Abstract
Background
Hypoxic burden (HB) has emerged as a strong predictor of cardiovascular risk in obstructive sleep apnoea (OSA). We aimed to assess the potential of HB to predict the cardiovascular benefit of treating OSA with continuous positive airway pressure (CPAP).
Methods
This was a post hoc analysis of the ISAACC trial (ClinicalTrials.gov: NCT01335087) including non-sleepy patients with acute coronary syndrome (ACS) diagnosed with OSA (apnoea–hypopnoea index ≥15 events·h−1) by respiratory polygraphy. Patients were randomised to CPAP or usual care and followed for a minimum of 1 year. HB was calculated as the total area under all automatically identified desaturations divided by total sleep time. Patients were categorised as having high or low baseline HB according to the median value (73.1%min·h−1). Multivariable Cox regression models were used to assess whether the effect of CPAP on the incidence of cardiovascular outcomes was dependent on the baseline HB level.
Results
The population (362 patients assigned to CPAP and 365 patients assigned to usual care) was middle-aged (mean age 59.7 years), overweight/obese and mostly male (84.5%). A significant interaction was found between the treatment arm and the HB categories. In the high HB group, CPAP treatment was associated with a significant reduction in the incidence of cardiovascular events (HR 0.57, 95% CI 0.34–0.96). In the low HB group, CPAP-treated patients exhibited a trend toward a higher risk of cardiovascular outcomes than those receiving usual care (HR 1.33, 95% CI 0.79–2.25). The differential effect of the treatment depending on the baseline HB level followed a dose–response relationship.
Conclusion
In non-sleepy ACS patients with OSA, high HB levels were associated with a long-term protective effect of CPAP on cardiovascular prognosis.
Tweetable abstract
Hypoxic burden stands out as an obstructive sleep apnoea severity metric with potential clinical utility to identify patients most likely to benefit from continuous positive airway pressure treatment for secondary cardiovascular prevention https://bit.ly/3szv4hf
Introduction
Obstructive sleep apnoea (OSA) is a chronic sleep disorder affecting approximately 1 billion people worldwide [1]. OSA is characterised by recurrent episodes of upper airway collapse leading to large intrathoracic pressure swings and repetitive cycles of oxygen desaturation and hypercapnia followed by re-oxygenation and/or arousal from sleep [2]. These perturbations initiate a cascade of downstream consequences, including sympathetic activation, inflammation, oxidative stress, metabolic dysregulation and endothelial dysfunction, which can ultimately trigger cardiovascular disease (CVD) [3].
Untreated OSA is associated with wide-ranging complications, especially in the cardiovascular sphere [4–10]. Together with lifestyle interventions, treatment with continuous positive airway pressure (CPAP) constitutes the first-line therapy for moderate-to-severe or symptomatic OSA patients. Previous evidence from randomised controlled trials (RCTs) has shown a favourable impact of CPAP treatment on a variety of CVD drivers, such as high blood pressure [11], inflammation [12], endothelial dysfunction [13] and atherosclerosis [14]. Nonetheless, despite the cumulative clinical and experimental data pointing to OSA as a modifiable risk factor for CVD [15], the effect of CPAP treatment on cardiovascular end-points remains controversial [16]. In the past decade, three large secondary prevention RCTs have explored the cardiovascular benefits of CPAP, failing to demonstrate a positive long-term effect of the treatment on the incidence of adverse cardiovascular outcomes [17–19]. The high variability of response among the available studies emphasises the existence of substantial heterogeneity in the impact of CPAP therapy on cardiovascular consequences [20]. This clinical scenario invites us to question whether specific subgroups of patients with OSA may benefit more from the therapy than others.
The current framework for the management of OSA reflects a “one size fits all” approach, where a single parameter, the apnoea–hypopnoea index (AHI), is used to define the presence, categorise the severity and guide the physician prescription for therapy. This generalist way of addressing a disease as complex as OSA can lead to important repercussions for patients and healthcare systems. Compelling data indicate that the AHI alone does not capture the heterogeneity of OSA and its underlying physiopathological consequences, particularly for the cardiovascular system [21]. Under this paradigm, substantial efforts are being made to develop alternative metrics that could effectively characterise OSA and predict adverse outcomes. A growing list of candidates have been proposed in recent years [22]. Promisingly, hypoxic burden (HB) has emerged as a strong predictor of CVD-related morbidity and mortality in several population-based and clinical cohorts [23–28]. However, the extent to which HB can be used to guide CPAP therapeutic decision making remains to be determined. In this study, we sought to assess the potential of HB to identify a specific subgroup of OSA patients who exhibit cardiovascular benefit from CPAP therapy.
Methods
Study population
This is a post hoc analysis of the ISAACC study (ClinicalTrials.gov: NCT01335087), a multicentre, open-label, parallel and prospective RCT [17]. A detailed description of the study aims and protocol, including inclusion and exclusion criteria, is outlined elsewhere [29]. Briefly, consecutive adult subjects admitted for acute coronary syndrome (ACS) to the coronary care unit or cardiology hospitalisation ward were eligible to participate in the study if they did not exhibit excessive daytime sleepiness, defined as Epworth Sleepiness Scale (ESS) ≤10. ACS was defined as the acute presentation of coronary disease with or without ST segment elevation infarction, unstable angina or type 1 myocardial infarction. Recruitment took place throughout 15 hospitals in Spain between 2011 and 2018, with the last end-of-study visit conducted in 2019. The clinical research ethics committee of each participating centre approved the trial protocol (approval number in the coordinating centre (University Hospital Arnau de Vilanova and Santa María, IRBLleida, Lleida, Spain): 2010/852) and every enrolled patient provided informed written consent to participate in the study.
Procedures and outcomes of the ISAACC trial
Baseline clinical evaluation
In the initial visit, detailed information regarding sociodemographic characteristics, health behaviours and medical history, including comorbidities and prescribed medications, was collected from all patients by trained clinicians. General physical and anthropometric parameters were recorded, and the degree of self-reported daytime sleepiness was assessed with the validated Spanish version of the ESS.
Sleep study
All patients who satisfied the selection criteria underwent respiratory polygraphy (Embletta; ResMed, Bella Vista, Australia) during the first 24–72 h after the time of hospitalisation for ACS. Sleep recordings included at least oronasal flow measurement (nasal pressure and thermistor), thoracoabdominal respiratory inductance plethysmography, heart rate measurement and fingertip pulse oximetry. All methods were performed according to the national clinical practice guidelines and regulations [30]. Apnoeas were defined as an interruption in oronasal airflow for ≥10 s. Hypopnoeas were defined as a reduction in oronasal airflow for ≥10 s associated with a decrease in arterial oxygen saturation (SaO2) ≥4%. Hypoxaemia parameters included the oxygen desaturation index (ODI), defined as the number of episodes of SaO2 decrease ≥4%, and the percentage of total sleep time spent with SaO2 <90% (TSat90). The definition of the study groups in the ISAACC trial was based on the AHI, which represents the average number of apnoea plus hypopnoea events per hour of sleep. Patients diagnosed with OSA (AHI ≥15 events·h−1) were randomly assigned (1:1) to the CPAP group or the usual care group. Usual care instructions comprised education on lifestyle and behavioural modifications.
