Abstract
The Asian Thyroid Working Group was founded in 2017 at the 12th Asia Oceania Thyroid Association (AOTA) Congress in Busan, Korea. This group activity aims to characterize Asian thyroid nodule practice and establish strict diagnostic criteria for thyroid carcinomas, a reporting system for thyroid fine needle aspiration cytology without the aid of gene panel tests, and new clinical guidelines appropriate to conservative Asian thyroid nodule practice based on scientific evidence obtained from Asian patient cohorts. Asian thyroid nodule practice is usually designed for patient-centered clinical practice, which is based on the Hippocratic Oath, “First do not harm patients,” and an oriental filial piety “Do not harm one’s own body because it is a precious gift from parents,” which is remote from defensive medical practice in the West where physicians, including pathologists, suffer from severe malpractice climate. Furthermore, Asian practice emphasizes the importance of resource management in navigating the overdiagnosis of low-risk thyroid carcinomas. This article summarizes the Asian Thyroid Working Group activities in the past 7 years, from 2017 to 2023, highlighting the diversity of thyroid nodule practice between Asia and the West and the background reasons why Asian clinicians and pathologists modified Western systems significantly.
Keywords: Asia, Thyroid neoplasms, Fine needle aspiration (FNA), Diagnostic criteria, Patient centered care, Active surveillance, Thyroidectomy
While universally guided by fundamental principles, the discipline of pathology is inherently influenced by regional variations. Asian pathologists and cytopathologists, despite being educated predominantly through international textbooks written by Western scholars, often find that the ground realities and nuances in their regions necessitate adaptations. Systems like the Bethesda System for Reporting Thyroid Cytology (TBSRTC) [1], the World Health Organization (WHO) classification of thyroid tumors [2,3], and the American Thyroid Association clinical guidelines [4] are indispensable. Nevertheless, their direct application in Asia occasionally yields outcomes that diverge from Western findings [5-16]. The intersection of these variances traces back to a myriad of intertwined scientific and non-scientific factors.
Population-based factors
Beyond genetic and biological differences, cultural practices and socio-environmental determinants in Asia can greatly influence thyroid pathology outcomes. For instance, dietary iodine intake, prevalent in many Asian diets, can significantly affect thyroid physiology and pathology.
Socio-economic and healthcare infrastructure
Diverse Asian nations, with their distinct historical trajectories, have developed unique healthcare infrastructures. The accessibility and quality of healthcare, including diagnostic facilities, can vary even within countries, let alone between them.
Economic burden of medical care
The juxtaposition of North American and Asian healthcare expenditure strategies underlines more profound socio-economic and policy-driven differences. These variations often dictate clinical decisions, with patient affordability playing a crucial role. Medical expenses vary significantly across countries, with North America typically experiencing higher costs. Immediate surgery is frequently chosen to reduce these expenses. However, in other parts of the world, the approach often leans towards risk stratification for surgery and long-term clinical monitoring, as these tend to be more cost-effective than surgical interventions compared to the United States.
Healthcare insurance variations
Health insurance schemes’ dynamics, influenced by public policies and private market forces, can significantly sway diagnostic and treatment choices.
Medical specialization and density of pathologists
The density of pathologists across countries significantly shapes the landscape of medical practice [17]. The number of pathologists in a country is intrinsically linked to the efficiency and effectiveness of its medical system.
Medico-legal climate and clinical guidelines
The medico-legal environment molds clinical practice considerably. In some countries, clinical practice guidelines are established with a defensive approach due to the potential for physicians to face malpractice lawsuits. While defensive medicine may seem prudent in litigious societies, it can inadvertently lead to overdiagnosis and overtreatment, escalating healthcare costs. Conversely, other nations resist such defensive medicine because it escalates societal costs and burdens patients financially [18-20].
Terminological and classificatory nuances
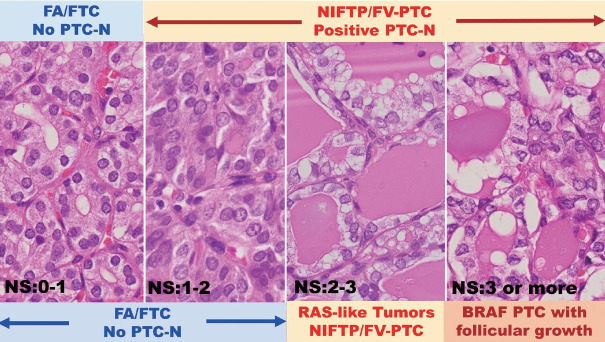
The evolution of medical terminologies and classifications, often reflecting deeper understandings of diseases, underscores the importance of constant knowledge exchange and adaptation among global medical communities. The noninvasive encapsulated follicular variant of papillary thyroid carcinoma (FVPTC) was once classified as a malignant tumor in American thyroid nodule practices. Its prevalence in the West is elevated due to a lower threshold for recognizing papillary thyroid carcinoma (PTC) nuclear features (Fig. 1) [21,22]. However, this classification was later changed to noninvasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) [2,23]. In many Asian practices, these are typically observed in benign follicular adenomas (FAs) due to a higher threshold for PTC-type nuclear features (Fig. 1), resulting in a lower prevalence of NIFTP in surgically removed nodules in Asia [2,8,11,12,14-16].
Fig. 1.
NS from 0 to 3 in encapsulated follicular pattern tumors results in variable benign, borderline, or malignant diagnoses between Asian and Western pathologists. Four illustrations (NS 0–1, NS 1–2, NS 2–3, and NS 3 or more) in encapsulated follicular pattern tumors often result in diverse benign, borderline, or malignant diagnoses among pathologists [5-7]. The two illustrations on the left are FA when noninvasive or FTC when invasive in Asian thyroid practice because those extremely delicate nuclear features are insufficient for PTC-type malignancy. Most Western pathologists accept nuclear features in the three illustrations (NS 1–2, NS 2–3, and NS 3 or more) on the right as positive for PTC-N and call them using the same diagnostic terminology, PTC type nuclear features, regardless of different genetic backgrounds, BRAF or RAS oncogene lineages [1-3,21-23]. Some Asian pathologists distinguish nuclear features in the two illustrations (NS of 2–3 and NS of 3 or more) on the right, either RAS-like FV-PTC or BRAF-like conventional PTC [14,16,35]. NS, nuclear score defined by Nikiforov et al. [2,23]. NS, nuclear scoring; FA, follicular adenoma; FTC, follicular thyroid carcinoma; NIFTP, noninvasive follicular thyroid neoplasm with papillarylike nuclear features; PTC, papillary thyroid carcinoma; PTC-N, papillary thyroid carcinoma type nuclear features; FV-PTC, follicular variant PTC; RAS, rat sarcoma virus; BRAF, v-Raf murine sarcoma viral oncogene homolog B.
Clinical management philosophies
The decision-making algorithms in medicine are based on clinical evidence, patient preferences, physician experiences, and systemic constraints. In Asian clinical practice, a more conservative management approach is typically adopted, emphasizing clinical risk stratification for high-risk indeterminate thyroid nodules, specifically follicular neoplasm (FN) [9-11,13-16]. Conversely, in North America, diagnostic surgery becomes the preferred course when patients cannot afford molecular tests [1,4,9, 10,11,13-15]. Consequently, in the West, indeterminate nodules, categorized as Bethesda III (atypia of undetermined significance [AUS]) and IV (FN), have a high resection rate (RR) of over 50% but a lower risk of malignancy (ROM) of less than 30%. In contrast, most Asian practices show a lower RR of less than 40% for these nodules, with a ROM exceeding 40%, even in the absence of molecular testing [9-11,13-16].
Country-specific approaches
Each country, influenced by its unique medical history, patient demographics, and healthcare policies, carves out its distinct approach to disease management. In Japan, a more conservative approach to management is prevalent [10,11,24-26]. Traditionally, lobectomy is the chosen procedure for low-risk small PTCs measuring 1–2 cm and for slightly larger low-risk PTCs, specifically, those sized less than 4 cm and categorized as T1–2, extrathyroidal extension (Ex) 0–1, N0–1, and M0 [24-26]. This conservative strategy offers notable benefits, most crucially the potential preservation of thyroid function. Such a conservative approach is particularly vital as patients who undergo total thyroidectomy and subsequently experience poorly managed hypothyroidism face severe risks, especially as they age and potentially struggle with proper hormone supplement management. Contrarily, in most other regions of Asia, as well as in North America and Europe, the predominant choice leans towards total thyroidectomy in the past [4,24-27].
Active surveillance vs. surgical intervention
The balance between surgical intervention and observation hinges on the collective experiences of a medical community, patient trust, and the broader healthcare framework. In Japan, over half of the patients with low-risk, small PTCs (less than 10 mm) opt for active surveillance, undergoing clinical follow-ups without surgery once a fine needle aspiration (FNA) confirms the malignancy [4,28-31]. Conversely, in the majority of other nations, FNA is typically not recommended for thyroid nodules that are small and low-risk, especially those under 1 cm [4]. This distinction arises from the fact that once an FNA identifies a nodule as malignant, surgery becomes the frequent choice, leading to potential overtreatment for patients with low-risk, small PTCs [4,14-16].
Grossing techniques for encapsulated follicular pattern thyroid tumors
As medicine advances, so does the need for precision. Nevertheless, precision must be juxtaposed against practicality in the light of resource optimization. Grossing techniques for encapsulated follicular pattern thyroid tumors vary globally. In Western countries, where pathologists often adopt defensive medical practices due to malpractice concerns, there is an emphasis on meticulously sampling the entire tumor capsules [2,15,16,32,33]. As recommended in Western textbooks, this practice has influenced the protocols in certain Asian laboratories, notably in China, Korea, Taiwan, and Thailand [32]. Conversely, Indian, Indonesian and Japanese labs generally select only representative sections, typically fewer than 10 per nodule, adhering to prior textbook guidelines [33,34]. This streamlined approach reduces the number of hematoxylin and eosin sections taken (from more than 20 sections to fewer than 10 per nodule) and conserves both pathologist time and societal resources.
The reasoning behind this more economical approach in India, Indonesia and Japan is multifaceted: (1) Lobectomy is deemed suitable for a range of nodules, from benign (e.g., FA) to borderline tumors (e.g., NIFTP, thyroid tumors of uncertain malignant potential), and even low-risk malignancies (like minimally invasive follicular thyroid carcinoma [FTC] or encapsulated conventional PTC) [4,24-26,33]. (2) Only pronounced, clear invasions carry significant implications for a patient’s prognosis. With this method focused on Sustainable Development Goals, there is virtually no risk of overlooking clinically consequential carcinomas, such as extensively angio-invasive or widely invasive carcinomas [2,3,33]. (3) Conducting exhaustive histological examinations to identify ambiguous invasions can result in the overtreatment of low-risk tumors, such as those termed minimally invasive FTCs or encapsulated FVPTCs (EFVPTCs). (4) Restricting the number of samples (to less than 10 sections) mirrors the clinical guideline of refraining from FNAs on low-risk thyroid nodules smaller than 1 cm. This approach encourages a clinical follow-up, subsequently reducing surgeries for small papillary carcinomas. Moreover, limiting samples can mitigate the unintended identification of questionable capsular invasions and, consequently, the diagnoses of minimally invasive FTCs and EFVPTCs. This, in turn, lessens the likelihood of excessive treatments, like completion thyroidectomy or radioactive iodine abrasion, particularly for very low-risk thyroid tumors with negligible prognostic impact [36-39].
