Abstract
Idiopathic mediastinal fibrosis, also called sclerosing or fibrosing mediastinitis, is a very rare and aggressive fibroinflammatory process characterized by fibrous tissue proliferation in the mediastinal region. Herein, we present a rare case of idiopathic mediastinal fibrosis presenting with esophageal obstruction, most likely associated with immunoglobulin G (IgG4)-related disease, affecting the posterior mediastinum with intrapulmonary infiltration. Computed tomography revealed a narrowed lumen and thickened wall of the distal esophagus surrounded by a necrotic mass with infiltration into the nearby structures, suggesting a locally advanced malignant process. Positron emission tomography revealed intense accumulation of 18F-fluorodeoxyglucose, indicating an active inflammatory component, which complicates further differential diagnosis of mediastinal masses. Thoracoscopic biopsy and immunohistochemical analysis confirmed a fibroinflammatory process with perivascular lymphoid cell infiltration that was cluster of differentiation (CD)3 (++) and CD20 (++), with massive numbers of IgG4-immunoreactive plasma cells. Although a benign condition, sclerosing mediastinitis is a close mimicker of esophageal carcinoma, which cannot be differentiated by computed tomography or positron emission tomography and must be considered in a differential diagnosis.
Keywords: Idiopathic mediastinal fibrosis, computed tomography, positron emission tomography, IgG4-related disease, esophageal carcinoma, differential diagnosis, case report
Introduction
Fibrosing mediastinitis (FM) is a fibroinflammatory disease involving the mediastinum. FM is characterized by the expansive growth of fibroinflammatory tissue that surrounds and involves the major airways, vascular structures, or the esophagus. 1 The clinical presentation of the disorder depends on the involved structures, with the most common symptoms being dyspnea, hemoptysis, chest pain, superior (SVC) and inferior vena cava syndrome, dysphagia, and pulmonary hypertension.2,3 In the secondary form, FM occurs most often owing to previous histoplasmosis or tuberculosis infection. Other infections that can cause this condition are aspergillosis, cryptococcosis, uncommon mycobacterial infections, and malignant conditions, such as lymphoma and mesothelioma.1,4 The term idiopathic mediastinal fibrosis (IMF) was introduced by Flieder et al. as a condition without a clearly identified cause and pathogenesis. 5 However, according to numerous published case reports, there is a strong association between fibroinflammatory and autoimmune diseases. 4
Herein, we present a case of a male patient with progressive and active IMF that involved the mediodistal thoracic esophagus, with luminal obstruction misdiagnosed as locally advanced carcinoma on CT and PET.
Case report
The patient was a man in his late 60s who was admitted to our hospital owing to dysphagia. He had no fever. Blood analyses revealed mild leukocytosis (white blood cells (WBC): 13.9 × 109/L) with neutrophilia, normal eosinophil (0.07 × 109/L) and platelet (PLT) counts (323 × 109/L), and mildly elevated C-reactive protein (CRP) (18.3 mg/L) and D-dimer (3.62 mg/L; cut-off value: 0.5 mg/L fibrinogen equivalent units (FEU)) concentrations. Anemia of chronic disease was present as normocytic anemia (low hemoglobin: 131 g/L; normal mean corpuscular volume (MCV); low serum iron: 5.5 μmol/L (11.0–30 μmol/L), low total iron-binding capacity: 35.9 μmol/L (44.8–80.6 μmol/L), and elevated ferritin: 428.2 μmol/L (30–300 μmol/L). Values for other laboratory tests were unremarkable (glucose, urea, creatinine, sodium, potassium, calcium, phosphorus, protein, albumin, glutamic pyruvic transaminase, glutamic oxaloacetic transaminase, alkaline phosphatase, gamma-glutamyl transferase, lactate dehydrogenase). Immunological laboratory tests were normal (Sjogren's Syndrome antigens A (SSA) and B (SSB), myeloperoxidase (MPO)- or proteinase 3‐antineutrophil cytoplasmic antibodies (PR3-ANCA), double-strand (ds)DNA, ribonucleoprotein (RNP), Smith (Sm), cryoglobulins, anti-thyroperoxidase (TPO), anti-thyroglobulin (Tg)). Endoscopy revealed narrowing of the mediodistal thoracic esophagus. It was barely possible to pass the endoscope because of fibrin deposition distally. CT revealed an extensive, irregular, partly necrotic, infiltrative mass in the distal esophagus involving the paraesophageal fat tissue and mediastinal pleura (Figure 1a). These findings corresponded to a locally advanced esophageal carcinoma (Figure 1b). No other pathological findings or signs of retroperitoneal fibrosis were observed on abdominal and pelvic CT.