Follow-up evaluations
The primary end-point of the ISAACC trial was a composite of death from any cardiovascular cause or non-fatal events, including acute myocardial infarction, non-fatal stroke, hospital admission for heart failure and new hospitalisations for unstable angina or transient ischaemic attack. Follow-up visits were scheduled for all patients at 1, 3, 6, 12, 18, 24, 30 and 36 months and annually thereafter. All patients were monitored and followed up for a minimum of 1 year. Each clinic visit included a physical examination, detailed record of current medication use, unhealthy life habits and assessment of the rate of cardiovascular events. A blinded committee adjudicated both fatal and non-fatal cardiovascular outcomes specified in the protocol. The follow-up time was defined as the time between the baseline visit and the end of the study or the occurrence of a cardiovascular event, whichever occurred first. Each patient's level of adherence to the treatment was measured by means of the internal clock of the CPAP device, dividing the hours of CPAP use by the days of treatment. Good compliance was defined as the use of the CPAP device for an average of ≥4 h per day.
Calculation of the oximetry-based HB
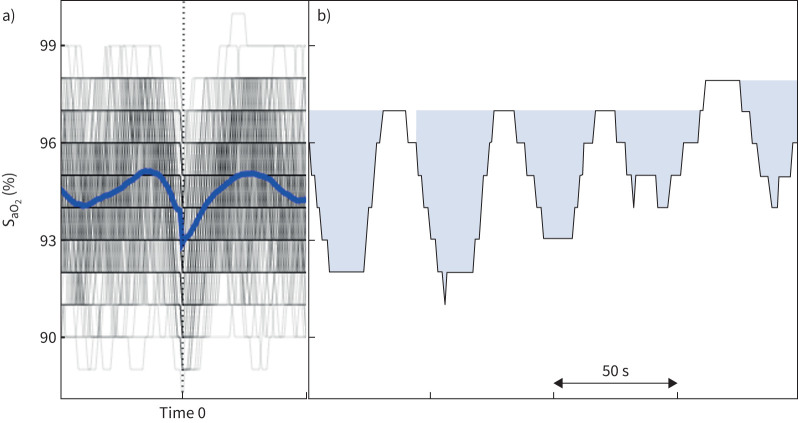
The original HB calculation method has been modified to calculate the HB using automatically identified desaturations from pulse oximetry, regardless of scored events. In the Sleep Heart Health Study (SHHS), this method has been proven to have a strong correlation with the HB obtained through manually scored apnoeas and hypopnoeas, and was significantly associated with CVD mortality, hypertension and excessive daytime sleepiness with similar effect sizes [31]. Similar to our published algorithm, all oxygen desaturation levels exceeding a 2% drop were automatically identified from the SaO2 signal. The identified desaturations were then synchronised with respect to the minimum oxygen saturation and were ensemble averaged to obtain the individual-specific search window, defined as the time interval between the two peaks around the minimum value of the ensemble average signal (figure 1). HB was then determined by summing all the individual areas under the desaturation curve restricted by the search window and dividing by the total sleep time [31].
FIGURE 1.
Calculation of the oximetry-based hypoxic burden (HB). a) All automatically detected desaturations are synchronised at their minimum saturation levels (time 0) and ensemble averaged to obtain the search window (duration between two maximum peaks around time 0). b) HB is calculated as the normalised summation of the coloured arterial oxygen saturation (SaO2) areas within the search window for all desaturation episodes divided by total sleep time.
Statistical analysis
Patients were categorised according to the baseline HB level as the “low HB group” or “high HB group” when the HB was below or above the median value of the population (≤73.1%min·h−1 or >73.1%min·h−1, respectively). To test the primary hypothesis that the effect of CPAP treatment on the primary outcome is moderated by the baseline HB level (i.e. that the effect of CPAP differs between those patients with high HB and those with low HB at baseline), a Cox regression model was used to assess the interaction between treatment arm and HB category (high HB×CPAP, with high HB being above the median value). Kaplan–Meier survival curves were plotted to illustrate incident CVD per treatment arm in the overall sample and in the subgroups with low or high HB. Log-rank tests were used to assess differences between survival curves. Additional multivariable Cox regression analyses were performed after adjusting for confounders (age, sex, body mass index and presence of CVD prior to admission to the study). For comparison, these analyses were repeated for the AHI, ODI and TSat90. Additionally, in an exploratory analysis, the CPAP group was divided into two subgroups based on median adherence to CPAP (“lower” or “higher” adherence subgroups). In the overall sample as well as in the low and high HB groups, the Kaplan–Meier survival curves compared the incidence of CVD of lower and higher adherence subgroups with that of the usual care group. Finally, in another exploratory analysis, to assess the dose–response relationship between HB and incident CVD in each treatment arm, HB was modelled continuously and the risk of incident CVD was predicted (relative to the average baseline characteristics of the OSA patients in the ISAACC trial). All statistical analyses were conducted using the R statistical package (www.r-project.org) and statistical significance was accepted at p<0.05 for the primary analysis.
Results
Description of the study cohort
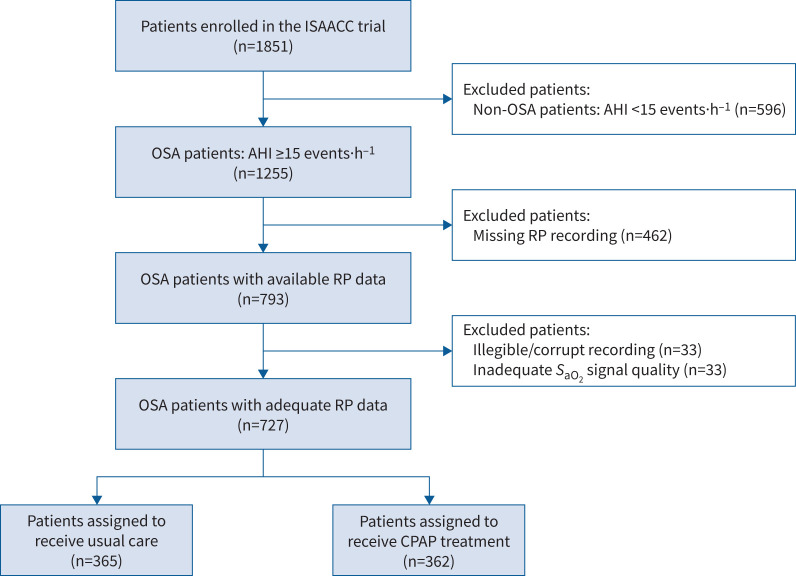
Of the 1255 patients with OSA enrolled in the ISAACC trial, all patients with available respiratory polygraphy data were included in this post hoc analysis. After excluding 33 patients due to irregularities in the sleep recording and 33 due to inadequate SaO2 signal quality, the final study cohort comprised 727 OSA patients (figure 2). No clinically relevant differences were observed between the included and excluded patients regarding baseline clinical and sleep characteristics, percentage of individuals allocated to each treatment arm or incidence of outcomes during the follow-up (supplementary table S1).