Categorization of cyst fluid only samples
How we classify and interpret diagnostic samples evolves with our understanding of disease processes and the nuances of clinical implications. In past cytological practices, including in the United States, cyst fluid only samples from thyroid FNA cytology were typically categorized as benign. However, current Western practices have moved these samples to the inadequate category due to the substantial risk they pose for false-negative diagnoses and the potential inability to completely exclude cystic PTC [1,14-16]. In contrast, some countries in Asia/Oceania still classify these samples as benign, while European countries might place them in a specific subcategory under the inadequate category. This reclassification in Europe is attributed to the extremely low malignancy risk associated with these samples, which is on par with or even lower than the risk associated with samples in the benign category [9,15,16].
VARIATION IN THYROID NODULE PRACTICES ACROSS REGIONS
In 2002, some members of the Asian Thyroid Working Group observed notable discrepancies between Japanese and American pathologists concerning the classification of encapsulated follicular pattern thyroid tumors [5,6]. The majority of noninvasive encapsulated follicular pattern thyroid nodules exhibiting subtle nuclear atypia consistent with RAS-like tumors (RAS-like dysplasia) were classified as malignant PTCs by American pathologists. In contrast, Japanese pathologists often categorized them as benign FAs or hyperplastic nodules (Fig. 1) [5,6,16,21,22]. This observation was further corroborated by multiple subsequent studies [7,15,21,22]. Following a comprehensive review of 109 cases, which exhibited neither recurrence nor metastasis over an average 14-year follow-up period, the international NIFTP working group reclassified these noninvasive encapsulated follicular pattern tumors in 2016. Previously deemed as malignant (FVPTC), they were redefined as borderline tumors (NIFTP) [23]. Despite this, considerable disparities persist between Asian and Western practices, notably in the prevalence of NIFTP— a statistic that remains higher in the West [2,8,11,12,14-16,22,40-43] compared to Asia [12,14,44-49].
The Asian Thyroid Working Group was inaugurated in 2017 during the 12th Asia Oceania Thyroid Association (AOTA) Congress in Busan, Korea [49,50]. The group’s initial endeavors explored the distinctions between Asian and Western approaches to thyroid nodule management. In the West, FVPTC (often synonymous with RAS-like PTC) represents a significant portion of PTC diagnoses [1-4,12,22,23,37,51]. Consequently, the frequency of the BRAF V600E mutation is somewhat diminished in Western PTC cases, given that EFVPTCs are typically non-BRAF (RAS-like) tumors [40]. Conversely, Asian PTC samples manifest a higher prevalence of the BRAF V600E mutation, largely because EFVPTCs are scarcely identified in Asian thyroid evaluations. A majority of encapsulated follicular pattern tumors with mild RAS-like dysplastic nuclear alterations are designated as benign FA in prevalent Asian practices [6-8,11,14-16,40,41].
This manuscript chronicles the endeavors and discoveries of the Asian Thyroid Working Group from 2017 to 2023, underscoring the nuanced distinctions in thyroid nodule practices between Asia and the West. A comprehensive exploration of these global thyroid nodule practice variances will be the centerpiece of an upcoming textbook titled “Thyroid FNA Cytology, Differential Diagnoses, and Pitfalls: 3rd Edition” [52].
GROUP STUDY INSIGHTS
Asian perspectives on NIFTP [12,44-48]
A noninvasive encapsulated follicular pattern tumor, which exhibited worrisome nuclear features characteristic of PTC (RAS-like dysplasia), was previously categorized as a malignant tumor (FVPTC). This classification was predominant due to the lower threshold set for malignancy based on RAS-like PTC nuclear features in earlier Western thyroid nodule practices [5-8,21-23, 40-45]. This classification was later revised to “NIFTP” and was categorized as a borderline tumor in the 4th edition of the WHO classification [2,23,42,43]. Comprehensive data regarding the global incidence of NIFTP was derived from a thorough review of published studies and collaborative research efforts by members of the Asian Thyroid Working Group. These studies ascertained that the global occurrence of NIFTP is notably less than what was initially anticipated [12,47,48]. Furthermore, the incidence rates of NIFTP in Asia are substantially lower than those observed in North American and European regions. This discrepancy stems from the perception of NIFTP in Asia—it isn’t deemed to necessitate surgical intervention. Instead, a conservative approach that emphasizes risk stratification for surgical interventions is the conventional clinical protocol for managing indeterminate thyroid nodules in Asia [9-11,14-16]. This practice is also influenced by the stringent criteria for identifying PTC-type nuclear features in the region (Fig. 1) [5,6,11,14-16,21,22,40].
Malignant lymphoma in Asian practice [53]
This study analyzed 153 cases of primary thyroid lymphoma (PTL) collected from 10 institutions represented by members of the Asian Thyroid Working Group. The reported prevalence was 0.54% of all malignant thyroid tumors. The breakdown indicated that mucosa-associated lymphoid tissue lymphoma accounted for 54.9%, while diffuse large B-cell lymphoma represented 38.6%. Ultrasound examination and FNA cytology were the primary preoperative diagnostic tools, with flow cytometry conducted in five institutions. The findings suggest that the prevalence of PTL in non-Western countries is lower than previously reported in other studies.
Oncocytic (Hürthle cell) lesions in Asian practice [54]
Of 42,190 thyroid aspirates, 760 (1.8%) exhibited a predominance of Hürthle cells. These samples were sourced from nine hospitals across six Asian countries. The majority, or 61%, were categorized as “atypia of undetermined significance, Hürthle cell type” (AUS-H); 35% were identified as “follicular neoplasm, Hürthle cell type” (FN-H); and 4% were classified as “suspicious for malignancy” (SFM). Histologic follow-up was conducted for 288 of these aspirates (equivalent to 38%). Of these, a significant majority (66%) were determined to be benign upon resection, with the most common histologic diagnosis being Hürthle cell adenoma at 28.5%. The ROM for AUS-H, FN-H, and SFM, based on the resected nodules, was 32%, 31%, and 71%, respectively. Meanwhile, the risk of neoplasm was calculated to be 47%, 81%, and 77% for the respective categories.
Medullary (C cell) thyroid carcinoma in Asia practice [55-57]
From 13 hospitals across 8 Asia-Pacific countries, 145 cases of medullary (C cell) thyroid carcinoma (MTC) with accessible FNA slides were gathered for this study. Of these cases, 99 (68.3%) were preliminarily interpreted as either suspicious for MTC (SMTC) or confirmed as MTC. While cytological detection alone for MTC showed limitations, the combined application with auxiliary tests substantially enhanced diagnostic proficiency. The staining techniques employed varied across institutions and included Papanicolaou, hematoxylin-eosin, and Romanowsky methods. Liquid-based cytology was implemented in merely three of the countries. Following a comprehensive review of all cases, the diagnostic rate for MTC or S-MTC rose to 91.7% (133 out of the 145 cases). Based solely on cytomorphologic data, a plausible scoring system has been suggested to ensure optimal diagnostic precision. Additionally, Jung et al. [57] provided an overview of the latest advancements and modifications related to MTC as delineated in the WHO classification.
Capsular invasion study [58]
This investigation assessed interobserver concordance in evaluating capsular invasion among 11 thyroid pathologists from five Asian countries. This was accomplished using 20 cases presented as virtual slides. The levels of agreement for definitive invasive and noninvasive classifications were fair, evidenced by kappa values of 0.578 and 0.404, respectively. However, concordance was poor for cases with ambiguous invasion, as indicated by a kappa value of 0.186. The discrepancies in invasion assessment led to divergent final pathological conclusions. In summary, the research highlighted significant interobserver variability in the assessment of capsular invasion, particularly in FNs where the invasion was debatable.
Anaplastic thyroid carcinoma studies [59,60]
PAX8 in anaplastic thyroid carcinoma [59]
PAX8 immunohistochemistry using the MRQ-50 antibody was performed in whole tissue slides (n = 147) or tissue microarray sections (n = 35). The study found PAX8 expression in 54.4% of the cases, significantly lower than those reported in prior studies with the polyclonal antibody. PAX8 expression was positively correlated with an epithelial pattern (63.6% vs. 37.5%) and a coexisting differentiated thyroid carcinoma component (71.6% vs. 44.3%). Pathologists should be aware that PAX8 expression in anaplastic thyroid carcinoma (ATC) is less than those reported in early studies to avoid misdiagnosis.
Primary and secondary ATCs [60]
This study searched for ATCs in our institutional databases and the Surveillance, Epidemiology, and End Result (SEER) database. The multi-institutional database retrieved 22 primary (de novo) and 23 secondary ATCs (the patient had a history of differentiated thyroid cancer [DTC] or coexisting DTC components at the time of diagnosis). Compared to primary ATCs, secondary ATCs were not statistically different regarding demographics, clinical manifestations, and patient survival. The only clinical discrepancy between the two groups was a significantly larger tumor diameter of the primary ATCs. The prevalence of TERT promoter, PIK3CA, and TP53 mutations was comparable between the two subtypes. In comparison to primary ATCs, however, BRAF mutations were more prevalent (odds ratio [OR], 4.70; 95% confidence interval [CI], 2.84 to 7.78), whereas RAS mutations were less frequent (OR, 0.43; 95% CI, 0.21 to 0.85) in secondary tumors.
BRAF-like nuclear features and RAS-like dysplasia [35]
This study examined whether pathologists could distinguish BRAF-like and RAS-like nuclear features morphologically. This analysis suggests that nuclear pseudo-inclusions and high nuclear scores have diagnostic utility as rule-in markers for differentiating PTC with BRAF V600E mutation from benign or borderline follicular tumors with RAS-like mutations. Relaxation of rigid criteria for nuclear features resulted in an overdiagnosis of PTC. Immunostaining or molecular testing for BRAF V600E mutation is a valuable adjunct for cases with high nuclear scores to identify true PTC.
FA with papillary architecture and a proposal for a new borderline tumor, noninvasive encapsulated papillary RAS-like thyroid tumor [61,62]
The term “noninvasive encapsulated papillary RAS-like thyroid tumor (NEPRAS)” was introduced by Ohba et al. in 2019 [61] to describe a noninvasive thyroid tumor characterized by a complete fibrous capsule, a predominantly papillary architecture, and the presence of a RAS mutation, yet exhibiting only subtle nuclear features consistent with PTC (RAS-like dysplasia). This tumor poses a challenge for pathologists as it lies at the intersection between an encapsulated conventional BRAF-like PTC and the FA with papillary architecture, which was recognized as a benign tumor entity in the 5th edition of the WHO classification [2,3].
