Abstract
Inactivation of transcription factor ς54, encoded by rpoN (glnF), restores high-temperature growth in Luria-Bertani (LB) medium to strains containing the heat-sensitive cell division mutation ftsZ84. Mutational defects in three other genes involved in general nitrogen control (glnD, glnG, and glnL) also suppress lethal filamentation. Since addition of glutamine to LB medium fully blocks suppression by each mutation, the underlying cause of suppression likely derives from a stringent response to the limitation of glutamine. This model is supported by several observations. The glnL mutation requires RelA-directed synthesis of the nutrient alarmone ppGpp to suppress filamentation. Artificially elevated levels of ppGpp suppress ftsZ84, as do RNA polymerase mutations that reproduce global effects of the ppGpp-induced state. Both the glnF null mutation and an elevated copy number of the relA gene similarly affect transcription from the upstream (pQ) promoters of the ftsQAZ operon, and both of these genetic conditions increase the steady-state level of the FtsZ84 protein. Physiological suppression of ftsZ84 by a high salt concentration was also shown to involve RelA. Additionally, we found that the growth of a glnF or glnD strain on LB medium depends on RelA or supplemental glutamine in the absence of RelA function. These data expand the roles for ppGpp in the regulation of glutamine metabolism and the expression of FtsZ during cell division.
Septation is the bacterial equivalent of cytokinesis and involves the regulated formation of a specialized cell wall at the midpoint of the dividing cell during vegetative growth. In Escherichia coli, loss of septation causes the formation of multinucleate cell filaments and eventual death. FtsZ is a GTPase and is the most abundant of several cell division proteins whose activity is essential for septation (for reviews, see references 29 and 51). FtsZ appears to guide septal invagination. Its subcellular distribution cycles with growth, coalescing at the division site during cytokinesis and then dispersing into the cytosol afterward (33, 40). This bimodal property could reflect a cyclic progression of FtsZ through active and inactive states. Much evidence supports the idea that the cellular activity of FtsZ depends on a dynamic self-polymerization reaction involving changes in localized concentration and a GTPase cycle akin to the tubulin proteins of eukaryotic cells (4, 6, 11, 35, 44, 60, 65). The thermosensitive allele ftsZ84 has been the subject of numerous investigations into the nature of septation since its initial description more than 2 decades ago (47). Genetic, biochemical, and microscopic studies of ftsZ84 strains have helped to generate a model that assigns a structural, and perhaps kinetic, role to FtsZ throughout cytokinesis, as well as a regulatory role for the commencement of septation. The process of discovery and characterization of additional factors affecting FtsZ continue to refine our understanding of its function in the cell cycle.
Purified FtsZ84 protein has reduced GTPase activity at elevated temperature, and cell filamentation is the consequence of this defect in vivo. This filamentation is relieved by growth media of high osmotic strength, a phenomenon originally called “salt repair” (47). The mechanism of salt repair for ftsZ84 is not understood. Conditional lethality imparted by ftsZ84 is genetically suppressed by increased dosages of some genes. Most notably, when present in multiple copies, the ftsZ84 allele suppresses itself, verifying that lethality owes to its reduced activity (39). Three extragenic loci have been characterized as suppressors of ftsZ84 when present in high dosages: the gene encoding the transcription factor SdiA (66) and two genes for regulators of capsular polysaccharide synthesis, rcsB and rcsF (20, 21). The connection between capsule gene regulation and septation is not clear. Too much FtsZ activity also disrupts normal septation (13, 67). Interestingly, this condition of FtsZ excess suppresses filamentation caused by abnormally high expression of another cell division gene, ftsA, which suggests the necessity of a dosage equilibrium between FtsA and FtsZ for their proper functioning (10, 12, 13).
The ftsZ and ftsA genes and another cell division gene, ftsQ, form a complex operon which comprises the distal end of a cluster of 16 aligned and overlapping genes concerning cell division and cell wall metabolism (3). The ftsQAZ operon contains several promoters, and there is genetic evidence for the influence of factors at some of these promoters. The majority of transcripts containing ftsZ appear to originate from promoters located 5′ of the operon (19, 69). One of these promoters (Qp2) is up-regulated by SdiA (66), and another (Qp1) is positively regulated by the stationary growth phase sigma factor ςs (2, 56). Also contributing to FtsZ expression are four internal promoters (Ap, Zp2, Zp3, and Zp4) located within the ftsQ and ftsA coding regions (30) and antisense RNAs that are complementary to the 5′ region of ftsZ (14, 50, 58). The activities of some of these promoters vary inversely with the growth rate (2), and the levels of ftsZ transcripts appear to oscillate with the cell cycle (19). The short-lived nucleotide guanosine tetraphosphate has recently received attention as a possible effector of cell division (61–63). Guanosine tetraphosphate and the related compound guanosine pentaphosphate, together referred to herein simply as ppGpp for brevity, function as an alarmone system and are believed to integrate cellular responses to various forms of nutrient stress (8). Natural synthesis of ppGpp in Escherichia coli occurs exclusively from the RelA and SpoT proteins, and the principal contribution is made by RelA. SpoT normally degrades ppGpp but has synthetic activity under certain conditions (27, 68). In the classic stringent response to the presence of uncharged tRNA, ppGpp increases the fidelity of protein translation (36). ppGpp also globally regulates gene expression by affecting initiation or pausing of RNA transcription and can have a positive or negative effect, depending on the targeted promoter. The identification of mutant ς70, β, and β′ subunits of RNA polymerase that mimic the ppGpp-induced state supports the idea that this signal exerts an important effect at the level of transcription (8, 26).
Until now, no mutation has been reported that suppresses the high-temperature lethality of ftsZ84 on Luria-Bertani (LB) medium. Several chromosomal mutations are identified here that restore septation and high-temperature growth on LB medium to strains having the ftsZ84 mutation. Four of these suppressors are loss-of-function alleles for different genes that regulate general nitrogen assimilation and glutamine metabolism during nitrogen-restricted growth. The data presented here indicate that the likely mechanism of suppression by these mutations involves RelA-dependent synthesis of the nutrient starvation signal ppGpp in response to glutamine limitation. This introduces evidence associating ppGpp with glutamine regulation on LB medium.
MATERIALS AND METHODS
Strains, plasmids, bacteriophages, and growth media.
The bacterial strains, plasmids, and bacteriophages used in this study are listed in Table 1. The ftsZ84 strains used in this study have the genetic background of W3110 Δ(lac)U169 (Court laboratory strain WJW45), except as specifically noted otherwise. Parental strain BSP610 and its derivatives were constructed by P1vir transductions using standard procedures (34) in the following steps. To make BSP610, the ftsZ84 allele of strain JFL100 was first linked to a leu::Tn10 marker and then cotransduced into WJW45 by selection for tetracycline resistance. Heat-sensitive transductants were then converted back to Leu+ prototrophy by using a P1 lysate of WJW45 as the donor and selection on minimal-salts medium plus glucose medium, with subsequent confirmation of heat sensitivity and tetracycline sensitivity. Mutations of glnA, glnD, glnE, glnF, glnG, and gltB were introduced into BSP610 and congenic FtsZ+ parental strain WJW45 by P1vir transductions from phage stocks prepared on donor strains containing the given mutation, employing selection for the drug resistance marker associated with each mutation. Unmarked mutations in glnL, glnB, and gltBDF were introduced, respectively, by transductions into the glnA, glyA, and glnF derivatives of BSP610 and WJW45 by selection for prototrophic conversion of these linked mutations.
TABLE 1.
