Abstract
The imbalance between regulatory T (Treg) cells and efficient T cells plays an important role in psoriasis. Low-dose interleukin (IL)-2 can preferentially activate Treg cells and ameliorate the imbalance of Treg/efficient T cells. This study focused on the status of circulating CD4+ T subsets and the clinical efficacy of low-dose IL-2 therapies in psoriasis. This retrospective study included peripheral blood samples obtained from 45 psoriatic patients and 40 healthy controls. The 45 psoriatic patients received three cycles of subcutaneous low-dose IL-2 treatment (0.5 million IU/day for 2 weeks) combined with conventional therapies. Inflammatory indices, CD4+ T-lymphocyte subsets, and cytokines were measured in all patients before and after treatment. The percentage of Treg cells was dramatically decreased in the psoriasis group compared to the healthy group, and the percentage of Treg cells negatively correlated with the disease indices and the Psoriasis Area and Severity Index (PASI) (P < 0.001). The Th17/Treg ratio was significantly increased in the psoriasis group compared to the healthy group, and the Th17/Treg ratio positively correlated with disease indices and PASI (P < 0.001). Low-dose IL-2 treatment significantly amplified the percentage of Treg cells and restored the Th17 and Treg immune balance in psoriasis (P < 0.001). Low-dose IL-2 combination therapy effectively improved the clinical manifestations of psoriasis but decreased the inflammatory indicators of the disease activity, with no apparent side effects. Thus, low-dose IL-2 provides a new strategy for the treatment of psoriasis.
Supplementary Information
The online version contains supplementary material available at 10.1007/s10753-023-01883-6.
KEY WORDS: interleukin-2, immunotherapy, treg cells, psoriasis
INTRODUCTION
Psoriasis is an immune-mediated systemic inflammatory disease. Its aetiology and pathogenesis are not fully understood. The imbalance between T-helper 1 (Th1) and T-helper 2 (Th2) cells is considered as the main pathogenesis of psoriasis, and growing evidence suggests that T-helper 17 (Th17) cells and CD4+CD25+ regulatory T (Treg) cells play important roles in the pathogenesis of psoriasis [1–4].
Interleukin-2 (IL-2) is an important immune regulatory factor. High-dose IL-2 has anti-tumour and enhanced immune effects, while low-dose IL-2 has immunosuppressive effects. Treg cells express CD25 on their surfaces and can bind the IL-2 receptor (α, β, and γ) with high affinity. Low-dose IL-2 can activate Treg cells, while other subsets of T cells require higher doses of IL-2 for activation. This forms the molecular basis of the low-dose IL-2 regulation of Treg cell functions [5]. Although the role of low-dose IL-2 in the treatment of autoimmune diseases has also attracted more attention, there are rare direct studies on the efficacy of low-dose IL-2 in psoriasis.
In this study, we evaluated the Th1, Th2, Th17, and Treg cells, cytokines, and inflammatory indices in patients with different types of psoriasis to explore the roles of Th1/Th2 and Th17/Treg imbalance in the pathogenesis of psoriasis. To investigate new concepts for the treatment of psoriasis, we observed the inflammatory and immunological indices of patients with psoriasis before and after treatment with low-dose IL-2 to clarify the clinical efficacy and safety.
METHODS
Subjects
In this retrospective study, peripheral blood samples were obtained from 45 psoriatic patients (24 men and 21 women; 21–58 years of age; 17 psoriasis vulgaris (PV), 18 psoriasis arthritis (PA), 5 erythrodermic psoriasis (EP), and 5 generalized pustular psoriasis (GPP)) and 40 healthy controls (19 men and 21 women; 23–53 years of age) from February 2020 to November 2021. The inclusion criteria were a documented diagnosis of psoriasis and a Psoriasis Area and Severity Index (PASI) ≥ 10. The exclusion criteria were another severe or progressive autoimmune/inflammatory disease; haematological disorders; vital organ failure; cancer; and active HIV, hepatitis B virus, or Epstein–Barr virus infections.
Study Design and Treatment
The 45 psoriatic patients received methotrexate at 7.5 mg per week for 12 weeks to control disease activity. From 13 to 24 weeks, the patients received conventional therapy combined with three cycles of low-dose IL-2 therapy using recombinant human IL-2 (rhIL-2, Beijing, China). Each of the three cycles consisted of daily subcutaneous injections of IL-2 for 2 consecutive weeks, followed by a 2-week break. The daily dose in every cycle was 0.5 million international units (MIU) of IL-2 for all patients. This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the Second Hospital of Shanxi Medical University (No. 2021-YX-010). Written informed consent was obtained from all participates prior to their enrolment in this study.
Measurement of Inflammatory Indices
The peripheral neutrophil count, leukocyte count, lymphocyte count, platelet count, neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), erythrocyte sedimentation rate (ESR), and c-reactive protein (CRP) concentration in serum were recorded.
Blood Sample Collection and Preparation
Peripheral blood mononuclear cells (PBMCs) were isolated from heparinized peripheral blood by density gradient centrifugation with Ficoll–Paque PREMIUM 1.077 (GE Healthcare Life Sciences, Pittsburgh, PA). The cells were then resuspended in RPMI 1640 medium containing phorbol 12-myristate13-acetate (PMA) (50 ng/ml), GolgiStop (1 µl), and ionomycin (1 µg/ml) for 5 h in an incubator at 37 °C with 5% CO2.
Cell Staining
The stimulated cells were stained with fluorescein isothiocyanate (FITC)–labelled CD4 antibody for 30 min at 4 °C. After surface staining, the cells were fixed, permeabilized, and stained with interferon-γ (IFN-γ)-phycoerythrin (PE) (Th1), IL-4-PE (Th2), and IL-17-PE (Th17). For Treg cells, the PBMCs were incubated with FITC–labelled CD4 and allophycocyanin (APC)-labelled CD25 antibodies at 4 °C for 30 min. The cells were then treated with 1 ml of freshly prepared fixation/permeabilization solution followed by staining with PE-labelled Foxp3 antibody.
Flow Cytometric Analysis
The percentages of Th1, Th2, Th17, and Treg cells were detected by flow cytometry (Calibur, BD, USA) and analysed by fluorescence-activated cell sorting (FACS) (Canto II, BD Biosciences, San Jose, CA) followed by data analysis using FlowJo 7.6 software (TreeStar, San Carlos, CA).
Cytometric Bead Array
The cytokines in serum from peripheral blood were analysed using a cytometric bead array human Th1, Th2, Th17, and Treg cytokine kit (Becton Dickinson, USA) along with a FACS Calibur flow cytometer. The beads were coated with antibodies that specifically reacted with each of the cytokines to be detected in the multiplex system. The beads could be differentiated by their sizes and their distinct spectral addresses. The serum concentrations of IL-2, IL-4, IL-6, IL-10, IL-17, IFN-γ, and tumour necrosis factor (TNF)-α were determined using the cytometric bead array technique.
Evaluation of Therapeutic Efficiency
Therapeutic efficiency was primarily evaluated using the quantitative PASI score, which measures the severity of psoriatic lesions based on area coverage and plaque appearance, including scaling, infiltration, and erythema. PASI 50 and PASI 75 indicated 50% and 75% improvements in PASI, respectively. PA patients were identified according to the criteria of the American College of Rheumatology (ACR20 response). At weeks 0 and 24 of treatment, all observation indices were reviewed, and the adverse reactions of patients were recorded in detail.
