Abstract
Background
Chimeric antigen receptor (CAR)-T cell therapy has been used to treat pediatric refractory or relapsed mature B-cell non-Hodgkin lymphoma (r/r MB-NHL) with significantly improved outcomes, but a proportion of patients display no response or experience relapse after treatment. To investigate whether tumor-intrinsic somatic genetic alterations have an impact on CAR-T cell treatment, the genetic features and treatment outcomes of 89 children with MB-NHL were analyzed.
Methods
89 pediatric patients treated at multiple clinical centers of the China Net Childhood Lymphoma (CNCL) were included in this study. Targeted next-generation sequencing for a panel of lymphoma-related genes was performed on tumor samples. Survival rates and relapse by genetic features and clinical factors were analyzed. Survival curves were calculated using a log-rank (Mantel-Cox) test. The Wilcox sum-rank test and Fisher’s exact test were applied to test for group differences.
Results
A total of 89 driver genes with somatic mutations were identified. The most frequently mutated genes were TP53 (66%), ID3 (55%), and ARID1A (31%). The incidence of ARID1A mutation and co-mutation of TP53 and ARID1A was high in patients with r/r MB-NHL (P = 0.006; P = 0.018, respectively). CAR-T cell treatment significantly improved survival in r/r MB-NHL patients (P = 0.00081), but patients with ARID1A or ARID1A and TP53 co-mutation had poor survival compared to those without such mutations.
Conclusion
These results indicate that children with MB-NHL harboring ARID1A or TP53 and ARID1A co-mutation are insensitive to initial conventional chemotherapy and subsequent CAR-T cell treatment. Examination of ARID1A and TP53 mutation status at baseline might have prognostic value, and risk-adapted or more effective therapies should be considered for patients with these high-risk genetic alterations.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12935-023-03122-2.
Keywords: Pediatric B-cell non-hodgkin Lymphomas, CAR-T cell therapies, Gene mutations
Introduction
Mature B-cell non-Hodgkin lymphoma (MB-NHL) accounts for 50–60% of non-Hodgkin lymphomas (NHLs) in children and adolescents [1–3] and is highly aggressive, with unique epidemiology and pathological features [1]. Despite its high intensity, short-course multichemotherapy has significantly improved the cure rate of patients [4, 5]; nevertheless, some children experience induction failure, refractoriness or recurrence (r/r), and the prognosis of r/r MB-NHL is generally poor, with a cure rate of less than 30% [6–8].
Chimeric antigen receptor (CAR)-T cell therapy is an emerging therapy that has been used to treat patients with r/r lymphoma, with significant improvement in clinical outcomes and an overall response rate of 52–82% [9–11]. In our previous study, the 18-month progression-free survival (EFS) rate with sequential CAR-T cell therapy was estimated to be 78% for r/r BL [12]. Although high remission rates after CAR-T cell therapy can be achieved in patients with r/r MB-NHL, some patients still experience relapse [11, 13–15]. In recent years, much attention has been given to clinical research on factors related to CAR-T cell therapy failure. Some studies have shown that intrinsic genetic changes may affect the treatment and prognosis of patients with lymphoma [16, 17]. However, to date, the effect of the preexisting genetic landscape of MB-NHL on CAR-T cell therapy has remained unclear, and despite recent studies in DLBCL [18], few studies have been based on children.
Therefore, we selected a cohort of 89 Chinese pediatric MB-NHL patients to analyze the relationship between mutation status and genetic characteristics and CAR-T cell therapy. We sought to understand the intrinsic molecular characteristics of r/r MB-NHL patients who are not sensitive to CAR-T cell therapy in an attempt to identify molecular predictors associated with this treatment. Identifying patients at high risk of relapse who may not benefit from immunochemotherapy and/or CAR-T cell therapy can provide a clinical basis for improving clinical management and treatment strategies for these lymphoma patients.
Patients and methods
Patients
A total of 89 patients treated at multiple clinical centers of the China Net Childhood Lymphoma (CNCL) from February 2019 to September 2021 were included in this retrospective study. Targeted next-generation sequencing (t-NGS) with a panel of lymphoma-related genes was performed on tumor samples (diagnostic [19] and staging criteria [20] are described in supplementary materials 1.1). All patients received at least first-line chemotherapy and were evaluated and followed up after treatment [21]. There were 41 patients with initial remission and 48 patients with r/r. Among the r/r patients 40 (details of the inclusion and exclusion (I/E) criteria are shown in supplementary materials 1.2) received CAR-T cell infusion between February 2019 and September 2021 and were evaluated for responses and adverse effects (see Supplementary material 1.3, 1.4 and 1.5). The cohort included 9 patients who received CD19 CAR-T cell therapy (ClinicalTrials #: ChiCTR-1,800,014,457), 1 patient who received CD20 CAR-T cell therapy (ClinicalTrials #: ChiCTR-1,800,014,457), 22 patients who received sequential CD19-22 CAR-T cell therapy (ClinicalTrials #: ChiCTR-1,800,014,457), and 8 patients who received CD19&22 or CD20&22 CAR-T cell therapy (ClinicalTrials #: ChiCTR-2,100,045,864). The lentiviral vector was prepared by Shanghai Yake Biotechnology Co., Ltd. This study was approved by the institutional review board of Beijing GoBroad Boren Hospital, in accordance with the Declaration of Helsinki (Approval numbers: 20,210,312-KS-001Y and 20,180,114-PJ-001). Patients (or their guardians) were required to provide written consent.
Definitions
Initial remission refers to achieving complete response (CR) after first-line chemotherapy. Relapsed or refractory (r/r) was defined as disease that was refractory (never obtaining a CR) or relapsed after first-line chemotherapy and had a partial response (PR) or no response (NR) as the best response to at least 2 cycles of salvage chemotherapy. Poor prognostic outcome was defined as meeting at least one of the following criteria: deceased, unresponsive to treatment and/or disease relapse.
Targeted next-generation sequencing and mutational analysis
t-NGS was used to detect the mutation status of 262 driver genes related to lymphoma to identify somatic mutations, including single-nucleotide variants (SNVs) and short insertions and deletions (InDels) in the patients. First, DNA was extracted from tumor tissue samples. The genomic DNA was fragmented to approximately 200 bp by enzyme digestion, and then end repair, adaptor ligation and PCR (polymerase chain reaction) were carried out to complete prelibrary construction. A complete set of probes provided by Agilent was used to capture the sequences of 262 genes (all coding exons), and the target fragments were enriched to generate the library. A NextSeq550 sequencer (Illumina) was used for 2*150 bp sequencing analysis. The human genome hg19 was used as a reference for sequence alignment and base identification. Mutation information was annotated by the information screening function based on public databases (dbSNP, 1000 Genomes, and ESP6500). The variant allele frequency (VAF) was calculated by the number of mutants reads over the number of total reads (Supplementary 1.6).
