Abstract
It is is well known that insulin stimulates glucose transport and epithelial Na+ channel (ENaC)-mediated Na+ reabsorption; however, the action of insulin on Cl− secretion is not fully understood. In this study, we investigated the action of insulin on Na+–K+–2Cl− cotransporter (NKCC)-mediated Cl− secretion in epithelial A6 cells. Interestingly, insulin treatment remarkably enhanced the forskolin-stimulated Cl− secretion associated with an increase in apical Cl− conductance by upregulating mRNA expression of both CFTR and NKCC, although insulin treatment alone had no effect on the basal Cl− secretion or apical Cl− conductance without forskolin application. We next elucidated a role of phosphoinositide 3-kinase (PI3K) in the insulin-induced enhancement of the Cl− secretion, since insulin actually activated PI3K, resulting in activation of Akt, a downstream molecule of PI3K. LY294002 (a PI3K inhibitor) reduced the Cl− secretion by suppressing mRNA expression of NKCC, whereas insulin still had a stimulatory action on mRNA expression of CFTR even in the presence of LY294002. On the other hand, we found that a MEK inhibitor (PD98059) further enhanced the insulin-stimulated CFTR mRNA expression and the Cl− secretion in forskolin-stimulated A6 cells and that insulin induced slight, transient activation of ERK followed by significant inactivation of ERK. These observations suggest that: (1) insulin respectively upregulates mRNA expression of NKCC and CFTR through activation of PI3K and inactivation of ERK; (2) insulin signals on mRNA expression of NKCC and CFTR are not enough to stimulate transepithelial Cl− secretion, but enhance the stimulatory action of cAMP on transepithelial Cl− secretion.
Keywords: Insulin, NKCC, CFTR, Cl− secretion
Introduction
Transepithelial Cl− secretion from the interstitial space to the luminal one carries negative charges followed by movement of cations, such as Na+, K+ and H+ secretion [1–10]. The NaCl or KCl secretion, essentially driven by the transepithelial Cl− secretion, generates an osmotic gradient followed by water secretion. Thus, Cl− secretion is a crucial driving force for water secretion, forms an epithelial surface liquid protecting our bodies from bacterial and viral infections, and controls body fluid contents [1–4]. Therefore, understanding the regulatory mechanisms of transepithelial Cl− secretion is important for controlling the volume of fluid covering the epithelial surface and extracellular fluid (ECF). Transepithelial Cl− secretion is composed of two steps: a Cl− uptake step through active transporters such as Na+–K+–2Cl− cotransporter (NKCC) in the basolateral membrane and a Cl− releasing step through Cl− channels in the apical membrane [3, 4, 11]. To increase transepithelial Cl− secretion, at least one of the two steps should be stimulated [12–16]. Therefore, discovering factors regulating Cl− transporters and/or Cl− channels and clarifying the regulatory mechanism of transepithelial Cl− secretion are important for understanding the mechanisms of body fluid homeostasis.
Evidence accumulated by recent studies indicates physiological roles of transepithelial Cl− secretion, Cl− transporters and Cl− channels in water movement [17], cell differentiation [18–21], cell proliferation [22–26], and cell signaling and gene expression [27–32]. For instance, proliferation of poorly differentiated gastric cancer cells (MKN45) with higher expression of NKCC1 is faster than that of moderately differentiated gastric cancer cells (MKN28) with lower expression of NKCC1 [22]. These observations suggest that expression levels of NKCC are correlated to the rate of proliferation and that growth factors possibly regulate cell growth via induction of NKCC1. In rat pheochromocytoma (PC12) cells, neuron growth factor (NGF) stimulates neurite elongation through increased NKCC expression, which is associated with intracellular Cl−-dependent tubulin polymerization [33]. These observations suggest the importance of transepithelial Cl− secretion, Cl− transporters and Cl− channels for various cellular functions, not only for transepithelial ion transport.
Insulin is a well-known hormone that regulates glucose metabolism (blood sugar) through its binding to the insulin receptor in the target tissue. Insulin receptor is a transmembrane receptor tyrosine kinase, which is activated through autophosphorylation by ligand binding and transduces the signal into the intracellular space through a phosphoinositide 3-kinase (PI3K)-Akt-dependent cascade [34], which in turn coordinates cell growth [35], cell survival [35], glucose metabolism [35] and ion/glucose transport [34–44]. Insulin is also known to stimulate ENaC-mediated Na+ reabsorption in the distal nephron by increasing ENaC surface expression in the apical membrane [43, 45, 46]. The hypertension frequently observed in type 2 diabetes [47] is considered to be explained by hyperstimulation of ENaC-mediated Na+ reabsorption in hyperinsulinemia. Thus, abnormal regulation or hyperstimulation of ENaC-mediated Na+ reabsorption is considered to be strongly correlated to hypertension. However, the action of insulin on transepithelial Cl− secretion is still poorly understood. In this study, we investigated the transcriptional action of insulin on Cl− secretion and clarified that insulin-enhanced forskolin-stimulated Cl− secretion by upregulating mRNA expression of NKCC and CFTR and that translocation/activation of NKCC and CFTR might be independently regulated.
Materials and methods
Materials
Transwell-Clear permeable supports (PET membrane Transwell-Clear 6.5 and 24 mm) were obtained from Corning, Inc. (Lowell, MA, USA). NCTC-109 medium, benzamil, NPPB, insulin and forskolin were purchased from Sigma (St Louis, MO, USA). LY294002 and PD98059 were obtained from Calbiochem (San Diego, CA, USA).
Solutions
The isotonic test solution contained 120 mM NaCl, 3.5 mM KCl, 1 mM CaCl2, 1 mM MgCl2, 5 mM glucose and 10 mM N-2-hydroxy-ethylpiperazine-N′-2-ethanesulfonic acid (HEPES) adjusted to pH 7.4.
Cell culture
Renal epithelial A6 cells derived from Xenopus laevis [48–50] were purchased from the American Type Culture Collection (ATCC). A6 cells (passage 73–84) were cultured as previously described [11, 51]. Namely, A6 cells (passage 73–84) were grown on plastic flasks at 27 °C in a humidified incubator with 1.0 % CO2 in air in a culture medium that contained 75 % (v/v) NCTC-109, 15 % (v/v) distilled water and 10 % (v/v) fetal bovine serum. For preparation of total RNA and cell lysate, A6 cells were cultured for 13–14 days on 24-mm Transwell-Clear permeable supports. For measurement of short-circuit current (Isc), A6 cells were cultured for 13–14 days on 6.5-mm Transwell-Clear permeable supports.
Measurement of the short-circuit current
Isc was measured as described previously [11, 51]. Monolayers of A6 cells subcultured on Transwell-Clear filter cups were transferred to a modified Ussing chamber (Jim’s Instrument Manufacturing, Iowa City, IA, USA). Transepithelial potential (PD) was continuously measured by a high-impedance millivoltmeter that could function as a voltage clamp with automatic fluid resistance compensation (VCC-600, Physiologic Instruments, San Diego, CA, USA) with a pair of calomel electrodes that were bridged to the modified Ussing chamber by a pair of polyethylene tubes filled with a solution of 2 % (w/v) agarose in a 2-M KCl solution. Isc was measured by the amplifier, VCC-600, with a pair of silver–silver chloride electrodes that were bridged to the modified Ussing chamber by a pair of polyethylene tubes filled with a solution of 2 % (w/v) agarose in a 2-M NaCl solution. When the Isc was measured, the PD was clamped to 0 mV for 1 s by the amplifier. Under a steady condition, the Isc was stable and did not change even if the transepithelial voltage was clamped to 0 mV for 1 min. In a non-steady state, the value of Isc measured at 1 s after clamping the PD to 0 mV is shown as Isc in the present study. A positive current represents a net flow of anions from the basolateral to the apical solution. To detect the transepithelial Cl− secretion, we added 100 μM NPPB (a non-selective Cl− channel blocker [52–54]) to the apical solution and measured an NPPB-sensitive Isc by calculating the difference between the Isc just before and 30 min after application of 100 μM NPPB. We show this difference of Isc as an NPPB-sensitive Isc.
