Graphical abstract

Keywords: Pembrolizumab, Lenvatinib, MMR deficient, Endometrial cancer
Highlights
-
•
dMMR endometrial cancer patients who failed to respond to pembrolizumab alone were treated with the combination of pembrolizumab and lenvatinib.
-
•
These patients demonstrated a 75% response rate (CR 12.5%, PR 62.5%) to the combination of pembrolizumab and lenvatinib.
-
•
The combination was well-tolerated, allowing patients to receive multiple cycles of therapy and have durable responses.
Abstract
To evaluate the efficacy of the combination of pembrolizumab and lenvatinib in MMR deficient (dMMR) endometrial cancer (EC) patients who previously failed to respond to single-agent pembrolizumab. A retrospective review of MMR deficient endometrial cancer patients was performed. Patients who failed to respond to pembrolizumab as a single-agent and subsequently received a combination of pembrolizumab and lenvatinib were analyzed. RECIST 1.1 criteria was used to establish clinical response (complete response, partial response, stable disease, and progression) based on CT and/or PET, comparing imaging before and after the addition of lenvatinib. Radiologic review was conducted by an independent radiologist. Eight patients with dMMR EC meeting treatment criteria were identified. The patients’ ages ranged from 54 to 80 and all tumors identified were of endometrioid histology. Initial pathologic stage ranged from FIGO stage IB to IVB and recurrence confirmed via imaging or tissue biopsy. Patients received a median of 14 cycles of therapy with pembrolizumab and lenvatinib (range 1–39). All patients had decrease in measurable disease with an objective response of 75 % (PR 62.5 %, CR 12.5 %). Both patients who received the initial recommended dose of 20 mg daily required a dose reduction. Based on this retrospective study, patients with dMMR EC without significant benefit from pembrolizumab monotherapy have a significant clinical response after the addition of lenvatinib. Combination therapy should be considered for dMMR EC patients who fail pembrolizumab monotherapy.
1. Introduction
Immunotherapy has changed the landscape of endometrial cancer (EC) treatment, particularly is those tumors deficient in mismatch repair (dMMR) proteins. The landmark Keynote 158 study first demonstrated a benefit to dMMR endometrial cancers, with an observed response rate of 48 %, with 14 % complete responses and 34 % partial responses. (O'Malley et al., 2022) These promising findings led to an FDA approval for pembrolizumab for dMMR patients whose disease progressed following prior treatment and who have no satisfactory alternative treatment options. After this, Keynote 775 demonstrated a substantial benefit when lenvatinib is added to pembrolizumab for mismatch repair proficient (pMMR) patients, thus expanding the role of immunotherapy to EC patients regardless of MMR status. (Makker and N, Casado Herraez, 2022) Most recently, the phase III NRG-GY018 study moved pembrolizumab to the frontline setting. In this study, the addition of immunotherapy to a platinum-based regimen led to an improved progression-free survival (PFS) in both pMMR and dMMR patients. (Eskander et al., 2023).
Despite these promising results, the median PFS indicates that a significant portion of patients will progress or recur on these treatments. Thus, effective second-line therapies after platinum-based chemotherapy, and now PD-1 inhibition, remain a significant unmet need. Alternative treatment strategies have poor response rates in pre-treated EC. Ixabepilone was inferior to doxorubicin as second line therapy in a large randomized trial. (McMeekin et al., 2015) Doxorubicin which was once felt to be one of the most active drugs endometrial cancer has 0 % response rate, in patients previously treated with a platinum and taxane. (McMeekin et al., 2015, Di Legge et al., 2011)The single agent response rate with lenvatinib in EC was only 14.3 %. (Vergote et al., 2013) When given in combination with pembrolizumab the overall response rate was 39.6 %. (Makker et al., 2019) Recently, Mimura et al. reported a single dMMR endometrial cancer patient who progressed on single agent pembrolizumab but responded to the combination of pembrolizumab and lenvatinib. (Mimura et al., 2022) However, in this case report only 22 days of therapy was administered before lenvatinib was discontinued because of a planned urinary diversion for a vesico-vaginal fistula. The overall response rate of Lenvatinib and pembrolizumab in Keynote 775 for dMMR patients was 41.5 % (95 % CI 29.4 – 54.4), supporting the potential activity of this regimen after standard chemotherapy. (Makker and N, Casado Herraez, 2022) Recognizing that efficacious treatment options for recurrent EC after chemotherapy and immunotherapy remain limited, we elected to add lenvatinib to a pembrolizumab backbone in dMMR EC patients who had limited to no benefit to pembrolizumab monotherapy.
