryo transfer | 3 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 GM501 versus Sydney IVF | 1 | 158 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 1.2 G2 versus BlastAssist | 1 | 129 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.68 [0.15, 3.17] |
| 1.3 Cook versus Vitrolife | 1 | 449 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.78 [0.40, 1.53] |
| 2 Blastocyst‐stage embryo transfer | 2 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only |
3.1. Analysis.
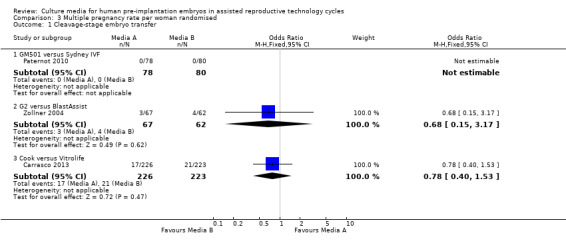
Comparison 3 Multiple pregnancy rate per woman randomised, Outcome 1 Cleavage‐stage embryo transfer.
3.2. Analysis.
Comparison 3 Multiple pregnancy rate per woman randomised, Outcome 2 Blastocyst‐stage embryo transfer.
Comparison 4. Miscarriage rate per woman randomised.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Cleavage‐stage embryo transfer | 4 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 GM501 versus ISMI | 1 | 172 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.62 [0.19, 1.97] |
| 1.2 GM501 versus Sydney IVF | 1 | 158 | Odds Ratio (M‐H, Fixed, 95% CI) | 5.26 [0.25, 111.37] |
| 1.3 G2 versus BlastAssist | 1 | 129 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.21 [0.04, 1.02] |
| 1.4 PI versus IVF | 1 | 182 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.35 [0.29, 6.20] |
| 2 Blastocyst‐stage embryo transfer | 3 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 2.1 ECM/Multiblast versus Global | 1 | 79 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.01, 8.43] |
| 2.2 GM501 versus Sydney IVF | 1 | 12 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2.3 G2 versus BlastAssist | 1 | 47 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.89 [0.24, 34.31] |
4.1. Analysis.

Comparison 4 Miscarriage rate per woman randomised, Outcome 1 Cleavage‐stage embryo transfer.
4.2. Analysis.
Comparison 4 Miscarriage rate per woman randomised, Outcome 2 Blastocyst‐stage embryo transfer.
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Arenas 2007.
| Methods | Randomisation: Yes Way of randomisation: Unknown Setting: Medical Centre Multicentre study: Unknown Time frame: Unknown |
|
| Participants | Country: Mexico, USA (Connecticut) Participants: 191 women Inclusion and exclusion criteria: Unknown Baseline characteristics: Mean oocytes retrieved per patient = 6 |
|
| Interventions | Culture media: IVC (In Vitro Care), G2 (Vitrolife) Duration of culture: Unknown Method of fertilisation: ICSI Mean number of embryos transferred: Unknown |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, embryo quality, implantation rate Other outcomes: pregnancy rate |
|
| Notes | Study is presented as an abstract All outcomes are presented as percentages, which percentage corresponds to which medium is not clear Pregnancy is reported, but it is not clear whether it is biochemical, clinical or ongoing |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not reported. States only "a total of 191 patients undergoing ICSI treatment were included in this study and randomised in two groups"; no further details |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Numbers analysed not reported. All outcomes presented only as percentages. |
| Selective reporting (reporting bias) | Unclear risk | Too few details reported to thoroughly assess this risk (published only as abstract). Does not report live birth or adverse effects |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). No funding received |
Artini 2004.
| Methods | Randomisation: Yes Way of randomisation: Unknown Setting: University Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: Italy Participants: 1014 oocytes Inclusion and exclusion criteria: Women with consensus to cryopreservation and more than 4 retrieved oocytes were included Baseline characteristics: Mean female age = 35.4, mean years of infertility = 4.2, mean oocytes retrieved per patient = 9.6 |
|
| Interventions | Culture media: HTF (Irvine Scientific), P1 (Irvine Scientific) Duration of culture: 2 to 3 days Method of fertilisation: IVF or ICSI Mean number of embryos transferred: Unknown |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, embryo quality, implantation rate, pregnancy rate | |
| Notes | It was not clear whether pregnancy rate refers to biochemical, clinical or ongoing pregnancies | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not reported |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Not reported whether there was any attrition |
| Selective reporting (reporting bias) | Unclear risk | Not reported |
| Other bias | Unclear risk | Poor reporting of methods, inconsistencies in units of analysis. Funding was not reported |
Barak 1998.
| Methods | Randomisation: Yes Way of randomisation: Unknown Setting: Medical Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: Israel Participants: 182 cycles undergoing routine IVF (however, unit of randomisation not entirely clear) Inclusion and exclusion criteria: Unknown Baseline characteristics: Mean maternal age = 34 |
|
| Interventions | Culture media: HTF (Irvine Scientific), P1 (Irvine Scientific), M3 (MediCult), Universal IVF (MediCult) Duration of culture: 2 to 3 days Method of fertilisation: IVF or ICSI Mean number of embryos transferred: 3.7 |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, pregnancy rate Other outcomes: embryo development rate |
|
| Notes | It is not clear whether the reported pregnancy was biochemical, clinical or ongoing | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States only "Aspirated oocytes from each patient were randomly allocated... into culture dishes". No further details. Wide disparity between sample sizes (37, 22, 54, 69) |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Results reported for 182 IVF treatment cycles, but number of women and unit of randomisation not entirely clear |
| Selective reporting (reporting bias) | High risk | It is stated in the aim of the study that implantation rate will be considered, but it is not reported in the article. Resulting pregnancies might be suggestive of implantation, but it is not clear whether the reported pregnancy was biochemical, clinical or ongoing |
| Other bias | Unclear risk | Methods poorly reported. Unit of analysis was cycles or oocytes, no per woman data . Funding was unclear. |
Baum 2004.
| Methods | Randomisation: Yes Way of randomisation: Randomly Setting: Medical Centre Multicentre study: Unknown Time frame: Unknown |
|
| Participants | Country: Israel Participants: Oocytes ‐ sample size not stated Inclusion and exclusion criteria: Unknown Baseline characteristics: Unknown |
|
| Interventions | Culture media: HTF (Irvine Scientific), Sydney IVF (Cook) Duration of culture: 2 to 3 days Method of fertilisation: IVF or ICSI Mean number of embryos transferred: 1.4 |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, embryo quality | |
| Notes | Study is published as abstract Exact numbers for fertilisation and good quality embryos are not reported, embryo quality reported as score |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States only "Sibling oocytes were randomly divided to two culture media"; no further details. |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Findings reported "per cumulus and corona surrounded oocyte" findings reported only as means and standard deviations with P values, no sample sizes stated. |
| Selective reporting (reporting bias) | Unclear risk | Too few details reported to assess this risk (published only as abstract) |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). Randomised oocytes, therefore no per woman data. Funding not reported |
Bird 2012.
| Methods | Randomisation: Yes Way of randomisation: Unknown Setting: Unknown Multicentre study: Unknown Time frame: Unknown |
|
| Participants | Country: UK, USA (Massachusetts) Participants: 257 zygotes (28 women) Inclusion and exclusion criteria: consecutive first cycle women aged ≤ 37 years with complete cohorts of ≥ 6 normal zygotes were included Baseline characteristics: Mean maternal age = 32.6, mean zygotes per woman = 9.2 |
|
| Interventions | Culture media: Global (IVF Online), Quinn's advantage (SAGE) Duration of culture: 5 days Method of fertilisation: Unknown Mean number of embryos transferred: Unknown |
|
| Outcomes | Outcomes considered in this review: embryo quality | |
| Notes | Study is presented as abstract No numbers provided |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States only "...normal zygotes were randomly allocated to group culture in equal‐sized microdroplets of either Group A: Global medium or Group B: Quinn's Advantage cleavage medium..." |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | No numbers provided |
| Selective reporting (reporting bias) | Low risk | Aim of the study was to assess carefully the morphologic development of sibling human zygotes |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). Randomised zygotes, therefore no per woman data. Funding not reported |
Bungum 2003.
| Methods | Randomisation: Yes Way of randomisation: Unknown Setting: Medical Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: Denmark Participants: 200 women Inclusion and exclusion criteria: All type of infertility Baseline characteristics: Unknown |
|
| Interventions | Culture media: G2 (Vitrolife), G3 (Vitrolife) Duration of culture: Unknown Method of fertilisation: Unknown Number of embryos transferred: 1 to 2 |
|
| Outcomes | Outcomes considered in this review: none Other outcomes: biochemical pregnancy rate |
|
| Notes | Study is published as abstract | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States only "200 patients with all types of infertility diagnoses were randomised". No further details. |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Preliminary results for 140 women reported in abstract. Follow‐up rate unclear |
| Selective reporting (reporting bias) | Unclear risk | Too few details reported to thoroughly assess this risk (published only as abstract). Biochemical pregnancy data for first 140 women reported; data on adverse effects and other outcomes collected but not reported in published abstract. Live birth not an outcome. |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). Funding not reported. |
Campo 2010.
| Methods | Randomisation: Yes Way of randomisation: Envelopes, stratification by age (< 36 and > 36) Setting: Medical Centre Multicentre study: No Time frame: four‐month period |
|
| Participants | Country: Belgium Participants: 172 women Inclusion and exclusion criteria: Blastocyst culture, pre‐implantation genetic diagnosis patients, testicular sperm extraction and egg donation procedures were excluded Base line characteristics: mean female age = 34.6, mean oocytes retrieved per cycle = 7.3 |
|
| Interventions | Culture media: GM501 (Gynemed), ISM (MediCult) Duration of culture: 2 to 3 days Method of fertilisation: IVF or ICSI Number of embryos transferred: 1.6 |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, implantation rate, cryopreservation rate, clinical pregnancy rate, miscarriages, ongoing pregnancy rate, live birth rate Other outcomes: embryo fragmentation |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Method of generation not stated |
| Allocation concealment (selection bias) | Unclear risk | States "Randomization was blinded by providing envelopes filled with an equal amount of adhesive labels printed with either 'GM501' or 'ISM1.'" Does not state that envelopes were opaque and sequentially numbered. |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Bliding not described |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 97% of randomised women included in analysis (167/172). The other 5 women had no transfer performed |
| Selective reporting (reporting bias) | Low risk | All outcomes stated in the methods section were reported. Live birth reported. |
| Other bias | Unclear risk | Funding was not reported |
Cano 2001.
| Methods | Randomisation: Yes Way of randomisation: Unknown Setting: Medical Centre Multicentre study: No Time frame: 2000 |
|
| Participants | Country: Spain Participants: 496 cycles, donor oocytes included Inclusion and exclusion criteria: Unknown Baseline characteristics: mean age and causes of infertility similar in both groups (numbers not reported) |
|
| Interventions | Culture media: unknown name of medium (MediCult media), IVF (Scandinavian IVF) Duration of culture: Unknown Method of fertilisation: Unknown Mean number of embryos transferred: 3.0 |
|
| Outcomes | Outcomes considered in this review: embryo quality, pregnancy rate, implantation rate, miscarriage rate | |
| Notes | Name of compared medium is not known It is not stated whether pregnancy rate refers to biochemical, clinical or ongoing pregnancies Exact numbers for implantation rate and embryo quality are missing |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States only "The treatments were randomly distributed into two groups"; no further details |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | 496 cycles randomised, 406 analysed (81%) |
| Selective reporting (reporting bias) | Unclear risk | Too few details reported to assess this risk (published only as abstract) |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). Randomised cycles, therefore no 'per woman' data. Funding was unclear. |
Carrasco 2013.
| Methods | Randomisation: Yes Way of randomisation: Computer‐generated randomisation table Setting: University Medical Centre Multicentre study: No Time frame: August 2008 to December 2008 |
|
| Participants | Country: Spain Participants: 449 couples Inclusion and exclusion criteria: All couples performing a cycle of IVF or ICSI with autologous oocytes were included. Oocyte donation cycles and pre‐implantation genetic diagnosis cycles were excluded. Baseline characteristics: mean maternal age = 36.4, mean maternal BMI = 22.5 |
|
| Interventions | Culture media: K‐SIMC (Cook), IVF/G3 (Vitrolife) Duration of culture: 2 days Method of fertilisation: IVF or ICSI Mean number of embryos transferred: 1.9 |
|
| Outcomes | Outcomes considered in this review: clinical pregnancy rate, miscarriage rate, live birth rate | |
| Notes | For IVF cycles, IVF medium from Vitrolife was used while for ICSI cycles G3 medium from Vitrolife was used. However the results for the Vitrolife group are added together Multiple pregnancy was not reported although clearly these data available. Cook: CPR=82; singleton pregnancies=65; multiple pregnancies must therefore equal 17 Vitrolife: CPR=80; singleton pregnancies=59; multiple pregnancies must therefore equal 21 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated randomisation table |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Apparently there was no attrition |
| Selective reporting (reporting bias) | High risk | Live birth data resulting from multiple pregnancies were not reported |
| Other bias | Low risk | No other potential bias identified. Academic funding |
Chatziioannou 2010.
