Abstract
Objectives
This study aims to assess current cardiovascular disease risk and prevalence of risk factors in patients with rheumatoid arthritis (RA), psoriatic arthritis (PsA) and axial spondyloarthritis (SpA).
Methods
2050 consecutive patients with inflammatory arthritis (IA) and 939 controls were included, with 1308 patients with RA, 356 patients with PsA and 386 patients with SpA. In a prospective cohort setting, questionnaires regarding previous cardiovascular events and risk factors were used to assess cardiovascular risk and prevalence in patients with IA by calculating ORs using logistic regression models.
Results
‘Traditional’ cardiovascular (CV) risk factors were significantly elevated in patients with IA compared with controls. Cardiovascular disease (CVD) ORs were increased in patients with RA and PsA compared with controls, 1.61 (95% CI: 1.04 to 2.48) and 2.12 (95% CI: 1.23 to 3.66), respectively, and a trend towards increased odds was observed in patients with SpA (OR 1.43; 95% CI: 0.79 to 2.59). After adjusting for traditional risk factors, CV risk was not increased in patients with RA (OR; 0.95, 95% CI: 0.58 to 1.55), PsA (OR 1.19; 95% CI: 0.64 to 2.22) and SpA (OR; 0.91, 95% CI: 0.47 to 1.77).
Conclusion
CVD is currently still more prevalent in patients with IA compared with healthy controls and, more importantly, this elevated risk is highly influenced by an increased prevalence of ‘traditional’ CV risk factors. More attention to, as well as improvements in, identification and treatment of ‘traditional’ risk factors, need to be made for not only RA, but other IA conditions as well.
Keywords: rheumatoid arthritis; arthritis, psoriatic; spondylitis, ankylosing; cardiovascular diseases
WHAT IS ALREADY KNOWN ON THIS TOPIC
The increased cardiovascular disease risk in patients with inflammatory arthritis has diminished in recent years.
WHAT THIS STUDY ADDS
We found that the cardiovascular risk in patients with inflammatory arthritis is still increased and is highly influenced by ‘traditional’ risk factors, less so by inflammation.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
All patients with inflammatory arthritis need better, structural screening for and treatment of ‘traditional’ cardiovascular risk factors, to attenuate their increased risk.
Introduction
The worldwide prevalence of inflammatory arthritis (IA) is approximately 3%, with rheumatoid arthritis (RA), psoriatic arthritis (PsA), axial spondyloarthritis (SpA) being the most common subtypes of IA. IA is associated with an elevated risk of cardiovascular burden.1–3 It has been well established that patients with RA have a higher risk of developing cardiovascular diseases (CVD) than the general population.4 Also, patients with RA have an increased overall mortality, primarily due to cardiovascular (CV) events.5 While the CVD risk of other inflammatory rheumatic diseases such as PsA and SpA has been disputed, and data is limited, recent research shows an increased risk compared with healthy controls, with ORs up to 1.43 and 1.97, respectively,6 7 while other studies found CV morbidity similar to that in patients with RA.8–12 Furthermore, a large population-based cohort recently found higher HRs for patients with ankylosing spondylitis compared with RA (1.97 vs 1.83).6
However, most guidelines do not yet recommend the same CV screening strategies for RA, PsA and SpA, despite the results of these above-mentioned studies. While the European Alliance of Associations for Rheumatology advises CV risk assessment of all patients with IA every 5 years, it advises to apply a multiplication factor of 1.5 to calculated risk scores only in patients with RA.13 The European Society of Cardiology states that a multiplication factor in patients with RA should be considered depending on disease severity.14
The increased CVD risk in patients with IA is partly due to systemic inflammation as well as an increased prevalence of ‘traditional’ risk factors, such as hypertension and dyslipidaemia.15 Inflammation can also affect the development of these risk factors.16 In the past two decades, more efficacious anti-inflammatory treatment options have become available, that is, biological and targeted synthetic disease modifying anti-rheumatic drugs (b-/tsDMARDs). Treatment with these drugs has resulted in a decreased, but still elevated, CVD risk in these patients by lowering systemic inflammation.17–19 bDMARD therapy has also been shown to reduce risk of recurrent CVD in patients without IA, but with a history CVD.20
The aim of the current study was to evaluate whether the risk of developing CVD in patients with IA remains elevated compared with the general population, despite advances in anti-inflammatory therapy, in a real-world setting.
Methods
Study participants
Data were derived from a prospective cohort study (Netherlands Trial Register, trial ID NL8513), that was primarily designed to compare the disease severity of COVID-19 between patients with rheumatic diseases and healthy controls.21 Between April 2020 and March 2021, all adult patients with rheumatic immune-mediated inflammatory diseases (IMID) from the Amsterdam Rheumatology and Immunology Center, were asked to participate in this study. Participants were asked, but not obliged, to register an age-matched and sex-matched healthy control without a rheumatic IMID. In the current study we focused on patients with RA, PsA and SpA.