Figure 1.
Computed tomography of the chest, axial view, showing a solid, infiltrative mass in the mediodistal esophagus with luminal obstruction and spread to the pericardium, aortic wall, and mediastinal pleura (a). Coronal scan showing a narrowed esophageal lumen and a mass that resembled esophageal carcinoma (b).
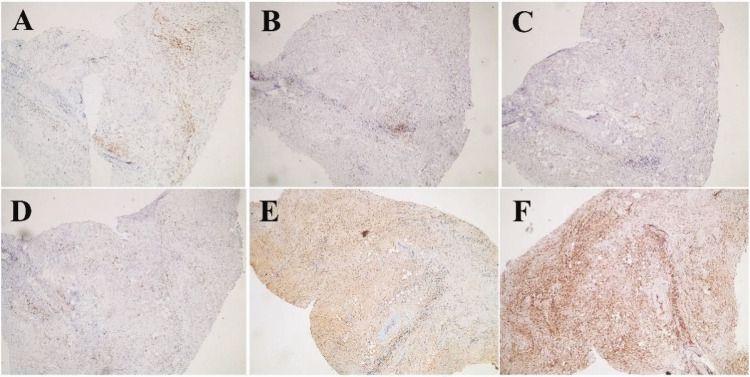
A biopsy was obtained during endoscopy but did not provide a definitive diagnosis. An attempted laparoscopic biopsy was also unsuccessful. Finally, thoracoscopic biopsy resulted in a sufficient quantity of tumor tissue. This irregular tumor mass was composed of sclerosing hyalinized fibrocollagenous bundles with a predominant lymphoplasmacytic infiltrate (Figure 2). Immunohistochemical analysis of the mass showed perivascular lymphoid cell infiltration that was CD3 (++) and CD20 (++), with a massive amount of IgG4-immunoreactive cells (Figure 3). These results were conclusive for sclerosing mediastinitis, and highly suggestive of IgG4-related disease despite the normal level of serum IgG4 (0.3 g/L). There were no signs of systemic vasculitis or connective tissue disease, and SSA, SSB, MPO- or PR3-ANCA, and dsDNA antibodies were absent. A percutaneous endoscopic gastrostomy tube was placed, and the patient was transferred to the Department of Immunology and Allergology. Corticosteroid therapy was administered with methylprednisolone at 1000 mg for 3 consecutive days. Prednisone was then prescribed at 40 mg once per day. After 2 weeks, the dose was decreased from 40 mg to 30 mg once per day; and 1 week later, the dose was decreased to 20 mg once per day. The therapeutic plan was to maintain a stable dose of 20 mg once per day for 4 weeks, thereafter. Unfortunately, the patient decided not to proceed with further treatment. Two months later, his clinical status worsened, and he complained of severe dysphagia and significant weight loss. Follow-up CT was performed, which revealed disease progression. A 3-mm defect in the esophageal wall was clearly observed, with communication between the lumen and a necrotic lesion (Figure 4a). The fibroinflammatory mass had increased in size and included the esophagus and the basal segments of the lungs with distal spread below the esophagogastric junction (Figure 4b). Moreover, inflammatory foci were seen in the right lower lung lobe (Figure 4c).
Figure 2.
Histopathological examination showing mainly fibroinflammatory changes with low cellularity, strong collagenases, and dense fibrohyaline areas within a polymorphic inflammatory infiltrate with a predominance of plasmacytes and eosinophils (×4, hematoxylin and eosin).
Figure 3.
Immunohistochemical findings showing oligocellular infiltrate with occasional strong perivascular infiltration and lymphoid cells with moderate immunoreactivity: CD3++ (a) and CD20++ (b). Some areas of infiltration are positive for fascin (c) and, rarely, for EMA (d). Abundant cells that are immunoreactive for IgG4 are visible (e, f) (×4). CD3, cluster of differentiation 3; CD20, cluster of differentiation 20; EMA, epithelial membrane antigen; IgG4, immunoglobulin G4.
