Abstract
In systemic lupus erythematosus (SLE), IL-2 production by T lymphocytes in vitro is impaired. Deficient IL-2 production may be an outcome of a primary SLE T cell disorder that is due to impaired signal transduction. In this issue of the JCI, evidence is presented that an anti-TCR/CD3 complex autoantibody present in SLE sera can bind to T cells and activate the Ca2+-calmodulin kinase IV (CaMKIV) signaling cascade, resulting in downregulation of IL-2 transcription and IL-2 production. Because IL-2 may contribute to the maintenance of T cell tolerance, deficient IL-2 production could promote a breach of T cell tolerance that results in autoantibody production in SLE.
Recently, it has been recognized that diverse autoantibodies directed against intra- and extracellular autoantigens exist in patients with systemic lupus erythematosus (SLE) for years before the clinical diagnosis is made (1); this suggests that physiologic mechanisms that maintain tolerance to self antigens have been breached. Tolerance to self antigens is established and preserved by a subpopulation of T lymphocytes known as Tregs (2), and the loss of tolerance is a pathologic process giving rise to autoimmunity. This circumstance raises the possibility of the existence of abnormal T cell clones that mediate defective helper and suppressor effector functions, which result in autoantibody generation by forbidden B cell clones. In SLE, defective signaling cascades are believed to give rise to a primary T cell disorder that is characterized by impaired effector functions (3). These effector dysfunctions are, at least in part, a result of skewed expression of various effector molecules, including CD40 ligand (e.g., CD154) and multiple cytokines, and may reflect an imbalance of gene expression. An extracellular factor(s) in the microenvironment that interacts with T cells and exacerbates these dysfunctions has not been previously identified.
Tregs, skewed cytokine production, and loss of tolerance
Impaired effector T cell functions due to skewed cytokine production may create a microenvironment that promotes a strong Th2 immune response relative to Th1 and Treg activity. Relative overproduction of IL-4, IL-6, and IL-10 by Th2 cells and underproduction of IL-2, IL-12, TGF-β, and IFN-γ by Th1 cells and Tregs can result in imbalanced autocrine and paracrine effects on T and B cells in the microenvironment. Because of the reduced numbers of CD4+CD25+ Tregs (4) as well as the diminished generation of IL-2 and TGF-β, there may be insufficient suppressor activity in SLE to counterbalance the enhanced Th2 effect on B cell antibody production. Taken together, these conditions create a microenvironment that promotes a dysregulated immune response driving both physiologic and forbidden B cell clones to overproduce antibodies and autoantibodies, which results in hypergammaglobulinemia. Moreover, these events occur despite the existence of other primary counterregulatory mechanisms, including expression of the cell surface molecule cytotoxic T lymphocyte antigen 4 (CTLA-4) (5).
IL-2 is a key cytokine that has been held to function predominantly as a growth factor. This cytokine is largely produced by activated CD4+ and CD8+ T cells and binds to high-affinity cell surface IL-2 receptors (IL-2Rs) expressed by T cells, B cells, NK cells, and APCs. However, current evidence from analyses of IL-2–/– and IL-2R–/– knockout mice supports the notion that IL-2 may operate, not as a principal growth factor in vivo, but as a third signal that stimulates clonal expansion of effector cells to promote tolerogenic responses and to regulate development and function of CD4+CD25+ Tregs and, possibly, CD8+ Tregs to maintain tolerance (6, 7). Although much less is known about the mechanisms of IL-2 function in humans, it seems reasonable to suppose that IL-2 may serve a parallel role in immune homeostasis.
Mechanisms of deficient IL-2 production by SLE T cells
Deficient IL-2 production may predispose individuals to impaired immunoregulation, loss of tolerance, and the development of SLE owing to the abrogation of suppressor mechanisms that maintain tolerance to self antigens. Two lines of evidence support this concept. First, it has been demonstrated that T cells from animal models of lupus as well as patients with SLE produce low amounts of IL-2 in vitro (8, 9). Second, vaccination of MRL/lpr lupus mice with live vaccinia recombinant viruses expressing the human IL-2 gene ameliorated disease activity (10). However, at the time that the IL-2 deficiency was discovered, the mechanisms leading to deficient IL-2 production by SLE T cells were unknown.