Strains, plasmids, and phages used in this study
| Strain, plasmid, or phage | Genotype and markers | Derivation, source, or reference |
|---|---|---|
| Strains | ||
| W3110 | E. coli K-12 | Laboratory stock |
| WJW45 | W3110 Δ(lac)U169 | Laboratory stock |
| BSP396 | WJW45 glnF208(rpoN)::Tn10 | 41 |
| BSP440 | WJW45 rpoN::ΔTn10-kan | 41 |
| BSP546 | WJW45 Δ(rpoN operon)::kan | 41 |
| BSP609 | WJW45 leu::Tn10 | WJW45 × P1/SG13082 |
| BSP610 | WJW45 ftZ84 | BSP609 × P1/JFL100 |
| BSP621 | BSP610 rpoN::ΔTn10-kan | BSP610 × P1/BSP440 |
| BSP623 | BSP610 glnF208(rpoN)::Tn10 | BSP610 × P1/BSP396 |
| BSP624 | BSP610 Δ(rpoN-npr)::kan | BSP610 × P1/BSP546 |
| BSP640 | BSP610 glnA::Tn5 | BSP610 × P1/TH16 |
| BSP641 | BSP610 glnD99::Tn10 | BSP610 × P1/RB9040 |
| BSP642 | BSP610 glnG10::Tn5 | BSP610 × P1/YMC12 |
| BSP656 | BSP610 relA251::kan | BSP610 × P1/CF1693 |
| BSP666 | BSP610 ΔglnL2001 | BSP640 × P1/RB9132 |
| BSP672 | BSP610 glnE::Tn5-131 (tetR) | BSP610 × P1/EB2853 |
| BSP677 | BSP610 glyA::Tn5 | BSP610 × P1/NC124 |
| BSP685 | BSP610 glnL2302 | BSP640 × P1/YMC15 |
| BSP686 | BSP610 ΔglnB2307 | BSP677 × P1/RB9011 |
| BSP690 | BSP666 ΔrpoS::kan | BSP666 × P1/CF5006 |
| BSP701 | BSP610 gltBDF500 | BSP624 × P1/EB2904 |
| BSP718 | BSP666 relA251::kan | BSP666 × P1/CF1693 |
| BSP732 | W3110 glnA::Tn5 | W3110 × P1/TH16 |
| BSP742 | BSP610 gltF226Ωkan | BSP610 × P1/MX3003 |
| BSP743 | W3110 ΔglnL2001 | BSP732 × P1/RB9132 |
| BSP747 | BSP610 rpoD504 zgh-3075::Tn10-specR | BSP610 × P1/BSP760 |
| BSP748 | BSP610 rpoD506 zgh-3075::Tn10-specR | BSP610 × P1/BSP761 |
| BSP760 | CF3744 Tn10-specR | CF3744 × λTSK |
| BSP761 | CF3746 Tn10-specR | CF3746 × λTSK |
| BSP762 | WJW45 relA251::kan | WJW45 × P1/CF1693 |
| BSP785 | BSP701 glnG10::Tn5 | BSP701 × P1/YMC12 |
| BSP808 | WJW45 glnD99::Tn10 | WJW45 × P1/RB9040 |
| BSP809 | BSP747 relA251::kan | BSP747 × P1/CF1693 |
| BSP810 | BSP762 glnD99::Tn10 | BSP762 × P1/RB9040 |
| BSP813 | BSP809 spoT207::cat | BSP809 × P1/CF1693 |
| BSP814 | BSP762 spoT207::cat | BSP762 × P1/CF1693 |
| BSP815 | BSP813 rpoS::Tn10 | BSP813 × P1/CF5025 |
| BSP816 | BSP762 glnF208(rpoN)::Tn10 | BSP762 × P1/BSP396 |
| BSP818 | BSP814 rpoD504 zgh-3075::Tn10-specR | BSP814 × P1/BSP760 |
| BSP821 | BSP814 glnF208(rpoN)::Tn10 | BSP814 × P1/BSP396 |
| BSP822 | BSP814 glnD99::Tn10 | BSP814 × P1/RB9040 |
| BSP823 | BSP821 glnF208(rpoN)::Tn10 | BSP821 × P1/BSP396 |
| BSP824 | BSP822 glnD99::Tn10 | BSP822 × P1/RB9040 |
| BSP828 | BSP743 relA251::kan | BSP743 × P1/CF1693 |
| BSP838 | BSP640 glnF208(rpoN)::Tn10 | BSP640 × P1/BSP396 |
| BSP845 | BSP640 glnD99::Tn10 | BSP640 × P1/RB9040 |
| BSP846 | W3110 relA251::kan | W3110 × P1/CF1693 |
| CF1693 | relA251::kan spoT207::cat | M. Cashel |
| CF5006 | ΔrpoS::kan | D. Gentry |
| CF3744 | rpoD504 zgh-3075::Tn10 | J. Hernandez |
| CF3746 | rpoD506 zgh-3075::Tn10 | J. Hernandez |
| CF5025 | rpoS::Tn10 | D. Gentry |
| EB2853 | glnE::Tn5-131 (tetR) | B. Bender |
| EB2904 | gltBDF500 | B. Bender |
| JFL100 | ftsZ84 ilv his thyA deo ara(Am) lac125(Am) galU42(Am) tyrT supF(Ts)A81 | J. Lutkenhaus |
| MX3003 | gltF226Ωkan | F. Valle |
| NC124 | glyA::Tn5 | N. Costantino |
| RB9040 | glnD99::Tn10 | L. Reitzer |
| RB9132 | ΔglnL2001 | L. Reitzer |
| RB9011 | ΔglnB2307 | L. Reitzer |
| SG13082 | leu::Tn10 | S. Gottesman |
| TH16 | glnA::Tn5 | L. Reitzer |
| YMC12 | glnG10::Tn5 | L. Reitzer |
| YMC15 | glnL2302 | L. Reitzer |
| Plasmids | ||
| pRS414 | Protein fusion vector, Apr | R. Simons |
| pRS415 | Operon fusion vector, Apr | R. Simons |
| pCX | pZ-lacZ | L. Rothfield |
| pCX32 | pQ-lacZ | L. Rothfield |
| pCX39 | Qp2-lacZ | L. Rothfield |
| pCX40 | Qp1-lacZ | L. Rothfield |
| pCX38 | pQ-pZ-lacZ | L. Rothfield |
| pGB2 | pSC101 aadA | Laboratory stock |
| pHM675 | pGB2(tacp relA′) | M. Cashel |
| pALS10 | pBR322(tacp relA) | M. Cashel |
| pALS14 | pBR322(tacp relA") | M. Cashel |
| pBP133 | pRS414(Zp2 ftsZ′-lacZ)PF | This study |
| pBP134 | pRS415(Zp2 ftsZ′-lacZ)OF | This study |
| pBP124 | pRS415(orf-241 rpoN orf-95 ptsN orf-284 npr) | 41 |
| pBP130 | pRS415(orf-241 rpoN orf-95 ptsN′) | 41 |
| pBP131 | pRS415(orf-241 ΔrpoN orf-95 ptsN orf-284 npr) | 41 |
| Phages | ||
| λTSK | λ cI857 Tn10 tetA′Ω(kanR specR) | Laboratory stock |
| λGL100 | plac ftsZ | J. Garcia-Lara |
| λBDC531 | λ lacYZ′ supF ′bla att imm21 Aps | Laboratory stock |
| λBP100 | λBDC531 lacYZ+ bla+ | 41 |
| λBP124 | λBDC531 bla+ (orf-241 rpoN orf-95 ptsN orf-284 npr-lacZY) | 41 |
| λBP124.1 | λBDC531 bla+ (orf-241 rpoN orf-95 ptsN::kan orf-284 npr-lacZY) | 41 |
| λBP124.2 | λBDC531 bla+ (orf-241 rpoN::kan orf-95 ptsN orf-284 npr-lacZY) | 41 |
| λBBP133 | λBDC531 bla+(Zp2 ftsZ′-lacZ)PF | λBDC531 × pBP133 |
| λBBP134 | λBDC531 bla+(Zp2 ftsZ′-lacZ)OF | λBDC531 × pBP134 |
Strains that lacked ppGpp, either partially (relA251::kan) or completely (relA251::kan spoT207::cat), were constructed by selection for the drug resistance marker associated with either disruption (68). The order of introduction of various mutations and discovery of certain medium requirements were determined empirically. The following restrictions were employed for construction of the final test strains. LB medium containing glutamine at 0.2% (LBQ) was used for the construction and propagation of ftsZ84 and/or relA-derived strains having the Ntr− phenotype, i.e., with a mutation of glnF, glnD, glnG, or glnL. Tetracycline was sometimes included in the medium during the construction of ftsZ84 relA glnF strains.
The phenotypes of glutamine prototrophy versus auxotrophy and the presence versus the lack of nitrogen control were tested, respectively, by plating at 32°C on M63 salts with glucose with or without glutamine (0.2% [wt/vol] each) and on ammonium-free W salts plus glucose medium containing either glutamine or arginine at 0.2% (wt/vol) as the sole source of nitrogen (57). Amino acid supplementation tests on ftsZ84 suppressors used LB plates that individually contained each amino acid at a 0.05% (wt/vol) final concentration.
Thermosensitivity tests.
While the thermosensitivity of ftsZ84 is known to vary among laboratories, often requiring LB medium without added sodium chloride, we have found that our standard LB medium (10 g of Bacto Tryptone, 5 g of NaCl, 8 g of yeast extract; sterilized via autoclave) consistently produces filamentation and lethality at 42°C for ftsZ84 strains in our W3110 genetic background. We observed that other ftsZ84 strains of different genetic backgrounds were less sensitive on this LB medium, and so all heat sensitivity tests were performed on BSP610 and its derivatives. Heat sensitivities and filamentation were assayed by monitoring colony formation on LB plates and by microscopic inspection of suspended colonies or liquid LB cultures.
Assays for ftsQAZ operon expression.
Reporter constructs containing ftsQAZ fusions to lacZ were introduced into tester strains either on low-copy-number plasmids (9) or in single copy on lambda prophages (Table 1). Phages λBBP133 and λBBP134 contain 342 nucleotides 5′ of the ftsZ start codon, which includes promoter Zp2, along with the first 11 codons of ftsZ joined to lacZ in protein and operon fusion configurations, respectively. To make these, DNA was amplified from the chromosome by using the oligonucleotide primers gttgacgaattcaagcttcgccaacaaggggttaaacatcac and actttaggatccgcgtcattggtaagttccattggttcaaac and ULTma DNA polymerase (Perkin Elmer) and then digested with EcoRI and BamHI and cloned into plasmids pRS414 or pRS415 (55), making plasmids pBP133 and pBP134, respectively. The absence of potential errors acquired during cloning was confirmed by direct sequencing of plasmids with the ABI Prism DNA Sequencing kit (Perkin Elmer) and a 373A DNA sequencer (Perkin Elmer). DNA sequences were assembled and analyzed by using Sequencher 3.0 (Gene Codes Corp.) and the Genetics Computer Group (Madison, Wis.) package, version 8.0. Each fusion was recombined from a plasmid onto λBDC531 to make phages λBBP133 and λBBP134 and used to infect BSP610, and prophage lysogens were screened for unit copy number as previously described (42). These strains were used as parental strains for all related strain constructions. β-Galactosidase activities were assayed by method B of Miller (34) with the following modification. Cultures were harvested by twofold dilution into ice-cold stop solution comprised of Z buffer plus 60-μg/ml chloramphenicol and 0.04% sodium azide.
