Abstract
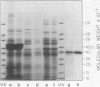
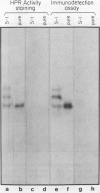
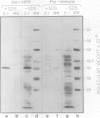
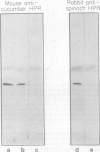
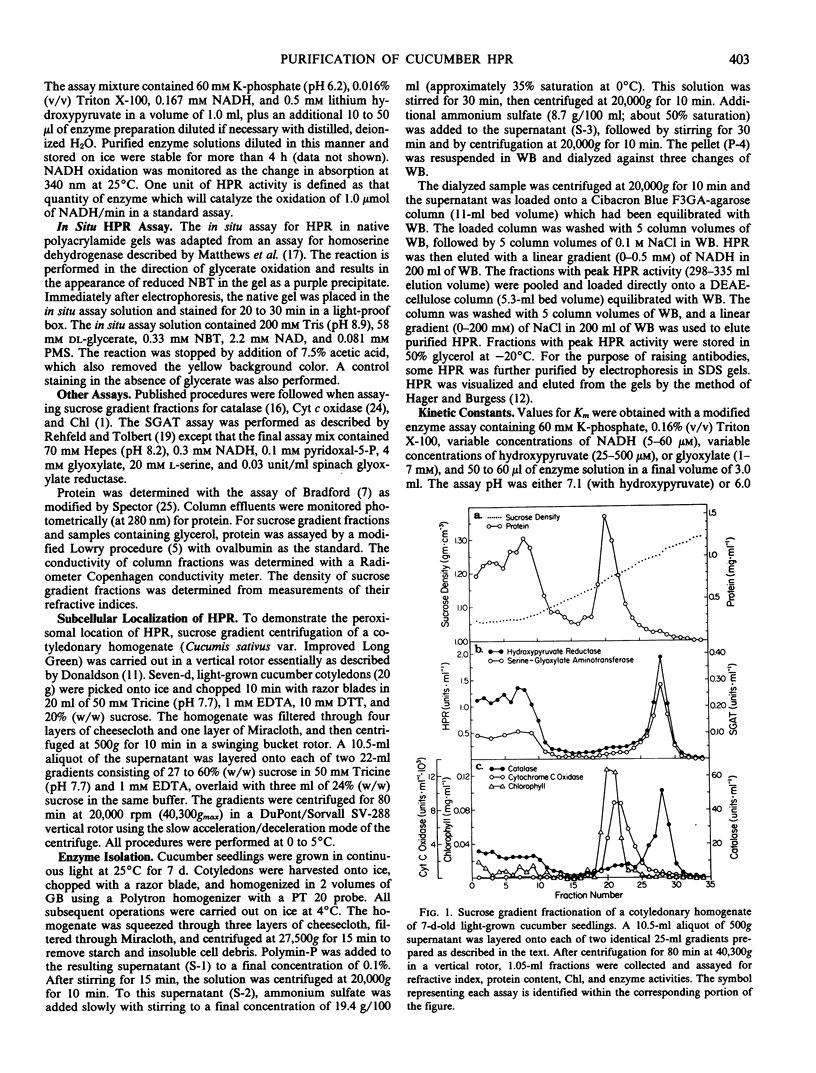
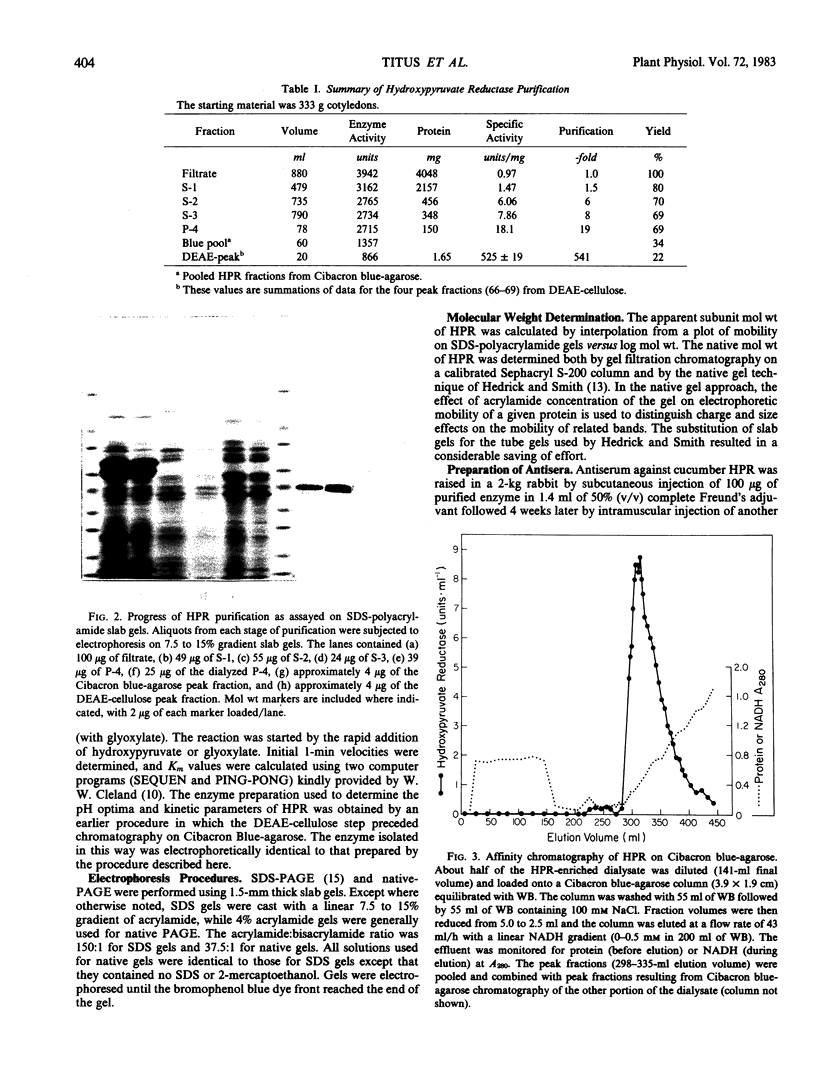
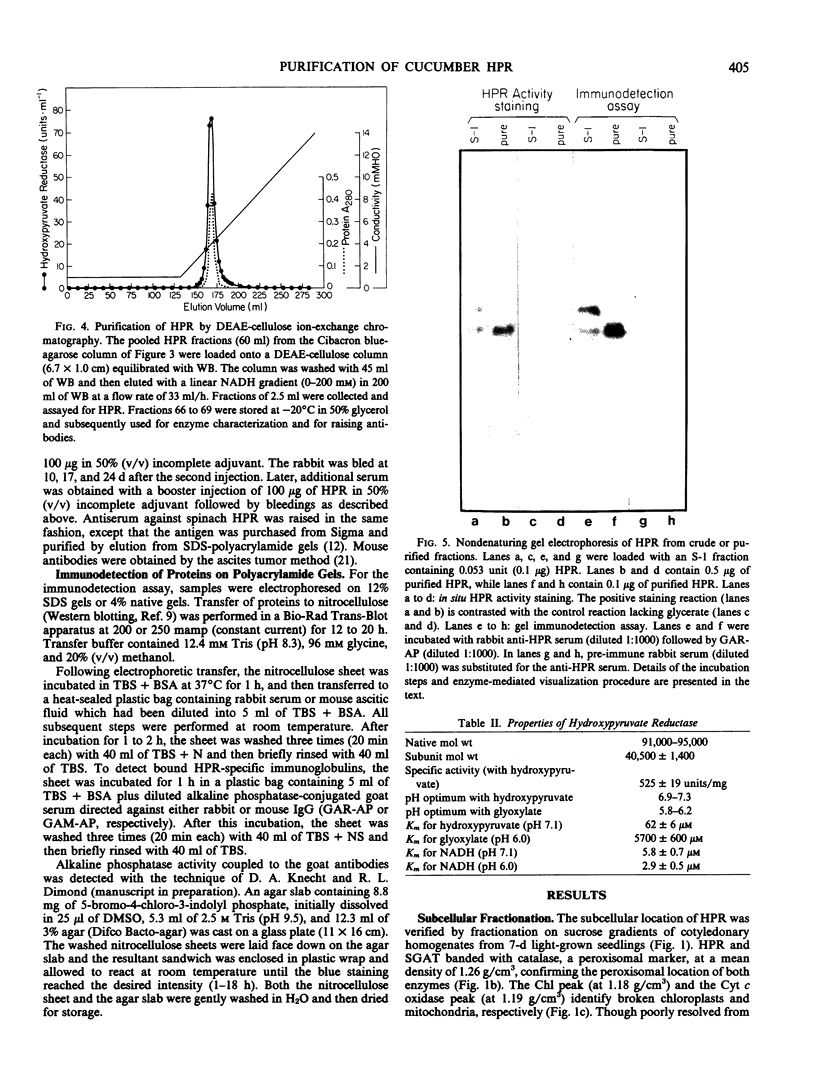
Hydroxypyruvate reductase (HPR), a marker enzyme of peroxisomes, has been purified to homogeneity from cotyledons of light-grown cucumber seedlings (Cucumis sativus var. Improved Long Green). In addition, the peroxisomal location of both HPR and serine-glyoxylate aminotransferase has been confirmed in cucumber cotyledons. The isolation procedure involved Polymin-P precipitation, a two-step precipitation with ammonium sulfate (35 and 50% saturation), affinity chromatography on Cibacron Blueagarose, and ion-exchange chromatography on DEAE-cellulose. HPR was purified 541-fold to a final specific activity of 525 ± 19 micromoles per minute per milligram of protein. Enzyme homogeneity was established by native and sodium dodecyl sulfate-polyacrylamide gel electrophoresis. The native molecular weight was 91 to 95 kilodaltons, approximately double the apparent subunit molecular weight of 40,500 ± 1,400. With hydroxypyruvate as substrate, the pH optimum was 7.1 and Km values were 62 ± 6 and 5.8 ± 0.7 micromolar for hydroxypyruvate and NADH, respectively. With glyoxylate as substrate, the pH optimum was 6.0, and the Km values for glyoxylate and NADH were 5700 ± 600 and 2.9 ± 0.5 micromolar, respectively. Antibodies to HPR were raised in mice (by the ascites tumor method) and in rabbits, and their monospecificity was demonstrated by a modified Western blot immunodetection technique.