Abstract
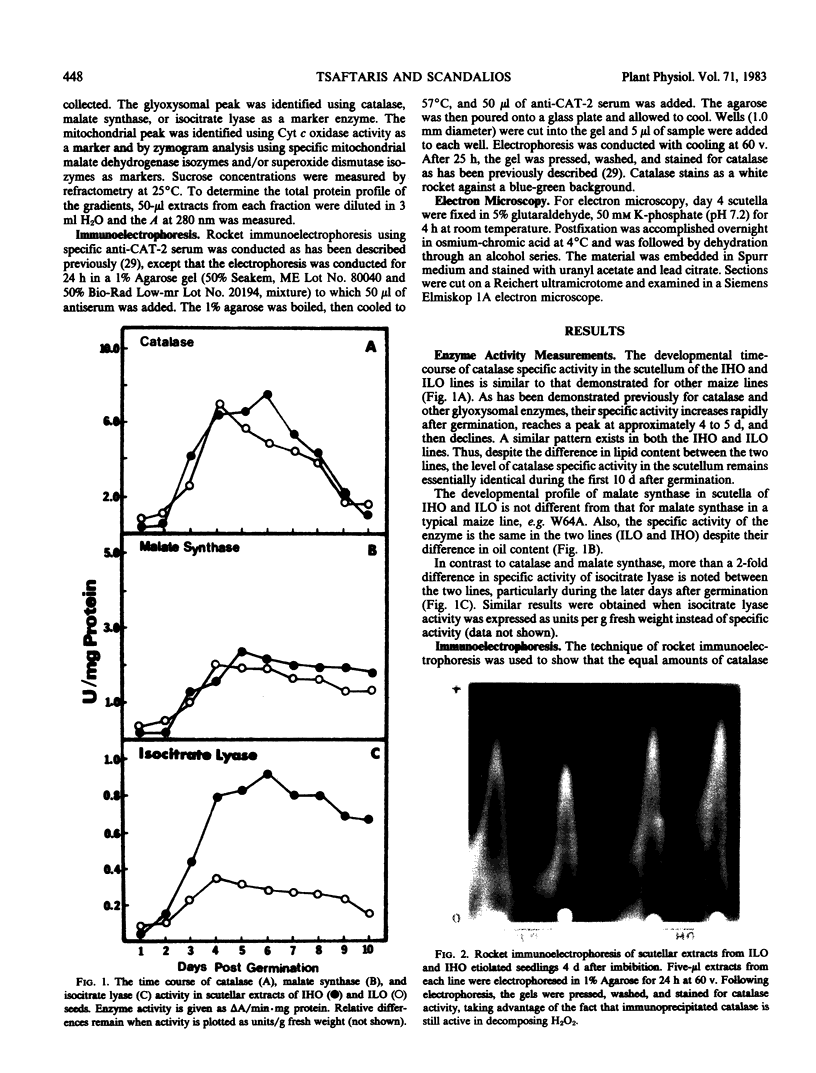
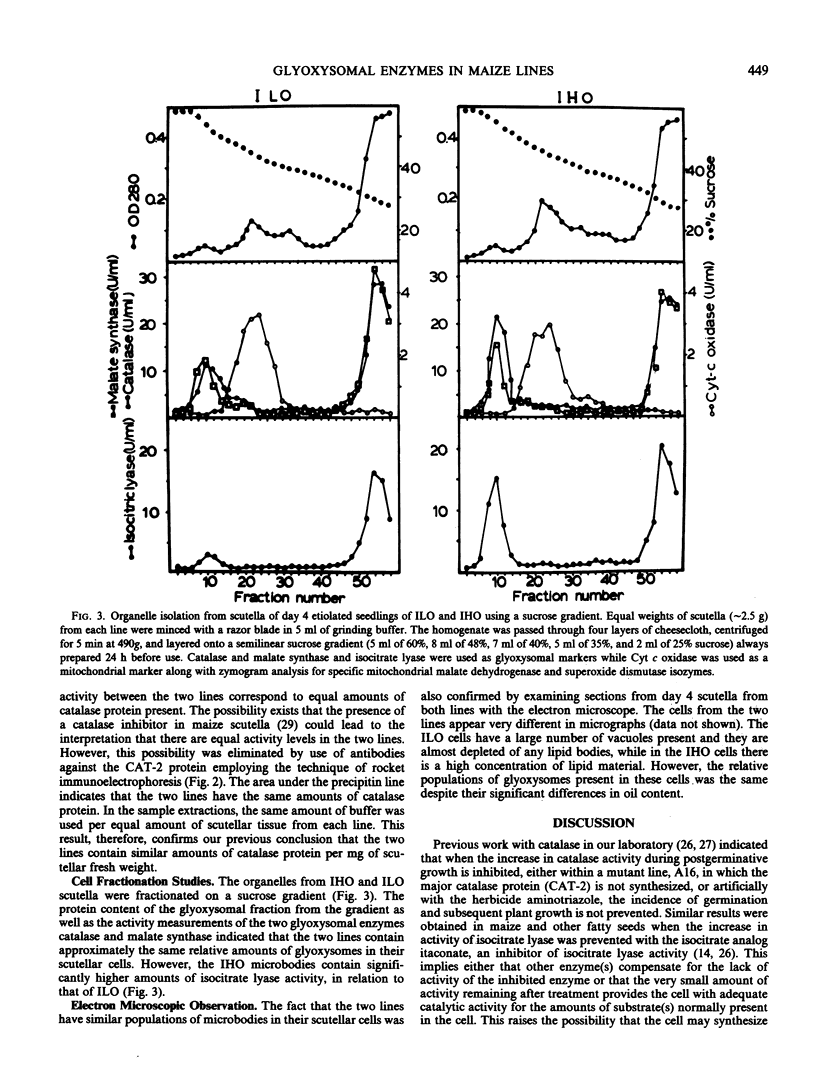
The developmental profile of the glyoxysomes and their component enzymes catalase, malate synthase, and isocitrate lyase were compared in the scutellum of two maize (Zea mays) lines, Illinois High Oil (IHO, approximately 20% lipid content) and Illinois Low Oil (ILO, less than 0.5% lipid content). The microbodies participate in the catabolism of the seed lipids and are responsible for leading the catabolic products (acetyl-Coenzyme A) into gluconeogenesis. The aim of this study was to determine whether changes in lipid content of the seed resulted in changes in the levels of the glyoxysomal enzymes. Enzyme activity measurements, immunological measurements (in the case of catalase), cell fractionation studies, and electron microscopic observations indicated that the IHO and ILO lines contain similar populations of glyoxysomes and exhibit similar catalase and malate synthase specific activities, despite the significant difference (40-fold) in their lipid content. Only the specific activity of isocitrate lyase was higher (2-fold higher) in the IHO seeds as compared to the ILO.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BEERS R. F., Jr, SIZER I. W. A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. J Biol Chem. 1952 Mar;195(1):133–140. [PubMed] [Google Scholar]
- Becker W. M., Leaver C. J., Weir E. M., Riezman H. Regulation of Glyoxysomal Enzymes during Germination of Cucumber: I. Developmental Changes in Cotyledonary Protein, RNA, and Enzyme Activities during Germination. Plant Physiol. 1978 Oct;62(4):542–549. doi: 10.1104/pp.62.4.542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beevers H. Glyoxysomes of castor bean endosperm and their relation to gluconeogenesis. Ann N Y Acad Sci. 1969 Dec 19;168(2):313–324. doi: 10.1111/j.1749-6632.1969.tb43118.x. [DOI] [PubMed] [Google Scholar]
- Breidenbach R. W., Beevers H. Association of the glyoxylate cycle enzymes in a novel subcellular particle from castor bean endosperm. Biochem Biophys Res Commun. 1967 May 25;27(4):462–469. doi: 10.1016/s0006-291x(67)80007-x. [DOI] [PubMed] [Google Scholar]