Quantitation of FtsZ protein.
The steady-state level of FtsZ84 protein in various strains was measured by Western blot assay done by standard procedures (23) and the protocols specified by the manufacturers of the electrophoretic and immunologic reagents. Overnight LB cultures were refreshed by 200-fold dilution and grown at 30°C to an optical density at 600 nm of 0.2 and then shifted to 42°C and grown for 2.5 h. Aliquots were collected at equivalent points in their respective growth curves, and volumes were normalized by optical density (5 ml at an optical density at 600 nm of 0.2). Cells were collected by centrifugation, washed in stop solution (described above), frozen immediately on dry ice, and then stored frozen until later use. To prepare extracts, cell pellets were thawed in 0.5 ml of buffer (50 mM sodium phosphate [pH 7.4], 10 μl of 10-mg/ml phenylmethylsulfonyl fluoride [Boehringer Mannheim]), mixed gently, passed through another freeze-thaw cycle, and then disrupted by sonication. Total protein was estimated via protein assay (Bio-Rad), and then cell extracts were normalized among each other by total protein concentration. Extracts were serially diluted into sodium dodecyl sulfate-polyacrylamide gel electrophoresis loading buffer and boiled briefly. Several dilutions of extract from each strain were tested. Proteins were fractionated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to nitrocellulose (Schleicher & Schuell Inc.) or Immobilon (Millipore) for immunoblot analysis. FtsZ was detected with anti-FtsZ serum and compared to purified FtsZ (gifts from the Rothfield laboratory) by using a peroxide-labeled secondary antibody and a LumiGlo chemiluminescent substrate kit (Kierkegaard & Perry). Band densities on X-Omat AR autoradiograph films (Kodak) were quantified with an LKB Ultroscan XL enhancer laser densitometer (Pharmacia LKB Biotechnologies). To check for consistency of measurements, total proteins were compared to estimates made with bicinchoninic acid and Coomassie Plus Protein Assay reagents (Pierce), and antigen detection was compared to immunoblots analyzed with a Storm 820 Phosphorimage Analyzer and the Vistra Systems Western Blot kit (Molecular Dynamics). Units representing the percent maximum signal of the desired protein band were compiled for FtsZ and an internal standard, NusA, detected with anti-NusA serum (59). FtsZ values from several measurements were normalized against their respective NusA values, adjusted for their respective loading volumes and exposure times, and finally summed to calculate an average FtsZ value for each strain.
RESULTS
Suppression correlates with the Ntr− phenotype.
While exploring the functions of newly identified genes of the rpoN operon (41), we discovered that mutations which disrupt the rpoN gene (41) allowed ftsZ84 strains to grow on LB agar at 42°C. Examination of these suppressor mutations by complementation tests showed that plasmid or lambda clones that replenish only the wild-type rpoN gene could abolish suppression (Table 2). Thus, loss of RNA polymerase sigma factor ς54 appeared to be the causative defect for suppression. From this information, the mechanism of ftsZ84 suppression could be direct or indirect; attributable either directly to an inability to induce promoters under ς54 control or indirectly to the physiological state of diminished glutamine metabolism, since the loss of ς54 activity creates both deficiencies. The relevance of each effect for suppression was tested as described next.
TABLE 2.
Effects of extrachromosomal genes of the rpoN operon on suppression of ftsZ84
| Strain | Relevant genotype | Growth at 42°Cb
|
|||
|---|---|---|---|---|---|
| λBP100a | λBP124 | λBP124.1 | λBP124.2 | ||
| WJW45 | + | + | + | + | |
| BSP610 | ftsZ84 | − | − | − | − |
| BSP621 | ftsZ84 rpoN::ΔTn10-kan | + | − | − | + |
Clones contain various extents of the rpoN operon as diagrammed in Fig. 1 of Powell et al. (41): λBP100, vector; λBP124, rpoN operon; λBP124.1, same as λBP124 but with a transposon in the ptsN gene downstream of rpoN; λBP124.2, same as λBP124 but with a transposon in the rpoN gene. Strains containing a different rpoN mutation (BSP623) or a large deletion of the rpoN operon (BSP624) behaved similarly to BSP621 in these tests. Plasmids containing wild-type rpoN (e.g., pBP130) also reversed suppression, while those containing rpoN mutations (e.g., pBP131) did not.
LB medium was used as described in Materials and Methods. Average colony size was scored as follows after 24 h of growth at 32 or 42°C: +, good growth at both temperatures; −, growth only at the lower temperature.
Mutations of several other genes affecting the regulation of glutamine metabolism were tested singly or in combination for the ability to suppress ftsZ84. This enabled us to distinguish between components causing a general defect in nitrogen regulation, called Ntr−, and those causing glutamine auxotrophy, called Gln− (for reviews of nitrogen regulation see references 28, 31, 32, and 45). Table 3 lists the regulatory genes and their protein products that are commonly known to be important for general nitrogen control and ammonium assimilation in E. coli. The Gln and Ntr phenotypes of strains having mutations in these genes are tabulated alongside findings of whether or not they suppressed ftsZ84. As shown, four single mutations, glnD, glnF, glnG, and glnL, allowed high-temperature growth with significant reductions in cell filamentation. Examination of data from all of these strains revealed that suppression generally correlated with the Ntr− phenotype and not with the Gln− phenotype (Table 3). The only exceptions to this correlation were strains containing gltBDF operon mutations. We reasoned that this apparent inconsistency may reflect the unusual physiology of gltBDF operon mutants. Such mutants possess substantially greater-than-normal levels of free glutamine due to the absence of glutamate synthase activity, which consumes glutamine in making glutamate (45). By combining either of the two different gltBDF operon mutations with the glnG null allele, we observed that suppression of ftsZ84 by glnG was reversed genetically (Table 3, footnote e; compare BSP742 with BSP785). Since such strains are still defective for Ntr regulation, these findings indicated that suppression of filamentation likely relates to glutamine limitation as a consequence of the loss of general nitrogen control. This disfavored the possibility of a more direct effect on FtsZ transcription by ς54. Interestingly, loss of glutamine synthetase (glnA), which is the only biosynthetic enzyme for glutamine in E. coli, does not suppress, and is itself unimportant for suppression by the glnF or glnD mutation (Table 3). It is not well understood how cells lacking both glutamine synthetase and ς54-dependent activation of the Ntr pathway survive on autoclaved LB medium, which contains no detectable free glutamine. The absence of free glutamine in our LB medium was verified by physiological high-pressure liquid chromatography (HPLC) analysis (data not shown). The survival of this mutant and certain other Ntr− mutants on LB medium, however, was found to require active RelA, suggesting a role for RelA in glutamine metabolism, as discussed below.
TABLE 3.
Effect on ftsZ84 exerted by inactivating genes for regulation of glutamine metabolism
| ftsZ84 strain | Mutation(s) | Protein(s) lost | Description or function | Phenotypea
|
Suppression of ftsZ84b | |
|---|---|---|---|---|---|---|
| Gln | Ntr | |||||
| BSP640 | glnA::Tn5 | GS | Glutamine synthetase | − | + | − |
| BSP686 | ΔglnB2307 | PII | Activates NRII phosphatase | + | + | − |
| BSP641 | glnD99::Tn10 | UT/URase | Regulates PII by uridylation | − | − | + |
| BSP672 | glnE::Tn5-131 | AT/ARase | Regulates GS by adenylation | + | + | − |
| BSP623 | glnF208::Tn10 | ς54 | RNA polymerase sigma factor | − | − | + |
| BSP642 | glnG10::Tn5 | NRI | Enhancer protein for ς54 | + | − | + |
| BSP666 | ΔglnL2001c | NRII | Kinase/phosphatase of NRI | + | − | + |
| BSP838 | glnF208::Tn10, glnA::Tn5d | ς54, GS | As described above | − | − | + |
| BSP701 | gltBDF500e | GOGAT, GltF | Glutamate synthase, regulator | ++ | − | − |
| BSP785 | gltBDF, glnG10::Tn5 | GOGAT, GltF, NRI | As described above | ++ | − | − |
Glutamine prototrophy (Gln) and nitrogen gene regulation (Ntr) phenotypes are as follows: Gln+, glutamine prototroph; Gln−, glutamine auxotroph; Gln++, glutamine prototroph with very high constitutive levels of glutamine; Ntr+, able to use poor nitrogen sources; Ntr−, unable to use poor nitrogen sources.
Suppression of ftsZ84 was measured and scored as for Table 2.
The glnL2302 mutant (BSP685) encoding NRII∗ with constitutive NRI kinase activity (Gln+ Ntr+) was also tested and did not affect ftsZ84 temperature sensitivity.
