Abstract
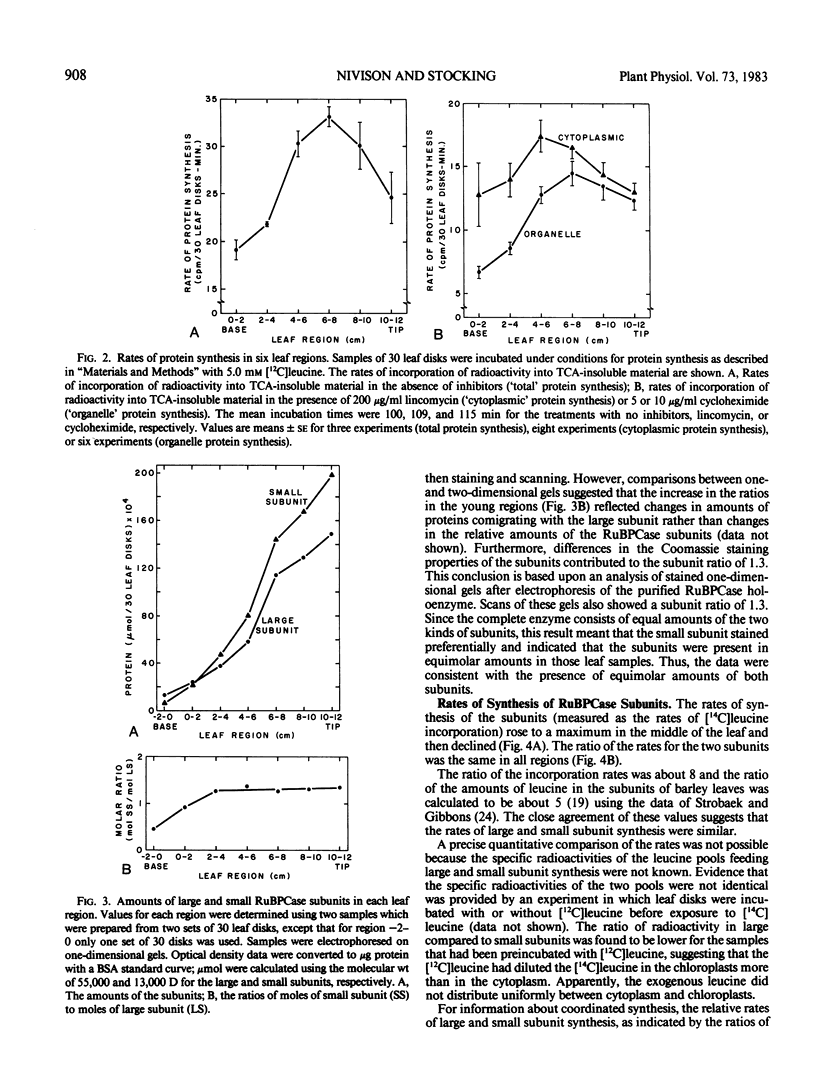
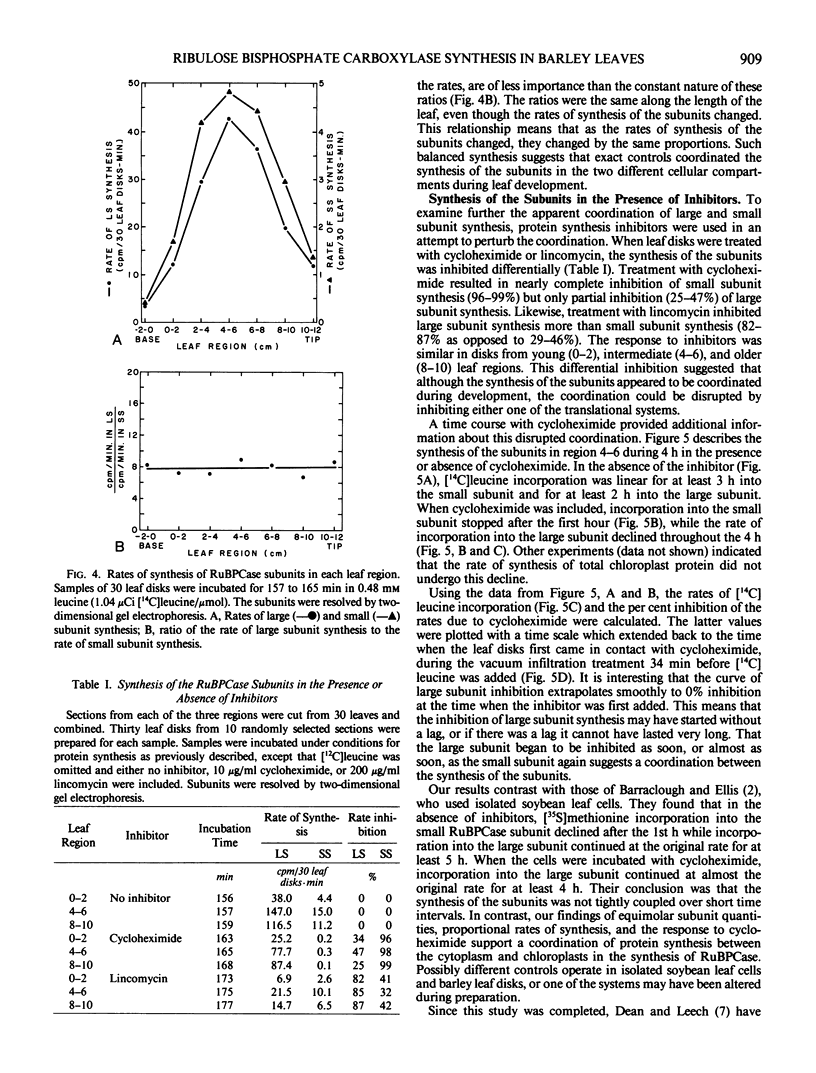
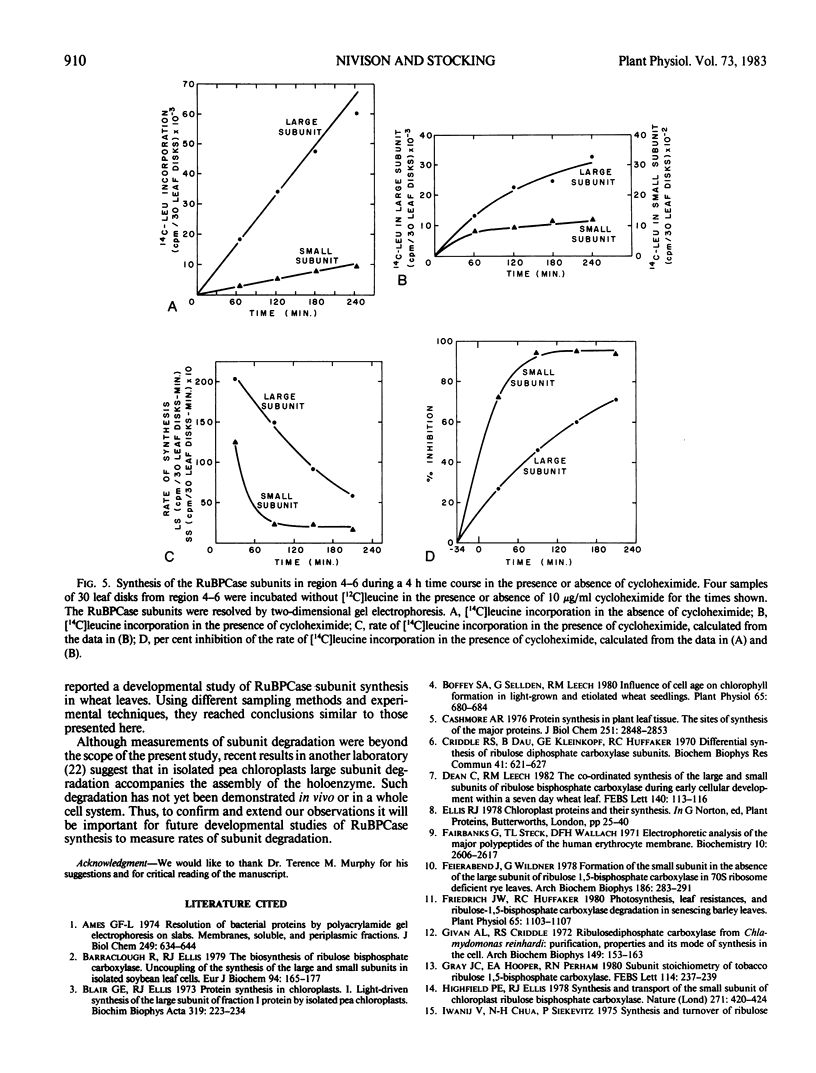
The coordination of the synthesis of the large and small subunits of ribulose 1,5-bisphosphate carboxylase (RuBPCase) was studied in young light-grown barley (Hordeum vulgare L. var. UC566) leaves. Since a barley leaf is a continuum of different aged cells with the youngest cells at the base and the oldest at the tip, developmental changes could be investigated by comparing different leaf regions. The rate of total cytoplasmic protein synthesis increased to a maximum before the rate of total organelle protein synthesis. The different positions of the maxima suggested that the synthesis of the small RuBPCase subunit on cytoplasmic ribosomes and the large RuBPCase subunit on chloroplast ribosomes might not be coupled during barley leaf development. However, measurements of the amounts and rates of synthesis of the subunits showed that they were coupled. Although the amounts of the RuBPCase subunits increased from the younger to the older leaf regions, the subunits were present in an equimolar ratio. While the rates of synthesis of both subunits increased to a maximum in a midleaf region and then declined, the ratio of the rates remained constant. That the subunit amounts remained equimolar and the synthetic rates proportional while total RuBPCase synthesis was changing indicated that the synthesis of the subunits was closely coordinated during leaf development. A close coordination was also supported by the kinetics of the inhibition of subunit synthesis in the presence of cycloheximide.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ames G. F. Resolution of bacterial proteins by polyacrylamide gel electrophoresis on slabs. Membrane, soluble, and periplasmic fractions. J Biol Chem. 1974 Jan 25;249(2):634–644. [PubMed] [Google Scholar]
