Abstract
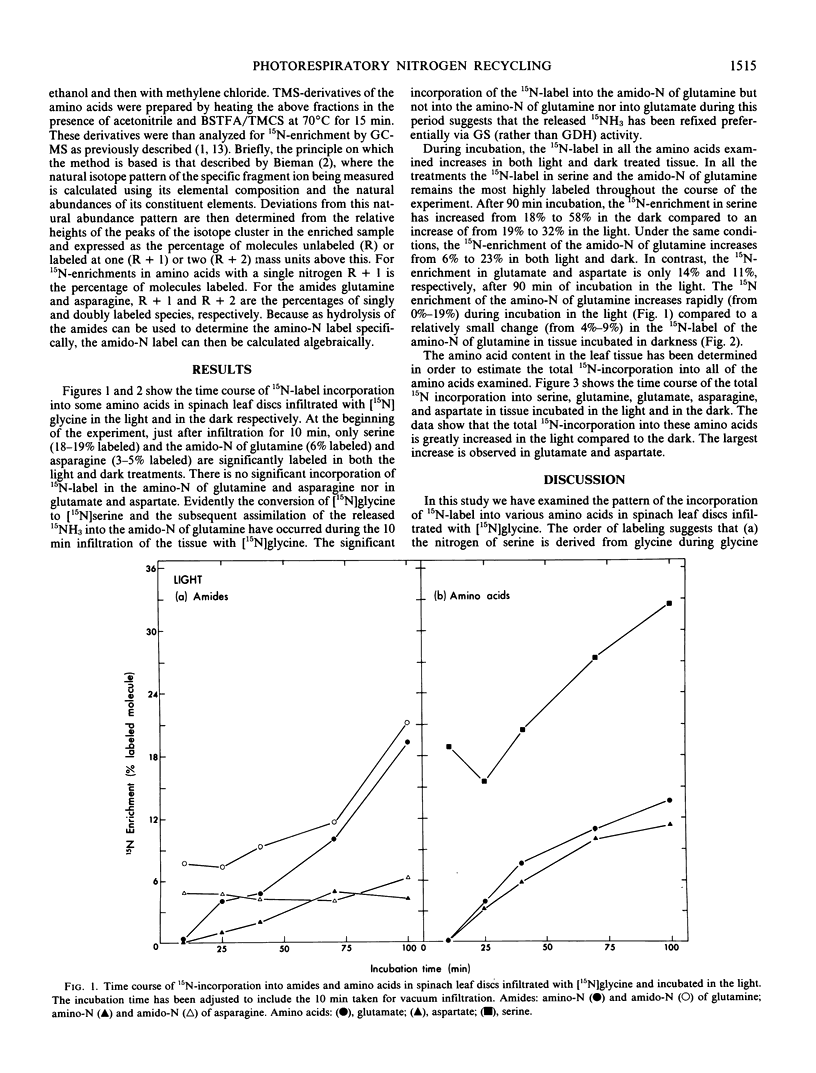
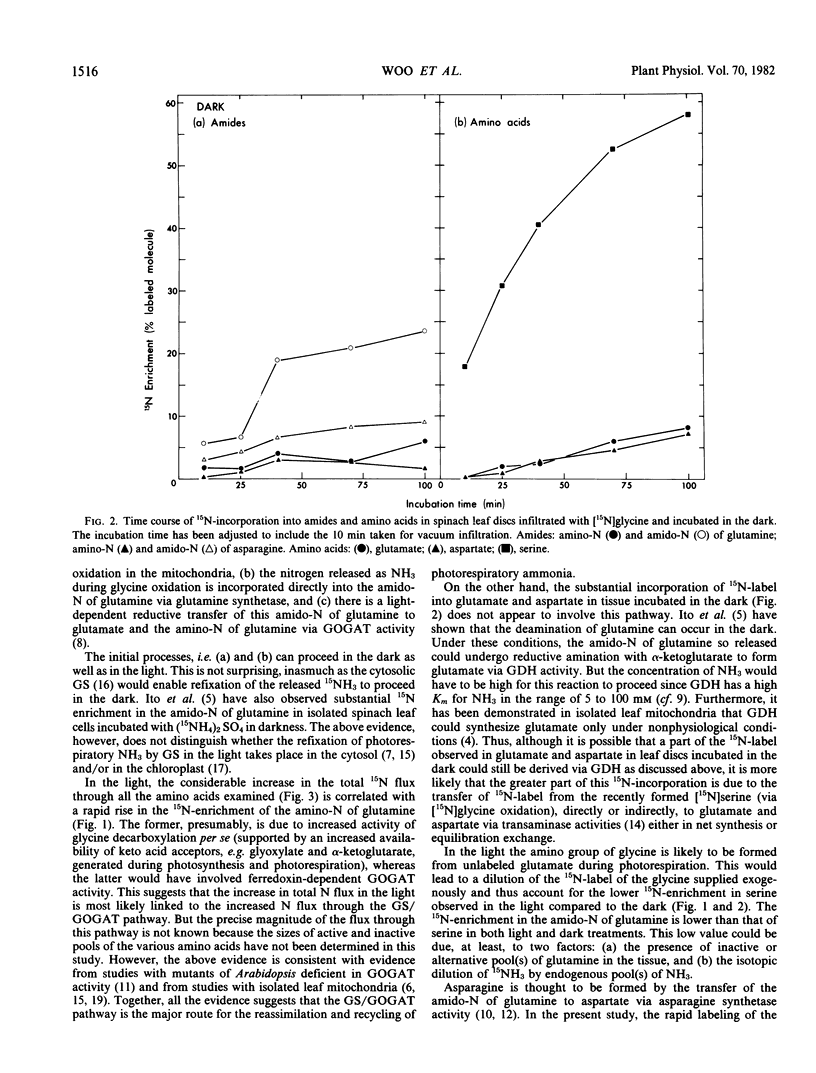
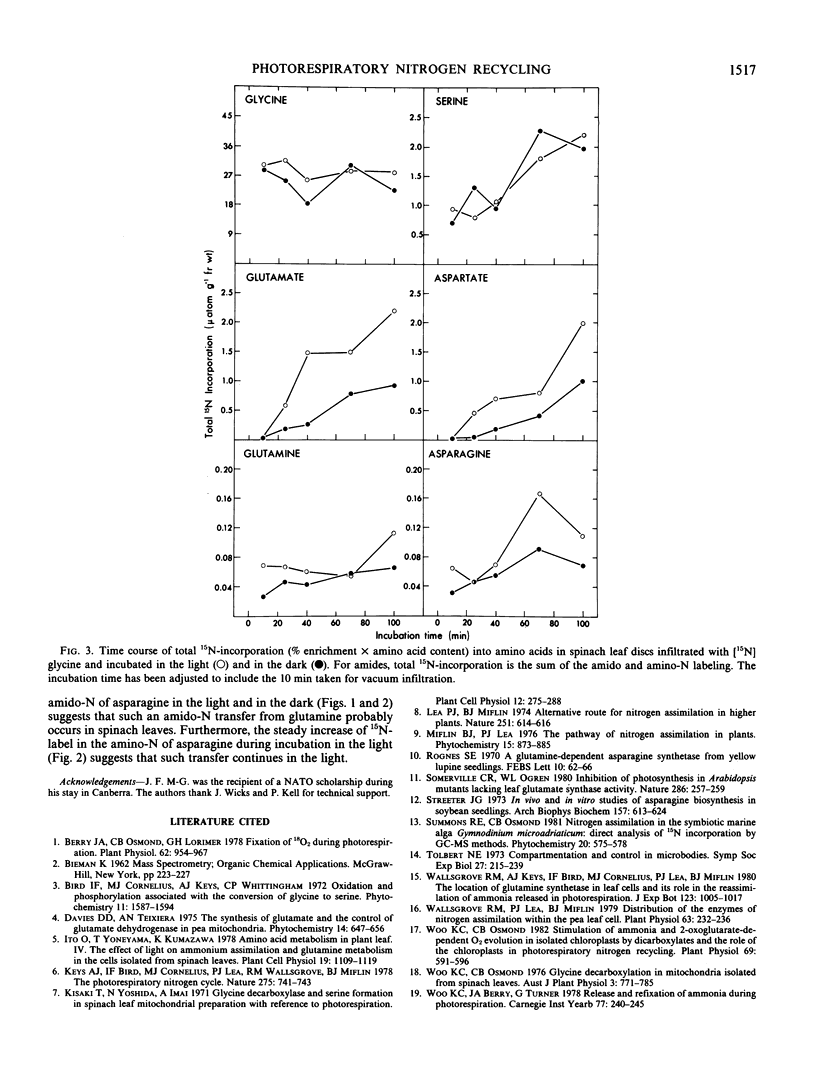
Spinach leaf (Spinacia oleracea L.) discs infiltrated with [15N]glycine were incubated at 25°C in the light and in darkness for 0, 30, 60 and 90 minutes. The kinetics of 15N-incorporation into glutamine, glutamate, asparagine, aspartate, and serine from [15N]glycine was determined. At the beginning of the experiment, just after infiltration (0 min incubation) serine, and the amido-N of glutamine and asparagine were the only compounds significantly labeled in both light- and dark-treated leaf discs. Incorporation of 15N-label into the other amino acids was observed at longer incubation time. The per cent 15N-enrichment in all amino acids was found to increase with incubation. However, serine and the amido-N of glutamine remained the most highly labeled products in all treatments. The above pattern of 15N-labeling suggests that glutamine synthetase was involved in the initial refixation of 15NH3 derived from [15N]glycine oxidation in spinach leaf discs.
