Abstract
The genome of Yersinia pestis, the causative agent of plague, contains at least 30 copies of an element, designated IS1541, which is structurally related to IS200 (85% identity). One such element is inserted within the chromosomal inv gene (M. Simonet, B. Riot, N. Fortineau, and P. Berche, Infect. Immun. 64:375–379, 1996). We characterized other IS1541 insertions by cloning 14 different Y. pestis 6/69M loci carrying a single copy of this insertion sequence (IS) into Escherichia coli and, for each element, sequencing 250 bp of both flanking regions. In no case was this IS element inserted into large open reading frames; however, in eight cases, it was detected downstream (17 to 139 bp) of genes thought to be transcribed monocistronically or which constituted the last gene of an operon, and in only one case was it detected upstream (37 bp) of the first gene of an operon. Sequence analysis revealed stem-loop structures (ΔG, <−10 kcal) resembling rho-independent transcription terminators in 8 of the 14 insertion sites. These motifs might constitute hot spots for insertion of this IS1541 element within the Y. pestis genome.
Transposition of insertion sequences (IS) is known to cause a number of effects, including insertions, deletions, and cointegrate formation, which can result in silent mutations, gene inactivation, or the modulation of expression of downstream genes (8). Several classes of IS elements are found in Yersinia pestis, the causative agent of bubonic and pneumonic plague. They include IS100, IS285, and IS1541 (previously designated IS200-like) elements (7, 24, 30), which are integrated into the chromosome and/or within the three virulence plasmids found in this bacterium, pYV, pFra, and pPst (for a review, see references 3 and 23). Guiyoule et al. (12) demonstrated by pulsed-field gel electrophoresis analysis that Y. pestis is subject to numerous and spontaneous DNA rearrangements. These events might be due to the presence of multiple IS in the bacterial genome allowing the fusion of replicons, such as the integration of virulence plasmids pYV and pFra into the chromosome (25, 32), or the deletion of fragments, such as the 102-kb chromosomal pigmentation (pgm) locus. This locus is a pathogenicity island involved in iron acquisition and can be lost spontaneously, at high frequency, by Y. pestis (6).
IS1541 (approximately 708 bp) was recently discovered within the chromosomal inv gene of Y. pestis, and gene disruption results in the inability of this bacterium to invade cultured mammalian cells (30). It displays 85% nucleotide identity with IS200 (approximately 709 bp) found in Salmonella spp., Shigella spp., and Escherichia coli, and both elements encode homologous (93% identity) 152-amino-acid putative transposases (1, 11, 14). Unlike most IS elements, IS200 and IS1541 do not contain inverted terminal repeats (15, 21, 30). As a consequence of this unusual feature, the ends of these elements have not been precisely characterized up to now. The IS1541 element has been found in multiple copies in the genome of Y. pestis (30), and in this report, we present additional sequence data for 14 insertion sites of the element other than in the inv gene in Y. pestis 6/69M.
Distribution of IS1541 in the Y. pestis 6/69M genome.
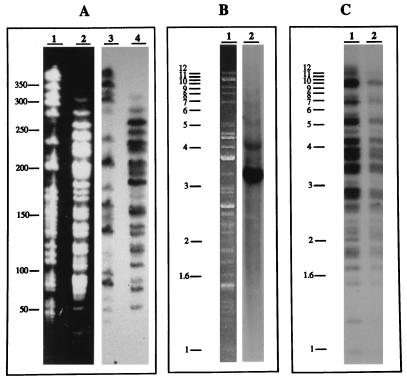
We have previously shown that the genome of Y. pestis 6/69M, which belongs to biotype Orientalis and ribotype B (12), contains multiple copies of the IS1541 element (30). By hybridization with an IS1541 probe of bacterial DNA digested by NotI or SpeI, restriction endonucleases that infrequently cut Y. pestis DNA (12, 17) and do not cut the IS element, we found that 18 of 27 NotI fragments and 17 of 29 SpeI fragments hybridized with this probe (Fig. 1A). This result demonstrates that the IS1541 elements are not clustered in the genome.
FIG. 1.
