Abstract
Purpose
To investigate the relationship between corneal tomographic or biomechanical parameters and risk of keratoconus in very asymmetric ectasia (VAE).
Methods
This retrospective case-control single-centre study included patients with VAE and normal controls. The VAE group had clinical ectasia in one eye and normal topography (VAE-NT) in the fellow eye; VAE-NT eyes were selected for analysis. The control group was selected from corneal refractive surgery candidates; the right eye was enrolled. Scheimpflug-based corneal tomography (Pentacam) and corneal biomechanical assessment (Corvis ST) were performed. Univariate and multivariable logistic regression were performed using Cox proportional hazards models to evaluate keratoconus-associated risk factors. A two-piecewise linear regression model was applied to examine the threshold effect of selected vital paragmeters on the risk of keratoconus according to a smoothing plot.
Results
Threshold effect between tomographic integration and risk of keratoconus was observed. Discrepancy between the central corneal thickness and thinnest corneal thickness (discrepancy CCT vs TCT) greater than 5 μm, discrepancy between the apex corneal thickness and thinnest corneal thickness (discrepancy ACT vs TCT) greater than 3 μm, vector distance between CCT and TCT (distance CCT vs TCT) greater than 0.65 mm indicated a significant increased risk of keratoconus. Risk of keratoconus decreased when distance CCT vs TCT was less than 0.65 mm.
Conclusion
Discrepancy CCT vs TCT, discrepancy ACT vs TCT, and distance CCT vs TCT can be used as indicators for risk assessment of early keratoconus.
Keywords: keratoconus, corneal tomographic, corneal biomechanical, very asymmetric ectasia
Introduction
Keratoconus is an ocular disorder characterized by bilaterality and asymmetric corneal thinning. Its pathophysiology is multifactorial, having genetic, biochemical (including pro-inflammatory mediators), biomechanical, and environmental components (mainly eye rubbing and nocturnal ocular compression).1–4 In a 2015 consensus among ophthalmology experts, it was agreed that true unilateral keratoconus does not exist, but a unilateral clinical presentation may occur in predisposed individuals due to asymmetric environmental factors, such as eye rubbing.5 As shown in the review by Rabinowitz et al, a few completely unilateral cases have been reported, the majority of fellow eyes in unilateral keratoconus tend to eventually develop keratoconus, or are diagnosed with forme fruste keratoconus (FFK). Therefore, ongoing monitoring is advisable.2,6,7
Corneal tomography is the standard procedure for diagnosing keratoconus. It has served as a sensitive and accurate diagnostic indicator in the early stages of the disease. Pentacam (Oculus Optikgeräte GmbH; Wetzlar, Germany) was found to be an efficient and precise means in corneal imaging; it has high repeatability and reproducibility. Corneal biomechanical parameters in vivo are valuable for the diagnosis of keratoconus.8 Compared with healthy eyes, keratoconic corneas show lower resistance to deformation.9 Changes of data related to biomechanical properties may be detectable before the appearance of tomographic and clinical signs and can be used as a diagnostic basis of early keratoconus.
The identification of patients with subclinical signs of keratoconus is crucial. This is a critical contraindication for corneal refractive surgery, and among refractive surgery candidates, the frequency of keratoconus or FFK is notably higher than in the general population.10–12 Previous studies have demonstrated differences in several corneal topographic and biomechanical values between eyes with keratoconus and normal eyes;13–15 however, the parameters and threshold values for keratoconus diagnosis were not investigated. Thus, this study aimed to develop a precise personalized keratoconus risk using cost-effective and readily accessible parameters for the functional link between selected risk factors and keratoconus.
Methods
Study Population
Patients from the Eye and ENT Hospital of Fudan University, Shanghai, China, were enrolled in this retrospective case-control study. The Ethics Committee of the Eye and ENT Hospital of Fudan University approved this study (approval number 2021118–1). This study adhered to the tenets of the Declaration of Helsinki. All participants provided informed consent. For pediatric participants, written informed consent was obtained from at least one parent or legal guardian of each subject after a detailed explanation of the procedure, and all the procedures were carried out with the subjects’ consent.
The apparently healthy eyes of patients diagnosed with very asymmetric ectasia (VAE) were included in the study. These patients must exhibit definite clinical ectasia in one eye, characterized by tomographic features and slit lamp findings such as Munson’s sign, Vogt’s striae, Fleischer’s ring, apical thinning, or Rizutti’s sign.16 In addition, the contralateral eye, referred to as VAE-NT (Very Asymmetric Ectasia - Normal Tomography), had to meet the criteria of having an ABCD keratoconus-specific grading categorized as stage 0, determined based on assessments of anterior corneal curvature, posterior corneal curvature, pachymetry at the thinnest area of the cornea, and corrected distance visual acuity (CDVA) using Pentacam topometry/KC staging.17 Patients with a history of corneal surgery, pregnancy, ophthalmologic disease, or systemic diseases with ocular manifestations were excluded.
A control group with no ocular disorders, except refractive errors, was selected from patients underwent corneal refractive surgery in our clinic. The included participants had normal slit-lamp biomicroscopy findings, overall subjectively normal tomography examinations, and no evidence of ectasia throughout a 1-year follow-up period. The right eyes of the control patients were included in this study.