To address the diagnostic challenge and reduce the psychological impact on patients, the term “NEPRAS” was proposed, echoing the approach taken with the NIFTP terminology as introduced by Nikiforov et al. [23]. Subsequently, Jung et al. [62] documented three additional cases, suggesting that a favorable prognosis could be anticipated following surgical resection of such tumors. This optimism is grounded in the understanding that most encapsulated thyroid tumors, when not invasive, tend to be indolent, mirroring the behavior of FAs regardless of their growth patterns (be it NIFTP in follicular or NEPRAS in papillary patterns) [37,38,63]. However, it is noteworthy that extensive long-term follow-up data on a large patient cohort still needs to be available [62].
RR and ROM of indeterminate (Bethesda III and IV) cytology [13,64-66]
Without molecular tests [13,64]
Increasing evidence shows that clinicians employ different management strategies in their use of TBSRTC. This meta-analysis investigated the differences in diagnosis frequency, RR, and ROM between Western and Asian cytopathology practices. This study demonstrates a difference in Western and Asian thyroid cytology practice, especially regarding the indeterminate categories. Lower RR (51.3% vs. 37.6%) and higher ROM (25.4% vs. 41.9%) suggest that Asian clinicians adopt a more conservative approach, whereas immediate diagnostic surgery is favored in Western practice for indeterminate nodules.
With gene panel tests [65]
Compared with Afirma microarray-based Gene Expression Classifier, Gene Sequencing Classifier (GSC) had a higher benign call rate (BCR) (65.3% vs. 43.8%), a lower RR (26.8% vs. 50.1%), and a higher ROM (60.1% vs. 37.6%). The BCR of Hürthle cell-predominant nodules was significantly elevated (73.7% vs. 21.4%). In addition, the specificity (43.0% vs. 25.1%) and positive predictive value (63.1% vs. 41.6%) of Afirma GSC were significantly improved while it still maintained a high sensitivity (94.3%) and a high negative predictive value (90.0%). With an increased BCR and improved diagnostic performance, GSC could reduce the rate of unnecessary surgical interventions and better tailor the clinical decisions of patients with indeterminate thyroid FNA results.
With molecular tests in Asia [66]
This meta-analysis study included a total of 34 studies with 7,976 indeterminate nodules. The multigene panel testing methods were exclusively used in the United States. Compared with the non-molecular era, molecular testing was associated with a significantly increased ROM (47.9% vs. 32.1%). The ROM of indeterminate nodules in Asian institutes was significantly higher than in Western countries (75.3% vs. 36.6%). Institutes employing single-gene tests achieved a higher ROM (59.8% vs. 37.9%). Molecular testing is a promising method to tailor the clinical management for indeterminate thyroid FNA. The combination of molecular testing and active surveillance enhances the accuracy of case selection for surgery in Asian countries.
Pediatric thyroid carcinoma [67-69]
TBSRTC outputs, including frequency and ROM for most categories, were not statistically different from data in adult patients. However, the RR in the pediatric group was significantly higher in most of the categories compared with published adult data: benign, 23.2% vs. 13.0%; AUS, 62.6% vs. 36.2%; FN, 84.3% vs. 60.5%; and SFM, 93.8% vs. 69.7%. Pediatric patients with benign and indeterminate thyroid nodules had a higher RR than their adult counterparts, but the ROM of these categories in adults and children was not statistically different, suggesting a potential risk of overtreatment in pediatric patients. Determining the best treatment guidelines and additional tools for risk stratification must be a top priority to identify the target patient groups for surgical intervention precisely. Our study further demonstrated that Asian pediatric thyroid nodules had higher ROM than those from adults.
Coronavirus disease 2019 pandemic in Asian cytology practice [70]
This study examined the impact of coronavirus disease 2019 (COVID-19) on cytology practice in the Asia-Pacific region involving 167 cytopathology laboratories from 24 countries. The majority reported that restrictive measures that limited the accessibility of health care services had been implemented in their cities and/or countries (80.8%) and their hospitals (83.8%). Approximately one-half of the participants reported the implementation of new biosafety protocols (54.5%) and improving laboratory facilities (47.3%). The majority of the respondents reported a significant reduction (> 10%) in caseload associated with both gynecological (82.0%) and nongynecological specimens (78.4%). Ten out of 14 authors were from the Asian Thyroid Working Group members.
Other meta-analyses of the literature [71-74]
Major fusion oncogenes in PTC [71]
This meta-analysis using 27 studies showed NTRK-, RET-, BRAF-, and ALK-rearranged PTCs had a unique demographic/clinicopathological profile but similar progression-free survival (PFS) and overall survival. NTRK1-positive PTCs demonstrated more aggressive clinical behaviors and shorter PFS than NTRK3-positive PTCs, whereas RET rearrangement variants shared comparable clinicopathological backgrounds. This study provides new insights and facilitates our understanding of clinicopathological features and survival outcomes of different fusion oncogenes in PTCs.
The metastatic pattern of thyroid carcinomas [72]
We included 2,787 M1 thyroid cancers for statistical analyses, and the incidence of distant metastasis at presentation was 2.4%. Lung was the most common metastatic site for ATC, poorly differentiated thyroid carcinoma, PTC, and oncocytic cell carcinoma, whereas bone is the favorable disseminated site of FTC and MTC. Patients with multi-organ metastases had the worst survival, whereas bone metastases were associated with a favorable outcome. Significant differences exist in distant metastasis patterns of thyroid cancer subtypes and their corresponding survival.
Malignant thyroid teratoma [73]
We incorporated the SEER data with published malignant thyroid teratoma (MTT) cases in the literature to analyze the characteristics and prognostic factors of MTTs. Our results showed that MTT is typically seen in adult females. These neoplasms were associated with an aggressive clinical course with high rates of extrathyroidal extension (80%) and nodal involvement (62%). During follow-up, the development of recurrence and metastases were common (42% and 46%, respectively), and one-third of patients died at the last follow-up.
Mucoepidermoid carcinoma and sclerosing mucoepidermoid carcinoma with eosinophilia [74]
This multicenter study of mucoepidermoid carcinoma (MEC) and MEC with eosinophilia (SMECE) integrated our data with published literature to further investigate these tumors’ clinicopathological characteristics and prognoses. Histopathologically, MECs and SMECEs comprised two main cell types, including epidermoid and mucin-secreting cells, arranged in cords, nests, and tubules. SMECEs were characterized by a densely sclerotic stroma with abundant eosinophils. SMECEs had a superior disease-specific survival rate compared to MECs, suggesting that they are low-grade cancers. This could help clinicians better evaluate patient outcomes and decide appropriate treatment plans.
Future group studies in the Asian Thyroid Working Group
One of the most promising and valuable projects for pathologists is revising thyroid tumor classification, such as refining new tumor entities and establishing a molecular classification of thyroid tumors. The current WHO classification is insufficient for genuine molecular classification, and Western pathologists distinguish RAS-mutated encapsulated follicular pattern thyroid tumors into two tumor groups, FA/tumors of uncertain malignant potential (UMP)/FTC and NIFTP/UMP/FVPTC, according to the absence (NS 0–1) or presence (NS 2–3) of RAS-like dysplastic nuclear features (Fig. 1). This distinction has very little clinical imprecation because both are treated similarly, and the outcomes are almost identical [2,4]. In Asian thyroid practice, the distinction between FA/UMP/FTC and NIFTP/UMP/FVPTC is not strict and often handles them into one tumor lineage, FA/FTC. Thus, NIFTP and FVPTC are rare in Asia [12], while FTC is a vanishing tumor entity and is frequently classified as FVPTC in recent Western practice [75,76]. The lack of uniformity in diagnosing encapsulated follicular pattern tumors with delicate RAS-nuclear features among pathologists creates serious confusion in referring clinicians in some instances and hinders data sharing among institutes. It is a time to combine both RAS-like tumors into one tumor entity, as there are no benefits in the strict sub-classification of RAS-like tumors (FA/UMP/FTC vs. NIFTP/UMP/FVPTC) for treating physicians and patients. The author believes that universally accepted handling of the same RAS oncogene-mutated thyroid tumors is essential to minimize observer variation of encapsulated follicular pattern tumors and establish better communication between Asia and the West, harmonizing them into one world.
SPECIAL ISSUES CONDUCTED BY THE ASIAN THYROID WORKING GROUP MEMBERS
Since 2016, multiple Asian Thyroid Working Group members have curated special issues in various journals. These special issues include:
Journal of Basic & Clinical Medicine
In 2016, a pivotal moment in pathology occurred with Nikiforov’s publication of a seminal paper introducing the borderline tumor entity known as NIFTP [23]. Recognizing the importance and potential implications of this new classification, Dr. Kakudo, in the same year, invited a distinguished cohort of 15 international authors, inclusive of four members from the Asian Thyroid Working Group, to present their perspectives and insights on NIFTP in the Journal of Basic & Clinical Medicine (Table 1). It is unfortunate that the journal above subsequently ceased its operations, and its digital presence has vanished. Nevertheless, most of the seminal works, including those discussing NIFTP, are retrievable online, specifically at Dr. Kakudo’s website (http://www.kakudok.jp/english/basic_and_ clinical_medicine/) and the individual ResearchGate profiles of the respective authors.
Table 1.
Publications on NIFTP in the Journal of Basic & Clinical Medicine, 2017
| No. | Author(s) | Title | Volume, page |
|---|---|---|---|
| 1 | Kakudo K [77] | Non-invasive encapsulated/well-circumscribed follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) and precursor thyroid tumors | 6(1):1-2 |
| 2 | Kakudo K [78] | Unsettled issues in non-invasive encapsulated/well-circumscribed follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) and precursor thyroid tumors | 6(1):3-7 |
| 3 | LiVolsi VA, Baloch ZW [79] | Coming to terms with diagnosis “Non-invasive follicular neoplasm with papillary-like nuclear features (NIFTP)’: practice changer in endocrine pathology | 6(1):8-13 |
| 4 | Kakudo K, Liu Z, Satoh S, Higuchi M, Hirokawa M [80] | Non-invasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP): diagnosis and differential diagnoses | 6(1):14-21 |
| 5 | Liu Z, Song Y, Han B, Zhang X, Su P, Cui X [81] | Non-invasive follicular thyroid neoplasm with papillary-like nuclear features and the practice in Qilu Hospital of Shandong University, China | 6(1):22-25 |
| 6 | Jung CK, Kim C [82] | Effect of lowering the diagnostic threshold for encapsulated follicular variant of papillary thyroid carcinoma on the prevalence of non-invasive follicular thyroid neoplasm with papillary-like nuclear features: a single-institution experience in Korea | 6(1):26-28 |
| 7 | Pusztaszeri MP, Triponez F, Meyer P, Sadowski SM [83] | Noninvasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP): report of an institutional experience with 86 cases | 6(1):29-35 |
| 8 | Rossi ED [84] | NIFTP diagnosis: roses and thorns for cytopathologists and histopathologists | 6(1):36-37 |
| 9 | Canberk S, Baloch ZW, Ince U, Schmitt F [85] | Diagnosis of non-invasive follicular tumor with papillary-like nuclear features (NIFTP): a practice changer for thyroid fine-needle aspiration interpretation | 6(1):38-43 |
| 10 | Maletta F, Volante M, Papotti M [86] | Experience on NIFTP cytology, with a mini meta-analysis of the literature | 6(1):44-50 |
| 11 | Ng D, Can NT, Ma ZV, van Zante A, Ljung BM, Khanafshar E [87] | Cytomorphologic features of noninvasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP): a comparison with infiltrative follicular variant of papillary thyroid carcinoma | 6(1):51-56 |
| 12 | Saglietti C, Bongiovanni M [88] | The value of cytological examination in the diagnosis of noninvasive thyroid neoplasm with papillary-like nuclear features (NIFTP) | 6(1):57-60 |
| 13 | Wu HH [89] | The impact of NIFTP on FNA cytology: can we still diagnose papillary thyroid carcinoma? | 6(1):61-62 |
| 14 | Yang GCH [90] | Ultrasound is cytopathologist’s best friend in the era of noninvasive follicular thyroid neoplasm with papillary-like nuclear features | 6(1):65-67 |
| 15 | Poller DN, Johnson SJ, Stephenson TJ [91] | Diagnosis of NIFTP in the UK | 6(1):63-64 |
NIFTP, noninvasive follicular thyroid neoplasm with papillary-like nuclear features.
