Introduction
Key Teaching Points.
-
•
A diseased atrioventricular (AV) conduction system may harbor concealed/decremental conduction and is quite susceptible to “long-short” sequences.
-
•
Application of a magnet to a dual-chamber pacemaker in patients not only alters the pacing mode (synchronous DDD to asynchronous DOO) but also suddenly accelerates the rate of AV pacing.
-
•
This creates a long-short sequence in both atrium and ventricle, which is expected to be reflected as a long-short sequence to the diseased conduction system as well, resulting in varying electrocardiogram (ECG) manifestations.
-
•
A systematic analysis of device tracings/ECGs, familiarity with magnet rate responses, and appreciation of complex electrophysiological mechanisms at play is imperative to “troubleshoot” unusual pacemaker-related responses.
Asynchronous pacing is the expected response after application of a magnet to interrogate a pacemaker. An unanticipated response as illustrated in our case initially led to a diagnostic dilemma. However, a systematic approach with careful analysis of electrograms allowed us to clarify the complex electrophysiological mechanisms involved.
Case report
An asymptomatic 48-year-old man presented to our device clinic for a routine interrogation of his pacemaker. He had undergone a coronary artery bypass grafting and mitral valve replacement 5 years ago. Unfortunately, he developed infective endocarditis of his aortic valve within a year, which necessitated mechanical aortic valve replacement. This was complicated by persistent postoperative intermittent high-grade atrioventricular (AV) block. Therefore, we implanted a dual-chamber transvenous pacemaker (ADVISA MRI DR; Medtronic Inc, Minneapolis, MN) with right atrial and right ventricular apical leads. Previous device interrogations had not detected any sensing/pacing abnormalities. The pacemaker was programmed to a DDDR mode with a lower rate of 70 beats per minute (bpm), upper tracking rate of 120 bpm, upper sensor rate of 130 bpm, and sensed and paced AV delay of 150 and 180 milliseconds (ms) respectively.
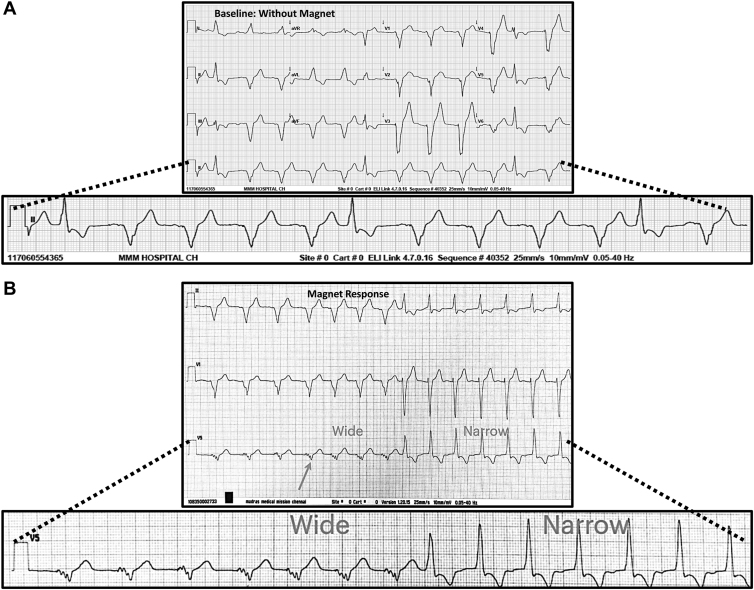
A 12-lead electrocardiogram (ECG) at baseline (Figure 1A) followed by magnet application (Figure 1B) is shown. Figure 1A shows a predominantly wide QRS rhythm (70 bpm) interspersed with shorter-coupled, relatively narrower QRS complexes (140 ms) of a different morphology. These were initially suspected to be premature ventricular complexes. When the magnet is applied, there is transition from the slower wide QRS rhythm (70 bpm) to the relatively narrower QRS rhythm at a slightly faster rate of 85 bpm (Figure 1B). A previous ECG showing intrinsic rhythm without pacing is also compared to the current one obtained after magnet application (Figure 1C). What is the most likely mechanism for the transition from wide to relatively narrower QRS rhythm pursuant to magnet application?