Variable Measurement
All participants had comprehensive ophthalmologic examinations performed by experienced technicians. Scheimpflug-based corneal tomography and corneal biomechanical assessment were performed using the Pentacam and Corvis ST (Oculus Optikgeräte GmbH; Wetzlar, Germany), respectively. Both measurements were performed by experienced examiners under the same lighting conditions. Parameters from the Pentacam and Corvis ST were obtained for each eye.
Statistical Analysis
Data distributions of each covariate were compared between the VAE-NT and normal groups using a t-test or the Kruskal–Wallis rank sum test for continuous variables and χ2 for categorical data. We performed univariate logistic regression and multivariable logistic regression using Cox proportional hazards models to evaluate the odds ratio (OR) and 95% confidence intervals (CIs) for risk factors associated with keratoconus. We applied multiple regression analyses to estimate the independent relationship between keratoconus risk and corneal tomographic parameters as well as biomechanical parameters, with adjustment for age, sex, and other potential confounders. A two-piecewise linear regression model was further applied to examine the threshold effect of selected vital parameters on the risk of keratoconus according to the smoothing plot. The trial inflection point was determined by moving along a predefined interval and detected the inflection point that gave the maximum model likelihood.
A two-tailed P <0.05 was considered significant in all analyses. EmpowerStats (http://www.empowerstats.com) and the R software, version 3.3.1 (http://www.R-project.org/), were used for all statistical analyses.
Results
Ninety-four eyes (VAE-NT) of 94 patients with VAE, and 202 eyes of 202 controls were analyzed. The average ages of the VAE-NT and control group patients were 21.89±6.18 years and 28.00±7.16 years, respectively. The clinical characteristics for the included patients are summarized in Table 1. For tomographic parameters, significant differences between the VAE-NT and control groups were found for flat keratometry (Kflat), thinnest corneal thickness (TCT), absolute value of discrepancy between the central corneal thickness and thinnest corneal thickness (discrepancy CCT vs TCT), absolute value of the discrepancy between the apex corneal thickness and TCT (discrepancy ACT vs TCT), absolute value of vector distance between the CCT and TCT (distance CCT vs TCT), back elevation at the thinnest pachymetry (BE.at.TP), maximum Ambrósio relational thickness (ARTmax), posterior radius of curvature (PRC) in 3-mm zone, deviation of average pachymetric progression (Dp), deviation of minimum thickness (Dt), deviation of ARTmax (Da), and Belin/Ambrósio Enhanced Ectasia Deviation (BAD D). For biomechanical parameters, significant differences between the two groups were found for IOPnct, biomechanically corrected IOP (bIOP), stress–strain index (SSI), deformation amplitude ratio (DA.Ratio), integrated radius (Integr. Radius), and the tomography and biomechanical index (TBI).
Table 1.
Demographic and Clinical Characteristics of Participants
| Normal (Mean+SD / N(%)) | VAE-NT (Mean+SD / N(%)) | P-value | |
|---|---|---|---|
| Number | 202 | 94 | |
| Age | 28.00 ± 7.16 | 21.89 ± 6.18 | <0.001 |
| Female | 126 (62.38%) | 22 (23.40%) | - |
| Male | 76 (37.62%) | 72 (76.60%) | - |
| Corneal tomographic parameters | |||
| Kflat (D) | 42.70 ± 1.45 | 42.24 ± 1.19 | 0.008 |
| Ksteep (D) | 43.90 ± 1.55 | 43.58 ± 1.40 | 0.155 |
| Kmax (D) | 44.43 ± 1.64 | 44.27 ± 1.44 | 0.558 |
| CCT (μm) | 538.05 ± 26.15 | 533.81 ± 22.46 | 0.126 |
| ACT (μm) | 537.77 ± 26.23 | 533.11 ± 22.51 | 0.094 |
| TCT (μm) | 534.27 ± 26.14 | 527.93 ± 22.73 | 0.029 |
| Discrepancy CCT vs TCT (μm) | 3.78 ± 1.93 | 5.88 ± 3.64 | <0.001 |
| Discrepancy ACT vs TCT (μm) | 3.50 ± 2.09 | 5.18 ± 3.42 | <0.001 |