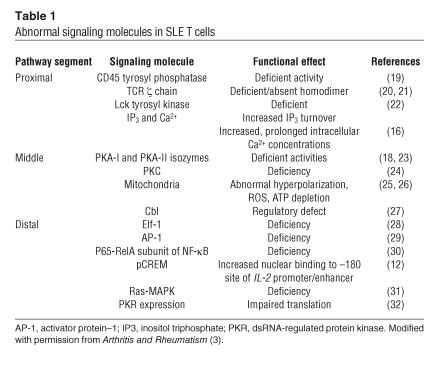
In SLE, a primary T cell disorder has been proposed to exist based on the identification of multiple discrete signaling abnormalities at the level of the TCR/CD3 complex, the cytosol, and the nucleus (3, 11) (summarized in Table 1). Tsokos, Kammer, and their colleagues first proposed that a primary failure of T cells due to defective signaling could hinder IL-2 gene transcription and IL-2 production and contribute to impaired T cell effector functions in SLE (12). To date, the data support this notion (13). Figure 1 presents a schematic of the mechanisms identified to date in SLE T cells that contribute to the downregula-tion of IL-2 production. However, extra-cellular factors in the microenviron-ment that impinge on the SLE T cell to further modify its immunoregulatory functions could also exist.
Table 1.
Abnormal signaling molecules in SLE T cells
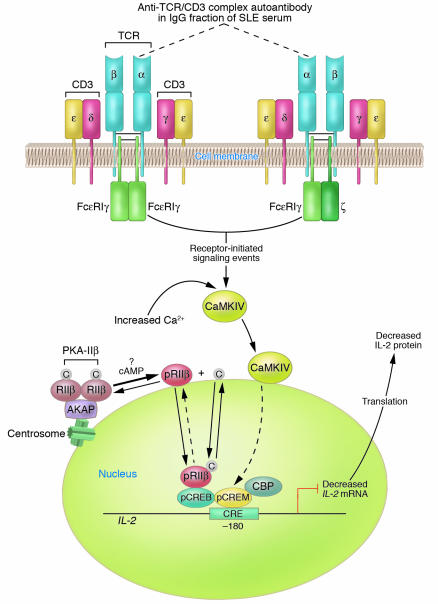
Figure 1.
Diagram of established and proposed mechanisms contributing to reduced IL-2 production by SLE T cells. A primary T cell disorder characterized by multiple abnormal signaling molecules has been identified in human SLE. A proportion of TCR ζ chains are replaced with FcεRIγ chains through which signaling can take place. In the microenvironment, anti-TCR/CD3 autoantibody(s) can bind to SLE T cells via the TCR/CD3 complex; however, the subunit(s) of the TCR/CD3 complex against which the autoantibody(s) is directed has not yet been identified. For the purpose of this illustration, the autoantibody is proposed to be against the α subunit of the TCR (dotted lines). Autoantibody binding to the TCR/CD3 complex results in heightened intracellular Ca2+ concentrations and activation of the CaMKIV signaling cascade. CaMKIV translocates to the nucleus, is proposed to phosphorylate CREM, and induces increased pCREM binding to the CRE located at the –180 region of the IL-2 promoter/enhancer. Because there is overexpression of the RIIβ in the T cell nuclei of some patients with SLE, it is proposed that PKA-IIβ is spontaneously activated by an unknown mechanism and that RIIβ becomes autophosphorylated; pRIIβ translocates to the nucleus, where it mediates downregulation of IL-2 transcription and IL-2 production. Together, pCREM and pRIIβ inhibit IL-2 transcription, thus yielding reduced IL-2 production. Note that proposed mechanisms have not been experimentally demonstrated. C, catalytic subunit; CBP, CREB binding protein.
IgG anti-TCR/CD3 autoantibodies and reduced IL-2 generation
In this issue of the JCI, Tsokos and colleagues present an incisive series of experiments that demonstrate that there is a factor in the microenvironment that can induce inhibition of IL-2 production by SLE T cells (14). Here, these authors show for what is believed to be the first time that the IgG fraction of SLE serum possesses an anti-TCR/CD3 complex autoantibody(s) that stimulates translocation of Ca2+-calmodulin kinase IV (CaMKIV) from the cytosol to the nucleus. This event induces upregulation of the CREM (cAMP response element [CRE] modulator) transcript and CREM protein, proposed phosphorylation of CREM (to pCREM), binding of pCREM homodimers as well as pCREM–phosphorylated CRE-binding protein (pCREB) heterodimers to the –180 region of the IL-2 promoter/enhancer, and leads to reduced IL-2 transcription and IL-2 production (Figure 1).