Journal of Pathology and Translational Medicine
In the AOTA Busan meeting, the Asian Thyroid Working Group commenced its operations in 2017. To begin their collaborative efforts, the Asian Thyroid Working Group summarized and reported the prevailing status of thyroid FNA cytology practices across seven Asian nations. These findings were subsequently published in a special issue of the Journal of Pathology and Translational Medicine, sponsored by the Korean Society of Pathologists and the Korean Society for Cytopathology. The articles from this special issue are delineated below in Table 2.
Table 2.
Articles on thyroid fine needle aspiration practices in Asia from Journal of Pathology and Translational Medicine, 2017
| No. | Author(s) | Title | Volume, page |
|---|---|---|---|
| 1 | Bychkov A, Kakudo K, Hong S [49] | Current practices of thyroid fine-needle aspiration in Asia: a missing voice | 51(6):517-520 |
| 2 | Cha YJ, Pyo JY, Hong SW, et al. [92] | Thyroid fine-needle aspiration cytology practice in Korea | 51(6):521-527 |
| 3 | Liu Z, Dongge D, Ma B, et al. [93] | History and practice of thyroid fine-needle aspiration in China, based on retrospective study of the practice in Shandong University Qilu Hospital | 51(6):528-532 |
| 4 | Agarwal S, Jain D [94] | Thyroid cytology in India: contemporary review and meta-analysis | 51(6):533-547 |
| 5 | Satoh S, Yamashita H, Kakudo K [95] | Thyroid cytology: the Japanese system and experience at Yamashita Thyroid Hospital | 51(6):548-554 |
| 6 | Abelardo AD [96] | Thyroid fine-needle aspiration practice in the Philippines | 51(6):555-559 |
| 7 | Hang JF, Hsu CY, Lai CR [97] | Thyroid fine-needle aspiration in Taiwan: the history and current practice | 51(6):560-564 |
| 8 | Keelawat S, Rangdaeng S, Koonmee S, Jitpasutham T, Bychkov A [98] | Current status of thyroid fine-needle aspiration practice in Thailand | 51(6):565-570 |
| 9 | Jung CK, Hong S, Bychkov A, Kakudo K [50] | The use of fine-needle aspiration (FNA) cytology in patients with thyroid nodules in Asia: a brief overview of studies from the Working Group of Asian Thyroid FNA Cytology | 51(6):571-578 |
Gland Surgery
“Asian and Western Practice in Thyroid Pathology: Similarities and Differences” was a themed issue published in Gland Surgery (Table 3). It addressed critical differences observed when Western systems for thyroid pathology and cytology were adopted in Asian practice. Dr. Kakudo invited 25 international authors, of which 19 are members of the Asian Thyroid Working Group. These original articles, reviews, and meta-analyses highlight significant differences. Some of these disparities are entirely understandable and expected, while others are deemed scientifically unacceptable and not suitable for patient-centered care.
Table 3.
Articles on “Asian and Western Practice in Thyroid Pathology: Similarities and Differences” in Gland Surgery, 2020
| No. | Author(s) | Title | Volume, page |
|---|---|---|---|
| 1 | Kakudo K [14] | Asian and Western practice in thyroid pathology: similarities and differences | 9(5):1614-1627 |
| 2 | Ohori NP [99] | Molecular testing and thyroid nodule management in North America | 9(5):1628-1638 |
| 3 | Kumarasinghe MP [100] | Standardisation of thyroid cytology terminology and practice: are modifications necessary? -a narrative review | 9(5):1639-1647 |
| 4 | Poller DN [101] | Litigation in thyroid cytology and histopathology in England: a very brief overview | 9(5):1648-1652 |
| 5 | Hirokawa M, Suzuki A, Higuchi M, et al. [102] | The Japanese reporting system for thyroid aspiration cytology 2019 (JRSTAC2019) | 9(5):1653-1662 |
| 6 | Ito Y, Miyauchi A [103] | Active surveillance of low-risk papillary thyroid microcarcinomas | 9(5):1663-1673 |
| 7 | Zhu Y, Wu H, Huang B, Shen X, Cai G, Gu X [104] | BRAFV600E mutation combined with American College of Radiology thyroid imaging report and data system significantly changes surgical resection rate and risk of malignancy in thyroid cytopathology practice | 9(5):1674-1684 |
| 8 | Pusztaszeri MP, Tamilia M, Payne RJ [105] | Active surveillance for low-risk small papillary thyroid cancer in North American countries: past, present and future (bridging the gap between North American and Asian practices) | 9(5):1685-1697 |
| 9 | Okamoto T, Omi Y, Yoshida Y, Horiuchi K, Abe K [106] | Radioactive iodine treatment of papillary thyroid carcinoma in Japan | 9(5):1698-1707 |
| 10 | Michael CW, Kameyama K, Kitagawa W, Azar N [107] | Rapid on-site evaluation (ROSE) for fine needle aspiration of thyroid: benefits, challenges and innovative solutions | 9(5):1708-1715 |
| 11 | Liu Z, Sui S, Su P, et al. [108] | The effect of implementing pre-surgical ultrasound-guided fine-needle aspiration biopsy on thyroid surgery, a 6-year interrupted time series analysis in Qilu Hospital of Shandong University | 9(5):1716-1723 |
| 12 | Canberk S [109] | Precursor and borderline lesions of the thyroid (indolent lesions of epithelial origin): from theory to practice | 9(5):1724-1734 |
| 13 | Nguyen TPX, Truong VT, Kakudo K, Vuong HG [64] | The diversities in thyroid cytopathology practices among Asian countries using the Bethesda System for Reporting Thyroid Cytopathology | 9(5):1735-1746 |
| 14 | Oo ZP, Hlaing AM, Kyi KCS, Fukuoka J, Bychkov A [110] | An overview of thyroid fine-needle aspiration practice in Myanmar | 9(5):1747-1753 |
| 15 | Tangnuntachai N, Rangdaeng S, Koonmee S, Tangjaturonrasme N, Keelawat S [111] | Pathological practice and management of thyroid nodules: a Thai perspective | 9(5):1754-1763 |
| 16 | Ooi LY, Nga ME [112] | Atypia of undetermined significance/follicular lesion of undetermined significance: Asian vs. non-Asian practice, and the Singapore experience | 9(5):1764-1787 |
| 17 | Abelardo AD, Sotalbo KCJ [113] | Clinical management of thyroid aspirates diagnosed as atypia of undetermined significance in the Philippines | 9(5):1788-1796 |
| 18 | Guleria P, Mani K, Agarwal S [114] | Indian experience of AUS/FLUS diagnosis: is it different from rest of Asia and the West? -A systematic review and meta-analysis | 9(5):1797-1812 |
| 19 | Odate T, Oishi N, Vuong HG, Mochizuki K, Kondo T [115] | Genetic differences in follicular thyroid carcinoma between Asian and Western countries: a systematic review | 9(5):1813-1826 |
| 20 | Nakra T, Jain D, Agarwal S [116] | Thyroid lymphoproliferative lesions in Asia | 9(5):1827-1837 |
| 21 | Li Y, Inomata K, Nishihara E, Kakudo K [117] | IgG4 thyroiditis in the Asian population | 9(5):1838-1846 |
| 22 | Bai Y, Niu D, Yao Q, Lin D, Kakudo K [118] | Updates in the advances of sporadic medullary thyroid carcinoma: from the molecules to the clinic | 9(5):1847-1856 |
| 23 | Jung CK, Lee S, Bae JS, Lim DJ [119] | Late-onset distant metastases confer poor prognosis in patients with well-differentiated thyroid cancer | 9(5):1857-1866 |
| 24 | Choden S, Keelawat S, Jung CK, Bychkov A [120] | An affordable immunohistochemical approach to estimate the prevalence of BRAFV600E in large cohort studies—establishing the baseline rate of BRAF mutation in an institutional series of papillary thyroid carcinoma from Thailand | 9(5):1867-1877 |
| 25 | Rashid FA, Munkhdelger J, Fukuoka J, Bychkov A [121] | Prevalence of BRAFV600E mutation in Asian series of papillary thyroid carcinoma— a contemporary systematic review | 9(5):1878-1900 |
THE ASIAN THYROID WORKING GROUP COMPANION MEETINGS
The inaugural Asian Thyroid Working Group Companion Meeting was convened at the 18th AOTA Congress in Busan, Korea, on March 16, 2017. Subsequent Companion Meetings have been held annually, as detailed in Tables 4–8. However, the meetings scheduled for 2020 and 2021 were canceled due to the COVID-19 pandemic.
Table 4.
Pre-Congress Joint Symposium by Working Group of Asian Thyroid FNA Cytology held on March 16, 2017 in Busan, Korea
| Chairpersons | Topic | Presenter | |
|---|---|---|---|
| Part 1: Thyroid FNA Cytology in Asian Countries, Current and Future | |||
| SoonWon Hong (Korea) and Chiung-Ru Lai (Taiwan) | Thyroid FNA practice in Korea | SoonWon Hong (Korea) | |
| Cytological-histological correlation studies on thyroid FNA from Thailand | Somboon Keelawat (Thailand) | ||
| Thyroid FNA practice in China | Zhiyan Liu (China) | ||
| Current status of thyroid FNA cytology in Taiwan | Chiung-Ru Lai (Taiwan) | ||
| The Japanese system of thyroid FNA cytology | Shinya Satoh (Japan) | ||
| Time travel of an old entity with a new name: NIFTP. A cytomorphologist’s perspective and data analyze of institutes from Turkey | Sule Canberk (Turkey) | ||
| Part 2: Impacts of NIFTP on Thyroid FNA Cytology | |||
| Chan Kwon Jung (Korea) and Mitsuyoshi Hirokawa (Japan) | Proposal of NIFTP and borderline thyroid tumors | Kennichi Kakudo (Japan) | |
| Impact of NIFTP on rates of malignancy for FNA diagnostic categories | Andrey Bychkov (Thailand) | ||
| Cytological Findings and Diagnostic Significance of NIFTP and WDT-UMP | Mitsuyoshi Hirokawa (Japan) | ||
| Diagnostic criteria of PTC-N, NIFTP and WDT-UMP among Asian pathologists | Zhiyan Liu (China) | ||
| Molecular correlates and rate of lymph node metastasis of NIFTP and invasive EFVPTC | Chan Kwon Jung (Korea) | ||
FNA, fine needle aspiration; NIFTP, noninvasive follicular thyroid neoplasm with papillary-like nuclear features; WDT, well differentiated tumor; UMP, uncertain malignant potential; PTC-N, papillary thyroid carcinoma type nuclear features; EFVPTC, encapsulated follicular variant of papillary thyroid carcinoma.