An ftsZ84 mutant containing glnA plus glnD (BSP845) behaved similarly to BSP838.
A different gltB operon mutant (BSP742 with gltF226Ω-kan) was also tested and behaved identically to gltBDF500 whether alone or with glnG10::Tn5.
Suppression of ftsZ84 by Ntr− mutants is blocked by externally added glutamine.
It is known that LB medium contains no free glutamine due to deamidation that occurs during autoclaving (49). We confirmed this absence by physiological HPLC (16a). glnA mutants presumably acquire glutamine from glutamine-containing oligopeptides in the medium. The hypothesis of glutamine limitation for the Ntr− mutants was first tested by enriching LB medium separately with each of the 20 standard amino acids. The added presence of glutamine reversed suppression by all four mutations, as exemplified for the glnF mutation in Table 4. These mutants were not identically sensitive, however, since the glnF strain required an amount of glutamine (0.25% [wt/vol]) five times as great as that required by the other mutants to completely restore the high-temperature lethality of ftsZ84. No other amino acid fully blocked suppression, although leucine, glycine, and methionine produced mild effects (Table 4). Since a return to glutamine sufficiency reversed suppression in all of the suppressor mutants, the physiological state created by glutamine limitation seemed to be important for their suppression of ftsZ84 heat sensitivity.
TABLE 4.
Effect of added amino acids on suppression of ftsZ84 temperature sensitivity
| Strain | Relevant genotype | Growth at 42°Ca
|
|
|---|---|---|---|
| LB | LBQb | ||
| WJW45 | W3110 (parental strain) | + | + |
| BSP610 | ftsZ84 | − | − |
| BSP623 | ftsZ84 glnFc | + | − |
Suppression of ftsZ84 was measured and scored as for Table 2.
LB plates were used without additional supplements or prepared separately with each of 20 amino acids as described in Materials and Methods. Shown here are data for LBQ plates. Leucine affected suppression similarly to glutamine but to a much lesser degree. Glycine and methionine caused a much weaker effect than leucine. No other single amino acid affected suppression of ftsZ84.
Shown here are data for glnF suppression. Suppression of an isogenic strain with a substitution of a glnD (BSP641), glnG (BSP642), or glnL (BSP666) mutation for glnF was also fully blocked by glutamine.
Dependence of ppGpp for suppression of ftsZ84.
Because ppGpp is a global regulator induced by nutrient restriction, we tested whether suppression is affected by changes in the level of ppGpp. This association was first explored by artificially increasing ppGpp with plasmid pALS10. The RelA protein produced from this plasmid is unregulated and gratuitously expresses ppGpp in direct relation to its transcription from a tac promoter (53). Induction of RelA from this plasmid allowed high-temperature growth of all of the ftsZ84 strains tested (Table 5). This indicated that all types of suppression may resemble or be identical to effects caused by the induction of ppGpp.
TABLE 5.
Importance of relA or a ppGpp-like effect for genetic suppression of ftsZ84
| Strain/plasmid | Relevant genotypea | LBb
|
|
|---|---|---|---|
| 32°C | 42°C | ||
| WJW45 | Wild type | + | + |
| BSP610/pALS14 | ftsZ84/vector | + | − |
| BSP610/pALS10 | ftsZ84/RelA | + | + |
| BSP610 | ftsZ84 | + | − |
| BSP743 | glnL | + | + |
| BSP666 | ftsZ84 glnL | + | + |
| BSP846 | relA | + | + |
| BSP828 | glnL relA | (+) | (+) |
| BSP718 | ftsZ84 glnL relA | (+) | − |
| BSP747 | ftsZ84 rpoD* | + | + |
| BSP813 | ftsZ84 relA spoT rpoD* | + | + |
Plasmid pALS10 expresses functional RelA′ from a tacp promoter, and pALS14 expresses an inactive RelA" fragment and served as a vector control. These plasmids were used without isopropyl-β-d-thiogalactopyranoside induction since the multicopy nature of the plasmids is sufficient to produce elevated ppGpp levels. The test medium contained 15-μg/ml ampicillin for plasmid maintenance. Two rpoD* mutations, rpoD504 (shown) and rpoD506, were tested, and both suppressed ftsZ84, although rpoD504 did so better. See Table 1 for complete genotypes.
Colony growth: +, large colonies; (+), tiny colonies; −, no colonies.
Next, we tested whether suppression by the Ntr mutants requires RelA activity. For this, we wished to remove natural sources of ppGpp in the tester strains; however, this proved to be difficult (see Materials and Methods). FtsZ84 mutant strains combining relA with either glnF or glnD required glutamine enrichment to survive on LB medium (i.e., LBQ medium), even at low temperature, and therefore were not useful for suppression tests. The same effect was found for congenic ftsZ+ strains. In contrast to the glnD relA and glnF relA strains (which are both phenotypically Gln− and Ntr−), the glnL relA strain (Gln+ Ntr−) did grow on plain LB at all of the temperatures tested. We reasoned that this was because the glnL mutant has slightly higher levels of internal glutamine (17, 46). The glnL mutation was therefore used to test the importance of relA. While the ftsZ84 glnL strain grew at 42°C, the congenic relA derivative did not (Table 5). We interpret this to mean that suppression of ftsZ84 by glnL depends on relA activity, as predicted. This conclusion presumably extends to the other Ntr− mutants, although this was not tested due to difficulties in constructing the desired multiply defective strains. These data show that increased ppGpp suppresses ftsZ84 and concur with the idea that RelA-directed ppGpp synthesis is of primary importance for ftsZ84 suppression by mutations conferring the Ntr− phenotype.
Finally, we employed mutant forms of sigma factor ς70, rpoD* (Table 4; see Table 7), that constitutively convert RNA polymerase into a form that behaves as though it had been modified by ppGpp (26). These mutations also suppressed ftsZ84 and could do so in the complete absence of cellular ppGpp, i.e., in relA spoT double null mutants (Table 5). This indicated that suppression is caused by an effect generated through ppGpp on RNA polymerase and not by ppGpp itself.
TABLE 7.
Levels of FtsZ84 and NusA proteins in temperature-sensitive and temperature-resistant strains
| Strain/plasmid | Genotype/plasmid | Normalized relativea amt of FtsZ84
|
|
|---|---|---|---|
| Avg (SD) | Ratio | ||
| BSP610 | ftsZ84 | 0.23 (0.02) | 1 |
| BSP623 | ftsZ84 glnF | 0.92 (0.17) | 3.9 |
| BSP610/pGB2 | ftsZ84/vector | 0.35 (0.02) | 1.5 |
| BSP610/pHM675 | ftsZ84/relA′ | 0.78 (0.16) | 3.4 |
Levels of the FtsZ84 and NusA proteins relative to the total protein level were estimated by quantitative immunoblot analysis as described in the text. Raw FtsZ84 values were normalized by division by the respective NusA values and averaged among four appropriately weighted measurements per strain. The average relative amount of NusA among 16 measurements was 0.23, and the standard deviation was 0.03.
Suppression of ftsZ84 by a high salt concentration also depends on RelA function.
The ftsZ84 relA strain was used to explore the necessity of ppGpp for a different but familiar kind of suppression, i.e., that of salt repair (47). The presence of additional NaCl in the LB medium suppressed the filamentation of our parental ftsZ84 strain but not that of its derivative which lacks RelA activity (Table 6). Furthermore, a requirement for RelA activity was also observed for suppression by (NH4)2SO4 and NH4Cl salts, indicating that the connection between suppression and relA is not limited to sodium salts (52). It is known that high sodium chloride levels are associated with accumulation of ppGpp (24).
TABLE 6.
Importance of relA for salt suppression of ftsZ84
| Strain | Relevant genotype | LB
|
LB + salta
|
||
|---|---|---|---|---|---|
| 32°C | 42°C | 32°C | 42°C | ||
| BSP610 | ftsZ84 | + | − | + | + |
| BSP656 | ftsZ84 relA | + | − | + | − |
Colony formation was scored as for Table 5. Salt was added to LB medium at 0.15 to 0.3% (wt/vol). The following salts suppressed ftsZ84 temperature sensitivity: NaCl, NH4Cl, (NH4)2SO4, and Na(NH4)HPO4. Na2HPO4 and NaH2PO4 did not suppress temperature sensitivity.
Suppression does not involve some other known effectors of ftsZ.
To explore the possible involvement of well-known effectors of the FtsZ protein, ftsZ84 strains were constructed that combined either glnL, glnD, or glnF with a mutation of sdiA, sulA, rcsB, rcsF, or rpoS. Perhaps of most relevance is our finding that suppression by glnL is unaffected by a null mutation of rpoS since strain BSP690 (ftsZ84 glnL rpoS) grew at 42°C as well as parental strain BSP666 (ftsZ84 glnL). Importantly, loss of the ςs transcription factor did not alter suppression by a ς70 (rpoD*) mutation (strain BSP815) or by a high salt concentration (data not shown). The added disruption of either sdiA, sulA, rcsB, or rcsF had no effect on the suppression of ftsZ84 by glnF. A potential involvement of the leucine regulatory protein Lrp was tested because we had seen a slight effect of added leucine on suppression (Table 4). Since an lrp glnF ftsZ84 strain grew well at all temperatures and an lrp ftsZ84 strain was still heat sensitive, Lrp appears to be unimportant for this kind of suppression by ppGpp on LB medium. Surprisingly, however, the ftsZ84 lrp strain acquired a heat-sensitive phenotype on M63 minimal salts plus glucose medium where none exists for the parental ftsZ84 (Lrp+) strain.