It has not been previously shown that an IgG autoantibody in SLE serum can activate a signaling cascade in T cells, although it is well known that serum factors can activate signaling pathways in diverse cell types. Indeed, we have previously found that SLE serum does not induce the defects of PKA signaling in normal T cells that have been identified in SLE T cells (15). To determine whether such an activating factor exists in SLE serum, Tsokos and his associates first investigated whether SLE sera could cause increased expression of nuclear pCREM relative to pCREB in normal T cells, just as had been observed in SLE T cells. Indeed, in normal T cells cultured in the presence of SLE sera, but not sera from patients with rheumatoid arthritis or from healthy controls, the authors observed increased levels of the CREM transcript, CREM protein, and pCREM binding to the –180 CRE region of the IL-2 promoter/enhancer. These findings revealed that SLE sera contain a factor capable of inducing nuclear CREM expression (14).
In subsequent experiments, the authors identified the protein kinase that mediates the overexpression of CREM. Based on their previous recognition that signals from the TCR/CD3 complex significantly stimulate the production of increased concentrations of intracellular Ca2+ in SLE T cells (16), they searched for a Ca2+-dependent kinase and found that SLE T cells exhibited increased amounts of nuclear CaMKIV compared to nuclear CaMKII in normal T cells. When normal T cells were then incubated with SLE or normal control sera, transiently transfected with a CaMKIV expression plasmid, and activated with a phorbol ester and ionomycin, T cells exposed to SLE sera exhibited significantly greater pCREM binding to the –180 region of the IL-2 promoter/enhancer (14). When these T cells were also transiently transfected with an IL-2 promoter–reporter luciferase construct, the cells incubated with SLE sera expressed significantly lower IL-2 promoter luciferase activity and IL-2 production compared with cells incubated in normal sera. Taken together, these results provide firm evidence that an extracellular factor in SLE sera can activate CaMKIV and induce its nuclear translocation from the cytosol, where it upregulates CREM expression, pCREM binding to the IL-2 promoter/enhancer, and inhibition of IL-2 transcription and IL-2 production.
To identify the SLE serum factor, the authors postulated that the TCR/CD3 complex is the target for a circulating IgG autoantibody(s) in SLE serum. They performed a subtraction experiment using wild-type and mutant Jurkat T cells that do not express the surface TCR/CD3 complexes. Absorption of the IgG autoantibody(s) by wild-type Jurkat T cells but not TCR/CD3– mutant Jurkat T cells eliminated the ligand(s) for the TCR/CD3 complex, reducing CaMKIV nuclear translocation and pCREM binding to the IL-2 promoter/enhancer (14). Although the authors did not perform an experiment to identify the subunit of the TCR/CD3 complex against which the autoantibody(s) is directed, the results support the notion that SLE sera possess an anti-TCR/CD3 complex autoantibody(s) that can bind to a subunit(s) of the TCR/CD3 complex and activate the CaMKIV signaling cascade in T cells (Figure 1).
Movement of signaling molecules between intracellular compartments is a well-known concept in cell biology. Under homeostatic conditions, signaling molecules are localized to specific intracellular regions; in response to a stimulus, these molecules can translocate to a target to perform a specific function, such as phosphorylation. cAMP-dependent activation of type IIβ PKA (PKA-IIβ) releases the β type II regulatory subunit (RIIβ) from its binding site on A-kinase anchor proteins (AKAPs) and its interaction with the catalytic subunit, permitting its autophosphorylation and translocation from the cytosol to the nucleus in T cells (17) (Figure 1). Once in the nucleus, phosphorylated RIIβ (pRIIβ) binds to pCREB, inhibits activator protein–1 formation, and strongly inhibits IL-2 transcription and IL-2 production. In SLE, the accumulation of RIIβ in the nuclei of T cells from some patients seems likely to contribute to reduced IL-2 production (18) (Figure 1). The identification of CaMKIV in the nuclei of SLE T cells is the second example of cytosol-to-nuclear translocation of a protein kinase. Despite our incomplete understanding of the repressor mechanisms involved at the level of the basal transcription machinery, the recognition that both nuclear CaMKIV and pRIIβ have the effect of downregulating IL-2 production suggests complementary functions with a powerful outcome (14, 17).