Distribution of IS1541 elements in Y. pestis 6/69M. Bacterial DNA was digested by appropriate restriction endonucleases according to the manufacturer’s recommendations (New England Biolabs), and DNA fragments were separated by electrophoresis. Pulsed-field gel electrophoresis of macrorestricted genomic DNA was carried out as described by Guiyoule et al. (12). After separation, DNA fragments were depurinated, denaturated, and then transferred to nylon membranes (Boehringer Gmbh, Mannheim, Germany) by the Southern technique (28). The IS probe preparation, [32P]dCTP (Amersham France, Courtaboeuf, France) labeling, and DNA-DNA hybridization were performed as previously described (20). (A) Lanes 1 and 2, pulsed-field gel electrophoresis of DNA digested by NotI (lane 1) or SpeI (lane 2); lanes 3 and 4, Southern blot hybridization of DNA digested by NotI (lane 3) or SpeI (lane 4) with the radiolabeled IS probe. (B) Lane 1, agarose gel electrophoresis of a total plasmid extract (pYV, pPst, and pFra) prepared by an alkaline lysis procedure and digested by the restriction endonuclease EcoRV; lane 2, Southern blot hybridization of EcoRV-digested DNA with the radiolabeled IS probe. (C) Southern blot hybridization of HincII-digested DNA from Pgm+ (lane 1) and Pgm− (lane 2) cells with the radiolabeled IS probe. Molecular size markers (in kilobases) are indicated on the left. A strong hybridization signal given by some bands may correspond to the presence of multiple IS.
As in almost all strains of Y. pestis (5), strain 6/69M contains one plasmid each of 110 (pFra), 70 (pYV), and 9.5 (pPst) kb. Plasmid DNAs were extracted from this strain, digested with EcoRV (which does cleave within IS1541), separated by electrophoresis, transferred onto a nylon membrane, and hybridized with a 32P-labeled IS1541-specific DNA probe (28). A single hybridizing band of about 3.2 kb was detected, and on the basis of the known restriction maps of individual plasmids, this fragment was assigned to pFra (Fig. 1B). Thus, almost all IS1541 copies were located on the bacterial chromosome.
Finally, we looked for the presence of the IS within the pgm locus. This was indirectly achieved by comparing the IS1541 hybridization profiles of DNA from the parental strain Pgm+ and its derivative strain Pgm−, digested by HincII, an enzyme which does not cleave within IS1541. As depicted in the autoradiogram presented in Fig. 1C, there was no detectable difference in the IS patterns exhibited by the two strains. This result suggested that there is no IS1541 element within the pgm locus. Moreover, E. coli recombinant clones carrying DNA fragments spanning the pgm locus did not hybridize with an IS1541-specific DNA probe (4).
Cloning of IS1541 flanking regions of the Y. pestis 6/69M genome.
The autoradiogram presented in Fig. 1C revealed the presence of numerous hybridizing bands, mostly ranging from 2 to 6 kb in size. To identify regions of the Y. pestis genome in which the IS element was inserted, HincII-restricted DNA from strain 6/69M was separated by electrophoresis through a 1% agarose gel in Tris-acetate buffer, and 2- to 6-kb DNA fragments were extracted from the gel by using β-agarase (New England Biolabs, Beverly, Mass.) and ligated with T4 DNA ligase into a SmaI-digested pUC19 vector (Appligene, Illkirch, France) (28). Recombinant plasmids were introduced into E. coli JM105 by transformation (28), and transformants were selected on Luria-Bertani agar containing ampicillin (100 μg ml−1) and X-Gal (5-bromo-4-chloro-3-indolyl-β-d-galactopyranoside). White transformants resistant to ampicillin were screened for the presence of the IS1541-related sequences by colony blot hybridization (28) with the specific probe. Endonuclease restriction (EcoRI, HindIII, and SphI) followed by Southern blot hybridization with the IS1541 probe of plasmids from recombinant clones allowed discrimination of 14 different inserts having a size less than 4.5 kb and containing a single copy of the IS element. Following digestion with the appropriate enzymes (EcoRI and/or SphI), these inserts were purified and put into M13mp18 and -mp19.
IS1541 insertion specificity in Y. pestis 6/69M.