| Methods | Randomisation: Yes Way of randomisation: Unknown Setting: University Medical Centre Multicentre study: No Time frame: January 2009 to November 2009 |
|
| Participants | Country: Greece Participants: 692 women Inclusion and exclusion criteria: All women treated by a fresh IVF or ICSI cycle with at least one cumulus oocyte complex were included Baseline characteristics: mean maternal age similar in both groups (numbers not reported) |
|
| Interventions | Culture media: Quinn's advantage (Sage), G5 (Vitrolife) Duration of culture: 2 or 3 days Method of fertilisation: IVF or ICSI Mean number of embryos transferred: 2.5 |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, embryo quality, clinical pregnancy rate Other outcomes: biochemical pregnancy |
|
| Notes | Exact numbers for fertilisation rate and embryo quality are missing Numbers for clinical pregnancy were deducted from the reported percentages |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | states "all patients treated by fresh IVF or ICSI (N = 692) with at least one cumulus oocyte complex (COC) retrieved were randomly allocated to 2 treatment groups". No further information |
| Allocation concealment (selection bias) | Unclear risk | As above |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Numbers for clinical pregnancy that was the primary outcome were deducted from the reported percentages |
| Selective reporting (reporting bias) | Unclear risk | Too few details reported to assess this risk (published only as abstract) |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract), funding not reported |
Chen 2009.
| Methods | Randomisation: Yes Way of randomisation: Randomly split in half Setting: University Medical Centre Multicentre study: Unknown Time frame: January 2008 to September 2008 |
|
| Participants | Country: China Participants: 201 cycles with 1958 oocytes Inclusion and exclusion criteria: Following inclusion criteria were used: Patient age ≥ 35 years, day 3 FSH level < 10 IU/L, PRL < 25 ng/ml, adequate semen parameters for IVF or ICSI, controlled ovarian stimulation was achieved using a long protocol, 2PN zygotes ≥ 4. Each patient was her own control. Baseline characteristics: Unknown |
|
| Interventions | Culture media: ECM (Irvine Scientific), P1 (Irvine Scientific) Duration of culture: 3 days Method of fertilisation: IVF Mean number of embryos transferred: Unknown |
|
| Outcomes | Outcomes considered in this review: top‐quality embryos Other outcomes: embryo‐cleavage rate, rate of normal‐cleaving embryos |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | states "After 16 to18 hour post‐insemination, when the 2PN zygotes were ≥ 4 per patient, half of them were transferred into culture dishes containing 0.5 ml of ECM media (group A) and half into culture dishes containing 0.5 ml of P1 media (group B) randomly". No further information |
| Allocation concealment (selection bias) | Unclear risk | As above |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All exact numbers for the reported outcomes are mentioned |
| Selective reporting (reporting bias) | Unclear risk | Too few details reported to assess this risk (published only as abstract) |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract), funding not reported |
Fechtali 2004.
| Methods | Randomisation: Yes Way of randomisation: Randomly Setting: University Medical Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: France Participants: 1790 oocytes Inclusion and exclusion criteria: Unknown Baseline characteristics: Unknown |
|
| Interventions | Culture media: FertiCult (FertiPro), ISM (MediCult) Duration of culture: Unknown Method of fertilisation: IVF or ICSI Mean number of embryos transferred: Unknown |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, embryo quality Other outcomes: mean number of blastomeres |
|
| Notes | Study published as abstract Exact numbers for fertilisation and embryo quality are missing |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States only "Sibling oocytes of all couples undergoing the IVF (N = 112) or ICSI (N = 131) program were randomly assigned to either Ferticult or ISM1TM culture media". No further details. |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Unclear whether all randomised oocytes included in analysis, as results given as proportions. |
| Selective reporting (reporting bias) | Unclear risk | Too few details reported to assess this risk (published only as abstract) |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). Randomised oocytes, therefore no "per woman" data. Funding was unclear. |
Findikli 2004.
| Methods | Randomisation: Yes Way of randomisation: Randomly Setting: Medical Centre Multicentre study: No Time frame: 2001 |
|
| Participants | Country: Turkey Participants: 1434 oocytes (113 women) Inclusion and exclusion criteria: Women undergoing ICSI for severe male factor infertility were included Base line characteristics: mean maternal age = 30.0, mean paternal age = 34.7, mean duration of infertility = 8.3 years, mean oocytes retrieved = 16.7 |
|
| Interventions | Culture media: G2 (Vitrolife), ISM (MediCult) Duration of culture: 6 days Method of fertilisation: ICSI Mean number of embryos transferred: 2.7 |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, embryo quality | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not reported |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Number of oocytes used to calculate fertilisation rate are reported |
| Selective reporting (reporting bias) | Unclear risk | Not reported |
| Other bias | Unclear risk | Funding was not reported |
Gimeno 2006.
| Methods | Randomisation: Yes Way of randomisation: Randomly Setting: Medical Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: Spain Participants: 118 women Inclusion and exclusion criteria: Women attending a program of oocyte donation for various reasons Base line characteristics: mean maternal age = 39.3 |
|
| Interventions | Culture media: HTF/CCM (Irvine Scientific/Vitrolife), ISM (MediCult) Duration of culture: 3 to 6 days Method of fertilisation: IVF or ICSI Mean number of embryos transferred: 2 |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, implantation rate, clinical pregnancy rate, live birth rate Other outcomes: number of cells and % fragmentation on days 2 and 3, blastocyst development |
|
| Notes | article in Spanish, abstract also in English | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | "Couples were assigned in random fashion to one of the two means of culture" |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Number of fertilized oocytes is different than number of embryos reported in the study. There is no explanation what happened with the remaining zygotes. Implantation rate reported as percentage and there is not enough information to calculate the exact number of embryos transferred and implanted. Analysis of pregnancy outcomes is per women who had transfer not per women randomised. Analysis of live birth data was not performed per day of transfer. |
| Selective reporting (reporting bias) | Unclear risk | Live birth data resulting from multiple pregnancies were not analysed. |
| Other bias | High risk | Discrepancy in data ‐ ongoing pregnancy rates lower than live birth rates. Academic funding |
Hazlett 2003.
| Methods | Randomisation: Yes Way of randomisation: Randomly Setting: Medical Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: USA (Illinois) Participants: 100 women Inclusion and exclusion criteria: Unknown Baseline characteristics: Mean maternal age = 32.75, mean oocytes retrieved = 12.5 |
|
| Interventions | Culture media: P1 (Irvine Scientific), IVC (In Vitro Care) Duration of culture: 3 days Method of fertilisation: IVF Mean number of embryos transferred: 2.5 |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, implantation rate, clinical pregnancy rate Other outcomes: embryo development |
|
| Notes | Study published as abstract Exact numbers for fertilisation, implantation and clinical pregnancies are missing Pregnancy rate reported per transfer rather than per woman. Live birth not reported. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States "One hundred patients were randomly assigned either P‐1 or IVC‐1 for fertilisation and embryo culture". No further details |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Data report results from 49 and 38 women; if 50 in each group, then significant unexplained loss from IVC group |
| Selective reporting (reporting bias) | Unclear risk | Too few details reported to assess this risk (published only as abstract) |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). No funding received |
Jamieson 1997.
| Methods | Randomisation: Yes Way of randomisation: Unknown Setting: Medical Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: UK Participants: 555 women Inclusion and exclusion criteria: Unknown Baseline characteristics: Unknown |
|
| Interventions | Culture media: IVF (MediCult), IVF‐50 (Scandinavian IVF) Duration of culture: 1 to 3 days Method of fertilisation: Unknown Mean number of embryos transferred: Unknown |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, embryo quality, pregnancy rate | |
| Notes | Study published as abstract Exact numbers for fertilisation, good quality embryos and pregnancies are missing. It is not sure whether pregnancies are biochemical, clinical or ongoing |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not reported |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Exact numbers of the outcomes not known |
| Selective reporting (reporting bias) | Unclear risk | The outcomes were not pre‐specified |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). |
Khan 2004.
| Methods | Randomisation: Yes Way of randomisation: Pre‐selected randomised numbers Setting: Medical Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: USA (Michigan) Participants: 57 women Inclusion and exclusion criteria: Unknown Baseline characteristics: Unknown |
|
| Interventions | Culture media: P1 (Irvine Scientific), ECM (Irvine Scientific) Duration of culture: 3 days Method of fertilisation: ICSI Mean number of embryos transferred: Unknown |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, cryopreservation rate, pregnancy rate Other outcomes: cleavage rate, blastocyst formation |
|
| Notes | Study published as abstract Exact numbers for fertilisation and cryopreservation rate are missing Pregnancy is not clear whether it is biochemical, clinical or ongoing |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States only "fifty‐seven consecutive patients were placed in either P1C group (29) or in the ECM group (28) according to preselected randomised numbers"; no further details. |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Number analysed not reported. |
| Selective reporting (reporting bias) | Unclear risk | Too few details reported to assess this risk (published only as abstract) |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). |
Kyono 2000.
| Methods | Randomisation: Yes Way of randomisation: Randomly divided Setting: Medical Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: Japan Participants: 181 zygotes (36 cycles) Inclusion and exclusion criteria: Unknown Baseline characteristics: Unknown |
|
| Interventions | Culture media: P1/Blastocyst (Irvine Scientific), G2 (Vitrolife), K‐SICM/K‐SIBM (Cook) Duration of culture: 5 days Method of fertilisation: ICSI Mean number of embryos transferred: Unknown |
|
| Outcomes | Outcomes considered in this review: none Other outcomes: blastocyst formation |
|
| Notes | Study published as abstract | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States only "they were randomly divided into three protocols of media"; no further details. |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All exact numbers were reported |
| Selective reporting (reporting bias) | Unclear risk | Too few details reported to assess this risk (published only as abstract) |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). |
Lambert 2005.
| Methods | Randomisation: Yes Way of randomisation: Randomly Setting: Medical Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: USA (Hawaii) Participants: Oocytes Inclusion and exclusion criteria: Women with at least one failed IVF attempt were included Baseline characteristics: Mean maternal age = 34, mean number of oocytes retrieved = 18 |
|
| Interventions | Culture media: unknown name of medium (MediCult), unknown name of medium (Vitrolife) Duration of culture: 5 days Method of fertilisation: IVF or CSI Mean number of embryos transferred: Unknown |
|
| Outcomes | Outcomes considered in this review: fertilisation rate Other outcomes: embryo development |
|
| Notes | Study published as abstract Exact numbers for fertilisation are missing The exact media names are missing |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States "sister oocytes were randomly selected according to maturational status after OPU and put into the two different types of culture system being evaluated". No further details. |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Low risk | Embryologists were blinded to the culture systems they were reading |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Numbers of oocytes randomised and analysed not reported; all results given as percentages |
| Selective reporting (reporting bias) | Unclear risk | Too few details reported to assess this risk (published only as abstract) |
| Other bias | Low risk | Insufficient details reported (published only as abstract). Randomised oocytes, therefore no 'per woman' data. The study was funded by a research grant, not from a company. |
Mauri 2001.
| Methods | Randomisation: Yes Way of randomisation: Drawing lots Setting: University Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: Brazil Participants: 182 women Inclusion and exclusion criteria: Unknown Baseline characteristics: mean maternal age = 34, mean oocytes retrieved = 11 |
|
| Interventions | Culture media: P1 (Irvine Scientific), IVF (Scandinavian IVF) Duration of culture: 2 days Method of fertilisation: ICSI Mean number of embryos transferred: 2.8 |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, implantation rate, miscarriage rate, pregnancy rate Other outcomes: embryo development |
|
| Notes | It is not clear whether reported pregnancies are biochemical, clinical or ongoing | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | States "Patient participation in each group was random, by drawing lots, using a randomisation table previously elaborated for the study" |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Pregnancy outcomes reported for all women randomised |
| Selective reporting (reporting bias) | Low risk | All expected outcomes reported |
| Other bias | Unclear risk | Funding was not reported |
Mayer 2003.
| Methods | Randomisation: Yes Way of randomisation: Unknown Setting: Academic Medical Centre Multicentre study: No Time frame: January 2000 to December 2002 |
|
| Participants | Country: USA (Virginia) Participants: 3211 oocytes Inclusion and exclusion criteria: All ICSI patients and patients with three or fewer oocytes were excluded Baseline characteristics: Unknown |
|
| Interventions | Culture media: G1.2./G1.2 (Vitrolife), P1/Blastocyst (Irvine Scientific), Quinn's Advantage/Blastocyst (Sage) Duration of culture: 3 or 5 days Method of fertilisation: IVF Mean number of embryos transferred: Unknown |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, clinical pregnancy rate Other outcomes: embryo development |
|
| Notes | Clinical pregnancy rate was excluded from the analysis since the unit of analysis (women) was different than the randomisation unit (oocytes) | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States only "Oocytes were randomly allocated during retrieval into two groups". No further details. |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Reports fertilisation rate for all randomised oocytes |
| Selective reporting (reporting bias) | Unclear risk | Too few details reported to assess this risk (published only as abstract) |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). Study conducted in two separate phases. Randomised oocytes, therefore no "per woman" data. Funding was unclear. |
Mendoza 2003.