Statistical Analysis
SPSS version 22.0 was used for all data analyses (SPSS Inc., Chicago, IL, USA), and P < 0.05 was considered statistically significant. Continuous variables were expressed as mean ± standard deviation, and the two groups were compared using Student’s t-test. One-way analysis of variance and least significant difference (LSD) test were used to analyse multiple groups. Median (interquartile range) and Wilcoxon test were used for abnormally distributed data when there were two groups. Kruskal–Wallis and LSD tests were used for multiple groups. For categorical variables, counts (percentages) were described and compared using the χ2 test. Relationships between two variables were evaluated by Spearman correlation analysis.
RESULTS
Baseline Characteristics of All Subjects
In total, 45 psoriatic patients and a group of 40 healthy subjects were enrolled. The difference between the ages of the psoriasis and healthy groups was not statistically significant (37.67 ± 5.54 vs. 36.00 ± 8.66, respectively; P > 0.05). The percentage of men in the psoriasis group was not significantly different from that in the healthy group (52.12% vs. 47.5%, respectively; P > 0.05). The years of median disease duration were 6.0 (7.5) in the psoriasis group (P < 0.001). The median PASI score was 19.500 (11.900) in the psoriasis group (P < 0.001; Table 1).
Table 1.
Baseline Characteristics and Inflammatory Parameters of All Subjects [mean ± SD, M(IQR)]
| Parameter | Healthy groups | Psoriasis groups | t/χ2 | P |
|---|---|---|---|---|
| N | 40 | 45 | - | - |
| Age (year) | 36.000 ± 8.659 | 37.667 ± 9.535 | –0.840 | 0.403 |
| Men (%) | 47.500 | 53.300 | 0.288 | 0.591 |
| Duration (year) | 0.000 (0.000) | 6.000 (7.500) | 8.387 | < 0.001 |
| PASI | 0.000 (0.000) | 19.500 (11.900) | –13.871 | < 0.001 |
| Leukocytes (× 109/L) | 6.798 ± 2.064 | 7.030 (3.410) | 1.985 | 0.047 |
| Neutrophil (× 109/L) | 4.566 ± 1.951 | 4.920 (3.140) | 2.157 | 0.031 |
| Lymphocytes (× 109/L) | 1.946 ± 0.520 | 1.947 ± 0.540 | –0.007 | 0.994 |
| Platelets (× 109/L) | 235 ± 60 | 235 (189) | 1.444 | 0.149 |
| ESR (mm/h) | 11 (6) | 30 (63) | 4.949 | < 0.001 |
| CRP (mg/L) | 5.740 (5.530) | 18.560 (43.645) | 5.212 | < 0.001 |
| NLR | 2.105 (1.416) | 3.440 (1.670) | 5.115 | < 0.001 |
| PLR | 127.941 ± 43.173 | 144.630 (96.175) | 1.259 | 0.208 |
NLR neutrophil-to-lymphocyte ratio, PASI Psoriasis Area and Severity Index, PLR platelet-to-lymphocyte ratio
The psoriasis group included 17 PV, 18 PA, 5 EP, and 5 GPP patients. The ages and sexes of the four types in the psoriasis group were not significant (P > 0.05). The PASI scores of the different psoriasis groups are shown in Table 2 (P < 0.001; Table 2).
Table 2.
Clinical Characteristics and Inflammatory Parameters Among the Different Psoriasis Groups [Mean ± SD, M(IQR)]
| Parameter | Healthy groups | PV | PA | EP | GPP | F/H | P |
|---|---|---|---|---|---|---|---|
| N | 40 | 17 | 18 | 5 | 5 | - | - |
| Age (year) | 36.000 ± 8.659 | 37.059 ± 9.202 | 35.278 ± 9.809 | 42.200 ± 8.408 | 43.800 ± 9.039 | 1.406 | 0.240 |
| Men (%) | 47.500 | 52.941 | 55.556 | 60.000 | 40.000 | 0.773 | 0.942 |
| Duration (year) | 0.000 (0.000)* | 6.147 ± 0.803* | 5.000 (8.500)* | 11.200 ± 1.985* | 7.000 ± 2.168* | 71.581 | < 0.001 |
| PASI | 0.000 (0.000)* | 18.171 ± 1.490* | 14.250 (9.550)* | 42.700 ± 2.337* | 20.620 ± 3.896* | 74.417 | < 0.001 |
| Leukocytes (× 109/L) | 6.798 ± 2.064* | 6.871 ± 1.671* | 6.806 ± 1.435* | 17.792 ± 4.086* | 10.536 ± 1.926* | 37.147 | < 0.001 |
| Neutrophil (× 109/L) | 4.566 ± 1.951* | 4.634 ± 1.397* | 4.679 ± 1.451* | 14.874 ± 3.985* | 8.398 ± 1.551* | 37.473 | < 0.001 |
| Lymphocytes (× 109/L) | 1.946 ± 0.520 | 1.971 ± 0.704 | 1.965 (0.350) | 2.004 ± 0.615 | 1.988 ± 0.518 | 0.212 | 0.995 |
| Platelets (× 109/L) | 235 ± 60* | 239 (64)* | 263 ± 101* | 460 ± 100* | 316 ± 83 | 15.410 | 0.004 |
| ESR (mm/h) | 12 (6)* | 19 ± 11* | 44 ± 34* | 118 ± 48* | 82 ± 23* | 38.109 | < 0.001 |
| CRP (mg/L) | 5.740 (5.530)* | 8.330 (11.135)* | 16.945 (40.160)* | 73.484 ± 9.991* | 65.334 ± 58.328* | 42.070 | < 0.001 |
| NLR | 2.105 (1.416)* | 3.306 ± 0.815* | 3.292 ± 0.734* | 9.180 ± 2.262* | 5.196 ± 0.925* | 36.933 | < 0.001 |
| PLR | 127.941 ± 43.173* | 139.940 (70.320)* | 146.414 ± 75.668* | 240.004 ± 62.587* | 162.852 ± 41.455 | 12.680 | 0.013 |
NLR neutrophil-to-lymphocyte ratio, PASI Psoriasis Area and Severity Index, PLR platelet-to-lymphocyte ratio
*P < 0.05
NLR and PLR Combined With ESR and CRP May Indicate Systemic Inflammation and Immune Status in Psoriasis
Compared with the healthy group, circulating leukocyte count, neutrophil count, NLR, ESR, and CRP were higher in the psoriasis group (P < 0.05). No significant differences were observed in lymphocyte count, platelet count, and PLR between the psoriasis and healthy groups (P > 0.05; Table 1). In the four types of the psoriasis group, PLR, platelet count, leukocyte count, and neutrophil count in peripheral blood were significantly increased in EP patients compared to PV patients, PA patients, and the healthy group (P < 0.05). The ESR and the CRP serum concentration were significantly higher in PA, EP, and GPP patients than in the healthy group (P < 0.001). No significant differences were observed in lymphocyte count among the psoriasis groups (P > 0.05; Table 2).