| Distance CCT vs TCT (mm) | 0.66 ± 0.20 | 0.73 ± 0.24 | 0.047 |
| FE.at.TP (μm) | 2.60 ± 1.32 | 2.80 ± 1.61 | 0.457 |
| BE.at.TP (μm) | 5.32 ± 3.10 | 6.77 ± 3.77 | 0.001 |
| ARTmax | 430.03 ± 65.99 | 373.91 ± 75.65 | <0.001 |
| ARC (3mm Zone) (mm) | 7.79 ± 0.26 | 7.84 ± 0.22 | 0.134 |
| PRC (3mm Zone) (mm) | 6.37 ± 0.24 | 6.31 ± 0.22 | 0.021 |
| Df | 0.47 ± 0.95 | 0.47 ± 1.17 | 0.590 |
| Db | 0.11 ± 0.81 | 0.09 ± 0.82 | 0.926 |
| Dp | 0.71 ± 0.78 | 1.36 ± 0.99 | <0.001 |
| Dt | 0.14 ± 0.75 | 0.32 ± 0.66 | 0.031 |
| Da | 0.53 ± 0.60 | 1.04 ± 0.69 | <0.001 |
| BAD D | 1.11 ± 0.55 | 1.44 ± 0.64 | <0.001 |
| Corneal biomechanical parameters | |||
| IOPnct | 17.55 ± 2.98 | 15.02 ± 2.02 | <0.001 |
| bIOP | 17.68 ± 2.74 | 15.45 ± 1.71 | <0.001 |
| SSI | 0.94 ± 0.15 | 0.89 ± 0.15 | 0.020 |
| DA.Ratio (2mm) | 4.36 ± 0.82 | 4.66 ± 0.81 | <0.001 |
| Integr. Radius | 8.33 ± 0.95 | 9.26 ± 0.98 | <0.001 |
| ARTh | 471.10 ± 83.98 | 490.30 ± 92.65 | 0.068 |
| SP-A1 | 104.40 ± 14.48 | 103.24 ± 15.97 | 0.335 |
| CBI | 0.12 ± 0.20 | 0.19 ± 0.28 | 0.306 |
| TBI | 0.25 ± 0.19 | 0.42 ± 0.33 | <0.001 |
Abbreviations: K, keratometry; D, diopter; CCT, central corneal thickness; ACT, apex corneal thickness; TCT, thinnest corneal thickness; Discrepancy CCT vs TCT, absolute value of discrepancy between central corneal thickness and thinnest corneal thickness; Discrepancy ACT vs TCT, absolute value of discrepancy between apex corneal thickness and thinnest corneal thickness; Distance CCT vs TCT, absolute value of vector distance between central corneal thickness and thinnest corneal thickness; FE.at.TP, front elevation at thinnest pachymetry; BE.at.TP, back elevation at thinnest pachymetry; ARTmax, maximum Ambrósio relational thickness; ARC, anterior radius of curvature; PRC, posterior radius of curvature; Df, deviation of front elevation difference map; Db, derivation of back elevation difference map; Dp, deviation of average pachymetric progression; Dt, deviation of minimum thickness; Da, deviation of ARTmax; BAD D, Belin/Ambrósio Enhanced Ectasia Deviation; IOPnct, intraocular pressure measured with non-contact tonometer; bIOP, biomechanically-corrected IOP; SSI, stress-strain index; DA, deformation amplitude; Integr. Radius, integrated radius; ARTh, Ambrósio’s relational thickness in the horizontal profile; SP-A1, stiffness parameter at first applanation; CBI, corvis biomechanical index; TBI, Tomography and Biomechanical Index.
The results of the univariate and multivariate logistic regression are shown in Table 2. After adjusting for sex, age, and corneal biomechanical parameters, positive correlations with the risk of keratoconus were found for discrepancy CCT vs TCT (OR 1.34, 95% CI 1.15–1.56, P for linear trend <0.001), discrepancy ACT vs TCT (OR 1.19, 95% CI 1.02–1.37, P for linear trend = 0.023), Dp (OR 7.38, 95% CI 3.28–16.64, P for linear trend <0.001), Dt (OR 6.06, 95% CI 1.63–22.49, P for linear trend = 0.007), Da (OR 7.23, 95% CI 2.78–18.76, P for linear trend <0.001), and BAD D (OR 1.12, 95% CI 1.01–1.23, P for linear trend = 0.024). However, an inverse correlation was shown for CCT (OR 0.96, 95% CI 0.92–1.00, P for linear trend = 0.043), ACT (OR 0.95, 95% CI 0.92–0.99, P for linear trend = 0.014), TCT (OR 0.95, 95% CI 0.91–0.98, P for linear trend = 0.004), ARTmax (OR 0.83, 95% CI 0.76–0.91, P for linear trend <0.001), PRC (OR 0.09, 95% CI 0.01–0.68, P for linear trend = 0.020), and Db (OR 0.52, 95% CI 0.30–0.91, P for linear trend = 0.021). For biomechanical parameters, the Integr. Radius (OR 4.64, 95% CI 2.48–8.67, P for linear trend <0.001) and ARTh (OR 1.28, 95% CI 1.16–1.41, P for linear trend <0.001) showed a positive correlation with the risk of keratoconus. However, the IOPnct (OR 0.58, 95% CI 0.46–0.73, P for linear trend <0.001) and bIOP (OR 0.53, 95% CI 0.41–0.69, P for linear trend <0.001) showed an inverse correlation with the risk of keratoconus.
Table 2.