FIGURE 2.
Study flowchart. Of the 1851 participants enrolled in the ISAACC trial, 1255 patients with obstructive sleep apnoea (OSA) were considered for the present post hoc analysis. Respiratory polygraphy (RP) data were available for a total of 793 patients, of whom 33 had irregularities in the sleep recording and 33 involved inadequate arterial oxygen saturation (SaO2) signal quality. The final study cohort comprised 727 OSA patients. AHI: apnoea–hypopnoea index; CPAP: continuous positive airway pressure.
The analytic sample comprised individuals who were mainly middle-aged (mean age 59.7 years) and overweight/obese, and most participants were male (84.5%). Baseline characteristics of the study cohort by categories of HB and treatment allocation are summarised in table 1. A total of 365 individuals were assigned to the usual care arm and 362 individuals were assigned to the CPAP treatment arm. The treatment groups were similar in terms of sociodemographic/anthropometric characteristics, unhealthy life habits, comorbidities, prescribed medications and sleep parameters, including HB (supplementary table S2).
TABLE 1.
Baseline characteristics of the study cohort by categories of hypoxic burden (HB) and treatment allocation
| Usual care | CPAP treatment | |||
| Low HB (n=187) | High HB (n=178) | Low HB (n=177) | High HB (n=185) | |
| Clinical data | ||||
| Demographic/anthropometric | ||||
| Age (years) | 60.0 (53.0–66.0) | 59.0 (53.0–68.0) | 60.0 (53.0–67.0) | 58.0 (52.0–65.0) |
| Sex | ||||
| Male | 161 (86.1) | 151 (84.8) | 143 (80.8) | 159 (85.9) |
| Female | 26 (13.9) | 27 (15.2) | 34 (19.2) | 26 (14.1) |
| BMI (kg·m−2) | 28.3 (25.7–30.8) | 30.1 (27.9–32.8) | 27.8 (25.5–30.8) | 29.4 (26.8–33.1) |
| Smoking status | ||||
| Never | 46 (24.6) | 40 (22.5) | 41 (23.2) | 46 (24.9) |
| Former | 50 (26.7) | 53 (29.8) | 45 (25.4) | 47 (25.4) |
| Current | 91 (48.7) | 85 (47.8) | 91 (51.4) | 92 (49.7) |
| Drinking status | ||||
| Never | 142 (78.9) | 132 (75.9) | 124 (71.3) | 138 (76.2) |
| Former | 2 (1.11) | 2 (1.15) | 1 (0.57) | 1 (0.55) |
| Current | 36 (20.0) | 40 (23.0) | 49 (28.2) | 42 (23.2) |
| Comorbidities | ||||
| Diabetes | 38 (20.3) | 50 (28.1) | 52 (29.4) | 54 (29.2) |
| Hypertension | 95 (50.8) | 111 (62.4) | 97 (54.8) | 105 (56.8) |
| Dyslipidaemia | 104 (55.6) | 101 (56.7) | 110 (62.1) | 109 (58.9) |
| Chronic pneumopathy | 13 (6.95) | 14 (7.87) | 11 (6.21) | 10 (5.41) |
| Previous CVD | 29 (15.5) | 42 (23.6) | 33 (18.6) | 37 (20.0) |
| Medication use | ||||
| Insulin | 7 (3.74) | 11 (6.18) | 6 (3.39) | 13 (7.03) |
| Oral antidiabetic drugs | 30 (16.0) | 42 (23.6) | 37 (20.9) | 48 (25.9) |
| ACE inhibitors | 41 (21.9) | 47 (26.4) | 40 (22.6) | 38 (20.5) |
| β-blockers | 37 (19.8) | 37 (20.8) | 28 (15.8) | 40 (21.6) |
| Diuretic agents | 30 (16.0) | 38 (21.3) | 35 (19.8) | 34 (18.4) |
| Calcium-channel blockers | 20 (10.7) | 36 (20.2) | 23 (13.0) | 22 (11.9) |
| Angiotensin II receptor blockers | 24 (12.8) | 30 (16.9) | 28 (15.8) | 35 (18.9) |
| Lipid-lowering drugs | 60 (32.1) | 69 (38.8) | 67 (37.9) | 78 (42.2) |
| Antiplatelet drugs | 39 (20.9) | 43 (24.2) | 40 (22.6) | 42 (22.7) |
| Anticoagulants | 9 (4.81) | 8 (4.49) | 7 (3.95) | 8 (4.32) |
| Sleep data | ||||
| AHI (events·h−1) | 22.0 (17.2–33.0) | 42.0 (32.0–55.5) | 23.0 (18.2–29.4) | 44.0 (30.0–59.0) |
| ODI (events·h−1) | 19.1 (14.4–26.7) | 41.2 (32.6–54.3) | 18.0 (12.8–24.6) | 37.7 (25.6–52.6) |
| Mean SaO2 (%) | 93.7 (92.3–94.9) | 92.4 (90.8–93.6) | 93.3 (92.0–94.4) | 92.7 (91.3–93.6) |
| Minimum SaO2 (%) | 87.0 (82.0–88.0) | 82.0 (76.0–85.0) | 85.0 (81.0–88.0) | 82.0 (78.0–85.0) |
| TSat90 (%) | 0.75 (0.10–3.65) | 8.95 (2.68–27.3) | 1.30 (0.20–6.40) | 6.60 (1.60–22.0) |
| HB (%min·h−1) | 50.6 (40.6–61.2) | 106 (85.0–142) | 53.3 (43.3–63.1) | 102 (84.4–139) |
| Somnolence (ESS) | 4.00 (3.00–6.00) | 5.00 (4.00–8.00) | 5.00 (3.00–6.00) | 5.00 (3.00–8.00) |
Data are presented as median (interquartile range) or n (%). CPAP: continuous positive airway pressure; BMI: body mass index; CVD: cardiovascular disease; ACE: angiotensin-converting enzyme; AHI: apnoea–hypopnoea index; ODI: oxygen desaturation index; SaO2: arterial oxygen saturation; TSat90: percentage of total sleep time with SaO2 <90%; ESS: Epworth Sleepiness Scale. The pre-intervention HB was categorised as “low” or “high” based on the median value (≤73.1%min·h−1 or >73.1%min·h−1, respectively).
