Abstract
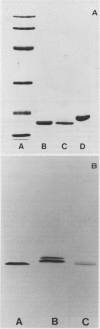
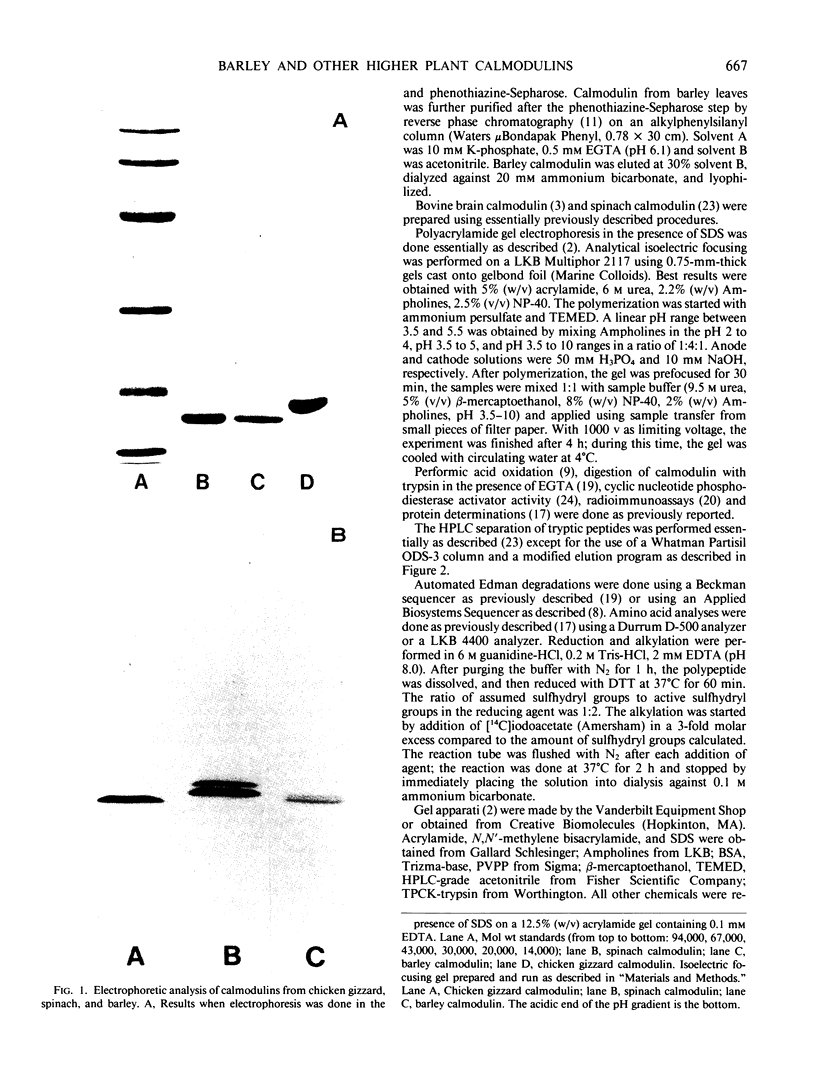
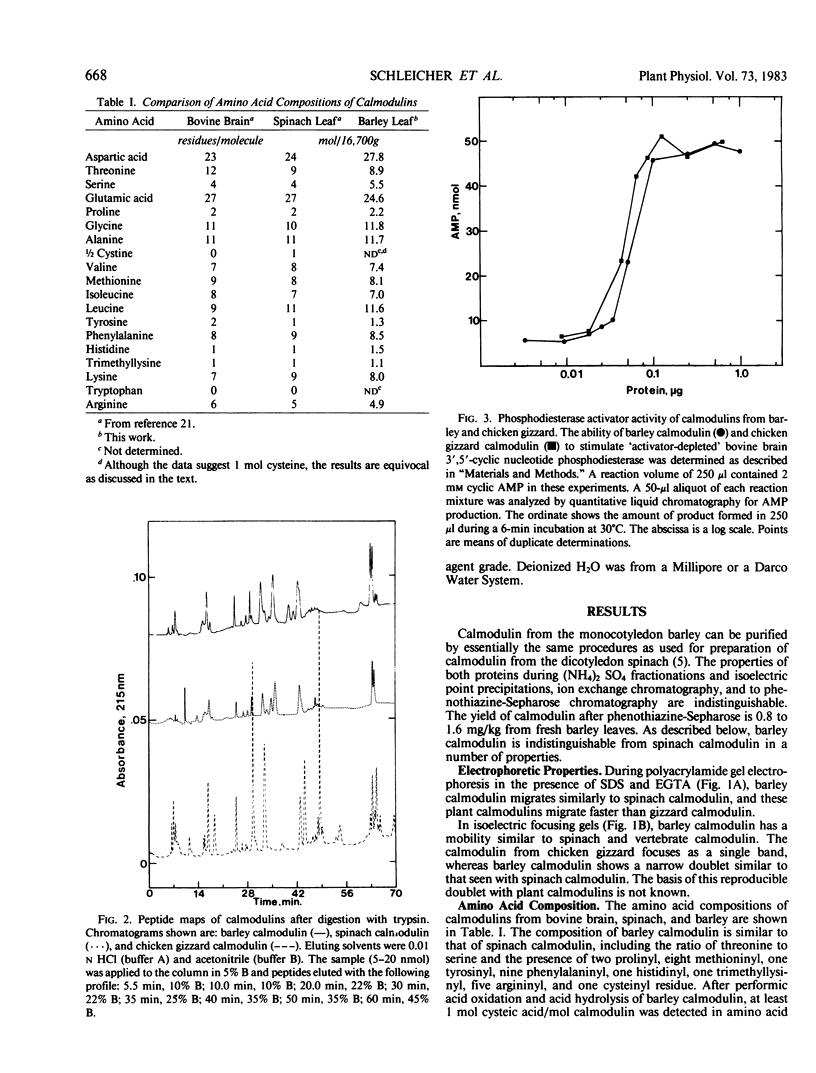
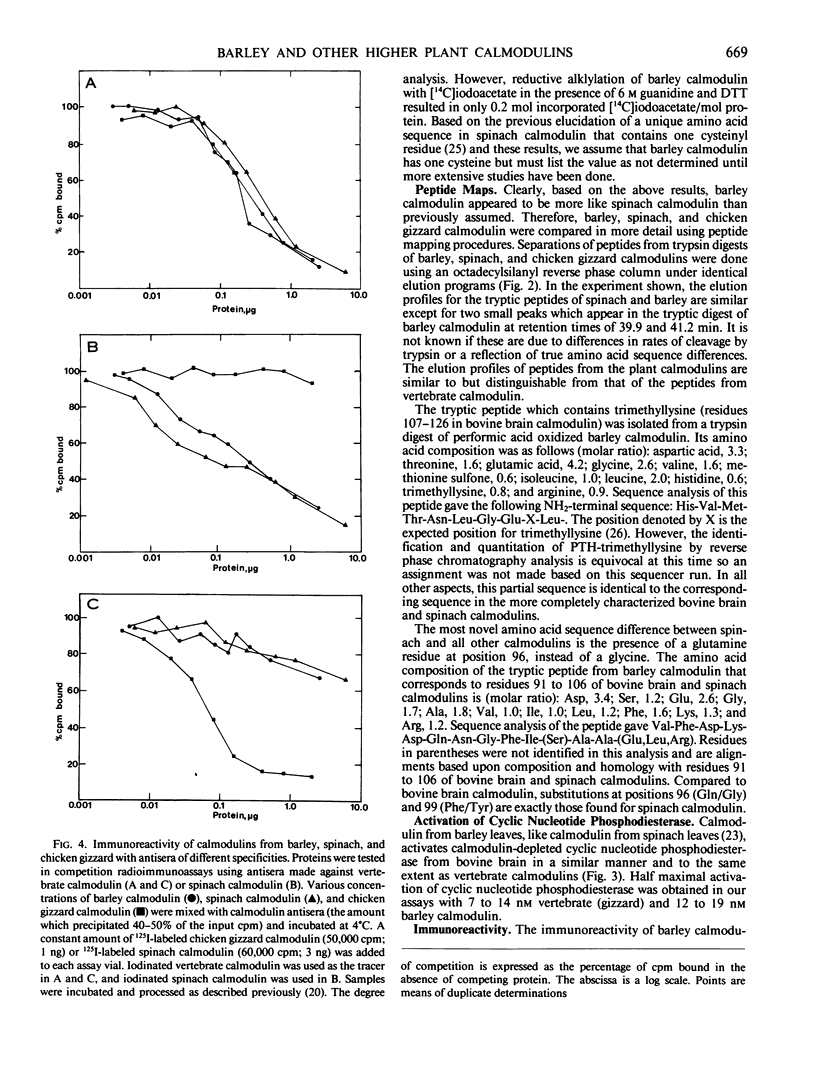
We report here that calmodulin isolated from the monocotyledon barley is indistinguishable by a variety of criteria from calmodulin isolated from the dicotyledon spinach. In contrast to previous reports, we find that barley (Hordeum vulgare) calmodulin has an amino acid composition similar to that of vertebrate and spinach calmodulins, including the presence of a single trimethyllysinyl residue, and that barley calmodulin quantitatively activates cyclic nucleotide phosphodiesterase. Furthermore, spinach and barley calmodulins are similar in terms of tryptic peptide maps and immunoreactivity with various antisera that differ in their molecular specificities for calmodulins. Limited amino acid sequence analysis demonstrates that the region around the single histidinyl and trimethyllysinyl residues is identical among barley, spinach, and vertebrate calmodulins and that barley calmodulin, like spinach calmodulin, has a novel glutamine residue at position 96. We conclude that calmodulin is highly conserved among higher plants and that detailed sequence analysis is required before significant differences, if any, can be assigned to barley or other higher plant calmodulins. These studies suggest that calmodulin's fundamental importance to the eukaryotic cell may have been established prior to the evolutionary emergence of higher plants.