2. Methods
This is a retrospective IRB-approved study of MMR-deficient endometrial cancer patients treated at the Cleveland Clinic or in one patient treated at Parkside Hospital in Indiana and subsequently received care at the Cleveland Clinic with the combination of pembrolizumab and lenvatinib following progression or failure to respond to pembrolizumab. Disease progression by iRECIST criteria was documented in six of the patients and one patient had stable disease with elevating CA 125 levels (consistent with disease progression). (Mönch, et al., 2023) One patient with stable disease was treated with the combination due to physician discretion. Radiologic review by RECIST criteria was conducted post hoc by an independent radiologist. All patients or their families gave consent to publish their medical history.
3. Case Presentations
The patient's age, stage, MMR deficiency, and tumor grade, duration of pembrolizumab, response to pembrolizumab, duration of pembrolizumab and lenvatinib, lenvatinib starting and final dosage, response to pembrolizumab and lenvatinib are all listed on Table 1.
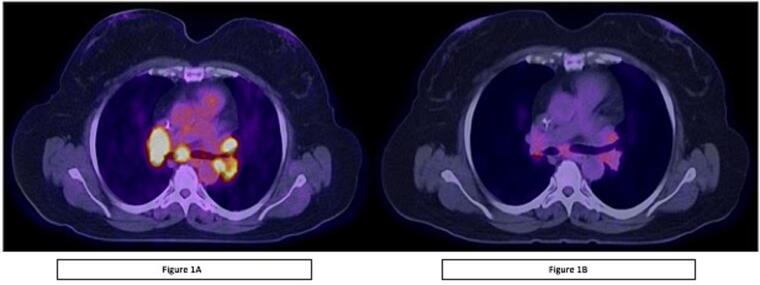
The patient was initiated on pembrolizumab with an MMR deficiency and a tumor mutational burden > 10. PET/CT scan at 3 months 6/8/2021 demonstrated a mixed response with some lymph nodes decreasing in others increasing. The patient was initiated on lenvatinib 20 mg/day. Three cycles later a PET/CT demonstrated a 44 % decrease in disease. Fig. 1 She received a total of 25 cycles until disease progression.
The patient was initiated on pembrolizumab. Disease progression was documented radiologically 3 months later. The patient was initiated on pembrolizumab and lenvatinib initially at 20 mg daily but subsequently decreased to 10 mg daily after cycle 5 due to toxicity (fatigue, weight loss, hypertension). She has now received 39 courses of therapy and her disease decreased by 58 %.
The patient was initiated on pembrolizumab. After 3 cycles her CA125 remained elevated at 396 U/ml. Her imaging demonstrated only stationary disease and lenvatinib 10 mg/day was added to pembrolizumab. Her CA125 rapidly normalized and CT imaging demonstrated a complete response. After cycle 8 the lenvatinib dose reduced to 4 mg daily on 11/8/2022 for uncontrolled hypertension and the patient has now completed 22 cycles.
The patient was initiated on pembrolizumab for 45 cycles. The patient’s imaging demonstrated worsening carcinomatosis and lenvatinib 10 mg daily was added. She only took this for only 21 days because of poor tolerance (fatigue, diarrhea, vomiting, and weight loss). Her CT scan demonstrated decrease in her measurable disease and her CA125 decreased. A plan to reinitiate the lenvatinib at 4 mg/day but the patient did not restart this and was admitted to an outside hospital 10 days later with leukocytosis and hypotension. CT imaging demonstrated no significant abdominal findings. The family elected comfort care and the patient expired.