Assessments
Demographic and clinical data were collected using a series of digital questionnaires, which were primarily focused on COVID-19. Data on patient characteristics were collected at baseline and included age, sex, height, weight, smoking status, educational level, ethnicity, type of IMID and presence of comorbidities. In all questionnaires participants were asked to report disease activity, medication use and COVID-19-related characteristics and symptoms. Disease activity was measured using patient-reported outcome measures (PROMs), including the Routine Assessment of Patient Index Data-3 and two Visual Analogue Scales, for disease activity and pain, all scored 1–10. For the present study, we included additional questions which focused on CV risk factors and CVD in the questionnaire that was distributed in January 2021. The prevalence of CV risk factors was defined as either self-reported presence of and/or medication use against a risk factor. Self-reported CVD were assessed as coronary heart disease, peripheral arterial disease or cerebral arterial disease, according to the International Statistical Classification of Diseases and related health problems ninth revision (codes, 410.0–410.9, 435.9, 436, 443.9 and 798).
Statistical analysis
Baseline characteristics were presented as mean±SD, median (inner quartiles) or frequencies (percentages), depending on the distribution of data. ORs of CVD in patients with IA compared with controls were calculated using logistic regression techniques. Analyses were adjusted for sex and age in model 1, and additionally for traditional risk factors of CVD, including body mass index (BMI), hypertension (self-reported or usage of antihypertensive medication), dyslipidaemia (self-reported or usage of lipid-lowering medication), diabetes mellitus (DM; self-reported or usage of antidiabetics), and smoking in model 2, and only these traditional risk factors in model 3. Full association models are shown in online supplementary table S1. All statistical analyses were performed with IBM SPSS Statistics for Windows, V.23 (IBM, Armonk, New York, USA).
rmdopen-2023-003485supp001.pdf (50.2KB, pdf)
Results
Baseline characteristics
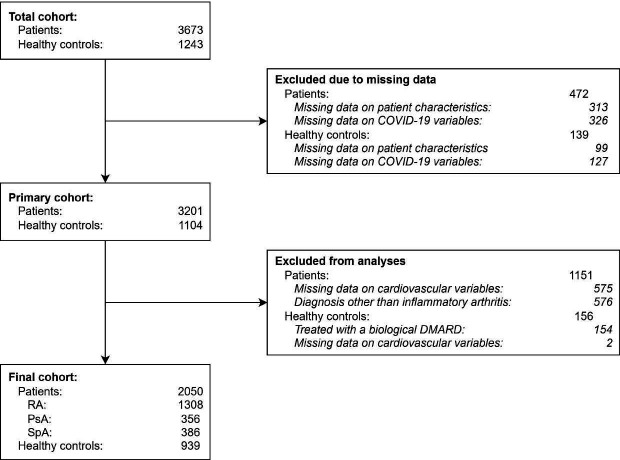
In total, 3201 patients with rheumatic IMIDs and 1104 healthy controls received the questionnaires. After exclusion of patients without IA and participants who had not filled out the questionnaire, data was analysed from 2050 patients with IA (1308 patients with RA, 356 patients with PsA and 386 patients with SpA), and 939 healthy controls (figure 1). The mean age of patients with IA was 57±13 years compared with 55±13 years for controls. Sixty-three per cent of patients and 69% of controls were women. Median disease duration was slightly longer in patients with SpA compared with RA and PsA (15 vs 13 years), and median disease activity was slightly higher in SpA compared with RA and PsA (scored 0.5 higher in all three PROMs). Patients with SpA more often used non-steroidal anti-inflammatory drugs and used fewer conventional synthetic DMARDs (csDMARDs) than patients with RA and PsA. 39% of patients with RA used bDMARDs, compared with 47% of patients with PsA and 57% of patients with SpA. Baseline characteristics and details on current anti-rheumatic treatment are shown in table 1.
Figure 1.
Flow diagram of study design. DMARD, disease-modifying anti-rheumatic drug; PsA, psoriatic arthritis, RA, rheumatoid arthritis, SpA, (axial) spondyloarthritis.
Table 1.