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson J. M., Charbonneau H., Jones H. P., McCann R. O., Cormier M. J. Characterization of the plant nicotinamide adenine dinucleotide kinase activator protein and its identification as calmodulin. Biochemistry. 1980 Jun 24;19(13):3113–3120. doi: 10.1021/bi00554a043. [DOI] [PubMed] [Google Scholar]
- Burgess W. H. Characterization of calmodulin and calmodulin isotypes from sea urchin gametes. J Biol Chem. 1982 Feb 25;257(4):1800–1804. [PubMed] [Google Scholar]
- Burgess W. H., Jemiolo D. K., Kretsinger R. H. Interaction of calcium and calmodulin in the presence of sodium dodecyl sulfate. Biochim Biophys Acta. 1980 Jun 26;623(2):257–270. doi: 10.1016/0005-2795(80)90254-8. [DOI] [PubMed] [Google Scholar]
- Eldik L. J., Grossman A. R., Iverson D. B., Watterson D. M. Isolation and characterization of calmodulin from spinach leaves and in vitro translation mixtures. Proc Natl Acad Sci U S A. 1980 Apr;77(4):1912–1916. doi: 10.1073/pnas.77.4.1912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gitelman S. E., Witman G. B. Purification of calmodulin from Chlamydomonas: calmodulin occurs in cell bodies and flagella. J Cell Biol. 1980 Dec;87(3 Pt 1):764–770. doi: 10.1083/jcb.87.3.764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grand R. J., Nairn A. C., Perry S. V. The preparation of calmodulins from barley (Hordeum sp.) and basidiomycete fungi. Biochem J. 1980 Mar 1;185(3):755–760. doi: 10.1042/bj1850755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hewick R. M., Hunkapiller M. W., Hood L. E., Dreyer W. J. A gas-liquid solid phase peptide and protein sequenator. J Biol Chem. 1981 Aug 10;256(15):7990–7997. [PubMed] [Google Scholar]