| Methods | Randomisation: Yes Way of randomisation: Table of random numbers Setting: Medical Centre Multicentre study: No Time frame: February 2003 to April 2003 |
|
| Participants | Country: Spain Participants: 118 women Inclusion and exclusion criteria: Unknown Baseline characteristics: mean maternal age = 33.8, mean years of infertility = 5.0, mean oocytes retrieved = 11.7 |
|
| Interventions | Culture media: G3 (Vitrolife), (Cook), (MediCult) Duration of culture: 2 to 4 days Method of fertilisation: IVF or ICSI Mean number of embryos transferred: 3 |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, embryo quality, implantation rate Other outcomes: biochemical pregnancy rate |
|
| Notes | All outcomes were reported in mean numbers rather than exact numbers. The exact names of the compared media were not provided, although the manufacturers are listed. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | It states that a table of random numbers was used, no further details |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Paper states that 37 women randomised to Vitrolife, 47 to Medicut and 34 to Cook, and does not mention dropouts. |
| Selective reporting (reporting bias) | Low risk | All outcomes mentioned in the aim are provided in the results |
| Other bias | Low risk | No external funding was received |
Paternot 2010.
| Methods | Randomisation: Yes Way of randomisation: Sealed envelopes Setting: University Centre Multicentre study: No Time frame: 2008 to 2009 |
|
| Participants | Country: Belgium Participants: 170 women Inclusion and exclusion criteria: Women younger than 36 years, during the first or second cycle were included.Cycles with pre‐implantation genetic diagnosis, sperm or oocyte donation, failed fertilisation, embryo transfer at day 2, cryopreservation of all fertilized oocytes were excluded Baseline characteristics: Mean maternal age = 30, mean oocytes retrieved = 11 |
|
| Interventions | Culture media: Sydney IVF (Cook), GM501 (Gynemed) Duration of culture: 3 or 5 days Method of fertilisation: IVF or ICSI Number of embryos transferred: 1 to 2 |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, embryo quality, implantation rate Other outcomes: utilization rate Obtained through contact with the primary investigator: live birth rate, ongoing pregnancy rate, clinical pregnancy rate, miscarriage rate, multiple pregnancy rate |
|
| Notes | ClinicalTrials.gov identifier: NCT01094314 | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States "a total of 170 patients were randomised, after informed consent, by a sealed envelope system". No further details. |
| Allocation concealment (selection bias) | Unclear risk | States "a total of 170 patients were randomised, after informed consent, by a sealed envelope system." No further details. |
| Blinding (performance bias and detection bias) All outcomes | Low risk | Doctors, nurses and participants were blinded |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 86% of randomised women (147/170) included in analysis of embryo morphology in published article. Reasons for dropouts stated and comparable across groups. Outcomes analysed by review authors based on total randomised women |
| Selective reporting (reporting bias) | Low risk | Data on all expected outcomes obtained from primary investigator (although not in published article) |
| Other bias | Low risk | No funding was received |
Rubino 2004.
| Methods | Randomisation: Yes Way of randomisation: Unknown Setting: Medical Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: Italy Participants: 175 women Inclusion and exclusion criteria: Unknown Baseline characteristics: Mean maternal age = 36.4, mean number of oocytes retrieved = 8.5 |
|
| Interventions | Culture media: IVF (Scandinavian IVF), Quinn's Advantage (Sage) Duration of culture: 3 Method of fertilisation: IVF or ICSI Mean Number of embryos transferred: 2.6 |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, embryo quality, implantation rate, clinical pregnancy rate | |
| Notes | Exact numbers for fertilisation, embryo quality and implantation are missing | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States "a prospective, controlled, randomised study has been started in patients undergoing IVF or ICSI treatment". No further details |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Number analysed not reported; results given as proportions only |
| Selective reporting (reporting bias) | Unclear risk | Too few details reported to assess this risk (published only as abstract) |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). Funding source was not reported |
Sepulveda 2009.
| Methods | Randomisation: Yes Way of randomisation: Randomly Setting: Private centre Multicentre study: No Time frame: March 2006 to October 2006 |
|
| Participants | Country: Peru Participants: 80 women Inclusion and exclusion criteria: Women with oocyte donation were included Baseline characteristics: Mean donor age = 25.6, mean recipient age = 40.5 |
|
| Interventions | Culture media: ECM/Multiblast (Irvine Scientific), Global (IVF Online) Duration of culture: 5 days Method of fertilisation: IVF or ICSI Mean Number of embryos transferred: 2 |
|
| Outcomes | Outcomes considered in this review: embryo quality, implantation rate, clinical pregnancy rate, ongoing pregnancy rate | |
| Notes | Embryo quality could not be analysed as exact numbers were missing Miscarriage rate was calculated by the review authors, based on differences between clinical and ongoing pregnancies |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States "A total of 80 patients, 40 per group, were randomly assigned to have their embryos cultured in either Global medium (IVFonline, Guelph, ON, Canada) or in Irvine Early Cleavage Medium (ECM)/Multiblast (Irvine Scientific)". No details reported. |
| Allocation concealment (selection bias) | Unclear risk | Was not reported clearly |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Was not reported clearly |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Pregnancy results reported for 79/80 randomised women, reason given for missing data for one woman. |
| Selective reporting (reporting bias) | Unclear risk | Adverse effects not reported |
| Other bias | Unclear risk | Embryo culture media were supplied free of charge from IVFonline |
Shih 2014.
| Methods | Randomisation: Yes Way of randomisation: Sibling oocytes or embryos from individual patients were randomly assigned to two culture groups Setting: University Medical Centre Multicentre study: No Time frame: February to May 2010 |
|
| Participants | Country: Taiwan Participants: 159 women undergoing IVF or ICSI Inclusion and exclusion criteria: Only cycles from female participants younger than 38 years of age, which contained at least six mature oocytes available for randomly splitting cultures were included. Cycles from oocyte and sperm donation programs were excluded. Baseline characteristics: Mean age 31 years, duration of infertility 3.2 to 3.3 years |
|
| Interventions | Culture media: Quinn's Advantage Cleavage and Blastocyst media (QAC/QAB) versus GIII series cleavage and blastocyst media (G1.3/G2.3). Duration of culture: 5 days Method of fertilisation: IVF or ICSI Mean number of embryos transferred: 2.6 to 2.8 |
|
| Outcomes | No outcomes of interest to this review | |
| Notes | Aim of study was to determine the correlation between the levels of reactive oxygen species (ROS) in prepared culture media and the early development of human embryos. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Method of sequence generation not described |
| Allocation concealment (selection bias) | Unclear risk | Method of allocation concealment not described |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Blinding not mentioned |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Data appear complete |
| Selective reporting (reporting bias) | Unclear risk | Does not report any clinical outcomes of interest to this review |
| Other bias | Low risk | No other potential source of bias noted |
Sifer 2009.
| Methods | Randomisation: Yes Way of randomisation: Sequences of embryo culture media Setting: University Medical Centre Multicentre study: No Time frame: April 2005 to December 2006 |
|
| Participants | Country: France Participants: 5133 oocytes (512 women) Inclusion and exclusion criteria: Women with less than four oocytes, fertilisation failure in both groups, no metaphase II oocytes in one of the groups were excluded Baseline characteristics: Mean maternal age = 33.2, mean years of infertility = 3.8, IVF or ICSI attempt rank = 2.1 |
|
| Interventions | Culture media: G3 (Vitrolife), ISM (MediCult) Duration of culture: 3 days Method of fertilisation: IVF or ICSI Mean Number of embryos transferred: 1.6 |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, embryo quality, implantation rate, multiple pregnancy rate, clinical pregnancy rate | |
| Notes | Exact numbers for fertilisation, embryo quality and implantation rate were missing Pregnancy outcomes were not included since the unit of analysis (women) was different from the randomisation unit (oocytes) |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States "Sibling oocytes of all couples undergoing an IVF (N = 286) or ICSI (N = 390) attempt were randomly assigned to either GIII series (Vitrolife) or ISM (Medicult) media". No further details. |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 96% of randomised oocytes analysed (4910/5133) |
| Selective reporting (reporting bias) | Unclear risk | Adverse effects not reported |
| Other bias | Unclear risk | Randomised oocytes, therefore no usable 'per woman' data. No funding was received |
Stevens 2000.
| Methods | Randomisation: Yes Way of randomisation: Computer‐generated randomisation table Setting: Medical Center Multicentre study: No Time frame: January 1999 to December 1999 |
|
| Participants | Country: USA (Colorado) Participants: 72 women Inclusion and exclusion criteria: Unknown Baseline characteristics: Mean maternal age = 37.5 |
|
| Interventions | Culture media: Ham's F10 (Gibco), G1.2/G2.2 (Vitrolife) Duration of culture: 3 days Method of fertilisation: IVF Mean Number of embryos transferred: Unknown |
|
| Outcomes | Outcomes considered in this review: implantation rate, ongoing pregnancy rate Other outcomes: biochemical pregnancy rate |
|
| Notes | Exact numbers for implantation rate and ongoing pregnancies were missing Data on ongoing pregnancies were calculated |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer generated randomisation table |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Number of ongoing pregnancies reported as percentage, but since number of women were reported, we could calculate the exact numbers. |
| Selective reporting (reporting bias) | Unclear risk | Not reported in methods section |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). Funding not reported |
Tedesco 1990.
| Methods | Randomisation: Yes Way of randomisation: Randomly placed into two media Setting: University Medical Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: USA (Florida) Participants: 142 oocytes Inclusion and exclusion criteria: Unknown Baseline characteristics: Unknown |
|
| Interventions | Culture media: Ham's F10 (Gibco), HTF (Irvine Scientific), Menezo B (Api‐System) Duration of culture: Unknown Method of fertilisation: IVF Mean Number of embryos transferred: Unknown |
|
| Outcomes | Outcomes considered in this review: fertilisation rate Other outcomes: cleavage rate |
|
| Notes | Number of oocytes fertilized were reported. Cleavage rate outcome was not separated from fertilisation outcome | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Article stated that oocytes were randomly placed in different culture media. No other information available |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Information insufficient to assess this |
| Selective reporting (reporting bias) | Unclear risk | Unable to assess due to poor reporting of methods |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). Funding not reported |
Von During 2004.
| Methods | Randomisation: Yes Way of randomisation: Unknown Setting: Univeristy Medical Centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: Norway Participants: 340 cycles (unknown number of women) Inclusion and exclusion criteria: All patients entering the IVF or ICSI program during the study period were included Baseline characteristics: Unknown |
|
| Interventions | Culture media: Universal IVF (MediCult), Sydney IVF (Cook) Duration of culture: 2 days Method of fertilisation: IVF or ICSI Mean Number of embryos transferred: 1.8 |
|
| Outcomes | Outcomes considered in this review: embryo quality, cryopreservation rate, implantation rate, clinical pregnancy rate | |
| Notes | Exact numbers for implantation rate and clinical pregnancies were missing | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not reported |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Number of women included not reported, exact number or outcomes not reported |
| Selective reporting (reporting bias) | Unclear risk | Primary outcome reported, secondary outcomes not pre‐specified |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). Funding not reported |
Yamamoto 2006.
| Methods | Randomisation: Yes Way of randomisation: Oocytes were split Setting: Private infertility centre Multicentre study: No Time frame: Unknown |
|
| Participants | Country: Japan Participants: Oocytes (34 women) Inclusion and exclusion criteria: Unknown Baseline characteristics: Unknown |
|
| Interventions | Culture media: MultiBlast (Irvine Scientific), BlastAssist (MediCult) Duration of culture: 3 or 5 days Method of fertilisation: Unknown Mean number of embryos transferred: Unknown |
|
| Outcomes | Outcomes considered in this review: embryo quality, clinical pregnancy rate | |
| Notes | We used embryo quality assessed per day 3 for our analysis, as it was not clear whether the blastocysts assessed on day 5 were excluded for the day 3 assessment. Outcome of clinical pregnancies was not used for analysis as the study randomised oocytes |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | States "Prospective randomised study at private setting fertility clinic ...All oocytes in each cycle were split into two groups and cultured in each medium." No further details |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Restuls presented as percentages with no denominator |
| Selective reporting (reporting bias) | Unclear risk | Outcomes not clearly stated in methods section |
| Other bias | Unclear risk | Insufficient details reported (published only as abstract). Funding was not received |
Zollner 2004.