The weight of evidence to date has led investigators to propose that the signaling abnormalities previously identified in SLE T cells reflect a disorder primary to the T cell (3, 11). Here, Tsokos and his colleagues present the first credible evidence that an extracellular factor(s) in the microenvironment can trigger a signaling cascade that ultimately modulates IL-2 transcription in T cells (14). It may be that extracellular factors further hinder an already impaired T cell by activating functional pathways via cell surface receptors.
Reduced IL-2 production, effector and Treg functions, and tolerance in SLE
Two fundamental concepts seem to be emerging from the IL-2–/– and IL-2R–/– knockout murine models that may become applicable to the study of SLE in humans. The first is that IL-2 is necessary for effective Treg maintenance of homeostasis via mechanisms involving anergy and apoptosis. Loss of IL-2 in the murine system is associated with reduced numbers of CD4+CD25+ T cells and autoimmunity; restoration of IL-2 prevents autoimmunity and generates homeostasis. The second is that IL-2 may operate as a third signal for the expansion, development, and function of antigen-dependent T cell effectors (6, 7).
In human SLE, reduced IL-2 generation may be a key factor underlying the impaired T cell effector functions and breach of tolerance that result in autoantibody production. In the murine system, development of CD4+CD25+ Tregs in the thymus and their subsequent export to and expansion in the periphery are dependent on IL-2 (7). Whether there is a similar requirement for IL-2 in the development and expansion of CD4+CD25+ Tregs in humans remains uncertain (6, 7). Although there is still limited information about CD4+CD25+ Treg function in human SLE, one might hypothesize that the reduced numbers of these Tregs in the periphery in SLE (4) reflect a defect in their development, export, and/or proliferation in the periphery.
Future directions
Clearly there is a paucity of information about IL-2 function in human immunity and tolerance, the molecular mechanisms regulating its expression in T cells, and its contribution to the immunopathogenesis of SLE. This gap in knowledge presents a compelling opportunity to investigate these issues. Among possible future directions, establishing the mechanisms of IL-2 regulation of homeostasis in humans should generally be emphasized. Because there is consensus that a breach of T cell tolerance contributes to the persistence of autoreactive T cells and autoantibody production, mechanisms that may result in this failure should be intensively investigated. In this vein, the contribution by Tsokos and his colleagues in this issue of the JCI (14) provides a new appreciation and insight into how the microenvironment in SLE can further impinge on a defective T cell to inhibit IL-2 production. From such studies will come the inspiration and novel approaches necessary to develop therapeutic tools to abate disease and improve the quality of life of our patients.
Footnotes
See the related article beginning on page 996.
Nonstandard abbreviations used: CaMKIV, Ca2+-calmodulin kinase IV; CRE, cAMP response element; CREB, CRE-binding protein; CREM, CRE modulator; IL-2R, IL-2 receptor; pCREB, phosphorylated CREB; pCREM, phosphorylated CREM; PKA-IIβ, type IIβ PKA; pRIIβ, phosphorylated RIIβ; RIIβ, β type II regulatory subunit; SLE, systemic lupus erythematosus.
Conflict of interest: The author has declared that no conflict of interest exists.