Baseline characteristics
| Patient characteristics | IA (n=2050) | RA (n=1308) | PsA (n=356) | SpA (n=386) | Controls (n=939) |
| Age, years | 57±13 | 60±12 | 57±12 | 51±13 | 55±13 |
| Women | 1266 (63) | 920 (70) | 167 (47) | 179 (46) | 636 (69) |
| Disease characteristics | |||||
| Disease duration, years | 13 (6–22) | 12 (6–21) | 13 (6–21) | 15 (7–27) | n/a |
| PROMs (0–10) | |||||
| RAPID-3 | 3.0 (1.5–5.5) | 3.0 (1.5–5.5) | 3.0 (1.5–5.5) | 3.5 (2.0–5.3) | n/a |
| VAS disease activity | 3.0 (2.0–4.0) | 3.0 (2.0–4.0) | 3.0 (2.0–4.0) | 3.5 (2.0–5.0) | n/a |
| VAS pain | 3.5 (1.5–6.0) | 3.5 (1.0–6.0) | 3.5 (1.0–6.0) | 4.0 (2.0–6.0) | n/a |
| NSAIDs | 450 (22) | 213 (16) | 60 (17) | 177 (46) | 18 (2) |
| Prednisone | 202 (10) | 174 (13) | 18 (5) | 10 (3) | n/a |
| DMARD | 1588 (78) | 1073 (82) | 276 (78) | 239 (62) | n/a |
| csDMARD | 1118 (55) | 900 (69) | 186 (52) | 32 (8) | n/a |
| Methotrexate | 906 (44) | 733 (56) | 164 (46) | 9 (2) | n/a |
| Hydroxychloroquine | 225 (11) | 218 (17) | 7 (2) | 0 (0) | n/a |
| Leflunomide | 63 (3) | 51 (4) | 9 (3) | 3 (1) | n/a |
| Sulfasalazine | 120 (6) | 90 (7) | 11 (3) | 19 (5) | n/a |
| Other | 16 (1) | 9 (1) | 5 (1) | 2 (1) | n/a |
| bDMARD | 895 (44) | 508 (39) | 169 (47) | 218 (57) | n/a |
| TNF-inhibitor | 727 (36) | 383 (29) | 147 (42) | 197 (51) | n/a |
| IL-6 inhibitor | 32 (2) | 32 (2) | 0 (0) | 0 (0) | n/a |
| IL-12/23 inhibitor | 6 (0) | 0 (0) | 6 (1) | 0 (0) | n/a |
| IL-17 inhibitor | 37 (2) | 0 (0) | 15 (4) | 22 (6) | n/a |
| Rituximab | 48 (2) | 47 (4) | 0 (0) | 0 (0) | n/a |
| Abatacept | 48 (2) | 46 (4) | 2 (1) | 0 (0) | n/a |
| tsDMARD | 20 (1) | 11 (1) | 5 (1) | 4 (1) | n/a |
| JAK-inhibitor | 17 (1) | 11 (1) | 2 (1) | 4 (1) | n/a |
| Apremilast | 3 (0) | 0 (0) | 3 (1) | 0 (0) | n/a |
Data are reported as mean±SD, median (inner quartiles) or frequency (percentage) where appropriate.
bDMARD, biological DMARD; csDMARD, conventional synthetic DMARD; DMARD, disease-modifying anti-rheumatic drug; IA, inflammatory arthritis; IL, interleukin; JAK, janus kinase; NSAIDs, non-steroidal anti-inflammatory drugs; PROM, patient-reported outcome measure; PsA, psoriatic arthritis; RA, rheumatoid arthritis; RAPID, Routine Assessment of Patient Index Data; SpA, (axial) spondyloarthritis; TNF, tumour necrosis factor; tsDMARD, targeted synthetic DMARD; VAS, Visual Analogue Scale.
Cardiovascular risk factors
Median BMI was significantly higher in the IA group (25.3) compared with the healthy control (24.4), with the highest BMI found in patients with PsA (26.3). Patients with IA exercised less (420 min/week for RA/PsA, 390 min/week for SpA) compared with controls who exercised 490 min/week. Thirty-four per cent of patients with RA had never smoked, compared with 38% of patients with PsA, 36% of patients with SpA and 44% of controls. A higher prevalence of hypertension (38% vs 25%), dyslipidaemia (31% vs 22%) and diabetes (5% vs 1%) was reported in patients with IA as compared with controls. Overall patients also used more antihypertensive (20% vs 14%), lipid-lowering medication (16% vs 10%), antidiabetic (2% vs 1%) and anticoagulant medication than controls (14% and 10%, respectively). An overview of CV risk factors is shown in table 2.
Table 2.
Traditional risk factors in patients with inflammatory arthritis compared with healthy controls
| CV risk factors | IA (n=2050) | RA (n=1308) | PsA (n=356) | SpA (n=386) | Controls (n=939) |
| BMI | 25.3 (22.8–28.4) | 25.3 (22.6–28.7) | 26.3 (23.5–29.4) | 24.8 (23.0–27.7) | 24.4 (22.1–17.7) |
| Smoking status | |||||
| Current, >1×/week | 197 (10) | 136 (10) | 21 (6) | 40 (10) | 49 (5) |
| Current, <1×/week | 58 (3) | 31 (2) | 8 (2) | 19 (5) | 22 (2) |
| Quit | 925 (45) | 599 (46) | 166 (47) | 160 (42) | 379 (40) |
| Never | 716 (35) | 442 (34) | 136 (38) | 138 (36) | 411 (44) |
| Alcohol | |||||
| Yes | 1165 (57) | 719 (55) | 215 (60) | 231 (60) | 669 (71) |
| Incidental | 442 (22) | 288 (22) | 66 (19) | 88 (23) | 170 (18) |
| Never | 386 (19) | 263 (20) | 62 (17) | 61 (16) | 82 (9) |
| Exercise (min/week) | 420 (211–720) | 420 (181–740) | 420 (220–660) | 390 (210–653) | 490 (220–660) |
| <30 min/day | 534 (26) | 359 (27) | 83 (23) | 92 (24) | 160 (17) |
| 30–60 min/day | 535 (26) | 314 (24) | 103 (29) | 118 (31) | 247 (26) |
| >60 min/day | 981 (48) | 635 (49) | 170 (48) | 176 (46) | 532 (57) |
| Hypertension | 770 (38) | 496 (38) | 140 (39) | 134 (35) | 231 (25) |
| Antihypertensives | 411 (20) | 268 (21) | 80 (23) | 63 (16) | 131 (14) |
| Dyslipidaemia | 641 (31) | 422 (32) | 110 (31) | 109 (28) | 207 (22) |
| Lipid-lowering medication | 335 (16) | 222 (17) | 59 (17) | 54 (14) | 98 (10) |
| Diabetes mellitus | 109 (5) | 67 (5) | 26 (7) | 16 (4) | 13 (1) |
| Antidiabetics | 46 (2) | 28 (2) | 10 (3) | 8 (2) | 6 (1) |
| Anticoagulants | 246 (12) | 179 (14) | 34 (10) | 33 (9) | 74 (8) |
Data are reported as mean±SD, median (inner quartiles) or frequency (percentage) where appropriate.