Measurement of transepithelial conductance (Gt)
We measured Gt using the same method as that previously reported in our study [11] using a high-impedance millivoltmeter (VCC-600). Namely, we applied an 1-μA constant current pulse every 10 s for 0.5 s to the A6 monolayer under open-circuit conditions, enabling us to evaluate the Gt by measuring the change in the PD (ΔPD) caused by the 1 μA constant-current pulse using Ohm’s law (Gt = 1 μA/ΔPD mV). To detect the apical Cl− conductance, we added 100 μM NPPB (a non-selective Cl− channel blocker [52–54]) to the apical solution and measured an NPPB-sensitive conductance by calculating the difference between the Gt just before and 30 min after application of 100 μM NPPB. We show this difference of Gt as an NPPB-sensitive conductance (i.e., the apical Cl− conductance), since the apical Cl− conductance is much smaller than the basolateral Cl− conductance [55]. We have already confirmed that the apical Cl− conductance is diminished by apical application of 100 μM NPPB, but not the basolateral Cl− conductance [55, 56]. Thus, we indicate that the NPPB-sensitive conductance can be used as the apical Cl− conductance [55, 56].
Western blotting
The confluent A6 monolayers grown on Transwell-Clear permeable supports were lysed by lysis buffer (50 mM HEPES, 120 mM NaCl, 1.5 mM MgCl2, 1 mM EGTA, 10 % glycerol, 1 % Triton X-100, 100 mM NaF, 10 mM pyrophosphate, 250 μg/ml leupeptin, 0.1 mM phenyl methylsulfonyl fluoride, 100 Kallokein inactivator units/ml aprotinin, pH 7.4) after various experimental treatments. Then, cells were homogenized by sonication and centrifuged at 21,880 g for 10 min at 4 °C to remove insoluble debris. The cell lysates containing 50 μg protein were boiled in SDS sample buffer [60 mM Tris-HCl, 2 % (w/v) SDS, 5 % (v/v) glycerol, pH 6.8] and then subjected to SDS-polyacrylamide gel electrophoresis (SDS-PAGE). After electrophoresis, proteins were transferred to nitrocellulose membranes. Nonspecific binding was blocked by incubation in 5 % (w/v) nonfat milk in Tris-buffered saline (TBS) containing 0.1 % Tween 20 (TBST) at room temperature for 60 min. Membranes were blotted with anti-phospho-ERK (Thr202/Tyr204), anti-phospho-Akt (Ser473) and anti-β-tubulin antibodies (Cell Signaling Technology, Beverly, MA, USA) in 5 % BSA in TBST at 4 °C overnight. The membranes were then washed with TBST and incubated for 60 min at room temperature with horseradish peroxidase (HRP)-conjugated anti-rabbit IgG (Cell Signaling Technology, Denver, MA, USA) in 5 % (w/v) non-fat milk in TBST. After washing with TBST, the blots were detected with ECL prime chemiluminescent reagent (GE Healthcare Bio-Sciences Corp., Piscataway, NJ, USA). Each blot presented was performed at least three times.
Quantitative real-time PCR
To compare relative amounts of mRNA for NKCC1 and the CFTR Cl− channel in A6 monolayers treated under different conditions, we performed quantitative real-time PCR. Total RNA was extracted from A6 monolayers using an RNeasy kit (QIAGEN, Tokyo, Japan) with DNase digestion according to the manufacture’s protocol. Reverse transcription (RT) was performed on 1 μg total RNA, calculated from absorbance measured at 260 nm, using a High Capacity Coda transcription kit (Applied Biosystems, Rotkreuz, Switzerland) according to the manufacturer’s protocol. Primers (SIGMA Genosis) and TaqMan probes (Applied Biosystems, Rotkreuz, Switzerland) unique for each Xenopus NKCC, Xenopus CFTR Cl− channel or Xenopus Β-actin were the following: xNKCC forward 5′-TGCCTCCCTTGCCAAGTCT-3′, reverse 5′-CGAGTGACACCCACATGTTGTA-3′, TaqMan probe 5′-AGGATGGCGTCCCGCCTTCAGATA-3′, xCFTR forward 5′-ATGCTATTCTACATGCCCCAATG-3′, reverse 5′-TCCAATATAGCTGTATCCTTTGAAAATC-3′, TaqMan probe 5′-ACCTTCAATACAATGAGAGCTGGTCGGATACTTAA-3′, xβ-actin forward 5′-GATGCTCCCCGTGCTGTTT-3′, reverse 5′-TTCCAACCATGACACCCTGA-3′, TaqMan probe 5′-CCCATCTATTGTGGGTCGCCCAAGA-3′. Quantitative real-time PCR with TaqMan probes using the ABI 7300 real-time PCR system was performed according to the manufacturer’s protocol. Diluted RT samples (100 ng total RNA) were amplified in a final volume of 25 μl. Primers were used at a concentration of 800 nM and probes at a concentration of 200 nM. β-actin were used as an internal control. The thermal cycling conditions were as follows: 50 °C for 2 min and 95 °C for 10 min, followed by 40 cycles of 95 °C for 15 s and 60 °C for 1 min. Relative amounts of NKCC and CFTR Cl− channel mRNA were determined by normalization with β-actin mRNA expression.
Temperature
All experiments for electrophysiological and other measurements were performed at 24–25 °C unless otherwise indicated.
Data presentation
All data are presented as mean ± standard error of the mean (SEM). Where SEM bars are not visible, they are smaller than the symbol. Student’s t test and ANOVA were used for statistical analysis as appropriate, and p < 0.05 was considered significant.
Results
Enhancement of forskolin-stimulated Cl− secretion by insulin pretreatment
The Cl− secretion is generally composed of two steps: (1) the step of Cl− uptake via the Na+–K+–2Cl− cotransporter 1 (NKCC1) at the basolateral membrane and (2) the step of Cl− release via the cystic fibrosis transmembrane conductance regulator (CFTR) Cl− channel at the apical membrane [11, 57]. Elevation of cytosolic cAMP by bilateral application of 10 μM forskolin stimulated the Cl− secretion through activation of both the apical Cl− channels and basolateral NKCC1 in A6 cells. In this study, transepithelial Cl− secretion and apical Cl− channel activities are respectively determined as NPPB (a Cl− channel blocker)-sensitive Isc and NPPB-sensitive conductance (Gt) by addition of 100 μM NPPB to the apical solution. In all of the experiments, to isolate the Cl− secretion from the Na+ absorption, we added 10 μM benzamil, which is a specific inhibitor of ENaC and completely blocks the ENaC at the concentration (10 μM) [30, 58], to the apical solution 10 min before application of forskolin. In this study, we measured NPPB-sensitive Isc and Gt after pretreatment of A6 monolayers with insulin, which shows its maximum effect on ion transport within 100 nM–1 μM [59, 60]. Thus, we applied 1 μM of insulin to obtain its maximum effect in the present study. We preincubated A6 cells with or without 1 μM insulin in the absence or presence of inhibitors in the culture medium for various time periods, then washed out insulin and inhibitors, and set the pretreated A6 monolayers to the modified Ussing chamber in 120 mM NaCl solution without any insulin and inhibitors. Then, we stimulated the A6 monolayers in 120 mM NaCl solution with 10 μM forskolin 10 min after addition of 10 μM benzamil. Finally, NPPB of 100 μM was added to the apical solution 60 min after application of forskolin, and the differences in Isc and Gt between just before and 30 min after addition of NPPB were measured as NPPB-sensitive Isc and Gt.