- Barraclough R., Ellis R. J. The biosynthesis of ribulose bisphosphate carboxylase. Uncoupling of the synthesis of the large and small subunits in isolated soybean leaf cells. Eur J Biochem. 1979 Feb 15;94(1):165–177. doi: 10.1111/j.1432-1033.1979.tb12883.x. [DOI] [PubMed] [Google Scholar]
- Blair G. E., Ellis R. J. Protein synthesis in chloroplasts. I. Light-driven synthesis of the large subunit of fraction I protein by isolated pea chloroplasts. Biochim Biophys Acta. 1973 Aug 24;319(2):223–234. doi: 10.1016/0005-2787(73)90013-0. [DOI] [PubMed] [Google Scholar]
- Boffey S. A., Selldén G., Leech R. M. Influence of Cell Age on Chlorophyll Formation in Light-grown and Etiolated Wheat Seedlings. Plant Physiol. 1980 Apr;65(4):680–684. doi: 10.1104/pp.65.4.680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cashmore A. R. Protein synthesis in plant leaf tissue. The sites of synthesis of the major proteins. J Biol Chem. 1976 May 10;251(9):2848–2853. [PubMed] [Google Scholar]
- Criddle R. S., Dau B., Kleinkopf G. E., Huffaker R. C. Differential synthesis of ribulosediphosphate carboxylase subunits. Biochem Biophys Res Commun. 1970 Nov 9;41(3):621–627. doi: 10.1016/0006-291x(70)90058-6. [DOI] [PubMed] [Google Scholar]
- Fairbanks G., Steck T. L., Wallach D. F. Electrophoretic analysis of the major polypeptides of the human erythrocyte membrane. Biochemistry. 1971 Jun 22;10(13):2606–2617. doi: 10.1021/bi00789a030. [DOI] [PubMed] [Google Scholar]