Univariate Analysis and Multivariate Logistic Regression Model for Risk Factors Associated with Keratoconus
| Crude (95% CI) | P-value | Adjust I (95% CI) | P-value | Adjust II (95% CI) | P-value | |
|---|---|---|---|---|---|---|
| Corneal tomographic parameters | ||||||
| Kflat (D) | 0.78 (0.64, 0.94) | 0.009 | 1.00 (0.79, 1.26) | 0.990 | 0.99 (0.73, 1.35) | 0.968 |
| Ksteep (D) | 0.87 (0.74, 1.02) | 0.094 | 1.00 (0.82, 1.22) | 0.979 | 0.89 (0.68, 1.17) | 0.407 |
| Kmax (D) | 0.94 (0.80, 1.10) | 0.413 | 1.07 (0.89, 1.29) | 0.484 | 0.92 (0.71, 1.19) | 0.521 |
| CCT (μm) | 0.99 (0.98, 1.00) | 0.176 | 0.99 (0.98, 1.01) | 0.358 | 0.96 (0.92, 1.00) | 0.043 |
| ACT (μm) | 0.99 (0.98, 1.00) | 0.138 | 0.99 (0.98, 1.00) | 0.267 | 0.95 (0.92, 0.99) | 0.014 |
| TCT (μm) | 0.99 (0.98, 1.00) | 0.045 | 0.99 (0.98, 1.00) | 0.118 | 0.95 (0.91, 0.98) | 0.004 |
| Discrepancy CCT vs TCT (μm) | 1.35 (1.21, 1.51) | <0.001 | 1.38 (1.23, 1.56) | <0.001 | 1.34 (1.15, 1.56) | <0.001 |
| Discrepancy ACT vs TCT (μm) | 1.26 (1.15, 1.40) | <0.001 | 1.25 (1.12, 1.39) | <0.001 | 1.19 (1.02, 1.37) | 0.023 |
| Distance CCT vs TCT# (mm) | 1.14 (1.02, 1.28) | 0.024 | 1.15 (1.01, 1.31) | 0.040 | 1.03 (0.87, 1.23) | 0.719 |
| FE.at.TP (μm) | 1.10 (0.93, 1.31) | 0.261 | 1.20 (0.98, 1.48) | 0.071 | 0.97 (0.74, 1.27) | 0.826 |
| BE.at.TP (μm) | 1.14 (1.05, 1.22) | 0.001 | 1.14 (1.05, 1.24) | 0.003 | 0.99 (0.88, 1.13) | 0.922 |
| ARTmax* | 0.88 (0.84, 0.92) | <0.001 | 0.88 (0.84, 0.93) | <0.001 | 0.83 (0.76, 0.91) | <0.001 |
| ARC (3mm Zone) (mm) | 2.24 (0.83, 6.04) | 0.112 | 0.70 (0.21, 2.40) | 0.575 | 1.17 (0.22, 6.28) | 0.853 |
| PRC (3mm Zone) (mm) | 0.32 (0.11, 0.93) | 0.037 | 0.10 (0.03, 0.37) | 0.001 | 0.09 (0.01, 0.68) | 0.020 |
| Df | 1.00 (0.78, 1.26) | 0.969 | 1.13 (0.86, 1.48) | 0.397 | 0.96 (0.66, 1.39) | 0.823 |
| Db | 0.98 (0.72, 1.32) | 0.892 | 0.96 (0.69, 1.35) | 0.832 | 0.52 (0.30, 0.91) | 0.021 |
| Dp | 2.36 (1.73, 3.22) | <0.001 | 2.35 (1.65, 3.36) | <0.001 | 7.38 (3.28, 16.64) | <0.001 |
| Dt | 1.40 (0.99, 1.98) | 0.054 | 1.36 (0.91, 2.02) | 0.132 | 6.06 (1.63, 22.49) | 0.007 |
| Da | 4.07 (2.52, 6.58) | <0.001 | 3.97 (2.32, 6.78) | <0.001 | 7.23 (2.78, 18.76) | <0.001 |
| BAD D# | 1.11 (1.06, 1.16) | <0.001 | 1.12 (1.06, 1.18) | <0.001 | 1.12 (1.02, 1.23) | 0.024 |
| Corneal biomechanical parameters | ||||||
| IOPnct | 0.65 (0.57, 0.75) | <0.001 | 0.66 (0.57, 0.77) | <0.001 | 0.58 (0.46, 0.73) | <0.001 |
| bIOP | 0.63 (0.54, 0.73) | <0.001 | 0.61 (0.52, 0.73) | <0.001 | 0.53 (0.41, 0.69) | <0.001 |
| SSI# | 0.80 (0.68, 0.95) | 0.010 | 0.85 (0.70, 1.03) | 0.089 | 0.93 (0.70, 1.23) | 0.611 |
| DA.Ratio | 1.95 (1.13, 3.34) | 0.016 | 2.03 (1.05, 3.91) | 0.035 | 1.42 (0.94, 2.16) | 0.099 |
| Integr. Radius | 2.74 (2.02, 3.71) | <0.001 | 2.66 (1.89, 3.76) | <0.001 | 4.64 (2.48, 8.67) | <0.001 |
| ARTh* | 1.03 (1.00, 1.05) | 0.079 | 1.02 (0.99, 1.06) | 0.151 | 1.28 (1.16, 1.41) | <0.001 |
| SP-A1 | 0.99 (0.98, 1.01) | 0.537 | 0.99 (0.97, 1.01) | 0.163 | 0.98 (0.95, 1.02) | 0.384 |
| CBI# | 1.14 (1.03, 1.26) | 0.015 | 1.13 (1.00, 1.27) | 0.053 | 1.12 (0.92, 1.37) | 0.254 |
| TBI# | 1.29 (1.16, 1.43) | <0.001 | 1.28 (1.14, 1.43) | <0.001 | 1.04 (0.84, 1.30) | 0.706 |
Notes: Adjust I model (corneal tomographic parameters): adjusted for sex; age. Adjust II model (corneal tomographic parameters): adjusted for sex; age; IOPnct; bIOP; SSI; DA.Ratio; Interger Radius; ARTh; SP-A1; CBI; TBI. Adjust I model (corneal biomechanical parameters): adjusted for sex; age. Adjust II model (corneal biomechanical parameters): adjusted for sex; age; Kflat; Ksteep; Kmax; CCT; ACT; TCT; Discrepancy CCT vs TCT; Discrepancy ACT vs TCT; Distance CCT vs TCT; FE.at.TP; BE.at.TP; ARTmax; ARC; PRC; Df; Db; Dp; Dt; Da; BAD D. *Every 10 unit increase. #Every 0.1 unit increase.