With the elimination of participation by the proteins tested as described above, the following model for suppression of ftsZ84 emerged: growth on LB medium without Ntr-dependent regulation causes a limitation of glutamine and increased levels of ppGpp. This leads to increased functional activity of FtsZ, which then restores septation. To test this hypothesis, it was sensible to determine whether ppGpp affects FtsZ expression.
Suppression causes changes in transcription of the ftsZ operon.
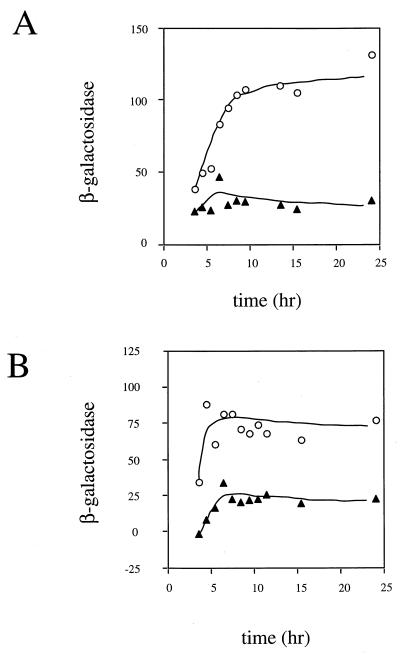
Since filamentation caused by the ftsZ84 allele is known to be suppressed by its own overexpression, we tested whether the glnF mutation or artificially high RelA activity could alter patterns of transcription within the ftsQAZ operon. Transcription of two sets of ftsQAZ operon promoters was measured by using reporter plasmids developed and tested elsewhere (66). These low-copy-number plasmids separately place lacZ under the transcriptional control of ftsZ promoters Zp2, Zp3, and Zp4 (herein called the pZ promoters) or ftsQ promoters Qp1 and Qp2 (herein called the pQ promoters). Contrary to what we expected, the activity of the pQ set of promoters was two- to threefold lower in the glnF strain than in the wild-type strain (Fig. 1A). glnD, glnG, and glnL mutants produced similar effects (data not shown). The effects on ftsZ operon transcription caused by artificially elevated levels of ppGpp were measured by using a compatible plasmid that overproduces RelA′ as the genetic condition of suppression. As was seen for the Ntr− mutations, constitutively high RelA activity also reduced transcription from the pQ promoters about twofold (Fig. 1B). Plasmids that separately contained lacZ fusions to either Qp1 or Qp2 (pCX39 and pCX40, respectively) were tested, and both had decreased β-galactosidase activity in the glnF mutant compared to the wild type (data not shown). In contrast, no consistent change in activity of the pZ set of promoters was observed that was common to both conditions of suppression (data not shown). The activities of protein and operon fusions to the Zp2 promoter carried on λBBP133 and λBBP134, respectively, did not differ between the wild-type and glnF genetic backgrounds (data not shown). This suggested that any change in FtsZ activity caused by suppression is not due to alterations at the level of ftsZ translation and is not observable by using an isolated pZ promoter. Thus, induction of RelA activity correlates with a two- or threefold decrease in the activity of the Qp1 and Qp2 promoters.
FIG. 1.
Activity of the Qp1 and Qp2 promoters in ftsZ84 heat-sensitive and heat-resistant strains. Transcription as measured by β-galactosidase activity of plasmid pCX32 (66) is plotted as a function of growth at 32°C, beginning from a culture density of 107 cells/ml and growth with selection for plasmids. (A) Suppression by glnF. Symbols: ○, BSP610 (ftsZ84); ▴, BSP623 (ftsZ84 glnF). (B) Suppression by multicopy relA′. Symbols: ○, BSP610/pGB2 (ftsZ84/vector); ▴, BSP610/pHM675 (ftsZ84/relA′). We noted that the strains containing plasmid pHM675 grew slowly, and this may reflect the toxic effect of high RelA′ concentrations observed by others (53).
Suppression correlates with increased levels of FtsZ84 protein.
To estimate possible changes in the amount of FtsZ protein that may accompany suppression, the steady-state level of FtsZ84 protein in these strains was measured by immunoblot analysis of whole-cell extracts. The amount of FtsZ84 protein was measured relative to that of an internal standard, NusA. A weighted average among several different loadings and measurements was calculated so as to minimize variations due to sample handling and antigen detection and finally compared between strains. The amount of FtsZ84 relative to the total amount of protein was greater in both of the suppressed strains than in their respective parental strains (Table 7). The glnF mutant strain had 3.9 times more FtsZ84 than the heat-sensitive parent, and the multicopy relA′ strain had 3.4 times more FtsZ84 protein. It appears, then, that the operative effect of suppression by ppGpp is an increase in the steady-state amount of FtsZ84 protein.
E. coli requires ppGpp to grow on LB medium in the absence of a functional Ntr system.
While investigating the role of RelA in the suppression of filamentation, we found that our ftsZ84 strains that lack ppGpp and genes for nitrogen regulation (Ntr−) do not grow in LB medium. To further explore this effect, we constructed Ntr− ftsZ+ derivatives of strains having different levels of ppGpp and tested them for growth on various media. The growth of a relA glnF double mutant strain (BSP816) on LB medium, was severely affected compared to that of its relA+ parental strain (BSP396), while a relA glnD strain (BSP810) was completely nonviable on LB medium (Table 8). Furthermore, the ppGpp0 state rendered both Ntr− strains (BSP821 and BSP822) fully nonviable on LB medium. The lethality of this LB effect was relieved genetically by the rpoD* mutations (Table 8) or physiologically by addition of glutamine to the medium (Table 8). Therefore, ppGpp appears to be critical for fulfillment of the glutamine requirement of Ntr− cells grown on LB medium. This introduces the possibility of a ppGpp-dependent survival mechanism for making glutamine available from LB medium that functions independently of ς54-dependent nitrogen regulation. Interestingly, these relA Ntr− (ftsZ+) strains filamented soon after being transferred from LBQ medium to plain LB medium. However, neither septation nor survival was restored by artificially increasing ftsZ expression (data not shown). This suggests that this effect is not identical to the ppGpp effect on ftsZ under investigation or may involve an earlier stage of septation. In contrast to the glnF and glnD derivatives, a glnA relA strain, which is simply defective for glutamine synthesis (Gln− Ntr+), grew well on LB medium. Since the glnA strain is not stressed on LB medium, as surmised from its inability to suppress ftsZ84, while Ntr− mutants are, it stands to reason that the Ntr system functions in LB medium to provide glutamine in spite of the conventional understanding that the Ntr system is not active in this regard during growth in LB medium (46). From our data, we propose that both the Ntr regulon and an undefined ppGpp-dependent pathway facilitate the supplying of glutamine from a source(s) in LB medium other than de novo synthesis or the transport of free glutamine, which is absent from standard LB medium.
TABLE 8.
Effects of relA, spoT, and rpoD mutations on the growth of a glnF or glnD strain on LB
| Strain | Allele presenta
|
Avg colony sizeb on:
|
|||||
|---|---|---|---|---|---|---|---|
| glnF | glnD | relA | spoT | rpoD | LB | LBQ | |
| Parental | WT | WT | WT | WT | WT | ++ | ++ |
| BSP396 | 208 | WT | WT | WT | WT | ++ | ++ |
| BSP808 | WT | 99 | WT | WT | WT | ++ | ++ |
| BSP762 | WT | WT | 251 | WT | WT | ++ | ++ |
| BSP816 | 208 | WT | 251 | WT | WT | (+) | ++ |
| BSP810 | WT | 99 | 251 | WT | WT | − | ++ |
| BSP814 | WT | WT | 251 | 207 | WT | + | + |
| BSP821 | 208 | WT | 251 | 207 | WT | − | + |
| BSP822 | WT | 99 | 251 | 207 | WT | − | + |
| BSP818 | WT | WT | 251 | 207 | 504 | + | + |
| BSP823 | 208 | WT | 251 | 207 | 504 | + | + |
| BSP824 | WT | 99 | 251 | 207 | 504 | (+) | + |
Strains contain mutations in the listed genes, as indicated by allele number, or have the wild-type (WT) locus. The parental strain is WJW45. Full strain genotypes are listed in Table 1.
LB medium was used with or without glutamine (Q) at 6.8 mM. Average colony sizes were scored after 24 h of incubation at 32°C as follows: ++, large to medium (0.5- to 0.2-mm diameter); +, small (0.1-mm diameter); (+), tiny (<0.05-mm diameter); −, no growth or no individual colonies.
DISCUSSION
This study demonstrates that an aspect of septation directed by FtsZ is influenced positively by nutritional stress and that the mechanism of this effect likely depends on the synthesis of ppGpp by RelA. We propose that ppGpp may effect suppression of FtsZ84 by increasing the total activity of the protein through an altered pattern of transcription, and we offer the following arguments in support of this hypothesis.
