Abstract
Background
The effectiveness of cerebral embolic protection devices (CEPD) in mitigating stroke after transcatheter aortic valve implantation (TAVI) remains uncertain, and therefore CEPD may be utilized differently across US hospitals. This study aims to characterize the hospital-level pattern of CEPD use during TAVI in the US and its association with outcomes.
Methods
Patients treated with nontransapical TAVI in the 2019 Nationwide Readmissions Database were included. Hospitals were categorized as CEPD non-users and CEPD users. The following outcomes were compared: the composite of in-hospital stroke or transient ischemic attack (TIA), in-hospital ischemic stroke, death, and cost of hospitalization. Logistic regression models were used for risk adjustment of clinical outcomes.
Results
Of 41,822 TAVI encounters, CEPD was used in 10.6% (n = 4422). Out of 392 hospitals, 65.8% were CEPD non-user hospitals and 34.2% were CEPD users. No difference was observed between CEPD non-users and CEPD users in the risk of in-hospital stroke or TIA (adjusted odds ratio (OR) = 0.99 [0.86-1.15]), ischemic stroke (adjusted OR = 1.00 [0.85-1.18]), or in-hospital death (adjusted OR = 0.86 [0.71-1.03]). The cost of hospitalization was lower in CEPD non-users.
Conclusions
Two-thirds of hospitals in the US do not use CEPD for TAVI, and no significant difference was observed in neurologic outcomes among patients treated at CEPD non-user and CEPD user hospitals.
Keywords: CEPD, Stroke, TAVI
Introduction
Stroke remains a major potential adverse event after transcatheter aortic valve implantation (TAVI), and it is associated with high mortality and morbidity.1,2 Based on the strong association of stroke with 1-year mortality after TAVI, stroke is now a key component of the composite outcome for benchmarking site performance in TAVI used by the American College of Cardiology (ACC)/Society of Thoracic Surgeons (STS) Transcatheter Valve Therapy (TVT) Registry.3 In the TVT Registry, the 30-day stroke rate following TAVI was 2.3% and did not decline significantly during the first 5 years of commercial TAVI in the United States (US) despite declining patient risk profiles.4 Cerebral embolic protection devices (CEPD)s were developed to minimize neurological complications during TAVI. Although the first CEPD was approved by the US Food and Drug Administration for commercial use in 2017, prior studies have shown conflicting results regarding the clinical benefits of CEPDs.5, 6, 7, 8, 9 It remains unclear whether CEPD use is a valuable strategy for hospitals to improve TAVI outcomes. The recently published PROTECTED TAVR randomized trial of CEPD use during TAVI did not show a significant reduction in periprocedural stroke, adding more uncertainty around the benefit of CEPD during TAVI.10 It is unknown how hospitals should incorporate CEPD into TAVI programs, and whether CEPD use impacts hospital-level TAVI outcomes. The purpose of this study was to characterize the pattern of CEPD use at the hospital level in the US, and to evaluate whether treatment with TAVI at a hospital that uses CEPD is associated with a lower risk of in-hospital adverse events.
Methodology
This was a retrospective multicenter cohort study. We queried the Nationwide Readmissions Database (NRD) for the year 2019. NRD is a publicly available database of all-payer inpatient stays developed by the Agency for Healthcare Research and Quality (AHRQ) as part of the Healthcare Cost and Utilization Project (HCUP). The NRD is drawn from the State Inpatient Databases that contain reliable and verified patient linkage numbers that can be used to track a patient across hospitals within a state while adhering to strict privacy guidelines. The NRD includes data from 30 states for the year 2019. These data account for 49.3% of the total US resident population and 49.1% of all hospitalizations. The NRD includes all discharge records of patients treated in US community hospitals excluding rehabilitation and long-term acute care facilities. Discharge weights are provided to obtain national estimates. This study was deemed exempt by the Saint Luke’s Hospital institutional review board as the NRD is a publicly available database that contains deidentified patient information.
Study Population
The NRD database was queried for patients who underwent percutaneous non-transapical TAVI. International Classification of Diseases 10 Clinical Modification procedure codes 02RF37Z, 02RF38Z, 02RF3JZ, or 02RF3KZ were used to identify all non-apical TAVI patients aged ≥18 years. Code X2A5312 was used to identify CEPD use. Codes used to identify cerebrovascular events and other covariates are provided in Supplemental Table 1. Prior validation studies have shown a sensitivity of 67%, a specificity of 99%, a positive predictive value of >80% to 90%, and a negative predictive value of ≥95% for these codes for the diagnosis of transient ischemic attack (TIA) or stroke and stroke subtypes. Patients with missing records or patients with missing data were excluded from the analysis.