As expected, patients in the high HB groups presented with more severe OSA than those in the low HB groups, reflected in greater values of AHI and the other respiratory polygraphy parameters. Likewise, patients with high HB values were more obese than those with low HB values (supplementary table S3). Among CPAP-treated individuals, 32.0% were good compliers in the low HB group, while 44.1% were good compliers in the high HB group (p=0.025).
Differential effect of CPAP treatment on cardiovascular prevention according to the baseline HB level
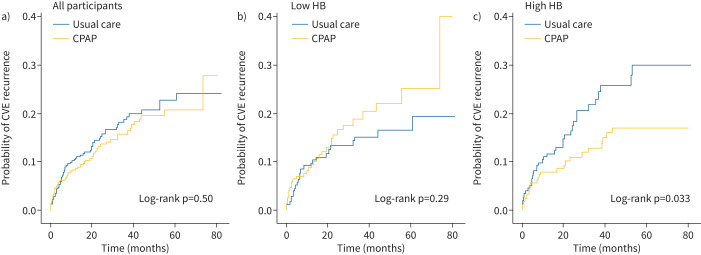
Over a mean±sd follow-up of 32.31±22.46 months, a total of 117 OSA subjects experienced a major adverse cardiovascular event. A significant interaction was found between CPAP treatment and the HB categories (p=0.023), indicating a differential effect of the treatment depending on the baseline HB level. The result was maintained after adjustment for confounding factors (p=0.035). Figure 3 and table 2 compare the impact of CPAP treatment versus usual care on the incidence of the primary end-point by categories of pre-intervention HB. While there was no association of CPAP therapy with cardiovascular prevention in the entire cohort, substantial heterogeneity in the effect of the treatment was observed when the population was stratified based on the HB. In the high HB group, there was a significant CPAP-related reduction in the incidence of adverse cardiovascular events during the follow-up (HR 0.57, 95% CI 0.34–0.96; p=0.035). Conversely, in the low HB category, CPAP-treated patients exhibited a trend toward a higher risk of cardiovascular outcomes during the follow-up than those receiving usual care (HR 1.33, 95% CI 0.79–2.25; p=0.281). The magnitude of the effect size was maintained after adjustment for confounders.
FIGURE 3.
Differential patterns of cardiovascular response to continuous positive airway pressure (CPAP) treatment according to the baseline hypoxic burden (HB) level. Unadjusted Kaplan‒Meier curves showing the cumulative incidence of cardiovascular events (CVEs) in the CPAP versus usual care groups for a) all participants (n=727), b) participants with low HB (n=364) and c) participants with high HB (n=363). The pre-intervention HB was categorised as “low” or “high” based on the median value (≤73.1%min·h−1 or >73.1%min·h−1, respectively).
TABLE 2.
Cox regression models evaluating the effect of continuous positive airway pressure (CPAP) treatment on the primary end-point according to the baseline hypoxic burden (HB) level
| Events | Follow-up (months) |
Unadjusted HR
(95% CI) |
p-value |
Adjusted# HR
(95% CI) |
p-value | |
| Low HB | ||||||
| Usual care | 26 (13.9) | 33.34 (15.03–54.04) | Reference | Reference | ||
| CPAP | 31 (17.4) | 24.34 (13.80–43.37) | 1.33 (0.79–2.25) | 0.281 | 1.31 (0.78–2.22) | 0.308 |
| High HB | ||||||
| Usual care | 36 (20.3) | 24.26 (12.27–41.86) | Reference | Reference | ||
| CPAP | 24 (13.0) | 32.00 (13.27–58.22) | 0.57 (0.34–0.96) | 0.035 | 0.61 (0.36–1.03) | 0.063 |
Data are presented as n (%) or median (interquartile range), unless otherwise stated. The pre-intervention HB was categorised as “low” or “high” based on the median value (≤73.1%min·h−1 or >73.1%min·h−1, respectively). Usual care and low baseline HB are the reference categories for the CPAP and high HB groups, respectively. #: adjustment for age, sex, body mass index and previous cardiovascular disease. Significant p-values (p<0.05) are presented in bold.
Furthermore, we evaluated the dose–response relationship between the baseline HB as a continuous variable and the incidence of cardiovascular events during the follow-up, according to the treatment arm (figure 4). Participants who received CPAP treatment had a reduced cardiovascular risk as the HB level increased. The opposite trend was observed in the usual care group, where the risk for cardiovascular events increased as the HB level increased.
FIGURE 4.
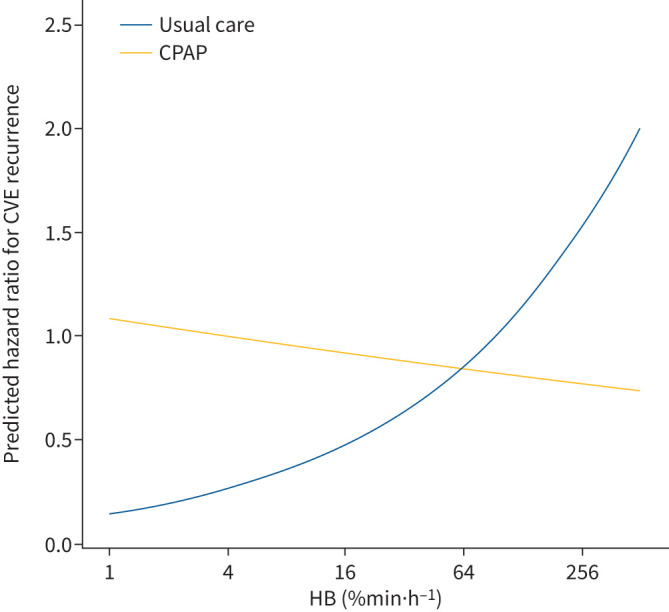
Dose–response relationship between baseline hypoxic burden (HB) and predicted risk for cardiovascular events (CVEs) stratified by treatment arm. The curves illustrate the predicted hazard for the primary end-point in the continuous positive airway pressure (CPAP) and the usual care groups, according to the continuous HB level. The predicted hazard ratios were estimated using the average baseline characteristics: average age (59.7 years), male sex (majority), average body mass index (29.3 kg·m−2) and absence of previous cardiovascular disease (majority). The x-axis is log-scaled.
Considering the higher values of CPAP compliance in the high HB group, we additionally explored the potential association between adherence to CPAP treatment and cardiovascular prognosis. In the low HB group, participants with higher adherence to CPAP treatment exhibited a non-significant higher cardiovascular risk, whereas in the high HB group, both adherence groups showed the same trend of protection against cardiovascular events compared with the usual care group (supplementary figure S1).