Statistical methods
Statistical analyses were carried out with SPSS statistics v.25 and R software v3.6.2. Actuarial survival analysis was estimated by the Kaplan–Meier method, and survival curves were generated using a log-rank (Mantel‒Cox) test. The Wilcox sum-rank test and Fisher’s exact test were applied to test for group differences. p values of 0.05 or below were considered statistically significant.
Overall survival (OS) was calculated from the date of the last CAR-T cell infusion to the date of last follow-up or death, and disease-free survival (DFS) was defined only for patients who achieved CR and was calculated from the date of CR after the last CAR-T cell treatment to the date of relapse or death regardless of cause. Follow-up visits for MB-NHL cases are scheduled every 3 to 6 months for the first 2 years and once every 6 months after 5 years. All patients were followed up until 30 September 2021.
Results
Clinical characteristics
The clinical data of the 89 patients are summarized in Table 1. The median age was 8 years old (range 0–18 years old). Of the patients, 63 had BL, accounting for most cases (73.8%), 11 (12.4%) had HGBL, 10 (11.2%) had DLBCL, and 5 had other types. This cohort predominantly consisted of males (73, 82.0%), approximately 5 times the number of females (16, 18.0%), and the sex composition was consistent with that of another study [22]. The major clinicopathological stages of the patients were stage III (43, 48.3%) and stage IV (42, 47.2%). Twenty-eight patients who achieved complete recovery or partial remission (CR) and achieved remission lasting more than 6 months after initial treatment were defined as the initial remission group (IR). Thirteen patients had CR after initial treatment, but remission lasted less than 6 months; these patients are still under follow-up. Forty-eight cases were r/r MB-NHL (r/r MB-NHL) (the groups are shown in Supplementary Table S1).
Table 1.
Clinical data of pediatric patients with MB-NHL (n = 89)
| Level | Overall n = 89 |
|
|---|---|---|
| Age (mean (SD)) | 8.94 (4.39) | |
| Gender (%) | Female | 16 (18.0) |
| Male | 73 (82.0) | |
| Disease subtype (%) | BL | 63 (70.8) |
| HGBL | 11 (12.4) | |
| DLBCL | 10 (11.2) | |
| Others | 5 (5.6) | |
| Stage (%) | II | 2 (2.2) |
| III | 43 (48.3) | |
| IV | 42 (47.2) | |
| Unknown | 2 (2.2) | |
| CNS (%) | CNS1 | 52 (58.4) |
| CNS2 | 15 (16.9) | |
| CNS3 | 20 (22.5) | |
| Unknown | 2 (2.2) | |
| Group (%) | r/r | 48 (53.9) |
| initial remission (≥ 6mounths) | 28 (31.5) | |
| initial remission (<6mounths) | 13 (14.6) | |
| CAR-T or not | No | 49 (55.1) |
| Yes | 40 (44.9) | |
| CAR-T Response (n = 40) | NR | 15 (37.5) |
| R | 25 (62.5) |
BL, Burkitt Lymphoma; HGBL, High-grade B cell Lymphoma; DLBCL, Diffuse Large B Cell Lymphoma; CNS,Central Nervous System; NR, no response; R, response
The median follow-up was 11.4 months (95% CI: 9.9–13.5 months). The one-year OS of r/r MB-NHL was 43.5% (95% CI, 29.4–64.3).
The mutation landscape of MB-NHL in children
The mutational profiles of 89 MB-NHL patients are presented in Supplemental Figure S1. We detected 324 somatic driver mutations involving 89 genes that have been reported in lymphomas with an impact on treatment decisions, diagnosis, or prognosis. On average, each patient carried 5 mutations, ranging between 1 and 13. Among patients with each mutation, 73 harbored at least three variants (82.0%), and 4 of them carried ≥ 10 mutations; one patient carried 13 mutations, the highest number detected.
The most commonly mutated genes were TP53 (in 59 of 89 patients; 66%), followed by ID3 (in 49 patients; 55%), ARID1A (in 28 patients; 31%), CCND3 (in 26 patients; 29%), DDX3X (in 23 patients; 26%), and GNA13 (in 14 patients; 16%) (Supplementary Fig. S1). The mutated genes are mainly related to the TP53 signaling pathway, chromatin remodeling, the cell cycle, epigenetics and the NF-κβ signaling pathway.
Differences between IR MB-NHL and r/r MB-NHL patients
By comparing the clinical characteristics of the patients with IR MB-NHL (n = 28) and those with r/r MB-NHL (n = 48), it was found that the age of the latter was significantly higher than that of the former (IR MB-NHL: median = 6.5 years, IQR: 4.25-10 years; r/r MB-NHL: median = 9 years, IQR: 7-13.75, P = 0.00043) (Fig. 1b and Supplementary Table S2). There are few studies on the correlation between age and prognosis in pediatric lymphoma, but our results are supported by evidence showing that pediatric NHL patients have a significantly better prognosis than adult patients with the same histological subtype [23]. There were no significant differences in clinicopathological stage, sex or CNS between the two groups (not shown).
Fig. 1.