Based on CEPD use during TAVI procedures, the hospitals identified from NRD were classified into CEPD non-user and CEPD user groups. Hospitals with <10 TAVI procedures were excluded from the analysis (Figure 1).
Figure 1.
Study Population.
Abbreviations: CEPD, cerebral embolic protection device; TAVI, transcatheter aortic valve implantation.
Outcomes
The primary outcome was the composite of in-hospital TIA or stroke (ischemic and hemorrhagic). Secondary outcomes included in-hospital mortality, in-hospital ischemic stroke, and total cost of hospitalization. The data include the total charge by the hospital for each admission. To calculate the estimated costs of hospitalization, data were merged with the costs:charge ratio available for each hospital from HCUP. The total costs for hospitalization were calculated by multiplying total charges with the appropriate costs:charge ratio. This methodology has been previously validated.11
Statistical Analysis
Categorical variables are expressed as percentages and continuous variables as mean ± standard deviation or median (interquartile range [IQR]) as appropriate. Unadjusted comparisons of categorical variables were made using Pearson X2 tests or Fisher’s exact tests, and unadjusted comparisons of continuous variables were made using Student’s t-tests. In-hospital TIA or stroke, mortality, and ischemic stroke were risk-adjusted using multivariable logistic regression models to account for potential confounding due to varying patient risk profiles between hospital groups. Multivariable logistic regression models were fit using variables derived from the TVT Registry stroke and mortality risk models,12,13 which could be ascertained in the NRD. These variables included age, sex, diabetes mellitus, hypertension, peripheral vascular disease, prior myocardial infarction, home oxygen, dialysis, tricuspid insufficiency, heart failure with reduced ejection fraction, prior stroke, and atrial fibrillation or atrial flutter. Effect sizes were reported as adjusted odds ratio (OR) and 95% confidence interval (CI). All p values were 2-sided with a significance threshold of p < 0.05. Statistical analyses were performed using SAS 9.4 (Cary, NC).
Results
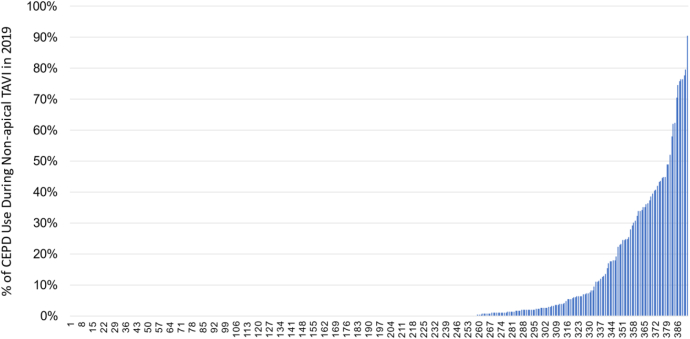
The study cohort included 41,666 patient admissions at 392 hospitals. CEPD was used in 4416 admissions (10.6% of admissions) at 134 hospitals (34.2% of hospitals). Among the 134 hospitals using CEPD, there was a large amount of variability in the rate of CEPD use which was observed (Figure 2). Only 12 hospitals (3.1% of all hospitals) used CEPD in >50% of procedures, while 122 hospitals (31.1% of all hospitals) used CEPD but did so in <50% of procedures. There were 258 CEPD non-user hospitals (65.8% of all hospitals).
Figure 2.
Histogram of Hospital Level CEPD Use for TAVI. Individual hospitals are plotted on the X axis with each hospital’s % of CEPD use during TAVI in 2019 plotted on the Y axis. Among 392 total hospitals, there were 258 CEPD non-user hospitals (65.8%) and 134 CEPD user hospitals (34.2%). Among the CEPD user hospitals there was considerable variability in CEPD use with some hospitals using CEPD in <10% of procedures and others using CEPD in >50% of procedures.
Abbreviations: CEPD, cerebral embolic protection device; TAVI, transcatheter aortic valve implantation.
The baseline characteristics of the study population stratified by hospital CEPD use are demonstrated in Table 1. At CEPD user hospitals, CEPD was used in 21.3% of all procedures. Baseline characteristics were otherwise well balanced between CEPD use groups, although small statistically significant differences were observed in diabetes mellitus, hypertension, peripheral vascular disease, obesity, chronic pulmonary disease, and tricuspid regurgitation between groups.