Differences in CD4+ T Cell Subset Numbers and Cytokines Between the Psoriasis and Healthy Groups
The Psoriasis Group Was Associated With a Higher Proportion of Th17 Cells, a Lower Proportion of Treg Cells, and an Increased Th17/Treg Ratio Compared to the Healthy Group
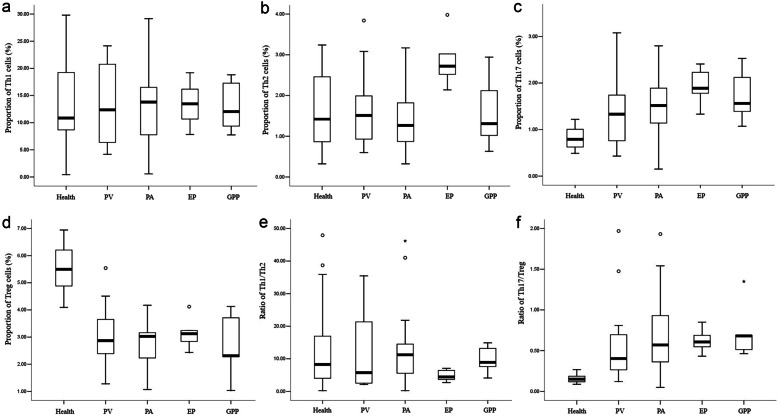
The proportion of Th17 cells in peripheral blood was significantly higher in the psoriasis group compared with the healthy group (P < 0.001; Table 3), which increased in the following order: PV < PA < GPP < EP (P < 0.001; Fig. 1c). In contrast, the percentage of Treg cells was dramatically lower in the psoriasis group compared to the healthy group (P < 0.001; Table 3), which increased in the following order: GPP < PA < PV < EP (P < 0.001; Fig. 1d). As a result, the Th17/Treg ratio was increased in the psoriasis group compared with the healthy group (P < 0.001; Table 3), which increased in the following order: PV < PA < EP < GPP (P < 0.001; Fig. 1f). However, no significant differences in Th1, Th2, or the Th1/Th2 ratio were observed between the psoriasis and healthy groups (P > 0.05, Table 3; Fig. 1a, b and e).
Table 3.
Differences in CD4+ T Cell Subset Numbers in the Psoriasis and Healthy Group [Mean ± SD, M(IQR)]
| Group | Healthy group | Psoriasis group | t/Z | P |
|---|---|---|---|---|
| N | 40 | 45 | - | - |
| Th1 (%) | 13.235 ± 6.835 | 13.114 ± 6.864 | 0.081 | 0.935 |
| Th2 (%) | 1.420 (1.618) | 1.510 (1.405) | −0.445 | 0.657 |
| Th17 (%) | 0.826 ± 0.215 | 1.546 ± 0.681 | −6.728 | < 0.001 |
| Treg (%) | 5.483 ± 0.802 | 2.859 ± 0.991 | 13.314 | < 0.001 |
| Th1/Th2 | 8.273 (13.898) | 7.598 (11.016) | 71.581 | 74.417 |
| Th17/Treg | 0.154 ± 0.045 | 0.547 (0.364) | −6.947 | < 0.001 |
Fig. 1.
a Proportion of Th1 cells. b Proportion of Th2 cells. c Proportion of Th17 cells. d Proportion of Treg cells. e Ratio of Th1/Th2. f Ratio of Th17/Treg.
Psoriasis Decreased IL-10 Concentrations, Increased IL-17 Concentrations, and Increased the IL-17/IL-10 Ratio Compared to the Healthy Group
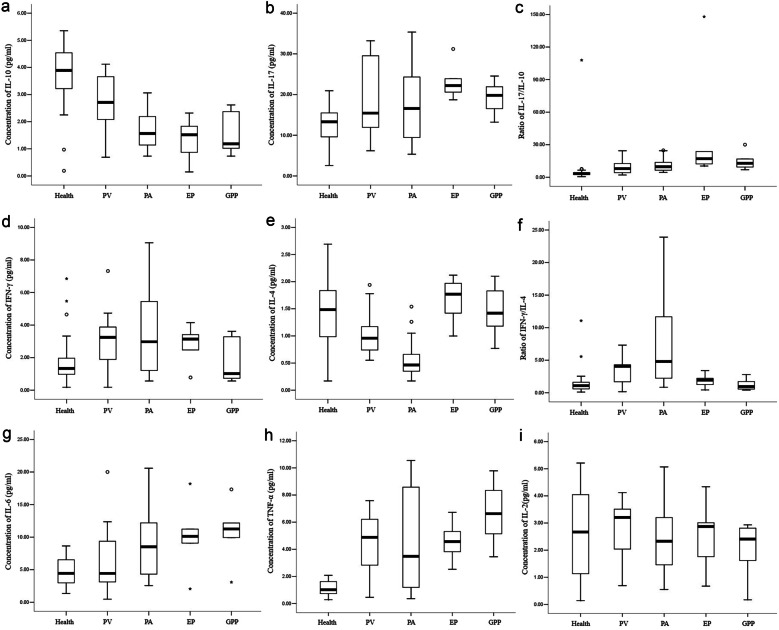
The serum concentrations of IL-10 in peripheral blood were lower in the psoriasis group than in the healthy group (P < 0.001; Table 4), which decreased in the following order: GPP < EP < PA < PV (P < 0.001; Fig. 2a). In contrast, the serum concentrations of IL-17 were higher in the psoriasis group compared to the healthy group (P < 0.001; Table 4), which decreased in the following order: PV < PA < EP, whereas the difference in IL-17 between the GPP patients and the healthy group was not significant (P > 0.05; Fig. 2b). It may be related to the difference in the distribution of IL-17 in peripheral blood and local tissues and the small sample size of GPP patients. As a result, the IL-17/IL-10 ratio was increased in the psoriatic patients compared with the healthy group (P < 0.001; Table 4), which increased in the following order: PV < PA < GPP < EP (P < 0.001; Fig. 2c).
Table 4.
Differences in Cytokine Levels Between the Psoriasis and Healthy Groups (pg/ml) [Mean ± SD, M(IQR)]
| Group | Healthy group | Psoriasis group | t/Z | P |
|---|---|---|---|---|
| N | 40 | 45 | - | - |
| IL-10 | 3.807 ± 1.094 | 2.012 ± 0.973 | 8.005 | < 0.001 |
| IL-17 | 12.778 ± 4.395 | 19.262 ± 8.678 | –4.415 | < 0.001 |
| IFN-γ | 1.335 (1.040) | 3.007 ± 2.069 | 3.002 | 0.003 |
| IL-4 | 1.454 ± 0.565 | 0.976 ± 0.551 | 3.948 | < 0.001 |
| IL-6 | 4.753 ± 2.207 | 8.330 (7.500) | 3.372 | 0.001 |
| TNF-α | 1.168 ± 0.521 | 4.888 ± 2.824 | –8.673 | < 0.001 |
| IL-2 | 2.670 (2.953) | 2.518 ± 1.219 | –0.735 | 0.462 |
| IL-17/IL-10 | 3.247 (2.027) | 10.644 (8.780) | 6.084 | < 0.001 |
| IFN-γ/IL-4 | 1.097 (1.025) | 3.019 (4.321) | 4.393 | < 0.001 |
Fig. 2.
Concentrations of a IL-10, b IL-17, d IFN-γ, e IL-4, g IL-6, h TNF-α, and i IL-2. Ratios of c IL-17/IL-10 and f IFN-γ/IL-4.