The patient received pembrolizumab for 6 cycles. After 3 months there was some tumor enlargement (possible pseudo-progression). Three months later her CT scan demonstrated disease progression. She was initiated on lenvatinib but therapy was delayed for an abdominal wall abscess and Covid infection. She has received 8 cycles before progression and her CT scans after 6 cycles demonstrate a radiologic partial response with a 45 % decrease in disease.
The patient was placed on pembrolizumab. Her CT scan after 3 months demonstrated largely stable disease, but some lung lesions were slightly larger (possible pseudo-progression). Following 3 additional cycles of pembrolizumab CT imaging demonstrated progression. Lenvatinib 10 mg/day was added. She has received 15 cycles of pembrolizumab and lenvatinib and achieved a partial response following 6 cycles.
The patient was treated with pembrolizumab for 3 cycles. A CT scan demonstrated disease progression. Lenvatinib 14 mg daily was added. After 3 cycles of pembrolizumab and lenvatinib her CT imaging demonstrated radiologic stable disease with a 9 % decrease in disease. The patient however developed a brain metastasis and suffered a major cerebrovascular accident, and the family chose to pursue hospice.
The patient was initiated pembrolizumab for 11 cycles. Due to CT evidence of progression confirmed by a confirmatory CT scan she was initiated on the pembrolizumab/lenvatinib 14 mg daily. Cycle 3 delayed due to an elevated amylase. She has received 13 cycles of pembrolizumab and lenvatinib with her CT imaging demonstrated radiologic stable disease with a 19 % decrease in disease.
Table 1.
Patient Tumor and Treatment Characteristics.
| Patient number | MMRd | MLH 1 methyl | Age* | Stage FIGO 2009 | Grade | Prior Systemic Therapy | Duration of Pembro | Response to Pembro | Duration of Pembro/ lenvima | Date initiated Lenvima Dose Starting/Final (# courses) | Response |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | MLH1/PMS2 | Present | 54 | IIIC1 Recurrent | FIGO 2 | CP x 6 | 3 Cycles | Stable | 25 cycles | 7/1/2021 20 (1) 10 (24) | Radiologic PR (↓ 44 %) |
| 2 | MLH1/PMS2 | Present | 56 | IVB | FIGO G2 | CPB x 9 B x 4 | 5 cycles | Progression | 39 cycles+ | 7/23/2021 20 (6) 10 (33) | Radiologic PR (↓ 58 %) |
| 3 | MLH1/PMS2 | Present | 73 | IVB | FIGO G1 | CP x 6 Letro x3 | 3 Cycles | Stable CA125 Progression | 22 cycles + | 7/11/2022 10 (2) 4 (20) | Radiologic CR ↓ CA125 396 to 9 U/ml |
| 4 | MLH1/PMS2 | Present | 71 | IIIC2 recurrent | FIGO G3 | CA x 4 Letro x 13 B x 45 | 47 cycles | Progression | 1 cycle | 8/5/2022 10 (1) | Radiologic PR (40 %) ↓ CA125 59.6 to 44.4 U/ml |
| 5 | MLH1/PMS2 | Present | 62 | IIIC1 recurrent | FIGO G2 | CDDP x 1CX1 CP X4 | 9 cycles | Progression | 8 cycles | 9/26/2022 10 (7) 4 (1) | Radiologic PR (45 %) |
| 6 | MLH1/PMS2 | Present | 71 | IB Recurrent | FIGO G3 | CP x 7 Letro x 7 | 5 cycles | Progression | 15 cycles + | 10/31/2022 10 (15) | Radiologic PR (↓ 32 %) |
| 7 | PMS2 | Lynch testing - | 80 | IB Recurrent | FIGO G3 | CIS x 2 CP x 4 | 3 cycles | Progression | 3 cycles | 11/7/2022 14 (3) | Radiologic Stable (↓ 9 %) |
| 8 | MLH1/PMS2 | Present | 79 | IB Recurrent | FIGO G1 | CPx6 | 11 cycles | Progression | 13 cycles + | 1/5/2023 14 (12) 10 (1) | Radiologic Stable (↓ 10 %) |
Abbreviations: * Age when Pembo/lenvima started, A = Abraxane, B = Bevacizumab, C = Carboplatin, Cis = Cisplatin, Lenvima = Lenvatinib, Letro = Letrozole MLH1Methyl = MLH1 promoter hypermethylation, P = Paclitaxel, Pembro = Pembrolizumab, -=negative, + currently in active treatment.