Table 1.
Baseline characteristics by hospital CEPD use during TAVI
| Patients treated at CEPD non-user hospitals n = 20,939 | Patients treated at CEPD user hospitals n = 20,727 | p | |
|---|---|---|---|
| CEPD Use | 0 (0.0%) | 4416 (21.3%) | <0.001 |
| Age in years | 78.6 ± 8.4 | 78.7 ± 8.7 | 0.16 |
| Female sex | 9176 (43.8%) | 9137 (44.1%) | 0.60 |
| Diabetes mellitus | 8000 (38.2%) | 7622 (36.8%) | 0.002 |
| Hypertension | 18,478 (88.2%) | 18,536 (89.4%) | <0.001 |
| Peripheral vascular disease | 4218 (20.1%) | 4475 (21.6%) | <0.001 |
| Obesity | 4435 (21.2%) | 4220 (20.4%) | 0.04 |
| Chronic pulmonary disease | 5580 (26.6%) | 5216 (25.2%) | 0.001 |
| Home oxygen | 750 (3.6%) | 751 (3.6%) | 0.82 |
| Dialysis | 490 (2.3%) | 466 (2.2%) | 0.53 |
| Dementia | 775 (3.7%) | 784 (3.8%) | 0.66 |
| Coronary artery disease | 13,843 (66.1%) | 13,803 (66.6%) | 0.30 |
| Prior myocardial infarction | 2066 (9.9%) | 2130 (10.3%) | 0.16 |
| Prior stroke | 2105 (10.1%) | 2081 (10.0%) | 0.96 |
| Atrial fibrillation or flutter | 7386 (35.3%) | 7422 (35.8%) | 0.25 |
| Heart failure with reduced ejection fraction | 2092 (10.0%) | 2030 (9.8%) | 0.50 |
| Tricuspid regurgitation | 378 (1.8%) | 447 (2.2%) | 0.01 |
| Median household income national quartile for patient ZIP Code | <0.001 | ||
| 1 | 4891 (23.6%) | 3091 (15.1%) | |
| 2 | 5958 (28.8%) | 4409 (21.5%) | |
| 3 | 5702 (27.6) | 5928 (29.0%) | |
| 4 | 4143 (20.0%) | 7043 (34.4%) | |
| Missing | 245 | 256 | |
| Primary expected payer | <0.001 | ||
| Medicare | 18,549 (88.7%) | 18,533 (89.5%) | |
| Medicaid | 292 (1.4%) | 287 (1.4%) | |
| Private Ins | 1475 (7.1%) | 1519 (7.3%) | |
| Self-Pay | 45 (0.2%) | 74 (0.4%) | |
| No Charge | 17 (0.1%) | 0 (0.0%) | |
| Other | 539 (2.6%) | 298 (1.4%) | |
| Missing | 22 | 16 | |
| Bed size of hospital | <0.001 | ||
| Small | 1461 (7.0%) | 428 (2.1%) | |
| Medium | 5058 (24.2%) | 4232 (20.4%) | |
| Large | 14,420 (68.9%) | 16,067 (77.5%) | |
| Control/ownership of the hospital | <0.001 | ||
| Government | 2320 (11.1%) | 1385 (6.7%) | |
| Private not-profit | 16,201 (77.4%) | 17,990 (86.8%) | |
| Private invest-own | 2418 (11.5%) | 1352 (6.5%) | |
| Teaching status of urban hospital | <0.001 | ||
| Metro non-teaching | 3104 (14.8%) | 1207 (5.8%) | |
| Metro teaching | 17,613 (84.1%) | 19,391 (93.6%) | |
| Non-Metro | 222 (1.1%) | 129 (0.6%) |
Abbreviations: CEPD, cerebral embolic protection device; TAVI, transcatheter aortic valve implantation.
There were notable differences observed in patient socioeconomic position and hospital characteristics between groups. CEPD non-user hospitals had a higher proportion of patients in the first quartile of median household income by zip code, while CEPD user hospitals had a higher proportion of patients in the fourth quartile of median household income. The CEPD non-user hospitals were more likely to be small, privately owned for-profit hospitals, and non-teaching sites compared with the CEPD user hospitals.