Suppression of heat sensitivity on LB medium by loss of nitrogen control, i.e., the Ntr− phenotypic condition, correlates with an increased steady-state concentration of the FtsZ84 protein (Table 7). This concurs with other findings that the cellular amount of FtsZ is normally rate limiting (38, 67) and that an increase of as little as a 1.5-fold can suppress heat-sensitive filamentation of ftsZ84 (66). It is worth noting here that suppression of filamentation by ppGpp appears to be allele specific since the Ntr− mutants did not abate heat sensitivity caused by the ftsZ26 allele, which cannot suppress itself by increased copy number (5) or by the ftsA12 mutation (data not shown). We postulate that the increase in FtsZ protein that accompanies loss of nitrogen control probably involves an elevated basal level of the nutrient stress signal ppGpp for the following reasons. The glnL mutant requires RelA to suppress ftsZ84 (Table 5), an abundance of glutamine annuls suppression by the nitrogen control mutations (Tables 2 and 4), and genetic conditions that overproduce RelA or mimic the ppGpp-induced state (rpoD*) suppress ftsZ84 (Table 5). The requirement of RelA for salt repair shows separately that the relief of ftsZ84 heat sensitivity by ppGpp operates there as well (Table 6).
A reasonable mechanism for RelA-dependent suppression would invoke changes in the transcription of ftsZ due to the known effect of ppGpp on RNA polymerase (8, 26). Indeed, the restoration of high-temperature growth of ftsZ84 strains by SdiA correlates with altered transcription and increased FtsZ protein levels (66). However, the mechanism of SdiA suppression is not similar to that described here since ppGpp decreases expression at both promoters Qp1 and Qp2 (Fig. 1), while SdiA activates the Qp2 promoter. Furthermore, SdiA is not needed for suppression by ppGpp. We note that there is no measured effect of ppGpp on transcription from the downstream pZ promoters or on translation of FtsZ.
We offer two possible scenarios of suppression that might accommodate the decrease in promoter activity upstream of ftsZ84. If the pZ promoters adjacent to ftsZ function independently of the pQ promoters upstream, then suppression might be the consequence of an increase in the ratio of FtsZ to FtsA and/or FtsQ. This scenario alone, however, does not explain why the absolute amount of FtsZ84 protein increases upon suppression. Alternatively, the phenomenon of promoter occlusion may account for the effect of decreased promoter activity, as well as that of increased FtsZ84 protein levels. Promoter occlusion is the inhibitory effect that elongating RNA polymerase molecules have upon transcription initiation at relatively weaker promoters downstream. This was originally suggested with respect to the activities of multiple trp operon promoters (25) and substantiated with respect to the lambda pL promoter and nearby pGal promoters (1). Promoter occlusion is a well-known phenomenon often called transcription interference at eukaryotic promoters (18, 43). By comparison, then, the predominant transcription arising early in the ftsQAZ operon may inhibit the activity of promoters nearer to ftsZ. Thus, downstream promoters may be activated by reducing promoter activity upstream. A corollary of this model is that translation is more efficient from mRNA originating at the downstream promoter (1). The predicted activation of the reporter-linked promoters on prophages λBBP133 and λBBP134 would not be detectable in these experiments since they do not contain the upstream promoters in cis. Plasmids carrying reporter fusions that do contain both sets of promoters in cis (68) are not stable under the genetic conditions tested here. Perhaps such constructs adversely affect the cell by introducing extra copies of ftsQ and ftsA. If promoter occlusion explains ftsZ84 suppression by decreased activity of promoters Qp1 and Qp2, then how can it agree with SdiA suppression by increased activity of promoter Qp2? We predict that although activation of Qp2 would further occlude pZ, the increase in total ftsZ translation from an increased amount of the longer transcripts must allow sufficient FtsZ expression. These models do not necessarily exclude one another and, in fact, there are probably many ways to activate FtsZ. For example, a cis-dependent model for regulation of the activities of ftsQAZ operon promoters based on DNA-looping and putative multiple operator sites has also been proposed (15). Early on, we had considered the possibility of a direct effect by ppGpp on the GTPase activity of FtsZ, but this idea was dismissed by the finding that the rpoD* mutation suppresses ftsZ84 in the complete absence of ppGpp (Table 5). Thus, it appears that the essence of suppression by ppGpp may be an increase in the total activity of FtsZ84 protein through transcriptional regulation. These data, however, do not distinguish between a direct and an indirect effect of ppGpp on ftsZ transcription.
Connections between ppGpp and septation have been suggested previously. Studies on the first ppGpp0 strains, which filament upon nutritional downshift (68), provided the first direct indication that ppGpp may normally stimulate septation. Gervais et al. mentioned unpublished data showing that multiple copies of relA suppress ftsZ84 (21). ppGpp has been shown to help confer resistance to the cell wall inhibitor mecillinam via several different mutations (62–64). In these examples, it is proposed that FtsZ levels could be especially limiting due to the abnormally wide diameter of these cells, and ppGpp may exert a suppressive effect by increasing the total amount of FtsZ. A similar explanation is given for suppression by ppGpp of heat-sensitive filamentation caused by a mutant of the β subunit of RNA polymerase (61). Perhaps this rpoB mutation affects the same step in transcription as the rpoD* mutants used here, except that it creates an RNA polymerase that is less responsive to ppGpp. Another rpoB mutant appears to resemble rpoD* and increases expression of ftsZ (7). The work reported here concurs with these findings and is the first direct evidence that RelA-mediated synthesis of ppGpp suppresses the ftsZ84 mutant on LB medium. Other indications of a role for ppGpp have come from its effect on growth rate control. It has been known for some time that E. coli cell size decreases under poorer growth conditions. In concordance with this, FtsZ expression has been shown to vary inversely with growth (2, 16, 48) and, by inference, with ppGpp, although a direct connection has not been shown until now. Thus, increased ppGpp correlates with increased FtsZ protein levels in E. coli cultures that enter stationary growth (2, 19, 56) or are otherwise under nutritional stress (54).
Altogether, the data presented here may elucidate some previously unexplained phenomena. One example is the suppression of ftsZ84 by multiple copies of the capsule regulators rcsB and rcsF (20, 21). Since suppression involved high-copy plasmids containing both an intact rcs regulator gene and its ς54 promoter, it is plausible that the plasmids may titrate a finite and constitutive supply of ς54 (41), thus leading to glutamine insufficiency, elevated ppGpp, and, hence suppression. The physiological suppression of ftsZ84 by high-osmotic-strength medium (Table 6) probably operates partly through a similar mechanism, since osmotic stress induces ppGpp (24). Interestingly, salt and ppGpp act oppositely on the isomerization reaction that converts the RNA polymerase holoenzyme into the elongating conformation at the rrn promoters of E. coli (22, 37). In this regard, it would be informative to measure the activities of ftsQAZ promoters as a function of medium osmolarity.
During this study, we found evidence that Ntr regulation is important for supplying glutamine in cells grown in LB medium since the absence of Ntr activity requires the nutrient stress factor ppGpp or enrichment of the LB medium with glutamine. This also reveals that ppGpp-dependent gene induction comprises an auxiliary path for supplying glutamine on LB medium. Peptide-bound glutamine can be less susceptible than free glutamine to nonenzymatic deamidation under harsh conditions (e.g., autoclaving) (49), and so the source of glutamine in LB made accessible by the ppGpp pathway may derive from the abundant supply of oligopeptides. Specific mutants which bypass the ppGpp requirement of this peptide transport and degradation pathway might be found by using combinations of Ntr− glnA relA strains and selecting for growth on LB medium.
These findings interrelate nitrogen control and cell division by a global signal of nutrient stress, ppGpp, and begin to explain some of the previously observed difficulty and variability encountered in working with Ntr− strains, as well as with ftsZ84 strains. While glnF, glnG, glnD, glnL, and rpoD* are the first mutations reported to suppress ftsZ84 on LB medium, the detailed mechanism of the ppGpp effect on FtsZ is not readily apparent and the promoter occlusion model proposed here awaits more direct experimental examination.
ACKNOWLEDGMENTS
We are thankful to the sources of strains, lambda phages, antisera, and plasmids listed in Table 1. We are particularly indebted to the following people for kindly responding to numerous requests and for helpful discussions: L. Reitzer, J. Garcia-Lara, L. Rothfield, J. Hernandez, D. Gentry, D. Vinella, and M. Cashel. We sincerely thank R. Dinterman for generously performing the amino acid analysis of LB medium by physiological HPLC.
This research was sponsored by the National Cancer Institute under contract with ABL.