Notably, we did not find an association between CPAP treatment and cardiovascular prevention according to the baseline levels of AHI or other conventional OSA severity metrics, such as the ODI or TSat90 (supplementary figure S2).
Secondary analyses were used to assess the effect of CPAP on the cardiac components of the primary end-point (i.e. cardiovascular death, non-fatal acute myocardial infarction and new hospitalisation for unstable angina) in the low and high HB groups (table 3). As with the primary outcome, there was a significant interaction between the treatment arm and the HB categories (p=0.025). CPAP therapy showed a stronger protective effect against cardiac events only in participants with high HB (HR 0.52, 95% CI 0.29–0.92; p=0.025). This result did not change after adjustment for covariates.
TABLE 3.
Cox regression models evaluating the effect of continuous positive airway pressure (CPAP) treatment on the cardiac end-point according to the baseline hypoxic burden (HB) level
| Events | Follow-up (months) |
Unadjusted HR
(95% CI) |
p-value |
Adjusted# HR
(95% CI) |
p-value | |
| Low HB | ||||||
| Usual care | 18 (9.6) | 34.91 (16.78–55.95) | Reference | Reference | ||
| CPAP | 22 (12.4) | 25.96 (14.32–47.57) | 1.36 (0.73–2.54) | 0.331 | 1.36 (0.72–2.55) | 0.339 |
| High HB | ||||||
| Usual care | 31 (17.4) | 24.6 (12.85–47.04) | Reference | Reference | ||
| CPAP | 19 (10.3) | 34.29 (17.50–58.45) | 0.52 (0.29–0.92) | 0.025 | 0.53 (0.30–0.95) | 0.034 |
Data are presented as n (%) or median (interquartile range), unless otherwise stated. The pre-intervention HB was categorised as “low” or “high” based on the median value (≤73.1%min·h−1 or >73.1%min·h−1, respectively). Usual care and low baseline HB are the reference categories for the CPAP and high HB groups, respectively. #: adjustment for age, sex, body mass index and previous cardiovascular disease. Significant p-values (p<0.05) are presented in bold.
Discussion
In the present post hoc analysis of the ISAACC trial, we report that in non-sleepy patients with ACS, there is a differential pattern of response to OSA treatment with CPAP depending on the baseline HB level. We observed that OSA patients with a high baseline HB not only had an increased risk for developing future adverse cardiovascular outcomes but also exhibited a significant long-term protective effect of CPAP on cardiovascular prognosis compared with those with a low baseline HB. These results suggest that this novel OSA severity metric could be useful to identify those patients who are most likely to experience a reduction in cardiovascular risk following treatment with CPAP.
The potential benefits of successfully treating OSA range from improving clinical symptomatology and reducing its associated morbidity and mortality to decreasing healthcare resource utilisation and consequent costs. CPAP is currently the mainstay of therapy for adults with OSA. There is high-quality evidence indicating that CPAP therapy reduces the frequency of respiratory events during sleep, decreases daytime sleepiness [32], improves systemic blood pressure [33], lowers the risk of traffic accidents [34] and improves quality of life [35, 36] across a range of disease severities. In contrast, RCTs have been unsuccessful in demonstrating a positive effect of treating OSA with CPAP on primary and secondary cardiovascular outcomes [37]. Several authors have discussed potential explanations that may shed light on these conflicting results [38, 39]. A substantial line of reasoning is based on questioning the use of the AHI as the “holy grail” to assess OSA [40]. Despite the standard use of this metric as the primary diagnostic, prognostic and treatment-deciding criterion for OSA [41, 42], available literature indicates that the AHI is an oversimplification of a complex and diverse disease phenomenon [40]. In recent years, the sleep medicine community has pointed to the need for a more tailored approach to optimise the management of this heterogeneous disease.
In the era of precision medicine, it has been a research priority to develop additional markers that fully capture the impact of OSA on different health outcomes, especially those that accurately foresee cardiovascular risk. A considerable number of alternatives have been proposed, where HB emerges as a physiologically informed metric that captures the depth, duration and frequency of night-time respiratory event-related hypoxaemia. Since its discovery [23], an increasing number of studies have demonstrated the capacity of HB for predicting CVD-related outcomes, including mortality [28], major cardiovascular events [23], stroke [25], heart failure [24], hypertension [26] and chronic kidney disease [27]. Nevertheless, to the best of our knowledge, there has been no study to date evaluating the potential of this OSA severity metric to predict the therapeutic benefit of CPAP [43]. In this secondary analysis of the ISAACC trial, we observed that the effect of OSA intervention with CPAP on the incidence of adverse cardiovascular outcomes was dependent on the baseline HB level. The cardiovascular response to CPAP treatment followed a dose–response relationship with the baseline HB levels and only those patients with a high HB exhibited a protective effect of the treatment, regardless of the level of adherence. In contrast, this protection conferred by CPAP against the recurrence of CVD was not evidenced by stratifying the population by the AHI or other metrics of nocturnal hypoxaemia, including the ODI and TSat90. Our findings reinforce the need to move beyond the AHI and conventional OSA severity metrics to fully characterise the disease.
Due to the large heterogeneity of this disease, it is increasingly recognised that the effect of CPAP on different health outcomes may not be uniform across the entire spectrum of the OSA population. The reliance on a single metric that does not capture this heterogeneity may prove detrimental to our therapeutic efforts as inadequate treatment allocation may result in inefficient utilisation of healthcare resources. Therefore, the accurate identification of individuals who are most likely to respond to CPAP treatment represents a hot topic in the field. This would be especially interesting for the large proportion of patients with sleep disordered breathing not reporting daytime sleepiness [44], for whom the prescription of CPAP as the treatment of choice remains a subject of great debate [45, 46]. This study provides further evidence of the existence of a specific subpopulation of patients who may be impacted differently by OSA and consequently have a different response to treatment. Our results facilitate a step toward personalised, guided therapeutic decision making in OSA. This could reduce healthcare costs by avoiding the administration of treatment that is unnecessary or ineffective for individuals with little probability of response, especially considering the tendency toward a harmful effect of CPAP observed in patients with low HB. Furthermore, the HB could not only help to capture the variability in OSA and guide CPAP treatment allocation but also inform enrolment criteria for the design of future RCTs.
For a novel disease metric to be useful, it should be easily incorporated into routine clinical practice. In this study, we used a validated new approach to calculate the HB based on automatically identified oxygen desaturations [31]. This approach eliminates the need for manual scoring of respiratory events, which is time consuming and prone to inter-scorer technical variability, which is a reported cause of inconsistencies in the calculation of conventional OSA indices among different sleep laboratories [47]. Other metrics have arisen in recent years. In particular, it has been shown that individuals with greater respiratory event-related pulse rate response (ΔHR) were at increased risk of cardiovascular morbidity and mortality [47]. Additionally, this metric was demonstrated to predict CPAP benefit in the RICCADSA trial [48]. Here, the original manual scoring of respiratory events was unavailable for some subjects and therefore we were unable to compute the ΔHR. Nevertheless, the simplicity of our novel HB measurement enables its calculation using wearable technology that only records the SaO2 signal, facilitating its inclusion as part of a clinical support system for OSA, not only to identify at-risk patients but also for targeted therapy.
