Abstract
Background and study aims Endoscopic resection (ER) is recommended for the management of duodenal neuroendocrine tumors (D-NETs) confined to the submucosal layer, without lymph node or distant metastasis. While this is accepted practice for lesions < 10 mm, consensus for larger lesions remains unclear. Although endoscopic submucosal dissection (ESD) has been proposed as the preferred ER technique for DNETs ≥10 mm, there are limited data on efficacy and safety, particularly in the Western setting.
Patients and methods We performed a retrospective analysis of patients with D-NETs who underwent ESD between 2012 and 2022 in three tertiary referral centers in Australia, France, and Belgium.
Results Fourteen patients with 15 D-NETs were evaluated. Median patient age was 64 years (interquartile range [IQR] 58–70 years). All D-NETs were confined to the duodenal bulb. Median D-NET size was 10 mm (IQR 7–12 mm) and specimen size was 15 mm (IQR 15–20 mm). Median procedure time was 60 minutes (IQR 25–90 minutes). The rate of en bloc resection was 100%. Intra-procedural perforation occurred in four patients (26.7%), with all closed endoscopically without long-term sequelae. There were no episodes of clinically significant bleeding. No local recurrence, lymph node or distant metastasis was observed at a median follow-up of 19.9 months (IQR 10.3–49.3 months).
Conclusions In experienced hands, ESD for D-NETs can achieve a 100% en bloc resection rate. There were no cases of local recurrence or distant metastatic spread, indicating that ESD may be a viable option for patients with D-NETs 10 to 15 mm that are not surgical candidates.
Keywords: Neoplasia, Quality and logistical aspects, Performance and complications, GI Pathology
Introduction
Duodenal neuroendocrine tumors (D-NETs) are uncommon neoplastic entities, primarily arising within the duodenal bulb. Between 1983 and 2010, there was a 400% increase in the incidence of D-NETs 1 , likely owing to improved optical evaluation and advancements in endoscope technology 2 . The majority of D-NETs (80%) are non-ampullary 3 , with current guidelines advocating for resection when ≤ 20 mm in size, in the absence of lymph node involvement 4 5 . This is primarily due to their metastatic potential, with the ability to spread to regional lymph nodes and distant organs such as the liver.
Although surgical intervention offers a greater chance of resection with clear margins when compared with endoscopic resection (ER), it is highly invasive and not without risk 4 . The utility of ER remains unclear, with relevant literature limited to small volume case-series 6 7 8 9 10 11 12 13 14 15 16 17 18 19 that describe a myriad of methods, including cold-snare polypectomy, endoscopic mucosal resection (EMR), band-EMR and band-EMR without resection (BWR). However, because D-NETs are subepithelial lesions (SELs), there is an inherent risk of incomplete resection with these techniques. Furthermore, any failed attempts at ER may promote formation of submucosal fibrosis and therefore render subsequent attempts difficult.
Endoscopic submucosal dissection (ESD) is an attractive alternative because it offers the potential to achieve en bloc resection with clear margins. However, the current evidence base pertaining to the outcomes of ESD for non-ampullary D-NETs is limited to case series of less than 10 patients, predominantly from Asia 6 20 21 22 . We sought to evaluate the performance and safety of ESD in the management of non-ampullary D-NETs in a multi-centered Western setting.
Methods
Study population
We reviewed prospectively maintained ESD databases from three tertiary referral centers in Australia, France, and Belgium between January 2012 and June 2022. A total of 14 patients with 15 D-NETs underwent ESD at one of the three expert endoscopy centers. In each case, a baseline gastroscopy, endoscopic ultrasound (EUS), and axial imaging in the form of computed tomography (CT) and/or somatostatin receptor imaging were performed prior to ESD to ensure that there was no evidence of regional lymph node enlargement or distant metastasis. The primary goal of ESD was to obtain an enbloc excision, with clear horizontal and vertical margins. Thus, the indications for ESD a were lesion size 10 to 15 mm, difficult access or suspected submucosal fibrosis as demonstrated by a poor lift with suctioning. Approval to maintain prospective databases was provided by the research and ethics committees of each respective hospital. Informed consent was gained from each patient prior to ESD.