Psoriasis Increased IFN-γ Concentrations, Decreased IL-4 Concentrations, and Increased the IFN-γ/IL-4 Ratio Compared to the Healthy Group
Compared with the healthy group, the IFN-γ concentrations were significantly elevated in the psoriasis group (P = 0.003). In contrast, the serum concentrations of IL-4 were significantly lower in the psoriasis group than in the healthy group (P < 0.001). As a result, the IFN-γ/IL-4 ratio was higher in the psoriasis group than in the healthy group (P < 0.001; Table 4). PV and PA patients had increased IFN-γ concentrations, decreased IL-4 concentrations, and greatly increased IFN-γ/IL-4 ratio compared to the healthy group. In contrast, no significant differences in IFN-γ, IL-4, or the IFN-γ/IL-4 ratio were observed between the EP and GPP patients compared with the healthy group (P > 0.05; Fig. 2d–f).
IL-6 and TNF-α Concentrations Were Significantly Elevated in Psoriatic Patients Compared to the Healthy Group
IL-6 is a critical factor in the homeostatic balance between Th17 and Treg cells. The serum concentrations of IL-6 were significantly elevated in PA, EP, and GPP patients compared with the healthy group, which was the highest in GPP patients (P < 0.001). However, the difference in IL-6 between the PV patients and the healthy group was not significant (P > 0.05; Fig. 2g).
The TNF-α concentrations were significantly increased in the psoriatic patients compared with the healthy group (P < 0.001; Table 4), which increased in the following order: PA < EP < PV < GPP (P < 0.001; Fig. 2h). The IL-2 levels in the psoriasis group were not significantly different than those in the healthy group (P > 0.05, Table 4; Fig. 2i).
Correlation Analysis of CD4+ T Lymphocyte Subsets With Inflammatory Indicators in Psoriasis
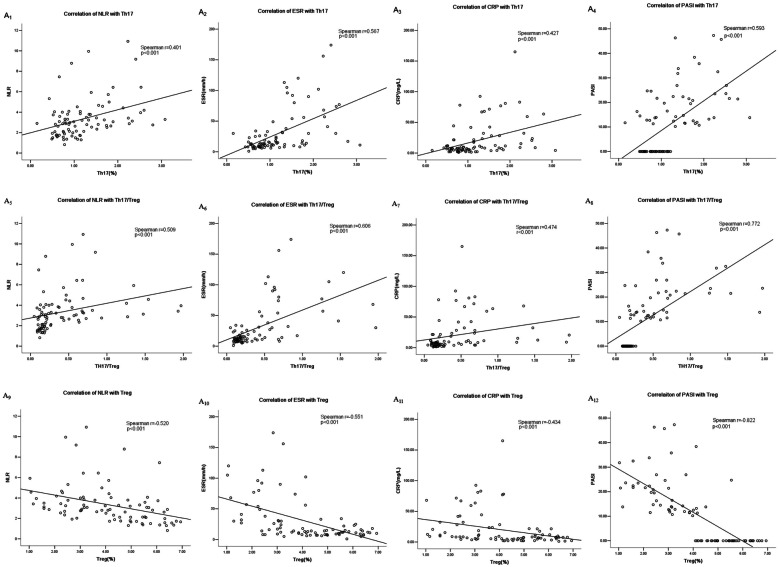
The percentage of Th17 cells positively correlated with NLR, ESR, CRP, and PASI (P < 0.001; Fig. 3A1–A4). The percentage of Treg cells negatively correlated with NLR, ESR, CRP, and PASI (P < 0.001; Fig. 3A9–A12). The Th17/Treg ratio positively correlated with NLR, ESR, CRP, and PASI (P < 0.001; Fig. 3A5–A8). The inflammatory indices did not significantly correlate with the Th1 percentage, Th2 percentage, or Th1/Th2 ratio (P > 0.05).
Fig. 3.
A1–A12 Correlation analysis of CD4+ T lymphocyte subsets with inflammatory indicators in psoriasis. B1–B23 Correlation analysis of cytokines with inflammatory indicators in psoriasis.
Correlation Analysis of Cytokines With Inflammatory Indicators in Psoriasis
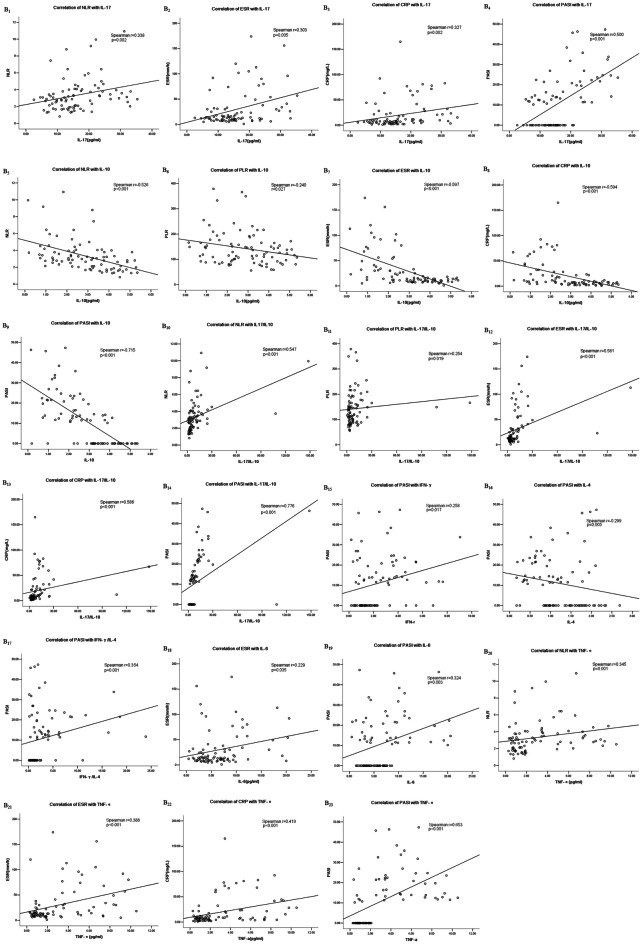
The IL-17 serum concentrations positively correlated with NLR, ESR, CRP, and PASI (P < 0.001; Fig. 3B1–B4). The serum concentrations of IL-10 negatively correlated with NLR, PLR, ESR, CRP, and PASI (P < 0.001; Fig. 3B5–B9). The IL-17/IL-10 ratio positively correlated with NLR, PLR, ESR, CRP, and PASI (P < 0.001; Fig. 3B10–B14).
The serum concentrations of IFN-γ and the IFN-γ/IL-4 ratio positively correlated with PASI (P < 0.001; Fig. 3B15, B17). The serum concentrations of IL-4 negatively correlated with PASI (P < 0.001; Fig. 3B16). The serum concentrations of IL-6 positively correlated with ESR and PASI (P < 0.001; Fig. 3B18, B19). The TNF-α serum concentrations positively correlated with NLR, ESR, CRP, and PASI (P < 0.001; Fig. 3B20–B23). The inflammatory indices did not significantly correlate with the IL-2 serum concentrations (P > 0.05).
Effects of Low-dose IL-2 in Psoriasis
Inflammatory Indicators After IL-2 Combination Therapies in Psoriasis
Next, we observed whether the Treg defects in psoriatic patients could be reversed with low-dose IL-2. The 45 psoriatic patients received methotrexate at 7.5 mg weekly for 12 weeks. From 13 to 24 weeks, they received methotrexate combined with three cycles of low-dose IL-2 therapy (0.5 MIU/d, subcutaneous injections). After treatment, inflammatory indicators of disease activity, including the leukocyte count, neutrophil count, ESR, CRP, and NLR in serum, decreased significantly compared with before treatment (P < 0.05). The lymphocyte count, platelet count, and PLR in serum did not change after treatment (P > 0.05; Table 5).