References
- 1.Arbuckle MR, et al. Development of autoantibodies before the clinical onset of systemic lupus erythematosus. N. Engl. J. Med. 2003;349:1526–1533. doi: 10.1056/NEJMoa021933. [DOI] [PubMed] [Google Scholar]
- 2.Thompson C, Powrie F. Regulatory T cells. Curr. Opin. Pharmacol. 2004;4:408–414. doi: 10.1016/j.coph.2004.05.001. [DOI] [PubMed] [Google Scholar]
- 3.Kammer GM, Perl A, Richardson BC, Tsokos GC. Abnormal T cell signal transduction in systemic lupus erythematosus. Arthritis Rheum. 2002;46:1139–1154. doi: 10.1002/art.10192. [DOI] [PubMed] [Google Scholar]
- 4.Liu MF, Wang C-R, Fung L-L, Wu C-R. Decreased CD4+CD25+ T cells in peripheral blood of patients with systemic lupus erythematosus. Scand. J. Immunol. 2004;59:198–202. doi: 10.1111/j.0300-9475.2004.01370.x. [DOI] [PubMed] [Google Scholar]
- 5.Jiang H, Chess L. An integrated view of suppressor T cell subsets in immunoregulation. J. Clin. Invest. 2004;114:1198–1208. doi:10.1172/JCI200423411.. doi: 10.1172/JCI23411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nelson BH. IL-2, regulatory T cells, and tolerance. J. Immunol. 2004;172:3983–3988. doi: 10.4049/jimmunol.172.7.3983. [DOI] [PubMed] [Google Scholar]
- 7.Malek TR, Bayer AL. Tolerance, not immunity, crucially depends on IL-2. Nat. Rev. Immunol. 2004;4:665–674. doi: 10.1038/nri1435. [DOI] [PubMed] [Google Scholar]
- 8.Wofsy D, et al. Deficient interleukin 2 activity in MRL/Mp and C57BL/6J mice bearing the lpr gene. J. Exp. Med. 1981;154:1671–1680. doi: 10.1084/jem.154.5.1671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Alcocer-Varela J, Alarcon-Segovia D. Decreased production of and response to interleukin-2 by cultured lymphocytes from patients with systemic lupus erythematosus. J. Clin. Invest. 1982;69:1388–1392. doi: 10.1172/JCI110579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gutierrez-Ramos JC, Andreu JL, Revilla Y, Vinuela E, Martinez C. Recovery from autoimmunity of MRL/lpr mice after infection with an interleukin-2/vaccinia recombinant virus. Nature. 1990;346:271–274. doi: 10.1038/346271a0. [DOI] [PubMed] [Google Scholar]
- 11.Dayal AK, Kammer GM. The T cell enigma in lupus. Arthritis Rheum. 1996;39:23–33. doi: 10.1002/art.1780390104. [DOI] [PubMed] [Google Scholar]
- 12.Solomou EE, Juang Y-T, Gourley MF, Kammer GM, Tsokos GC. Molecular basis of deficient IL-2 production in T cells from patients with systemic lupus erythematosus. J. Immunol. 2001;166:4216–4222. doi: 10.4049/jimmunol.166.6.4216. [DOI] [PubMed] [Google Scholar]
- 13.Kyttaris VC, Tsokos GC. T lymphocytes in systemic lupus erythematosus: an update. Curr. Opin. Rheumatol. 2004;16:548–552. doi: 10.1097/01.bor.0000132646.55056.e0. [DOI] [PubMed] [Google Scholar]
- 14.Juang Y-T, et al. Systemic lupus erythematosus serum IgG increases CREM binding to the IL-2 promoter and suppresses IL-2 production through CaMKIV. J. Clin. Invest. 2005;115:996–1005. doi:10.1172/JCI200522854.. doi: 10.1172/JCI200522854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kammer GM, Haqqi TM, Hasler P, Malemud CJ. The effect of circulating serum factors from patients with systemic lupus erythematosus on protein kinase A (PKA) activity and PKA-dependent protein phosphorylation in T lymphocytes. Clin. Immunol. Immunopathol. 1993;67:8–16. doi: 10.1006/clin.1993.1039. [DOI] [PubMed] [Google Scholar]
- 16.Vassilopoulos D, Kovacs B, Tsokos GC. TCR/CD3 complex-mediated signal transduction pathway in T cells and T cell lines from patients with systemic lupus erythematosus. J. Immunol. 1995;155:2269–2281. [PubMed] [Google Scholar]
- 17.Elliott MR, et al. Down-regulation of IL-2 production in T lymphocytes by phosphorylated protein kinase A-RIIb. J. Immunol. 2004;172:7804–7812. doi: 10.4049/jimmunol.172.12.7804. [DOI] [PubMed] [Google Scholar]
- 18.Mishra N, Khan IU, Tsokos GC, Kammer GM. Association of deficient type II protein kinase A activity with aberrant nuclear translocation of the RIIb-subunit in systemic lupus erythematosus T lymphocytes. J. Immunol. 2000;165:2830–2840. doi: 10.4049/jimmunol.165.5.2830. [DOI] [PubMed] [Google Scholar]