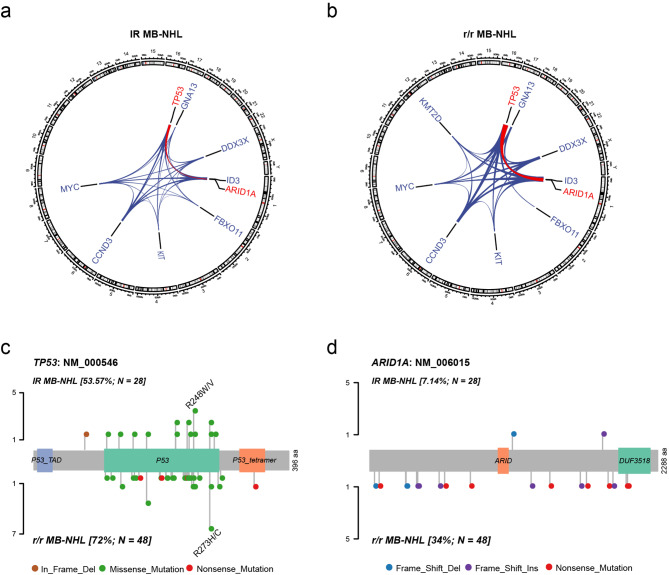
Comparison between r/r MB-NHL and initial remission patients. (a). The mutational spectrum of r/r MB-NHL (n = 48; left) and initial remission patients (n = 28; right). Overview of the top 30 mutated genes with different forms of mutation and their frequencies. Side bar plots indicate the incidence of mutations in a gene in 76 patients. Top bar plots indicate the number of mutated genes per participant. (b). The box plot indicates a comparison of ages between r/r MB-NHL (left, red) and initial remission patients (right, blue). (c). Comparison of mutation profiles between r/r MB-NHL (left, red) and initial remission patients (right, blue). (d). Comparison of the incidence of ARID1A mutations in r/r MB-NHL (left) and initial remission patients (right). Wt, wild-type ARID1A; Mut, ARID1A mutation
In 28 IR MB-NHL patients, the most commonly mutated genes were TP53 (54%, 15/28), ID3 (50%, 14/28), CCND3 (36%, 10/28), DDX3X (25%, 7/28), MYC (25%, 7/28), and GNA13 (18%, 5/28). In 48 r/r patients, the most commonly mutated gene was TP53 (75%, 36/48), followed by ID3 (56%, 27/48), ARID1A (35%, 17/48), CCND3 (25%, 12/48), DDX3X (25%, 12/48), and GNA13 (17%, 8/48). Compared with MB-NHL, TP53 (75% vs. 54%) and ARID1A (35% vs. 7%) showed a higher incidence of mutation than IR MB-NHL (Fig. 1a and c), with the incidence of ARID1A mutation reaching a significant difference (P = 0.006, Fig. 1d). The results suggest that ARID1A mutations are enriched in resistant disease.
Focusing on the mutation patterns of TP53 and ARID1A, point mutations in the DNA-binding domain (DBD) were the main type for TP53. The TP53 R248 W/V/Q (4 vs. 2) mutation was more common in IR MB-NHL, but the TP53 R273H/C (8 vs. 3) mutation was more common in r/r MB-NHL patients. Patients with r/r MB-NHL showed a higher prevalence of frameshift and truncating mutations than IR MB-NHL, and these mutation patterns were the major pathogenic mutations found for ARID1A, leading to a loss of its function (Fig. 2c and d).
Fig. 2.
Gene mutations of TP53 and ARID1A in r/r MB-NHL and initial remission patients. (a) Co-mutations between genes in cases with IR Mb-NHL (left) and (b) r/r MB-NHL (right). The red lines indicate co-mutations of TP53 and ARID1A. (c) The location of amino acid changes resulting from TP53 and (d) ARID1A mutations. The upper part indicates IR MB-NHL and the lower part indicates r/r MB-NHL patients. The length of the line indicates the number of mutations. R248 W/V (c) is a hot spot mutation in TP53 in initial remission, and R273H/C (c) is a hot spot mutation in TP53 in r/r MB-NHL
Fig. 3.
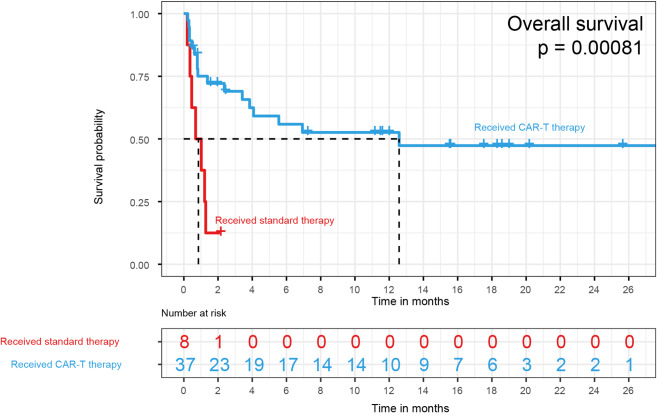
Kaplan‒Meier method comparing overall survival (OS) in r/r MB-NHL between patients who received CAR-T cell therapy (blue; n = 37; mOS = 12.6 months, 95% CI: 3.85-NA months) and those treated without CAR-T cell therapy (red; n = 8). Patients with no survival data were excluded
Further analysis of the VAF showed a significantly higher percentage of VAF for ARID1A and TP53 mutations in patients with r/r MB-NHLs than in patients with IR MB-NHL (TP53 VAF: IR MB-NHL: median = 20.33%, IQR: 0-47.4%, r/r MB-NHL: median = 53.1%, IQR: 0-83.8%, P = 0.0075; ARID1A VAF: IR MB-NHL: median = 0%, IQR: 0–0%, r/r MB-NHL: median = 0%, IQR: 0-43.4%, P = 0.0025).
Effects of mutations on the outcome of CAR-T cell therapy
Among the r/r MB-NHL patients, 40 were treated with CAR-T cell therapy. Twenty-two patients (55%) achieved CR and 12 patients (30%) achieved PR, with an Overall response rate (ORR) of 85% (34/40), according to the outcome assessment at the end of each CAR-T cell treatment.
The difference in r/r MB-NHL survival probability between patients treated with and without CAR-T cells was compared (patients with no survival data were excluded). It is worth noting that the median survival in patients treated with CAR-T cell therapy was more than one year (mOS = 12.6 months, 95% CI: 3.85-NA months), with a one-year survival rate of 52.6% and a two-year survival rate of more than 47.3% (Fig. 3). In comparison, patients who did not receive CAR-T cell treatment had poor prognosis, with a median survival of less than one month (P = 0.00081). The results showed that CAR-T cell treatment significantly improved the outcome of r/r MB-NHL.
These patients were divided based on the evaluation of the last CAR-T cell treatment into those who responded (CAR-T response patients (CR or PR), 25) and those who did not respond (CAR-T nonresponse patients (NR or PD), 15) to CAR-T cell therapy.
To further explore molecular markers associated with CAR-T cell treatment, we compared differences in gene mutation profiles between the patients who did and did not respond to CAR-T cell treatment. The results showed that the incidence of ARID1A mutation (65% vs. 24%) and the VAF of the gene mutation were significantly higher in patients who did not respond to CAR-T cell therapy than in patients who did respond (P = 0.042, Fig. 4d; P = 0.027, Fig. 4e, respectively).
Fig. 4.