- Choinski J. S., Trelease R. N. Control of Enzyme Activities in Cotton Cotyledons during Maturation and Germination: II. Glyoxysomal Enzyme Development in Embryos. Plant Physiol. 1978 Jul;62(1):141–145. doi: 10.1104/pp.62.1.141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerhardt B. P., Beevers H. Developmental studies on glyoxysomes in Ricinus endosperm. J Cell Biol. 1970 Jan;44(1):94–102. doi: 10.1083/jcb.44.1.94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gientka-Rychter A., Cherry J. H. De Novo Synthesis of Isocitritase in Peanut (Arachis hypogaea L.) Cotyledons. Plant Physiol. 1968 Apr;43(4):653–659. doi: 10.1104/pp.43.4.653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan F. R., McFadden B. A. Enzyme Profiles in Seedling Development and the Effect of Itaconate, an Isocitrate Lyase-directed Reagent. Plant Physiol. 1979 Aug;64(2):228–231. doi: 10.1104/pp.64.2.228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Longo C. P. Evidence for de novo synthesis of isocitratase and malate synthesis in germinating peanut cotyledons. Plant Physiol. 1968 Apr;43(4):660–664. doi: 10.1104/pp.43.4.660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miernyk J. A., Trelease R. N. Control of Enzyme Activities in Cotton Cotyledons during Maturation and Germination : IV. beta-OXIDATION. Plant Physiol. 1981 Feb;67(2):341–346. doi: 10.1104/pp.67.2.341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quail P. H., Scandalios J. G. Turnover of genetically defined catalase isozymes in maize. Proc Natl Acad Sci U S A. 1971 Jul;68(7):1402–1406. doi: 10.1073/pnas.68.7.1402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scandalios J. G., Chang D. Y., McMillin D. E., Tsaftaris A., Moll R. H. Genetic regulation of the catalase developmental program in maize scutellum: Identification of a temporal regulatory gene. Proc Natl Acad Sci U S A. 1980 Sep;77(9):5360–5364. doi: 10.1073/pnas.77.9.5360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trelease R. N., Becker W. M., Gruber P. J., Newcomb E. H. Microbodies (Glyoxysomes and Peroxisomes) in Cucumber Cotyledons: Correlative Biochemical and Ultrastructural Study in Light- and Dark-grown Seedlings. Plant Physiol. 1971 Oct;48(4):461–475. doi: 10.1104/pp.48.4.461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vigil E. L. Cytochemical and developmental changes in microbodies (glyoxysomes) and related organelles of castor bean endosperm. J Cell Biol. 1970 Sep;46(3):435–454. doi: 10.1083/jcb.46.3.435. [DOI] [PMC free article] [PubMed] [Google Scholar]