IS200 does not possess terminal inverted repeats and preferentially inserts into T/A-rich short segments (15, 21). Consequently, the ends of this element have not been definitively assigned and a duplication event at the target site, if any, has not been precisely characterized. We previously reported the insertion of IS1541 within the inv gene of Y. pestis, and in the present work we sequenced about 350 nucleotides of both IS-chromosome junction fragments in 14 different Y. pestis loci (MO951, MO952, MO953, MO954, MO955, MO956, MO957, MO958, MO959, MO960, MO962, MO963, MO964, and MO965). In all cases, the nucleotide sequence (100 bp) of both termini of IS1541 were identical to those of the prototype sequence (30). Sequence analysis revealed that the right extremity of IS1541, as defined in Fig. 2A, is almost identical to that of IS200, whereas their left extremities display sequence divergence within their outermost 7-bp segments. However, the ends of IS1541, like those of IS200, do not contain inverted repeats. Comparison of the sequences of the 14 target sites of IS1541 revealed that in all cases insertion occurred within a T/A-rich short (≤10-bp) segment (Fig. 2B). Two T’s were invariably present in both sides of the IS element, but since the sequence of targets prior to insertion is not known, it is not possible to determine if these motifs belong to the element or whether they result from an IS-mediated duplication event. More interestingly, in 13 of the 14 targets studied, sequence analysis revealed that the region upstream of the IS could form stem-loop structures having a minimum energy formation of <−10 kcal/mol. These motifs resemble rho-independent transcription terminators and might constitute hot spots for insertion of IS1541 within the Y. pestis genome. Precise characterization of the transposition behavior of IS1541 will require the development of a qualitative and quantitative transposition assay.
FIG. 2.
Nucleotide sequences adjacent to IS1541 in Y. pestis 6/69M. Sequencing of the IS flanking regions was achieved by the dideoxynucleotide chain termination method (29) with modified T7 DNA polymerase (Sequenase version 2.0; Amersham France) and primers located at the left (5′-CATTTGCAGTTGCCAG-3′) and right (5′-GTTTACGGGCCGTAA-3′) ends of the IS (30). In each case, the sequence of the complementary strand was carried out by using synthetic oligonucleotides designated from the sequence previously determined. (A) The 5′ and 3′ ends of IS200 and IS1541 are shown. Asterisks indicate identical nucleotides. (B) Nucleotide sequences (36 bp) upstream and downstream of 15 IS elements are shown. Inverted and complementary nucleotides are depicted by arrows below the sequences. The loop free energies (ΔG, in kilocalories) were calculated as reported by Zuker (33).
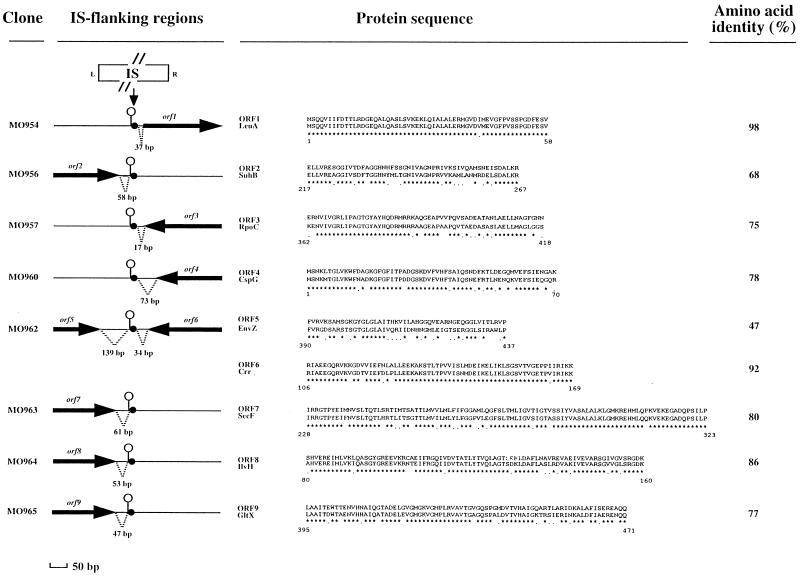
In none of the 14 target sequences was IS1541 inserted within large open reading frames (ORFs) (data not shown). However, sequence analysis using the BlastX program revealed that, in 9 of 14 cases, this IS inserted in the vicinity of an ORF (designated orf1 to orf9, of which only orf4 was entire) coding for a peptide highly homologous (47 to 98% identity) to a known bacterial protein from E. coli or Salmonella typhimurium (Fig. 3). In eight cases, IS1541 inserted downstream of genes thought to be either transcribed monocistronically (cspG, envZ, gltX, and suhB) or which constituted the last gene (crr, ilvH, rpoC, and secF) of an operon. These findings are consistent with our proposal that the hairpin structures present at the corresponding IS1541 insertion sites might correspond to transcriptional terminators. Insertion of IS1541 in the 5′ region of leuA, the first gene of the leucine operon, constitutes the only case of insertion of this IS upstream from a gene. By analogy with the S. typhimurium system, it is likely that this insertion has occurred in a hairpin structure belonging to the transcriptional regulatory region of the leu operon (10).