It has been proposed that OSA may have a different impact on the cerebrovascular system than on the coronary system [48], and therefore the weights of the cardiac and cerebrovascular components of the primary composite end-point could not be equally balanced. Based on this assumption, we additionally tested the prognostic value of HB focusing exclusively on the cardiac component of the primary end-point. This analysis yielded stronger associations between HB and the effect of CPAP treatment, with a calculated risk reduction of 47% in patients with high HB.
Strengths and limitations
The strengths of this study include its multicentre design, which enabled us to appropriately address the association of HB with the impact of CPAP treatment on long-term cardiovascular outcomes by evaluating a large number of patients with confirmed diagnoses of ACS and OSA. Standardised methods were applied for data collection across the participating centres. All sleep studies were performed with the same respiratory polygraphy model and were scored by certified technicians following international guidelines. Moreover, major adverse cardiovascular outcomes specified in the study protocol were documented and adjudicated by a blinded external committee.
Our study has several limitations that should be noted. First, although a causal relation of OSA to vascular disease has strong biological plausibility, due to the exploratory nature of this secondary analysis of an RCT, cause–effect relationships remain unproven. The results of this study indicate the possibility of identifying specific subgroups of OSA patients who could respond to CPAP treatment based on an oximetry-based metric. However, the validity of our findings should be further confirmed in independent cohorts. Second, the unavailability of raw respiratory polygraphy data for all OSA patients in the ISAACC cohort may pose a potential limitation. Although no clinically relevant differences were observed between the included and excluded patients, this aspect could have introduced bias into the results. Third, although the method for HB calculation applied here has been validated in the SHHS, the original approach for calculating HB could not be applied in this cohort due to the unavailability of all the original manually scored event files. Fourth, for ethical reasons, patients with excessive daytime sleepiness were excluded from the ISAACC trial. Therefore, the results should not be extrapolated to OSA patients with somnolence. Fifth, the study design is limited to patients admitted to the hospital for ACS, making it necessary to explore the clinical value of HB in different clinical settings. Sixth, due to insufficient statistical power to individually evaluate the cerebrovascular composite end-point, further research is needed to determine the potential of HB to predict the effect of CPAP on cerebrovascular consequences. Finally, the under-representation of women and subjects with other ethnicities may limit the generalisability of the results to the global population. Future studies should confirm whether the results are valid for women and individuals with other racial backgrounds.
Conclusions
In patients with ACS, OSA treatment with CPAP prevented CVD-related events in patients with a high baseline HB. Although further validation work is needed, our exploratory findings highlight the potential for this oximetry-based metric to identify OSA patients without excessive daytime sleepiness who are more likely to respond to CPAP therapy in the long term. The HB represents a simple, inexpensive and easily accessible metric that could feasibly be translated into routine clinical practice to guide CPAP treatment allocation for cardiovascular prevention in asymptomatic OSA.
Supplementary material
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary material ERJ-00828-2023.Supplement (558.3KB, pdf)
Shareable PDF
Acknowledgements
We thank the individuals who participated in this trial and their families, the clinical and research teams of the various sleep and cardiology departments, and the Spanish Sleep Network for their work on the ISAACC study.
Footnotes
This article has an editorial commentary: https://doi.org/10.1183/13993003.01741-2023
Ethics approval: The clinical research ethics committee of each participating centre approved the trial protocol (approval number in the coordinating centre: 2010/852).
Conflict of interest: M.Á. Martinez-Garcia received grants from VitalAire and Philips, Spain, and serves as a consultant for ResMed Inc., Australia, companies that develop products related to sleep apnoea. L. Messineo received a consultancy fee from Apnimed. A. Wellman works as a consultant for Apnimed, SomniFix and Nox, and has received grants from SomniFix and Sanofi; A. Wellman has a financial interest in Apnimed, a company developing pharmacological therapies for sleep apnoea. His interests were reviewed and are managed by Brigham and Women's Hospital and Partners HealthCare in accordance with their conflict of interest policies. S. Redline received grant support and consulting fees from Jazz Pharmaceuticals, and consulting fees from Apnimed and Lilly Pharma. S. Sands receives personal fees as a consultant for Nox Medical and Merck, outside the submitted work, and receives grant support from Apnimed and ProSomnus. F. Barbé received a research grant from ResMed Inc., Australia, a company that develops products related to sleep apnoea. A. Azarbarzin reports grant support from SomniFix and serves as a consultant for SomniFix, Respicardia, Eli Lilly and Apnimed. Apnimed is developing pharmacological treatments for obstructive sleep apnoea. A. Azarbarzin's interests were reviewed by Brigham and Women's Hospital and Mass General Brigham in accordance with their institutional policies. All other authors declare no competing interests.
Support statement: The ISAACC trial was funded by Instituto de Salud Carlos III (PI10/02763, PI10/02745, PI18/00449 and PI22/00636), co-funded by the European Union, Spanish Respiratory Society (SEPAR), Catalonian Cardiology Society, ResMed Ltd (Australia), EsteveTeijin (Spain), Oxigen Salud (Spain), and ALLER and CIBERES. The funding sources did not have any participation or decision in the study development. This research was also supported by the American Academy of Sleep Medicine Foundation (188-SR-17, SR-2217), American Heart Association (19CDA34660137) and NHLBI (R01HL153874 and R35HL135818). L. Pinilla has received financial support from the Ministry of Universities of Spain (Formación de Profesorado Universitario: FPU19/01555; Ayudas para Estancias Breves: EST22/00259), Sociedad Española de Neumología y Cirugía Torácica (SEPAR), Fundació Catalana de Pneumologia (FUCAP), CIBERES and IRBLleida. M. Sánchez-de-la-Torre has received financial support from Ministerio de Ciencia e Innovación – Agencia Estatal de Investigación (Ramón y Cajal: RYC2019-027831-I). Funding information for this article has been deposited with the Crossref Funder Registry.