Table 5.
The Working Group of Asian thyroid FNA cytology: recent achievements, current activities, and prospective directions was held on January 19, 2018 in Chiang Mai, Thailand, as the second companion meeting by the Asian Thyroid WG
| Chairpersons | Topic | Presenter |
|---|---|---|
| Samreung Rangdaeng (Thailand) and Kennichi Kakudo (Japan) | Thyroid FNA practice in the Philippines | Agustina Abelardo (Philippines) |
| A survey of current practice of thyroid FNA in Taiwan | Jen-Fan Hang (Taiwan) | |
| Experience of implementing Bethesda system of reporting in BPKIHS, Nepal | Sushil Dhakal (Nepal) | |
| Thyroid FNA in single vs. multiple unit service | Pichet Sampatanukul (Thailand) | |
| Active surveillance for indeterminate thyroid nodules and risk of malignancy | Kennichi Kakudo (Japan) | |
| Diagnostic impact of BRAF testing in thyroid FNA | Ju Yeon Pyo (Korea) | |
| Molecular profile of NIFTP | Chan Kwon Jung (Korea) | |
| How the introduction of NIFTP has changed our practice – Results of survey by the WG | Andrey Bychkov (Thailand) | |
| Where is the NIFTP in thyroid FNA | SoonWon Hong (Korea) | |
| Evaluation of follicular patterned lesions “including oncocytic” by FNA based on the new WHO classification | Sule Canberk (Turkey) | |
| Atypia of undetermined significance: Its cyto-histologic outcome and risk of malignancy | Annette Salillas (Philippines) | |
| Needle tract implantation after thyroid fine needle aspiration procedure | Toshitetsu Hayashi (Japan) |
FNA, fine needle aspiration; NIFTP, noninvasive follicular thyroid neoplasm with papillary-like nuclear features; WHO, World Health Organization.
Table 6.
The third Asian Thyroid Working Group Companion Meeting (Asian Practice of Thyroid FNA Cytology) was held on May 8, 2019 at the 20th International Congress of Cytology (ICC Sydney)
| Chairpersons | Topic | Presenter |
|---|---|---|
| Chiung-Ru Lai (Taiwan) and Chan Kwon Jung (Korea) | A welcome message and Introduction of Australian System for Reporting Thyroid FNA Cytology | Pryianthi Kumarasinghe (Australia) |
| The use of the Bethesda System for Reporting Thyroid Cytopathology in Korea: a nationwide multicenter survey by the Korean Society of Endocrine Pathologists | SoonWon Hong (Korea) | |
| Active surveillance for indeterminate thyroid nodules in China | Yun Zhu (China) | |
| Implementation of the Bethesda system for reporting thyroid cytopathology in Japan | Mitsuyoshi Hirokawa (Japan) | |
| Impact of NIFTP on the Bethesda system in Asian practice | Andrey Bychkov (Thailand) | |
| Hürthle cell lesions in Asian practice: multi-institutional studies in 2018 and 2019 | Deepali Jain (India) |
FNA, fine needle aspiration; NIFTP, noninvasive follicular thyroid neoplasm with papillary-like nuclear features.
Table 7.
On 16th of November 2019, one more (the 4th face to face meeting) companion meeting was held at the 58th Japanese Society of Clinical Cytology (JSCC) Fall Meeting in Okayama, Japan as the Global Asian Forum
| Chairpersons | Topic | Presenter |
|---|---|---|
| Andrey Bychkov (Japan) and Kennichi Kakudo (Japan) | Nuclear features of BRAF mutated PTCs and RAS-mutated neoplasms | Chan Kwon Jung (Korea) |
| AUS/FLUS in Indian experience with thyroid FNA | Shipra Agarwal (India) | |
| BRAF testing to refine cytology categories: how and when | Priyanthi Kumarasinghe (Australia) | |
| Resection rate and risk of malignancy among TBSRTC categories in Western vs. Asian practice | Huyen-Trang Vu (Vietnam) | |
| Cytological diagnosis of medullary thyroid carcinoma among Asian Working Group in Thyroid Cytology | Chih-Yi Liu (Taiwan) | |
| Observer variation study in the diagnosis of follicular neoplasms | Yaqiong Li (China) |
PTC, papillary thyroid carcinoma; AUS, atypia of undetermined significance; FLUS, follicular lesions of undetermined significance; FNA, fine needle aspiration; TBSRTC, The Bethesda System for Reporting Thyroid Cytopathology.
Table 8.
At the 21st International Congress of Cytology held at Baltimore, USA, the 5th face to face companion meeting by the Asian Thyroid Working Group was held on November 18, 2022, as a cytology short course 5: Why Are There Significant Differences Among Us in Thyroid Nodule Practices?
| Chairpersons | Topic | Presenter |
|---|---|---|
| Kennichi Kakudo (Japan) and Jen-Fan Hang (Taiwan) | A high diagnostic threshold of RAS-like nuclear features in Asian pathologists impacts thyroid nodule practice | Kennichi Kakudo (Japan) |
| Diagnoses of BRAF-like PTC and RAS-like PTC | Chan Kwon Jung (Korea) | |
| Molecular testing for cytologically indeterminate thyroid nodules | Jen-Fan Hang (Taiwan) | |
| Japanese thyroid practice using the Bethesda system | Mitsuyoshi Hirokawa (Japan) |
PTC, papillary thyroid carcinoma.
FUTURE PERSPECTIVES
Kennichi Kakudo served as the esteemed president of the Asian Thyroid Working Group from 2017 to 2023, concluding his tenure in July 2023. During this period, the core members who played a pivotal role alongside Dr. Kakudo were Chan Kwon Jung from Korea, Zhiyan Liu from China, Mitsuyoshi Hirokawa from Japan, and Andrey Bychkov, who had affiliations with Russia, Thailand, and Japan. In a significant transition, Chiung-Ru Lai (Taiwan) was nominated as the president in July 2023. This nomination was subsequently ratified during a web meeting held by the core members. As of August 2023, the core team under Dr. Lai’s leadership includes Chan Kwon Jung (Korea), Zhiyan Liu (China), Andrey Bychkov (Japan), Radhika Srinivasan (India), Mitsuyoshi Hirokawa (Japan), Somboon Keelawat (Thailand), with Kennichi Kakudo (Japan) serving as a consultant, and Jen-Fan Hang (Taiwan) fulfilling the role of secretary.
There are numerous group studies that are presently under-way, with their findings expected to be published in the near future. The Asian Thyroid Working Group remains committed to offering a collaborative platform for all its members, which is composed of 58 members from 14 countries in September 2023. A notable contribution to the academic community will be the 3rd edition of “Thyroid FNA Cytology, Differential Diagnosis, and Pitfalls,” slated for release in 2023. This edition will encompass an in-depth discussion of all topics presented in the article as mentioned above [52]. The volume will feature more than 60 chapters penned by esteemed Asian authors. Within these chapters, we present a reporting system tailored to the Asian medical context, given that high-cost gene panel tests are not a commonplace practice (as detailed in Table 9).
Table 9.
Histological type-oriented reporting system of thyroid FNA cytology, useful for ancillary tests
| Cytological category | |
|---|---|
| 1) Inadequate (non-diagnostic) | |
| Adequate specimen categories | |
| 2) Normal or benign | |
| 3) RAS-like (FA/FTC) tumor lineage | |
| A1: Favor benign (low risk) | |
| A2: Borderline (intermediate risk) | |
| A3: Favor malignant (high risk) | |
| A4: Suspicious for malignancy (probability of FTC, PDC, etc. should be stated) | |
| 4) BRAF-like (PTC) tumor lineage | |
| B1: Low risk dysplasia | |
| B2: Intermediate risk dysplasia | |
| B3: High risk dysplasia (suspicious for PTC) | |
| B4: Conclusive PTC type malignancy | |
| 5) Other types (unspecified lineage, or other than RAS-like and BRAF-like tumor lineages) | |
| C1: Low risk | |
| C2: Intermediate risk | |
| C3: High risk (probability of C cell carcinoma, ATC, ML and metastatic carcinoma should be stated) | |
The original idea of this histological type-oriented reporting system of thyroid FNA cytology was first proposed by Kakudo et al in 2019 [122] and updated in 2023 [52].
FNA, fine needle aspiration; RAS, rat sarcoma virus; FA, follicular adenoma; FTC, follicular thyroid carcinoma; PDC, poorly differentiated carcinoma; PTC, papillary thyroid carcinoma; BRAF, v-Raf murine sarcoma viral oncogene homolog B; ATC, anaplastic thyroid carcinoma; ML, malignant lymphoma.
The core team, along with the project leader, is enthusiastic about creating more avenues for budding Asian pathologists and cytopathologists to engage with the broader international pathology community. We ardently hope that the activities under the aegis of the Asian Thyroid Working Group will act as a springboard for the younger generation, integrating them with the global community and bolstering their careers in pathology and cytopathology. A list of publications by the Asian Thyroid Working members from 2017 to 2023 is available in Supplementary Table S1.
Acknowledgments
The authors thank all the members of the Asian Thyroid Working Group for their generous support and collaborations on thyroid disease studies.
Footnotes
Ethics Statement
Not applicable.
Availability of Data and Material
A publication list by Asian WG member is available as a Supplementary Table S1.
Code Availability
Not applicable.
Author contributions
Conceptualization: KK. CKJ. Data curation: KK, CKJ, HGV, JFH. Formal analysis: KK. Funding acquisition: KK. Investigation: KK. Methodology: all authors. Project administration: KK. Resources: KK. Supervision: KK. Validation: all authors. Visualization: KK, CKJ, HGV, JFH. Writing—original draft: KK. Writing—review & editing: KK, CKJ, HGV, JFH. Approval of final manuscript: all authors.
Conflicts of Interest
C.K.J., the editor-in-chief, along with K.K., Z.L., A.B., and C.-R.L., who are contributing editors of the Journal of Pathology and Translational Medicine, were not involved in the editorial evaluation or decision to publish this article. All other authors have declared no conflicts of interest.
Funding Statement
No funding to declare.
Supplementary Information
The Data Supplement is available with this article at https://doi.org/10.4132/jptm.2023.10.04.