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arnon D. I. COPPER ENZYMES IN ISOLATED CHLOROPLASTS. POLYPHENOLOXIDASE IN BETA VULGARIS. Plant Physiol. 1949 Jan;24(1):1–15. doi: 10.1104/pp.24.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becker W. M., Leaver C. J., Weir E. M., Riezman H. Regulation of Glyoxysomal Enzymes during Germination of Cucumber: I. Developmental Changes in Cotyledonary Protein, RNA, and Enzyme Activities during Germination. Plant Physiol. 1978 Oct;62(4):542–549. doi: 10.1104/pp.62.4.542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bensadoun A., Weinstein D. Assay of proteins in the presence of interfering materials. Anal Biochem. 1976 Jan;70(1):241–250. doi: 10.1016/s0003-2697(76)80064-4. [DOI] [PubMed] [Google Scholar]
- Betsche T., Gerhardt B. Apparent Catalase Synthesis in Sunflower Cotyledons during the Change in Microbody Function: A Mathematical Approach for the Quantitative Evaluation of Density-labeling Data. Plant Physiol. 1978 Oct;62(4):590–597. doi: 10.1104/pp.62.4.590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Burke J. J., Trelease R. N. Cytochemical demonstration of malate synthase and glycolate oxidase in microbodies of cucumber cotyledons. Plant Physiol. 1975 Nov;56(5):710–717. doi: 10.1104/pp.56.5.710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Cleland W. W. Statistical analysis of enzyme kinetic data. Methods Enzymol. 1979;63:103–138. doi: 10.1016/0076-6879(79)63008-2. [DOI] [PubMed] [Google Scholar]
- Donaldson R. P. Nicotinamide cofactors (NAD and NADP) in glyoxysomes, mitochondria, and plastids isolated from castor bean endosperm. Arch Biochem Biophys. 1982 Apr 15;215(1):274–279. doi: 10.1016/0003-9861(82)90305-8. [DOI] [PubMed] [Google Scholar]