FIG. 3.
Insertions of IS1541 in eight coding regions of the Y. pestis 6/69M genome. Arrows represent ORFs. Asterisks and periods indicate identical and similar amino acids, respectively. Symbols: •, IS insertion site; ○|, hairpin structure. Left and right extremities of IS1541 are indicated by L and R, respectively. LeuA is the E. coli α-isopropylmalate synthase (26); SuhB is the extragenic suppressor protein of E. coli (31) which possesses inositol monophosphatase activity (18); RpoC is the β′ subunit of the E. coli RNA polymerase (22); CspG is an E. coli cold shock protein (19); EnvZ is the osmotic sensor from the two-component system OmpR-EnvZ of S. typhimurium (16); Crr is the component III (IIIGlc) of the phosphoenolpyruvate-dependent phosphotransferase system of E. coli (27); SecF is a protein export membrane protein of E. coli (9); IlvH is an acetolactate synthase of S. typhimurium (13) involved in isoleucine and valine synthesis; GltX is the glutamyl-tRNA synthetase of E. coli (2).
Characterization of IS1541 insertions in unrelated Y. pestis strains.
The next step was to consider whether, in other strains of Y. pestis, the IS1541 element flanked the same genes as in strain 6/69M. For this purpose, strains Hambourg 12 (biotype Orientalis, ribotype A), PKR XXIV (biotype Medievalis, ribotype O), Sénégal Th (biotype Orientalis, ribotype B), and Saïgon 55-1239 (biotype Orientalis, ribotype E) from the Centre National de Référence des Yersinia (Institut Pasteur, Paris, France) were investigated. As previously reported (30), these strains, isolated from different periods of time and geographical areas, displayed different IS1541 hybridization patterns. Y. pestis DNA was amplified by PCR with different sets of primers, each of them consisting of (i) a forward primer located within one of the previously defined ORFs (F1, 5′-CTGAGCGATAGTACG-3′ [orf1]; F2, 5′-CTATTGGTTCGTGAG-3′ [orf2]; F3, 5′-CGTGATGAATTGCGTGGC-3′ [orf3]; F4, 5′-GAAGTATCTCTATGTC-3′ [orf4]; F5, 5′-CGTGTTAAATCAGCG-3′ [orf5]; F6, 5′-CGTATCGCTGAAGAA-3′ [orf6]; F7, 5′-CGTCGTGGAACGCCT-3′ [orf7]; F8, 5′-CTCACATGTTGAGCG-3′ [orf8]; or F9, 5′-CGCCATCACCGAATG-3′[orf9]) and (ii) a reverse primer chosen in the IS element sequence (R1, 5′-CATTTGCAGTTGCCAG-3′ [used in PCR with F2, F5, F7, F8, and F9] or R2, 5′-TGCGGTCTGGCAACT-3′ [used in PCR with F1, F3, F4, and F6]). PCRs were performed as previously described (30), and amplimers were analyzed by 1% agarose gel electrophoresis. For the five unrelated strains, the IS element was always found, on the basis of product size, in the vicinity of leuA, suhB, rpoC, crr, secF, ilvH, envZ, or gltX but was not associated with suhB in strain PKR XXIV (data not shown). We previously reported that an identical copy of IS1541 was inserted at the same nucleotide position within the inv gene of strains 6/69M, Hambourg 12, PKR XXIV, Sénégal Th, and Saïgon 55-1239 (30). This indicates that acquisition of IS1541 by these strains was prior to their divergence. Data also suggest that insertions of IS1541 at a given loci constitute a stable event, which, in turn, raises the question of the efficiency of the intracellular mobility of these IS elements in Y. pestis. This last point is currently being investigated in the laboratory.
Nucleotide sequence accession numbers.
The sequences of orf1, orf2, orf3, orf4, orf5, orf6, orf7, orf8, and orf9 have been submitted to the EMBL nucleotide sequence database under accession numbers Z97975, Z97976, Z97977, Z97978, Z97979, Z97980, Z97981, Z97982, and Z97983, respectively.
Acknowledgments
We thank C. Buchrieser and A. Guiyoule for their collaboration and E. Carniel for a critical reading of the manuscript. We are also grateful to P. Berche for his constant interest in this work.