Table 5.
Changes in the Inflammatory Indicator Levels Before and After Treatment [Mean ± SD, M(IQR)]
| Parameter | Before treatment | After treatment | t/Z | P |
|---|---|---|---|---|
| Leukocytes (× 109/L) | 7.030 (3.410) | 7.046 ± 1.589 | −2.258 | 0.024 |
| Neutrophil (× 109/L) | 4.920 (3.140) | 4.714 ± 1.560 | −2.664 | 0.008 |
| Lymphocytes (× 109/L) | 1.947 ± 0.540 | 2.015 ± 0.559 | 0.555 | 0.581 |
| Platelets (× 109/L) | 235 (189) | 240 ± 58 | −1.913 | 0.056 |
| ESR (mm/h) | 30 (63) | 13 (16) | −5.696 | <0.001 |
| CRP (mg/L) | 18.560 (43.645) | 8.290 (16.855) | −4.809 | <0.001 |
| NLR | 3.440 (1.670) | 2.262 (1.166) | −4.656 | <0.001 |
| PLR | 144.630 (96.175) | 115.672 (38.526) | −1.834 | 0.067 |
NLR neutrophil-to-lymphocyte ratio, PLR platelet-to-lymphocyte ratio
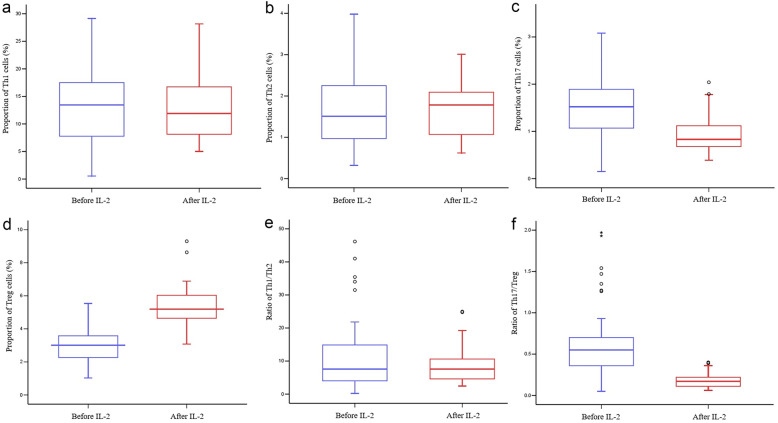
Low-dose IL-2 Can Effectively Promote Treg Cells and Reverse the Th17/Treg Immune Imbalance in Psoriasis
After IL-2 treatment, the percentage of Treg cells was greatly amplified (twofold increase) and decreased the Th17/Treg ratio (P < 0.001). The proportion of Th17 cells in peripheral blood was decreased after treatment (P < 0.001). The percentage of Treg cells was fivefold more than that of Th17 cells, which was the main reason for the decrease in the Th17/Treg ratio. No significant differences in Th1, Th2, or the Th1/Th2 ratio were observed after treatment (P > 0.05; Fig. 4).
Fig. 4.
Proportions of a Th1 cells, b Th2 cells, c Th17 cells, and d Treg cells. Ratios of e Th1/Th2 and f Th17/Treg.
Efficacy of Low-dose IL-2 Treatment on the Peripheral Cytokine Concentrations in Psoriasis
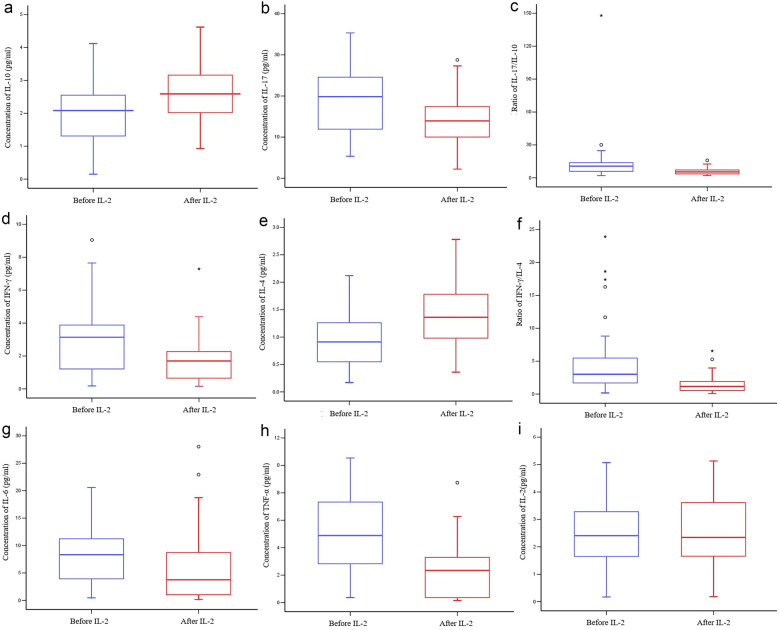
Low-dose IL-2 treatment significantly increased the concentrations of anti-inflammatory cytokines (IL-10 and IL-4) among the psoriatic patients (P < 0.05). In contrast, the concentrations of pro-inflammatory factors (IL-6, IL-17, IFN-γ, and TNF-α) in peripheral blood were significantly decreased after treatment (P < 0.05). As a result, the IL-17/IL-10 and IFN-γ/IL-4 ratios were decreased after treatment (P < 0.001). However, no significant differences in the IL-2 concentration were observed between before and after the treatment (P > 0.05; Fig. 5).
Fig. 5.
Concentrations of a IL-10, b IL-17, d IFN-γ, e IL-4, G IL-6, h TNF-α, and i IL-2. Ratios of c IL-17/IL-10 and f IFN-γ/IL-4.
Clinical Efficacy and Safety of Low-dose IL-2 Therapy
After 24 weeks of treatment, the PASI 50 and PASI 75 were 68.89% and 72.22%, respectively. The ACR20 percentages were 35% after treatment (P < 0.05). Low-dose IL-2 was well tolerated, and most adverse events were mild and of a transient nature, suggesting a favourable safety profile. Two patients experienced injection-site reactions on day 4 of treatment. The reactions subsided spontaneously within 1 week. No significant differences in the alanine transaminase, blood urea nitrogen, or serum creatinine were observed between the psoriatic patients and the treatment groups.
DISCUSSION
Th17/Treg immune imbalance has been reported to contribute to the pathogenesis of psoriasis [1–3]. Normally, naïve CD4+ T cells differentiate into Th17 and Treg cells. The Th17 cells produce IL-6, IL-17, and TNF-α to mediate the inflammatory reaction in psoriasis. Meanwhile, Treg cells produce suppressive cytokines such as IL-10, transforming growth factor-β, and cell–cell interaction, thereby playing an important role in immunosuppression. Th17 and Treg cells regulate each other to maintain immune balance [6–8].
In this study, the proportion of Th17 cells in peripheral blood was significantly increased in the patients with psoriasis compared with the healthy controls, and the Th17 cell proportion positively correlated with NLR, ESR, CRP, and PASI. Moreover, the Treg cell percentage was dramatically decreased and negatively correlated with NLR, ESR, CRP, and PASI. These changes resulted in a dramatic increase in the Th17/Treg ratio, which increased in the following order: PV < PA < EP < GPP. The Th17/Treg ratio positively correlated with NLR, ESR, CRP, and PASI. However, no significant differences in Th1, Th2, or the Th1/Th2 ratio were observed between the psoriasis and healthy groups. These results suggest that the percentages of Th17 and Treg cells are closely related to psoriasis disease severity.