A list of publications by the Asian Thyroid Working members from 2017 to 2023
References
- 1.Ali SZ, Vanderlaan P. The Bethesda System for Reporting Thyroid Cytopathology. 3rd ed. New York: Springer; 2023. [Google Scholar]
- 2.WHO Classification of Tumours Editorial Board . WHO classification of tumours of endocrine organs. 5th ed. Lyon: IARC Press; 2022. [Google Scholar]
- 3.Jung CK, Bychkov A, Kakudo K. Update from the 2022 World Health Organization classification of thyroid tumors: a standardized diagnostic approach. Endocrinol Metab (Seoul) 2022;37:703–18. doi: 10.3803/EnM.2022.1553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Haugen BR, Alexander EK, Bible KC, et al. 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016;26:1–133. doi: 10.1089/thy.2015.0020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kakudo K, Katoh R, Sakamoto A, et al. Thyroid gland: international case conference. Endocr Pathol. 2002;13:131–4. doi: 10.1385/ep:13:2:131. [DOI] [PubMed] [Google Scholar]
- 6.Hirokawa M, Carney JA, Goellner JR, et al. Observer variation of encapsulated follicular lesions of the thyroid gland. Am J Surg Pathol. 2002;26:1508–14. doi: 10.1097/00000478-200211000-00014. [DOI] [PubMed] [Google Scholar]
- 7.Lloyd RV, Erickson LA, Casey MB, et al. Observer variation in the diagnosis of follicular variant of papillary thyroid carcinoma. Am J Surg Pathol. 2004;28:1336–40. doi: 10.1097/01.pas.0000135519.34847.f6. [DOI] [PubMed] [Google Scholar]
- 8.Liu Z, Zhou G, Nakamura M, et al. Encapsulated follicular thyroid tumor with equivocal nuclear changes, so-called well-differentiated tumor of uncertain malignant potential: a morphological, immunohistochemical, and molecular appraisal. Cancer Sci. 2011;102:288–94. doi: 10.1111/j.1349-7006.2010.01769.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kakudo K, Kameyama K, Miyauchi A, Nakamura H. Introducing the reporting system for thyroid fine-needle aspiration cytology according to the new guidelines of the Japan Thyroid Association. Endocr J. 2014;61:539–52. doi: 10.1507/endocrj.ej13-0494. [DOI] [PubMed] [Google Scholar]
- 10.Kakudo K, Kameyama K, Hirokawa M, Katoh R, Nakamura H. Subclassification of follicular neoplasms recommended by the Japan thyroid association reporting system of thyroid cytology. Int J Endocrinol. 2015;2015:938305. doi: 10.1155/2015/938305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kakudo K, Higuchi M, Hirokawa M, Satoh S, Jung CK, Bychkov A. Thyroid FNA cytology in Asian practice: active surveillance for indeterminate thyroid nodules reduces overtreatment of thyroid carcinomas. Cytopathology. 2017;28:455–66. doi: 10.1111/cyt.12491. [DOI] [PubMed] [Google Scholar]
- 12.Bychkov A, Hirokawa M, Jung CK, et al. Low rate of noninvasive follicular thyroid neoplasm with papillary-like nuclear features in Asian practice. Thyroid. 2017;27:983–4. doi: 10.1089/thy.2017.0079. [DOI] [PubMed] [Google Scholar]
- 13.Vuong HG, Ngo HT, Bychkov A, et al. Differences in surgical resection rate and risk of malignancy in thyroid cytopathology practice between Western and Asian countries: a systematic review and meta-analysis. Cancer Cytopathol. 2020;128:238–49. doi: 10.1002/cncy.22228. [DOI] [PubMed] [Google Scholar]
- 14.Kakudo K. Asian and Western practice in thyroid pathology: similarities and differences. Gland Surg. 2020;9:1614–27. doi: 10.21037/gs-2019-catp-02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hirokawa M, Auger M, Jung CK, Callegari FM. Thyroid FNA cytology: the Eastern versus Western perspectives. Cancer Cytopathol. 2023;131:415–20. doi: 10.1002/cncy.22692. [DOI] [PubMed] [Google Scholar]
- 16.Kakudo K, Liu Z, Jung CK, Lai CR. Lessons learned from the diversity of thyroid nodule practice. Cancer Cytopathol. doi: 10.1002/cncy.22728. 2023 Jun 6 [Epub]. [DOI] [PubMed] [Google Scholar]
- 17.Bychkov A, Schubert M. Constant demand, patchy supply. Pathologist. 2023;88:18–27. [Google Scholar]
- 18.Reisch LM, Carney PA, Oster NV, et al. Medical malpractice concerns and defensive medicine: a nationwide survey of breast pathologists. Am J Clin Pathol. 2015;144:916–22. doi: 10.1309/AJCP80LYIMOOUJIF. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Labarge B, Walter V, Lengerich EJ, et al. Evidence of a positive association between malpractice climate and thyroid cancer incidence in the United States. PLoS One. 2018;13:e0199862. doi: 10.1371/journal.pone.0199862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kakudo K, Bychkov A, Abelardo A, Keelawat S, Kumarasinghe P. Malpractice climate is a key difference in thyroid pathology practice between North America and the rest of the world. Arch Pathol Lab Med. 2019;143:1171. doi: 10.5858/arpa.2019-0228-LE. [DOI] [PubMed] [Google Scholar]
- 21.Renshaw AA, Gould EW. Why there is the tendency to “overdiagnose” the follicular variant of papillary thyroid carcinoma. Am J Clin Pathol. 2002;117:19–21. doi: 10.1309/CJEU-XLQ7-UPVE-NWFV. [DOI] [PubMed] [Google Scholar]
- 22.Tallini G, Tuttle RM, Ghossein RA. The history of the follicular variant of papillary thyroid carcinoma. J Clin Endocrinol Metab. 2017;102:15–22. doi: 10.1210/jc.2016-2976. [DOI] [PubMed] [Google Scholar]
- 23.Nikiforov YE, Seethala RR, Tallini G, et al. Nomenclature revision for encapsulated follicular variant of papillary thyroid carcinoma: a paradigm shift to reduce overtreatment of indolent tumors. JAMA Oncol. 2016;2:1023–9. doi: 10.1001/jamaoncol.2016.0386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lee YS, Nam KH, Chung WY, et al. Practical management of well differentiated thyroid carcinoma in Korea. Endocr J. 2008;55:1015–24. doi: 10.1507/endocrj.k08e-188. [DOI] [PubMed] [Google Scholar]
- 25.Takami H, Ito Y, Okamoto T, Onoda N, Noguchi H, Yoshida A. Revisiting the guidelines issued by the Japanese Society of Thyroid Surgeons and Japan Association of Endocrine Surgeons: a gradual move towards consensus between Japanese and western practice in the management of thyroid carcinoma. World J Surg. 2014;38:2002–10. doi: 10.1007/s00268-014-2498-y. [DOI] [PubMed] [Google Scholar]
- 26.Ito Y, Onoda N, Okamoto T. The revised clinical practice guidelines on the management of thyroid tumors by the Japan Associations of Endocrine Surgeons: core questions and recommendations for treatments of thyroid cancer. Endocr J. 2020;67:669–717. doi: 10.1507/endocrj.EJ20-0025. [DOI] [PubMed] [Google Scholar]
- 27.Welch HG, Doherty GM. Saving thyroids: overtreatment of small papillary cancers. N Engl J Med. 2018;379:310–2. doi: 10.1056/NEJMp1804426. [DOI] [PubMed] [Google Scholar]
- 28.Ito Y, Uruno T, Nakano K, et al. An observation trial without surgical treatment in patients with papillary microcarcinoma of the thyroid. Thyroid. 2003;13:381–7. doi: 10.1089/105072503321669875. [DOI] [PubMed] [Google Scholar]
- 29.Castro MR, Morris JC, Ryder M, Brito JP, Hay ID. Most patients with a small papillary thyroid carcinoma enjoy an excellent prognosis and may be managed with minimally invasive therapy or active surveillance. Cancer. 2015;121:3364–5. doi: 10.1002/cncr.29468. [DOI] [PubMed] [Google Scholar]
- 30.Sugitani I, Ito Y, Miyauchi A, Imai T, Suzuki S. Active surveillance versus immediate surgery: questionnaire survey on the current treatment strategy for adult patients with low-risk papillary thyroid microcarcinoma in Japan. Thyroid. 2019;29:1563–71. doi: 10.1089/thy.2019.0211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sugitani I, Ito Y, Takeuchi D, et al. Indications and strategy for active surveillance of adult low-risk papillary thyroid microcarcinoma: consensus statements from the Japan Association of Endocrine Surgery Task Force on Management for Papillary Thyroid Microcarcinoma. Thyroid. 2021;31:183–92. doi: 10.1089/thy.2020.0330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ghossein R, Barletta JA, Bullock M, et al. Data set for reporting carcinoma of the thyroid: recommendations from the International Collaboration on Cancer Reporting. Hum Pathol. 2021;110:62–72. doi: 10.1016/j.humpath.2020.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shindo H, Kakudo K, Inomata K, et al. Additional tissue sampling trials did not change our thyroid practice. Cancers (Basel) 2021;13:1270. doi: 10.3390/cancers13061270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kakudo K, Li Y, Taniguchi E, Liu Z. Follicular neoplasms in the 4th edition WHO classification of endocrine organs. J Basic Clin Med. 2018;7:10–9. [Google Scholar]
- 35.Jung CK, Bychkov A, Song DE, et al. Molecular correlates and nuclear features of encapsulated follicular-patterned thyroid neoplasms. Endocrinol Metab (Seoul) 2021;36:123–33. doi: 10.3803/EnM.2020.860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chan J. Strict criteria should be applied in the diagnosis of encapsulated follicular variant of papillary thyroid carcinoma. Am J Clin Pathol. 2002;117:16–8. doi: 10.1309/P7QL-16KQ-QLF4-XW0M. [DOI] [PubMed] [Google Scholar]
- 37.Piana S, Frasoldati A, Di Felice E, Gardini G, Tallini G, Rosai J. Encapsulated well-differentiated follicular-patterned thyroid carcinomas do not play a significant role in the fatality rates from thyroid carcinoma. Am J Surg Pathol. 2010;34:868–72. doi: 10.1097/PAS.0b013e3181dbee07. [DOI] [PubMed] [Google Scholar]
- 38.Chetty R. A proposal for the classification of follicular-patterned neoplasms of the thyroid gland. Am J Surg Pathol. 2011;35:313. doi: 10.1097/PAS.0b013e318206eebb. [DOI] [PubMed] [Google Scholar]
- 39.Goffredo P, Cheung K, Roman SA, Sosa JA. Can minimally invasive follicular thyroid cancer be approached as a benign lesion?: a population-level analysis of survival among 1,200 patients. Ann Surg Oncol. 2013;20:767–72. doi: 10.1245/s10434-012-2697-4. [DOI] [PubMed] [Google Scholar]
- 40.Kakudo K. Different threshold of malignancy for RAS-like thyroid tumors causes significant differences in thyroid nodule practice. Cancers (Basel) 2022;14:812. doi: 10.3390/cancers14030812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kakudo K, El-Naggar AK, Hodak SP, et al. Noninvasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) in thyroid tumor classification. Pathol Int. 2018;68:327–33. doi: 10.1111/pin.12673. [DOI] [PubMed] [Google Scholar]