Endoscopic resection
All endoscopic procedures were performed by a study investigator (accredited gastroenterologist with interventional endoscopy training and an established tertiary referral practice). All ESD procedures were performed with general anesthesia with endotracheal intubation. A single-channel upper gastrointestinal endoscope with a water-jet system was used. Carbon dioxide insufflation was used during all ESD cases. A Dual Knife or Dual Knife J 1.5 mm (Olympus, Tokyo, Japan) was used to mark out the lesion using soft coagulation, with a 3- to 5-mm rim of normal duodenal mucosa to reduce the risk of positive horizontal margins. A submucosal injection of a colloid chromoinjectate was performed to lift the lesion. At the discretion of the proceduralist, adrenaline (1:100,000) was added to the chromoinjectate in some cases.
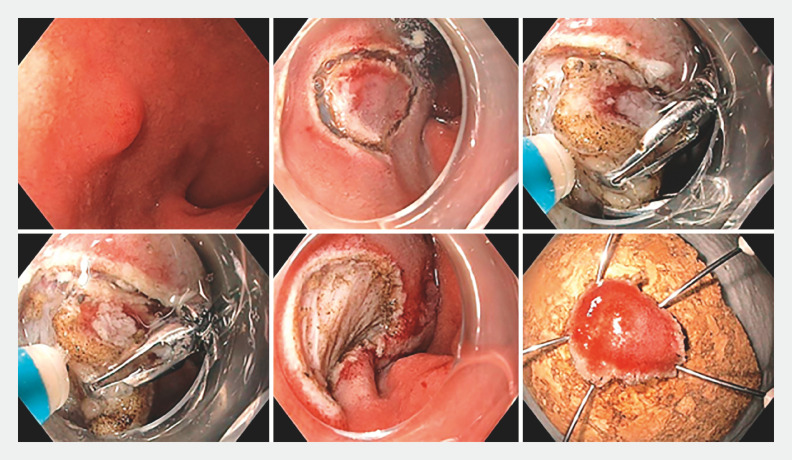
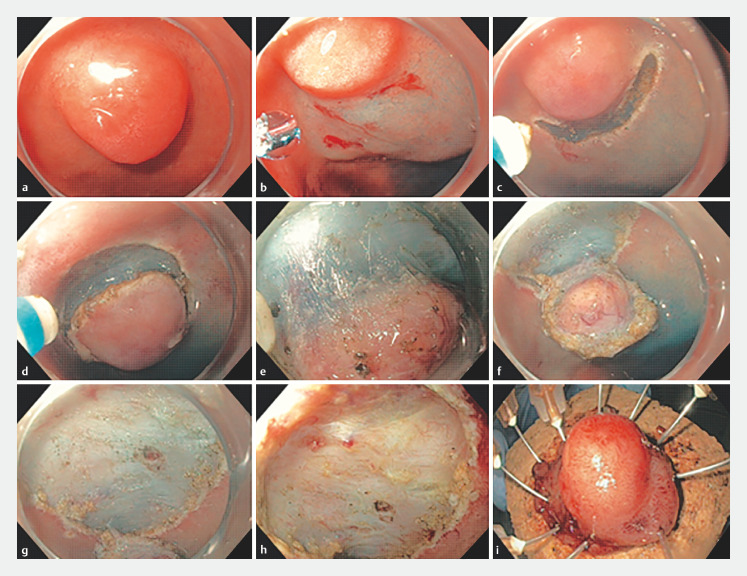
Subsequently, a mucosal incision was performed to expose the submucosal plane, ensuring the soft coagulation markings were kept within the field of resection. In the majority of cases, the mucosal incision was extended circumferentially, following which submucosal dissection was performed as close to the muscle layer as possible. In one case, due to the challenging location of the D-NET, a submucosal tunnel was created from the pylorus into the duodenal bulb. Once that was complete, a mucosal incision was created on either side of the tunnel to remove the lesion en bloc. Traction was employed if submucosal fibrosis was encountered ( Fig. 1 ). A microprocessor-controlled generator (ERBE VIO 300D or VIO3; ERBE, Tubingen, Germany) was used during mucosal incision and submucosal dissection ( Fig. 2 ). Large vessels were treated prophylactically with hemostatic forceps using soft coagulation. Minor intra-procedural bleeding was considered an accepted part of routine ESD and was commonly encountered. This was readily treated with the Dual Knife on a Swift coagulation setting or hemostatic forceps on a soft coagulation setting.
Fig. 1.
A 10-mm duodenal bulb neuroendocrine tumor removed by endoscopic submucosal dissection, employing endoclips for traction to expose the submucosal plane.
Fig. 2.
Endoscopic submucosal dissection technique. a Duodenal neuroendocrine tumor observed in duodenal bulb. b Submucosal injection. c Mucosal incision. d Circumferential incision completed. e Submucosal dissection. f Submucosal tumor completed dissected. g Completion of dissection. h Post-ESD defect. i En bloc pinned specimen.