Figure 4.
Follow-up CT 2 months after the initial diagnosis showing disease progression. Axial and coronal CT images show a large mediastinal, necrotic, mass that communicates with the esophageal lumen (the white arrow indicates a defect in the esophageal wall) and fistulization with peripheral airways of the right lower lung lobe (a, b). Pneumonic foci of lung parenchyma are also seen (c). CT, computed tomography.
Fluorodeoxyglucose positron emission tomography-CT (FDG PET-CT) was performed. Increased FDG uptake was noted in a large, partly necrotic, soft tissue mass in the lower mediastinum, suggesting the presence of active inflammation (Figure 5). Unfortunately, the patient refused any additional recommended treatment and decided not to proceed with follow-up in our hospital. Our hospital board and ethics committee approved this study. Written informed consent for publication was obtained from the patient, and all patient details are de-identified. This study conforms to the CARE guidelines. 6
Figure 5.
FDG PET-CT: PET, CT, combined PET-CT, and MIP images showing intense FDG uptake in a soft tissue mass abutting the lower esophagus (SUVmax: 20.6). Air bubbles and communication with the esophageal lumen are visible; infiltration into the upper abdomen is visible. FDG PET-CT, 18-fluorodeoxyglucose positron emission tomography-computed tomography; MIP, maximum-intensity projection; SUVmax, maximum standardized uptake value.
Discussion
FM is characterized by excessive fibrosis within the mediastinum 5 and affects mainly young patients. FM is represented by symptoms of obstruction of vital mediastinal structures. 5 On CT, two types of FM may be present: focal and diffuse. 5 The focal type is seen in 82% of patients and manifests as a localized, calcified mass, usually in the paratracheal and hilar regions. 5 This form is usually associated with Histoplasma capsulatum infection. 5 The diffuse type involves multiple mediastinal compartments and presents as a massive infiltrative lesion, rarely calcified. 5 This form occurs often with other idiopathic fibrosing disorders. 5 IMF can be associated with other fibroinflammatory diseases, such as idiopathic retroperitoneal fibrosis, or with ANCA-associated vasculitis or large-vessel arteritis as autoimmune and primary inflammatory conditions. In the isolated form, IMF is usually involved in the spectrum of IgG4-RD or non-IgG4-RD. 1
IMF has been described in patients with systemic vasculitis, Behcet’s disease, and granulomatosis, as well as idiopathic retroperitoneal fibrosis, sclerosing cholangitis, autoimmune pancreatitis, and other disorders within a spectrum of immunoglobulin G (IgG)4-related disease (IgG4-RD). 1 The diagnosis of FM can be very challenging. Using diagnostic imaging, it is very difficult to differentiate these lesions from mediastinal malignancies, such as lymphoma, and lung and esophageal carcinoma. 3 Computed tomography (CT) is the gold standard in the diagnostic evaluation of these patients. 3 Magnetic resonance imaging (MRI) and positron emission tomography (PET) can be used, with equally difficult differentiation from a malignant condition. 3 PET is important in assessing the activity of the inflammatory component as well as in monitoring the response to therapy. 3 Biopsy and histological study are mandatory in establishing a precise diagnosis. 7 There is no exact therapeutic protocol, and steroid therapy provides mainly symptomatic relief, often without a significant reduction in the size of the lesion. 1 Surgery and interventional radiology are reserved for patients with life-threatening symptoms. 1
Using cross-sectional imaging, it is very difficult to differentiate FM from other mediastinal masses, especially malignancies. 8 Differential diagnoses comprise other infiltrative mediastinal lesions, such as lung cancer, metastases, lymphoproliferative diseases, and desmoid tumors.1,8 These lesions appear similarly to FM on CT, with features of soft tissue masses that obliterate the fat plane and infiltrate the surrounding structures. 8 It is very rare that FM primarily involves the esophagus, with thickening and destruction of the wall, as in our patient. FDG PET-CT is widely used in oncology and indicates increased glucose metabolism and high expression of glucose transporters (GLUT) on the membranes of malignant cells. 9 FDG PET-CT is also gaining popularity in the evaluation of inflammatory and infectious diseases, as high glucose metabolism and FDG accumulation are observed in active inflammatory tissue. 9 Activated inflammatory cells exhibit increased glycolytic activity, as well as upregulation of GLUT, which may be caused by high concentrations of inflammatory mediators. 10 FDG PET-CT is very useful for monitoring the effect of therapy, and in some cases, unlike in ours, FDG uptake decreases after steroid therapy. 10 In our case, increased FDG uptake was noted in the large mediastinal soft tissue mass, suggesting the presence of activated inflammatory cells and active inflammation due to progressive FM.