BMI, body mass index; CV, cardiovascular ; IA, inflammatory arthritis; PsA, psoriatic arthritis; RA, rheumatoid arthritis; SpA, (axial) spondyloarthritis.
Cardiovascular disease
The number of patients with a history of CVD was higher among patients with IA (n=110; 5%) compared with healthy controls (n=31; 3%). CVD events were mainly cardiac in all groups, accounting for 69% of all events in patients with IA and 76% in controls. The odds of having CVD were 1.66 times higher in patients with IA (OR: 1.66, 95% CI: 1.11 to 2.49) compared with healthy controls. Similar findings were found for the prevalence of RA, PsA and SpA, respectively, 5%, 7% and 4%. We found that unadjusted ORs were significantly increased in RA (OR: 1.61, 95% CI: 1.04 to 2.48) and PsA (OR: 2.12, 95% CI: 1.23 to 3.66) compared with controls, while the OR in SpA did not reach statistical significance (OR: 1.43, 95% CI: 0.79 to 2.59). Adjustment for age and sex in model 1 decreased the CV risk in patients with RA and PsA, but not in patients with SpA (OR 1.49; 95% CI: 0.80 to 2.76). When additionally adjusting for ‘traditional’ CV risk factors (age, sex, BMI, smoking, hypertension, DM and dyslipidaemia) in model 2, the CV risk decreased in all patient groups, with only the OR in PsA remaining slightly, although not significantly, increased (OR 1.19; 95% CI: 0.64 to 2.22). Adjusting solely for these ‘traditional’ risk factors in model 3 yielded similar results, with slightly higher ORs in all patient groups. Results from logistic regression analyses are shown in table 3.
Table 3.
ORs of cardiovascular diseases in patients with inflammatory arthritis compared with healthy controls
| OR (crude) | P value | OR (model 1) | P value | OR (model 2) | P value | OR (model 3) | P value | |
| IA | 1.66 (1.11 to 2.49) | 0.01 | 1.38 (0.91 to 2.09) | 0.13 | 0.99 (0.63 to 1.56) | 0.96 | 1.05 (0.67 to1.65) | 0.82 |
| RA | 1.61 (1.04 to 2.48) | 0.03 | 1.27 (0.83 to 2.01) | 0.29 | 0.95 (0.58 to 1.55) | 0.85 | 0.97 (0.60 to 1.57) | 0.91 |
| PsA | 2.12 (1.23 to 3.66) | 0.01 | 1.68 (0.96 to 2.96) | 0.07 | 1.19 (0.64 to 2.22) | 0.57 | 1.37 (0.75 to 2.52) | 0.31 |
| SpA | 1.43 (0.79 to 2.59) | 0.23 | 1.49 (0.80 to 2.76) | 0.29 | 0.91 (0.47 to 1.77) | 0.78 | 1.05 (0.56 to 2.00) | 0.87 |
ORs are shown with 95% CIs. A p value of <0.05 was considered statistically significant. Model 1 was adjusted for age and sex, model 2 was adjusted for age, sex, BMI, smoking, diabetes mellitus, hypertension and dyslipidaemia, model 3 was adjusted for BMI, smoking diabetes mellitus, hypertension and dyslipidaemia.
BMI, body mass index; IA, inflammatory arthritis; PsA, psoriatic arthritis; RA, rheumatoid arthritis; SpA, (axial) spondyloarthritis.