Acute (0–60 min) or chronic (2–24 h) application of insulin to the basolateral solution had no effect on basal Cl− secretion detected as NPPB-sensitive Isc in A6 cells (data not shown). To clarify the action of insulin on cAMP-stimulated Cl− secretion, we pretreated A6 cells with 1 μM insulin for 0, 1, 3, 6 and 24 h in the culture medium and washed out the insulin. Just after washing out the insulin, 10 μM of forskolin was applied to the apical and basolateral solutions for elevation of cytosolic cAMP concentration. Then, we measured NPPB-sensitive Isc and Gt by adding 100 μM NPPB 60 min after application of forskolin in the absence of insulin. Pretreatment of monolayered A6 cells with 1 μM insulin for more than 3 h markedly enhanced the forskolin-stimulated Cl− secretion (NPPB-sensitive Isc) by increasing apical Cl− channel activities (conductances) detected as NPPB-sensitive Gt (Fig. 1). Pretreatment with insulin for 6 h had similar effects to that for 24 h, which showed the maximum effects on the enhancement of forskolin-stimulated Cl− secretion (NPPB-sensitive Isc) and conductance (NPPB-sensitive Gt) (Fig. 1). It is notable that application of insulin alone had no stimulatory action on Cl− secretion (NPPB-sensitive Isc) or conductance (NPPB-sensitive Gt) in the absence of forskolin. Namely, the NPPB-sensitive Isc was 0.30 ± 0.03 μA/cm2 (n = 7) in cells preincubated with insulin for 24 h, which was not significantly different from that without insulin (0.23 ± 0.03 μA/cm2 without insulin; n = 7): the NPPB-sensitive Gt was 22.04 ± 1.31 μA/cm2 (n = 7) in cells preincubated with insulin for 24 h, which was not significantly different from that without insulin (18.67 ± 1.38 μS/cm2 without insulin; n = 7).
Fig. 1.
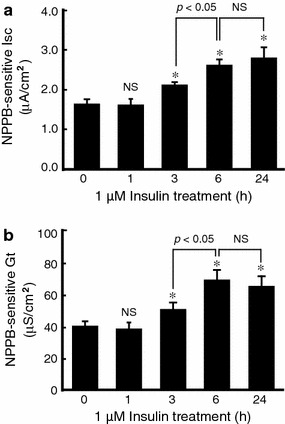
Time-dependent effect of insulin on forskolin-stimulated NPPB-sensitive Isc (a) and Gt (b) in A6 cells. After pretreating monolayer A6 cells with 1 μM insulin for 0, 1, 3, 6 and 24 h, 10 μM forskolin was applied to the apical and basolateral sites, and then NPPB-sensitive Isc and Gt were detected by the addition of 100 μM NPPB to the apical solution 60 min after application of forskolin. Data are presented as mean ± SEM. n = 5–7. *p < 0.05 vs. control (0 h; without insulin)
Insulin enhanced the Cl− secretion through transcriptional regulation of NKCC and CFTR
Incubation with insulin for at least 3 h is required for enhancement of the Cl− secretion, suggesting that transcriptional regulation in NKCC and/or CFTR might be involved in the enhancement of the Cl− secretion. To elucidate this suggestion, we next examined the effect of insulin on mRNA expression of NKCC and CFTR by quantitative real-time PCR. Pretreatment of monolayered A6 cells with 1 μM insulin for 24 h remarkably increased mRNA expression of both NKCC and CFTR (Fig. 2). This result suggests that insulin enhanced forskolin-stimulated Cl− secretion by increasing mRNA expression of both NKCC and CFTR.
Fig. 2.
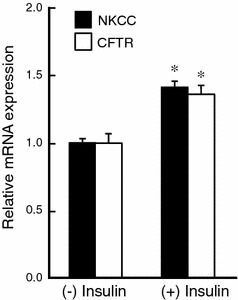
Effects of insulin on mRNA expression of NKCC and the CFTR Cl− channel. A6 monolayers were treated with 1 μM insulin for 24 h, and then mRNA expression of NKCC and the CFTR Cl− channel was detected by quantitative real-time PCR. Data are presented as mean ± SEM. n = 5–8. *p < 0.05 vs. insulin (−)
Involvement of PI3K in the transcriptional regulation of NKCC and CFTR
We next addressed the question of how insulin induces mRNA expression of NKCC and CFTR. As PI3K is a major signal molecule in the insulin cascade, we examined whether insulin activates PI3K in monolayered A6 cells by detecting Akt phosphorylation. Insulin increased phosphorylation of Akt at Ser473 (Fig. 3), which is known to be phosphorylated through a PI3K-dependent pathway. To clarify the involvement of PI3K in the insulin-induced mRNA expression of NKCC and CFTR, we examined the effects of LY294002 (a specific inhibitor of PI3K). Pretreatment with LY294002 markedly reduced mRNA expression of NKCC and CFTR in insulin-untreated and -treated A6 cells (Fig. 4a). The treatment with LY294002 blocked the stimulatory action of insulin on NKCC mRNA expression, but not CFTR mRNA expression (Fig. 4a). This observation suggests that insulin upregulates NKCC mRNA expression by mainly activating a PI3K-dependent pathway, but CFTR mRNA expression via a PI3K-independent pathway.
Fig. 3.
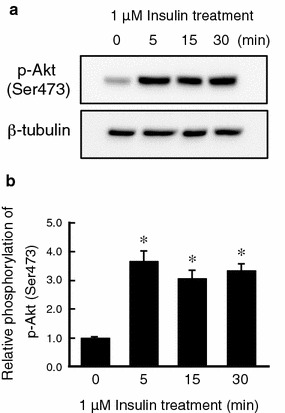
Effects of insulin on phosphorylation of Akt as an indicator of PI3K activity in A6 monolayered cells. a A6 monolayers were treated with 1 μM insulin for the indicated time, and then phosphorylation of Akt (p-Akt) at Ser473 was detected by immunoblotting with an anti-phospho Akt (Ser473)-specific antibody. b Relative amounts of phosphorylated Akt (p-Akt) at Ser473 are shown. Equal loading of cell lysate to each well was confirmed by measuring β-tubulin as an internal control. Data are presented as mean ± SEM. n = 3. *p < 0.05 vs. insulin (−)
Fig. 4.