For accurate diagnosis of FM, biopsy with histopathological analysis is mandatory, and a thoracoscopic procedure is often necessary to ensure an adequate sample, as in our case. The histopathological findings correspond to fibrous tissue with signs of chronic inflammation. 11 Immunohistochemical evidence of the prevalence of IgG4-positive plasma cell infiltration points to a possible diagnosis of IgG4-RD. 11 This immune-mediated fibroinflammatory disorder is characterized by the formation of tumefactive lesions, characteristic histopathological findings, and often, elevated serum IgG4 levels. 12 The clinical presentation is very heterogeneous. There are four distinct phenotypes described within the large spectrum of IgG4-RD: pancreatohepatobilliary disease, retroperitoneal fibrosis with or without aortitis, head and neck-limited disease, and Mikulicz’s syndrome with systemic involvement. 12 Owing to wide variable clinical, laboratory, and imaging features, it is very difficult to make a diagnosis of IgG4-RD. Therefore, the international guidelines for diagnosis and treatment are valuable. In 2020, Umehara at al. published the Revised Comprehensive Diagnostic (RCD) Criteria for IgG-4 RD. 13 In the guidelines, there are three criteria for establishing a diagnosis of this disease: (1) organ involvement, such as diffuse/localized swelling; (2) serum IgG4 level >135 mg/dL; and (3) positive for two of three pathological subitems: dense lymphocytic plasma cell infiltration with fibrosis; ratio of IgG4-positive plasma cells/IgG-positive cells >40% and number of IgG4-positive plasma cells >10 per high-power field; and typical tissue fibrosis, particularly storiform fibrosis, or obliterative phlebitis. When all three criteria are present, the diagnosis of IgG4-RD is established. When criteria (1) and (3) are met, IgG4-RD is considered probable, as in our case, while criteria (1) and (2) indicate the possibility of IgG4-RD. In 2019, the American College of Rheumatology/European League Against Rheumatism classification criteria were developed.11,14 To enter the process of classification, involvement of at least one of 10 possible organs in a manner consistent with IgG4-RD is needed, and exclusion criteria are then applied. If none of the exclusion criteria are met, the inclusion criteria are evaluated, and a score of ≥20 points classifies a patient as having IgG4-RD. Our patient met the entry criteria because there was pathological evidence of an inflammatory process accompanied by a lymphocytic infiltrate. Additionally, all of the exclusion criteria were met (no fever, no leukopenia, thrombocytopenia, peripheral eosinophilia, and no autoantibodies (SSA, SSB, MPO- or PR3-ANCA, dsDNA, RNP, Sm, cryoglobulins, anti-TPO, and anti-Tg). However, the score for our patient did not reach 20 points for the inclusion criteria. It is important to emphasize that the criteria are not diagnostic and were developed for clinical trials and research, not clinical settings.