Discussion
In line with previous findings, our results demonstrated that patients with IA more often had ‘traditional’ CV risk factors than age-matched and sex-matched controls.22 23 Patients with IA had a significantly higher prevalence of hypertension, dyslipidaemia and DM, had a greater BMI and exercised less compared with controls, and both patients with RA and SpA were more often current smokers. The systemic inflammation characteristic to all forms of IA seems to be partially responsible for the increased prevalence of hypertension, dyslipidaemia and DM.24 25 Inflammation causes oxidative stress and endothelial dysfunction with subsequent vascular resistance and finally hypertension.25 Inflammation also induces dyslipidaemia.26 27 Additionally, a higher prevalence of DM has been previously reported in all three forms of IA, with inflammation as a causal factor.28–30 Inflammation in the pancreas causes β-cell dysfunction and apoptosis, leading to insulin deficiency and eventually DM.28 The prevalence of these risk factors could be further increased by less physical activity of the patients with IA. Several studies have shown that sufficient physical activity can prevent CVD and has a favourable effect on all above-mentioned CV risk factors.31–33 Physical activity is therefore recommended as an integral part of standard care for patients with IA.34 Furthermore, smoking increases the chances of developing CVD, but also of IA.35 The link between smoking and RA has long been established, but there is also evidence that smoking increases the incidence of PsA and SpA.36 Both patients and controls in our cohort often had untreated CV risk factors, with nearly half of both groups not receiving preventive medical treatment. While patients in our cohort are otherwise rather healthy (few smokers, relatively low BMI), they have a large cumulative (IA) disease burden. Despite improved treatment of inflammation in recent years, this might have resulted in vascular damage, hypertension and eventually CVD. Other research confirms that screening and treatment of ‘traditional’ CV risk factors remains suboptimal in many patients with IA.37–39 To further decrease the prevalence of ‘traditional’ CV risk factors (and thus the incidence of CVD) we need to work towards timely control of inflammation, improve awareness of the CVD risk and provide structural CV risk screening and management in all patients with IA.
Moreover, we found that the prevalence of CVD was higher among patients with IA compared with controls. This enhanced risk is considered partially due to the chronic inflammation found in these patient groups, and due to the increased prevalence of ‘traditional’ risk factors. Adjusting our crude analyses for these ‘traditional’ risk factors highly attenuated the ORs for patients with IA versus healthy controls. These results suggest that the current CVD risk in patients with IA is nowadays highly influenced by the increased prevalence of these risk factors. This is in agreement with another recent study in patients with longstanding RA, where the contribution of these risk factors on the CVD risk increased over the years.40 The risk of CVD in patients with RA has similarly decreased, with the over twofold risk compared with the general population found more than 10 years ago lowering to a current increased risk of about 50–60%.22 40 A possible explanation could be that current treatment of systemic inflammation has improved, while screening and treatment of ‘traditional’ risk factors remains insufficient.37 41 Patients in our cohort used more bDMARDs than the overall population of patients with IA,42–44 whereby treatment with tumour necrosis factor inhibitor has been shown to decrease CVD events in RA, and possibly also PsA, patients.19 45 46 However, larger studies demonstrating the long-term effect of (b)DMARDs on CVD for all patients with IA are scarce.
Interestingly, in our cohort, the prevalence of CVD in patients with PsA was higher than in patients with RA, and the OR of patients with PsA compared with healthy controls was consistently the highest in all models. While there are few comparative studies, the CVD risk for patients with RA is generally found to be equal or higher than patients with PsA.10 13 A possible explanation for our findings might be that ‘traditional’ risk factors are not screened and treated as well in patients with PsA, due to lesser awareness. Thus far CVD comorbidity has been studied to a much greater extent in RA in comparison to PsA, and the guidelines regarding CVD risk in patients with RA are stricter than in patients with PsA.13 Furthermore, PsA is more often associated with metabolic syndrome and truncal obesity than RA, which contributes to a pro-inflammatory state and eventually arterial stiffness and CVD.47 48 Additionally, a number of pro-inflammatory cytokines are present in both PsA, metabolic syndrome and atherosclerosis, which helps explain the increased risk.49
Our study has several strengths. First, it covers a well-defined patient population with a large sample size and a diversity of patients in a prospective data collecting setting, with consistent data collection across groups. Second, patients were compared with age-matched and sex-matched controls to minimise bias. Finally, this study provides data regarding the CVD risk of the three most common inflammatory rheumatic diseases, of which there is limited data in previous studies.
A limitation of this study is that data regarding CVD were obtained by means of a self-completed questionnaire, and logistic constraints limited the collection of physical examination and laboratory data. Another limitation of this study is that data on CVD were cross-sectionally collected, which limits our ability to investigate causal pathways (ie, via mediation analyses). The relationship between IA, CV risk factors and CVD is complex, as chronic inflammation caused by IA conditions can contribute to CVD risk through a multitude of mediating factors.16 Adjusting for CV risk factors eliminates some effects of chronic inflammation on CVD risk, so it should be noted that our adjusted results likely underestimate the total effect on CVD risk. Interpreting these adjusted results should be done with the complex relationship between inflammation and atherosclerosis, and the paradoxical effect of inflammation on lipid levels in mind. However, previous studies clearly demonstrated that chronic inflammation independently contributes to the development of CVD’s, so we believe our adjusted results still provide valuable insights into the current magnitude of this association.50
In conclusion, CVD is currently still more prevalent in patients with IA compared with healthy controls and, more importantly, this elevated risk is highly related to an increased prevalence of ‘traditional’ CV risk factors. Current treatments better address systemic inflammation with combinations of csDMARDs and/or b-/tsDMARDs, reducing its detrimental effects on CVD in these patient groups. However, more attention should be paid to identifying and treating ‘traditional’ CVD risk factors, not only in RA, but also in other IA conditions.