Abbreviations: K, keratometry; D, diopter; CCT, central corneal thickness; ACT, apex corneal thickness; TCT, thinnest corneal thickness; Discrepancy CCT vs TCT, absolute value of discrepancy between central corneal thickness and thinnest corneal thickness; Discrepancy ACT vs TCT, absolute value of discrepancy between apex corneal thickness and thinnest corneal thickness; Distance CCT vs TCT, absolute value of vector distance between central corneal thickness and thinnest corneal thickness; FE.at.TP, front elevation at thinnest pachymetry; BE.at.TP, back elevation at thinnest pachymetry; ARTmax, maximum Ambrósio relational thickness; ARC, anterior radius of curvature; PRC, posterior radius of curvature; Df, deviation of front elevation difference map; Db, derivation of back elevation difference map; Dp, deviation of average pachymetric progression; Dt, deviation of minimum thickness; Da, deviation of ARTmax; BAD D, Belin/Ambrósio Enhanced Ectasia Deviation; IOPnct, intraocular pressure measured with non-contact tonometer; bIOP, biomechanically-corrected IOP; SSI, stress-strain index; DA, deformation amplitude; Integr. Radius, integrated radius; ARTh, Ambrósio’s relational thickness in the horizontal profile; SP-A1, stiffness parameter at first applanation; CBI, corvis biomechanical index; TBI, Tomography and Biomechanical Index; Crude: no adjustment.
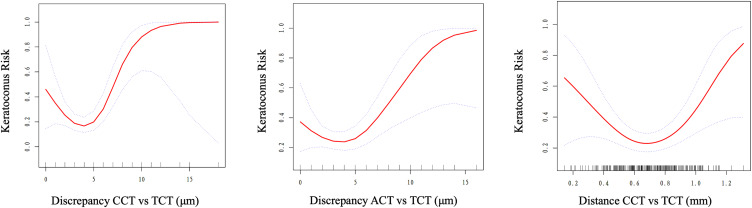
Further analysis of the nonlinear relationship and threshold effect was performed (Table 3). We observed the threshold effect for discrepancy CCT vs TCT; the turning point was 5 μm, with the risk of keratoconus increasing by 153% (P <0.001) with each 1-μm increment when discrepancy CCT vs TCT was >5 μm. A similar threshold effect was observed when discrepancy ACT vs TCT value was 3 μm (when discrepancy ACT vs TCT was >3 μm, the risk of keratoconus increased by 41% with each 1-μm increment). For distance CCT vs TCT, the optimal cut-off value was 0.65 mm, and an inverse correlation was observed when distance CCT vs TCT was <0.65 mm; the risk of keratoconus decreased by 45% with each 0.1-mm decrease. A positive correlation was observed when distance CCT vs TCT was >0.65 mm, with the risk of keratoconus increasing by 66% with each 0.1-mm increase (Figure 1). As for biomechanical parameters, we observed a threshold effect in TBI; the turning point was 0.43, with the risk of keratoconus decreasing by 34% (OR 0.66, 95% CI 0.46–0.95, P = 0.025) with each 0.1 decrement of TBI value when TBI was <0.43, and the risk of keratoconus increasing by 97% (OR 1.97, 95% CI 1.22–3.19, P = 0.006) with each 0.1 increment of TBI value when TBI was >0.43.
Table 3.
Threshold Effect Analysis on Risk of Keratoconus
| Variables in Model | OR (95% CI) | P-value |
|---|---|---|
| Discrepancy CCT vs TCT (μm) | ||
| < 5 | 0.72 (0.51, 1.00) | 0.051 |
| ≥ 5 | 2.53 (1.67, 3.85) | <0.001 |
| Discrepancy ACT vs TCT (μm) | ||
| < 3 | 0.59 (0.35, 1.01) | 0.056 |
| ≥ 3 | 1.41 (1.15, 1.73) | 0.001 |
| Distance CCT vs TCT# (mm) | ||
| < 0.65 | 0.55 (0.37, 0.83) | 0.004 |
| ≥ 0.65 | 1.66 (1.20, 2.30) | 0.002 |
Note: #Every 0.1 unit increase.
Abbreviations: Discrepancy CCT vs TCT, absolute value of discrepancy between central corneal thickness and thinnest corneal thickness; Discrepancy ACT vs TCT, absolute value of discrepancy between apex corneal thickness and thinnest corneal thickness; Distance CCT vs TCT, absolute value of vector distance between central corneal thickness and thinnest corneal thickness.
Figure 1.