This research was supported partly by INSERM, the Université René Descartes (Paris), and the Conseil Régional Nord Pas-de Calais.
REFERENCES
- 1.Biserčić M, Ochman H. Natural populations of Escherichia coli and Salmonella typhimurium harbor the same classes of insertion sequences. Genetics. 1993;133:449–454. doi: 10.1093/genetics/133.3.449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Breton R, Sanfaçon H, Papayannopoulos I, Biemann K, Lapointe J. Glutamyl-tRNA synthetase of Escherichia coli. Isolation and primary structure of the gltX gene and homology with other aminoacyl-tRNA synthetases. J Biol Chem. 1986;261:10610–10617. [PubMed] [Google Scholar]
- 3.Brubaker R R. Factors promoting acute and chronic diseases caused by yersiniae. Clin Microbiol Rev. 1991;4:309–324. doi: 10.1128/cmr.4.3.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Carniel, E. Personal communication.
- 5.Ferber D M, Brubaker R R. Plasmids in Yersinia pestis. Infect Immun. 1981;31:839–841. doi: 10.1128/iai.31.2.839-841.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fetherston J D, Schuetze P, Perry R D. Loss of the pigmentation phenotype in Yersinia pestis is due to the spontaneous deletion of 102 kb of chromosomal DNA which is flanked by a repetitive element. Mol Microbiol. 1992;6:2693–2704. doi: 10.1111/j.1365-2958.1992.tb01446.x. [DOI] [PubMed] [Google Scholar]
- 7.Filippov A A, Oleinikov P N, Motin V L, Protsenko O A, Smirnov G B. Sequencing of two Yersinia pestis IS elements, IS285 and IS100. Contrib Microbiol Immunol. 1995;13:306–309. [PubMed] [Google Scholar]
- 8.Galas D J, Chandler M. Bacterial insertion sequences. In: Berg D E, Howe M M, editors. Mobile DNA. Washington, D.C: American Society for Microbiology; 1989. pp. 109–162. [Google Scholar]
- 9.Gardel C, Johnson K, Jacq A, Beckwith J. The secD locus of E. coli codes for two membrane proteins required for protein export. EMBO J. 1990;9:3209–3216. doi: 10.1002/j.1460-2075.1990.tb07519.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gemmill R M, Wessler S R, Keller E B, Calvo J M. leu operon of Salmonella typhimurium is controlled by an attenuation mechanism. Proc Natl Acad Sci USA. 1979;76:4941–4945. doi: 10.1073/pnas.76.10.4941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gibert I, Barbé J, Casadesús J. Distribution of insertion sequence IS200 in Salmonella and Shigella. J Gen Microbiol. 1990;136:2555–2560. doi: 10.1099/00221287-136-12-2555. [DOI] [PubMed] [Google Scholar]
- 12.Guiyoule A, Grimont F, Iteman I, Grimont P A D, Lefèvre M, Carniel E. Plague pandemics investigated by ribotyping of Yersinia pestis strains. J Clin Microbiol. 1994;32:634–641. doi: 10.1128/jcm.32.3.634-641.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jahreis K, Postma P W, Lengeler J W. Nucleotide sequence of the ilvH-fruR gene region of Escherichia coli K12 and Salmonella typhimurium LT2. Mol Gen Genet. 1991;226:332–336. doi: 10.1007/BF00273623. [DOI] [PubMed] [Google Scholar]
- 14.Lam S, Roth J R. IS200: a Salmonella-specific insertion sequence. Cell. 1983;34:951–960. doi: 10.1016/0092-8674(83)90552-4. [DOI] [PubMed] [Google Scholar]
- 15.Lam S, Roth J R. Structural and functional studies of insertion element IS200. J Mol Biol. 1986;187:157–167. doi: 10.1016/0022-2836(86)90225-1. [DOI] [PubMed] [Google Scholar]
- 16.Liljeström P, Laamanen I, Palva E T. Structure and expression of the ompB operon, the regulatory locus for the outer membrane porin regulon in Salmonella typhimurium LT-2. J Mol Biol. 1988;201:663–673. doi: 10.1016/0022-2836(88)90465-2. [DOI] [PubMed] [Google Scholar]