- Jamieson G. A., Jr, Bronson D. D., Schachat F. H., Vanaman T. C. Structure and function relationships among calmodulins and troponin C-like proteins from divergent eukaryotic organisms. Ann N Y Acad Sci. 1980;356:1–13. doi: 10.1111/j.1749-6632.1980.tb29593.x. [DOI] [PubMed] [Google Scholar]
- Klee C. B., Vanaman T. C. Calmodulin. Adv Protein Chem. 1982;35:213–321. doi: 10.1016/s0065-3233(08)60470-2. [DOI] [PubMed] [Google Scholar]
- Sasagawa T., Ericsson L. H., Walsh K. A., Schreiber W. E., Fischer E. H., Titani K. Complete amino acid sequence of human brain calmodulin. Biochemistry. 1982 May 11;21(10):2565–2569. doi: 10.1021/bi00539a041. [DOI] [PubMed] [Google Scholar]
- Van Eldik L. J., Piperno G., Watterson D. M. Similarities and dissimilarities between calmodulin and a Chlamydomonas flagellar protein. Proc Natl Acad Sci U S A. 1980 Aug;77(8):4779–4783. doi: 10.1073/pnas.77.8.4779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Eldik L. J., Watterson D. M. Characterization of a calcium-modulated protein from transformed chicken fibroblasts. J Biol Chem. 1979 Oct 25;254(20):10250–10255. [PubMed] [Google Scholar]
- Van Eldik L. J., Watterson D. M. Reproducible production of antiserum against vertebrate calmodulin and determination of the immunoreactive site. J Biol Chem. 1981 May 10;256(9):4205–4210. [PubMed] [Google Scholar]