- 42.Lloyd RV, Osamura RY, Kloppel G, Rosai J. WHO classification of tumours of endocrine organs. 4th ed. Lyon: IARC Press; 2017. [Google Scholar]
- 43.Bai Y, Kakudo K, Jung CK. Updates in the pathologic classification of thyroid neoplasms: a review of the World Health Organization classification. Endocrinol Metab (Seoul) 2020;35:696–715. doi: 10.3803/EnM.2020.807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bychkov A, Keelawat S, Agarwal S, et al. Impact of non-invasive follicular thyroid neoplasm with papillary-like nuclear features on the Bethesda system for reporting thyroid cytopathology: a multiinstitutional study in five Asian countries. Pathology. 2018;50:411–7. doi: 10.1016/j.pathol.2017.11.088. [DOI] [PubMed] [Google Scholar]
- 45.Bychkov A, Jung CK, Liu Z, Kakudo K. Noninvasive follicular thyroid neoplasm with papillary-like nuclear features in Asian practice: perspectives for surgical pathology and cytopathology. Endocr Pathol. 2018;29:276–88. doi: 10.1007/s12022-018-9519-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Liu Z, Bychkov A, Jung CK, et al. Interobserver and intraobserver variation in the morphological evaluation of noninvasive follicular thyroid neoplasm with papillary-like nuclear features in Asian practice. Pathol Int. 2019;69:202–10. doi: 10.1111/pin.12779. [DOI] [PubMed] [Google Scholar]
- 47.Vuong HG, Tran TT, Bychkov A, et al. Clinical impact of non-Invasive follicular thyroid neoplasm with papillary-like nuclear features on the risk of malignancy in the Bethesda System for Reporting Thyroid Cytopathology: a meta-analysis of 14,153 resected thyroid nodules. Endocr Pract. 2019;25:491–502. doi: 10.4158/EP-2018-0506. [DOI] [PubMed] [Google Scholar]
- 48.Rana C, Vuong HG, Nguyen TQ, et al. The incidence of noninvasive follicular thyroid neoplasm with papillary-like nuclear features: a meta-analysis assessing worldwide impact of the reclassification. Thyroid. 2021;31:1502–13. doi: 10.1089/thy.2021.0158. [DOI] [PubMed] [Google Scholar]
- 49.Bychkov A, Kakudo K, Hong S. Current practices of thyroid fineneedle aspiration in Asia: a missing voice. J Pathol Transl Med. 2017;51:517–20. doi: 10.4132/jptm.2017.09.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Jung CK, Hong S, Bychkov A, Kakudo K. The use of fine-needle aspiration (FNA) cytology in patients with thyroid nodules in Asia: a brief overview of studies from the Working Group of Asian Thyroid FNA Cytology. J Pathol Transl Med. 2017;51:571–8. doi: 10.4132/jptm.2017.10.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Thompson LD. Ninety-four cases of encapsulated follicular variant of papillary thyroid carcinoma: a name change to Noninvasive Follicular Thyroid Neoplasm with Papillary-like Nuclear Features would help prevent overtreatment. Mod Pathol. 2016;29:698–707. doi: 10.1038/modpathol.2016.65. [DOI] [PubMed] [Google Scholar]
- 52.Kakudo K. Thyroid FNA cytology: differential diagnoses and pitfalls. 3rd ed. Singapore: Springer; 2023. [Google Scholar]
- 53.Hirokawa M, Suzuki A, Hashimoto Y, et al. Prevalence and diagnostic challenges of thyroid lymphoma: a multi-institutional study in non-Western countries. Endocr J. 2020;67:1085–91. doi: 10.1507/endocrj.EJ20-0202. [DOI] [PubMed] [Google Scholar]
- 54.Agarwal S, Bychkov A, Jung CK, et al. The prevalence and surgical outcomes of Hurthle cell lesions in FNAs of the thyroid: a multiinstitutional study in 6 Asian countries. Cancer Cytopathol. 2019;127:181–91. doi: 10.1002/cncy.22101. [DOI] [PubMed] [Google Scholar]
- 55.Liu CY, Bychkov A, Agarwal S, et al. Cytologic diagnosis of medullary thyroid carcinoma in the Asia-Pacific region. Diagn Cytopathol. 2021;49:60–9. doi: 10.1002/dc.24586. [DOI] [PubMed] [Google Scholar]
- 56.Liu CY, Chen CC, Bychkov A, et al. Constitutive cytomorphologic features of medullary thyroid carcinoma using different staining methods. Diagnostics (Basel) 2021;11:1396. doi: 10.3390/diagnostics11081396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Jung CK, Agarwal S, Hang JF, Lim DJ, Bychkov A, Mete O. Update on C-cell neuroendocrine neoplasm: prognostic and predictive histopathologic and molecular features of medullary thyroid carcinoma. Endocr Pathol. 2023;34:1–22. doi: 10.1007/s12022-023-09753-5. [DOI] [PubMed] [Google Scholar]
- 58.Zhu Y, Li Y, Jung CK, et al. Histopathologic assessment of capsular invasion in follicular thyroid neoplasms: an observer variation study. Endocr Pathol. 2020;31:132–40. doi: 10.1007/s12022-020-09620-7. [DOI] [PubMed] [Google Scholar]
- 59.Lai WA, Hang JF, Liu CY, et al. PAX8 expression in anaplastic thyroid carcinoma is less than those reported in early studies: a multiinstitutional study of 182 cases using the monoclonal antibody MRQ-50. Virchows Arch. 2020;476:431–7. doi: 10.1007/s00428-019-02708-4. [DOI] [PubMed] [Google Scholar]
- 60.Ngo TN, Le TTB, Le T, et al. Primary versus secondary anaplastic thyroid carcinoma: perspectives from multi-institutional and population-level data. Endocr Pathol. 2021;32:489–500. doi: 10.1007/s12022-021-09692-z. [DOI] [PubMed] [Google Scholar]
- 61.Ohba K, Mitsutake N, Matsuse M, et al. Encapsulated papillary thyroid tumor with delicate nuclear changes and a KRAS mutation as a possible novel subtype of borderline tumor. J Pathol Transl Med. 2019;53:136–41. doi: 10.4132/jptm.2018.12.07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Jung CK, Park SY, Kim JH, Kakudo K. New insights into classification and risk stratification of encapsulated thyroid tumors with a predominantly papillary architecture. J Pathol Transl Med. 2020;54:197–203. doi: 10.4132/jptm.2020.04.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Giani C, Torregrossa L, Ramone T, et al. Whole tumor capsule is prognostic of very good outcome in the classical variant of papillary thyroid cancer. J Clin Endocrinol Metab. 2021;106:e4072–83. doi: 10.1210/clinem/dgab396. [DOI] [PubMed] [Google Scholar]
- 64.Nguyen TP, Truong VT, Kakudo K, Vuong HG. The diversities in thyroid cytopathology practices among Asian countries using the Bethesda System for Reporting Thyroid Cytopathology. Gland Surg. 2020;9:1735–46. doi: 10.21037/gs-20-404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Vuong HG, Nguyen TP, Hassell LA, Jung CK. Diagnostic performances of the Afirma Gene Sequencing Classifier in comparison with the Gene Expression Classifier: a meta-analysis. Cancer Cytopathol. 2021;129:182–9. doi: 10.1002/cncy.22332. [DOI] [PubMed] [Google Scholar]
- 66.Ngo HT, Nguyen TP, Vu TH, et al. Impact of molecular testing on the management of indeterminate thyroid nodules among Western and Asian countries: a systematic review and meta-analysis. Endocr Pathol. 2021;32:269–79. doi: 10.1007/s12022-020-09643-0. [DOI] [PubMed] [Google Scholar]
- 67.Vuong HG, Suzuki A, Na HY, et al. Application of the Bethesda System for Reporting Thyroid Cytopathology in the pediatric population. Am J Clin Pathol. 2021;155:680–9. doi: 10.1093/ajcp/aqaa182. [DOI] [PubMed] [Google Scholar]
- 68.Vuong HG, Chung DG, Ngo LM, et al. The use of the Bethesda System for Reporting Thyroid Cytopathology in pediatric thyroid nodules: a meta-analysis. Thyroid. 2021;31:1203–11. doi: 10.1089/thy.2020.0702. [DOI] [PubMed] [Google Scholar]
- 69.Vuong HG, Jung CK, Kakudo K, Bychkov A. Response to Cherella et al. re: “the use of the Bethesda System for Reporting Thyroid Cytopathology in pediatric thyroid nodules: a meta-analysis”. Thyroid. 2021;31:1442–4. doi: 10.1089/thy.2021.0190. [DOI] [PubMed] [Google Scholar]
- 70.Wang YH, Bychkov A, Chakrabarti I, et al. Impact of the COVID-19 pandemic on cytology practice: an international survey in the Asia-Pacific region. Cancer Cytopathol. 2020;128:895–904. doi: 10.1002/cncy.22354. [DOI] [PubMed] [Google Scholar]
- 71.Vuong HG, Le HT, Le TT, Le T, Hassell L, Kakudo K. Clinicopathological significance of major fusion oncogenes in papillary thyroid carcinoma: an individual patient data meta-analysis. Pathol Res Pract. 2022;240:154180. doi: 10.1016/j.prp.2022.154180. [DOI] [PubMed] [Google Scholar]
- 72.Vuong HG, Le MK, Hassell L, Kondo T, Kakudo K. The differences in distant metastatic patterns and their corresponding survival between thyroid cancer subtypes. Head Neck. 2022;44:926–32. doi: 10.1002/hed.26987. [DOI] [PubMed] [Google Scholar]
- 73.Vuong HG, Nguyen TP, Ngo HT, Hassell L, Kakudo K. Malignant thyroid teratoma: an integrated analysis of case series/case reports. Endocr Relat Cancer. 2021;28:495–503. doi: 10.1530/ERC-21-0142. [DOI] [PubMed] [Google Scholar]
- 74.Le HT, Nguyen TP, Hirokawa M, et al. Primary thyroid mucoepidermoid carcinoma (MEC) is clinically, prognostically, and molecularly different from sclerosing MEC with eosinophilia: a multicenter and integrated study. Endocr Pathol. 2023;34:100–11. doi: 10.1007/s12022-022-09741-1. [DOI] [PubMed] [Google Scholar]
- 75.LiVolsi VA, Asa SL. The demise of follicular carcinoma of the thyroid gland. Thyroid. 1994;4:233–6. doi: 10.1089/thy.1994.4.233. [DOI] [PubMed] [Google Scholar]
- 76.Cipriani NA, Nagar S, Kaplan SP, et al. Follicular thyroid carcinoma: how have histologic diagnoses changed in the last half-century and what are the prognostic implications? Thyroid. 2015;25:1209–16. doi: 10.1089/thy.2015.0297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kakudo K. Non-invasive encapsulated/well-circumscribed follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) and precursor thyroid tumors. J Basic Clin Med. 2017;6:1–2. [Google Scholar]
- 78.Kakudo K. Unsettled issues in non-invasive encapsulated/wellcircumscribed follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) and precursor thyroid tumors. J Basic Clin Med. 2017;6:3–7. [Google Scholar]
- 79.LiVolsi VA, Baloch ZW. Coming to terms with diagnosis “non-invasive follicular neoplasm with papillary like nuclear features (NIFTP)’: practice changer in endocrine pathology. J Basic Clin Med. 2017;6:8–13. [Google Scholar]
- 80.Kakudo K, Liu Z, Satoh S, Higuchi M, Hirokawa M. Non-invasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP): diagnosis and differential diagnoses. J Basic Clin Med. 2017;6:14–21. [Google Scholar]