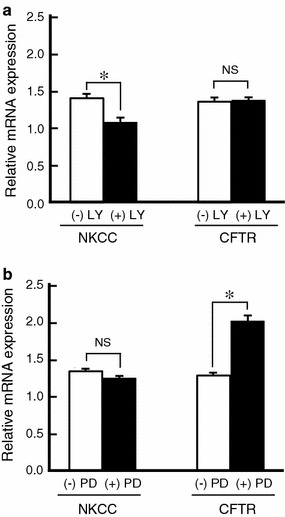
Effects of LY294002 (LY, a PI3K inhibitor, 30 μM) (a) and PD98059 (PD, a MEK inhibitor, 50 μM) (b) on insulin-induced mRNA expression of the NKCC and the CFTR Cl− channel. After pretreatment with or without LY294002 or PD98059 for 1 h in the culture medium, A6 monolayers were incubated in the culture medium with or without 1 μM insulin in the presence or absence of LY294002 or PD98059 for 6 h. The mRNA expression of NKCC and CFTR was detected by real-time PCR. Relative amounts of mRNA expression of NKCC and the CFTR Cl− channel were normalized to β-actin as an internal control. Amounts of mRNA expression of NKCC and the CFTR Cl− channel in the absence of insulin are shown as 1.0; therefore, the relative amounts of mRNA expression of NKCC and the CFTR Cl− channel shown here mean the insulin action on amounts of mRNA expression of NKCC and the CFTR Cl− channel compared with those without insulin pretreatment. Data are presented as mean ± SEM. n = 5–8. NS, no significance; *p < 0.05
To further elucidate a PI3K-independent pathway for CFTR mRNA expression, we examined a possible role of ERK in CFTR mRNA expression by using PD98059 (a MEK inhibitor blocking ERK activation). Pretreatment of monolayered A6 cells with PD98059 did not affect NKCC mRNA expression, but further increased CFTR mRNA expression in insulin-treated A6 cells (Fig. 4b). These observations suggest that ERK would be a negative regulator for CFTR mRNA expression and that insulin might inactivate ERK, leading to an increase in CFTR mRNA expression through a PI3K-independent pathway. To confirm this presumption, we examined whether insulin inactivates ERK by detecting activated (phosphorylated) ERK with immunoblotting. Insulin induced slight, transient activation followed by a remarkable inactivation (dephosphorylation) of ERK (Fig. 5). Based on these results, we suggest that insulin increases CFTR mRNA expression via suppression of ERK in a PI3K-independent manner.
Fig. 5.
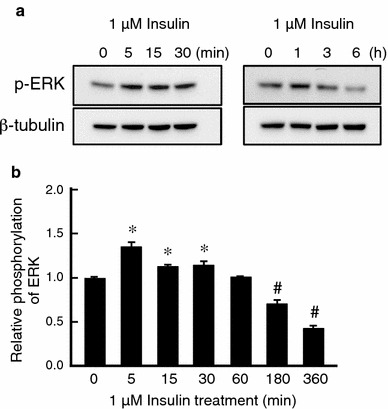
Effects of insulin on phosphorylation of ERK in A6 monolayered cells. a A6 monolayers were treated with 1 μM insulin for the indicated time, and then phosphorylation of ERK was detected by immunoblotting with an anti-phospho-ERK specific antibody. b Relative amounts of phosphorylated ERK are shown. Equal loading of cell lysate to each well was confirmed by measuring β-tubulin as an internal control. Data are presented as mean ± SEM. n = 3. * Significantly larger than that at 0 min (without insulin treatment) with p < 0.05. # Significantly smaller than that at 0 min (without insulin treatment) with p < 0.05
The regulatory mechanism of forskolin-stimulated Cl− secretion by insulin
In this study, we indicated that insulin enhanced the forskolin-stimulated Cl− secretion by increasing respectively mRNA expression of both NKCC and CFTR through PI3K activation and ERK inactivation. We next elucidated whether PI3K-dependent transcriptional regulation in NKCC and CFTR actually participates in the enhancement of forskolin-stimulated Cl− secretion by insulin in A6 cells. Pretreatment with LY294002 slightly decreased the forskolin-stimulated NPPB-sensitive Isc associated with a similar rate of reduction of NPPB-sensitive Gt in insulin-untreated A6 cells [(−) Insulin in Fig. 6a, b]. Likewise, LY294002 remarkably suppressed the insulin-induced enhancement of forskolin-stimulated NPBB-sensitive Isc associated with suppression of NPPB-sensitive Gt to a similar extent as Isc [(+) Insulin in Fig. 6a, b]. Consequently, most of the insulin-induced enhancement of forskolin-stimulated Cl− secretion and Cl− channel conductance (activity) was sensitive to LY294002, suggesting that insulin enhances the forskolin-stimulated Cl− secretion and Cl− channel conductance via a PI3K-dependent pathway (Fig. 6). On the contrary, pretreatment with PD98059 further increased the insulin-induced enhancement of forskolin-stimulated Cl− secretion accompanied by further elevation of apical Cl− channel conductance (activity) (Fig. 7), suggesting that complete inhibition of ERK forcibly caused by PD98059 further increased the insulin-pretreatment-stimulated Cl− secretion via further enhancement of CFTR expression in forskolin-stimulated cells.
Fig. 6.
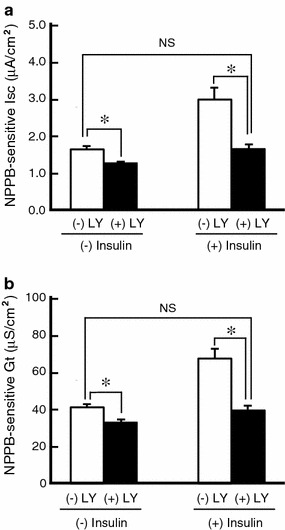
Effects of LY294002 (LY, a PI3K inhibitor, 30 μM) on forskolin-stimulated NPPB-sensitive Isc (a) and Gt (b) in A6 cells treated with or without insulin. After pretreatment with or without LY294002 for 1 h in the culture medium, A6 monolayers were incubated in the medium with or without 1 μM insulin in the presence or absence of LY294002 for 6 h. After each treatment, 10 μM forskolin was applied, and then NPPB-sensitive Isc and Gt were measured by adding NPPB 60 min after the application of forskolin. Data are presented as mean ± SEM. n = 5–8. *p < 0.05
Fig. 7.
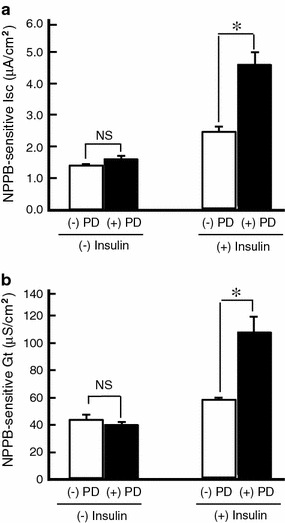
Effects of PD98059 (PD, a MEK inhibitor, 50 μM) on forskolin-stimulated NPPB-sensitive Isc (a) and Gt (b) in A6 cells treated with or without insulin. After pretreatment with or without 50 μM PD98059 for 1 h in the culture medium, A6 monolayers were incubated in the medium with or without 1 μM insulin for 6 h in the presence or absence of PD98059. After each treatment, 10 μM forskolin was applied, and then NPPB-sensitive Isc and Gt were measured by adding NPPB 60 min after application of forskolin. Data are presented as mean ± SEM. n = 5–8. *p < 0.05
cAMP-dependent regulation of NKCC and/or CFTR translocation
mRNA expression of NKCC and CFTR is upregulated by insulin; nevertheless, the basal Cl− secretion or apical Cl− channel conductance (activity) was not significantly increased. There are two possible explanations for these phenomena. One is that insulin induced too small increases in basal Cl− secretion and apical Cl− channel conductance (activity) to detect the increases. The other is that insulin signals alone are not enough to translocate proteins of NKCC and/or CFTR newly synthesized by insulin pretreatment to the plasma membrane for stimulation of Cl− secretion. To confirm this presumption, we examined the effects of brefeldin A (BFA; an inhibitor of protein translocation from ER to Golgi) on the insulin-induced enhancement of Cl− secretion and apical Cl− channel conductance (activity) in forskolin-stimulated cells. After pretreatment with insulin for 5 h, we further pretreated A6 cells with both insulin and BFA (5 μg/ml) for 1 h. Then, we measured Cl− secretion and apical Cl− channel conductance (activity) in forskolin-stimulated cells in the absence or presence of BFA. BFA abolished the stimulatory action of insulin on Cl− secretion (Fig. 8a) and apical Cl− channel conductance (activity: Fig. 8b) in forskolin-stimulated cells.