Figure 1.
A: Baseline 12-lead electrocardiogram (ECG) with lead II magnified in the lower panel showing wide complex rhythm at a rate of 70 beats per minute (bpm) (lower rate of pacemaker) with intermittent narrower QRS complexes (140 ms) of a different morphology. B: Long rhythm strip recorded during application of magnet showing transition from wide to narrower complex rhythm (85 bpm). The magnet response starts after the arrow. C: Upper left panel depicts a previous ECG without pacing (intrinsic rhythm). The upper right panel shows the 12-lead long rhythm strip ECG recorded during magnet response. The lower panel shows a magnified view of leads aVF, V1, and V2. Atrial pacing spikes (black arrows), onset of magnet response (blue arrow), and atrial/ventricular rates are also illustrated (also see discussion).
Discussion
Analysis of baseline ECG along with the magnified rhythm strip (Figure 1A) is compatible with sequential AV pacing at the lower rate of 70 bpm. Atrial and ventricular pacing spikes are associated with consistent capture, albeit with low-amplitude paced P waves. The atypical left bundle branch morphology of the wide QRS rhythm with its late precordial transition and left-superior axis is also compatible with right ventricular apical pacing. The narrower QRS complexes are followed by a delay in resumption of sequential AV pacing. This suggests that they are adequately sensed by the ventricular lead with appropriate reset of the pacemaker timing cycle. Also, the faster and narrower QRS rhythm after magnet application (Figure 1B) resembles the isolated beats seen in Figure 1A.
A thorough scrutiny of the magnet response (Figure 1B) reveals that the first 4 wide QRS beats (fourth beat denoted by arrow) occur at the lower pacemaker rate of 70 bpm and are preceded by tiny pacing spikes. This is consistent with DDD pacing, but the low-amplitude P waves are not easily discernable. This is followed by acceleration of the paced QRS complexes to 85 bpm. This is in fact the expected magnet rate response for a normally functioning newer-generation Medtronic pacemaker (asynchronous DOO pacing in this case). The first narrower QRS complex is coupled to the last paced QRS complex at a short cycle length of 600 ms (100 bpm). Subsequently the rate of the narrower QRS rhythm is also 85 bpm. Although a ventricular origin for the narrower QRS rhythm is possible, the exact rate of 85 bpm alludes to the possibility of a conducted rhythm.
Figure 1C is quite helpful in further clarifying the mechanism. The upper left panel demonstrates a slower intrinsic rhythm with shorter- and longer-coupled R-R intervals. There is a suggestion of low-amplitude P waves preceding some of the QRS complexes. It is, however, difficult to confidently annotate a P wave in front of every QRS. We can still infer that at least the shorter-coupled R-R intervals may be associated with sinus/atrial capture (conducted QRS) with prolonged AV conduction (better discernable in aVF).
The relatively narrower QRS complexes noted during magnet response (Figure 1C; upper right panel) exactly replicate (12/12-lead ECG match) the intrinsic QRS morphology (left panel). The magnified lower panel of Figure 1C shows tiny atrial pacing spikes (black arrows; at a rate of 85 bpm) preceding the narrower QRS complexes (also at a rate of 85 bpm) during the magnet response. The second black arrow is relatively shorter coupled (85 bpm) to the first black arrow and represents the onset of asynchronous DOO magnet response (blue arrow). The most likely inference is that there is 1:1 AV conduction with very prolonged PR interval during magnet response. The captured P waves are likely masked by the conducted QRS complexes.