| Methods | Randomisation: Yes Way of randomisation: Cube randomisation Setting: University Medical Centre Multicentre study: No Time frame: November 2000 to August 2001 |
|
| Participants | Country: Germany Participants: 176 women Inclusion and exclusion criteria: Couples unselected for age, sperm parameters and infertility criteria undergoing IVF or ICSI were included. Unsuccessful oocyte retrieval or fertilisation failure patients were excluded. Baseline characteristics: mean maternal age = 33, mean paternal age = 35, mean duration of infertility = 4 years, mean number of oocytes retrieved = 11 |
|
| Interventions | Culture media: G2 (Vitrolife), BlastAssist (MediCult) Duration of culture: 3,4 or 5 days Method of fertilisation: IVF or ICSI Number of embryos transferred: 1 to 3 |
|
| Outcomes | Outcomes considered in this review: fertilisation rate, embryo quality, implantation rate, clinical pregnancy rate Obtained through contact with the primary investigator: miscarriage rate, multiple pregnancy rate, live birth rate |
|
| Notes | Exact numbers for embryo quality were missing | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Cube randomisation (reported through contact with author) |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | High risk | No blinding was performed (confirmed by contact with author). |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Data for all women randomised supplied by author |
| Selective reporting (reporting bias) | Low risk | All outcomes stated in aims and methods are reported. Data on live birth supplied by author |
| Other bias | Unclear risk | This study was supported by Fa. MediCult |
IVF = in vitro fertilisation
ICSI = intracytoplasmic sperm injection
FSH = follicle‐stimulating hormone
PRL = prolactin
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Aoki 2005 | Retrospective study |
| Assaf 1998 | Quasi‐randomised trial; alternating patients |
| Atchajaroensatit 2005 | Quasi‐randomised trial; patients randomly divided according to availability of culture media |
| Balaban 2004 | Quasi‐randomised trial; patients randomised by alternating weekdays |
| Barrett 1997 | Study comparing different culture media and protein supplementations at the same time |
| Behr 2004 | Not relevant |
| Ben‐Yosef 2001 | Study on medium supplementation |
| Ben‐Yosef 2004 | Quasi‐randomised trial; randomisation was obtained by a computer‐generated randomization table prepared in advance. The randomisation assigned one or the other media systems for use during a given day for all patients undergoing oocyte retrieval on that day |
| Biggers 2002 | Quasi‐randomised trial; the embryos were alternately assigned |
| Bisioli 2003 | Quasi‐randomised trial; the randomisation was done by alternating IVF cases to each protocol |
| Blefco 2001 | Retrospective study |
| Bungum 2002 | Study on media supplementation |
| Choi 2004 | Study with cryopreserved embryos, not an RCT |
| Ciray 2012 | Quasi‐randomised trial; Author stated when contacted that "all injected oocytes were pooled into one microdrop of media and distributed into two groups, hence, we did not have an accurate method of sequence generation" |
| Coates 1999 | Media supplementation |
| Cooke 2002 | Second set of media is a modification of the first |
| Cummins 1986 | Quasi‐randomised trial; incomplete data |
| de Araujo 2009 | In vitro maturation media |
| Di Falco Cossiello 2011 | Quasi‐randomised trial; first zygote was randomised based on a computer‐generated list and the following zygotes were alternated |
| Dudkiewicz 2003 | Retrospective study |
| Dumoulin 2010 | Quasi‐randomised trial; oocytes and embryos randomised by alternating days |
| Eaton 2012 | Retrospective study |
| Fatemeh 2012 | Quasi‐randomised trial; women assigned according to the daily medium schedule for oocyte retrieval |
| Feichtinger 1986 | Comparison with using patient serum as medium |
| Fukunaga 2003 | Randomization was performed among experiments. Randomization between groups within an experiment not reported |
| Geber 2012 | Quasi‐randomised trial; women assigned sequentially |
| Gianaroli 1989 | Comparison with human amniotic fluid as medium |
| Gilson 2013 | Retrospective study |
| Hambiliki 2011 | Quasi‐randomised trial; oocytes assigned alternatively. |
| Hassani 2013 | Quasi‐randomized trial; patients were randomised according to the daily media schedule for oocyte retrieval |
| Hirai 2003 | Comparison with human follicular fluids (article in Japanese) |
| Hoogendijk 2007 | Quasi‐randomised trial |
| Ignoul Vanvuchelen 1996 | Comparison with in‐house media |
| Isiklar 1998 | Quasi‐randomized trial; patients were randomised according to their patient file |
| Janssens 1996 | Quasi‐randomised trial; alternating patients |
| Jindal 2005 | Retrospective study |
| Jones 1998 | no reference to randomisation |
| Kasai 2001 | no reference to randomisation for media |
| Kasterstein 1999 | Quasi‐randomised trial; culture media were alternately changed on a bi‐weekly basis |
| Khoury 2012 | Quasi‐randomised trial; patients were randomised according to their patient number |
| Kim 2002 | Not randomised |
| Kleijkers 2014 | Quasi‐randomised study |
| Komarovsky 2007 | Comparative study, not an RCT |
| Kyono 1999 | Quasi‐randomised trial; patients were assigned based on the medium batch in use at the time of oocyte retrieval |
| Kyono 2002 | Comparison of day of embryo transfer |
| Lane 2007 | Review |
| Liebermann 2012 | Retrospective study |
| Lin 2014 | Participants were mice |
| Lopes 2008 | Quasi‐randomised trial; patients were alternately assigned |
| Mahadevan 1996 | Comparison with in‐house media |
| Mantikou 2013 | Review |
| Mayer 1998 | No reference to randomisation |
| Mayer 1999 | Quasi‐randomised trial; patients were alternately assigned |
| Mayer 1999b | Quasi‐randomised trial |
| Mellon 2005 | Quasi‐randomised trial; consecutive patients were alternately assigned |
| Merry 2002 | Quasi‐randomised trial; there was no sequence generation or allocation concealment (according to contact with author) |
| Michaeli 2007 | Retrospective study |
| Moodie 2004 | Quasi‐randomised trial; cases were sequentially alternated |
| Mortimer 2003 | Quasi‐randomised trial |
| Nelissen 2012 | Quasi‐randomised trial; patients were randomised by alternation |
| Parinaud 1987 | Quasi‐randomised trial |
| Parinaud 1998 | Quasi‐randomised trial; patients were alternatively assigned |
| Parinaud 1999 | Quasi‐randomised trial; patients were alternatively assigned |
| Perri 2001 | Quasi‐randomised trial; patients were alternatively assigned according to the availability of media |
| Pool 2012 | Review |
| Prados 2002 | Retrospective study |
| Quinn 1985 | Quasi‐randomised study; patients were randomised to media alternating every two weeks |
| Quinn 1985b | Quasi‐randomised study, randomised according to weeks |
| Quinn 1995 | Prospective study, but not randomised |
| Quinn 1995b | Not randomised |
| Reed 2009 | Quasi‐randomised trial |
| Rinehart 1998 | Quasi‐randomised trial; random alternating allocation |
| Romano 2004 | Quasi‐randomised trial; the randomisation was according to the odd or even numbers of the birthday of the patients |
| Ruiz 1999 | Quasi‐randomised trial; "What we did for allocation concealment, was to culture half of the oocytes of each patient with each medium, the standard medium contained human albumin and was medium A, and the one without albumin was medium B, starting each patient with either medium A or B consecutively" (communication with author) |
| Sieren 2003 | Quasi‐randomised trial |
| Sieren 2006 | Quasi‐randomised trial; after contact with author: no adequate sequence generation, no allocation concealment |
| Staessen 1994 | Comparison with in‐house media |
| Staessen 1998 | Quasi‐randomised trial; alternate days |
| Steele 2006 | Quasi‐randomized trial; cycles were alternated in week blocks |
| Steinkampf 2001 | Retrospective study |
| Stoddart 2001 | Quasi‐randomised trial; media were used on weekly basis |
| Stoddart 2002 | Study on media supplementation |
| Summers‐Chase 2004 | Quasi‐randomised trial; media were used on weekly basis |
| Sun 2001 | Not an RCT |
| Szell 1998 | Quasi‐randomised trial; patients were alternately assigned to one of the two groups |
| Tekpetey 1998 | Quasi‐randomized trial; patient allocation was based on the medium batch in used at the time of oocyte retrieval |
| Thorsell 2003 | Retrospective study |
| Torres 2012 | No reference to randomisation |
| Utsunomiya 2002 | Quasi‐randomised trial; patients prospectively assigned according to a randomisation sequence |
| Van der Auwera 2001 | Patients randomised to two groups, embryos cultured in media from Vitrolife (IVF or G2) or in media from Cook. There is no information whether embryos were randomised between IVF and G2; the results for these two media are pooled together |
| Van Langenendonckt 2001 | Quasi‐randomised trial; patients were alternately assigned to one of the two groups |
| Vansteenburgge 2007 | Retrospective study |
| Verheyen 2003 | Quasi‐randomised trial; alternating days |
| Verza 2003 | Not an RCT. Mentions "blindly randomised" but reports further along in the report that the oocytes were split equally |
| Warnes 1997 | Study on media supplementation |
| Westin 2003 | Quasi‐randomised trial; patients randomised alternatively |
| Wirleitner 2010 | Retrospective study |
| Xella 2009 | Retrospective study |
| Zech 2006 | Quasi‐randomised trial; patients were randomised according to the date of birth; in the second study, fertilized sibling oocytes from each patient were randomly assigned |
| Zhang 2002 | Retrospective study |
Characteristics of ongoing studies [ordered by study ID]
Dal Canto 2013.
| Trial name or title | ISM1 Versus EmbrioGen Media for Embryo Culture With Previous Failure Implantation in ART Treatments |
| Methods | Randomisation: Unknown Way of randomisation: Unknown Setting: Medical Centre Multicentre study: Unknown Time frame: September 2012 to September 2013 |
| Participants | Country: Italy Participants: 100 infertile couples with an indication for standard IVF or intracytoplasmic sperm injection. Inclusion and exclusion criteria: women experiencing at least two (maximum 4) failures of implantation after ART treatments (with elective embryo transfer) will be included. Treatments with frozen spermatozoa, frozen oocytes, women with PCO and PCOS and women with previous pregnancy, miscarriage or biochemical pregnancy will be excluded Baseline characteristics: Unknown |
| Interventions | Culture media: EmbryoGen (Origio), ISM1 (Origio) Duration of culture: 2 days Method of fertilisation: IVF or ICSI Mean number of embryos transferred: Unknown |
| Outcomes | Primary Outcome Measures: Ongoing pregnancy rate after 12 weeks of gestation Secondary Outcome Measures: Ongoing implantation rate after 12 weeks of gestation |
| Starting date | September 2012 |
| Contact information | ClinicalTrials.gov identifier: NCT01689454 and NCT01689428 |
| Notes |
Hardarson 2014.
| Trial name or title | Comparison of Human Embryo Development Using Sequential and Single Media With Time‐lapse Imaging |
| Methods | Randomisation: Yes Way of randomisation: Unknown Setting: Medical Center Multicentre study: Yes Time frame: August 2013 to November 2014 |
| Participants | Country: USA (California and Texas), Sweden Participants: 150 women, randomisation of embryos Inclusion and exclusion criteria: women with 6 or more normal embryos following egg retrieval and fertilisation, aged 21 to 40 years or 21 to 55 years who are using an egg donor and fresh or frozen ejaculated sperm will be included. Women with previous history of cancer or day‐3 embryo transfer will be excluded. Baseline characteristics: Maternal age = 21 to 40 years or 21 to 55 years for an egg donor |
| Interventions | Culture media: Vitrolife media Duration of culture: 5 days Method of fertilisation: Unknown Mean number of embryos transferred: Unknown |
| Outcomes | Primary Outcome Measures: good quality blastocysts Secondary Outcome Measures: biochemical pregnancy rate, implantation rate, ongoing pregnancy rate, live birth rate, birth weight |
| Starting date | August 2013 |
| Contact information | ClinicalTrials.gov identifier: NCT01939626 |
| Notes | Sponsored by Vitrolife |
Mastenbroek 2013.
| Trial name or title | A multicentre, double‐blind randomised controlled trial of two media for the in vitro culture of human pre‐implantation embryos. |
| Methods | Randomisation: Yes Way of randomisation: centralised online computer generated randomisation table Setting: University Medical Centre Multicentre study: Yes Time frame: Jan 2009 to May 2013 |
| Participants | Country: the Netherlands Participants: 784 subfertile couples undergoing in vitro fertilisation (IVF) or intracytoplasmic sperm injection (ICSI). Inclusion and exclusion criteria: couples undergoing a PGD or a modified natural cycle and couples for which IVF will be used to prevent the transmission of HIV will be excluded Baseline characteristics: Unknown |
| Interventions | Culture media: HTF (Irvine Scientific), G5 (Vitrolife) Duration of culture: 2 to 4 days Method of fertilisation: IVF or ICSI Mean number of embryos transferred: Unknown |
| Outcomes | Primary Outcome Measures: Live birth rate Secondary Outcome Measures: embryo quality, clinical pregnancy rate, ongoing pregnancy rate, miscarriage rate, time to pregnancy, birth weight and percentage of children with congenital abnormalities. |
| Starting date | January 2009 |
| Contact information | trialregister.nl identifier: NTR1979 |
| Notes |
Werner 2014.