Fig. 8.
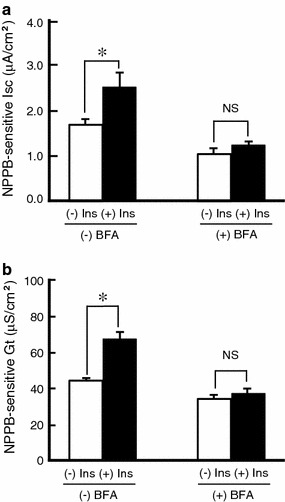
Effects of brefeldin A (BFA) on forskolin-stimulated Cl− secretion (a) and conductance (b) in A6 cells treated with or without insulin. After pretreatment with or without 1 μM insulin for 5 h in the culture medium, A6 monolayers were incubated in the medium with or without 5 μg/ml BFA in the presence or absence of insulin for 1 h. After each treatment, A6 monolayers were incubated with 10 μM forskolin under each condition without insulin, and then NPPB-sensitive Isc and Gt were measured by adding NPPB 60 min after application of forskolin. Data are presented as mean ± SEM. n = 4. Ins insulin, NS no significance; *p < 0.05
Discussion
In this study, we elucidate that insulin enhanced forskolin-stimulated Cl− secretion and apical Cl− channel conductance (activity) by respectively upregulating mRNA expression of NKCC and CFTR via activation of PI3K-Akt and inactivation of ERK in epithelial A6 cells. Previous studies indicated the action of growth factors including insulin on NKCC. Insulin activates NKCC in human fibroblast [61], 3T3-L1 fibroblast, adipocyte [62] and L6 cells [63] regulating cell volume and K+ uptake. IGF-1 (insulin-like growth factor-1) increases NKCC mRNA expression in pavement cells [64], while EGF (epidermal growth factor) and NGF increase NKCC protein expression in intestinal epithelial cells [65] and PC12 cells [18]. In human skin fibroblasts, EGF-induced activation of NKCC is crucial for mitogenic signaling pathways [66]. Thus, these studies indicate that growth factors regulate NKCC expression at transcriptional, translational and posttranslational levels in various types of cells. On the other hand, NKCC basically contributes to Cl− uptake from extracellular into intracellular space regulating the cell volume, intracellular Cl− concentration ([Cl−]i) and transepithelial Cl− secretion. In our previous studies, [Cl−]i was shown to have a crucial role in cell proliferation and differentiation in gastric cancer cells [22, 23, 25], prostate cancer cells [24], osteoblast-like cells [67], breast cancer cell [26] and neural cells [18, 19]. These studies suggest that growth factors might control cell proliferation through regulation of NKCC expression and activity.
In this study, pretreatment with insulin alone did not stimulate transepithelial Cl− secretion in A6 cells, even though insulin upregulated mRNA expression of NKCC and CFTR. On the other hand, in the presence of forskolin, the enhancement of Cl− secretion by insulin was revealed. To stimulate transepithelial Cl− secretion, at least either the step of NKCC-mediated Cl− uptake or CFTR-mediated Cl− release in the Cl− secretion must be stimulated. This means that elevation of cAMP regulating NKCC and CFTR would be essentially required for stimulation of Cl− uptake or release contributing to the Cl− secretion.
In the present study, we observed that application of insulin alone upregulated mRNA expression of NKCC and CFTR; nevertheless, its application showed no stimulatory action on the basal Cl− secretion or apical Cl− channel conductance (activity). To clarify these phenomena, we speculated that proteins of NKCC and/or CFTR newly synthesized via insulin-elevated mRNA expression of NKCC and CFTR would stay in intracellular stores sites, but neither protein would be translocated to the apical or basolateral membrane only by insulin signaling. To confirm this speculation, we applied BFA (an inhibitor of protein translocation from ER to Golgi). As expected, BFA blocked the forskolin action on Cl− secretion and apical Cl− channel conductance (activity) (Fig. 8). Further, we should state that even in the absence of insulin application, BFA reduced the stimulatory action of forskolin [Fig. 8a, b; (+) BFA]. This means that irrespective of insulin application forskolin would have a stimulatory action on the translocation of NKCC and CFTR proteins to the apical and basolateral membrane. We strongly suggest that insulin shows its enhancing action on forskolin-stimulated Cl− secretion and apical Cl− channel conductance (activity) by upregulating mRNA expression of NKCC and CFTR but that the insulin signaling is not large enough for translocation of the insulin-induced newly synthesized proteins of NKCC and CFTR.
In A6 cells, insulin increased cell proliferation, which was abolished by bumetanide (an NKCC inhibitor) (Niisato et al. unpublished data). cAMP stimulates translocation of CFTR Cl− channels to the apical membrane and increases the number of functional CFTR Cl− channels detected by patch clamp techniques [68]. Thus, insulin and cAMP cooperatively ativate Cl− transport regulating various cellular functions including epithelial Cl− secretion.
Conclusion
Taken together, these observations based on the present study suggest that: (1) insulin stimulates mRNA expression of NKCC and CFTR, which respectively contribute to transepithelial Cl− secretion as a Cl− uptake pathway across the basolateral membrane and a Cl− release pathway across the apical membrane; (2) the insulin-induced mRNA expression of NKCC and CFTR is respectively mediated through activation of PI3K stimulating NKCC mRNA expression and inactivation of ERK suppressing CFTR mRNA expression; (3) the insulin-increased NKCC and CFTR would be in the cytosolic store sites and cytosolic cAMP stimulates translocation of the insulin-increased NKCC and CFTR to the basolateral and apical membranes leading to an increases in transepithelial Cl− secretion associated with elevation of apical Cl− channel conductance (activity). Thus, insulin enhances the stimulatory action of cAMP on epithelial Cl− secretion via increases in new synthesis of NKCC and CFTR via PI3K- and ERK-mediated mechanisms (Fig. 9).
Fig. 9.
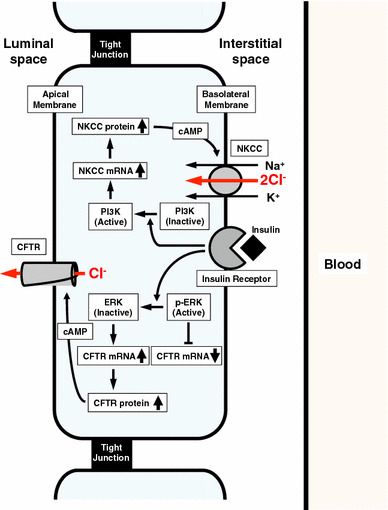
Summary of insulin action on NKCC and CFTR participating in epithelial Cl− secretion in epithelial A6 cells. (1) Insulin activates PI3K, resulting in stimulation of NKCC mRNA expression. (2) Insulin inactivates ERK, which suppresses CFTR mRNA expression. Insulin-induced inactivation of ERK releases suppression of CFTR mRNA expression, leading to elevation of CFTR mRNA expression. Both elevation of mRNA expression of NKCC and CFTR induced by insulin stimulates expression of NKCC and CFTR proteins, which stay in cytosolic store sites. cAMP stimulates translocation of insulin-induced NKCC and CFTR proteins staying in cytosolic store sites, leading to much larger epithelial Cl− secretion associated with much larger elevation of CFTR activity than that under the insulin-untreated condition
Acknowledgments
This work was supported by Grants-in-Aid from the Japan Society of the Promotion of Science (24590283 to NN, 25670111 to YM), Salt Science (1235 to YM and NN) and Cell Research Conference to YM.