Histological evaluation
Once retrieved, the lesion was fixed in 10% formalin. Following serial sectioning, the resected specimens were assessed independently by two subspecialist gastrointestinal pathologists. The histological type, macroscopic appearance, tumor size, depth of invasion, lymphatic and vascular involvement, and horizontal and vertical margins were assessed. The Ki-67 index was assessed in all cases to classify tumors as grade 1, 2 or 3 according to the World Health Organization (WHO) 2019 classification 23 .
Definitions
En bloc resection was defined as resection of the lesion in a single piece with no endoscopically visible residual tumor. R0 resection was defined as en bloc resection with histologically clear horizontal and vertical margins. Presence of any tumor cells at the resected margin was considered a positive margin. As per the 2023 European Neuroendocrine Tumor Society (ENETS) guideline, curative resection was defined as an R0 resection, lesion size ≤15 mm, WHO Grade 1 histology and without lymphovascular invasion 4 .
Peri-procedural adverse events, including bleeding, perforation, and stenosis were recorded. Clinically significant intra-procedural bleeding was defined as the requirement of hemostatic techniques other than the use of hemostatic forceps or the Dual Knife J on Swift coagulation. Delayed bleeding was defined as hematemesis or melena that required an additional endoscopic procedure and necessitated the use of hemostatic forceps or endo-clips within the first 2 weeks post-ESD.
Perforation was subdivided into intra-procedural and delayed. Delayed perforation was diagnosed when a patient presented with fevers and peritoneal or retroperitoneal free air on CT imaging, in the absence of intraoperative perforation. Stenosis was defined as inability to pass a standard gastroscope beyond the site of ESD at time of endoscopic follow-up or if a patient presented with clinical signs of obstruction.
Follow-up
Post-ESD, patients were kept fasted and observed in the hospital overnight on intravenous (IV) fluids and a continuous IV proton pump inhibitor(PPI) infusion. The following morning, patients were started on clear fluids. Patients were discharged if stable, afebrile, pain-free, and tolerating fluids. All patients were prescribed twice-daily oral PPI therapy for 2 months. At follow-up gastroscopy 6-months post-ESD, if there was endoscopic concern for recurrence, biopsies were taken to assess for histologic evidence of recurrence.
Statistical analysis
SPSS version 26.0 (IBM, Armonk, New York, United States) was used for data analysis. Variables were analyzed per lesion. Continuous variables were summarized using median and interquartile range (IQR). Categorical variables were summarized as frequencies (%). All analyses were exploratory and two-tailed tests with a 5% significance level were used throughout. To test for association between categorical variables, the Pearson x 2 or the Fisher Exact tests were used, where appropriate.
Results
Patient and lesion characteristics
A total of 15 D-NETs in 14 patients underwent ESD ( Table 1 ). The median age of the patients was 64 years (IQR 58–70 years) and the majority were male (n=9, 64.3%). All D-NETs were located within the duodenal bulb. There were two patients (14.3%) with concomitant D-NETs identified at index gastroscopy. One had a 2- to 3-mm lesion that was easily accessible and resected via EMR later. The other had their concomitant D-NET removed via ESD at a later date, and is included within this dataset.
Table 1 Patient and lesion characteristics.
| Characteristics | Value |
| IQR, interquartile range; EUS, endoscopic ultrasound; NET, neuroendocrine tumor. | |
| Age in years, median (IQR) | 64 (58–70) |
| Sex n (%) | |
|
9 (60%) |
|
6 (40%) |
| NET location n (%) | |
|
15 (100%) |
| Depth based on EUS examination n (%) | |
|
2 (13.3%) |
|
13 (86.7%) |
| Tumor morphology, n (%) | |
|
9 (60%) |
|
6 (40%) |
| Ulceration of NET n (%) | 1 (6.7%) |
| Tumor size on endoscopy, median mm (IQR) | 10 (7–12) |
Endoscopic morphology was either Paris Is (n=9, 60%) or Paris IIa (n=6, 40%). One lesion was ulcerated (6.7%). Most of the D-NETs appeared to originate from Layer 3 (submucosa) based on EUS examination (n=13, 86.7%) and demonstrated tumor avidity on 68-Ga-DOTATATE positron emission tomography (PET) scan imaging (n=10, 66.7%). The median size of the D-NETs at index gastroscopy was 10 mm (IQR 7–12 mm).