- Hager D. A., Burgess R. R. Elution of proteins from sodium dodecyl sulfate-polyacrylamide gels, removal of sodium dodecyl sulfate, and renaturation of enzymatic activity: results with sigma subunit of Escherichia coli RNA polymerase, wheat germ DNA topoisomerase, and other enzymes. Anal Biochem. 1980 Nov 15;109(1):76–86. doi: 10.1016/0003-2697(80)90013-5. [DOI] [PubMed] [Google Scholar]
- Hedrick J. L., Smith A. J. Size and charge isomer separation and estimation of molecular weights of proteins by disc gel electrophoresis. Arch Biochem Biophys. 1968 Jul;126(1):155–164. doi: 10.1016/0003-9861(68)90569-9. [DOI] [PubMed] [Google Scholar]
- Kohn L. D., Warren W. A. The kinetic properties of spinach leaf glyoxylic acid reductase. J Biol Chem. 1970 Aug 10;245(15):3831–3839. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lamb J. E., Riezman H., Becker W. M. Regulation of Glyoxysomal Enzymes during Germination of Cucumber: 2. Isolation and Immunological Detection of Isocitrate Lyase and Catalase. Plant Physiol. 1978 Nov;62(5):754–760. doi: 10.1104/pp.62.5.754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthews B. F., Gurman A. W., Bryan J. K. Changes in Enzyme Regulation during Growth of Maize: I. Progressive Desensitization of Homoserine Dehydrogenase during Seedling Growth. Plant Physiol. 1975 Jun;55(6):991–998. doi: 10.1104/pp.55.6.991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noguchi T., Fujiwara S. Development of glutamate:glyoxylate aminotransferase in the cotyledons of cucumber (Cucumis sativus) seedlings. Biochem J. 1982 Jan 1;201(1):209–214. doi: 10.1042/bj2010209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rehfeld D. W., Tolbert N. E. Aminotransferases in peroxisomes from spinach leaves. J Biol Chem. 1972 Aug 10;247(15):4803–4811. [PubMed] [Google Scholar]