Association of TP53 and ARID1A mutations with the therapeutic effect of CAR-T cell therapy. Comparison of the incidence of (a) TP53 mutations and (d) ARID1A mutations in the CAR-T cell response group (left) and CAR-T cell no response group (right). Wt, wild-type ARID1A; Mut, ARID1A mutation. Comparison of variant allele frequencies (VAFs) among (b) TP53 and (e) ARID1A mutations in the CAR-T cell response group (left, green box) and CAR-T cell no response group (right, orange box). (c) Patients with mutations (MUT) in TP53 (red line) showed worse survival than patients with wild-type (Wt) TP53 (blue line). (f). The survival probability was significantly lower in patients with ARID1A mutations (Mut, red line) than in patients with wild-type ARID1A (Wt, blue line) (p = 0.01)
Regarding the outcomes of patients treated with CAR-T cell therapy, survival analysis showed that compared with ARID1A wild-type patients, patients with ARID1A mutations had a poor response to CAR-T therapy and worse DFS (log-rank P = 0.0078, Figure S2b) and OS, with a median survival of 3.85 months (95% CI: 0.79-NA month; ARID1A-wt, mOS = not reached, log-rank P = 0.0095, Fig. 4c). These results show that ARID1A status may have an impact on CAR-T therapy.
It was also found that patients who did not respond to CAR-T cell therapy tended to have a higher incidence of TP53 mutations (93% vs. 54%) than those who did respond (P = 0.059, Fig. 4a) as well as a significantly higher VAF of TP53 (P = 0.0013, Fig. 4b). Survival analysis also showed that patients with TP53 mutations had a shorter median survival of 4.08 months (95% CI: 2.37-NA month) than those without TP53 mutations (TP53-wt, mOS = not reached, log-rank P = 0.11, Fig. 4f), which may indicate a trend of poor outcomes, but there was no significant difference in median OS (log-rank P = 0.11, Fig. 4f)) or DFS (log-rank P = 0.92, Figure S2a) between patients with TP53 mutations and patients without TP53 mutations.
Efficacy of TP53 and ARID1A co-mutation in CAR-T cell treatment
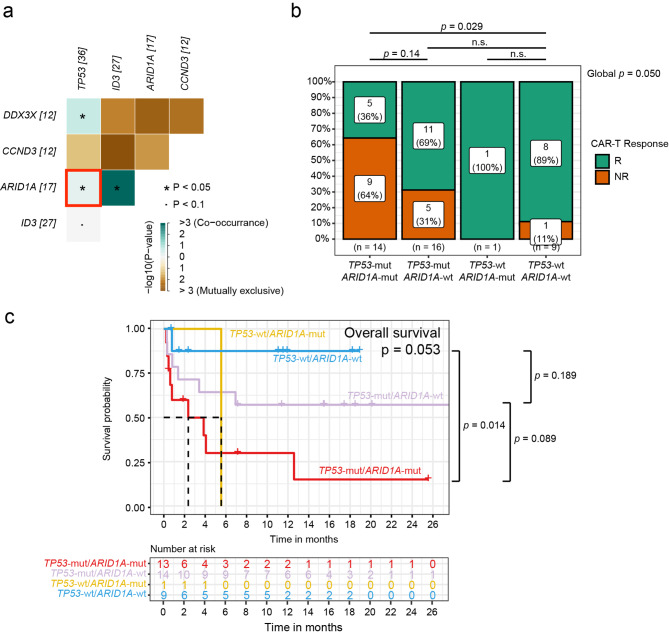
The above results show that the incidence of co-mutation of TP53 and ARID1A in patients with r/r MB-NHL was significantly higher than that in patients with initial remission (TP53-mut&ARID1A-mut: n = 16, TP53-mut&ARID1A-wt: n = 20, TP53-wt&ARID1A-mut: n = 1, TP53-wt&ARID1A-wt: n = 11, P = 0.018, Fig. 5a). Therefore, we sought to further understand whether TP53 and ARID1A co-mutations have an impact on CAR-T cell therapy. We found that patients (n = 14) with co-mutations of TP53 and ARID1A had a lower response rate to CAR-T cell treatment than the other groups. Only 5 of 14 patients showed a response,the overall response rate was only 36% (Fig. 5b).
Fig. 5.
Co-mutations in TP53 and ARID1A are associated with inferior clinical outcomes after CAR-T cell therapy. (a) Analysis of co-mutations in r/r MB-NHL. (b) Comparison of response rates of CAR-T cell therapy according to TP53 and ARID1A mutation status. (c) Survival analyses by Kaplan–Meier according to mutation status in patients treated with CAR-T cell therapy
Kaplan-Meier survival curves of the effects of ARID1A and TP53 status on OS and DFS showed significantly shorter OS and DFS in patients harboring ARID1A and TP53 co-mutations. (ARID1A-mut/TP53-mut: mOS = 2.37 months, 95% CI: 0.625 - NA months; TP53-wt/ARID1A-wt: mOS = not reached, log-rank p = 0.014, Fig. 5c) (ARID1A-mut/TP53-mut: mDFS = 4.06 months, 95% CI: 0.53-NA months; TP53-wt/ARID1A-wt: mDFS = not reached, log-rank p = 0.0039, Figure. S2c).
Therefore, although CAR-T cell treatment significantly improved survival in r/r MB-NHL patients, patients with ARID1A and TP53 co-mutation had poor outcome after treatment when compared to those without such mutations.
Discussion
The outcome of r/r MB-NHL in children is very poor [6, 8, 24, 25]. Although most patients improve after treatment with CAR-T cells, some patients still experience relapse and rapid progression [26]. We explored a critical clinical question: are there molecular markers that predict the outcome of CAR-T cell therapy in r/r MB-NHL pediatric patients? We analyzed the molecular variation characteristics of MB-NHL in 89 children. To the best of our knowledge, this is the first study to find that ARID1A mutations are associated with poor outcome after CAR-T cell therapy in children with r/r MB-NHL. At the same time, a significant finding was that co-mutation of ARID1A and TP53 in Chinese children with r/r MB-NHL is associated with insensitivity to chemotherapy and CAR-T cell treatment.
ARID1A is one of the most commonly mutated genes in cancers [27–29]. Expressing a subunit of the SWI/SNF chromatin remodeler, ARID1A impacts transcription initiation and elongation [30, 31], participates in control of the PI3K/AKT/mTOR pathway and is associated with EZH2 methyltransferase activity, steroid receptor modulation and regulation of p53 targets [32–34]. Studies have shown that ARID1A has a crucial role in regulating gene expression that drives oncogenesis or tumor suppression and that deletion of ARID1A promotes tumor progression. In this study, the incidence of ARID1A was higher in r/r MB-NHL patients than in IR patients (35% vs. 7%), indicating that ARID1A mutations are enriched in resistant disease and that these patients might be insensitive to initial chemotherapy. A study in ovarian cancer showed that ARID1A alterations may also mediate resistance to platinum chemotherapy and estrogen receptor degraders/modulators [35], which further supports our findings that ARID1A deficiency may render patients insensitive to initial chemotherapy and be associated with relapse. Moreover, consistent with other studies, most of the ARID1A mutations we detected are classified as loss-of-function mutations, which were dispersed throughout the coding sequence [36]. Most of them are truncating or frameshift mutations. These types may lead to disruption of protein functional domains or mediate mRNA degradation, thus disrupting ARID1A gene function [37, 38] and leading to tumor cell progression and patient recurrence.