Procedure and histologic outcomes
The majority of the patients underwent ESD at the lead site (n=9, 66.7%; Table 2 ). The median specimen size was 15 mm (IQR 15–20 mm) and time taken to complete ESD was 60 minutes (IQR 25–90 minutes). Histological analysis classified 5 (33.3%) D-NETs as Grade 1 and 10 (66.7%) as Grade 2.
Table 2 Procedural and short-term outcomes.
| Characteristic/outcome | Value |
| IQR, interquartile range; WHO, World Health Organization. | |
| ESD duration min (IQR) | 60 (25–90) |
| Specimen size, median mm (IQR) | 15 (15–20) |
| Technique n (%) | |
|
9 (60%) |
|
1 (6.7%) |
|
5 (33.3%) |
| WHO grade, n (%) | |
|
5 (33.3%) |
|
10 (66.7%) |
|
0 (0%) |
| Lymphovascular invasion n (%) | 1 (6.7%) |
| Complications n (%) | |
|
0 (0%) |
|
0 (0%) |
|
4 (26.7%) |
|
0 (0%) |
|
0 (0%) |
| En bloc r esection completed n (%) | 15 (100%) |
| R0 margins n (%) | |
|
15 (100%) |
|
7 (46.7%) |
| Curative resection n (%) | 3 (20%) |
| Follow-up time median, months (IQR) | 19.9 (10.3–49.3) |
| Local recurrence n (%) | 0 (0%) |
| Lymph metastasis n (%) | 0 (0%) |
| Distant metastasis n (%) | 0 (0%) |
En bloc resection was achieved in all 15 lesions (100%) ( Table 3 ). A conventional approach was utilized in nine (60%). One (6.7%) required the creation of a submucosal tunnel from the pylorus into the duodenal bulb. The remainder required traction (n=5, 33.3%) due to the presence of submucosal fibrosis.
Table 3 Characteristics of 15 ESDs performed in 14 patients with D-NETs located in the duodenal bulb.
| Case | Age (yr) | Sex | Size (mm) | Depth | Duration (min) | Method | Morphology (Paris) | Pathologic grade | LVI | R0 resection | Curative resection | Intra-procedural perforation |
| *Numbers 8 and 9 were the same patient. †Specimen contained two separate NETs sized 15 and 16 mm. ESD, endoscopic submucosal dissection; D-NET, duodenal neuroendocrine tumor; LVI, lymphovascular invasion; M, male; F, female; B, duodenal bulb; SM, submucosa; MM, muscular mucosa; S, standard ESD technique; Tr, traction technique; Tu, tunnelling technique. | ||||||||||||
| 1 | 61 | M | 16 † | SM | 240 | Tu | 0-Is | 1 | No | No | No (R1, size) | Yes |
| 2 | 77 | M | 15 | MM | 100 | S | 0-Is | 1 | No | Yes | Yes | No |
| 3 | 68 | M | 4 | SM | 28 | Tr | 0-IIa | 2 | No | Yes | No (Grade 2) | No |
| 4 | 58 | F | 10 | SM | 18 | Tr | 0-Is | 2 | Yes | Yes | No (Grade 2, LVI) | No |
| 5 | 58 | F | 5 | MM | 73 | Tr | 0-Is | 1 | No | No | No (R1) | No |
| 6 | 65 | F | 10 | SM | 25 | S | 0-IIa | 2 | No | No | No (Grade 2, R1) | No |
| 7 | 82 | M | 15 | SM | 60 | S | 0-Is | 2 | No | No | No (Grade 2, R1) | No |
| 8 * | 55 | M | 12 | SM | 60 | S | 0-Is | 2 | No | Yes | No (Grade 2) | No |
| 9 | 55 | M | 8 | SM | 60 | S | 0-Is | 2 | No | No | No (Grade 2, R1) | No |
| 10 | 70 | F | 7 | SM | 90 | Tr | 0-Is | 2 | No | Yes | No (Grade 2) | Yes |
| 11 | 77 | F | 12 | SM | 20 | S | 0-Is | 2 | No | No | No (Grade 2, R1) | No |
| 12 | 62 | M | 8 | SM | 91 | Tr | 0-IIa | 2 | No | No | No (Grade 2, R1) | Yes |
| 13 | 64 | F | 10 | SM | 60 | S | 0-IIa | 2 | No | No | No (Grade 2, R1) | No |
| 14 | 52 | F | 7 | SM | 60 | S | 0-IIa | 1 | No | Yes | Yes | Yes |
| 15 | 65 | F | 10 | SM | 22 | S | 0-IIa | 1 | No | Yes | Yes | No |
Horizontal R0 margins were achieved in all 15 (100%) and vertical R0 margins in seven patients (46.7%). Thus, R0 resection was achieved in seven patients total (46.7%). Of the seven cases with positive vertical margins, the length of the involved interface was a median of 0.90 mm (IQR 0.18–3.48 mm). Lymphovascular invasion was present in one Grade 2 D-NET (6.7%), which had clear margins. By ENETS 2023 criteria, a curative resection was achieved in three cases (20%). There were no predictors of R0 or curative resection.