REFERENCES
- 1.Adhya S, Gottesman M. Promoter occlusion: transcription through a promoter may inhibit its activity. Cell. 1982;29:939–944. doi: 10.1016/0092-8674(82)90456-1. [DOI] [PubMed] [Google Scholar]
- 2.Aldea M, Garrido T, Pla J, Vicente M. Division genes in Escherichia coli are expressed coordinately to cell septum requirements by gearbox promoters. EMBO J. 1990;9:3787–3794. doi: 10.1002/j.1460-2075.1990.tb07592.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ayala J A, Garrido T, de Pedro M A, Vicente M. Molecular biology of bacterial septation. In: Ghuysen J M, Hackenbeck R, editors. Bacterial cell wall. Amsterdam, The Netherlands: Elsevier Science; 1994. pp. 73–101. [Google Scholar]
- 4.Bi E, Lutkenhaus J. FtsZ regulates frequency of cell division in Escherichia coli. J Bacteriol. 1990;172:2765–2768. doi: 10.1128/jb.172.5.2765-2768.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bi E, Lutkenhaus J. Isolation and characterization of ftsZ alleles that affect septal morphology. J Bacteriol. 1992;174:5414–5423. doi: 10.1128/jb.174.16.5414-5423.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bi E F, Lutkenhaus J. FtsZ ring structure associated with division in Escherichia coli [see comments] Nature. 1991;354:161–164. doi: 10.1038/354161a0. [DOI] [PubMed] [Google Scholar]
- 7.Cam K, Cuzange A, Bouche J P. Sigma S-dependent overexpression of ftsZ in an Escherichia coli K-12 rpoB mutant that is resistant to the division inhibitors DicB and DicF RNA. Mol Gen Genet. 1995;248:190–194. doi: 10.1007/BF02190800. [DOI] [PubMed] [Google Scholar]
- 8.Cashel M, Gentry D R, Hernandez J, Vinella D. The stringent response. In: Neidhardt F C, Ingraham J L, Low K B, Magasanik B, Schaechter M, Umbarger H E, editors. Escherichia coli and Salmonella: cellular and molecular biology. Washington, D.C: ASM Press; 1996. pp. 1458–1496. [Google Scholar]
- 9.Cortay J C, Cozzone A J. A study of bacterial response to polypeptide antibiotics. FEBS Lett. 1983;157:307–310. doi: 10.1016/0014-5793(83)80566-3. [DOI] [PubMed] [Google Scholar]
- 10.Dai K, Lutkenhaus J. The proper ratio of FtsZ to FtsA is required for cell division to occur in Escherichia coli. J Bacteriol. 1992;174:6145–6151. doi: 10.1128/jb.174.19.6145-6151.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.de Boer P, Crossley R, Rothfield L. The essential bacterial cell-division protein FtsZ is a GTPase. Nature. 1992;359:254–256. doi: 10.1038/359254a0. [DOI] [PubMed] [Google Scholar]
- 12.Descoteaux A, Drapeau G R. Regulation of cell division in Escherichia coli K-12: probable interactions among proteins FtsQ, FtsA, and FtsZ. J Bacteriol. 1987;169:1938–1942. doi: 10.1128/jb.169.5.1938-1942.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dewar S J, Begg K J, Donachie W D. Inhibition of cell division initiation by an imbalance in the ratio of FtsA to FtsZ. J Bacteriol. 1992;174:6314–6316. doi: 10.1128/jb.174.19.6314-6316.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dewar S J, Donachie W D. Antisense transcription of the ftsZ-ftsA gene junction inhibits cell division in Escherichia coli. J Bacteriol. 1993;175:7097–7101. doi: 10.1128/jb.175.21.7097-7101.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dewar S J, Donachie W D. Regulation of expression of the ftsA cell division gene by sequences in upstream genes. J Bacteriol. 1990;172:6611–6614. doi: 10.1128/jb.172.11.6611-6614.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dewar S J, Kagan-Zur V, Begg K J, Donachie W D. Transcriptional regulation of cell division genes in Escherichia coli. Mol Microbiol. 1989;3:1371–1377. doi: 10.1111/j.1365-2958.1989.tb00118.x. [DOI] [PubMed] [Google Scholar]
- 16a.Dinterman, R., and B. Powell. Unpublished data.
- 17.Feng J, Atkinson M R, McCleary W, Stock J B, Wanner B L, Ninfa A J. Role of phosphorylated metabolic intermediates in the regulation of glutamine synthetase synthesis in Escherichia coli. J Bacteriol. 1992;174:6061–6070. doi: 10.1128/jb.174.19.6061-6070.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Friesen P D, Miller L K. Temporal regulation of baculovirus RNA: overlapping early and late transcripts. J Virol. 1985;54:392–400. doi: 10.1128/jvi.54.2.392-400.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Garrido T, Sanchez M, Palacios P, Aldea M, Vicente M. Transcription of ftsZ oscillates during the cell cycle of Escherichia coli. EMBO J. 1993;12:3957–3965. doi: 10.1002/j.1460-2075.1993.tb06073.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gervais F G, Drapeau G R. Identification, cloning, and characterization of rcsF, a new regulator gene for exopolysaccharide synthesis that suppresses the division mutation ftsZ84 in Escherichia coli K-12. J Bacteriol. 1992;174:8016–8022. doi: 10.1128/jb.174.24.8016-8022.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gervais F G, Phoenix P, Drapeau G R. The rcsB gene, a positive regulator of colanic acid biosynthesis in Escherichia coli, is also an activator of ftsZ expression. J Bacteriol. 1992;174:3964–3971. doi: 10.1128/jb.174.12.3964-3971.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hamming J, Ab G, Gruber M. E. coli RNA polymerase-rRNA promoter interaction and the effect of ppGpp. Nucleic Acids Res. 1980;8:3947–3963. doi: 10.1093/nar/8.17.3947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Harlow E, Lane D, editors. Antibodies: a laboratory manual. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory; 1988. [Google Scholar]
- 24.Harshman R B, Yamazaki H. MSI accumulation induced by sodium chloride. Biochemistry. 1972;11:615–618. doi: 10.1021/bi00754a023. [DOI] [PubMed] [Google Scholar]
- 25.Hausler B, Somerville R L. Interaction in vivo between strong closely spaced constitutive promoters. J Mol Biol. 1979;127:353–356. doi: 10.1016/0022-2836(79)90335-8. [DOI] [PubMed] [Google Scholar]
- 26.Hernandez V J, Cashel M. Changes in conserved region 3 of Escherichia coli sigma 70 mediate ppGpp-dependent functions in vivo. J Mol Biol. 1995;252:536–549. doi: 10.1006/jmbi.1995.0518. [DOI] [PubMed] [Google Scholar]
- 27.Johnson G S, Adler C R, Collins J J, Court D. Role of the spoT gene product and manganese ion in the metabolism of guanosine 5′-diphosphate 3′-diphosphate in Escherichia coli. J Biol Chem. 1979;254:5483–5487. [PubMed] [Google Scholar]
- 28.Kustu S, Santero E, Keener J, Popham D, Weiss D. Expression of sigma 54 (ntrA)-dependent genes is probably united by a common mechanism. Microbiol Rev. 1989;53:367–376. doi: 10.1128/mr.53.3.367-376.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lutkenhaus J, Mukherjee A. Cell division. In: Neidhardt F C, Ingraham J L, Low K B, Magasanik B, Schaechter M, Umbarger H E, editors. Escherichia coli and Salmonella: cellular and molecular biology. Washington, D.C: ASM Press; 1996. pp. 1615–1626. [Google Scholar]
- 30.Lutkenhaus J F, Wu H C. Determination of transcriptional units and gene products from the ftsA region of Escherichia coli. J Bacteriol. 1980;143:1281–1288. doi: 10.1128/jb.143.3.1281-1288.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Magasanik B. Genetic control of nitrogen assimilation in bacteria. Annu Rev Genet. 1982;16:135–168. doi: 10.1146/annurev.ge.16.120182.001031. [DOI] [PubMed] [Google Scholar]
- 32.Magasanik B. The regulation of nitrogen utilization in enteric bacteria. J Cell Biochem. 1993;51:34–40. doi: 10.1002/jcb.240510108. [DOI] [PubMed] [Google Scholar]
- 33.Magasanik B. Regulation of transcription of the glnALG operon of Escherichia coli by protein phosphorylation. Biochimie. 1989;71:1005–1012. doi: 10.1016/0300-9084(89)90104-1. [DOI] [PubMed] [Google Scholar]
- 34.Miller J, editor. A short course in bacterial genetics: a laboratory manual and handbook for Escherichia coli and related bacteria. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory Press; 1992. [Google Scholar]
- 35.Mukherjee A, Lutkenhaus J. Guanine nucleotide-dependent assembly of FtsZ into filaments. J Bacteriol. 1994;176:2754–2758. doi: 10.1128/jb.176.9.2754-2758.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nystom T. Role of guanosine tetraphosphate in gene expression and the survival of glucose or seryl-tRNA starved cells of Escherichia coli K12. Mol Gen Genet. 1994;245:355–362. doi: 10.1007/BF00290116. [DOI] [PubMed] [Google Scholar]
- 37.Ohlsen K L, Gralla J D. Interrelated effects of DNA supercoiling, ppGpp, and low salt on melting within the Escherichia coli ribosomal RNA rrnB P1 promoter. Mol Microbiol. 1992;6:2243–2251. doi: 10.1111/j.1365-2958.1992.tb01400.x. [DOI] [PubMed] [Google Scholar]