Footnotes
H. Sun and N. Niisato contributed equally to the present study.
Contributor Information
Naomi Niisato, Phone: 075-251-5311, Email: naomi@koto.kpu-m.ac.jp.
Yoshinori Marunaka, Email: marunaka@koto.kpu-m.ac.jp.
References
- 1.Bachmann O, Juric M, Seidler U, Manns MP, Yu H. Basolateral ion transporters involved in colonic epithelial electrolyte absorption, anion secretion and cellular homeostasis. Acta Physiol (Oxf) 2011;201:33–46. doi: 10.1111/j.1748-1716.2010.02153.x. [DOI] [PubMed] [Google Scholar]
- 2.Hong JH, Park S, Shcheynikov N, Muallem S. Mechanism and synergism in epithelial fluid and electrolyte secretion. Pflugers Arch. 2014;466:1487–1499. doi: 10.1007/s00424-013-1390-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Marunaka Y. Importance of expression and function of angiotensin II receptor type 1 in pulmonary epithelial cells. Respir Physiol Neurobiol. 2014;196:39–42. doi: 10.1016/j.resp.2014.02.013. [DOI] [PubMed] [Google Scholar]
- 4.Marunaka Y (2014) Characteristics and pharmacological regulation of epithelial Na+ channel (ENaC) and epithelial Na+ transport. J Pharmacol Sci. (in press). [PubMed]
- 5.Ikehara O, Hayashi H, Waguri T, Kaji I, Karaki S, Kuwahara A, Suzuki Y. Subepithelial trypsin induces enteric nerve-mediated anion secretion by activating proteinase-activated receptor 1 in the mouse cecum. J Physiol Sci. 2012;62:211–219. doi: 10.1007/s12576-012-0198-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ikehara O, Hayashi H, Waguri T, Kaji I, Karaki S, Kuwahara A, Suzuki Y. Luminal trypsin induces enteric nerve-mediated anion secretion in the mouse cecum. J Physiol Sci. 2014;64:119–128. doi: 10.1007/s12576-013-0302-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mizuno H, Suzuki Y, Watanabe M, Sokabe T, Yamamoto T, Hattori R, Gotoh M, Tominaga M. Potential role of transient receptor potential (TRP) channels in bladder cancer cells. J Physiol Sci. 2014;64:305–314. doi: 10.1007/s12576-014-0319-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fujii T, Minagawa T, Shimizu T, Takeguchi N, Sakai H. Inhibition of ecto-ATPase activity by curcumin in hepatocellular carcinoma HepG2 cells. J Physiol Sci. 2012;62:53–58. doi: 10.1007/s12576-011-0176-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fujii T, Awaka SY, Takahashi Y, Fujita K, Tsuji H, Shimizu T, Gomi T, Tsukada K, Sakai H (2013) Modulation of H+, K+ -ATPase activity by the molecular chaperone ERp57 highly expressed in gastric parietal cells. FEBS Lett 587:3898–3905 [DOI] [PubMed]
- 10.Takahashi Y, Fujii T, Fujita K, Shimizu T, Higuchi T, Tabuchi Y, Sakamoto H, Naito I, Manabe K, Uchida S, Sasaki S, Ikari A, Tsukada K, Sakai H. Functional coupling of chloride-proton exchanger ClC-5 to gastric H+, K+ -ATPase. Biol Open. 2014;3:12–21. doi: 10.1242/bio.20136205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sun H, Niisato N, Nishio K, Hamilton KL, Marunaka Y. Distinct action of flavonoids, myricetin and quercetin, on epithelial Cl− secretion: useful tools as regulators of Cl− secretion. Biomed Res Int. 2014;2014:902735. doi: 10.1155/2014/902735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sasamoto K, Niisato N, Marunaka Y. Analysis of epithelial ion transport by mathematical model. J Physiol Sci. 2012;62:S136. [Google Scholar]
- 13.Banga A, Flaig S, Lewis S, Winfree S, Blazer-Yost BL. Epinephrine stimulation of anion secretion in the Calu-3 serous cell model. Am J Physiol Lung Cell Mol Physiol. 2014;306:L937–L946. doi: 10.1152/ajplung.00190.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kim D, Kim J, Burghardt B, Best L, Steward MC. Role of anion exchangers in Cl− and HCO3 − secretion by the human airway epithelial cell line Calu-3. Am J Physiol Cell Physiol. 2014;307:C208–C219. doi: 10.1152/ajpcell.00083.2014. [DOI] [PubMed] [Google Scholar]
- 15.Lee RJ, Foskett JK. Ca2+ signaling and fluid secretion by secretory cells of the airway epithelium. Cell Calcium. 2014;55:325–336. doi: 10.1016/j.ceca.2014.02.001. [DOI] [PubMed] [Google Scholar]
- 16.Ponce-Coria J, Markadieu N, Austin TM, Flammang L, Rios K, Welling PA, Delpire E. A Novel Ste20-related proline/alanine-rich kinase (spak)-independent pathway involving calcium-binding protein 39 (Cab39) and serine threonine kinase with No lysine member 4 (WNK4) in the activation of Na-K-Cl cotransporters. J Biol Chem. 2014;289:17680–17688. doi: 10.1074/jbc.M113.540518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Asano J, Niisato N, Nakajima K, Miyazaki H, Yasuda M, Iwasaki Y, Hama T, Dejima K, Hisa Y, Marunaka Y. Quercetin stimulates Na+/K+/2Cl− cotransport via PTK-dependent mechanisms in human airway epithelium. Am J Respir Cell Mol Biol. 2009;41:688–695. doi: 10.1165/rcmb.2008-0338OC. [DOI] [PubMed] [Google Scholar]
- 18.Nakajima K, Miyazaki H, Niisato N, Marunaka Y. Essential role of NKCC1 in NGF-induced neurite outgrowth. Biochem Biophys Res Commun. 2007;359:604–610. doi: 10.1016/j.bbrc.2007.05.133. [DOI] [PubMed] [Google Scholar]
- 19.Nakajima K, Niisato N, Marunaka Y. Quercetin stimulates NGF-induced neurite outgrowth in PC12 cells via activation of Na+/K+/2Cl− cotransporter. Cell Physiol Biochem. 2011;28:147–156. doi: 10.1159/000331723. [DOI] [PubMed] [Google Scholar]
- 20.Okada S, Li Q, Whitin JC, Clayberger C, Krensky AM. Intracellular mediators of granulysin-induced cell death. J Immunol. 2003;171:2556–2562. doi: 10.4049/jimmunol.171.5.2556. [DOI] [PubMed] [Google Scholar]
- 21.Okada Y, Sato K, Numata T. Pathophysiology and puzzles of the volume-sensitive outwardly rectifying anion channel. J Physiol. 2009;587:2141–2149. doi: 10.1113/jphysiol.2008.165076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Shiozaki A, Miyazaki H, Niisato N, Nakahari T, Iwasaki Y, Itoi H, Ueda Y, Yamagishi H, Marunaka Y. Furosemide, a blocker of Na+/K+/2Cl− cotransporter, diminishes proliferation of poorly differentiated human gastric cancer cells by affecting G0/G1 state. J Physiol Sci. 2006;56:401–406. doi: 10.2170/physiolsci.RP010806. [DOI] [PubMed] [Google Scholar]