Our findings agree with Ma et al. who reported Th17/Treg immune imbalance in PV patients [9]. However, Zhang et al. found that Th17 and Treg cells were increased in psoriatic patients in the peripheral circulation, both of which positively correlated with PASI score [10, 11]. These distinct results may stem from differences in disease states, Treg definition, the time frame from which the PASI scores were derived, and the investigated psoriasis subtypes [12, 13]. Overall, these studies suggest that Th17/Treg immune imbalance potentially contributes to psoriasis pathogenesis.
The immune imbalance between Th17 and Treg cells is associated with the disorder of pro-inflammatory and anti-inflammatory cytokines in psoriasis. In this study, the serum concentrations of IL-10 in the psoriasis group were decreased compared to the healthy group, and the IL-10 concentrations negatively correlated with NLR, ESR, CRP, and PASI. Reduction in IL-10 concentrations indirectly showed that Treg cells’ suppressive function was impaired, and this could not effectively resist the pro-inflammatory effect of increased Th17 cells secretion of IL-17. The serum concentrations of IL-17 were higher in the psoriatic patients compared to the healthy controls. As a result, the IL-17/IL-10 ratio was increased in the psoriasis group compared to the healthy group, and it positively correlated with NLR, PLR, ESR, CRP, and PASI. The increased IL-17/IL-10 ratio further confirms the imbalance of Th17/Treg cells in psoriasis.
IL-6 is a critical factor in the homeostatic balance between Th17 and Treg cells, and high concentrations of IL-6 increase Th17 cell production in cooperation with transforming growth factor-β and decrease Treg cell activity [14, 15]. In this study, the concentrations of IL-6, IFN-γ, and TNF-α were significantly elevated in the psoriasis group compared with the healthy group. The serum concentrations of IL-6 positively correlated with ESR and PASI; the serum concentrations of IFN-γ positively correlated with PASI, and the TNF-α serum concentrations positively correlated with NLR, ESR, CRP, and PASI. Yang et al. reported that the secretion of IFN-γ and TNF-α by Tregs was increased in psoriatic patients, suggesting that the conversion of Tregs to Th1 cells may impair Treg cell function, leading to the excessive activation of conventional T cells in autoimmune diseases [16]. Josefowicz et al. also reported the impaired function of Treg cells, leading to Th2 cell overactivation [17]. These Treg defects were associated with disease severity, and the recovery of Treg may be an effective method for the treatment of psoriasis.
Low-dose IL-2 therapy aims to compensate for the shortage of Treg cells and expand their population. Treg cells are more sensitive to IL-2 and require by far much lower doses of IL-2 for their stimulation because Treg cells expressing CD25 on the surface can form high-affinity IL-2 receptor complex, which is composed of α (CD25), β (CD122), and γ chain (CD132). In contrast, CD4+ conventional T cells, CD8+ T cells, or natural killer cells do not express CD25 and need high-dose IL-2 for stimulation [5, 18, 19]. IL-2 directly activates and phosphorylates the transcription factor STAT5, which upregulates the key transcription factor Forkhead helix protein 3 (Foxp3) of Treg cells, thereby inducing the differentiation of Treg cells and maintaining their activity [20, 21]. However, IL-2 activation of STAT5 can inhibit the expression of retinoid-related orphan receptor γt and then inhibit the differentiation of Th17 cells [22, 23]. As mentioned above, IL-2 is a crucial factor in the production and maintenance of Treg cells and reverses the imbalance of Th17/Treg cells.
Low-dose IL-2 has attracted attention in the treatment of autoimmune diseases. Clinical studies have shown that many immune-related diseases, such as systemic lupus erythematosus (SLE), rheumatoid arthritis, polymyositis/dermatomyositis, Sjögren’s syndrome, Treg cell defects, and Th17/Treg cell imbalance, may benefit from low-dose IL-2 treatment [24–28]. Humrich et al. used low-dose IL-2 to treat SLE with moderate-to-severe disease activity, supporting that Treg defects were associated with disease severity and could be corrected using low-dose IL-2, especially for active and refractory SLE [24]. More recently, in an open-label, multicentre phase I/IIa clinical trial that used low-dose IL-2 across 11 individual diseases suffering from autoimmune inflammatory conditions, the level of Treg cells in patients significantly increased (2.0 ± 0.6) fold, without effector T cell activation, and the clinical symptoms improved significantly [29].
In this study, 45 psoriatic patients were treated with low-dose IL-2 combined with methotrexate, which greatly amplified the percentage of Treg cells. The proportion of Th17 cells and the Th17/Treg ratio decreased significantly. No significant differences in Th1, Th2, or the Th1/Th2 ratio were observed after treatment. The percentage of Treg cells was fivefold more than that of Th17 cells, which means anti-inflammatory Tregs play important roles in restoring the Th17 and Treg cell balance in psoriasis. Wang et al. reported that in PA patients that exhibited low Treg numbers, low-dose IL-2 combination with conventional therapy not only increased the absolute numbers of Tregs but also rapidly alleviated PA disease activity as indicated by the disease activity score without any apparent side effects [30]. Therefore, these findings strengthen the concept that low-dose IL-2 corrects Treg cell defects, promotes Treg cell expansion, and restores immune balance.
At the same time, low-dose IL-2 treatment also significantly increased the concentrations of anti-inflammatory cytokines (IL-4 and IL-10) but decreased the IFN-γ/IL-4 and IL-17/IL-10 ratios and regulated the production of pro-inflammatory factors (IFN-γ, IL-6, IL-17, and TNF-α), leading to significant decreases in the IFN-γ/IL-4 and IL-17/IL-10 ratios. These results indicate that low-dose IL-2 can not only restore the activity of Treg cells but also improve the pro-inflammatory environment in psoriasis and rebuild the immune balance.
Low-dose IL-2 in psoriatic patients can reduce disease activity by promoting the expansion of circulating Tregs and restoring immune balance. After low-dose IL-2 treatment, the clinical manifestations of psoriasis improved, and the PASI 50 and PASI 75 were 68.89% and 22.22% at week 24, respectively. The ACR20 percentages were 35% after treatment in PA. Low-dose IL-2 was well tolerated, and most adverse events were mild and of a transient nature, suggesting a favourable safety profile. He et al. reported low-dose IL-2 therapy in a randomised, double-blind, placebo-controlled clinical trial in 60 patients with active SLE. At week 12, the SLE responder index-4 response rates were 55.17% and 30.00% for the IL-2 and placebo groups, respectively. The prednisone dose was reduced by more than 50% at week 24. The levels of anti-dsDNA antibodies were decreased in the IL-2 group compared with the placebo group, whereas the complement C3 and C4 levels were increased. An expansion of the Treg cells was observed in this study. The number of Treg in peripheral blood was increased, the Tfh and Th17 cells were decreased, and the (Tfh+Th17)/Treg ratio was decreased [31, 32].