There were no significant differences observed in the unadjusted risk of in-hospital death, stroke or TIA, ischemic stroke, hemorrhagic stroke, or TIA among encounters at CEPD non-user and CEPD user hospitals (Table 2). Figure 3 shows risk-adjusted outcomes by hospital CEPD use. No significant difference was observed between CEPD non-user hospitals vs. CEPD user hospitals for in-hospital stroke or TIA (OR 0.99 [0.86-1.50], p = 0.99), ischemic stroke (OR 1.00 [0.85-1.18], p = 1.00), or death (OR 0.86 [0.71-1.03], p = 0.10).
Table 2.
Unadjusted in-hospital TAVI outcomes by hospital CEPD use
| Patients treated at CEPD non-user hospitals n = 20,939 | Patients treated at CEPD user hospitals n = 20,727 | p | |
|---|---|---|---|
| Death | 249 (1.2%) | 223 (1.1%) | 0.27 |
| Stroke or TIA | 364 (1.7%) | 366 (1.8%) | 0.83 |
| Ischemic stroke | 291 (1.4%) | 294 (1.4%) | 0.80 |
| Hemorrhagic stroke | 20 (0.1%) | 17 (0.1%) | 0.64 |
| TIA | 62 (0.3%) | 62 (0.3%) | 0.95 |
Abbreviations: CEPD, cerebral embolic protection device; TAVI, transcatheter aortic valve implantation; TIA, transient ischemic attack.
Figure 3.
Risk-Adjusted TAVI Outcomes by Hospital Level CEPD Use. All models were risk adjusted for the following variables: age, sex, diabetes mellitus, hypertension, peripheral vascular disease, prior myocardial infarction, home oxygen, dialysis, tricuspid insufficiency, heart failure with reduced ejection fraction, prior stroke, atrial fibrillation, atrial flutter, hospital size, hospital control/ownership, hospital location, and hospital teaching status.
Abbreviations: CEPD, cerebral embolic protection device; TAVI, transcatheter aortic valve implantation; TIA, transient ischemic attack.
A lower cost of hospitalization was observed in the CEPD non-user hospitals, while encounters for TAVI at CEPD user hospitals were associated with approximately $6000 higher cost per encounter (Figure 4).
Figure 4.
Cost for Index TAVI Admission by Hospital Level CEPD Use. Values are mean US dollars with error bar indicating standard deviation.
Abbreviations: CEPD, cerebral embolic protection device; TAVI, transcatheter aortic valve implantation; US, United States.
Discussion
This retrospective, observational study of a large national registry demonstrated the following principal findings (Figure 5). First, approximately two-thirds of US TAVI programs never use CEPD during TAVI (Figure 3). Second, hospitals which do not use CEPD are more commonly small, private for-profit, non-teaching sites compared with the hospitals that do use CEPD. CEPD non-user hospitals also treat patients from a lower socioeconomic position more commonly than CEPD user hospitals. Third, patients treated at CEPD user hospitals did not have lower rates of in-hospital adverse outcomes such as stroke or death. Finally, CEPD user hospitals had significantly higher cost of hospitalization for TAVI.
Figure 5.
Pattern of CEPD Use for TAVI in US Hospitals in 2019.
Abbreviations: CEPD, cerebral embolic protection device; TAVI, transcatheter aortic valve implantation; US, United States.
Six pivotal RCTs have evaluated CEPDs during TAVI: SENTINEL,6 CLEAN-TAVI,7 DEFLECT-III,14 EMBOL-X,15 MISTRAL-C16 and PROTECTED TAVR.10 The CLEAN TAVI trial included 100 patients and showed a significantly lower total number of lesions on MRI but no difference in clinical strokes. In the MISTRAL-C trial, the results suggested that CEPD use could reduce the number and size of MRI lesions. The SENTINEL trial showed that the CEPD was safe and reduced embolic burden in 99% of the patients but the reduction in lesions seen on MRI was not significant. However, a post hoc analysis did show a reduction in 72-hour stroke in the device arm, and these data contributed toward eventual commercial approval of the device in 2017. There have also been observational studies which have supported CEPD use with promising improvements in clinical outcomes observed. The recent analysis by Khan et al.17 from the Nationwide Inpatient Sample and NRD found that patient-level CEPD use was associated with reduced in-hospital mortality and neurological complications in a retrospective study involving 4380 patients.