- 19.Takeuchi T, Pang M, Amano K, Koide J, Abe T. Reduced protein tyrosine phosphatase (PTPase) activity of CD45 on peripheral blood lymphocytes in patients with systemic lupus erythematosus. Clin. Exp. Immunol. 1997;109:20–26. doi: 10.1046/j.1365-2249.1997.4371334.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liossis SNC, Ding XZ, Dennis GJ, Tsokos GC. Altered pattern of TCR/CD3-mediated protein-tyrosyl phosphorylation in T cells from patients with systemic lupus erythematosus. Deficient expression of the T cell receptor zeta chain. J. Clin. Invest. 1998;101:1448–1457. doi: 10.1172/JCI1457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Takeuchi T, et al. TCRz chain lacking exon 7 in two patients with systemic lupus erythematosus. Int. Immunol. 1998;10:911–921. doi: 10.1093/intimm/10.7.911. [DOI] [PubMed] [Google Scholar]
- 22.Matache C, et al. p56lck activity and expression in peripheral blood lymphocytes from patients with systemic lupus erythematosus. Autoimmunity. 1999;29:111–120. doi: 10.3109/08916939908995380. [DOI] [PubMed] [Google Scholar]
- 23.Kammer GM, Khan IU, Malemud CJ. Deficient type I protein kinase A isozyme activity in systemic lupus erythematosus T lymphocytes. J. Clin. Invest. 1994;94:422–430. doi: 10.1172/JCI117340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tada Y, Nagasawa K, Yamauchi Y, Tsukamoto H, Niho Y. A defect in the protein kinase C system in T cells from patients with systemic lupus erythematosus. Clin. Immunol. Immunopathol. 1991;60:220–231. doi: 10.1016/0090-1229(91)90065-i. [DOI] [PubMed] [Google Scholar]
- 25.Gergely P, Jr, et al. Persistent mitochondrial hyperpolarization, increased reactive oxygen intermediate production, and cytoplasmic alkalinization characterize altered IL-10 signaling in patients with systemic lupus erythematosus. J. Immunol. 2002;169:1092–1101. doi: 10.4049/jimmunol.169.2.1092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gergely P, Jr, et al. Mitochondrial hyperpolarization and ATP depletion in patients with systemic lupus erythematosus. Arthritis Rheum. 2002;46:175–190. doi: 10.1002/1529-0131(200201)46:1<175::AID-ART10015>3.0.CO;2-H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yi Y, McNerney M, Datta SK. Regulatory defects in Cbl and mitogen-activated protein kinase (extracellular signal-related kinase) pathways cause persistent hyperexpression of CD40 ligand in human lupus T cells. J. Immunol. 2000;165:6627–6634. doi: 10.4049/jimmunol.165.11.6627. [DOI] [PubMed] [Google Scholar]
- 28.Juang Y-T, Tenbrock K, Nambiar MP, Gourley MF, Tsokos GC. Defective production of functional 98-kDa form of Elf-1 is responsible for the decreased expression of TCR z-chain in patients with systemic lupus erythematosus. J. Immunol. 2002;169:6048–6055. doi: 10.4049/jimmunol.169.10.6048. [DOI] [PubMed] [Google Scholar]
- 29.Kyttaris VC, Juang Y-T, Tenbrock K, Weinstein A, Tsokos GC. Cyclic adenosine 5′-monophosphate response element modulator is responsible for the decreased expression of c-fos and activator protein-1 binding in T cells from patients with systemic lupus erythematosus. J. Immunol. 2004;173:3557–3563. doi: 10.4049/jimmunol.173.5.3557. [DOI] [PubMed] [Google Scholar]
- 30.Wong HK, Kammer GM, Dennis G, Tsokos GC. Abnormal NF-kB activity in T lymphocytes from patients with systemic lupus erythematosus is associated with decreased p65-RelA protein expression. J. Immunol. 1999;163:1682–1689. [PubMed] [Google Scholar]
- 31.Deng C, et al. Decreased ras-mitogen-activated protein kinase signaling may cause DNA hypomethylation in T lymphocytes from lupus patients. Arthritis Rheum. 2001;44:397–407. doi: 10.1002/1529-0131(200102)44:2<397::AID-ANR59>3.0.CO;2-N. [DOI] [PubMed] [Google Scholar]
- 32.Grolleau A, Kaplan MJ, Hanash SM, Beretta L, Richardson BC. Impaired translational response and increased protein kinase PKR expression in T cells from lupus patients. J. Clin. Invest. 2000;106:1561–1568. doi: 10.1172/JCI9352. [DOI] [PMC free article] [PubMed] [Google Scholar]