- Watterson D. M., Harrelson W. G., Jr, Keller P. M., Sharief F., Vanaman T. C. Structural similarities between the Ca2+-dependent regulatory proteins of 3':5'-cyclic nucleotide phosphodiesterase and actomyosin ATPase. J Biol Chem. 1976 Aug 10;251(15):4501–4513. [PubMed] [Google Scholar]
- Watterson D. M., Iverson D. B., Van Eldik L. J. Rapid separation and quantitation of 3',5'-cyclic nucleotides and 5'-nucleotides in phosphodiesterase reaction mixtures using high-performance liquid chromatography. J Biochem Biophys Methods. 1980 Mar;2(3):139–146. doi: 10.1016/0165-022x(80)90021-4. [DOI] [PubMed] [Google Scholar]
- Watterson D. M., Iverson D. B., Van Eldik L. J. Spinach calmodulin: isolation, characterization, and comparison with vertebrate calmodulins. Biochemistry. 1980 Dec 9;19(25):5762–5768. doi: 10.1021/bi00566a015. [DOI] [PubMed] [Google Scholar]
- Watterson D. M., Sharief F., Vanaman T. C. The complete amino acid sequence of the Ca2+-dependent modulator protein (calmodulin) of bovine brain. J Biol Chem. 1980 Feb 10;255(3):962–975. [PubMed] [Google Scholar]
- Yazawa M., Yagi K. The amino acid sequence of the calmodulin obtained from sea anemone (metridium senile) muscle. Biochem Biophys Res Commun. 1980 Sep 16;96(1):377–381. doi: 10.1016/0006-291x(80)91225-5. [DOI] [PubMed] [Google Scholar]
- Yazawa M., Yagi K., Toda H., Kondo K., Narita K., Yamazaki R., Sobue K., Kakiuchi S., Nagao S., Nozawa Y. The amino acid sequence of the Tetrahymena calmodulin which specifically interacts with guanylate cyclase. Biochem Biophys Res Commun. 1981 Apr 30;99(4):1051–1057. doi: 10.1016/0006-291x(81)90725-7. [DOI] [PubMed] [Google Scholar]