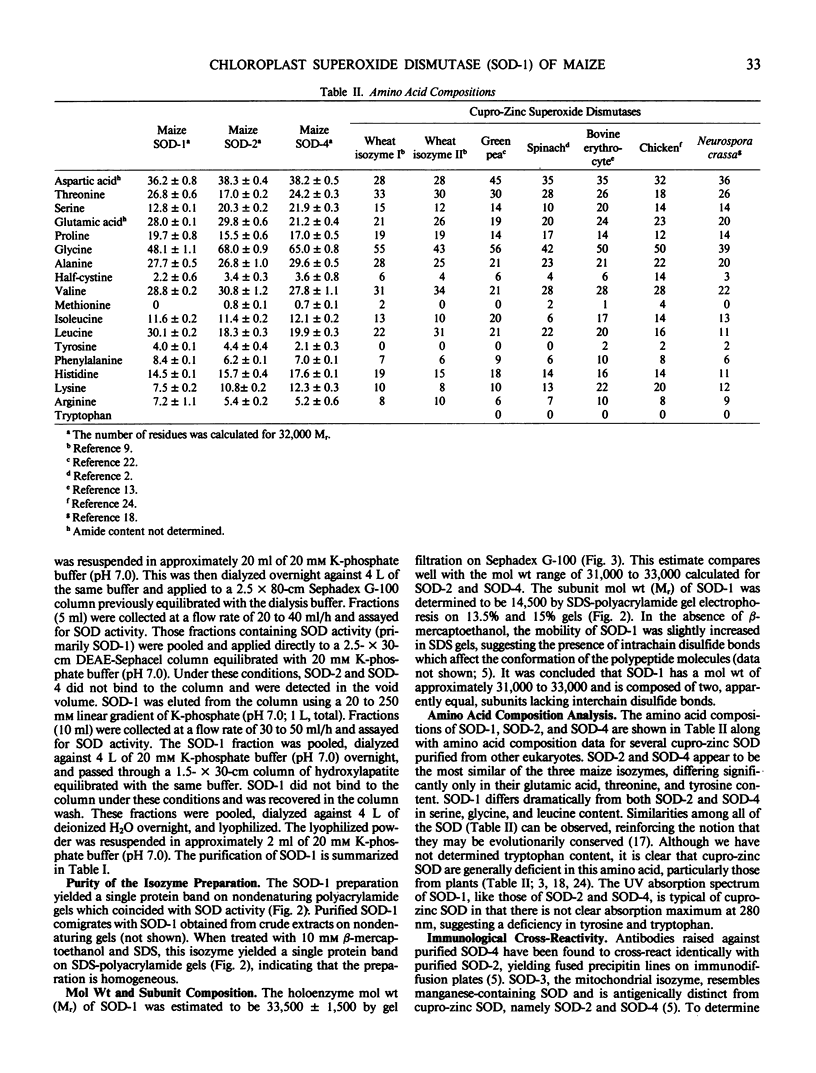
Abstract
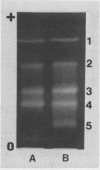
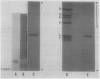
The chloroplast-associated form of superoxide dismutase from maize (Zea mays L.) (SOD-1) has been purified by a stepwise procedure consisting of (NH4)2SO4 fractionation, G-100 Sephadex gel filtration, DEAE-Sephacel chromatography, and hydroxylapatite chromatography. This procedure resulted in a single band on sodium dodecyl sulfate-polyacrylamide gels indicating that the preparation is homogeneous. The holoenzyme molecular weight was estimated at 31,000 to 33,000 by gel filtration. The subunit molecular weight of this dimeric protein was estimated at 14,500 on sodium dodecyl sulfate-polyacrylamide gels. Studies involving amino acid composition analysis, immunological cross-reactivity, in vitro subunit hybridizations, and H2O2 sensitivity indicate that SOD-1 differs significantly from SOD-2 and SOD-4, the other cupro-zinc forms of SOD from maize. The possible physiological role of SOD-1 within the chloroplast is discussed.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Asada K., Urano M., Takahashi M. Subcellular location of superoxide dismutase in spinach leaves and preparation and properties of crystalline spinach superoxide dismutase. Eur J Biochem. 1973 Jul 2;36(1):257–266. doi: 10.1111/j.1432-1033.1973.tb02908.x. [DOI] [PubMed] [Google Scholar]
- Baum J. A., Scandalios J. G. Isolation and characterization of the cytosolic and mitochondrial superoxide dismutases of maize. Arch Biochem Biophys. 1981 Feb;206(2):249–264. doi: 10.1016/0003-9861(81)90089-8. [DOI] [PubMed] [Google Scholar]
- Beauchamp C. O., Fridovich I. Isozymes of superoxide dismutase from wheat germ. Biochim Biophys Acta. 1973 Jul 12;317(1):50–64. doi: 10.1016/0005-2795(73)90198-0. [DOI] [PubMed] [Google Scholar]
- Beauchamp C., Fridovich I. Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem. 1971 Nov;44(1):276–287. doi: 10.1016/0003-2697(71)90370-8. [DOI] [PubMed] [Google Scholar]
- Fridovich I. Superoxide dismutases. Annu Rev Biochem. 1975;44:147–159. doi: 10.1146/annurev.bi.44.070175.001051. [DOI] [PubMed] [Google Scholar]
- Jackson C., Dench J., Moore A. L., Halliwell B., Foyer C. H., Hall D. O. Subcellular localisation and identification of superoxide dismutase in the leaves of higher plants. Eur J Biochem. 1978 Nov 15;91(2):339–344. doi: 10.1111/j.1432-1033.1978.tb12685.x. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lumsden J., Hall D. O. Soluble & membrane-bound superoxide dismutases in a blue-green algae (Spirulina)and spinach. Biochem Biophys Res Commun. 1974 May 7;58(1):35–41. doi: 10.1016/0006-291x(74)90887-0. [DOI] [PubMed] [Google Scholar]
- Misra H. P., Fridovich I. The purification and properties of superoxide dismutase from Neurospora crassa. J Biol Chem. 1972 Jun 10;247(11):3410–3414. [PubMed] [Google Scholar]
- Sawada Y., Oyama T., Yamazaki I. Preparation and physicochemical properties of green pea superoxide dismutase. Biochim Biophys Acta. 1972 May 12;268(2):305–312. doi: 10.1016/0005-2744(72)90325-7. [DOI] [PubMed] [Google Scholar]
- Weisiger R. A., Fridovich I. Superoxide dismutase. Organelle specificity. J Biol Chem. 1973 May 25;248(10):3582–3592. [PubMed] [Google Scholar]