CAR-T cell therapy is a good option for pediatric patients with r/r MB-NHL [9]; however, patients with ARID1A mutations have lower rates of response and survival after CAR-T cell therapy. CAR-T cell exhaustion is partly attributed to prolonged exposure to the immunosuppressive microenvironment, upregulation of inhibitory receptors, and persistent CAR stimulation by antigen [39, 40]. There is evidence that ARID1A can modulate the tumor immune microenvironment, which underlies its correlation with sensitivity to immunotherapy [40–42]. Julia A. Belk et al. demonstrated through gene editing that ARID1A improves T-cell persistence and that anti-ARID1A deficiency promotes mutability and potentiates therapeutic antitumor immunity, unleashing tumor immunity in vivo [43]; however, deletion of ARID1A gene function and decreased T-cell persistence may be barriers to immune checkpoint blockade and the effectiveness of CAR-T cell immunotherapy [44]. In addition, ARID1A mutation is associated with increased expression of PD-L1, which reduces the level of antitumor immune response and may promote immune escape of tumor cells [45, 46]. ARID1A is also reportedly closely related to DNA mismatch repair and microsatellite instability [41], which is a possible factor for poor immunotherapy effects caused by ARID1A mutation. These conclusions support our view that ARID1A mutations are associated with poorer clinical benefit in patients treated with CAR-T cells. Furthermore, ARID1A alterations may be a risk factor for insensitivity to chemotherapy or CAR-T cell therapy, and patients should be monitored and regularly followed up.
The negative prognostic impact of TP53 mutation and its association with drug resistance is well known in many malignancies [47], and some studies have shown that TP53 aberrations are valuable prognostic markers in CD19-CAR-T cell recipients [18]. Nonetheless, there have been few studies on children with B-NHL. In our study, children with TP53 mutations showed a trend of adverse outcomes after CAR-T cell therapy, but without reaching a significant level; hence, further research is needed. Multiple studies in solid tumors have demonstrated an inverse relationship between TP53 and ARID1A mutations and even found ARID1A and TP53 mutual exclusivity in ovarian clear cell and uterine endometrioid carcinomas [34, 48]. Remarkably, we found significant co-mutation of ARID1A and TP53 in r/r MB-NHL that was associated with CAR-T cell insensitivity. ARID1A has been shown to interact with ligand-bound nuclear hormone receptors and TP53 through its C-terminal domain and to stimulate the transcriptional activity of these transcription factors [34, 49]. A new study using genetic engineering has shown that ARID1A loss probably affects multiple aspects of TP53-regulated chromatin and promotes squamous differentiation and acquisition of invasive properties [50]. These may be the causes of the poor prognosis in patients with co-mutation of ARID1A and TP53, and their molecular mechanisms need to be further studied. In recent years, it has been shown that a high VAF load is associated with poor outcome in patients with hematologic tumors [51] and we also report similar findings.
We have identified molecular abnormalities that may affect the outcome of CAR-T cell therapy in r/r MB-NHL patients, and there is an urgent need to find new solutions. In recent years, with the development of drugs and clinical research, target drugs related to TP53 or ARID1A have been studied in the clinic and laboratory, including combined epigenetic inhibitors to form combined lethal targeted therapies and immune checkpoint inhibitor treatment [52, 53]. CAR-T cell therapy combined with immunosuppressants or cytokine inhibitors has been shown to enhance antitumor efficacy in the treatment of hematologic tumors [54]. In addition, multitarget CAR-T cell combination therapy or radiotherapy combined with CAR-T cell therapy may be more effective than CAR-T cell therapy alone. Our study provides a new clinical basis for further work. In the future, we will continue to pay attention to the follow-up treatment and outcome of lymphoma patients.
This study has several limitations. First, sequencing of tumor samples was driven by clinical decision-making, potentially leading to selection bias. However, we note that population features and outcomes were similar between patients who underwent sequencing and those who did not. Finally, to our knowledge, although this is the first analysis evaluating the role of ARID1A in children with r/r MB-NHL treated with CAR-T cells, the sample size was limited. Validation using larger cohorts and prospective trials are warranted to guide CAR-T cell product selection.
Conclusion
In summary, our study analyzed the genetic features and treatment outcomes of 89 children with MB-NHL, and ARID1A mutations are common in pediatric MB-NHL. We demonstrated that children with MB-NHL harboring ARID1A or TP53 and ARID1A co-mutation were insensitive to initial conventional chemotherapy and subsequent CAR-T treatment and were associated with disease progression or relapse. Our data suggest that ARID1A and TP53 mutation status should be considered when prognostic factors are evaluated before CAR-T treatment, and this finding would help clinicians customize tailored treatments for their patients.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
The authors thank all of the patients who participated in the study and their families, friends, and caregivers for their support, and the members of clinical centers of the China Net Childhood Lymphoma (CNCL) for their support in carrying out this work. The authors would also like to thank the members of the Molecular Diagnostics Laboratory of Beijing GoBroad Boren Hospital for the targeted deep sequencing and data analysis of the samples.
List of abbreviations
- MB-NHL
Mature B-cell non-Hodgkin lymphoma
- IR
Initial remission
- r/r
Refractory or relapsed
- CNCL
China Net Childhood Lymphoma
- CAR-T cell
Chimeric antigen receptor T cell
- EFS
Progression-free survival
- OS
Overall survival
- DFS
Disease-free survival
- CR
Complete response
- NR
Partial response
- PR
No response
- PD
Progressive disease
- SNVs
Single-nucleotide variants
- InDels
Short insertions and deletions
- t-NGS
Targeted next-generation sequencing
- PCR
Polymerase chain reaction
- VAF
Variant allele frequency
- BL
Burkitt Lymphoma
- HGBL
High-grade B cell Lymphoma
- DLBCL
Diffuse Large B Cell Lymphoma
- CNS
Central nervous system
- ORR
Overall response rate (ORR)
Authors’ contributions
QZ, YZ contributed to the conception, designed the study and revised the first draft; Y Li, Y Liu wrote the first draft of the manuscript, and contributed to data acquisition, analysis and interpretation; KY contributed to data analysis and interpretation; LJ, JY, SH, YL, BH, RL, WL and AL contributed to data acquisition. All authors have read and approved the final manuscript.