- 23.Miyazaki H, Shiozaki A, Niisato N, Ohsawa R, Itoi H, Ueda Y, Otsuji E, Yamagishi H, Iwasaki Y, Nakano T, Nakahari T, Marunaka Y. Chloride ions control the G1/S cell-cycle checkpoint by regulating the expression of p21 through a p53-independent pathway in human gastric cancer cells. Biochem Biophys Res Commun. 2008;366:506–512. doi: 10.1016/j.bbrc.2007.11.144. [DOI] [PubMed] [Google Scholar]
- 24.Hiraoka K, Miyazaki H, Niisato N, Iwasaki Y, Kawauchi A, Miki T, Marunaka Y. Chloride ion modulates cell proliferation of human androgen-independent prostatic cancer cell. Cell Physiol Biochem. 2010;25:379–388. doi: 10.1159/000303042. [DOI] [PubMed] [Google Scholar]
- 25.Ohsawa R, Miyazaki H, Niisato N, Shiozaki A, Iwasaki Y, Otsuji E, Marunaka Y. Intracellular chloride regulates cell proliferation through the activation of stress-activated protein kinases in MKN28 human gastric cancer cells. J Cell Physiol. 2010;223:764–770. doi: 10.1002/jcp.22088. [DOI] [PubMed] [Google Scholar]
- 26.Kitagawa M, Niisato N, Shiozaki A, Ohta-Fujimoto M, Hosogi S, Miyazaki H, Ichikawa D, Otsuji E, Marunaka Y. A regulatory role of K-Cl cotransporter in the cell cycle progression of breast cancer MDA-MB-231 cells. Arch Biochem Biophys. 2013;539:92–98. doi: 10.1016/j.abb.2013.06.014. [DOI] [PubMed] [Google Scholar]
- 27.Akita T, Okada Y. Characteristics and roles of the volume-sensitive outwardly rectifying (VSOR) anion channel in the central nervous system. Neuroscience. 2014;275c:211–231. doi: 10.1016/j.neuroscience.2014.06.015. [DOI] [PubMed] [Google Scholar]
- 28.Simon Bulley SB, Jaggar JH. Cl− channels in smooth muscle cells. Pflugers Arch. 2014;466:861–872. doi: 10.1007/s00424-013-1357-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.de Los Heros P, Alessi DR, Gourlay R, Campbell DG, Deak M, Macartney TJ, Kahle KT, Zhang J. The WNK-regulated SPAK/OSR1 kinases directly phosphorylate and inhibit the K+–Cl− co-transporters. Biochem J. 2014;458:559–573. doi: 10.1042/BJ20131478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Niisato N, Eaton DC, Marunaka Y. Involvement of cytosolic Cl− in osmoregulation of alpha-ENaC gene expression. Am J Physiol Renal Physiol. 2004;287:F932–F939. doi: 10.1152/ajprenal.00131.2004. [DOI] [PubMed] [Google Scholar]
- 31.Schobel N, Radtke D, Lubbert M, Gisselmann G, Lehmann R, Cichy A, Schreiner BS, Altmuller J, Spector AC, Spehr J, Hatt H, Wetzel CH. Trigeminal ganglion neurons of mice show intracellular chloride accumulation and chloride-dependent amplification of capsaicin-induced responses. PLoS One. 2012;7:e48005. doi: 10.1371/journal.pone.0048005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mohebbi N, Perna A, van der Wijst J, Becker HM, Capasso G, Wagner CA. Regulation of two renal chloride transporters, AE1 and pendrin, by electrolytes and aldosterone. PLoS One. 2013;8:e55286. doi: 10.1371/journal.pone.0055286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Nakajima K, Niisato N, Marunaka Y. Enhancement of tubulin polymerization by Cl−-induced blockade of intrinsic GTPase. Biochem Biophys Res Commun. 2012;425:225–229. doi: 10.1016/j.bbrc.2012.07.072. [DOI] [PubMed] [Google Scholar]
- 34.Hayata H, Miyazaki H, Niisato N, Yokoyama N, Marunaka Y. Lowered extracellular pH is involved in the pathogenesis of skeletal muscle insulin resistance. Biochem Biophys Res Commun. 2014;445:170–174. doi: 10.1016/j.bbrc.2014.01.162. [DOI] [PubMed] [Google Scholar]
- 35.Guo S. Insulin signaling, resistance, and the metabolic syndrome: insights from mouse models into disease mechanisms. J Endocrinol. 2014;220:T1–T23. doi: 10.1530/JOE-13-0327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Blazer-Yost BL, Cox M. Insulin-like growth factor 1 stimulates renal epithelial Na+ transport. Am J Physiol. 1988;255:C413–C417. doi: 10.1152/ajpcell.1988.255.3.C413. [DOI] [PubMed] [Google Scholar]
- 37.Blazer-Yost BL, Cox M, Furlanetto R. Insulin and IGF I receptor-mediated Na+ transport in toad urinary bladders. Am J Physiol. 1989;257:C612–C620. doi: 10.1152/ajpcell.1989.257.4.C612. [DOI] [PubMed] [Google Scholar]
- 38.Marunaka Y, Eaton DC. Effects of insulin and phosphatase on a Ca2+-dependent Cl− channel in a distal nephron cell line (A6) J Gen Physiol. 1990;95:773–789. doi: 10.1085/jgp.95.5.773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Marunaka Y, Hagiwara N, Tohda H. Insulin activates single amiloride-blockable Na channels in a distal nephron cell line (A6) Am J Physiol Renal Physiol. 1992;263:F392–F400. doi: 10.1152/ajprenal.1992.263.3.F392. [DOI] [PubMed] [Google Scholar]
- 40.Blazer-Yost BL, Shah N, Jarett L, Cox M, Smith RM. Insulin and IGF1 receptors in a model renal epithelium: receptor localization and characterization. Biochem Int. 1992;28:143–153. [PubMed] [Google Scholar]
- 41.Marunaka Y, Niisato N, Shintani Y. Protein phosphatase 2B-dependent pathway of insulin action on single Cl− channel conductance in renal epithelium. J Membr Biol. 1998;161:235–245. doi: 10.1007/s002329900330. [DOI] [PubMed] [Google Scholar]
- 42.Marunaka Y, Niisato N, O’Brodovich H, Post M, Tanswell AK. Roles of Ca2+ and protein tyrosine kinase in insulin action on cell volume via Na+ and K+ channels and Na+/K+/2Cl− cotransporter in fetal rat alveolar type II pneumocyte. J Membr Biol. 1999;168:91–101. doi: 10.1007/s002329900500. [DOI] [PubMed] [Google Scholar]
- 43.Butterworth MB, Edinger RS, Frizzell RA, Johnson JP. Regulation of the epithelial sodium channel by membrane trafficking. Am J Physiol Renal Physiol. 2009;296:F10–F24. doi: 10.1152/ajprenal.90248.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Marunaka Y, Niisato N, Taruno A, Ohta M, Miyazaki H, Hosogi S, Nakajima K, Kusuzaki K, Ashihara E, Nishio K, Iwasaki Y, Nakahari T. Kubota T (2011) Regulation of epithelial sodium transport via epithelial Na+ channel. J Biomed Biotechnol. 2011;2011:978196. doi: 10.1155/2011/978196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kamynina E, Staub O. Concerted action of ENaC, Nedd4-2, and Sgk1 in transepithelial Na+ transport. Am J Physiol Renal Physiol. 2002;283:F377–F387. doi: 10.1152/ajprenal.00143.2002. [DOI] [PubMed] [Google Scholar]