After administering low-dose IL-2 for psoriasis, it also decreased the inflammatory indicators of disease activity, including the leukocyte count, neutrophil count, ESR, CRP, and NLR in serum after treatment. NLR is a new inflammatory index calculated by dividing the number of neutrophils by the number of lymphocytes. Neutrophils reflect systemic inflammation, and lymphocytes reflect the immune status in psoriatic patients [33, 34]. We found that circulating NLR was significantly higher in the psoriasis group compared to the healthy group. NLR positively correlated with the levels of Th17, IL-17, and TNF-α along with the Th17/Treg and IL-17/IL-10 ratios. NLR negatively correlated with Treg cells and IL-10 concentrations. After low-dose IL-2 treatment, NLR levels decreased in the serum of psoriatic patients. Kim et al. reported that NLR was increased in psoriatic patients compared to the control group and that NLR positively correlated with PASI [35]. Altogether, these results indicate that NLR combined with conventional inflammatory indicators is useful markers for evaluating systemic inflammation in psoriatic patients. Thus, NLR may serve as simple, convenient, and cost-effective biomarkers to monitor the course of psoriasis before and after systemic therapy [33].
In conclusion, inflammatory diseases are associated with insufficient Tregs in psoriasis, leading to Th17/Treg immune imbalance, which contributes to psoriasis pathogenesis, supporting the regulation of Th17/Treg imbalance as a potential psoriasis treatment. Notably, low-dose IL-2 not only selectively activated and expanded Treg cells but also, with conventional therapies, may improve the anti-inflammatory environment and rebuild immune balance in psoriatic patients. Low-dose IL-2 can rapidly alleviate disease activity and improve clinical symptoms significantly. Low-dose IL-2 showed good safety and was well tolerated. As a new immunomodulatory therapy, low-dose IL-2 may be an effective treatment option for psoriatic patients.
Supplementary Information
Below is the link to the electronic supplementary material.
ACKNOWLEDGEMENTS
The authors thank AiMi Academic Services (www.aimieditor.com) for the English language editing and review services.
AUTHOR CONTRIBUTION
Zusha Qiao and Wengpeng Zhao contributed to the study conception and design. Material preparation, data collection were performed by Wenli Feng and Yan Ma. Data analyses were performed by Yan Liu. The first draft of the manuscript was written by Zusha Qiao and Wengpeng Zhao, and all authors commented on previous versions of the manuscript. Hongzhong Jin contributed to manuscript revision and read and approved the submitted version. All authors read and approved the final manuscript.
FUNDING
This study was supported by grants from the National Natural Science Foundation of China (No. 82072262), the National Key R&D Program of China (No. 2022YFC3601800), the National High Level Hospital Clinical Research Funding (No. 2022-PUMCH-B-092), and the Natural Science Foundation for Young Scientists of Shanxi Province (No. 20210302124693).
DATA AVAILABILITY
All data relevant to the study were included in the article and are available upon reasonable request.
DECLARATIONS
Ethics Approval
This study was conducted in accordance with the Declaration of Helsinki and approved by the Medical Ethics Committee of the Second Hospital of Shanxi Medical University (2021-YX-010). Written informed consent was obtained from all participates prior to their enrolment in this study.
Conflict of Interest
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Furiati SC, Catarino JS, Silva MV, Silva RF, Estevam RB, Teodoro RB, Pereira SL, Ataide M, Rodrigues V, Jr, Rodrigues DBR. Th1, Th17, and Treg responses are differently modulated by TNF-α inhibitors and methotrexate in psoriasis patients. Scientific Reports. 2019;9(1):7526. doi: 10.1038/s41598-019-43899-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Soleimani R, Mohammadi M, Saghebi SA, Taghipour A, Khorsand Vakilzadeh A, Tavakkol Afshari J. Comparison of Th1/Th2 and Treg/Th17 ratios between wet and dry cupping therapies in Persian medicine. Avicenna Journal of Phytomedicine. 2020;10(1):24–34. [PMC free article] [PubMed] [Google Scholar]
- 3.Komine M. Recent advances in psoriasis research; the clue to mysterious relation to gut microbiome. International Journal of Molecular Sciences. 2020;21(7):2582. doi: 10.3390/ijms21072582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhao X, Jiao J, Li X, Hou R, Li J, Niu X, Liu R, Yang X, Li J, Liang J, et al. Immunomodulatory effect of psoriasis-derived dermal mesenchymal stem cells on TH1/TH17 cells. European Journal of Dermatology: EJD. 2021;31(3):318–325. doi: 10.1684/ejd.2021.4050. [DOI] [PubMed] [Google Scholar]
- 5.Zhou P. Emerging mechanisms and applications of low-dose IL-2 therapy in autoimmunity. Cytokine and Growth Factor Reviews. 2022;67:80–88. doi: 10.1016/j.cytogfr.2022.06.003. [DOI] [PubMed] [Google Scholar]
- 6.Zhu X, Zhu J. CD4 T helper cell subsets and related human immunological disorders. International Journal of Molecular Sciences. 2020;21(21):8011. doi: 10.3390/ijms21218011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Spinner CA, Lazarevic V. Transcriptional regulation of adaptive and innate lymphoid lineage specification. Immunological Reviews. 2021;300(1):65–81. doi: 10.1111/imr.12935. [DOI] [PubMed] [Google Scholar]
- 8.Xu K, Yang WY, Nanayakkara GK, Shao Y, Yang F, Hu W, Choi ET, Wang H, Yang X. GATA3, HDAC6, and BCL6 regulate FOXP3+ Treg plasticity and determine Treg conversion into either novel antigen-presenting cell-like Treg or Th1-Treg. Frontiers in Immunology. 2018;9:45. doi: 10.3389/fimmu.2018.00045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ma L, Xue H, Gao T, Gao M, Zhang Y. Notch1 signaling regulates the Th17/Treg immune imbalance in patients with psoriasis vulgaris. Mediators of Inflammation. 2018;2018:3069521. doi: 10.1155/2018/3069521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhang L, Li Y, Yang X, Wei J, Zhou S, Zhao Z, Cheng J, Duan H, Jia T, Lei Q, et al. Characterization of Th17 and FoxP3(+) Treg cells in paediatric psoriasis patients. Scandinavian Journal of Immunology. 2016;83(3):174–180. doi: 10.1111/sji.12404. [DOI] [PubMed] [Google Scholar]
- 11.Zhang L, Yang XQ, Cheng J, Hui RS, Gao TW. Increased Th17 cells are accompanied by FoxP3(+) Treg cell accumulation and correlated with psoriasis disease severity. Clinical Immunology. 2010;135(1):108–117. doi: 10.1016/j.clim.2009.11.008. [DOI] [PubMed] [Google Scholar]
- 12.Nussbaum L, Chen YL, Ogg GS. Role of regulatory T cells in psoriasis pathogenesis and treatment. The British Journal of Dermatology. 2021;184(1):14–24. doi: 10.1111/bjd.19380. [DOI] [PubMed] [Google Scholar]