Funding
Not applicable.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
This study was approved by the institutional review board of Beijing GoBroad Boren Hospital, in accordance with the Declaration of Helsinki (Approval number: 20210312-KS-001Y and 20180114-PJ-001).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yang Li and Yang Liu contributed equally to this work.
Contributor Information
Qinlong Zheng, Email: zhengql@gobroadhealthcare.com.
Yonghong Zhang, Email: zhangyongh@gobroadhealthcare.com.
References
- 1.Egan G, Goldman S, Alexander S. Mature B-NHL in children, adolescents and young adults: current therapeutic approach and emerging treatment strategies. Br J Haematol. 2019;185(6):1071–85. doi: 10.1111/bjh.15734. [DOI] [PubMed] [Google Scholar]
- 2.Reiter A, Schrappe M, Parwaresch R, Henze G, Müller-Weihrich S, Sauter S, Sykora KW, Ludwig WD, Gadner H, Riehm H. Non-hodgkin’s Lymphomas of childhood and adolescence: results of a treatment stratified for biologic subtypes and stage–a report of the Berlin-Frankfurt-Münster Group. J Clin Oncology: Official J Am Soc Clin Oncol. 1995;13(2):359–72. doi: 10.1200/JCO.1995.13.2.359. [DOI] [PubMed] [Google Scholar]
- 3.Sandlund JT. Non-hodgkin Lymphoma in Children. Curr Hematol Malig Rep. 2015;10(3):237–43. doi: 10.1007/s11899-015-0277-y. [DOI] [PubMed] [Google Scholar]
- 4.Minard-Colin V, Brugières L, Reiter A, Cairo MS, Gross TG, Woessmann W, Burkhardt B, Sandlund JT, Williams D, Pillon M, et al. Non-hodgkin Lymphoma in Children and adolescents: Progress through effective collaboration, current knowledge, and challenges ahead. J Clin Oncology: Official J Am Soc Clin Oncol. 2015;33(27):2963–74. doi: 10.1200/JCO.2014.59.5827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.El-Mallawany NK, Cairo MS. Advances in the diagnosis and treatment of childhood and adolescent B-cell non-hodgkin Lymphoma. Clin Adv Hematol Oncology: H&O. 2015;13(2):113–23. [PubMed] [Google Scholar]
- 6.Jourdain A, Auperin A, Minard-Colin V, Aladjidi N, Zsiros J, Coze C, Gandemer V, Bertrand Y, Leverger G, Bergeron C, et al. Outcome of and prognostic factors for relapse in children and adolescents with mature B-cell Lymphoma and Leukemia treated in three consecutive prospective Lymphomes Malins B protocols. A Société Française Des Cancers De l’Enfant study. Haematologica. 2015;100(6):810–7. doi: 10.3324/haematol.2014.121434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rigaud C, Auperin A, Jourdain A, Haouy S, Couec ML, Aladjidi N. Outcome of relapse in children and adolescents with B-cell non-hodgkin Lymphoma and mature acute Leukemia: a report from the French LMB study. Pediatr Blood Cancer. 2019;66(9):e27873. doi: 10.1002/pbc.27873. [DOI] [PubMed] [Google Scholar]
- 8.Woessmann W, Zimmermann M, Meinhardt A, Müller S, Hauch H, Knörr F, Oschlies I, Klapper W, Niggli F, Kabickova E, et al. Progressive or relapsed Burkitt Lymphoma or Leukemia in children and adolescents after BFM-type first-line therapy. Blood. 2020;135(14):1124–32. doi: 10.1182/blood.2019003591. [DOI] [PubMed] [Google Scholar]
- 9.Foà R, Moleti ML, Testi AM. Treatment of relapsed/refractory paediatric aggressive B-cell non-hodgkin Lymphoma. Br J Haematol. 2020;189(5):826–43. doi: 10.1111/bjh.16461. [DOI] [PubMed] [Google Scholar]
- 10.Itzhaki O, Jacoby E, Nissani A, Levi M, Nagler A, Kubi A, Brezinger K, Brayer H, Zeltzer LA, Rozenbaum M et al. Head-to-head comparison of in-house produced CD19 CAR-T cell in ALL and NHL patients. J Immunother Cancer 2020, 8(1). [DOI] [PMC free article] [PubMed]
- 11.Harker-Murray PD, Pommert L, Barth MJ. Novel therapies potentially available for Pediatric B-Cell Non-hodgkin Lymphoma. J Natl Compr Cancer Network: JNCCN. 2020;18(8):1125–34. doi: 10.6004/jnccn.2020.7608. [DOI] [PubMed] [Google Scholar]
- 12.Liu Y, Deng B, Hu B, Zhang W, Zhu Q, Liu Y, Wang S, Zhang P, Yang Y, Yang J, et al. Sequential different B-cell antigen-targeted CAR T-cell therapy for pediatric refractory/relapsed Burkitt Lymphoma. Blood Adv. 2022;6(3):717–30. doi: 10.1182/bloodadvances.2021004557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schuster SJ, Bishop MR, Tam CS, Waller EK, Borchmann P, McGuirk JP, Jäger U, Jaglowski S, Andreadis C, Westin JR, et al. Tisagenlecleucel in adult relapsed or refractory diffuse large B-Cell Lymphoma. N Engl J Med. 2019;380(1):45–56. doi: 10.1056/NEJMoa1804980. [DOI] [PubMed] [Google Scholar]
- 14.Jain T, Bar M, Kansagra AJ, Chong EA, Hashmi SK, Neelapu SS, Byrne M, Jacoby E, Lazaryan A, Jacobson CA, et al. Use of Chimeric Antigen Receptor T Cell Therapy in clinical practice for Relapsed/Refractory aggressive B cell Non-hodgkin Lymphoma: an Expert Panel Opinion from the American Society for Transplantation and Cellular Therapy. Biology of Blood and Marrow Transplantation: Journal of the American Society for Blood and Marrow Transplantation. 2019;25(12):2305–21. doi: 10.1016/j.bbmt.2019.08.015. [DOI] [PubMed] [Google Scholar]
- 15.Wang N, Hu X, Cao W, Li C, Xiao Y, Cao Y, Gu C, Zhang S, Chen L, Cheng J, et al. Efficacy and safety of CAR19/22 T-cell cocktail therapy in patients with refractory/relapsed B-cell malignancies. Blood. 2020;135(1):17–27. doi: 10.1182/blood.2019000017. [DOI] [PubMed] [Google Scholar]
- 16.Sekimizu M, Mori T, Kikuchi A, Mitsui T, Sunami S, Kobayashi R, Fujita N, Inada H, Takimoto T, Saito AM, et al. Prognostic impact of cytogenetic abnormalities in children and adolescents with mature B-cell non-hodgkin Lymphoma: a report from the Japanese Pediatric Leukemia/Lymphoma Study Group (JPLSG) Pediatr Blood Cancer. 2015;62(7):1294–6. doi: 10.1002/pbc.25482. [DOI] [PubMed] [Google Scholar]