Acknowledgments
We would like to thank Carlijn Wagenaar for her assistance in finalising the manuscript.
Footnotes
Contributors: Conceptualisation: RR, LB. Formal analysis: RR, LCP. Investigating: RR, RH, LCP. Methodology: RR. Supervision: GJW, AWRvK, MTN. Validation: RR. Writing—original draft: RR, RH, LCP. Writing—review and editing: RR, RH, LB, FH, GJW, AWRvK, MTN. Guarantor: MTN.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request. Anonymised data can be shared if an agreement has been made with the authors.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
This study involves human participants and was approved by VU University Medical Center, registration number 2020.169. Participants gave informed consent to participate in the study before taking part.
References
- 1.Avina-Zubieta JA, Thomas J, Sadatsafavi M, et al. Risk of incident cardiovascular events in patients with rheumatoid arthritis: a meta-analysis of observational studies. Ann Rheum Dis 2012;71:1524–9. 10.1136/annrheumdis-2011-200726 [DOI] [PubMed] [Google Scholar]
- 2.Eriksson JK, Jacobsson L, Bengtsson K, et al. Is ankylosing spondylitis a risk factor for cardiovascular disease, and how do these risks compare with those in rheumatoid arthritis Ann Rheum Dis 2017;76:364–70. 10.1136/annrheumdis-2016-209315 [DOI] [PubMed] [Google Scholar]
- 3.Ogdie A, Yu Y, Haynes K, et al. Risk of major cardiovascular events in patients with Psoriatic arthritis, psoriasis and rheumatoid arthritis: a population-based cohort study. Ann Rheum Dis 2015;74:326–32. 10.1136/annrheumdis-2014-205675 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Crowson CS, Liao KP, Davis JM, et al. Rheumatoid arthritis and cardiovascular disease. Am Heart J 2013;166:622–8. 10.1016/j.ahj.2013.07.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Provan SA, Lillegraven S, Sexton J, et al. Trends in all-cause and cardiovascular mortality in patients with incident rheumatoid arthritis: a 20-year follow-up matched case-cohort study. Rheumatology (Oxford) 2020;59:505–12. 10.1093/rheumatology/kez371 [DOI] [PubMed] [Google Scholar]
- 6.Conrad N, Verbeke G, Molenberghs G, et al. Autoimmune diseases and cardiovascular risk: a population-based study on 19 autoimmune diseases and 12 cardiovascular diseases in 22 million individuals in the UK. Lancet 2022;400:733–43. 10.1016/S0140-6736(22)01349-6 [DOI] [PubMed] [Google Scholar]
- 7.Polachek A, Touma Z, Anderson M, et al. Risk of cardiovascular morbidity in patients with psoriatic arthritis: a meta-analysis of observational studies. Arthritis Care Res (Hoboken) 2017;69:67–74. 10.1002/acr.22926 [DOI] [PubMed] [Google Scholar]
- 8.Mathieu S, Pereira B, Soubrier M. Cardiovascular events in ankylosing spondylitis: an updated meta-analysis. Semin Arthritis Rheum 2015;44:551–5. 10.1016/j.semarthrit.2014.10.007 [DOI] [PubMed] [Google Scholar]
- 9.Liew JW, Ramiro S, Gensler LS. Cardiovascular morbidity and mortality in ankylosing spondylitis and Psoriatic arthritis. Best Pract Res Clin Rheumatol 2018;32:369–89. 10.1016/j.berh.2019.01.002 [DOI] [PubMed] [Google Scholar]
- 10.Zhu TY, Li EK, Tam LS. Cardiovascular risk in patients with psoriatic arthritis. Int J Rheumatol 2012;2012:714321. 10.1155/2012/714321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jamnitski A, Visman IM, Peters MJL, et al. Prevalence of cardiovascular diseases in psoriatic arthritis resembles that of rheumatoid arthritis. Ann Rheum Dis 2011;70:875–6. 10.1136/ard.2010.136499 [DOI] [PubMed] [Google Scholar]
- 12.Lauper K, Courvoisier DS, Chevallier P, et al. Incidence and prevalence of major adverse cardiovascular events in rheumatoid arthritis, psoriatic arthritis, and axial spondyloarthritis. Arthritis Care Res (Hoboken) 2018;70:1756–63. 10.1002/acr.23567 [DOI] [PubMed] [Google Scholar]
- 13.Agca R, Heslinga SC, Rollefstad S, et al. EULAR recommendations for cardiovascular disease risk management in patients with rheumatoid arthritis and other forms of inflammatory joint disorders: 2015/2016 update. Ann Rheum Dis 2017;76:17–28. 10.1136/annrheumdis-2016-209775 [DOI] [PubMed] [Google Scholar]
- 14.Visseren FLJ, Mach F, Smulders YM, et al. 2021 ESC guidelines on cardiovascular disease prevention in clinical practice. Eur Heart J 2021;42:3227–337. 10.1093/eurheartj/ehab484 [DOI] [PubMed] [Google Scholar]