- Feierabend J., Wildner G. Formation of the small subunit in the absence of the large subunit of ribulose 1,5-bisphosphate carboxylase in 70 S ribosome-deficient rye leaves. Arch Biochem Biophys. 1978 Mar;186(2):283–291. doi: 10.1016/0003-9861(78)90437-x. [DOI] [PubMed] [Google Scholar]
- Friedrich J. W., Huffaker R. C. Photosynthesis, leaf resistances, and ribulose-1,5-bisphosphate carboxylase degradation in senescing barley leaves. Plant Physiol. 1980 Jun;65(6):1103–1107. doi: 10.1104/pp.65.6.1103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Givan A. L., Criddle R. S. Ribulosediphosphate carboxylase from Chlamydomonas reinhardi: purification, properties and its mode of synthesis in the cell. Arch Biochem Biophys. 1972 Mar;149(1):153–163. doi: 10.1016/0003-9861(72)90309-8. [DOI] [PubMed] [Google Scholar]
- Gray J. C., Hooper E. A., Perham R. N. Subunit stoichiometry of tobacco ribulose 1.5-bisphosphate carboxylase. FEBS Lett. 1980 Jun 2;114(2):237–239. doi: 10.1016/0014-5793(80)81123-9. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Nishimura M., Akazawa T. Biosynthesis of ribulose-1,5-bisphosphate carboxylase in spinach leaf protoplasts. Plant Physiol. 1978 Jul;62(1):97–100. doi: 10.1104/pp.62.1.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Farrell P. H. High resolution two-dimensional electrophoresis of proteins. J Biol Chem. 1975 May 25;250(10):4007–4021. [PMC free article] [PubMed] [Google Scholar]
- Roy H., Bloom M., Milos P., Monroe M. Studies on the assembly of large subunits of ribulose bisphosphate carboxylase in isolated pea chloroplasts. J Cell Biol. 1982 Jul;94(1):20–27. doi: 10.1083/jcb.94.1.20. [DOI] [PMC free article] [PubMed] [Google Scholar]