However, despite approval of CEPD by the US Food and Drug Administration 6 years ago, there remains considerable uncertainty about the true effectiveness of this technology to mitigate neurologic risk during TAVI. The concerns regarding the benefits of CEPD during TAVI were renewed by the recent publication of the PROTECTED TAVR trial, which was the largest randomized trial of CEPD use during TAVI. That study did not show any reduction in the primary endpoint of periprocedural stroke within 72 hours after TAVI, although there was a reduction in the secondary endpoint of disabling stroke in the CEPD group. Similar negative findings have been previously reported in numerous observational studies as well. An NRD-based study evaluated the impact of patient-level CEPD use on postdischarge stroke prevention and found that the overall stroke incidence postdischarge within 6 months was similar between CEPD and non-CEPD groups.18 Another recent study by Kolte et al.9 showed no significant decrease in in-hospital stroke, ischemic stroke, hemorrhagic stroke, TIA, and all-cause mortality with CEPD use during TAVI. Similarly, a study from the STS/ACC TVT Registry found no association between CEPD use and in-hospital strokes.8 Taken together, the conflicting results from trials and registry studies has created uncertainty about whether CEPDs should be used during TAVI and in which patients.
The high cost of CEPD, when coupled with conflicting clinical effectiveness data, contributes to the challenges for US hospitals when deciding on whether to adopt CEPD use. A prior cost-effectiveness analysis which evaluated the cost of CEPDs in relation to their effect, showed that using a Sentinel device during TAVI is economically cost-effective from a Medicare payer perspective.19 The present study shows higher cost of hospitalization of approximately $6000 USD per admission for patients in hospitals that use CEPD. However, the magnitude of the difference in hospital costs in this study is more than the actual cost of the device, which implies that hospitals that do not use CEPD may be more attentive to other cost saving measures during TAVI as well. We observed that patients treated at CEPD non-user hospitals were more commonly in a lower socioeconomic position, and these hospitals are more commonly small or medium bed size with private ownership and non-teaching status. Our findings suggest that CEPD use may be associated with the cost sensitivity of the hospital.
Our results show that two-thirds of TAVI programs in the US have chosen not to use CEPD, which may be due to a lack of sufficient evidence and a lack of sufficient reimbursement to justify its use. While most hospitals never use CEPD, the proportion of TAVI cases performed with CEPD in the US is rising slowly. Kolte et al. found that CEPD use during TAVI in the US is 3.9%, but utilization increased from 2.3% in 2017 to 14.7% in 2018. Similarly, data from the TVT Registry show that the rate of CEPD use during TAVI increased from 5% to 13% in 2018-2019.13 Despite small but steady growth in CEPD use overall, our study shows large variability in CEPD practice patterns across US TAVI centers.
A new patient-focused TAVI quality score was recently introduced by the TVT Registry.3 In this score, risk-standardized results are estimated for 5 outcomes at 30 days, including mortality, stroke, major bleeding, acute kidney injury, or moderate to severe paravalvular regurgitation. These outcomes were selected in a hierarchical manner based on the strength of association with 1-year mortality. Scores are reported as “site difference” or “win-difference”, which is a method of reporting risk-standardized composite outcomes that have different weights based on clinical importance and timing. Publicly, this is represented by a “star rating,” which includes 1-3 stars for outcomes “better than expected,” “as expected,” or “worse than expected.” Based on this methodology for public reporting, site-level TAVI stroke rates have become a major driver in the measure of hospital performance.20 Our study shows that a strategy of selectively using CEPD during TAVI is not associated with reduced stroke rates, and adopting CEPD is unlikely to improve performance on the TVT Registry composite metric for TAVI quality.
Limitations
This was a retrospective observational design that is subject to residual confounding despite risk adjustment. The clustering of outcomes by site for reasons unrelated to CEPD use was not accounted for in this study. NRD is an administrative database based on coding data. Incorrect coding may lead to under-reporting or over-reporting of CEPD use and possible miscoding of outcomes. However, there is no reason to expect that these errors would disproportionately affect one study group more than another. The NRD lacks granular geographical, anatomic, procedural, and clinical data which may be related to neurologic outcomes. For example, data on alternative access types for percutaneous TAVI are absent, and it is known that nonfemoral access is associated with a higher risk of procedural stroke. Not all variables included in the TVT risk models are available in the data set and hence could not be adjusted for in the analysis. The NRD does not contain long-term outcome data unless they are related to hospital readmission to an NRD-participating hospital within the same calendar year.