- 81.Liu Z, Song Y, Han B, Zhang X, Su P, Cui X. Non-invasive follicular thyroid neoplasm with papillary-like nuclear features and the practice in Qilu Hospital of Shandong University, China. J Basic Clin Med. 2017;6:22–5. [Google Scholar]
- 82.Jung CK, Kim C. Effect of lowering the diagnostic threshold for encapsulated follicular variant of papillary thyroid carcinoma on the prevalence of non-invasive follicular thyroid neoplasm with papillary-like nuclear features: a single-institution experience in Korea. J Basic Clin Med. 2017;6:26–8. [Google Scholar]
- 83.Pusztaszeri MP, Triponez F, Meyer P, Sadowski SM. Noninvasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP): report of an institutional experience with 86 cases. J Basic Clin Med. 2017;6:29–35. [Google Scholar]
- 84.Rossi ED. NIFTP diagnosis: roses and thorns for cytopathologists and histopathologists. J Basic Clin Med. 2017;6:36–7. [Google Scholar]
- 85.Canberk S, Baloch ZW, Ince U, Schmitt F. Diagnosis of non-invasive follicular tumor with papillary-like nuclear features (NIFTP): a practice changer for thyroid fine-needle aspiration interpretation. J Basic Clin Med. 2017;6:38–43. [Google Scholar]
- 86.Maletta F, Volante M, Papotti M. Experience on NIFTP cytology, with a mini meta-analysis of the literature. J Basic Clin Med. 2017;6:44–50. [Google Scholar]
- 87.Ng D, Can NT, Ma ZV, van Zante A, Ljung BM, Khanafshar E. Cytomorphologic features of noninvasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP): a comparison with infiltrative follicular variant of papillary thyroid carcinoma. J Basic Clin Med. 2017;1:51–6. [Google Scholar]
- 88.Saglietti C, Bongiovanni M. The value of cytological examination in the diagnosis of noninvasive thyroid neoplasm with papillarylike nuclear features (NIFTP) J Basic Clin Med. 2017;6:57–60. [Google Scholar]
- 89.Wu HH. The impact of NIFTP on FNA cytology: can we still diagnose papillary thyroid carcinoma? J Basic Clin Med. 2017;6:61–2. [Google Scholar]
- 90.Yang GC. Ultrasound is cytopathologist’s best friend in the era of noninvasive follicular thyroid neoplasm with papillary-like nuclear features. J Basic Clin Med. 2017;6:65–7. [Google Scholar]
- 91.Poller DN, Johnson SJ, Stephenson TJ. Diagnosis of NIFTP in the UK. J Basic Clin Med. 2017;6:63–4. [Google Scholar]
- 92.Cha YJ, Pyo JY, Hong S, et al. Thyroid fine-needle aspiration cytology practice in Korea. J Pathol Transl Med. 2017;51:521–7. doi: 10.4132/jptm.2017.09.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Liu Z, Liu D, Ma B, et al. History and practice of thyroid fine-needle aspiration in China, based on retrospective study of the practice in Shandong University Qilu Hospital. J Pathol Transl Med. 2017;51:528–32. doi: 10.4132/jptm.2017.09.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Agarwal S, Jain D. Thyroid cytology in India: contemporary review and meta-analysis. J Pathol Transl Med. 2017;51:533–47. doi: 10.4132/jptm.2017.08.04. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Satoh S, Yamashita H, Kakudo K. Thyroid cytology: the Japanese system and experience at Yamashita Thyroid Hospital. J Pathol Transl Med. 2017;51:548–54. doi: 10.4132/jptm.2017.09.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Abelardo AD. Thyroid fine-needle aspiration practice in the Philippines. J Pathol Transl Med. 2017;51:555–9. doi: 10.4132/jptm.2017.07.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Hang JF, Hsu CY, Lai CR. Thyroid fine-needle aspiration in Taiwan: the history and current practice. J Pathol Transl Med. 2017;51:560–4. doi: 10.4132/jptm.2017.09.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Keelawat S, Rangdaeng S, Koonmee S, Jitpasutham T, Bychkov A. Current status of thyroid fine-needle aspiration practice in Thailand. J Pathol Transl Med. 2017;51:565–70. doi: 10.4132/jptm.2017.08.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Ohori NP. Molecular testing and thyroid nodule management in North America. Gland Surg. 2020;9:1628–38. doi: 10.21037/gs-2019-catp-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Kumarasinghe MP. Standardisation of thyroid cytology terminology and practice: are modifications necessary?: a narrative review. Gland Surg. 2020;9:1639–47. doi: 10.21037/gs-2019-catp-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Poller DN. Litigation in thyroid cytology and histopathology in England: a very brief overview. Gland Surg. 2020;9:1648–52. doi: 10.21037/gs.2020.02.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Hirokawa M, Suzuki A, Higuchi M, et al. The Japanese reporting system for thyroid aspiration cytology 2019 (JRSTAC2019) Gland Surg. 2020;9:1653–62. doi: 10.21037/gs-2019-catp-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Ito Y, Miyauchi A. Active surveillance of low-risk papillary thyroid microcarcinomas. Gland Surg. 2020;9:1663–73. doi: 10.21037/gs-2019-catp-03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Zhu Y, Wu H, Huang B, Shen X, Cai G, Gu X. BRAF(V600E) mutation combined with American College of Radiology thyroid imaging report and data system significantly changes surgical resection rate and risk of malignancy in thyroid cytopathology practice. Gland Surg. 2020;9:1674–84. doi: 10.21037/gs-20-535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Pusztaszeri MP, Tamilia M, Payne RJ. Active surveillance for lowrisk small papillary thyroid cancer in North American countries: past, present and future (bridging the gap between North American and Asian practices) Gland Surg. 2020;9:1685–97. doi: 10.21037/gs-20-389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Okamoto T, Omi Y, Yoshida Y, Horiuchi K, Abe K. Radioactive iodine treatment of papillary thyroid carcinoma in Japan. Gland Surg. 2020;9:1698–707. doi: 10.21037/gs-20-378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Michael CW, Kameyama K, Kitagawa W, Azar N. Rapid on-site evaluation (ROSE) for fine needle aspiration of thyroid: benefits, challenges and innovative solutions. Gland Surg. 2020;9:1708–15. doi: 10.21037/gs-2019-catp-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Liu Z, Sui S, Su P, et al. The effect of implementing pre-surgical ultrasound-guided fine-needle aspiration biopsy on thyroid surgery, a 6-year interrupted time series analysis in Qilu Hospital of Shandong University. Gland Surg. 2020;9:1716–23. doi: 10.21037/gs-20-348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Canberk S. Precursor and borderline lesions of the thyroid (indolent lesions of epithelial origin): from theory to practice. Gland Surg. 2020;9:1724–34. doi: 10.21037/gs-20-429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Oo ZP, Hlaing AM, Kyi KC, Fukuoka J, Bychkov A. An overview of thyroid fine-needle aspiration practice in Myanmar. Gland Surg. 2020;9:1747–53. doi: 10.21037/gs-20-414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Tangnuntachai N, Rangdaeng S, Koonmee S, Tangjaturonrasme N, Keelawat S. Pathological practice and management of thyroid nodules: a Thai perspective. Gland Surg. 2020;9:1754–63. doi: 10.21037/gs-20-395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Ooi LY, Nga ME. Atypia of undetermined significance/follicular lesion of undetermined significance: Asian vs. non-Asian practice, and the Singapore experience. Gland Surg. 2020;9:1764–87. doi: 10.21037/gs-20-555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Abelardo AD, Sotalbo KCJ. Clinical management of thyroid aspirates diagnosed as atypia of undetermined significance in the Philippines. Gland Surg. 2020;9:1788–96. doi: 10.21037/gs-20-426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Guleria P, Mani K, Agarwal S. Indian experience of AUS/FLUS diagnosis: is it different from rest of Asia and the West?: A systematic review and meta-analysis. Gland Surg. 2020;9:1797–812. doi: 10.21037/gs-20-392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Odate T, Oishi N, Vuong HG, Mochizuki K, Kondo T. Genetic differences in follicular thyroid carcinoma between Asian and Western countries: a systematic review. Gland Surg. 2020;9:1813–26. doi: 10.21037/gs-20-356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Nakra T, Jain D, Agarwal S. Thyroid lymphoproliferative lesions in Asia. Gland Surg. 2020;9:1827–37. doi: 10.21037/gs-20-432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Li Y, Inomata K, Nishihara E, Kakudo K. IgG4 thyroiditis in the Asian population. Gland Surg. 2020;9:1838–46. doi: 10.21037/gs-20-417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Bai Y, Niu D, Yao Q, Lin D, Kakudo K. Updates in the advances of sporadic medullary thyroid carcinoma: from the molecules to the clinic. Gland Surg. 2020;9:1847–56. doi: 10.21037/gs-2019-catp-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Jung CK, Lee S, Bae JS, Lim DJ. Late-onset distant metastases confer poor prognosis in patients with well-differentiated thyroid cancer. Gland Surg. 2020;9:1857–66. doi: 10.21037/gs-20-416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Choden S, Keelawat S, Jung CK, Bychkov A. An affordable immunohistochemical approach to estimate the prevalence of BRAF (V600E) in large cohort studies: establishing the baseline rate of BRAF mutation in an institutional series of papillary thyroid carcinoma from Thailand. Gland Surg. 2020;9:1867–77. doi: 10.21037/gs-20-388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Rashid FA, Munkhdelger J, Fukuoka J, Bychkov A. Prevalence of BRAF(V600E) mutation in Asian series of papillary thyroid carcinoma: a contemporary systematic review. Gland Surg. 2020;9:1878–900. doi: 10.21037/gs-20-430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Kakudo K, Pusztaszeri M, Bongiovanni M. Factors impacting thyroid fine needle aspiration cytology and algorithm for cytological diagnosis in the Japanese system for reporting FNA cytology. In: Kakudo K, editor. Thyroid FNA cytology, differential diagnoses and pitfalls. 2nd ed. Singapore: Springer Singapore; 2019. pp. 19–26. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
A list of publications by the Asian Thyroid Working members from 2017 to 2023