References
- 1.Benjafield AV, Ayas NT, Eastwood PR, et al. Estimation of the global prevalence and burden of obstructive sleep apnoea: a literature-based analysis. Lancet Respir Med 2019; 7: 687–698. doi: 10.1016/S2213-2600(19)30198-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Javaheri S, Barbe F, Campos-Rodriguez F, et al. Sleep apnea: types, mechanisms, and clinical cardiovascular consequences. J Am Coll Cardiol 2017; 69: 841–858. doi: 10.1016/j.jacc.2016.11.069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sánchez-de-la-Torre M, Campos-Rodriguez F, Barbé F. Obstructive sleep apnoea and cardiovascular disease. Lancet Respir Med 2013; 1: 61–72. doi: 10.1016/S2213-2600(12)70051-6 [DOI] [PubMed] [Google Scholar]
- 4.Gottlieb DJ, Yenokyan G, Newman AB, et al. Prospective study of obstructive sleep apnea and incident coronary heart disease and heart failure: the Sleep Heart Health Study. Circulation 2010; 122: 352–360. doi: 10.1161/CIRCULATIONAHA.109.901801 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wu X, Lv S, Yu X, et al. Treatment of OSA reduces the risk of repeat revascularization after percutaneous coronary intervention. Chest 2015; 147: 708–718. doi: 10.1378/chest.14-1634 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Marin JM, Carrizo SJ, Vicente E, et al. Long-term cardiovascular outcomes in men with obstructive sleep apnoea–hypopnoea with or without treatment with continuous positive airway pressure: an observational study. Lancet 2005; 365: 1046–1053. doi: 10.1016/S0140-6736(05)71141-7 [DOI] [PubMed] [Google Scholar]
- 7.Punjabi NM, Caffo BS, Goodwin JL, et al. Sleep-disordered breathing and mortality: a prospective cohort study. PLoS Med 2009; 6: e1000132. doi: 10.1371/journal.pmed.1000132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Peppard PE, Young T, Palta M, et al. Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med 2000; 342: 1378–1384. doi: 10.1056/NEJM200005113421901 [DOI] [PubMed] [Google Scholar]
- 9.Campos-Rodriguez F, Martinez-Garcia MA, de la Cruz-Moron I, et al. Cardiovascular mortality in women with obstructive sleep apnea with or without continuous positive airway pressure treatment: a cohort study. Ann Intern Med 2012; 156: 115–122. doi: 10.7326/0003-4819-156-2-201201170-00006 [DOI] [PubMed] [Google Scholar]
- 10.Nieto FJ, Young TB, Lind BK, et al. Association of sleep-disordered breathing, sleep apnea, and hypertension in a large community-based study. J Am Coll Cardiol 2000; 283: 1829–1836. doi: 10.1001/jama.283.14.1829 [DOI] [PubMed] [Google Scholar]
- 11.Bratton DJ, Gaisl T, Wons AM, et al. CPAP vs mandibular advancement devices and blood pressure in patients with obstructive sleep apnea a systematic review and meta-analysis. JAMA 2015; 314: 2280–2293. doi: 10.1001/jama.2015.16303 [DOI] [PubMed] [Google Scholar]
- 12.Baessler A, Nadeem R, Harvey M, et al. Treatment for sleep apnea by continuous positive airway pressure improves levels of inflammatory markers – a meta-analysis. J Inflamm 2013; 10: 13. doi: 10.1186/1476-9255-10-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schwarz EI, Puhan MA, Schlatzer C, et al. Effect of CPAP therapy on endothelial function in obstructive sleep apnoea: a systematic review and meta-analysis. Respirology 2015; 20: 889–895. doi: 10.1111/resp.12573 [DOI] [PubMed] [Google Scholar]
- 14.Drager LF, Bortolotto LA, Figueiredo AC, et al. Effects of continuous positive airway pressure on early signs of atherosclerosis in obstructive sleep apnea. Am J Respir Crit Care Med 2007; 176: 706–712. doi: 10.1164/rccm.200703-500OC [DOI] [PubMed] [Google Scholar]
- 15.Cowie MR, Linz D, Redline S, et al. Sleep disordered breathing and cardiovascular disease: JACC State-of-the-Art Review. J Am Coll Cardiol 2021; 78: 608–624. doi: 10.1016/j.jacc.2021.05.048 [DOI] [PubMed] [Google Scholar]
- 16.Dissanayake HU, Colpani JT, Sutherland K, et al. Obstructive sleep apnea therapy for cardiovascular risk reduction – time for a rethink? Clin Cardiol 2021; 44: 1729–1738. doi: 10.1002/clc.23747 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sánchez-de-la-Torre M, Sánchez-de-la-Torre A, Bertran S, et al. Effect of obstructive sleep apnoea and its treatment with continuous positive airway pressure on the prevalence of cardiovascular events in patients with acute coronary syndrome (ISAACC study): a randomised controlled trial. Lancet Respir Med 2020; 8: 359–367. doi: 10.1016/S2213-2600(19)30271-1 [DOI] [PubMed] [Google Scholar]
- 18.McEvoy RD, Antic NA, Heeley E, et al. CPAP for prevention of cardiovascular events in obstructive sleep apnea. N Engl J Med 2016; 375: 919–931. doi: 10.1056/NEJMoa1606599 [DOI] [PubMed] [Google Scholar]
- 19.Peker Y, Glantz H, Eulenburg C, et al. Effect of positive airway pressure on cardiovascular outcomes in coronary artery disease patients with nonsleepy obstructive sleep apnea: the RICCADSA randomized controlled trial. Am J Respir Crit Care Med 2016; 194: 613–620. doi: 10.1164/rccm.201601-0088OC [DOI] [PubMed] [Google Scholar]
- 20.Zinchuk A, Klar Yaggi H. Sleep apnea heterogeneity, phenotypes, and cardiovascular risk implications for trial design and precision sleep medicine. Am J Respir Crit Care Med 2019; 200: 412–413. doi: 10.1164/rccm.201903-0545ED [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pevernagie DA, Gnidovec-Strazisar B, Grote L, et al. On the rise and fall of the apnea–hypopnea index: a historical review and critical appraisal. J Sleep Res 2020; 29: e13066. doi: 10.1111/jsr.13066 [DOI] [PubMed] [Google Scholar]
- 22.Malhotra A, Ayappa I, Ayas N, et al. Metrics of sleep apnea severity: beyond the apnea–hypopnea index. Sleep 2021; 44: zsab030. doi: 10.1093/sleep/zsab072.072 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Azarbarzin A, Sands SA, Stone KL, et al. The hypoxic burden of sleep apnoea predicts cardiovascular disease-related mortality: the Osteoporotic Fractures in Men Study and the Sleep Heart Health Study. Eur Heart J 2019; 40: 1149–1157. doi: 10.1093/eurheartj/ehy624 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Azarbarzin A, Sands SA, Taranto-Montemurro L, et al. The sleep apnea-specific hypoxic burden predicts incident heart failure. Chest 2020; 158: 739–750. doi: 10.1016/j.chest.2020.03.053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Blanchard M, Gervès-Pinquié C, Feuilloy M, et al. Hypoxic burden and heart-rate variability predict stroke incidence in sleep apnoea. Eur Respir J 2020; 57: 2004022. doi: 10.1183/13993003.04022-2020 [DOI] [PubMed] [Google Scholar]