- 13.Kanda N, Hoashi T, Saeki H. The defect in regulatory T cells in psoriasis and therapeutic approaches. Journal of Clinical Medicine. 2021;10(17):3880. doi: 10.3390/jcm10173880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schwarz A, Philippsen R, Schwarz T. Induction of regulatory T cells and correction of cytokine disbalance by short-chain fatty acids: Implications for psoriasis therapy. The Journal of Investigative Dermatology. 2021;141(1):95–104.e2. doi: 10.1016/j.jid.2020.04.031. [DOI] [PubMed] [Google Scholar]
- 15.Goodman WA, Levine AD, Massari JV, Sugiyama H, McCormick TS, Cooper KD. IL-6 signaling in psoriasis prevents immune suppression by regulatory T cells. Journal of Immunology. 2009;183(5):3170–3176. doi: 10.4049/jimmunol.0803721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yang L, Li B, Dang E, Jin L, Fan X, Wang G. Impaired function of regulatory T cells in patients with psoriasis is mediated by phosphorylation of STAT3. Journal of Dermatological Science. 2016;81(2):85–92. doi: 10.1016/j.jdermsci.2015.11.007. [DOI] [PubMed] [Google Scholar]
- 17.Josefowicz SZ, Niec RE, Kim HY, Treuting P, Chinen T, Zheng Y, Umetsu DT, Rudensky AY. Extrathymically generated regulatory T cells control mucosal TH2 inflammation. Nature. 2012;482(7385):395–399. doi: 10.1038/nature10772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Graßhoff, H., S. Comdühr, L.R. Monne, A. Müller, P. Lamprecht, G. Riemekasten, and J.Y. Humrich. 2021. Low-dose IL-2 therapy in autoimmune and rheumatic diseases. Frontiers in Immunology 12. [DOI] [PMC free article] [PubMed]
- 19.Pol, J.G., P. Caudana, J. Paillet, E. Piaggio, and G. Kroemer. 2020. Effects of interleukin-2 in immunostimulation and immunosuppression. The Journal of Experimental Medicine 217 (1). [DOI] [PMC free article] [PubMed]
- 20.Toomer KH, Lui JB, Altman NH, Ban Y, Chen X, Malek TR. Essential and non-overlapping IL-2Rα-dependent processes for thymic development and peripheral homeostasis of regulatory T cells. Nature Communications. 2019;10(1):1037. doi: 10.1038/s41467-019-08960-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jones DM, Read KA, Oestreich KJ. Dynamic roles for IL-2-STAT5 signaling in effector and regulatory CD4+ T cell populations. Journal of Immunology. 2020;205(7):1721–1730. doi: 10.4049/jimmunol.2000612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nasrallah R, Imianowski CJ, Bossini-Castillo L, Grant FM, Dogan M, Placek L, Kozhaya L, Kuo P, Sadiyah F, Whiteside SK, et al. A distal enhancer at risk locus 11q13.5 promotes suppression of colitis by Treg cells. Nature. 2020;583(7816):447–452. doi: 10.1038/s41586-020-2296-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Luo J, Ming B, Zhang C, Deng X, Li P, Wei Z, Xia Y, Jiang K, Ye H, Ma W, Liu Z, Li H, Yang XP, Dong L. IL-2 inhibition of Th17 generation rather than induction of Treg cells is impaired in primary Sjögren’s syndrome patients. Frontiers in Immunology. 2018;9:1755. doi: 10.3389/fimmu.2018.01755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Humrich JY, Cacoub P, Rosenzwajg M, Pitoiset F, Pham HP, Guidoux J, Leroux D, Vazquez T, Riemekasten G, Smolen JS, et al. Low-dose interleukin-2 therapy in active systemic lupus erythematosus (LUPIL-2): a multicentre, double-blind, randomised and placebo-controlled phase II trial. Annals of the Rheumatic Diseases. 2022;81(12):1685–1694. doi: 10.1136/ard-2022-222501. [DOI] [PubMed] [Google Scholar]
- 25.Akbarzadeh R, Riemekasten G, Humrich JY. Low-dose interleukin-2 therapy: a promising targeted therapeutic approach for systemic lupus erythematosus. Current Opinion in Rheumatology. 2023;35(2):98–106. doi: 10.1097/BOR.0000000000000924. [DOI] [PubMed] [Google Scholar]
- 26.Zhang X, Miao M, Zhang R, Liu X, Zhao X, Shao M, Liu T, Jin Y, Chen J, Liu H, et al. Efficacy and safety of low-dose interleukin-2 in combination with methotrexate in patients with active rheumatoid arthritis: a randomized, double-blind, placebo-controlled phase 2 trial. Signal Transduction and Targeted Therapy. 2022;7(1):67. doi: 10.1038/s41392-022-00887-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang SX, Wang J, Sun HH, Zhang JQ, Liu GY, Luo J, He PF, Li XF. Circulating regulatory T cells were absolutely decreased in dermatomyositis/polymyositis patients and restored by low-dose IL-2. Annals of the Rheumatic Diseases. 2021;80(8):e130. doi: 10.1136/annrheumdis-2019-216246. [DOI] [PubMed] [Google Scholar]
- 28.Miao M, Hao Z, Guo Y, Zhang X, Zhang S, Luo J, Zhao X, Zhang C, Liu X, Wu X, et al. Short-term and low-dose IL-2 therapy restores the Th17/Treg balance in the peripheral blood of patients with primary Sjögren's syndrome. Annals of the Rheumatic Diseases. 2018;77(12):1838–1840. doi: 10.1136/annrheumdis-2018-213036. [DOI] [PubMed] [Google Scholar]
- 29.Rosenzwajg M, Lorenzon R, Cacoub P, Pham HP, Pitoiset F, El Soufi K, RIbet C, Bernard C, Aractingi S, Banneville B, et al. Immunological and clinical effects of low-dose interleukin-2 across 11 autoimmune diseases in a single, open clinical trial. Annals of the Rheumatic Diseases. 2019;78(2):209–217. doi: 10.1136/annrheumdis-2018-214229. [DOI] [PubMed] [Google Scholar]
- 30.Wang J, Zhang SX, Hao YF, Qiu MT, Luo J, Li YY, Gao C, Li XF. The numbers of peripheral regulatory T cells are reduced in patients with psoriatic arthritis and are restored by low-dose interleukin-2. Therapeutic Advances in Chronic Disease. 2020;11:2040622320916014. doi: 10.1177/2040622320916014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.He J, Zhang X, Wei Y, Sun X, Chen Y, Deng J, Jin Y, Gan Y, Hu X, Jia R, et al. Low-dose interleukin-2 treatment selectively modulates CD4(+) T cell subsets in patients with systemic lupus erythematosus. Nature Medicine. 2016;22(9):991–993. doi: 10.1038/nm.4148. [DOI] [PubMed] [Google Scholar]
- 32.He J, Zhang R, Shao M, Zhao X, Miao M, Chen J, Liu J, Zhang X, Zhang X, Jin Y, et al. Efficacy and safety of low-dose IL-2 in the treatment of systemic lupus erythematosus: a randomised, double-blind, placebo-controlled trial. Annals of the Rheumatic Diseases. 2020;79(1):141–149. doi: 10.1136/annrheumdis-2019-215396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.An I, Ucmak D, Ozturk M. The effect of biological agent treatment on neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio, mean platelet volume, and C-reactive protein in psoriasis patients. Postepy Dermatologii i Alergologii. 2020;37(2):202–206. doi: 10.5114/ada.2020.94838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.An İ, Aksoy M, Ayhan E, Ozturk M. The effect of secukinumab treatment on inflammatory parameters in patients with psoriasis: a multicentre retrospective study. International Journal of Clinical Practice. 2021;75(6):e14114. doi: 10.1111/ijcp.14114. [DOI] [PubMed] [Google Scholar]
- 35.Kim DS, Shin D, Lee MS, Kim HJ, Kim DY, Kim SM, Lee MG. Assessments of neutrophil to lymphocyte ratio and platelet to lymphocyte ratio in Korean patients with psoriasis vulgaris and psoriatic arthritis. The Journal of Dermatology. 2016;43(3):305–310. doi: 10.1111/1346-8138.13061. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data relevant to the study were included in the article and are available upon reasonable request.