Adverse events
There were no episodes of clinically significant intra-procedural bleeding, delayed bleeding, delayed perforation, or stenosis.
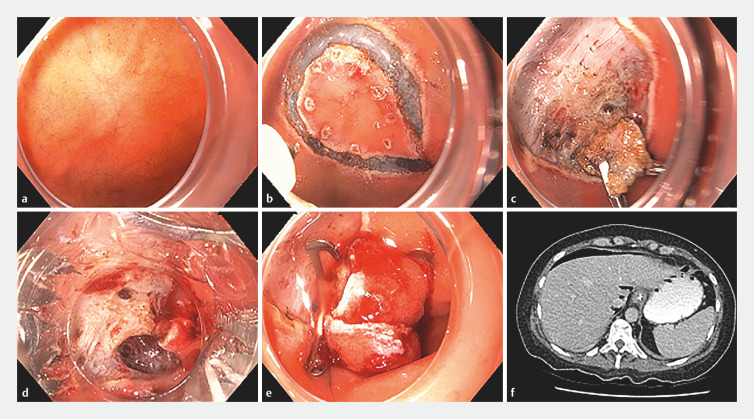
Intra-procedural perforation occurred in four cases (26.7%). All were managed conservatively at the time of the index-ESD, with through-the-scope endoscopic clip closure performed successfully in three cases. All four patients were admitted to the hospital for close observation. In addition to standard care with IV fluids and PPI therapy, they were started on IV antibiotics. Three of the four patients were discharged the next day. The fourth case ( Fig. 3 ) involved significant submucosal fibrosis due to a previous attempt at resection with EMR. During traction-assisted ESD, two small areas of perforation were identified and promptly closed with an over-the-scope clip (OTSC). Cross-sectional imaging confirmed the presence of capno-peritoneum. The patient was managed conservatively in consultation with the local upper gastrointestinal surgical team. Patient-controlled analgesia for pain and IV antibiotics were administered and patient was discharged on Day 7 post-ESD.
Fig. 3.
a Scar from prior D-NET removed by EMR with positive margins. b Circumferential incision completed. c Submucosal dissection with traction technique. d Two areas of deep mural injury (black arrows). e Over-the-scope endoscopic clip closure. f Evidence of pneumoperitoneum on CT post procedure.
ESD was significantly longer in patients experiencing perforation (120.3 ± 81.1 minutes vs. 47.8 ± 26.8 minutes; P =0.017). By univariate analysis, the only risk factor for perforation was non-avidity on PET imaging D-NETs (n=3, 75% vs. n=1, 10%; P=0.041). However, when adjusted for patient age, gender, layer of origin, and presence of ulceration, there was no significance ( P =0.999).
None of the patients with perforation required salvage surgical intervention and there were no long-term sequelae.
Short-term outcomes
Patients were followed up for a median of 19.9 months (IQR 10.3–49.3 months) after ESD ( Table 2 ). None of the patients showed evidence of local endoscopic recurrence, metachronous lymph node metastasis or distant metastasis on PET scan during the follow-up period.
Discussion
In this study, we assessed the procedural and short-term treatment outcomes of ESD for non-ampullary D-NETs. To the best of our knowledge, this is the largest case-series to date and the first in a Western setting. We showed that ESD is technically feasible in expert centers, achieving a 100% en bloc resection rate for D-NETs 10 to 15 mm. The rate of positive vertical margins was high (53.3%), although it did not translate into short-term local recurrence or lymph node/distant metastases at a median follow-up of 19.9 months. Reassuringly, all intra-procedural perforations were detected promptly within the index-ESD procedure and managed with clip closure, without any sequelae. While longer-term follow-up is required to determine the efficacy of ESD for D-NETs 10 to 15 mm, our findings suggest that this is a viable option to consider, particularly in those that are not candidates for surgical resection.