Fig. 1.
Axial PET CT image before treatment (a) show multiple, enlarged and FDG-avid mediastinal lymph nodes. Axial PET CT image from the same level after treatment (b) shows resolution of the FDG-avid mediastinal lymph nodes.
4. Results
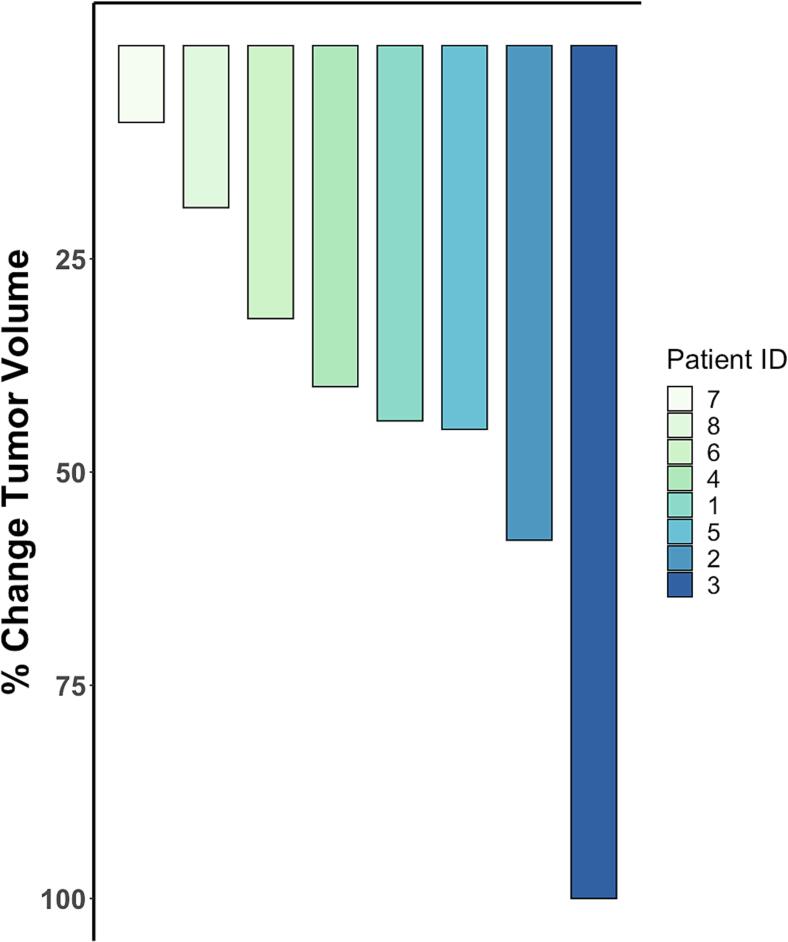
The patients’ ages ranged from 54 to 80 and all tumors identified were of endometrioid histology. Initial pathologic stage ranged from FIGO stage IB to IVB and recurrence confirmed via imaging or tissue biopsy. Patients received a median 14 cycles of therapy with pembrolizumab and lenvatinib (range 1–39). As demonstrated in the waterfall Plot, response to therapy with pembrolizumab and lenvatinib was observed in all patients, compared to baseline tumor measurement. (Fig. 2). Our RECIST 1.1 criteria overall response rate was 75 % (PR 62.5 %, CR 12.5 %). As demonstrated in the swimmer Plot, dose, duration of therapy and response is varied by patient. (Fig. 3) Only two patients initially received the FDA-approved lenvatinib daily dose of 20 mg, and both required dose reduction because of toxicity. Responses were maintained even at the lowest dose of lenvatinib 4 mg daily.