| Trial name or title | |
| Methods | Randomisation: Yes Way of randomisation: paired study design, zygotes were randomised after confirming fertilisation and half of each patient’s zygotes were cultured in S media (Quinn’s Advantage Cleavage Medium, Sage; Blast Assist, Origio) and half in M media (Continuous Single Culture; Irvine Scientific). Setting: Unknown Multicentre study: No Time frame: July 2013 to August 2016 |
| Participants | Country: USA (New Jersey) Participants: 200 women, randomisation of embryos Inclusion and exclusion criteria: Inclusion: Women undergoing IVF or CCS (no PGD banking) that meet ASRM guidelines for double embryo transfer (DET), with AMH ≥ 1.2, FSH ≤ 12, BAFC ≥12, maximum of 1 prior failed IVF cycle for patients 35 to 45 years old, patients < 35 years old must have 1 prior failed IVF cycle, donor sperm OK. Exclusion: Chronic endometrial insufficiency, use of oocyte donor or gestational carriers, Medical contraindications to DET, male factor (< 100,000 sperm or surgical sperm), communicating hydrosalpinx (on HSG), Single gene disorders or sex selection Baseline characteristics: Unknown |
| Interventions | Culture media: Monophasic media (Continuous Single Culture; Irvine Scientific), Sequential media (Quinn’s Advantage Cleavage Medium, Sage; Blast Assist, Origio) Duration of culture: 5 to 6 days Method of fertilisation: IVF Mean number of embryos transferred: 2 |
| Outcomes | Primary Outcome Measures: pregnancy rate |
| Starting date | |
| Contact information | |
| Notes | October 2014 publication notes that "paired euploid transfers are now being performed to assess if there is any difference in pregnancy outcome in relation to the type of media utilized". |
IVF = in vitro fertilisation
ICSI = intracytoplasmic sperm injection
PCO = polycystic ovaries
PCOS = polycystic ovary syndrome
ART = assisted reproductive technology
PGD = preimplantation genetic diagnosis
HIV = human immunodeficiency virus
CCS = comprehensive chromosome screening
ASRM = American Society of Reproductive Medicine
AMH = anti‐Mullerian hormone
FSH = follicle‐stimulating hormone
BAFC = basal antral follicle count
HSG = hysterosalpingogram
Differences between protocol and review
The Background has been updated in line with current Cochrane recommendations
Change to inclusion criteria for intervention: culture media must be commercially available
Change to inclusion criteria: We excluded studies involving the addition of supplements (such as globulin or human serum albumin) to commercially available media
Primary outcome (formerly live birth) changed to live birth or ongoing pregnancy. A sensitivity analysis was added to indicate the rationale that while we acknowledge that a live birth is the most important clinical outcome, addition of data on ongoing pregnancy increases the power of the analysis (should pooling be possible)
Added outcomes: fertilisation rate, miscarriage rate
Data on embryo outcomes from studies randomising women or oocytes were reported as additional outcomes
Cryopreservation rate reported per total number of embryos instead of per randomised woman
Decision to use Mantel‐Haenszel odds ratios only, option to use Peto odds ratios for rare events removed
Due to lack of data, we were unable to perform meta‐analyses, sub‐group or sensitivity analyses for any of the outcomes.
Contributions of authors
Mohamed AFM Youssef & Eleni Mantikou: conception and design of study, acquisition, analysis and interpretation of data, writing and revision of the manuscript; final approval of manuscript
Madelon van Wely: conception and study of design, interpretation of data, consultation on statistical issues, revision of the manuscript, final approval of the manuscript
Sjoerd Repping & Sebastiaan Mastenbroek: conception and design of study, interpretation of data, revision of the manuscript, final approval of the manuscript
Fulco Van der Veen and Hesham Al‐Inany: conception and design of study, consultation on clinical issues, revision of the manuscript, final approval of the manuscript
Sources of support
Internal sources
No sources of support supplied
External sources
MDSG, New Zealand.
Declarations of interest
None known
New
References
References to studies included in this review
Arenas 2007 {published data only}
- Arenas L, Santos R, Garcia G, Galachi P. Comparison of two embryo culture systems in an ICSI program. Fertility and Sterility 2007;88(1):s324. [Google Scholar]
Artini 2004 {published data only}
- Artini PG, Valentino V, Cela V, Cristello F, Vite A, Genazzani AR. A randomized controlled comparison study of culture media (HTF versus P1) for human in vitro fertilization. European Journal of Obstetrics and Gynecology and Reproductive Biology 2004;116:196‐200. [DOI] [PubMed] [Google Scholar]
- Cristello F, Valentino V, Vite A, Matteucci C, Taponeco F, Artini P. Human tubal fluid vs P1: a randomized control comparison study of culture media for human in vitro fertilization. Gynecological Endocrinology 2000;14(Suppl 2):148. [Google Scholar]
Barak 1998 {published data only}
- Barak Y, Goldman S, Gonen Y, Nevo Z, Bartoov B, Kogosowski A. Does glucose affect fertilization, development and pregnancy rates of human in‐vitro fertilized oocytes?. Human Reproduction (Oxford, England) 1998;13(S4):203‐11. [DOI] [PubMed] [Google Scholar]
Baum 2004 {published data only}
- Baum M, Seidman DS, Yonash M, Levron J, Levran D, Mashiach S, et al. A prospective, randomized comparison on sibling oocytes of two commercial media for ivf and embryo culture. Human Reproduction 2004;19:i23. [Google Scholar]
Bird 2012 {published data only}
- Bird S, Summers M, Mirzazadeh F, Ottolini C, Muriel Rios L, Rogers S, et al. A comparative morphologic assessment of sibling zygotes cultured in a single medium versus a sequential medium. Reproductive BioMedicine Online 2012;24:s11‐2. [Google Scholar]
Bungum 2003 {published data only}
- Bungum M, Povlsen B, Madsen K, Poulsen A L, Elliman P, Humaidan P. Comparison of G1.2/G2.2 and the GIII series, sequential media for culture of human embryos. A prospective, randomized study. Fertility and Sterility 2003;80(S3):198. [Google Scholar]
Campo 2010 {published data only}
- Binda M, Campo R, Kerkhoven G, Frederickx V, Serneels A, Roziers P, et al. Comparison of the effects of two in vitro culture media on embryo fragmentation and pregnancy outcome. Human Reproduction 2010;25(suppl 1):i88. [Google Scholar]
- Campo R, Binda MM, Kerkhoven G, Frederickx V, Serneels A, Roziers P, et al. Critical reappraisal of embryo quality as a predictive parameter for pregnancy outcome: a pilot study. Facts, Views & Vision in ObGyn 2010;2(2):289‐95. [PMC free article] [PubMed] [Google Scholar]
Cano 2001 {published data only}
- Cano F, Gomez E, Perez‐Cano I, Amorocho B, Landeras J, Ballesteros A, et al. Comparison of two sets of culture media (Medi‐Cult and Scandinavian IVF Science) for assisted reproduction. Human Reproduction 2001;16(suppl 1):161‐2. [Google Scholar]
Carrasco 2013 {published data only}
- Carrasco B, Boada M, Rodriguez I, Coroleu B, Barri P, Veiga A. Does culture medium influence offspring birth weight?. Fertility and Sterility 2013;100(5):1283‐8. [DOI] [PubMed] [Google Scholar]
- Carrasco B, Boada M, Rodriguez I, Coroleu B, Barri PN, Veiga A. Does embryo culture medium influence birthweight of the newborn‐a randomised control trial. Human Reproduction 2013;28:i98. [Google Scholar]
Chatziioannou 2010 {published data only}
- Chatziioannou E, Kolibianakis EM, Venetis CA, Paggou T, Tokmakidou A, Sanopoulou T, et al. The effect of 2 different embryo culture systems on the probability of pregnancy: a randomized controlled trial. Journal fur Reproduktionsmedizin und Endokrinologie 2010;7(4):267. [Google Scholar]
Chen 2009 {published data only}
- Chen Q, Zhang AJ, Feng Y, Lu XW, Ji DM, Xu ZP. Effect of the P1 Medium and the ECM Medium on Embryo Quality in IVF. Journal of Reproduction and Contraception 2009;20(2):67‐72. [Google Scholar]
Fechtali 2004 {published data only}
- Fechtali B, Moor P, Roman K, Tranchant S, Porcher R, Rudant J, et al. Comparison of two embryo culture media in IVF and ICSI therapy. Human Reproduction 2004;19(suppl 1):i147. [Google Scholar]
Findikli 2004 {published data only}
- Findikli N, Kahraman S, Donmez E, Melil SS, Benkhalifa M. Comparison of embryo development in sibling oocytes cultured in two different sequential media. Middle East Fertility Society Journal 2004;9(2):150‐6. [Google Scholar]
Gimeno 2006 {published data only}
- Gimeno L, Lopez MA, Meseguer M, Santos JM, Pellicer A, Remohi J, et al. Comparative study of two different embryo culture media HTF and ISM‐1: Repercussion on both embryo phenotype and reproductive outcome. Revista Iberoamericana de Fertilidad y Reproduccion Humana 2006;23(6):375‐82. [Google Scholar]
Hazlett 2003 {published and unpublished data}
- Hazlett W, Karande V, Jobanputra K, Vietzke M. Media comparison: in vitro care and Irvine scientific. Fertility and Sterility 2002;78(suppl 1):s143. [Google Scholar]
Jamieson 1997 {published data only}
- Jamieson ME, George MA, Grossart E, Mitchell PAL, Mowat LG. A prospective, randomized comparison of two commercial media for IVF and embryo culture. Human Reproduction 1997;12:142. [Google Scholar]
Khan 2004 {published data only}
- Khan I, Urich M, Shmoury M, Abozeid T, Ulah K, Say M. Randomized study to compare P1 culture medium with the new early cleavage medium (ECM) for day 3 transfers. Fertility and Sterility 2004;82(2):S329. [Google Scholar]
Kyono 2000 {published data only}
- Kyono K, Fukunaga N, Chiba S, Matsuo Y, Sato T, Haryu M. Blastocyst formation and development in three different sequential culture media. Human Reproduction 2000;15(1):147. [Google Scholar]
Lambert 2005 {published data only}
- Chesmore J, Lambert H, Lopez A, Vu K. The evaluation of two different types of sequential culture media systems for human in vitro fertilization using sister oocytes. Fertility and Sterility 2005;83(2):S31. [Google Scholar]
- Lambert H, Vu K, Lopez A, Chesmore J. The evaluation of two different types of sequential culture systems for human in vitro fertilization using sister oocytes: interim results. Human Reproduction 2005;20(suppl 1):i156. [Google Scholar]
Mauri 2001 {published data only}
- Mauri AL, Petersen CG, Baruffi RL, Franco JG, Jr. A prospective, randomized comparison of two commercial media for ICSI and embryo culture. Journal of Assisted Reproduction and Genetics 2001;18:378‐81. [DOI] [PMC free article] [PubMed] [Google Scholar]
Mayer 2003 {published data only}
- Mayer JF, Jones EL, Lacey DR, Gibbons WE, Stadtmauer LA, Oehninger SC. In vitro culture of sibling oocytes in three different culture media. Annual meeting of the American Society for Reproductive Medicine (ASRM). 2003; Vol. 80, issue 3:S298.
- Mayer JF, Jones EL, Nehchiri F, Muasher SJ, Gibbons WE, Oehninger SC. A randomized prospective comparison of sibling human oocytes/embryos cultured in two different media. Fertility and Sterility 2001;76s:229. [Google Scholar]
Mendoza 2003 {published data only}
- Mendoza R, Expósito A, Corcóstegui B, Ramón O, Etxanojauregi A, Matorras R, et al. Comparison of IVF/ICSI results with three sequential culture media: Vitrolife, Medi‐Cult and Cook. Revista Iberoamericana de Fertilidad y Reproduccion Humana 2003;20(5):311‐5. [Google Scholar]
Paternot 2010 {published data only}
- Paternot G, Debrock S, D'Hooghe TM, Spiessens C. Early embryo development in a sequential versus single medium: a randomized study. Reproductive Biology and Endocrinology: RB&E 2010;8:83‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Rubino 2004 {published data only}
- Rubino P, Scaggiante F, Boldi Cotti P. Outcome in IVF between using two different culture media systems. Human Reproduction 2004;19(suppl 1):i142. [Google Scholar]
Sepulveda 2009 {published data only}
- Sepulveda S, Garcia J, Arriaga E, Diaz J, Noriega‐Portella L, Noriega‐Hoces L. In vitro development and pregnancy outcomes for human embryos cultured in either a single medium or in a sequential media system. Fertility and Sterility 2009;91:1765‐70. [DOI] [PubMed] [Google Scholar]
Shih 2014 {published data only}
- Shih YF, Lee TH, Liu CH, Tsao HM, Huang CC, Lee MS. Effects of reactive oxygen species levels in prepared culture media on embryo development: a comparison of two media. Taiwanese Journal of Obstetrics and Gynecology 2014;53(4):504‐8. [DOI] [PubMed] [Google Scholar]
Sifer 2009 {published data only}
- Sifer C, Handelsman D, Grange E, Porcher R, Poncelet C, Martin‐Pont B, et al. An auto‐controlled prospective comparison of two embryos culture media (GIII series versus ISM) for IVF and ICSI treatments. Journal of Assisted Reproduction and Genetics 2009;26(11‐12):575‐81. [DOI] [PMC free article] [PubMed] [Google Scholar]
Stevens 2000 {published data only}
- Stevens J, Schoolcraft WB, Schlenker T, Wagley L, Oliver S, Baird M. Comparison of G1.2 and Ham’s F10 With Fetal Cord Serum in Day 3 Transfer After IVF. Fertility and Sterility 2000;74:S106. [Google Scholar]
Tedesco 1990 {published data only}
- Tedesco T, Clark B, Miller K, Poole L, Emery M, Bernhisel M, et al. IVF fertilization and cleavage rates were not significantly different in human tubal fluid or Hams F10 with different serum supplements or Menezo B2. Fertility and Sterility. 1990:S118.