- 17.Alexander M, Newman MZ, Peixun Zhou AE, Blain A, Erhorn A, Barnard RE, Crossland S, Wilkinson A, Enshaei JD, Zordi F, Harding M, Taj, Katrina M, Wood D, Televantou, Suzanne D, Turner GA, Amos Burke CJ, Harrison Simon Bomken, Chris M Bacon, Vikki Rand: genomic abnormalities of TP53 define distinct risk groups of paediatric B-cell non-hodgkin Lymphoma. Leukemia. 2022;36(3):781–9. doi: 10.1038/s41375-021-01444-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shouval R, Alarcon Tomas A. Impact of TP53 genomic alterations in large B-Cell Lymphoma treated with CD19-Chimeric Antigen receptor T-Cell therapy. 2022, 40(4):369–81. [DOI] [PMC free article] [PubMed]
- 19.Polyatskin IL, Artemyeva AS, Krivolapov YA. [Revised WHO classification of tumors of hematopoietic and lymphoid tissues, 2017 (4th edition):lymphoid tumors] Arkh Patol. 2019;81(3):59–65. doi: 10.17116/patol20198103159. [DOI] [PubMed] [Google Scholar]
- 20.Rosolen A, Perkins SL, Pinkerton CR, Guillerman RP, Sandlund JT, Patte C, Reiter A, Cairo MS. Revised International Pediatric Non-hodgkin Lymphoma Staging System. J Clin Oncology: Official J Am Soc Clin Oncol. 2015;33(18):2112–8. doi: 10.1200/JCO.2014.59.7203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sandlund JT, Guillerman RP, Perkins SL, Pinkerton CR, Rosolen A, Patte C, Reiter A, Cairo MS. International Pediatric Non-hodgkin Lymphoma Response Criteria. J Clin Oncology: Official J Am Soc Clin Oncol. 2015;33(18):2106–11. doi: 10.1200/JCO.2014.59.0745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Burkhardt B, Zimmermann M, Oschlies I, Niggli F, Mann G, Parwaresch R, Riehm H, Schrappe M, Reiter A. The impact of age and gender on biology, clinical features and treatment outcome of non-hodgkin Lymphoma in childhood and adolescence. Br J Haematol. 2005;131(1):39–49. doi: 10.1111/j.1365-2141.2005.05735.x. [DOI] [PubMed] [Google Scholar]
- 23.Reiter A, Klapper W. Recent advances in the understanding and management of diffuse large B-cell Lymphoma in children. Br J Haematol. 2008;142(3):329–47. doi: 10.1111/j.1365-2141.2008.06988.x. [DOI] [PubMed] [Google Scholar]
- 24.Cairo M, Auperin A, Perkins SL, Pinkerton R, Harrison L, Goldman S, Patte C. Overall survival of children and adolescents with mature B cell non-hodgkin Lymphoma who had refractory or relapsed Disease during or after treatment with FAB/LMB 96: a report from the FAB/LMB 96 study group. Br J Haematol. 2018;182(6):859–69. doi: 10.1111/bjh.15491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rigaud C, Auperin A, Jourdain A, Haouy S, Couec ML, Aladjidi N, Michon J, Leblanc T, Patte C, Gandemer V, et al. Outcome of relapse in children and adolescents with B-cell non-hodgkin Lymphoma and mature acute Leukemia: a report from the French LMB study. Pediatr Blood Cancer. 2019;66(9):e27873. doi: 10.1002/pbc.27873. [DOI] [PubMed] [Google Scholar]
- 26.Kenderian SS, Porter DL, Gill S. Chimeric Antigen Receptor T Cells and hematopoietic cell transplantation: how not to put the CART before the horse. Biology of Blood and Marrow Transplantation: Journal of the American Society for Blood and Marrow Transplantation. 2017;23(2):235–46. doi: 10.1016/j.bbmt.2016.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang K, Kan J, Yuen ST, Shi ST, Chu KM, Law S, Chan TL, Kan Z, Chan AS, Tsui WY, et al. Exome sequencing identifies frequent mutation of ARID1A in molecular subtypes of gastric cancer. Nat Genet. 2011;43(12):1219–23. doi: 10.1038/ng.982. [DOI] [PubMed] [Google Scholar]
- 28.Wu JN, Roberts CW. ARID1A mutations in cancer: another epigenetic Tumor suppressor? Cancer Discov. 2013;3(1):35–43. doi: 10.1158/2159-8290.CD-12-0361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gupta S, Alexander S, Pole JD, Sutradhar R, Crump M, Nagamuthu C, Baxter NN, Nathan PC. Superior outcomes with paediatric protocols in adolescents and young adults with aggressive B-cell non-hodgkin Lymphoma. Br J Haematol. 2022;196(3):743–52. doi: 10.1111/bjh.17862. [DOI] [PubMed] [Google Scholar]
- 30.Mullen J, Kato S, Sicklick JK, Kurzrock R. Targeting ARID1A mutations in cancer. Cancer Treat Rev. 2021;100:102287. doi: 10.1016/j.ctrv.2021.102287. [DOI] [PubMed] [Google Scholar]
- 31.Mathur R. ARID1A loss in cancer: towards a mechanistic understanding. Pharmacol Ther. 2018;190:15–23. doi: 10.1016/j.pharmthera.2018.05.001. [DOI] [PubMed] [Google Scholar]
- 32.Zegeye MM, Lindkvist M, Fälker K, Kumawat AK, Paramel G, Grenegård M, Sirsjö A, Ljungberg LU. Activation of the JAK/STAT3 and PI3K/AKT pathways are crucial for IL-6 trans-signaling-mediated pro-inflammatory response in human vascular endothelial cells. Cell Communication and Signaling: CCS. 2018;16(1):55. doi: 10.1186/s12964-018-0268-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.de la Serna IL, Carlson KA, Imbalzano AN. Mammalian SWI/SNF complexes promote MyoD-mediated muscle differentiation. Nat Genet. 2001;27(2):187–90. doi: 10.1038/84826. [DOI] [PubMed] [Google Scholar]