- 46.Diakov A, Nesterov V, Mokrushina M, Rauh R, Korbmacher C. Protein kinase B alpha (PKBalpha) stimulates the epithelial sodium channel (ENaC) heterologously expressed in Xenopus laevis oocytes by two distinct mechanisms. Cell Physiol Biochem. 2010;26:913–924. doi: 10.1159/000324000. [DOI] [PubMed] [Google Scholar]
- 47.Aoi W, Hosogi S, Niisato N, Yokoyama N, Hayata H, Miyazaki H, Kusuzaki K, Fukuda T, Fukui M, Nakamura N, Marunaka Y. Improvement of insulin resistance, blood pressure and interstitial pH in early developmental stage of insulin resistance in OLETF rats by intake of propolis extracts. Biochem Biophys Res Commun. 2013;432:650–653. doi: 10.1016/j.bbrc.2013.02.029. [DOI] [PubMed] [Google Scholar]
- 48.Perkins FM, Handler JS. Transport properties of toad kidney epithelia in culture. Am J Physiol. 1981;241:C154–C159. doi: 10.1152/ajpcell.1981.241.3.C154. [DOI] [PubMed] [Google Scholar]
- 49.Handler JS, Preston AS, Perkins FM, Matsumura M, Johnson JP, Watlington CO. The effect of adrenal steroid hormones on epithelia formed in culture by A6 cells. Ann N Y Acad Sci. 1981;372:442–454. doi: 10.1111/j.1749-6632.1981.tb15493.x. [DOI] [PubMed] [Google Scholar]
- 50.Handler JS, Perkins FM, Johnson JP. Hormone effects on transport in cultured epithelia with high electrical resistance. Am J Physiol. 1981;240:C103–C105. doi: 10.1152/ajpcell.1981.240.3.C103. [DOI] [PubMed] [Google Scholar]
- 51.Niisato N, Ohta M, Eaton DC, Marunaka Y. Hypotonic stress upregulates beta- and gamma-ENaC expression through suppression of ERK by inducing MKP-1. Am J Physiol Renal Physiol. 2012;303:F240–F252. doi: 10.1152/ajprenal.00198.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Asano J, Niisato N, Nakajima K, Miyazaki H, Yasuda M, Iwasaki Y, Hama T, Dejima K, Hisa Y, Marunaka Y. Quercetin stimulates Na+/K+/2Cl− cotransport via PTK-dependent mechanisms in human airway epithelium. Am J Respir Cell Mol Biol. 2009;41:688–695. doi: 10.1165/rcmb.2008-0338OC. [DOI] [PubMed] [Google Scholar]
- 53.Diener M, Scharrer E. Effects of short-chain fatty acids on cell volume regulation and chloride transport in the rat distal colon. Comp Biochem Physiol A Physiol. 1997;118:375–379. doi: 10.1016/S0300-9629(96)00322-2. [DOI] [PubMed] [Google Scholar]
- 54.Chen B, Nicol G, Cho WK. Role of calcium in volume-activated chloride currents in a mouse cholangiocyte cell line. J Membr Biol. 2007;215:1–13. doi: 10.1007/s00232-007-9000-9. [DOI] [PubMed] [Google Scholar]
- 55.Niisato N, Marunaka Y. Activation of the Na+-K+ pump by hyposmolality through tyrosine kinase-dependent Cl− conductance in Xenopus renal epithelial A6 cells. J Physiol. 1999;518(Pt 2):417–432. doi: 10.1111/j.1469-7793.1999.0417p.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Niisato N, Ito Y, Marunaka Y. Activation of Cl− channel and Na+/K+/2Cl− cotransporter in renal epithelial A6 cells by flavonoids: genistein, daidzein, and apigenin. Biochem Biophys Res Commun. 1999;254:368–371. doi: 10.1006/bbrc.1998.9952. [DOI] [PubMed] [Google Scholar]
- 57.Sun H, Niisato N, Marunaka Y. A role of PI3 kinase in insulin-induced enhancement of cAMP-stimulated Cl− secretion in renal epithelia A6 cells. J Physiol Sci. 2013;63:S197. [Google Scholar]
- 58.Niisato N, Driessche WV, Liu M, Marunaka Y. Involvement of protein tyrosine kinase in osmoregulation of Na+ transport and membrane capacitance in renal A6 cells. J Membr Biol. 2000;175:63–77. doi: 10.1007/s002320001055. [DOI] [PubMed] [Google Scholar]
- 59.Ueda-Nishimura T, Niisato N, Miyazaki H, Naito Y, Yoshida N, Yoshikawa T, Nishino H, Marunaka Y. Synergic action of insulin and genistein on Na+/K+/2Cl− cotransporter in renal epithelium. Biochem Biophys Res Commun. 2005;332:1042–1052. doi: 10.1016/j.bbrc.2005.05.046. [DOI] [PubMed] [Google Scholar]
- 60.Markadieu N, Crutzen R, Blero D, Erneux C, Beauwens R. Hydrogen peroxide and epidermal growth factor activate phosphatidylinositol 3-kinase and increase sodium transport in A6 cell monolayers. Am J Physiol. 2005;288:F1201–F1212. doi: 10.1152/ajprenal.00383.2004. [DOI] [PubMed] [Google Scholar]
- 61.Longo N (1996) Insulin stimulates the Na+, K+ -ATPase and the Na+/K+/Cl− cotransporter of human fibroblasts. Biochim Biophys Acta 1281:38–44 [DOI] [PubMed]
- 62.Sargeant RJ, Liu Z, Klip A. Action of insulin on Na+-K+-ATPase and the Na+-K+-2Cl− cotransporter in 3T3-L1 adipocytes. Am J Physiol. 1995;269:C217–C225. doi: 10.1152/ajpcell.1995.269.1.C217. [DOI] [PubMed] [Google Scholar]
- 63.Zhao H, Hyde R, Hundal HS. Signalling mechanisms underlying the rapid and additive stimulation of NKCC activity by insulin and hypertonicity in rat L6 skeletal muscle cells. J Physiol. 2004;560:123–136. doi: 10.1113/jphysiol.2004.066423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tse WK, Au DW, Wong CK. Effect of osmotic shrinkage and hormones on the expression of Na+/H+ exchanger-1, Na+/K+/2Cl− cotransporter and Na+/K+-ATPase in gill pavement cells of freshwater adapted Japanese eel, Anguilla japonica. J Exp Biol. 2007;210:2113–2120. doi: 10.1242/jeb.004101. [DOI] [PubMed] [Google Scholar]
- 65.O’Mahony F, Toumi F, Mroz MS, Ferguson G, Keely SJ. Induction of Na+/K+/2Cl− cotransporter expression mediates chronic potentiation of intestinal epithelial Cl− secretion by EGF. Am J Physiol Cell Physiol. 2008;294:C1362–C1370. doi: 10.1152/ajpcell.00256.2007. [DOI] [PubMed] [Google Scholar]
- 66.Panet R, Atlan H. Stimulation of bumetanide-sensitive Na+/K+/Cl− cotransport by different mitogens in synchronized human skin fibroblasts is essential for cell proliferation. J Cell Biol. 1991;114:337–342. doi: 10.1083/jcb.114.2.337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Maki M, Miyazaki H, Nakajima K, Yamane J, Niisato N, Morihara T, Kubo T, Marunaka Y. Chloride-dependent acceleration of cell cycle via modulation of Rb and cdc2 in osteoblastic cells. Biochem Biophys Res Commun. 2007;361:1038–1043. doi: 10.1016/j.bbrc.2007.07.131. [DOI] [PubMed] [Google Scholar]
- 68.Silvis MR, Bertrand CA, Ameen N, Golin-Bisello F, Butterworth MB, Frizzell RA, Bradbury NA. Rab11b regulates the apical recycling of the cystic fibrosis transmembrane conductance regulator in polarized intestinal epithelial cells. Mol Biol Cell. 2009;20:2337–2350. doi: 10.1091/mbc.E08-01-0084. [DOI] [PMC free article] [PubMed] [Google Scholar]