Absolute value of the discrepancy between the central corneal thickness and thinnest corneal thickness (discrepancy CCT vs TCT), absolute value of the discrepancy between the apex corneal thickness and thinnest corneal thickness (discrepancy ACT vs TCT), absolute value of the vector distance between the central corneal thickness and thinnest corneal thickness (distance CCT vs TCT), and keratoconus risk dose–response nonlinear relationship.
Discussion
Keratoconus is a complex and progressive disease, making it sometimes challenging to establish precise cut-off values for corneal tomographic and biomechanical parameters, crucial diagnostic markers for the condition. However, even subtle deviations in critical features like corneal pachymetry thickness and keratometry values may indicate FFK. Our study found that several newly suggested vital tomographic and biomechanical parameters exhibited an association with the presence of FFK in fellow eyes of patient with clinically evident seemingly unilateral keratoconus. We also identified a threshold effect in eyes with VAE-NT. To our knowledge, this is the first study to provide clear evidence of both linear and nonlinear associations between corneal tomographic findings, biomechanical parameters, and the presence of FFK.
It is reasonable to conclude that normal tomography does not rule out early keratoconus. A longitudinal study showed that over 20% of the eyes with FFK and unilateral keratoconus progressed into clinical keratoconus.18,19 Dramatic changes were noted in the past decade on the diagnosis of keratoconus and other ectatic diseases, with the emergence of corneal imaging indices especially Scheimpflug imaging. These improvements in corneal imaging can measure both the anterior and posterior corneal surfaces and reconstruct the anterior segment of the eye. Corvis ST is a useful instrument to analyse corneal biomechanical properties; it has a good capacity to detect the weakened mechanical strength of cornea. The sensitivity and specificity of keratoconus diagnosis have improved dramatically, but screening for subclinical keratoconus remains a clinical challenge. The contralateral eyes of patients with unilateral keratoconus have proven to be a good model to detect subclinical changes before developing keratoconus.20–22
This study aimed to identify the linear or nonlinear relationship between the selected tomographic and biomechanical parameters and risk of developing keratoconus. We demonstrated that the risk of keratoconus may increase when discrepancy CCT vs TCT and discrepancy ACT vs TCT were >5 μm and >3 μm, respectively. For distance CCT vs TCT, the absolute value of the vector distance between the two corneal thickness locations, the risk of keratoconus increased by 66% for every 0.1-mm increment in distance CCT vs TCT. Our study first proposed the existence of a non-linear relationship between discrepancy CCT vs TCT, discrepancy ACT vs TCT, distance CCT vs TCT, and the risk of keratoconus; changes in these corneal tomographic parameters may indicate abnormalities in corneal morphology and thickness distribution. Based on our study, discrepancy CCT vs TCT >5 μm, discrepancy ACT vs TCT >3 μm, or distance CCT vs TCT >0.65 mm should receive increased attention in clinical work, especially in the preoperative screening of subclinical keratoconus in corneal refractive surgery candidates and assessment of the risk of postoperative corneal ectasia. Our study results have important implications for clinical and research work, identifying new parameters for assessing the risk of keratoconus and providing quantitative indicators for early screening and diagnosis of keratoconus.
The early detection of subclinical keratoconus before the appearance of abnormal posterior ectasia, abnormal corneal thickness spatial distribution, and typical clinical manifestations is of great significance.23 Our study showed a linear relationship of the risk of keratoconus with the CCT, ACT, TCT, discrepancy CCT vs TCT, discrepancy ACT vs TCT, ARTmax, PRC, Db, Dp, Dt, Da, BAD D, IOPnct, bIOP, Integr. Radius, and ARTh. The multivariate logistic regression analysis indicated a decreased risk of keratoconus with increasing CCT, ACT, and TCT; the risk of keratoconus decreased by 4%, 5%, or 5% for every 1-μm increase, respectively. As for biomechanical parameters, we observed that the risk of keratoconus decreased by 34% with each 0.1 decrement of TBI value when TBI was <0.43, and the risk of keratoconus increased by 97% with each 0.1 increment of TBI value when TBI was >0.43; this provides a better understanding of this parameter. Several comparative clinical studies have been conducted to access biomechanical properties to identified specific quantitative indicators for detecting FFK.13,24–27 Herber et al confirmed that CBI was a sensitive parameter to distinguish between normal eye and FFK eyes by using a cut-off of 0.2.26 Other studies found that TBI has a higher AUROC curve value than CBI for normal controls versus FFK eyes.13,25 Deformation amplitudes were also reported discriminant functional variables.28 A previous study also suggested diminishing predictive effects from TBI or BAD D.8,24 The diagnostic efficacy of these biomechanical parameters differed in different studies and may be related to the inclusion of different ethnic groups, sample sizes, and diagnostic criteria for VAE-NT. The accuracy of this parameter and the underlying clinical significance should be explored. In our study, we quantified the increased risk of developing keratoconus in patients with a moderate abnormal TBI and provided a more informative normal range for the threshold value of TBI and a deeper understanding of the variation pattern of this parameter. By combining it with other examination parameters, we have provided a more comprehensive assessment of the patient’s risk of developing keratoconus.
The strength of our study is related to its accurate detection of early ectatic changes. Prospective patients could be notified, receive regular follow-ups, and avoid delaying necessary interventions, such as corneal cross-linking, before the development of significant loss of vision. The statistical functions could be useful tools for the early detection of subclinical keratoconus, help select patients appropriate for corneal refractive surgeries, ensure safe performance of the operation, and objectively assess the risk of developing keratoconus.