Fig. 2.
Fig. 3.
5. Discussion
We report dMMR patients who failed to have a meaningful response to single agent pembrolizumab but subsequently demonstrated a high rate of objective responses to the addition of Lenvatinib. Mimura et al previously reported one MMR deficient patient who demonstrated objective response to the combination of with pembrolizumab and lenvatinib, after 22 days of therapy. (Mimura et al., 2022) Our case series differs from that report in that more MMR deficient patients (n = 8) are reported and more courses of therapy (n = 126, range 1–39 cycles) were delivered. All but two of our patients initiated a lenvatinib below the FDA recommended dose of 20 mg/day. (Center for Drug Evaluation and Research, 2022) In Study 111/Keynote 146 by Makker et al, although the dose of 20 mg/ day was prescribed, due to dose reductions and delays, the median dose delivered was 14 mg/day. (Makker et al., 2019) Previously, How et al. studying patients who received the the pembrolizumab and lenvatinib combination, retrospectively compared the toxicity and efficacy of initial recommended (20 mg daily) and a reduced starting dose (14 mg daily) of lenvatinib. The response rate, progression-free and overall survival was not different between these two groups. In a subsequent publication on the toxicity of Study 111/Keynote 146, only 2 of the 31 patients who were treated for 6 months or more received the recommended dose of lenvatinib, with some patients receiving only 4 mg/day. (Makker et al., 2021).
Resistance to chemotherapy and immunotherapy will become more prevalent with the FDA approval of PD-1 inhibitors in combination with platinum-based chemotherapy. One way to overcome the poor response rate in certain malignancies is to combine immune checkpoint inhibitors with other agents, observed to cause direct damage to the cancer cell and increase exposure to tumor associated antigens. Via adaptive immunotherapy, tumor associated antigens are presented to the major histocompatibility complex and generate activated T cells which kill cells that have these antigens, ie the cancer cell. It has been reported that treatment with lenvatinib decreased the proportion of monocytes and macrophages population and increased that of CD8 + T cell populations. (Kimura et al., 2018) While lenvatinib has only modest activity in endometrial cancer as a single agent, when utilized in combination with immune checkpoint inhibition significantly higher antitumor activity is seen.
The strength of this article is that it provides an alternative immune therapy for patients who have MMR deficient endometrial cancer who have failed to respond to single agent pembrolizumab. The weaknesses of the study are that it is a small, single institution, and retrospective study. Nonetheless, this retrospective study demonstrates the combination of pembrolizumab and lenvatinib is active in MMR deficient endometrial cancer patients who failed to respond to single agent pembrolizumab. It seems appropriate based on toxicity, cost and our current knowledge to initiate treatment of MMR deficient patients with single agent pembrolizumab. In the event of disease progression on single agent pembrolizumab the combination with pembrolizumab and lenvatinib can be considered. A prospective study comparing to single agent pembrolizumab versus the combination of pembrolizumab and lenvatinib for MMR deficient endometrial cancer patients is planned.
CRediT authorship contribution statement
Peter G. Rose: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. Myra Feldman: Methodology, Project administration, Resources, Software. Iwona Podzielinski: Data curation, Writing – review & editing. Aaron P. Petty: Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization. Roberto Vargas: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Center for Drug Evaluation and Research, 2022. FDA grants regular approval to pembrolizumab and lenvatinib for advanced endometrial carcinoma [WWW Document]. FDA. URL https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-regular-approval-pembrolizumab-and-lenvatinib-advanced-endometrial-carcinoma (accessed 9.26.23).