- 26.Kim JS, Azarbarzin A, Wang R, et al. Association of novel measures of sleep disturbances with blood pressure: the Multi-Ethnic Study of Atherosclerosis. Thorax 2020; 75: 57–63. doi: 10.1136/thoraxjnl-2019-213533 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jackson CL, Umesi C, Gaston SA, et al. Multiple, objectively measured sleep dimensions including hypoxic burden and chronic kidney disease: findings from the Multi-Ethnic Study of Atherosclerosis. Thorax 2021; 76: 704–713. doi: 10.1136/thoraxjnl-2020-214713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Trzepizur W, Blanchard M, Ganem T, et al. Sleep apnea-specific hypoxic burden, symptom subtypes, and risk of cardiovascular events and all-cause mortality. Am J Respir Crit Care Med 2022; 205: 108–117. doi: 10.1164/rccm.202105-1274OC [DOI] [PubMed] [Google Scholar]
- 29.Esquinas C, Sánchez-De-La Torre M, Aldomá A, et al. Rationale and methodology of the impact of continuous positive airway pressure on patients with ACS and nonsleepy OSA: the ISAACC trial. Clin Cardiol 2013; 36: 495–501. doi: 10.1002/clc.22166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Consenso Nacional sobre el Síndrome de Apneas–Hipopneas del Sueño (SAHS) . Resumen. [Summary.] Arch Bronconeumol 2005; 41: Suppl. 4, 7–9. doi: 10.1016/S0210-5705(09)71003-9 [DOI] [Google Scholar]
- 31.Esmaeili N, Labarca G, Hu WH, et al. Hypoxic burden based on automatically identified desaturations is associated with adverse health outcomes. Ann Am Thorac Soc 2023; 20: 1633–1641. doi: 10.1513/AnnalsATS.202303-248OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Marshall NS, Barnes M, Travier N, et al. Continuous positive airway pressure reduces daytime sleepiness in mild to moderate obstructive sleep apnoea: a meta-analysis. Thorax 2006; 61: 430–434. doi: 10.1136/thx.2005.050583 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Martínez-García MA, Capote F, Campos-Rodríguez F, et al. Effect of CPAP on blood pressure in patients with obstructive sleep apnea and resistant hypertension. JAMA 2013; 310: 2407. doi: 10.1001/jama.2013.281250 [DOI] [PubMed] [Google Scholar]
- 34.Tregear S, Reston J, Schoelles K, et al. Continuous positive airway pressure reduces risk of motor vehicle crash among drivers with obstructive sleep apnea: systematic review and meta-analysis. Sleep 2010; 33: 1373–1380. doi: 10.1093/sleep/33.10.1373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Siccoli MM, Pepperell JCT, Kohler M, et al. Effects of continuous positive airway pressure on quality of life in patients with moderate to severe obstructive sleep apnea: data from a randomized controlled trial. Sleep 2008; 31: 1551–1558. doi: 10.1093/sleep/31.11.1551 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Campos-Rodriguez F, Queipo-Corona C, Carmona-Berna C, et al. Continuous positive airway pressure improves quality of life in women with obstructive sleep apnea. A randomized controlled trial. Am J Respir Crit Care Med 2016; 194: 1286–1294. doi: 10.1164/rccm.201602-0265OC [DOI] [PubMed] [Google Scholar]
- 37.Labarca G, Dreyse J, Drake L, et al. Efficacy of continuous positive airway pressure (CPAP) in the prevention of cardiovascular events in patients with obstructive sleep apnea: systematic review and meta-analysis. Sleep Med Rev 2020; 52: 101312. doi: 10.1016/j.smrv.2020.101312 [DOI] [PubMed] [Google Scholar]
- 38.Pack AI, Magalang UJ, Singh B, et al. Randomized clinical trials of cardiovascular disease in obstructive sleep apnea: understanding and overcoming bias. Sleep 2021; 44: zsaa229. doi: 10.1093/sleep/zsaa229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Javaheri S, Martinez-Garcia MA, Campos-Rodriguez F. CPAP treatment and cardiovascular prevention: we need to change the design and implementation of our trials. Chest 2019; 156: 431–437. doi: 10.1016/j.chest.2019.04.092 [DOI] [PubMed] [Google Scholar]
- 40.Punjabi NM. COUNTERPOINT: is the apnea–hypopnea index the best way to quantify the severity of sleep-disordered breathing? No. Chest 2016; 149: 16–19. doi: 10.1378/chest.14-2261 [DOI] [PubMed] [Google Scholar]
- 41.Kapur VK, Auckley DH, Chowdhuri S, et al. Clinical practice guideline for diagnostic testing for adult obstructive sleep apnea: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med 2017; 13: 479–504. doi: 10.5664/jcsm.6506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mediano O, González Mangado N, Montserrat JM, et al. Documento internacional de consenso sobre apnea obstructiva del sueño. [International Consensus Document on Obstructive Sleep Apnea.] Arch Bronconeumol 2022; 58: 52–68. doi: 10.1016/j.arbres.2021.03.017 [DOI] [PubMed] [Google Scholar]
- 43.Martinez-Garcia MA, Sánchez-de-la-Torre M, White DP,et al. Hypoxic burden in obstructive sleep apnea: present and future. Arch Bronconeumol 2022; 59: 36–43. doi: 10.1016/j.arbres.2022.08.005 [DOI] [PubMed] [Google Scholar]
- 44.Kapur VK, Baldwin CM, Resnick HE,et al. Sleepiness in patients with moderate to severe sleep-disordered breathing. Sleep 2005; 28: 472–477. doi: 10.1093/sleep/28.4.472 [DOI] [PubMed] [Google Scholar]
- 45.Vakulin A, Chai-Coetzer CL, McEvoy RD. Con: should asymptomatic patients with moderate-to-severe OSA be treated? Breathe 2019. 15: 11–14. doi: 10.1183/20734735.0347-2018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ryan S. Pro: should asymptomatic patients with moderate-to-severe OSA be treated? Breathe 2019; 15: 7–10. doi: 10.1183/20734735.0340-2018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Collop NA. Scoring variability between polysomnography technologists in different sleep laboratories. Sleep Med 2002; 3: 43–47. doi: 10.1016/S1389-9457(01)00115-0 [DOI] [PubMed] [Google Scholar]
- 48.Freemantle N, Calvert M, Wood J,et al. Composite outcomes in randomized trials: greater precision but with greater uncertainty? JAMA 2003; 289: 2554–2559. doi: 10.1001/jama.289.19.2554 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary material ERJ-00828-2023.Supplement (558.3KB, pdf)
This one-page PDF can be shared freely online.
Shareable PDF ERJ-00828-2023.Shareable (594.1KB, pdf)