- 15.Crowson CS, Rollefstad S, Ikdahl E, et al. Impact of risk factors associated with cardiovascular outcomes in patients with rheumatoid arthritis. Ann Rheum Dis 2018;77:48–54. 10.1136/annrheumdis-2017-211735 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Myasoedova E, Chandran A, Ilhan B, et al. The role of rheumatoid arthritis (RA) flare and cumulative burden of RA severity in the risk of cardiovascular disease. Ann Rheum Dis 2016;75:560–5. 10.1136/annrheumdis-2014-206411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Myasoedova E, Davis JM, Roger VL, et al. Improved incidence of cardiovascular disease in patients with incident rheumatoid arthritis in the 2000S: a population-based cohort study. J Rheumatol 2021;48:1379–87. 10.3899/jrheum.200842 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Peters MJL, van Sijl AM, Voskuyl AE, et al. The effects of tumor necrosis factor inhibitors on cardiovascular risk in rheumatoid arthritis. Curr Pharm Des 2012;18:1502–11. 10.2174/138161212799504786 [DOI] [PubMed] [Google Scholar]
- 19.Roubille C, Richer V, Starnino T, et al. The effects of tumour necrosis factor inhibitors, methotrexate, non-steroidal anti-inflammatory drugs and corticosteroids on cardiovascular events in rheumatoid arthritis, psoriasis and psoriatic arthritis: a systematic review and meta-analysis. Ann Rheum Dis 2015;74:480–9. 10.1136/annrheumdis-2014-206624 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ridker PM, Everett BM, Thuren T, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med 2017;377:1119–31. 10.1056/NEJMoa1707914 [DOI] [PubMed] [Google Scholar]
- 21.Boekel L, Hooijberg F, Vogelzang EH, et al. Antibody development and disease severity of COVID-19 in non-immunised patients with rheumatic immune-mediated inflammatory diseases: data from a prospective cohort study. RMD Open 2022;8:e002035. 10.1136/rmdopen-2021-002035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Peters MJL, van Halm VP, Voskuyl AE, et al. Does rheumatoid arthritis equal diabetes mellitus as an independent risk factor for cardiovascular disease? A prospective study. Arthritis Rheum 2009;61:1571–9. 10.1002/art.24836 [DOI] [PubMed] [Google Scholar]
- 23.Ogdie A, Haynes K, Troxel AB, et al. Risk of mortality in patients with psoriatic arthritis, rheumatoid arthritis and psoriasis: a longitudinal cohort study. Ann Rheum Dis 2014;73:149–53. 10.1136/annrheumdis-2012-202424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liao KP, Solomon DH. Traditional cardiovascular risk factors, inflammation and cardiovascular risk in rheumatoid arthritis. Rheumatology (Oxford) 2013;52:45–52. 10.1093/rheumatology/kes243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Willerson JT, Ridker PM. Inflammation as a cardiovascular risk factor. Circulation 2004;109:II2–10. 10.1161/01.CIR.0000129535.04194.38 [DOI] [PubMed] [Google Scholar]
- 26.Feingold KR, Grunfeld C. The effect of inflammation and infection on lipids and lipoproteins. In: Feingold KR, Anawalt B, Boyce A, et al., eds. Endotext. South Dartmouth (MA), 2019. [Google Scholar]
- 27.Charles-Schoeman C, Gonzalez-Gay MA, Kaplan I, et al. Effects of tofacitinib and other DMARDs on lipid profiles in rheumatoid arthritis: implications for the rheumatologist. Semin Arthritis Rheum 2016;46:71–80. 10.1016/j.semarthrit.2016.03.004 [DOI] [PubMed] [Google Scholar]
- 28.Tian Z, Mclaughlin J, Verma A, et al. The relationship between rheumatoid arthritis and diabetes mellitus: a systematic review and meta-analysis. Cardiovasc Endocrinol Metab 2021;10:125–31. 10.1097/XCE.0000000000000244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhao SS, Robertson S, Reich T, et al. Prevalence and impact of comorbidities in axial Spondyloarthritis: systematic review and meta-analysis. Rheumatology (Oxford) 2020;59:iv47–57. 10.1093/rheumatology/keaa246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dal Bello G, Gisondi P, Idolazzi L, et al. Psoriatic arthritis and diabetes mellitus: a narrative review. Rheumatol Ther 2020;7:271–85. 10.1007/s40744-020-00206-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bensimhon DR, Kraus WE, Donahue MP. Obesity and physical activity: a review. Am Heart J 2006;151:598–603. 10.1016/j.ahj.2005.03.005 [DOI] [PubMed] [Google Scholar]
- 32.Kelemen MH, Effron MB, Valenti SA, et al. Exercise training combined with antihypertensive drug therapy. Effects on lipids, blood pressure, and left ventricular mass. JAMA 1990;263:2766–71. [PubMed] [Google Scholar]
- 33.Wallberg-Henriksson H, Rincon J, Zierath JR. Exercise in the management of non-insulin-dependent diabetes mellitus. Sports Med 1998;25:25–35. 10.2165/00007256-199825010-00003 [DOI] [PubMed] [Google Scholar]