A limitation of this study was the relatively small number of cases, which may lead to statistical bias; thus, subgroup or individual analyses were not feasible. A larger clinical observation and cohort study is needed to investigate the long-term relationships of the tomography and biomechanical parameters on the risk of keratoconus.
Conclusion
Our study identified specific quantitative indicators for detecting FFK in eyes with seemingly normal corneal tomography, inpatients presenting with apparently unilateral keratoconus. These indicators include a CCT vs TCT discrepancy of >5 μm, an ACT vs TCT discrepancy of >3 μm, and a CCT vs TCT distance of >0.65 mm. However, further research focusing on these parameters is necessary.
Funding Statement
This study was supported by the National Natural Science Foundation of China (Grant No. 82371096), Joint Research Project of New Frontier Technology in Municipal Hospitals (SHDC12018103), Project of Shanghai Science and Technology (Grant No.20410710100), Clinical Research Plan of SHDC (SHDC2020CR1043B), and Project of Shanghai Xuhui District Science and Technology (XHLHGG202104).
Data Sharing Statement
Data is available on request from the corresponding author.
Compliance with Ethics Guidelines
The Ethics Committee of the Eye and ENT Hospital of Fudan University approved this study (approval number 2021118-1). This study adhered to the tenets of the Declaration of Helsinki. All participants provided informed consent.
Disclosure
The authors declare that there are no competing interests. Yishan Qian and Xingtao Zhou contributed equally to the work and should therefore be regarded as equal corresponding authors.
References
- 1.Mas Tur V, MacGregor C, Jayaswal R, O’Brart D, Maycock N. A review of keratoconus: diagnosis, pathophysiology, and genetics. Surv Ophthalmol. 2017;62(6):770–783. doi: 10.1016/j.survophthal.2017.06.009 [DOI] [PubMed] [Google Scholar]
- 2.Rabinowitz YS, Galvis V, Tello A, Rueda D, Garcia JD. Genetics vs chronic corneal mechanical trauma in the etiology of keratoconus. Exp Eye Res. 2021;202:108328. doi: 10.1016/j.exer.2020.108328 [DOI] [PubMed] [Google Scholar]
- 3.Galvis V, Tello A, Barrera R, Nino CA. Inflammation in Keratoconus. Cornea. 2015;34:e22–e23. [DOI] [PubMed] [Google Scholar]
- 4.Gatinel D, Galvis V, Tello A, et al. Obstructive sleep apnea-hypopnea syndrome and keratoconus: an epiphenomenon related to sleep position? Cornea. 2020;39(4):e11–e12. doi: 10.1097/ICO.0000000000002219 [DOI] [PubMed] [Google Scholar]
- 5.Gomes JA, Tan D, Rapuano CJ, et al.; Group of Panelists for the Global Delphi Panel of K, Ectatic D. Global consensus on keratoconus and ectatic diseases. Cornea. 2015;34(4):359–369. doi: 10.1097/ICO.0000000000000408 [DOI] [PubMed] [Google Scholar]
- 6.Temstet C, Sandali O, Bouheraoua N, et al. Corneal epithelial thickness mapping using Fourier-domain optical coherence tomography for detection of form fruste keratoconus. J Cataract Refract Surg. 2015;41(4):812–820. doi: 10.1016/j.jcrs.2014.06.043 [DOI] [PubMed] [Google Scholar]
- 7.Li X, Rabinowitz YS, Rasheed K, Yang H. Longitudinal study of the normal eyes in unilateral keratoconus patients. Ophthalmology. 2004;111(3):440–446. doi: 10.1016/j.ophtha.2003.06.020 [DOI] [PubMed] [Google Scholar]
- 8.Ferreira-Mendes J, Lopes BT, Faria-Correia F, Salomao MQ, Rodrigues-Barros S, Ambrosio R. Enhanced ectasia detection using corneal tomography and biomechanics. Am J Ophthalmol. 2019;197:7–16. doi: 10.1016/j.ajo.2018.08.054 [DOI] [PubMed] [Google Scholar]
- 9.Ali NQ, Patel DV, McGhee CN. Biomechanical responses of healthy and keratoconic corneas measured using a noncontact scheimpflug-based tonometer. Invest Ophthalmol Vis Sci. 2014;55(6):3651–3659. doi: 10.1167/iovs.13-13715 [DOI] [PubMed] [Google Scholar]
- 10.Lafond G, Bazin R, Lajoie C. Bilateral severe keratoconus after laser in situ keratomileusis in a patient with forme fruste keratoconus. J Cataract Refract Surg. 2001;27(7):1115–1118. doi: 10.1016/S0886-3350(00)00805-1 [DOI] [PubMed] [Google Scholar]
- 11.Galvis V, Tello A, Jaramillo JA, Gutierrez AJ, Rodriguez L, Quintero MP. Prevalence of keratoconus patients who consulted with a desire refractive surgery in ophthalmology center reference Bucaramanga, Colombia. Rev Soc Colomb Oftal. 2011;44:129–134. [Google Scholar]