Although D-NETs are uncommon, they can lead to serious consequences, and thus, mandate intervention. While current guidelines recommend ER for non-ampullary D-NETs <10 mm 2 24 , for larger lesions it remains unclear whether a surgical or ER approach is superior 24 . Traditional ER methods, such as cold-snare polypectomy, EMR, band-EMR and BWR, are limited by high rates of positive margins 6 11 14 15 16 17 18 19 20 21 25 . One advantage of ESD over traditional ER techniques is that we were able to achieve 100% R0 horizontal margins. However, we noted that eight (53.3%) had a positive vertical margin. Because the submucosal layer of the duodenal cap is thin, subepithelial lesions such as D-NETs are in close proximity to the muscularis propria ( Fig. 1 ). Thus, to achieve a histologically free vertical margin is extremely challenging. Interestingly, the median length of the involved interface at this margin was only 0.9 mm. Furthermore, some D-NETs may be encapsulated 26 27 28 29 , and therefore, with resection it is possible that the margin is void of tumor cells. Reassuringly, disease-free survival in our cohort was 100%, with none of the patients in our series demonstrating local endoscopic recurrence or lymph node/distant metastasis during a median follow-up period of 19.9 months. Comparatively, the rates of local recurrence with EMR are reported upwards of 18% 6 9 10 11 12 13 30 . Although longer follow-up of data is required, our results demonstrate that the ESD of D-NETs 10 to 15 mm in size is technically feasible.
The low rate of curative resection (20%) achieved in our study was predominantly independent of the resection technique and rather driven by tumor biology, with 10 of the 12 non-curative cases (83.3%) due to WHO Grade 2 histology. Following discussion at local multi-disciplinary team meetings, surveillance was chosen in all non-curative cases due to a combination of factors, including patient age, underlying comorbidities, and personal preference. As aforementioned, there were no cases of endoscopic recurrence noted at follow-up, in keeping with studies in an Eastern setting 6 20 21 25 30 . As an alternative to ESD, endoscopic full-thickness resection (EFTR) may be an option for D-NETs because it may address the issue of positive vertical margins. However, in comparison to ESD, precise control over depth and lateral margins of excision are not assured. Furthermore, the technique is limited by expense, lesion size, maneuverability of the device within the duodenum, and the risk of delayed perforation 19 . Alternatively, the risk of invasive surgery may well exceed the potential for recurrence after ESD for 10- to 15-mm D-NETs. Therefore, further studies are required, particularly comparing short- and long-term outcomes of ESD with EFTR.
ESD for non-ampullary D-NETs is more technically difficult than for stomach, esophageal or rectal lesions owing to the fact that the duodenal wall is very thin and the lumen is narrower, therefore affecting maneuverability 31 32 . This may account for the higher rates of intra-procedural perforation experienced with ESD for D-NETs, ranging from 13% to 67% 20 21 22 . In the present study, our intra-procedural perforation rate was similar at 26.7%. Reassuringly, all perforations were identified and managed promptly. One case required an OTSC closure; however, this was a technically challenging case with significant submucosal fibrosis secondary to a prior EMR attempt. Importantly, there was no need for salvage surgery or long-term sequelae. Although non-avidity on PET imaging was identified as a risk factor for perforation, this is likely a Type 1 statistical error due to the small number of cases. Furthermore, there was no significance noted when adjusted for age, gender, layer of origin, and presence of ulceration. Thus, we have demonstrated that when ESD is performed by experienced endoscopists in tertiary referral centers, complications such as perforation can be readily managed, without long-term impact on the patient.
The present study had several limitations. First, it was a retrospective study that assessed the outcomes of ESD for D-NETs. Thus, we did not have access to data pertaining to the total number of D-NETs that were detected or resected by other means during the study period. Second, despite being multicenter, the overall number of patients was small. However, to the best of our knowledge, this is the largest number of patients undergoing ESD for D-NETs reported to date. Third, the follow-up period and method of follow-up were variable. The duration was somewhat insufficient for detecting metastasis in all cases. A larger number of patients enrolled prospectively involving multiple centers of excellence with standardized follow-up for a minimum of 5 years is required to best determine the efficacy and safety of ESD for D-NETs.
Conclusions
In conclusion, if there is no evidence of lymph node or distant metastasis during workup for a D-NET, ESD is an effective and safe ER technique when performed in an expert center. ESD should be the preferred modality for 10- to 15-mm D-NETs. Although tumor biology negatively impacted curative resection rates, current alternatives include highly comorbid surgeries. While the lack of short-term recurrence is encouraging, the impact of positive vertical margins with ESD is yet to be determined. Further studies are required, including with longer-term follow-up and comparisons with EFTR.