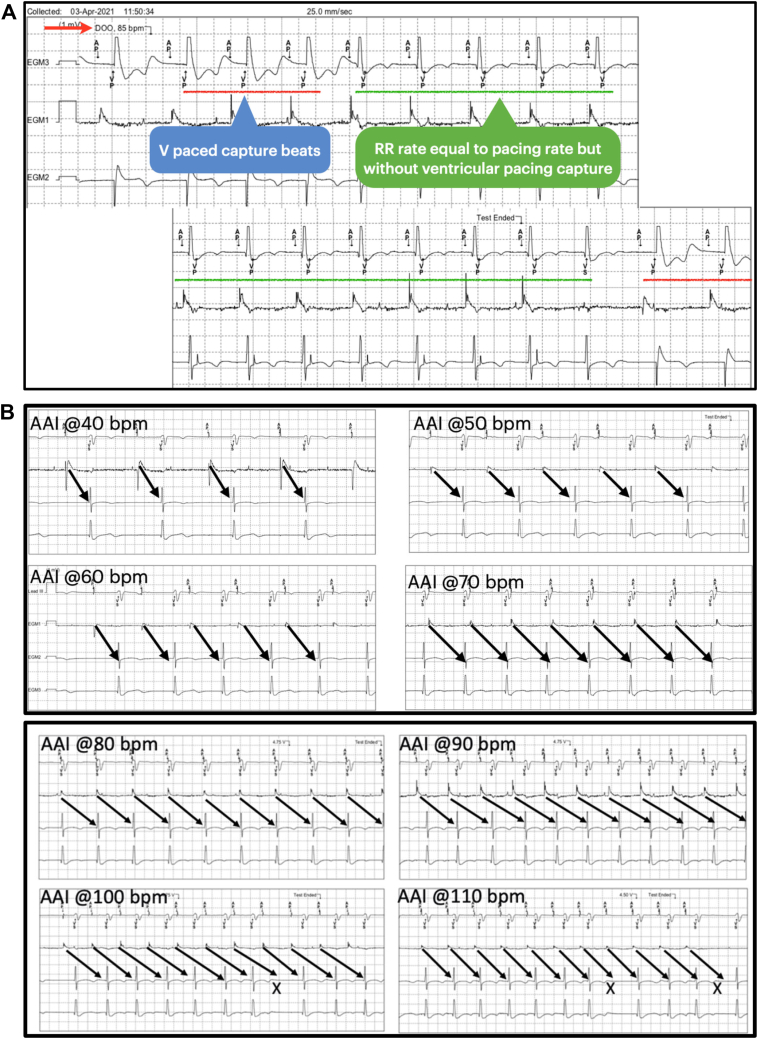
A mechanistic explanation is provided in subsequent Figures 2 and 3. Figure 2A clearly depicts onset and termination of magnet response. The conducted QRS complexes are not sensed during asynchronous DOO pacing. Ventricular pacing with functional noncapture occurs at the end of each conducted QRS. When the magnet test ends, the last atrial paced beat (rate of 85 bpm) conducts with a very long PR interval, resulting in a sensed QRS (annotated by the device as VS). Subsequently there is resumption of DDD pacing (70 bpm) with sequential atrial and ventricular capture. Atrial pacing at faster rates (from 40 bpm to 90 bpm) confirms decremental AV conduction with progressive prolongation of the PR interval and, eventually, early onset of AV Wenckebach (at and above atrial pacing rate of 100 bpm) (Figure 2B).
Figure 2.
A: Device electrogram showing onset and end of magnet response is shown. Atrial pacing (AP), ventricular pacing (VP), and ventricular sensing (VS) are annotated by the device. B: Device tracing demonstrating responses to atrial pacing at different rates (40–110 beats/min).
Figure 3.