- 12.Gilevska F, Kostovska B, Osmani I, et al. Prevalence of keratoconus in refractive surgery practice population in North Macedonia. Int Ophthalmol. 2022;42(10):3191–3198. doi: 10.1007/s10792-022-02319-0 [DOI] [PubMed] [Google Scholar]
- 13.Koc M, Aydemir E, Tekin K, Inanc M, Kosekahya P, Kiziltoprak H. Biomechanical analysis of subclinical keratoconus with normal topographic, topometric, and tomographic findings. J Refract Surg. 2019;35(4):247–252. doi: 10.3928/1081597X-20190226-01 [DOI] [PubMed] [Google Scholar]
- 14.Catalan-Lopez S, Cadarso-Suarez L, Lopez-Raton M, Cadarso-Suarez C. Corneal biomechanics in unilateral keratoconus and fellow eyes with a scheimpflug-based tonometer. Optom Vis Sci. 2018;95(7):608–615. doi: 10.1097/OPX.0000000000001241 [DOI] [PubMed] [Google Scholar]
- 15.Ahmadi Hosseini SM, Abolbashari F, Niyazmand H, Sedaghat MR. Efficacy of corneal tomography parameters and biomechanical characteristic in keratoconus detection. Cont Lens Anterior Eye. 2014;37(1):26–30. doi: 10.1016/j.clae.2013.07.007 [DOI] [PubMed] [Google Scholar]
- 16.Rabinowitz YS. Keratoconus. Surv Ophthalmol. 1998;42(4):297–319. doi: 10.1016/S0039-6257(97)00119-7 [DOI] [PubMed] [Google Scholar]
- 17.Belin MW, Duncan JK. Keratoconus: the ABCD grading system. Klin Monbl Augenheilkd. 2016;233(06):701–707. doi: 10.1055/s-0042-100626 [DOI] [PubMed] [Google Scholar]
- 18.Ambrosio R Jr, Randleman JB. Screening for ectasia risk: what are we screening for and how should we screen for it? J Refract Surg. 2013;29(4):230–232. doi: 10.3928/1081597X-20130318-01 [DOI] [PubMed] [Google Scholar]
- 19.Li X, Yang H, Rabinowitz YS. Keratoconus: classification scheme based on videokeratography and clinical signs. J Cataract Refract Surg. 2009;35(9):1597–1603. doi: 10.1016/j.jcrs.2009.03.050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bae GH, Kim JR, Kim CH, Lim DH, Chung ES, Chung T-Y. Corneal topographic and tomographic analysis of fellow eyes in unilateral keratoconus patients using Pentacam. Am J Ophthalmol. 2014;157(1):103–109 e101. doi: 10.1016/j.ajo.2013.08.014 [DOI] [PubMed] [Google Scholar]
- 21.Shajari M, Jaffary I, Herrmann K, et al. Early tomographic changes in the eyes of patients with keratoconus. J Refract Surg. 2018;34(4):254–259. doi: 10.3928/1081597X-20180124-01 [DOI] [PubMed] [Google Scholar]
- 22.Buhren J, Kook D, Yoon G, Kohnen T. Detection of subclinical keratoconus by using corneal anterior and posterior surface aberrations and thickness spatial profiles. Invest Ophthalmol Vis Sci. 2010;51(7):3424–3432. doi: 10.1167/iovs.09-4960 [DOI] [PubMed] [Google Scholar]
- 23.Ambrosio R Jr, Caiado AL, Guerra FP, et al. Novel pachymetric parameters based on corneal tomography for diagnosing keratoconus. J Refract Surg. 2011;27(10):753–758. doi: 10.3928/1081597X-20110721-01 [DOI] [PubMed] [Google Scholar]
- 24.Ambrosio R Jr, Lopes BT, Faria-Correia F, et al. Integration of scheimpflug-based corneal tomography and biomechanical assessments for enhancing ectasia detection. J Refract Surg. 2017;33(7):434–443. doi: 10.3928/1081597X-20170426-02 [DOI] [PubMed] [Google Scholar]
- 25.Asroui L, Dagher SA, Elsheikh A, et al. Biomechanical evaluation of topographically and tomographically normal fellow eyes of patients with keratoconus. J Refract Surg. 2022;38(5):318–325. doi: 10.3928/1081597X-20220225-01 [DOI] [PubMed] [Google Scholar]
- 26.Herber R, Hasanli A, Lenk J, et al. Evaluation of corneal biomechanical indices in distinguishing between normal, very asymmetric, and bilateral keratoconic eyes. J Refract Surg. 2022;38(6):364–372. doi: 10.3928/1081597X-20220601-01 [DOI] [PubMed] [Google Scholar]
- 27.Zhang X, Ding L, Sun L, et al. Prognostic nomograms predicting risk of keratoconus in very asymmetric ectasia: combined corneal tomographic and biomechanical assessments. Front Bioeng Biotechnol. 2022;10:839545. doi: 10.3389/fbioe.2022.839545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pena-Garcia P, Peris-Martinez C, Abbouda A, Ruiz-Moreno JM. Detection of subclinical keratoconus through non-contact tonometry and the use of discriminant biomechanical functions. J Biomech. 2016;49(3):353–363. doi: 10.1016/j.jbiomech.2015.12.031 [DOI] [PubMed] [Google Scholar]