Von During 2004 {published data only}
- During V, Kjoetroedn S, Romundstad LB, Bakkan F, Lippe B, Remen A, et al. A prospective randomized study comparing culture media from COOK‐IVF and from MediCult in IVF/ICSI. Human Reproduction 2004;19(suppl 1):i148. [Google Scholar]
Yamamoto 2006 {published data only}
- Yamamoto M, Sugihara K, Nagata F, Nakaoka Y, Fukuda A, Morimoto Y. Comparison of two commercially available sequential media for their laboratory and clinical outcomes in split oocytes: a prospective and randomized study. Fertility and Sterility 2006;86(2):S228. [Google Scholar]
Zollner 2004 {published data only}
- Zollner KP, Zollner U, Schneider M, Dietl J, Steck T. Comparison of two media for sequential culture after IVF and ICSI shows no differences in pregnancy rates: a randomized trial. Medical Science Monitor. 2004; Vol. 10:CR1‐7. [PubMed]
- Zollner KP, Zollner U, Schneider M, Steck T, Dietl J. Randomized comparison of two media for extended culture after IVF and ICSI. Human Reproduction. 2002; Vol. 17, issue Suppl 1:39.
References to studies excluded from this review
Aoki 2005 {published data only}
- Aoki VW, Wilcox AL, Peterson CM, Parker‐Jones K, Hatasaka HH, Gibson M, et al. Comparison of four media types during 3‐day human IVF embryo culture. Reproduction Biomedicine Online 2005;10(5):600‐6. [DOI] [PubMed] [Google Scholar]
Assaf 1998 {published data only}
- Assaf A. Comparison of Medicult medium and BM1 medium for human IVF. Human Reproduction 1998;13:312. [Google Scholar]
Atchajaroensatit 2005 {published data only}
- Atchajaroensatit P, Jetsawagsri U, Tawiwong W, Bunyatin S, Ketcharoon, Lunta L, et al. A prospective randomized study comparing three culture media; COOK, Vitrolife and LifeGlobal in fresh and frozen‐thawed cycle. Human Reproduction 2005;20(suppl 1):i156‐60. [Google Scholar]
Balaban 2004 {published data only}
- Balaban B, Urman B. Comparison of two sequential media for culturing cleavage‐stage embryos and blastocysts: embryo characteristics and clinical outcome. Reproductive BioMedicine Online 2005;10:485‐91. [DOI] [PubMed] [Google Scholar]
- Balaban B, Urman B, Yakin K, Isiklar A, Bozdag H, Mumcu A. A prospective randomized comparison of two sequential culture media;G1.2/G2.2 versus GIII series. Fertility and Sterility 2004;82(2):S221. [Google Scholar]
- Urman B, Balaban B, Yakin K, Isiklar A, Mercan R, Mumcu A. GIII series compared with G1.2 and G2.2 media result in higher implantation and clinical pregnancy rates in frozen thawed embryo transfer cycles. Fertility and Sterility 2004;82(2):S151‐2. [Google Scholar]
Barrett 1997 {published data only}
- Barrett CB, Penzias AS, Powers RD. P1/SSS embryo culture medium yields a higher pregnancy rate than HTF/Plasmanate in IVF/ET. Fertility and Sterility 1997;68(1):s132. [Google Scholar]
Behr 2004 {published data only}
- Behr B, Wang H. Effects of culture conditions on IVF outcome. European Journal of Obstetrics, Gynecology, and Reproductive Biology 2004;115(Suppl 1):S72‐6. [DOI] [PubMed] [Google Scholar]
Ben‐Yosef 2001 {published data only}
- Ben‐Yosef D, Yovel I, Schwartz T, Azem F, Lessing JB, Amit A. Increasing synthetic serum substitute concentrations in P1 glucose/phosphate‐free medium improves implantation rate: a comparative study. Journal of Assisted Reproduction and Genetics 2001;18:588‐92. [DOI] [PMC free article] [PubMed] [Google Scholar]
Ben‐Yosef 2004 {published data only}
- Ben Yosef D, Azem F, Yovel I, Wagmann I, Lessing J, Amit A. Prospective randomized comparison of two embryo culture systems: P1 medium (Irvine) and Cook IVF medium. 17th World Congress on Fertility and Sterility. 2001:226.
- Ben‐Yosef D, Amit A, Azem F, Schwartz T, Cohen T, Mei‐Raz N, et al. Prospective randomized comparison of two embryo culture systems: P1 medium by Irvine Scientific and the Cook IVF medium. Journal of Assisted Reproduction and Genetics 2004;21(8):291‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
Biggers 2002 {published data only}
- Biggers JD, Racowsky C. The development of fertilized human ova to the blastocyst stage in KSOM (AA) medium: is a two‐step protocol necessary?. Reproductive Biomedicine Online 2002;5(2):133‐40. [DOI] [PubMed] [Google Scholar]
Bisioli 2003 {published data only}
- Bisioli C, Valcarcel A, Tiveron M, Quintana R, Young E, Sueldo E. Better embryo quality and reproductive performance using sequential media in day 3 transfers. Fertility and Sterility 2003;80(3):S57. [Google Scholar]
Blefco 2001 {published data only}
- Blefco DA, Devaux A, Junca AM, Plachot M, Royere D. Comparison between IVF and ICSI blastocyst culture and freezing: a French retrospective multicentre study. Human Reproduction (Oxford) 2001;16(abstract Book 1):167. [Google Scholar]
Bungum 2002 {published data only}
- Bungum M, Humaidan P, Bungum L. Recombinant human albumin as protein source in culture media used for IVF: a prospective randomized study. Reproductive Biomedicine Online 2002;4(3):233‐6. [DOI] [PubMed] [Google Scholar]
- Bungum M, Humaiden P, Bungum L. Recombinant human albumin as protein source in culture media used for IVF ‐ a prospective randomized study. Human Reproduction 2002;17:37‐8. [DOI] [PubMed] [Google Scholar]
- Bungum M, Humaiden P, Bungum L. Recombinant human albumin as protein source in culture media used for in vitro fertilization, a prospective randomized study. Fertility and Sterility 2002;78(3):s56‐7. [DOI] [PubMed] [Google Scholar]
Choi 2004 {published data only}
- Choi SJ, Kim SK, Chun KW, Koong MK, Kang IS, Yang KM, et al. Efficacy of sequential culture media system in the frozen‐thawed embryo transfer of human pro‐nuclear zygotes. Human Reproduction 2004;19(suppl 1):i108. [Google Scholar]
Ciray 2012 {published data only}
- Ciray HN, Aksoy T, Goktas C, Ozturk B, Bahceci M. Time‐lapse evaluation of human embryo development in single versus sequential culture media—a sibling oocyte study. Journal of Assisted Reproduction and Genetics 2012;29:891‐900. [DOI] [PMC free article] [PubMed] [Google Scholar]
Coates 1999 {published data only}
- Coates A, Rutherford AJ, Hunter H, Leese HJ. Glucose‐free medium in human in vitro fertilization and embryo transfer: a large‐scale, prospective, randomized clinical trial. Fertility and Sterility 1999;72:229‐32. [DOI] [PubMed] [Google Scholar]
Cooke 2002 {published data only}
- Cooke S, Quinn P, Kime L, Ayres C, Tyler JPP, Driscoll GL. Improvement in early human embryo development using new formulation sequential stage‐specific culture media. Fertility and Sterility 2002;78(6):2002. [DOI] [PubMed] [Google Scholar]
Cummins 1986 {published data only}
- Cummins JM, Breen TM, Fuller SM, Harrison KL, Wilson LM, Hennessey JF, et al. Comparison of two media in a human in vitro fertilization program: lack of significant differences in pregnancy rate. Journal of In Vitro Fertilization and Embryo Transfer 1986;3(5):326‐30. [DOI] [PubMed] [Google Scholar]
de Araujo 2009 {published data only}
- Araujo CH, Nogueira D, Araujo MC, Martins Wde P, Ferriani RA, dos Reis RM. Supplemented tissue culture medium 199 is a better medium for in vitro maturation of oocytes from women with polycystic ovary syndrome than human tubal fluid. Fertility and Sterility 2009;91:509‐13. [DOI] [PubMed] [Google Scholar]
Di Falco Cossiello 2011 {published data only}
- Falco Cossiello R, Aggelis A, Faundes D, Petta CA. Morphological differences in human zygotes and embryos cultured in different media. Zygote 2011;30:1‐7. [DOI] [PubMed] [Google Scholar]
Dudkiewicz 2003 {published data only}
- Dudkiewicz A, Poole E, Novotny M, Zhu J, Kim H, Cohen D. Do early cleavage‐stage embryos demonstrate a culture media preference?. Fertility and Sterility 2003;80(3):S166. [Google Scholar]
Dumoulin 2010 {published data only}
- Dumoulin JC, Land JA, Montfoort AP, Nelissen EC, Coonen E, Derhaag JG, et al. Effect of in vitro culture of human embryos on birthweight of newborns. Human Reproduction 2010;25(3):605‐12. [DOI] [PubMed] [Google Scholar]
Eaton 2012 {published data only}
- Eaton JL, Lieberman ES, Stearns C, Chinchilla M, Racowsky C. Embryo culture media and neonatal birthweight following IVF. Human Reproduction 2012;27(2):375‐9. [DOI] [PubMed] [Google Scholar]
Fatemeh 2012 {published data only}
- Fatemeh H, Eftekhari‐Yazdi P, Karimian L, Fazel M, Foulafi H, Johansson L. Improved embryo quality, higher birth weight and length of babies born after culture in ISM1 (Medicult, Denmark) in comparison to G‐1TM v5 (Vitrolife, Sweden). Human Reproduction 2012;27(suppl 2):ii137. [Google Scholar]
Feichtinger 1986 {published data only}
- Feichtinger W, Kemeter P, Menezo Y. The use of synthetic culture medium and patient serum for human in vitro fertilization and embryo replacement. Journal of In Vitro Fertilization and Embryo Transfer 1986;3(2):87‐92. [DOI] [PubMed] [Google Scholar]
Fukunaga 2003 {published data only}
- Fukunaga N, Kyono K, Nakajo Y, Chiba S, Sasaki T. The rate of blastocyst formation and development between using three different sequential culture media. Human Repruduction 2003;18(suppl 1):147‐8. [Google Scholar]
Geber 2012 {published data only}
- Geber S, Bossi R, Guimarães F, Valle M, Sampaio M. Effects of transfer of embryos independently cultured in essential and sequential culture media on pregnancy rates in assisted reproduction cycles. Journal of Assisted Reproduction and Genetics 2012;29(10):1097‐1101. [DOI] [PMC free article] [PubMed] [Google Scholar]
Gianaroli 1989 {published data only}
- Gianaroli L, Trounson A, King C, Ferraretti A, Chiappazzo L, Bafaro G. Human amniotic fluid for fertilization and culture of human embryos: results of clinical trials in human in vitro fertilization (IVF) programs. Journal of In Vitro Fertilization and Embryo Transfer 1989;6(4):213‐7. [DOI] [PubMed] [Google Scholar]
Gilson 2013 {published data only}
- Gilson A, Piront N, Heens B, Vastersaegher C, Vansteenbrugge A, Pauwels PCP. Clinical outcomes following the transfer of vitrified blastocysts derived from a single or sequential culture medium. Human Reproduction 2013;28(suppl 1):172. [Google Scholar]
Hambiliki 2011 {published data only}
- Hambiliki F, Sandell P, Yaldir F, Stavreus‐Evers A. A prospective randomized sibling oocyte study of two media systems for culturing cleavage‐stage embryos‐impact on fertilization rate. Journal of Assisted Reproduction and Genetics 2011;28(4):335‐41. [DOI] [PMC free article] [PubMed] [Google Scholar]
Hassani 2013 {published data only}
- Hassani F, Eftekhari‐Yazdi P, Karimian L, Valojerdi MR, Movaghar B, Fazel M, et al. The effects of ISM1 medium on embryo quality and outcomes of IVF/ICSI cycles. International Journal of Fertility and Sterility 2013;7(2):108‐15. [PMC free article] [PubMed] [Google Scholar]
Hirai 2003 {published data only}
- Hirai K, Utsunomiya T, Araki Y. Clinical application of a new medium product of 'HFF99' to human in vitro fertilization. Japanese Journal of Fertility & Sterility 2003;48:17‐22. [Google Scholar]
Hoogendijk 2007 {published data only}
- Hoogendijk CF, Kruger TF, Beer MB, Siebert TI, Henkel R. A study of two sequential culture media ‐ impact on embryo quality and pregnancy rates. South African Journal of Obstetrics and Gynaecology 2007;13(2):52‐9. [Google Scholar]
Ignoul Vanvuchelen 1996 {published data only}
- Ignoul Vanvuchelen R, Dumoulin JCM, Bergers Janssen JM, Brass M, Wissen LCP, Geraedts JPM, et al. Comparison of IVF‐50 commercial medium with in house prepared human tubal fluid medium for human IVF. Human Reproduction. 1996; Vol. 11:17.