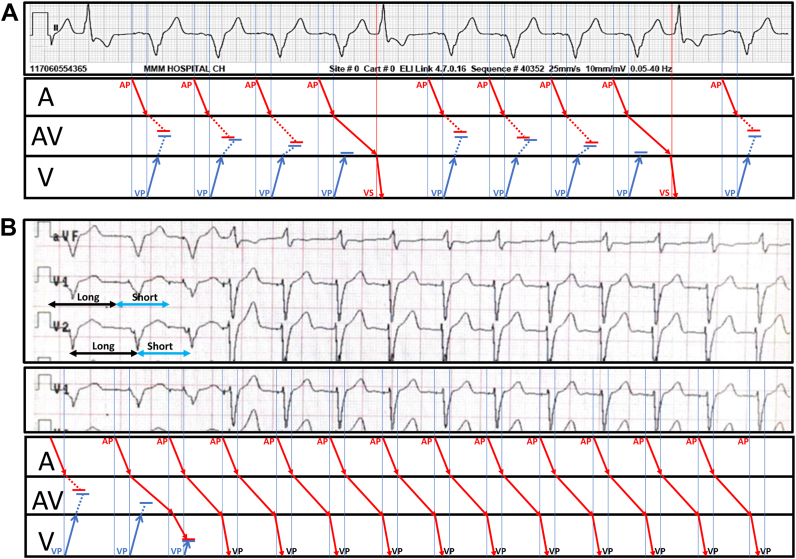
A: Baseline long rhythm electrocardiogram strip (upper panel) with concurrent ladder diagram explanation (lower panel). Annotations for atrium (A), atrioventricular conduction system (AV), ventricle (V), atrial pacing (AP), ventricular pacing (VP), and ventricular sensing (VS) are noted in the lower panel. B: Magnet response: long rhythm strip recorded during application of magnet (upper and mid panel) with concurrent ladder diagram explanation (lower panel). Long-short sequence created by magnet application at the atrial and ventricular level is denoted in the upper panel (see discussion).
A ladder diagram (Figure 3) is furnished to explicate the proposed electrophysiological mechanisms involved. During sequential AV pacing at baseline (Figure 3A) there is likely antegrade and retrograde concealed conduction into the AV conduction system. Decremental retrograde concealed conduction with retrograde block for every fourth paced beat (Wenckebach pattern) is postulated.1 This allows the preceding atrial paced beat to conduct to the ventricle with a very prolonged AV delay. This is followed by resumption of sequential AV pacing with both antegrade and retrograde concealed conduction, as described above.
Application of a magnet (Figure 3B) not only alters the pacing mode (synchronous DDD to asynchronous DOO) but also suddenly accelerates the rate of AV pacing (70 bpm to 85 bpm). This creates a long-short sequence in both atrium and ventricle, which is expected to be reflected as a long-short sequence to the diseased conduction system as well. This interrupts concealed conduction from the ventricle, but allows prolonged AV conduction (without Wenckebach) to manifest itself. The captured P wave is masked by the conducted QRS complexes. Upon close inspection, the ventricular pacing spikes can be appreciated toward the end of each conducted QRS (with functional noncapture), consistent with expected magnet response.
The asynchronous magnet rate response before elective replacement index is reached varies between different models of Medtronic pacemakers. The more familiar response consists of 3 paced beats at a rate of 100 bpm for threshold margin testing followed by pacing at 85 bpm.2 In the newer generation of pacemakers such as ours, it is set at 85 bpm without any threshold margin testing (https://www.medtronicacademy.com/features/magnet-mode-feature). The interval between the last captured ventricular beat (third beat) and first conducted QRS complex (fourth beat) is noted to be 600 ms (100 bpm) in Figure 1B. Not purely coincidental, this abbreviated interval compared to the magnet rate serves as a pointer toward the possibility of intrinsic AV conduction driving the subsequent QRS closer to the last paced one.
Conclusion
A diseased AV conduction system, as encountered in our patient, may harbor concealed/decremental conduction and is susceptible to “long-short” sequences. This can yield interesting interactions between conducted and paced rhythms even at relatively slower pacing cycle lengths. However, as showcased here, (1) a systematic analysis of device tracings/ECGs, (2) familiarity with magnet rate responses, and (3) appreciation of complex electrophysiological mechanisms at play are imperative to “troubleshoot” any unusual pacemaker-related responses.
Acknowledgments
Funding Sources
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Disclosures
None.
References
- 1.Jazayeri M.R. Role of concealed conduction and allied phenomena in the genesis, maintenance, and termination of cardiac arrhythmias. Pacing Clin Electrophysiol. 2019;42:779–804. doi: 10.1111/pace.13699. [DOI] [PubMed] [Google Scholar]
- 2.Jacob S., Panaich S.S., Maheshwari R., et al. Clinical applications of magnets on cardiac rhythm management devices. Europace. 2011;13:1222–1230. doi: 10.1093/europace/eur137. [DOI] [PubMed] [Google Scholar]