- 34.Guan B, Wang TL, Shih Ie M. ARID1A, a factor that promotes formation of SWI/SNF-mediated chromatin remodeling, is a Tumor suppressor in gynecologic cancers. Cancer Res. 2011;71(21):6718–27. doi: 10.1158/0008-5472.CAN-11-1562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lyu C, Zhang Y, Zhou X, Lang J. ARID1A gene silencing reduces the sensitivity of ovarian clear cell carcinoma to cisplatin. Experimental and Therapeutic Medicine. 2016;12(6):4067–71. doi: 10.3892/etm.2016.3863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wu RC, Wang TL, Shih Ie M. The emerging roles of ARID1A in Tumor suppression. Cancer Biol Ther. 2014;15(6):655–64. doi: 10.4161/cbt.28411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mamo A, Cavallone L, Tuzmen S, Chabot C, Ferrario C, Hassan S, Edgren H, Kallioniemi O, Aleynikova O, Przybytkowski E, et al. An integrated genomic approach identifies ARID1A as a candidate tumor-suppressor gene in Breast cancer. Oncogene. 2012;31(16):2090–100. doi: 10.1038/onc.2011.386. [DOI] [PubMed] [Google Scholar]
- 38.Guan B, Gao M, Wu CH, Wang TL, Shih Ie M. Functional analysis of in-frame indel ARID1A mutations reveals new regulatory mechanisms of its Tumor suppressor functions. Neoplasia (New York NY) 2012;14(10):986–93. doi: 10.1593/neo.121218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tang L, Zhang Y, Hu Y. T Cell Exhaustion and CAR-T Immunotherapy in Hematological Malignancies. 2021, 2021:6616391. [DOI] [PMC free article] [PubMed]
- 40.Gumber D, Wang LD. Improving CAR-T immunotherapy: overcoming the challenges of T cell exhaustion. EBioMedicine. 2022;77:103941. doi: 10.1016/j.ebiom.2022.103941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hu G, Tu W, Yang L, Peng G, Yang L. ARID1A deficiency and immune checkpoint blockade therapy: from mechanisms to clinical application. Cancer Lett. 2020;473:148–55. doi: 10.1016/j.canlet.2020.01.001. [DOI] [PubMed] [Google Scholar]
- 42.Li N, Liu Q. ARID1A loss induces polymorphonuclear myeloid-derived suppressor cell chemotaxis and promotes Prostate cancer progression. Nat Commun. 2022;13(1):7281. doi: 10.1038/s41467-022-34871-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Belk JA, Yao W, Ly N, Freitas KA, Chen YT, Shi Q, Valencia AM, Shifrut E, Kale N, Yost KE, et al. Genome-wide CRISPR screens of T cell exhaustion identify chromatin remodeling factors that limit T cell persistence. Cancer Cell. 2022;40(7):768–786e767. doi: 10.1016/j.ccell.2022.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Weber EW, Parker KR, Vandris P, Sotillo E, Lynn RC, Qi Y, Sandor K, Malipatlolla M, Xu P, Bashti M et al. Transient rest restores functionality in exhausted CAR-T cells through epigenetic remodeling. Science (New York, NY) 2021, 372(6537). [DOI] [PMC free article] [PubMed]
- 45.Wang L, Qu J, Zhou N, Hou H, Jiang M, Zhang X. Effect and biomarker of immune checkpoint blockade therapy for ARID1A deficiency cancers. Biomed Pharmacotherapy = Biomedecine Pharmacotherapie. 2020;130:110626. doi: 10.1016/j.biopha.2020.110626. [DOI] [PubMed] [Google Scholar]
- 46.Shen J, Ju Z, Zhao W, Wang L, Peng Y, Ge Z, Nagel ZD, Zou J, Wang C, Kapoor P, et al. ARID1A deficiency promotes mutability and potentiates therapeutic antitumor immunity unleashed by immune checkpoint blockade. Nat Med. 2018;24(5):556–62. doi: 10.1038/s41591-018-0012-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Vazquez A, Bond EE, Levine AJ, Bond GL. The genetics of the p53 pathway, apoptosis and cancer therapy. Nat Rev Drug Discovery. 2008;7(12):979–87. doi: 10.1038/nrd2656. [DOI] [PubMed] [Google Scholar]
- 48.Liang H, Cheung LW, Li J, Ju Z, Yu S, Stemke-Hale K, Dogruluk T, Lu Y, Liu X, Gu C, et al. Whole-exome sequencing combined with functional genomics reveals novel candidate driver cancer genes in endometrial cancer. Genome Res. 2012;22(11):2120–9. doi: 10.1101/gr.137596.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Inoue H, Furukawa T, Giannakopoulos S, Zhou S, King DS, Tanese N. Largest subunits of the human SWI/SNF chromatin-remodeling complex promote transcriptional activation by steroid hormone receptors. J Biol Chem. 2002;277(44):41674–85. doi: 10.1074/jbc.M205961200. [DOI] [PubMed] [Google Scholar]
- 50.Jake J, Reske MR, Wilson J, Holladay RA, Siwicki H, Skalski S, Harkins M, Adams JI, Risinger G, Hostetter Co-existing TP53 and ARID1A mutations promote aggressive endometrial tumorigenesis. PLoS Genet. 2021;17(12):e1009986. doi: 10.1371/journal.pgen.1009986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sasaki K, Kanagal-Shamanna R, Montalban-Bravo G, Assi R, Jabbour E, Ravandi F, Kadia T, Pierce S, Takahashi K, Nogueras Gonzalez G, et al. Impact of the variant allele frequency of ASXL1, DNMT3A, JAK2, TET2, TP53, and NPM1 on the outcomes of patients with newly diagnosed acute Myeloid Leukemia. Cancer. 2020;126(4):765–74. doi: 10.1002/cncr.32566. [DOI] [PubMed] [Google Scholar]
- 52.Karube K, Enjuanes A, Dlouhy I, Jares P, Martin-Garcia D, Nadeu F, Ordóñez GR, Rovira J, Clot G, Royo C, et al. Integrating genomic alterations in diffuse large B-cell Lymphoma identifies new relevant pathways and potential therapeutic targets. Leukemia. 2018;32(3):675–84. doi: 10.1038/leu.2017.251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Li Z, Lin J, Zhang L, Li J, Zhang Y, Zhao C, Wang H. Comprehensive analysis of multiple parameters associated with Tumor immune microenvironment in ARID1A mutant cancers. Future Oncol (London England) 2020;16(29):2295–306. doi: 10.2217/fon-2020-0243. [DOI] [PubMed] [Google Scholar]
- 54.Giulino-Roth L, Goldman S. Recent molecular and therapeutic advances in B-cell non-hodgkin Lymphoma in children. Br J Haematol. 2016;173(4):531–44. doi: 10.1111/bjh.13969. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.