Isiklar 1998 {published data only}
- Isiklar A, Balaban B, Alatas C, Aksoy S, Mercan R, Urman B. Comparison of IVF‐50 and S1 medium for human IVF. Human Reproduction 1998;13:332. [Google Scholar]
- Isiklar A, Balaban B, Alatas C, Aksoy S, Mercan R, Urman B. Comparison of IVF‐50 and S2 medium for in‐vitro culture of ICSI embryos from day 2 to day 3. Human Reproduction 1998;13(suppl 1):235. [Google Scholar]
Janssens 1996 {published data only}
- Janssens R, Carle M, Clerck E, Henderix P, LaurierK, Lebrun R, et al. Comparison of Menezo B2 medium with IVF‐50™ medium for human IVF. Human Reproduction 1996;11(Suppl 1):19. [Google Scholar]
Jindal 2005 {published data only}
- Jindal SK, Zapantis A, Nihsen M, Lieman HJ, Barad D. Comparison of embryo quality from sibling zygotes cultured in G1v3 or HTF media. Fertility and Sterility 2005;84(1):S18‐9. [Google Scholar]
Jones 1998 {published data only}
- Jones GM, Trounson AO, Vella P, Kausche A, Lolatgis N, Wood C. Development of viable human embryos to the blastocyst stage following initial culture in a simple or a complex culture medium. Human Reproduction 1998;13:216. [Google Scholar]
Kasai 2001 {published data only}
- Kasai G, Fujii S, Fukui A. Morula/blastocyst transfer using two sequential media. 17th World Congress on Fertility and Sterility. 2001:164.
Kasterstein 1999 {published data only}
- Kasterstein E, Strassburger D, Komarovksy D, Bern O, Friedler S, Raziel A, et al. Blastocyst formation and development in different culture media.. Human Reproduction 1999;14(Suppl 1):198‐9. [Google Scholar]
Khoury 2012 {published data only}
- Khoury C, Coffler M, Potter D, Frederick J, Battaglia D. Improved blastocyst development using a single step medium versus a sequential medium. Fertility and Sterility 2012;97(3 suppl 1):S4‐5. [Google Scholar]
Kim 2002 {published data only}
- Kim SH, Ku SY, Oh SK, Choi YM, Moon SY, Lee JY. Blastocyst transfer following in vitro culture for 5 days. Human Reproduction 2002;17(suppl 1):138‐9. [Google Scholar]
Kleijkers 2014 {published data only}
- Kleijkers SH, Montfoort AP, Smits LJ, Viechtbauer W, Roseboom TJ, Nelissen EC, et al. IVF culture medium affects post‐natal weight in humans during the first 2 years of life. Human Reproduction 2014;29(4):661‐9. [DOI] [PubMed] [Google Scholar]
- Kleijkers SH, Montfoort AP, Smits LJ, Viechtbauer W, Roseboom TJ, Nelissen EC, et al. IVF culture medium affects post‐natal weight in humans during the first 2 years of life. Human Reproduction 2014;29(4):661‐9. [DOI] [PubMed] [Google Scholar]
Komarovsky 2007 {published data only}
- Komarovsky D, Strassburger D, Bern O, Kasterstein E, Komsky E, Raziel A, et al. Comparison of two human embryo culture systems: SAGE versus Vitrolife ART line of products. Human Reproduction 2007;22(suppl 1):i167. [Google Scholar]
Kyono 1999 {published data only}
- Kyono K, Momma K, Chiba S, Otomo Y, Haryu M. Comparison of human tubal fluid (HTF) and P1 culture media for human IVF and ICSI. 11th World Congress on In Vitro Fertilization and Human Reproductive Genetics. 1999:202.
Kyono 2002 {published data only}
- Kyono K, Fukunaga N, Haigo K, Chiba S, Nakajo Y, Fuchinoue K. Two step (day 1 to 3, day 5) embryo transfers improve on‐going pregnancy rate in women with multiple failures of conception after ART. Fertility and Sterility 2002;78(3):S230. [Google Scholar]
Lane 2007 {published data only}
- Lane M, Gardner DK. Embryo culture medium: which is the best?. Best Practice & Research. Clinical Obstetrics & Gynaecology 2007;21:83‐100. [DOI] [PubMed] [Google Scholar]
Liebermann 2012 {published data only}
- Liebermann J, Pelts EJ, Brohammer R, Wagner Y, Sipe C, Lederer K. Single‐step vs. two‐step media: Are there any differences in outcome and implantation?. Fertility and Sterility 2012;98(3):166. [Google Scholar]
Lin 2014 {published data only}
- Lin YH, Hwang JL, Huang LW, Seow KM, Hsieh BC, Tzeng CR. Comparison of Quinn’s Advantage fertilization medium and tissue culture medium 199 for in vitro maturation of oocytes. Taiwanese Journal of Obstetrics & Gynecology 2014;53:17‐20. [DOI] [PubMed] [Google Scholar]
Lopes 2008 {published data only}
- Lopes VM, Pereira TR, Lopes JRC, Café TC, Almodin CG, Minguetti‐Camara VC. A comparison between GV and HTF/P1 media in IVF in‐vitro [Estudo comparativo entre meio GV e HTF/P1 em fertilizacao in‐vitro]. Jornal Brasileiro de Reproducao Assistida 2008;12:11‐4. [Google Scholar]
Mahadevan 1996 {published data only}
- Mahadevan MM, Miller MM, Moutos DM. Improved human zygote development in a modified Ham's F10 medium in vitro. Journal of Assisted Reproduction and Genetics 1996;13(9):722‐5. [DOI] [PubMed] [Google Scholar]
Mantikou 2013 {published data only}
- Mantikou E, Youssef MAFM, Wely M, Veen F, Al‐inany HG, Repping S, et al. Embryo culture media and IVF/ICSI success rates: A systematic review. Human Reproduction Update 2013;19:210‐20. [DOI] [PubMed] [Google Scholar]
Mayer 1998 {published data only}
- Mayer J, Weedon V, Nehchiri F, Jones E, Oehninger S, Gardner DK, et al. Blastocyst development of human embryos with poor morphological grades: a comparison of two different culture systems. Fertility and Sterility 1998;30:S118. [Google Scholar]
Mayer 1999 {published data only}
- Mayer IF, Nehchiri F, Kalin HL, Jones EL, Weedon VM, Lanzendorf SE, et al. Prospective randomized study of two culture media on pregnancy rates with cryo preserved zygotes. Fertility and Sterility 1999;72(3):s145. [Google Scholar]
Mayer 1999b {published data only}
- Mayer JF, Nehchiri F, Kalin HL, Jones EL, Weedon VM, Lanzendorf SE, et al. Prospective randomized study of two culture media on pregnancy rates with cryo preserved zygotes. Fertility and Sterility 1999;72(3):145. [Google Scholar]
Mellon 2005 {published data only}
- Mellon JA, Salvador CJ, Fluker MR, Yuzpe AA, Kelk DA. A randomized prospective comparison of a sequential versus single media culture system. Fertility and Sterility 2005;84(suppl 1):S449‐50. [Google Scholar]
Merry 2002 {published data only}
- Merry N, Attard M, Tucker K, Edgar DH. A comparison of early embryo development in two culture systems. 21st Annual Scientific Meeting of the Fertility Society of Australia. 2002:56.
Michaeli 2007 {published data only}
- Michaeli M, Ellenbogen A, Rotfarb N, Karchovsky E, Shalom‐Paz E, Hallack M. In vitro fertilization outcome using three types of culture media containing different concentrations of glucose for developing cleavage stage embryos. A comparative study. Fertility and Sterility 2007;88(suppl 1):S324. [Google Scholar]
Moodie 2004 {published data only}
- Moodie G, Schenkman E, Chao S, Sinclair P, Brenner S, Kenigsberg D. A comparison of Global, P1 and Quinn’s Advantage culture media:There was no significant difference in clinical outcome. Fertility and Sterility 2004;82(2):328. [Google Scholar]
Mortimer 2003 {published data only}
- Mortimer ST, Fluker MR, Yzupe AA. Effect of culture system upon fertilization rate and embryo development for patients 40 years and older. Fertility and Sterility 2003;80(1):S168. [Google Scholar]
Nelissen 2012 {published data only}
- Nelissen E, Moontfoort APA, Menheere PPCA, Evers JLH, Peeters LL, Dumoulin JCM. Effect of IVF culture medium on human fetal growth. Human Reproduction 2011;26(suppl 1):i67. [Google Scholar]
- Nelissen EC, Montfoort AP, Coonen E, Derhaag JG, Geraedts JP, Smits LJ, et al. Further evidence that culture media affect perinatal outcome: findings after transfer of fresh and cryopreserved embryos. Human Reproduction 2012;27(7):1966‐76. [DOI] [PubMed] [Google Scholar]
- Nelissen ECM, Montfoort APA, Coonen E, Derhaag JG, Evers JLH, Dumoulin JCM. In vitro culture affects the birthweight of human singletons. Human Reproduction 2010;25(suppl 1):i238. [Google Scholar]
Parinaud 1987 {published data only}
- Parinaud J, Monrozoies X, Reme JM, Sarramon MF, Grandjean H, Pontonnier G. Randomized trail of two media used for in vitro fertilization. European Journal of Obstetrics, Gynecology, and Reproductive Biology 1987;25:203‐8. [DOI] [PubMed] [Google Scholar]
Parinaud 1998 {published data only}
- Parinaud J, Veiga A, Vieitez G, Busquets A, Milhet P, Calderon C, et al. Randomized comparison of three media used for embryo culture after intracytoplasmic sperm injection. Human reproduction (Oxford, England) 1998;13:212‐17. [DOI] [PubMed] [Google Scholar]
Parinaud 1999 {published data only}
- Parinuard J, Milhet P, Vieitez G, Richoilley G. Use of a medium devoid of any human animal compound (SMART2) for embryo culture in intracytoplasmic sperm injection. Journal of Assisted Reproduction and Genetics 1999;16(1):13‐6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Perri 2001 {published data only}
- Perri T, Yoeli R, Mor L, Shelef M, Schwartz A, Orvieto R, et al. Culture media in assisted reproduction technology practice, a comparison study. Human Reproduction 2001;16(Suppl 1):19‐21. [Google Scholar]
Pool 2012 {published data only}
- Pool TB, Schoolfield J, Han D. Human embryo culture media comparisons.. Methods in Molecular Biology 2012;912:367‐86. [DOI] [PubMed] [Google Scholar]
Prados 2002 {published data only}
- Prados N, Calderon G, Crespo M, Remohi J, Caligara C, Navarro J. Culture media and protein sources in the optimisation of human embryo quality. Fertility and Sterility 2002;78(3):S245. [Google Scholar]
Quinn 1985 {published data only}
- Quinn P, Kerin JF, Warnes GM. Improved pregnancy rate in human in vitro fertilization with the use of a medium based on the composition of human tubal fluid. Fertility and Sterility 1985;44:493‐8. [DOI] [PubMed] [Google Scholar]
Quinn 1985b {published data only}
- Quinn P, Warnes GM, Kerin JF, Kirby C. Culture factors affecting the success rate of in vitro fertilization and embryo transfer. Annals of the New York Academy of Sciences 1985;442:195‐204. [DOI] [PubMed] [Google Scholar]
Quinn 1995 {published data only}
- Quinn P. Enhanced results in mouse and human embryo culture using a modified human tubal fluid medium lacking glucose and phosphate 1. Journal of Assisted Reproduction and Genetics 1995;12(2):97‐105. [DOI] [PubMed] [Google Scholar]
Quinn 1995b {published data only}
- Quinn P, Moinipanah R, Steinberg JM, Weathersbee PS. Successful human in vitro fertilization using a modified human tubal fluid medium lacking glucose and phosphate ions. Fertility and Sterility 1995;63:922‐4. [DOI] [PubMed] [Google Scholar]
Reed 2009 {published data only}
- Reed ML, Hamic A, Thompson DJ, Caperton CL. Continuous uninterrupted single medium culture without medium renewal versus sequential media culture: a sibling embryo study. Fertility and Sterility 2009;92(5):1783‐6. [DOI] [PubMed] [Google Scholar]
Rinehart 1998 {published data only}
- Rinehart J, Chapman C, McKiernan S, Bavister B. A protein‐free chemically defined embryo culture medium produces pregnancy rates similar to human tubal fluid (HTF) supplemented with 10% synthetic serum substitute (SSS). Human Reproduction 1998;13:59. [Google Scholar]
- Rinehart J, Chapman CA, McKiernan SA, Bavister B. Birth weights are similar in children from embryos cultured in a protein‐free chemically‐defined medium compared to embryos cultured in human tubal fluid (HTF) supplemented with 10% synthetic serum substitute (SSS). Fertility and Sterility 1998;70(3):S290. [Google Scholar]
Romano 2004 {published data only}
- Romano S, Scarselli F, Minasi MG, Martinez F, Icobelli M, Sapienza F, et al. Effect of different culture systems on embryo development potential. Human Reproduction 2004;19(suppl 1):i6‐7. [Google Scholar]
Ruiz 1999 {published data only}
- Ruiz A, Cobo AC, Perez‐Cano I, Herrer R, Ochoa S, Moreno D, et al. In vitro culture medium without albumin for an in vitro fertilization program. Fertility and Sterility 1999;72(suppl 1):S245‐6. [Google Scholar]
Sieren 2003 {published data only}
- Sieren K, Barnett B, Douglas J, Rodriguez A, Madden J, Meintjes M. Clinical outcomes of a randomized trial between G‐media versions two and three in a predominantly blastocyst program. Fertility and Sterility 2003;80(Suppl 3):S294. [Google Scholar]
Sieren 2006 {published data only}
- Sieren KR, Grainger DA, Tjadena BL, Balla GD. In vitro outcome of a prospective, randomized trial comparing the GIII Series™ and Quinn’s Advantage® sequential media systems for embryo development. Fertility and Sterility 2006;86:s233‐4. [Google Scholar]
Staessen 1994 {published data only}
- Staessen C, Abbeel E, Janssenswillen C, Devroey P, Steirteghem AC. Controlled comparison of Earle's balanced salt solution with Mėnėzo B2 medium for human in‐vitro fertilization performance. Human Reproduction 1994;10:1915‐9. [DOI] [PubMed] [Google Scholar]
Staessen 1998 {published data only}
- Staessen C, Janssenswillen C, Clerck E, Steirteghem A. Controlled comparison of commercial media for human in‐vitro fertilization: Menezo B2 medium versus Medi‐Cult universal and BM1 medium. Human Reproduction 1998;13(9):2448‐54. [DOI] [PubMed] [Google Scholar]
- Clerck E, Carle M, Henderix P, Janssens R, Laurier K, Lebrun F, et al. Comparison of Menezo B2 medium with BMI medium for human IVF. Human Reproduction 1996;11:18‐9. [Google Scholar]
Steele 2006 {published data only}
- Steele MG, Kopakaki A, Sriskandakumar R, Papanikou C, Sciorio R, Mary N, et al. Comparison of Medicult SequentialMedium ISM1 with non‐Sequential universalIVF Medium in an IVF/ICSI programme. Human Fertility 2006;9(4):259. [Google Scholar]
Steinkampf 2001 {published data only}
- Steinkampf MP, Hubbard JA, Kretzer PA, Hammond KR, Tucker M. A comparison of two sequential IVF media systems on fertilization rates and embryo quality. Fertility and Sterility 2001;76(3):S215. [Google Scholar]
Stoddart 2001 {published data only}
- Stoddard NR, Fleming SD. A comparison of P1/P2 and G12/G22 sequential culture media systems. 17th World Congress on Fertility and Sterility. 2001; Vol. November 2001:165.