Considering that there is no established biomarker, histopathology remains the most important diagnostic procedure for IgG4-RD. 15 Our patient was diagnosed with FM, with immunohistochemical findings of possible IgG4-RD. FM has been described in case reports as a part of IgG4-RD. Two cases of IgG4-related FM have been published by Japanese authors, who also reviewed 15 published cases. 16 The authors reported the following involved areas, clinically: retroperitoneal fibrosis (47%), lymph nodes (13.3%), pancreas (6.6%) and dacryosialadenitis (6.6%). IgG4-related FM was found as a distinct fibrotic subtype of IgG4-RD in a prospective study of 20 patients with IgG4-related FM who were compared with IgG4-RD patients without FM. 17 A case series of IMF patients by Rossi et al. showed that four of nine patients had proven IgG4-RD. 1 Among these patients, three had additional extramediastinal foci of fibrosis in the retroperitoneal and cervical regions, and within the seminal vesicles. One patient had a solitary prevertebral lesion in the posterior mediastinum, without esophageal involvement. 1 Schowengerdt et al. published a review and analysis of 180 cases of granulomatous and chronic FM and reported only two cases with esophageal obstruction due to FM. 18 The SVC is the structure most often affected by this process, which results in symptoms of SVC syndrome.6,18
FM can have an uncertain course, with both spontaneous remission or exacerbation of symptoms, and the disease course is usually slowly progressive. 19 There is no proven successful medical treatment for idiopathic FM. Corticosteroids, rituximab, or tamoxifen have been effective in some cases. 19 There are three available treatment options: systemic drug therapy comprising antifungal or corticosteroid drugs, surgical resection, and local therapy for complications. 20 Glucocorticoid and/or immunosuppressant therapy in some cases may lead to a decrease in mass size and resolution of inflammatory syndrome. 1 Surgery and interventional radiology are reserved for patients with life-threatening symptoms. 1 In our case, after steroid therapy, which was suspended in accordance with the patient’s wishes, an increase in the lesion size as well as persistent metabolic activity were clearly observed on follow-up imaging.
Conclusion
IMF can have an aggressive and progressive course despite being a benign condition. Contrast-enhanced CT and FDG PET-CT can effectively reveal the extent of fibrosis, presence of complications, which comprise mainly SVC syndrome and airway or rarely, esophageal constriction, as well as the effect of medical treatment. For a definitive diagnosis, a biopsy sample of sufficient quantity, with immunohistochemical analysis, is necessary because FM is an exceptional mimicker of malignancy on diagnostic imaging.
Supplemental Material
Supplemental material, sj-pdf-1-imr-10.1177_03000605231213212 for CT and 18FDG-PET/CT findings in progressive mediastinal idiopathic fibrosis as a benign mimicker of esophageal carcinoma: a case report by Milica Mitrovic-Jovanovic, Ognjan Skrobic, Strahinja Odalovic, Aleksandra Djikic Rom, Aleksandra Plavsic, Aleksandra Jankovic, Milena Kostadinovic, Nenad Ivanovic, Aleksandar Simic, Aleksandra Djuric-Stefanovic and Jelena Djokic Kovac in Journal of International Medical Research
Footnotes
Author contributions: Conceptualization: M.M and O.S.; Methodology, A.J, N.I., and S.O.; Resources: A.D.R; Writing—original draft preparation: M.M., J.D.K., and A.P; Writing—review and editing, M.M, M.K, and J.D.K.; Supervision, A.S., and A.D.S.
The authors declare that there is no conflict of interest.
Funding: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
ORCID iD
Milica Mitrovic-Jovanovic https://orcid.org/0000-0002-7525-7615
References
- 1.Rossi GM, Emmi G, Corradi D, et al. Idiopathic mediastinal fibrosis: a systemic immune-mediated disorder. A case series and a review of the literature. Clin Rev Allergy Immunol 2017; 52: 446–459. [DOI] [PubMed] [Google Scholar]
- 2.Hanley PC, Shub C, Lie JT. Constrictive pericarditis associated with combined idiopathic retroperitoneal and mediastinal fibrosis. Mayo Clin Proc 1984; 59: 300–304. [DOI] [PubMed] [Google Scholar]