Footnotes
Conflict of Interest Michael J. Bourke: Research Support: Olympus, Cook Medical, Boston Scientific. The remaining authors have no conflicts of interest to disclose.
References
- 1.Fitzgerald TL, Dennis SO, Kachare SD et al. Increasing incidence of duodenal neuroendocrine tumors: Incidental discovery of indolent disease? Surgery. 2015;158:466–471. doi: 10.1016/j.surg.2015.03.042. [DOI] [PubMed] [Google Scholar]
- 2.Delle Fave G, Kwekkeboom DJ, Van Cutsem E et al. ENETS Consensus Guidelines for the management of patients with gastroduodenal neoplasms. Neuroendocrinology. 2012;95:74–87. doi: 10.1159/000335595. [DOI] [PubMed] [Google Scholar]
- 3.Randle RW, Ahmed S, Newman NA et al. Clinical outcomes for neuroendocrine tumors of the duodenum and ampulla of Vater: a population-based study. J Gastrointest Surgery. 2014;18:354–362. doi: 10.1007/s11605-013-2365-4. [DOI] [PubMed] [Google Scholar]
- 4.Panzuto F, Ramage J, Pritchard DM et al. European Neuroendocrine Tumor Society (ENETS) 2023 Guidance Paper for Gastroduodenal NET G1-G3. J Neuroendocrinology. 2023;35:e13306. doi: 10.1111/jne.13306. [DOI] [PubMed] [Google Scholar]
- 5.Kulke MH, Shah MH, Benson AB et al. Neuroendocrine tumors, version 1.2015. Journal of the National Comprehensive Cancer Network. 2015;13:78–108. doi: 10.6004/jnccn.2015.0011. [DOI] [PubMed] [Google Scholar]
- 6.Kim GH, Kim JI, Jeon SW et al. Endoscopic resection for duodenal carcinoid tumors: a multicenter, retrospective study. J Gastroenterol Hepatol. 2014;29:318–324. doi: 10.1111/jgh.12390. [DOI] [PubMed] [Google Scholar]
- 7.Dasari BVM, Al-Shakhshir S, Pawlik TM et al. Outcomes of surgical and endoscopic resection of duodenal neuroendocrine tumours (NETs): a systematic review of the literature. J Gastrointest Surg. 2018;22:1652–1658. doi: 10.1007/s11605-018-3825-7. [DOI] [PubMed] [Google Scholar]
- 8.Massironi S, Campana D, Partelli S et al. Heterogeneity of duodenal neuroendocrine tumors: an italian multi-center experience. Ann Surg Oncol. 2018;25:3200–3206. doi: 10.1245/s10434-018-6673-5. [DOI] [PubMed] [Google Scholar]
- 9.Mahmud N, Tomizawa Y, Stashek K et al. Endoscopic resection of duodenal carcinoid tumors: a single-center comparison between simple polypectomy and endoscopic mucosal resection. Pancreas. 2019;48:60–65. doi: 10.1097/MPA.0000000000001197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gincul R, Ponchon T, Napoleon B et al. Endoscopic treatment of sporadic small duodenal and ampullary neuroendocrine tumors. Endoscopy. 2016;48:979–986. doi: 10.1055/s-0042-112570. [DOI] [PubMed] [Google Scholar]
- 11.Oono Y, Shinmura K, Hori K et al. Endoscopic submucosal resection using a ligation device without injection for duodenal neuroendocrine tumors. Surg Endosc. 2019;33:2008–2014. doi: 10.1007/s00464-018-06642-5. [DOI] [PubMed] [Google Scholar]
- 12.Min B-H, Kim ER, Lee JH et al. Management strategy for small duodenal carcinoid tumors: does conservative management with close follow-up represent an alternative to endoscopic treatment? Digestion. 2013;87:247–253. doi: 10.1159/000349958. [DOI] [PubMed] [Google Scholar]
- 13.Park SB, Kang DH, Choi CW et al. Clinical outcomes of ligation-assisted endoscopic resection for duodenal neuroendocrine tumors. Medicine. 2018;97:e0533. doi: 10.1097/MD.0000000000010533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fujimoto A, Sasaki M, Goto O et al. Treatment results of endoscopic mucosal resection with a ligation device for duodenal neuroendocrine tumors. Intern Med. 2019;58:773–777. doi: 10.2169/internalmedicine.1517-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lee SW, Sung JK, Cho YS et al. Comparisons of therapeutic outcomes in patients with nonampullary duodenal neuroendocrine tumors (NADNETs): a multicenter retrospective study. Medicine. 