Conclusion
Most US hospitals have not adopted CEPD for prevention of strokes during TAVI. Although CEPD use is associated with increased cost, there is no evidence of better neurologic outcomes for patients treated in hospitals using CEPD.
Ethics Statement
This research was conducted ethically and all the authors are in agreement with this statement. The NRD is a publicly available database with no individually identifiable data, and thus does not constitute research with human subjects (as defined under 45CFR46:102).
Funding
This research did not receive any specific grant from funding agencies.
CRediT Authorship Contributions
Aamer Ubaid, MD (Conceptualization; Writing – original draft; Writing – review & editing),
Kevin F. Kennedy, MS (Formal analysis),
Adnan K. Chhatriwalla, MD (Writing – review & editing),
John T. Saxon, MD (Writing – review & editing),
Anthony Hart, MD (Writing – review & editing),
Keith B. Allen, MD (Writing – review & editing),
Corinne Aberle, MD (Writing – review & editing),
Islam Shatla, MD (Writing – original draft (supporting)),
Abdelrhman Abumoawad, MD (Writing – original draft (supporting)),
Satya Preetham Gunta, MD (Writing – original draft (supporting)),
David Skolnick, MD (Supervision),
Chetan P. Huded, MD, MSc (Conceptualization; Writing – original draft (equal); writing – review & editing; Supervision).
Disclosure Statement
J. T. Saxon is a proctor for Edwards Lifesciences and Medtronic Inc and is on the speaker Bbreau for Abbott Vascular, Medtronic Inc, and Edwards Lifesciences. Dr A. K. Chhatriwalla has a grant from Boston Scientific, is a proctor for Edwards Lifesciences and Medtronic Inc, and is on the speaker bureau for Abbott Vascular, Edwards Lifesciences, and Medtronic Inc. Dr K. B. Allen is an institutional research supporter for Abbott, Medtronic, Edwards, and Boston Scientific and is a proctor and on speaker bureau for Abbott, Medtronic, and Edwards with all payments to the institution and none to him personally. Dr C. P Huded has consulting fees from Boston Scientific and a grant from Abbott Vascular.
Footnotes
Supplemental data for this article can be accessed on the publisher’s website.
Supplementary Material
References
- 1.Arnold S.V., Manandhar P., Vemulapalli S., et al. Impact of short-term complications of transcatheter aortic valve replacement on longer-term outcomes: results from the STS/ACC transcatheter valve therapy registry. Eur Heart J Qual Care Clin Outcomes. 2021;7(2):208–213. doi: 10.1093/ehjqcco/qcaa001. [DOI] [PubMed] [Google Scholar]
- 2.Kapadia S., Agarwal S., Miller D.C., et al. Insights into timing, risk factors, and outcomes of stroke and transient ischemic attack after transcatheter aortic valve replacement in the PARTNER trial (placement of aortic transcatheter valves) Circ Cardiovasc Interv. 2016;9(9):e002981. doi: 10.1161/CIRCINTERVENTIONS.115.002981. [DOI] [PubMed] [Google Scholar]
- 3.Desai N.D., O’Brien S.M., Cohen D.J., et al. Composite metric for benchmarking site performance in transcatheter aortic valve replacement: results from the STS/ACC TVT registry. Circulation. 2021;144(3):186–194. doi: 10.1161/CIRCULATIONAHA.120.051456. [DOI] [PubMed] [Google Scholar]
- 4.Huded C.P., Tuzcu E.M., Krishnaswamy A., et al. Association between transcatheter aortic valve replacement and early postprocedural stroke. JAMA. 2019;321(23):2306. doi: 10.1001/jama.2019.7525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bagur R., Solo K., Alghofaili S., et al. Cerebral embolic protection devices during transcatheter aortic valve implantation. Stroke. 2017;48(5):1306–1315. doi: 10.1161/STROKEAHA.116.015915. [DOI] [PubMed] [Google Scholar]
- 6.Kapadia S.R., Kodali S., Makkar R., et al. Protection against cerebral embolism during transcatheter aortic valve replacement. J Am Coll Cardiol. 2017;69(4):367–377. doi: 10.1016/j.jacc.2016.10.023. [DOI] [PubMed] [Google Scholar]