- 3.Wu Z, Jarvis H, Howard LS, et al. Post-tuberculous fibrosing mediastinitis: a review of the literature. BMJ Open Respir Res 2017; 4: E000174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dechambre S, Dorzee J, Fastrez J, et al. Bronchial stenosis and sclerosing mediastinitis: an uncommon complication of external thoracic radiotherapy. Eur Respir J 1998; 11: 1188–1190. [DOI] [PubMed] [Google Scholar]
- 5.Miyata T, Takahama M, Yamamoto R, et al. Sclerosing mediastinitis mimicking anterior mediastinal tumor. Ann Thorac Surg 2009; 88: 293–295. [DOI] [PubMed] [Google Scholar]
- 6.Gagnier JJ, Kienle G, Altman DG, et al. The CARE Guidelines: Consensus-based clinical case reporting guideline development. Glob Adv Health Med 2013; 2: 38–43. DOI: 10.7453/gahmj.2013.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Novella Sanchez L, Sanz Herrero F, Berraondo Fraile J, et al. Mediastinal fibrosis and superior vena cava syndrome. Arch Bronconeumol 2013; 49: 340–342. [DOI] [PubMed] [Google Scholar]
- 8.Devaraj A, Griffin N, Nicholson AG, et al. Computed tomography findings in fibrosing mediastinitis. Clin Radiol 2007; 62: 781–786. [DOI] [PubMed] [Google Scholar]
- 9.Casali M, Lauri C, Altini C, et al. State of the art of 18F-FDG PET/CT application in inflammation and infection: a guide for image acquisition and interpretation. Clin Transl Imaging 2021; 9: 299–339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pijl JP, Nienhuis PH, Kwee TC, et al. Limitations and pitfalls of FDG-PET/CT in infection and inflammation. Semin Nucl Med 2021; 51: 633–645. [DOI] [PubMed] [Google Scholar]
- 11.Rossi GM, Rocco R, Accorsi Buttini E, et al. Idiopathic retroperitoneal fibrosis and its overlap with IgG4-related disease. Intern Emerg Med 2017; 12: 287–299. [DOI] [PubMed] [Google Scholar]
- 12.Wallace ZS, Zhang Y, Perugino CA, et al.; ACR/EULAR IgG4-RD Classification Criteria Committee. Clinical phenotypes of IgG4-related disease: an analysis of two international cross-sectional cohorts. Ann Rheum Dis 2019; 78: 406–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Umehara H, Okazaki K, Kawa S, et al.; Research program for intractable disease by the Ministry of Health, Labor and Welfare (MHLW) Japan. The 2020 revised comprehensive diagnostic (RCD) criteria for IgG4-RD. Mod Rheumatol 2021; 31: 529–533. [DOI] [PubMed] [Google Scholar]
- 14.Wallace ZS, Naden RP, Chari S, et al.; Members of the ACR/EULAR IgG4-R Classification Criteria Working Group. The 2019 American College of Rheumatology/European League Against Rheumatism classification criteria for IgG4-related disease. Ann Rheum Dis 2020; 79: 77–87. [DOI] [PubMed] [Google Scholar]
- 15.Lanzillotta M, Mancuso G, Della-Torre E. Advances in the diagnosis and management of IgG4 related disease. BMJ 2020; 16: 369. [DOI] [PubMed] [Google Scholar]
- 16.Takanashi S, Akiyama M, Suzuki K, et al. IgG4-related fibrosing mediastinitis diagnosed with computed tomography-guided percutaneous needle biopsy: two case reports and a review of the literature. Medicine (Baltimore) 2018; 97: E10935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang P, Han X, Li J, et al. IgG4- related fibrosing mediastinitis: clinical presentation, treatment efficacy and comparison with IgG4-RD without fibrosing mediastinitis. Clin Exp Rheumatol 2020; 6: 1206–1214. [PubMed] [Google Scholar]
- 18.Schowengerdt CG, Suyemoto R, Main FB. Granulomatous and fibrous mediastinitis. A review and analysis of 180 cases. J Thorac Cardiovasc Surg 1969; 57: 365–379. [PubMed] [Google Scholar]
- 19.Kang H, Jung MJ. Aggressive and progressive fibrosing mediastinitis involving the thoracic spine mimicking malignancy: a case report. Radiol Case Rep 2019; 14: 490–494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gorospe L, Ayala-Carbonero AM, Fernandez-Mendez MA, et al. Idiopathic fibrosing mediastinitis: spectrum of imaging findings with emphasis on its association with IgG4-related disease. Clin Imaging 2015; 39: 993–999. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-pdf-1-imr-10.1177_03000605231213212 for CT and 18FDG-PET/CT findings in progressive mediastinal idiopathic fibrosis as a benign mimicker of esophageal carcinoma: a case report by Milica Mitrovic-Jovanovic, Ognjan Skrobic, Strahinja Odalovic, Aleksandra Djikic Rom, Aleksandra Plavsic, Aleksandra Jankovic, Milena Kostadinovic, Nenad Ivanovic, Aleksandar Simic, Aleksandra Djuric-Stefanovic and Jelena Djokic Kovac in Journal of International Medical Research