- 17.Lucier T S, Brubaker R R. Determination of genome size, macrorestriction pattern polymorphism, and nonpigmentation-specific deletion in Yersinia pestis by pulsed-field gel electrophoresis. J Bacteriol. 1992;174:2078–2086. doi: 10.1128/jb.174.7.2078-2086.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Matsuhisa A, Suzuki N, Noda T, Shiba K. Inositol monophosphatase activity from the Escherichia coli suhB gene product. J Bacteriol. 1995;177:200–205. doi: 10.1128/jb.177.1.200-205.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Nakashima K, Kanamaru K, Mizuno T, Horikoshi K. A novel member of the cspA family of genes that is induced by cold shock in Escherichia coli. J Bacteriol. 1996;178:2994–2997. doi: 10.1128/jb.178.10.2994-2997.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Odaert M, Berche P, Simonet M. Molecular typing of Yersinia pseudotuberculosis by using an IS200-like element. J Clin Microbiol. 1996;34:2231–2235. doi: 10.1128/jcm.34.9.2231-2235.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.O’Reilly C, Black G W, Laffey R, McConnell D J. Molecular analysis of an IS200 insertion in the gpt gene of Salmonella typhimurium LT2. J Bacteriol. 1990;172:6599–6601. doi: 10.1128/jb.172.11.6599-6601.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ovchinnikov Y A, Monastyrskaya G S, Gubanov V V, Guryev S O, Chertov O Y, Modyanov N N, Grinkevich V A, Makarova I A, Marchenko T V, Polovnikova I N, Lipkin V M, Sverdlov E D. The primary structure of Escherichia coli RNA polymerase. Nucleotide sequence of the rpoB gene and amino-acid sequence of the β-subunit. Eur J Biochem. 1981;116:621–629. doi: 10.1111/j.1432-1033.1981.tb05381.x. [DOI] [PubMed] [Google Scholar]
- 23.Perry R D, Fetherston J D. Yersinia pestis—etiologic agent of plague. Clin Microbiol Rev. 1997;10:35–66. doi: 10.1128/cmr.10.1.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Podladchikova O N, Dikhanov G G, Rakin A V, Heesemann J. Nucleotide sequence and structural organization of Yersinia pestis insertion sequence IS100. FEMS Microbiol Lett. 1994;121:269–274. doi: 10.1111/j.1574-6968.1994.tb07111.x. [DOI] [PubMed] [Google Scholar]
- 25.Protsenko O A, Filippov A A, Kutyrev V V. Integration of the plasmid encoding the synthesis of capsular antigen and murine toxin into Yersinia pestis chromosome. Microb Pathog. 1991;11:123–128. doi: 10.1016/0882-4010(91)90005-u. [DOI] [PubMed] [Google Scholar]
- 26.Ricca E, Calvo J M. The nucleotide sequence of leuA from Salmonella typhimurium. Nucleic Acids Res. 1990;18:1290. doi: 10.1093/nar/18.5.1290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Saffen D W, Presper K A, Doering T L, Roseman S. Sugar transport by the bacterial phosphotransferase system. Molecular cloning and structural analysis of the Escherichia coli ptsH, ptsI, and crr genes. J Biol Chem. 1987;262:16241–16253. [PubMed] [Google Scholar]
- 28.Sambrook J, Fritsch E F, Maniatis T. Molecular cloning: a laboratory manual. 2nd ed. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory Press; 1989. [Google Scholar]
- 29.Sanger F, Nicklen S, Coulson A R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA. 1977;12:5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Simonet M, Riot B, Fortineau N, Berche P. Invasin production by Yersinia pestis is abolished by insertion of an IS200-like element within the inv gene. Infect Immun. 1996;64:375–379. doi: 10.1128/iai.64.1.375-379.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yano R, Nagai H, Shiba K, Yura T. A mutation that enhances synthesis of ς32 and suppresses temperature-sensitive growth of the rpoH15 mutant of Escherichia coli. J Bacteriol. 1990;172:2124–2130. doi: 10.1128/jb.172.4.2124-2130.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zsigray R M, Hopper J B, Zukowski K, Chesbro W R. Integration of the Vwa plasmid into the chromosome of Yersinia pestis strains harboring F′ plasmids of Escherichia coli. Infect Immun. 1985;47:670–673. doi: 10.1128/iai.47.3.670-673.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zuker M. On finding all suboptimal foldings of an RNA molecule. Science. 1989;244:48–52. doi: 10.1126/science.2468181. [DOI] [PubMed] [Google Scholar]