- 7.Haussig S., Mangner N., Dwyer M.G., et al. Effect of a cerebral protection device on brain lesions following transcatheter aortic valve implantation in patients with severe aortic stenosis. JAMA. 2016;316(6):592. doi: 10.1001/jama.2016.10302. [DOI] [PubMed] [Google Scholar]
- 8.Butala N.M., Makkar R., Secemsky E.A., et al. Cerebral embolic protection and outcomes of transcatheter aortic valve replacement: results from the transcatheter valve Therapy registry. Circulation. 2021;143(23):2229–2240. doi: 10.1161/CIRCULATIONAHA.120.052874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kolte D., Khera S., Nazir S., Butala N.M., Bhatt D.L., Elmariah S. Trends in cerebral embolic protection device use and association with stroke following transcatheter aortic valve implantation. Am J Cardiol. 2021;152:106–112. doi: 10.1016/j.amjcard.2021.04.038. [DOI] [PubMed] [Google Scholar]
- 10.Kapadia S.R., Makkar R., Leon M., et al. Cerebral embolic protection during transcatheter aortic-valve replacement. N Engl J Med. 2022;387(14):1253–1263. doi: 10.1056/NEJMoa2204961. [DOI] [PubMed] [Google Scholar]
- 11.Cost-to-charge ratio files: user guide for nationwide readmissions database (NRD) CCRs. 2020. https://www.hcup-us.ahrq.gov/db/ccr/costtocharge.jsp
- 12.Thourani V.H., O’Brien S.M., Kelly J.J., et al. Development and application of a risk prediction model for in-hospital stroke after transcatheter aortic valve replacement: a report from the Society of Thoracic Surgeons/American College of Cardiology transcatheter valve therapy registry. Ann Thorac Surg. 2019;107(4):1097–1103. doi: 10.1016/j.athoracsur.2018.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Carroll J.D., Mack M.J., Vemulapalli S., et al. STS-ACC TVT registry of transcatheter aortic valve replacement. Ann Thorac Surg. 2021;111(2):701–722. doi: 10.1016/j.athoracsur.2020.09.002. [DOI] [PubMed] [Google Scholar]
- 14.Lansky A.J., Schofer J., Tchetche D., et al. A prospective randomized evaluation of the TriGuardTM HDH embolic DEFLECTion device during transcatheter aortic valve implantation: results from the DEFLECT III trial. Eur Heart J. 2015;36(31):2070–2078. doi: 10.1093/eurheartj/ehv191. [DOI] [PubMed] [Google Scholar]
- 15.Banbury M.K., Kouchoukos N.T., Allen K.B., et al. Emboli capture using the Embol-X intraaortic filter in cardiac surgery: a multicentered randomized trial of 1,289 patients. Ann Thorac Surg. 2003;76(2):508–515. doi: 10.1016/S0003-4975(03)00530-7. [DOI] [PubMed] [Google Scholar]
- 16.Mieghem N.M.V., van Gils L., Ahmad H., et al. Filter-based cerebral embolic protection with transcatheter aortic valve implantation: the randomised MISTRAL-C trial. EuroIntervention. 2016;12(4):499–507. doi: 10.4244/EIJV12I4A84. [DOI] [PubMed] [Google Scholar]
- 17.Khan M.Z., Zahid S., Khan M.U., et al. Use and outcomes of cerebral embolic protection for transcatheter aortic valve replacement: a US nationwide study. Catheter Cardiovasc Interv. 2021;98(5):959–968. doi: 10.1002/ccd.29842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Majmundar M., Kumar A., Kalra A. Impact of embolic protection device placed during transcatheter aortic valve implantation on postdischarge stroke prevention. Am J Cardiol. 2021;158:150–152. doi: 10.1016/j.amjcard.2021.08.004. [DOI] [PubMed] [Google Scholar]
- 19.Alkhouli M., Ganz M., Mercaldi K., McGovern A., Griffiths R., Kapadia S. TCT-305 cost-effectiveness of cerebral embolic protection with the SENTINEL device in transcatheter aortic valve replacement: a US Medicare payer perspective. J Am Coll Cardiol. 2021;78(19):B125. doi: 10.1016/j.jacc.2021.09.1158. [DOI] [Google Scholar]
- 20.The Society of Thoracic Surgeons (STS)/American College of Cardiology (ACC) STS/ACC TVT registry. An initiative of the STS natioanl database and teh ACCs NCDR. Companion guide for public reporting. 2020. https://www.ncdr.com/WebNCDR/docs/default-source/tvt-public-page-documents/tvt_registry_public_reporting_participant_companion_guide_11_8_2021.pdf?sfvrsn=610ed69f_8
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.