- Di Legge A., Trivellizzi I.N., Moruzzi M.C., Pesce A., Scambia G., Lorusso D. Phase 2 trial of nonpegylated doxorubicin (Myocet) as second-line treatment in advanced or recurrent endometrial cancer. Int J Gynecol Cancer. 2011;21(8):1446–1451. doi: 10.1097/IGC.0b013e31822d754e. [DOI] [PubMed] [Google Scholar]
- Eskander R.N., Sill M.W., Beffa L., et al. Pembrolizumab plus chemotherapy in advanced endometrial cancer. N. Engl. J. Med. 2023;388:2159–2170. doi: 10.1056/NEJMoa2302312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura T., Kato Y.u., Ozawa Y., Kodama K., Ito J., Ichikawa K., Yamada K., Hori Y., Tabata K., Takase K., Matsui J., Funahashi Y., Nomoto K. Immunomodulatory activity of lenvatinib contributes to antitumor activity in the Hepa1-6 hepatocellular carcinoma model. Cancer Sci. 2018;109(12):3993–4002. doi: 10.1111/cas.13806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makker V., Rasco D., Vogelzang N.J., Brose M.S., Cohn A.L., Mier J., Di Simone C., Hyman D.M., Stepan D.E., Dutcus C.E., Schmidt E.V., Guo M., Sachdev P., Shumaker R., Aghajanian C., Taylor M. Lenvatinib plus pembrolizumab in patients with advanced endometrial cancer: An interim analysis of a multicentre, open-label, single-arm, phase 2 trial. Lancet Oncol. 2019;20(5):711–718. doi: 10.1016/S1470-2045(19)30020-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makker V., Columbo N., Herraez Casado, et al. Lenvatinib plus pembrolizumab for advanced endometrial cancer. N. Engl. J. Med. 2022;386:437–448. doi: 10.1056/NEJMoa2108330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makker V., Taylor M.H., Oaknin A., et al. Characterization and Management of Adverse Reactions in Patients with Advanced Endometrial Carcinoma Treated with Lenvatinib Plus Pembrolizumab. Oncologist. 2021;26:e1599–e1608. doi: 10.1002/onco.13883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMeekin S., Dizon D., Barter J., Scambia G., Manzyuk L., Lisyanskaya A., Oaknin A., Ringuette S., Mukhopadhyay P., Rosenberg J., Vergote I. Phase III randomized trial of second-line ixabepilone versus paclitaxel or doxorubicin in women with advanced endometrial cancer. Gynecol Oncol. 2015;138(1):18–23. doi: 10.1016/j.ygyno.2015.04.026. [DOI] [PubMed] [Google Scholar]
- Mimura K., Shimomura A., Gota T., Ando K., Kawamura Y., Taniyama T., Oishi H., Shimizu C. Response to lenvatinib and pembrolizumab combination therapy in pembrolizumab-pretreated relapsed endometrial cancer. Gynecologic Oncology Reports. 2022;44:101084. doi: 10.1016/j.gore.2022.101084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mönch S., Heimer MM., Winkelmann M., et al. Patterns of pseudoprogression across different cancer entities treated with immune checkpoint inhibitors. Cancer Imaging. 2023;23:58. doi: 10.1186/s40644-023-00580-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Malley D.M., Bariani G.M., Cassier P.A., Marabelle A., Hansen A.R., De Jesus Acosta A., Miller W.H., Safra T., Italiano A., Mileshkin L., Xu L., Jin F., Norwood K., Maio M. Pembrolizumab in patients with microsatellite instability-high advanced endometrial cancer: Results from the KEYNOTE-158 study. J Clin Oncol. 2022;40(7):752–761. doi: 10.1200/JCO.21.01874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vergote I., Teneriello M., Powell M.A., Miller D.S., Garcia A.A., Mikheeva O.N., Pinter T., Bidzinski M., Cebotaru C.L., Fan J., Ren M., Meneses N., Funahashi Y., Kadowaki T., O'Brien J.P., Penson R.T. A phase II trial of lenvatinib in patients with advanced or recurrent endometrial cancer: Angiopoietin-2 as a predictive marker for clinical outcomes. JCO. 2013;31(15_suppl):5520. [Google Scholar]