Stoddart 2002 {published data only}
- Stoddart NR, Rho HC, Hussain A, Fleming SD, Bernhisel M. A comparison of the effects of short‐term oocyte culture in glucose/phosphate‐free medium or in glucose/ phosphate‐supplemented medium prior to intracytoplasmic sperm injection (ICSI) on clinical outcome. Fertility and Sterility 2002;78(1):S142‐3. [Google Scholar]
Summers‐Chase 2004 {published data only}
- Summers‐Chase D, Check JH, Swenson K, Yuan W, Brittingham D, Barci H. A comparison of in vitro fertilization outcome by culture media used for developing cleavage‐stage embryos. Clinical and Experimental Obstetrics & Gynecology 2004;31:179‐82. [PubMed] [Google Scholar]
- Summers‐Chase D, Swenson K, Cochrane H, Yuan W, Brittingham D, Check JH. A random comparison of a new non‐sequential media, a new sequential media, and a standard non‐sequential media on in vitro fertilization outcome. Fertility and Sterility 2002;78:S287. [Google Scholar]
Sun 2001 {published data only}
- Sun M, Feng Y, Gao Q. Application of sequential culture in assisted reproductive technique. Zhonghua Fu Chan Ke Za Zhi 2001;36(2):92‐4. [PubMed] [Google Scholar]
Szell 1998 {published data only}
- Szell AZ, Antaran JM, Chetkowski RJ. Pregnancy and implantation rates from the transfer of human embryos cultured in P1 or human tubal fluid medium and transferred with or without assisted hatching. Fertility and Sterility 1998;30:S495. [Google Scholar]
Tekpetey 1998 {published data only}
- Tekpetey FR, Martin JSB, Tummon IS, Nisker J, Power S. Randomised control comparison of human tubal fluid (HTF) and P1 culture media for human IVF. Fertility and Sterility 1998;70:S17. [Google Scholar]
Thorsell 2003 {published data only}
- Thorsell LP, Iwaszko MA, Palmer MA, Hook R, Dorfmann AD, Blauer KL. Use of two commercially available sequential media culture systems does not affect pregnancy rates. Fertility and Sterility 2003;80(3):296‐7. [Google Scholar]
Torres 2012 {published data only}
- Torres P, Peinado I, Orden M, Monzo A, Rubio JM, Pellicer A. Evaluation of the efficiency of different commercial systems of embryo culture [Evaluacion de la eficacia de diferentes sistemas comerciales de cultivo embrionario]. Revista Iberoamericana de Fertilidad y Reproduccion Humana 2012;27(5):417‐24. [Google Scholar]
Utsunomiya 2002 {published data only}
- Utsunomiya T, Naitu T, Nagaki M. A prospective trial of blastocyst culture and transfer. Human Reproduction 2002;17:1846‐51. [DOI] [PubMed] [Google Scholar]
Van der Auwera 2001 {published data only}
- Auwera I, Spiessens C, Debrock S, Afschrift H, Meeuwis I, Meuleman C, et al. IVF outcome in two different sequential culture media. 17th World Congress on Fertility and Sterility. 2001:94.
Van Langenendonckt 2001 {published data only}
- Langendonckt A, Godin PA, Demylle D, Beliard A, Wyns C. Pregnancy outcome following human embryo culture in G11/G22 and Sydney IVF cleavage/blastocyst sequential media: a prospective randomized comparative study. Human Reproduction 2000;15:141‐2. [Google Scholar]
- Langenendonckt A, Demylle D, Wyns C, Nisolle M, Donnez J. Comparison of G1.2/G2.2 and Sydney IVF cleavage /blastocyst sequential media for the culture of human embryos: a prospective randomized comparative study. Fertility and Sterility 2001;76:1023‐31. [DOI] [PubMed] [Google Scholar]
Vansteenburgge 2007 {published data only}
- Vansteenbrugge A, Vastersaegher C, Fontanie P, Klay C, Rodez D, Pauwels PC. Comparison of a single medium with sequential media for the development of human zygotes to the blastocyst stage. Fertility and Sterility 2007;88(suppl 1):S319. [Google Scholar]
Verheyen 2003 {published data only}
- Verheyen G, Landuyt L, Joris H, Devroey P, Steirtegham A. Different embryo development patterns in different sequential media appear to result in similar pregnancy rates. Human Reproduction 2003;18(Suppl 1):xviii36‐8. [Google Scholar]
Verza 2003 {published data only}
- Verza S, Schneider DT, Esteves SC. Comparison of two systems to culture human embryos up to day 3: a prospective randomized study. Fertility and Sterility 2003;88(suppl 1):149‐50. [Google Scholar]
Warnes 1997 {published data only}
- Warnes GM, Payne D, Jerrrey R, Hourigan L, Kirby C, Kerin J, et al. Reduced pregnancy rates following the transfer of human embryos frozen or thawed in culture media supplemented with normal serum albumin. Human Reproduction 1997;12(7):1525‐30. [DOI] [PubMed] [Google Scholar]
Westin 2003 {published data only}
- Westin C, Borg K, Hillensjo T, Jakobsson AH, Olofsson J, Reismer E, et al. A comparison between a conventional medium and a new generation of sequential media (GIII) for transfer of early cleavage embryo. Human Reproduction 2003;18(1):151. [Google Scholar]
Wirleitner 2010 {published data only}
- Wirleitner B, Vanderzwalmen P, Stecher A, Zech MH, Zintz M, Zech NH. Individual demands of human embryos on IVF culture medium: influence on blastocyst development and pregnancy outcome. Reproductive Biomedicine Online 2010;21(6):776‐82. [DOI] [PubMed] [Google Scholar]
Xella 2009 {published data only}
- Xella S, Marsella T, Tagliasacchi D, Giulini S, Marca A, Tirelli A, et al. Embryo quality and implantation rate in two different culture media: ISM1 versus Universal IVF Medium. Fertility and Sterility 2009;93(6):1859‐63. [DOI] [PubMed] [Google Scholar]
Zech 2006 {published data only}
- Zech N, Stecher A, Zech H, Uher P, Vanderzwalmen P. Prospective analysis of embryo development to day 5 and transfer outcome in sequential medium (G1.3‐G2.3) vs. a one step protocol (Global medium). Human Reproduction 2006;21(suppl 1):i162‐3. [Google Scholar]
Zhang 2002 {published data only}
- Zhang X, Horan K, Kallmann K, Confino E, Kazer RR. A comparison between culture media from two commercial vendors for their ability to support embryo development. Human Reproduction 2002;17(suppl 1):137. [Google Scholar]
References to ongoing studies
Dal Canto 2013 {published and unpublished data}
- Dal Canto, et al. ISM1 Versus EmbrioGen Media for Embryo Culture With Previous Failure Implantation in ART Treatments. http://clinicaltrials.gov/ct2/show/NCT01689454?term=ivf+culture&rank=5.
Hardarson 2014 {published data only}
- Harsarson, et al. Comparison of Human Embryo Development Using Sequential and Single Media With Time‐lapse Imaging. http://www.clinicaltrials.gov/ct2/show/record/NCT01939626?term=ivf+culture&rank=26.
Mastenbroek 2013 {published data only}
- Mastenbroek, et al. A multi‐center double‐blind randomized controlled trial of two media for the in vitro culture of human preimplantation embryos.. http://www.trialregister.nl/trialreg/admin/rctview.asp?TC=1979.
Werner 2014 {published data only}
- Werner MD, Hong KH, Franasiak JM, Reda CV, et al. Sequential (S) vs. monophasic (M) media impact trial (Summit): A paired randomized controlled trial comparing blastulation rates or aneuploidy risk amongst sibling zygotes.. Fertility and Sterility. 2014; Vol. 102(3 SUPPL 1):e129‐30. [DOI] [PubMed]
- Werner, et al. Sequential vs. Monophasic Media Impact Trial (SuMMIT). http://www.clinicaltrials.gov/ct2/show/record/NCT01917240?term=ivf+culture&rank=41. [DOI] [PubMed]
Additional references
Blake 2007
- Blake DA, Farquhar CM, Johnson N, Proctor M. Cleavage stage versus blastocyst stage embryo transfer in assisted conception. Cochrane Database of Systematic Reviews 2007, Issue 4. [DOI: 10.1002/14651858.CD002118.pub3] [DOI] [PubMed] [Google Scholar]
Doherty 2000
- Doherty AS, Mann MR, Tremblay KD, Bartolomei MS, Schultz RM. Differential effects of culture on imprinted H19 expression in the preimplantation mouse embryo. Biology of Reproduction 2000;62:1526‐35. [DOI] [PubMed] [Google Scholar]
Fauque 2007
- Fauque P, Jouannet P, Lesaffre C, Ripoche M, Dandolo L, Vaiman D, et al. Assisted reproductive technology affects developmental kinetics, H19 Imprinting control region methylation and H19 gene expression in individual mouse embryos. BMC Developmental Biology 2007;7:116. [DOI: 10.1186/1471-213X-7-116] [DOI] [PMC free article] [PubMed] [Google Scholar]
Gardner 2002 a
- Gardner DK, Lane M, Schoolcraft WB. Physiology and culture of the human blastocyst. Journal of Reproductive Immunology 2002;55(1‐2):85‐100. [DOI] [PubMed] [Google Scholar]
Gardner 2002 b
- Gardner DK, Lane M. Development of viable mammalian embryos in vitro: evolution of sequential media. Principles of Cloning. New York: Academic Press, 2002:187–213. [Google Scholar]
Higgins 2011
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. Available from www.cochrane‐handbook.org 2011.
Hoelker 2010
- Hoelker M, Rings F, Lund Q, Phatsara C, Schellander K, Tesfaye D. Effect of embryo density on in vitro developmental characteristics of bovine preimplantative embryos with respect to micro and macroenvironments. Reproduction in Domestic Animals 2010;45:138‐45. [DOI] [PubMed] [Google Scholar]
Khosla 2001
- Khosla S, Dean W, Reik W, Feil R. Culture of preimplantation embryos and its long‐term effects on gene expression and phenotype. Human Reproduction Update 2001;7(4):419‐27. [DOI] [PubMed] [Google Scholar]
Menezo 2004
- Menezo Y, Testart J, Perrone D. Serum is not necessary in human invitro fertilization, early embryo culture, and transfer. Fertility and Sterility 1984;42:750‐5. [DOI] [PubMed] [Google Scholar]
Zander 2006
- Zander DL, Thompson JG, Lane M. Perturbations in mouse embryo development and viability caused by ammonium are more severe after exposure at the cleavage stages. Biology of Reproduction 2006;74(2):288‐94. [DOI] [PubMed] [Google Scholar]