2019;98:e16154. doi: 10.1097/MD.0000000000016154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tashima T, Ryozawa S, Tanisaka Y et al. Endoscopic resection using an over-the-scope clip for duodenal neuroendocrine tumors. Endosc Int Open. 2021;9:E659–E666. doi: 10.1055/a-1374-6141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Khara HS, Shovlin GJ, Johal AS et al. Endoscopic banding without resection (BWR) technique for treatment of diminutive neuroendocrine tumors in the duodenum. Endosc Int Open. 2019;7:E302–E307. doi: 10.1055/a-0684-9563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Karagiannis S, Eshagzaiy K, Duecker C et al. Endoscopic resection with the cap technique of a carcinoid tumor in the duodenal bulb. Endoscopy. 2009;41:E288–E289. doi: 10.1055/s-0029-1215123. [DOI] [PubMed] [Google Scholar]
- 19.Bauder M, Schmidt A, Caca K. Endoscopic full-thickness resection of duodenal lesions-a retrospective analysis of 20 FTRD cases. United European Gastroenterol J. 2018;6:1015–1021. doi: 10.1177/2050640618773517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Suzuki S, Ishii N, Uemura M et al. Endoscopic submucosal dissection (ESD) for gastrointestinal carcinoid tumors. Surg Endosc. 2012;26:759–763. doi: 10.1007/s00464-011-1948-y. [DOI] [PubMed] [Google Scholar]
- 21.Nishio M, Hirasawa K, Ozeki Y et al. Short- and long-term outcomes of endoscopic submucosal dissection for non-ampullary duodenal neuroendocrine tumors. Ann Gastroenterol. 2020;33:265–271. doi: 10.20524/aog.2020.0477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Matsumoto S, Miyatani H, Yoshida Y. Endoscopic submucosal dissection for duodenal tumors: a single-center experience. Endoscopy. 2013;45:136–137. doi: 10.1055/s-0032-1310123. [DOI] [PubMed] [Google Scholar]
- 23.Nagtegaal ID, Odze RD, Klimstra D et al. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76:182. doi: 10.1111/his.13975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Delle Fave G, O'Toole D, Sundin A et al. ENETS Consensus Guidelines Update for Gastroduodenal Neuroendocrine Neoplasms. Neuroendocrinology. 2016;103:119–124. doi: 10.1159/000443168. [DOI] [PubMed] [Google Scholar]
- 25.Li Q-L, Zhang Y-Q, Chen W-F et al. Endoscopic submucosal dissection for foregut neuroendocrine tumors: an initial study. World J Gastroenterol. 2012;18:5799. doi: 10.3748/wjg.v18.i40.5799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Orttz H, Ortega JS, Bernat R et al. Carcinoids of the first part of the duodenum. J Br Surgery. 1976;63:235–236. doi: 10.1002/bjs.1800630318. [DOI] [PubMed] [Google Scholar]
- 27.Bourke M. Endoscopic resection in the duodenum: current limitations and future directions. Endoscopy. 2013;45:127–132. doi: 10.1055/s-0032-1326177. [DOI] [PubMed] [Google Scholar]
- 28.Kaliounji A, Alkoutami S, Farraj M et al. A rare presentation of a duodenal neuroendocrine tumor. Cureus. 2023;15 doi: 10.7759/cureus.33747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Neumann H, Ramesh J, Wilcox C et al. Resection of carcinoids in the duodenal bulb using the band ligation technique with the Duette mucosectomy device. Endoscopy. 2013;45:E365–E366. doi: 10.1055/s-0033-1344770. [DOI] [PubMed] [Google Scholar]
- 30.Kim SH, Park CH, Ki HS et al. Endoscopic treatment of duodenal neuroendocrine tumors. Clin Endosc. 2013;46:656–661. doi: 10.5946/ce.2013.46.6.656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hoteya S, Furuhata T, Takahito T et al. Endoscopic submucosal dissection and endoscopic mucosal resection for non-ampullary superficial duodenal tumor. Digestion. 2017;95:36–42. doi: 10.1159/000452363. [DOI] [PubMed] [Google Scholar]
- 32.Pérez-Cuadrado-Robles E, Quénéhervé L, Margos W et al. ESD versus EMR in non-ampullary superficial duodenal tumors: a systematic review and meta-analysis. Endosc Int Open. 2018;6:E998–E1007. doi: 10.1055/a-0579-9050. [DOI] [PMC free article] [PubMed] [Google Scholar]