- Riezman H., Weir E. M., Leaver C. J., Titus D. E., Becker W. M. Regulation of Glyoxysomal Enzymes during Germination of Cucumber: 3. IN VITRO TRANSLATION AND CHARACTERIZATION OF FOUR GLYOXYSOMAL ENZYMES. Plant Physiol. 1980 Jan;65(1):40–46. doi: 10.1104/pp.65.1.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnarrenberger C., Oeser A., Tolbert N. E. Development of Microbodies in Sunflower Cotyledons and Castor Bean Endosperm during Germination. Plant Physiol. 1971 Nov;48(5):566–574. doi: 10.1104/pp.48.5.566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spector T. Refinement of the coomassie blue method of protein quantitation. A simple and linear spectrophotometric assay for less than or equal to 0.5 to 50 microgram of protein. Anal Biochem. 1978 May;86(1):142–146. doi: 10.1016/0003-2697(78)90327-5. [DOI] [PubMed] [Google Scholar]
- Tolbert N. E. Metabolic pathways in peroxisomes and glyoxysomes. Annu Rev Biochem. 1981;50:133–157. doi: 10.1146/annurev.bi.50.070181.001025. [DOI] [PubMed] [Google Scholar]
- Trelease R. N., Becker W. M., Gruber P. J., Newcomb E. H. Microbodies (Glyoxysomes and Peroxisomes) in Cucumber Cotyledons: Correlative Biochemical and Ultrastructural Study in Light- and Dark-grown Seedlings. Plant Physiol. 1971 Oct;48(4):461–475. doi: 10.1104/pp.48.4.461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weir E. M., Riezman H., Grienenberger J. M., Becker W. M., Leaver C. J. Regulation of glyoxysomal enzymes during germination of cucumber. Temporal changes in translatable mRNAs for isocitrate lyase and malate synthase. Eur J Biochem. 1980 Dec;112(3):469–477. [PubMed] [Google Scholar]
- ZELITCH I. The isolation and action of crystalline glyoxylic acid reductase from tobacco leaves. J Biol Chem. 1955 Oct;216(2):553–575. [PubMed] [Google Scholar]