- 34.Rausch Osthoff A-K, Niedermann K, Braun J, et al. 2018 EULAR recommendations for physical activity in people with inflammatory arthritis and osteoarthritis. Ann Rheum Dis 2018;77:1251–60. 10.1136/annrheumdis-2018-213585 [DOI] [PubMed] [Google Scholar]
- 35.Chang K, Yang SM, Kim SH, et al. Smoking and rheumatoid arthritis. Int J Mol Sci 2014;15:22279–95. 10.3390/ijms151222279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhao SS, Goodson NJ, Robertson S, et al. Smoking in spondyloarthritis: unravelling the complexities. Rheumatology (Oxford) 2020;59:1472–81. 10.1093/rheumatology/keaa093 [DOI] [PubMed] [Google Scholar]
- 37.Raadsen R, Hansildaar R, van Kuijk AWR, et al. Male rheumatoid arthritis patients at substantially higher risk for cardiovascular mortality in comparison to women. Semin Arthritis Rheum 2023;62:152233. 10.1016/j.semarthrit.2023.152233 [DOI] [PubMed] [Google Scholar]
- 38.Semb AG, Ikdahl E, Wibetoe G, et al. Atherosclerotic cardiovascular disease prevention in rheumatoid arthritis. Nat Rev Rheumatol 2020;16:361–79. 10.1038/s41584-020-0428-y [DOI] [PubMed] [Google Scholar]
- 39.Eder L, Harvey P, Chandran V, et al. Gaps in diagnosis and treatment of cardiovascular risk factors in patients with psoriatic disease: an International Multicenter Study. J Rheumatol 2018;45:378–84. 10.3899/jrheum.170379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Raadsen R, Agca R, Boers M, et al. In RA patients without prevalent CVD, incident CVD is mainly associated with traditional risk factors: A 20-year follow-up in the CARRE cohort study. Semin Arthritis Rheum 2023;58:152132. 10.1016/j.semarthrit.2022.152132 [DOI] [PubMed] [Google Scholar]
- 41.van den Oever IAM, Heslinga M, Griep EN, et al. Cardiovascular risk management in rheumatoid arthritis patients still suboptimal: the implementation of cardiovascular risk management in rheumatoid arthritis project. Rheumatology (Oxford) 2017;56:1472–8. 10.1093/rheumatology/kew497 [DOI] [PubMed] [Google Scholar]
- 42.Mease PJ, Stryker S, Liu M, et al. Treatment patterns in rheumatoid arthritis patients newly initiated on biologic and conventional synthetic disease-modifying antirheumatic drug therapy and enrolled in a North American Clinical Registry. Arthritis Res Ther 2021;23:236. 10.1186/s13075-021-02599-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lee MP, Lii J, Jin Y, et al. Patterns of systemic treatment for Psoriatic arthritis in the US: 2004-2015. Arthritis Care Res (Hoboken) 2018;70:791–6. 10.1002/acr.23337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Perrone V, Losi S, Filippi E, et al. Analysis of the prevalence of Ankylosing Spondylitis and treatment patterns and drug utilization among affected patients: an Italian real-world study. Expert Rev Pharmacoecon Outcomes Res 2022;22:327–33. 10.1080/14737167.2022.2032663 [DOI] [PubMed] [Google Scholar]
- 45.Nair S, Singh Kahlon S, Sikandar R, et al. Tumor necrosis factor-alpha inhibitors and cardiovascular risk in rheumatoid arthritis: a systematic review. Cureus 2022;14:e26430. 10.7759/cureus.26430 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nurmohamed M, Bao Y, Signorovitch J, et al. Longer durations of antitumour necrosis factor treatment are associated with reduced risk of cardiovascular events in patients with rheumatoid arthritis. RMD Open 2015;1:e000080. 10.1136/rmdopen-2015-000080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lin YC, Dalal D, Churton S, et al. Relationship between metabolic syndrome and carotid intima-media thickness: cross-sectional comparison between psoriasis and psoriatic arthritis. Arthritis Care Res (Hoboken) 2014;66:97–103. 10.1002/acr.22144 [DOI] [PubMed] [Google Scholar]
- 48.Loganathan A, Kamalaraj N, El-Haddad C, et al. Systematic review and meta-analysis on prevalence of metabolic syndrome in psoriatic arthritis, rheumatoid arthritis and psoriasis. Int J Rheum Dis 2021;24:1112–20. 10.1111/1756-185X.14147 [DOI] [PubMed] [Google Scholar]
- 49.Ferguson LD, Siebert S, McInnes IB, et al. Cardiometabolic Comorbidities in RA and PSA: lessons learned and future directions. Nat Rev Rheumatol 2019;15:461–74. 10.1038/s41584-019-0256-0 [DOI] [PubMed] [Google Scholar]
- 50.Alfaddagh A, Martin SS, Leucker TM, et al. Inflammation and cardiovascular disease: from mechanisms to therapeutics. Am J Prev Cardiol 2020;4:100130. 10.1016/j.ajpc.2020.100130 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
rmdopen-2023-003485supp001.pdf (50.2KB, pdf)
Data Availability Statement
Data are available upon reasonable request. Anonymised data can be shared if an agreement has been made with the authors.