The 15N-enrichment of the amino-N of glutamine was found to increase rapidly from 0 to 19% during incubation in the light. There was a comparatively smaller increase (4-9%) in the 15N-label of the amino-N of glutamine in tissue incubated in darkness. Furthermore the total flux of 15N-label into each of the amino acids examined was found to be greater in tissue incubated in the light than those in the dark. The above evidence indicates the involvement of the glutamine synthetase/glutamate synthase pathway in the recycling of photorespiratory NH3 during glycine oxidation in spinach leaves.
Full text
PDF
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berry J. A., Osmond C. B., Lorimer G. H. Fixation of O(2) during Photorespiration: Kinetic and Steady-State Studies of the Photorespiratory Carbon Oxidation Cycle with Intact Leaves and Isolated Chloroplasts of C(3) Plants. Plant Physiol. 1978 Dec;62(6):954–967. doi: 10.1104/pp.62.6.954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lea P. J., Miflin B. J. Alternative route for nitrogen assimilation in higher plants. Nature. 1974 Oct 18;251(5476):614–616. doi: 10.1038/251614a0. [DOI] [PubMed] [Google Scholar]
- Rognes Sven Erik. A glutamine-dependent asparagine synthetase from yellow lupine seedlings. FEBS Lett. 1970 Sep 18;10(1):62–66. doi: 10.1016/0014-5793(70)80416-1. [DOI] [PubMed] [Google Scholar]
- Streeter J. G. In vivo and in vitro studies on asparagine biosynthesis in soybean seedlings. Arch Biochem Biophys. 1973 Aug;157(2):613–624. doi: 10.1016/0003-9861(73)90681-4. [DOI] [PubMed] [Google Scholar]
- Tolbert N. E. Compartmentation and control in microbodies. Symp Soc Exp Biol. 1973;27:215–239. [PubMed] [Google Scholar]
- Wallsgrove R. M., Lea P. J., Miflin B. J. Distribution of the Enzymes of Nitrogen Assimilation within the Pea Leaf Cell. Plant Physiol. 1979 Feb;63(2):232–236. doi: 10.1104/pp.63.2.232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woo K. C., Osmond C. B. Stimulation of ammonia and 2-oxoglutarate-dependent o(2) evolution in isolated chloroplasts by dicarboxylates and the role of the chloroplast in photorespiratory nitrogen recycling. Plant Physiol. 1982 Mar;69(3):591–596. doi: 10.1104/pp.69.3.591. [DOI] [PMC free article] [PubMed] [Google Scholar]