- 38.Palacios P, Vicente M, Sanchez M. Dependency of Escherichia coli cell-division size, and independency of nucleoid segregation on the mode and level of ftsZ expression. Mol Microbiol. 1996;20:1093–1098. doi: 10.1111/j.1365-2958.1996.tb02549.x. [DOI] [PubMed] [Google Scholar]
- 39.Phoenix P, Drapeau G R. Cell division control in Escherichia coli K-12: some properties of the ftsZ84 mutation and suppression of this mutation by the product of a newly identified gene. J Bacteriol. 1988;170:4338–4342. doi: 10.1128/jb.170.9.4338-4342.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pla J, Sanchez M, Palacios P, Vicente M, Aldea M. Preferential cytoplasmic location of FtsZ, a protein essential for Escherichia coli septation. Mol Microbiol. 1991;5:1681–1686. doi: 10.1111/j.1365-2958.1991.tb01915.x. [DOI] [PubMed] [Google Scholar]
- 41.Powell B S, Court D L, Inada T, Nakamura Y, Michotey V, Cui X, Reizer A, Saier M H, Jr, Reizer J. Novel proteins of the phosphotransferase system encoded within the rpoN operon of Escherichia coli. Enzyme IIANtr affects growth on organic nitrogen and the conditional lethality of an erats mutant. J Biol Chem. 1995;270:4822–4839. doi: 10.1074/jbc.270.9.4822. [DOI] [PubMed] [Google Scholar]
- 42.Powell B S, Rivas M P, Court D L, Nakamura Y, Rivas M P, Turnbough C L., Jr Rapid confirmation of single copy lambda prophage integration by PCR. Nucleic Acids Res. 1994;22:5765–5766. doi: 10.1093/nar/22.25.5765. . (Erratum, 11:1278, 1995.) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Proudfoot N J. Transcriptional interference and termination between duplicated alpha-globin gene constructs suggests a novel mechanism for gene regulation. Nature. 1986;322:562–565. doi: 10.1038/322562a0. [DOI] [PubMed] [Google Scholar]
- 44.RayChaudhuri D, Park J T. Escherichia coli cell-division gene ftsZ encodes a novel GTP-binding protein. Nature. 1992;359:251–254. doi: 10.1038/359251a0. [DOI] [PubMed] [Google Scholar]
- 45.Reitzer L J. Ammonium assimilation and the biosynthesis of glutamine, glutamate, aspartate, asparagine, l-alanine, and d-alanine. In: Neidhardt F C, Ingraham J L, Low K B, Magasanik B, Schaechter M, Umbarger H E, editors. Escherichia coli and Salmonella: cellular and molecular biology. Washington, D.C: ASM Press; 1996. pp. 391–433. [Google Scholar]
- 46.Reitzer L J. Sources of nitrogen and their utilization. In: Neidhardt F C, Ingraham J L, Low K B, Magasanik B, Schaechter M, Umbarger H E, editors. Escherichia coli and Salmonella: cellular and molecular biology. Washington, D.C: ASM Press; 1996. pp. 380–390. [Google Scholar]
- 47.Ricard M, Hirota Y. Process of cellular division in Escherichia coli: physiological study on thermosensitive mutants in cell division. J Bacteriol. 1973;116:314–322. doi: 10.1128/jb.116.1.314-322.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Robin A, Joseleau-Petit D, D’Ari R. Transcription of the ftsZ gene and cell division in Escherichia coli. J Bacteriol. 1990;172:1392–1399. doi: 10.1128/jb.172.3.1392-1399.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Robinson A B, Rudd C J. Deamidation of glutaminyl and asparaginyl residues in peptides and proteins. Curr Top Cell Regul. 1974;8:247–295. doi: 10.1016/b978-0-12-152808-9.50013-4. [DOI] [PubMed] [Google Scholar]
- 50.Robinson A C, Kenan D J, Hatfull G F, Sullivan N F, Spiegelberg R, Donachie W D. DNA sequence and transcriptional organization of essential cell division genes ftsQ and ftsA of Escherichia coli: evidence for overlapping transcriptional units. J Bacteriol. 1984;160:546–555. doi: 10.1128/jb.160.2.546-555.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rothfield L I, Cook W R, de Boer P A. Biogenesis of the Escherichia coli cell division system. Cold Spring Harbor Symp Quant Biol. 1991;56:751–756. doi: 10.1101/sqb.1991.056.01.084. [DOI] [PubMed] [Google Scholar]
- 52.Rowbury R J, Goodson M, Lazim Z, Humphrey T J. Sensitization to acid induced by sodium ions in Escherichia coli: dependence of (p)ppGpp and cAMP and suppression of the relA-associated defect by mutations in envZ. Microbios. 1996;85:161–177. [PubMed] [Google Scholar]
- 53.Schreiber G, Metzger S, Aizenman E, Roza S, Cashel M, Glaser G. Overexpression of the relA gene in Escherichia coli. J Biol Chem. 1991;266:3760–3767. [PubMed] [Google Scholar]
- 54.Schreiber G, Ron E Z, Glaser G. ppGpp-mediated regulation of DNA replication and cell division in Escherichia coli. Curr Microbiol. 1995;30:27–32. doi: 10.1007/BF00294520. [DOI] [PubMed] [Google Scholar]
- 55.Simons R, Houman F, Kleckner N. Improved single and multicopy lac-based cloning vectors for protein and operon fusions. Gene. 1987;53:85–96. doi: 10.1016/0378-1119(87)90095-3. [DOI] [PubMed] [Google Scholar]
- 56.Sitnikov D M, Schineller J B, Baldwin T O. Control of cell division in Escherichia coli: regulation of transcription of ftsQA involves both rpoS and SdiA-mediated autoinduction. Proc Natl Acad Sci USA. 1996;93:336–341. doi: 10.1073/pnas.93.1.336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Smith G R, Halpern Y S, Magasanik B. Genetic and metabolic control of enzymes responsible for histidine degradation in Salmonella typhimurium 4-imidazolone-5-propionate amidohydrolase and N-formimino-l-glutamate formiminohydrolase. J Biol Chem. 1971;246:3320–3329. [PubMed] [Google Scholar]
- 58.Tetart F, Bouche J P. Regulation of the expression of the cell-cycle gene ftsZ by DicF antisense RNA. Division does not require a fixed number of FtsZ molecules. Mol Microbiol. 1992;6:615–620. doi: 10.1111/j.1365-2958.1992.tb01508.x. [DOI] [PubMed] [Google Scholar]
- 59.Tsugawa A, Saito M, Court D L, Nakamura Y. nusA amber mutation that causes temperature-sensitive growth of Escherichia coli. J Bacteriol. 1988;170:908–915. doi: 10.1128/jb.170.2.908-915.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.van Heeswijk W C, Rabenberg M, Westerhoff H V, Kahn D. The genes of the glutamine synthetase adenylylation cascade are not regulated by nitrogen in Escherichia coli. Mol Microbiol. 1993;9:443–457. doi: 10.1111/j.1365-2958.1993.tb01706.x. [DOI] [PubMed] [Google Scholar]
- 61.Vinella D, D’Ari R. Thermoinducible filamentation in Escherichia coli due to an altered RNA polymerase beta subunit is suppressed by high levels of ppGpp. J Bacteriol. 1994;176:966–972. doi: 10.1128/jb.176.4.966-972.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Vinella D, D’Ari R, Jaff A. Penicillin binding protein 2 is dispensable in Escherichia coli when ppGpp synthesis is induced. EMBO J. 1992;11:1493–1501. doi: 10.1002/j.1460-2075.1992.tb05194.x. . (Erratum, 11:5137, 1992.) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Vinella D, Gagny B, Joseleau-Petit D, D’Ari R, Cashel M. Mecillinam resistance in Escherichia coli is conferred by loss of a second activity of the AroK protein. J Bacteriol. 1996;178:3818–3828. doi: 10.1128/jb.178.13.3818-3828.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Vinella D, Joseleau-Petit D, Thevenet D, Bouloc P, D’Ari R. Penicillin-binding protein 2 inactivation in Escherichia coli results in cell division inhibition, which is relieved by FtsZ overexpression. J Bacteriol. 1993;175:6704–6710. doi: 10.1128/jb.175.20.6704-6710.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Wang X, Lutkenhaus J. The FtsZ protein of Bacillus subtilis is localized at the division site and has GTPase activity that is dependent upon FtsZ concentration. Mol Microbiol. 1993;9:435–442. doi: 10.1111/j.1365-2958.1993.tb01705.x. [DOI] [PubMed] [Google Scholar]
- 66.Wang X D, de Boer P A, Rothfield L I. A factor that positively regulates cell division by activating transcription of the major cluster of essential cell division genes of Escherichia coli. EMBO J. 1991;10:3363–3372. doi: 10.1002/j.1460-2075.1991.tb04900.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ward J E, Jr, Lutkenhaus J. Overproduction of FtsZ induces minicell formation in E. coli. Cell. 1985;42:941–949. doi: 10.1016/0092-8674(85)90290-9. [DOI] [PubMed] [Google Scholar]
- 68.Xiao H, Kalman M, Ikehara K, Zemel S, Glaser G, Cashel M. Residual guanosine 3′,5′-bispyrophosphate synthetic activity of relA null mutants can be eliminated by spoT null mutations. J Biol Chem. 1991;266:5980–5990. [PubMed] [Google Scholar]
- 69.Zhou P, Helmstetter C E. Relationship between ftsZ gene expression and chromosome replication in Escherichia coli. J Bacteriol. 1994;176:6100–6106. doi: 10.1128/jb.176.19.6100-6106.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]