Abstract
Platelet mitochondrial MAO-A activity of male albino rats (Wistar strain) was significantly inhibited with an inhibition of its only V max during aging. This age-induced inhibition of platelet MAO-A activity became reversed following the application of higher dosages (2.0–2.5 μg/kg/day, i.t. for 21 consecutive days) of carnosine. Though carnosine at lower dosage (0.5 μg/kg/day, i.t. for 21 consecutive days) was ineffective to platelet mitochondrial MAO-A activity in both young and aged rats, at higher dosages (2.0–2.5 μg/kg/day, i.t. for 21 consecutive days) under similar condition this enzyme activity was significantly enhanced. Carnosine at 1.0 μg/kg/day, i.t. for 21 consecutive days significantly enhanced MAO-A activity only in aged (18 and 24 months) rats. These results suggest that carnosine withdraws the aging-induced inhibition of mammalian blood platelet MAO-A activity and restores its activity towards that (MAO-A activity) observed in young mammalian blood platelets.
Keywords: Blood platelet, Monoamine oxidase-A (MAO-A), Aging, Carnosine
Introduction
Platelet is an important blood component, having a plasma membrane, platelet contractile protein surrounded by a glycocalyx outer layer, mitochondria and other organelles without a nucleus inside [1]. The monoamine oxidase (MAO) which oxidizes monoamines [2] is a platelet mitochondrial outer membrane bound enzyme [E.C. 1.4.3.4] [1, 3]. MAO, depending on substrate specificities and inhibitor sensitivities, has been classified into two classes: MAO-A and MAO-B [4]. Platelet MAO activity has been found to be reduced [5] in chronic neurodegenerative disorders like schizophrenia [6], but in Alzheimer which is also a neurodegenerative disease, platelet MAO activity is increased [7, 8]. Alexopoulos et al. [8] have shown that brain and platelet MAO activity is increased without affecting their MAO-A activity. The platelet MAO-A activity of bipolar depressed patients [9] and in children with hyperactive, inattentive and combined subtype of attention deficit hyperactive disorder (ADHD) [10] has been also shown to be reduced.
Carnosine, a dipeptide (β-Ala-l-His), an endogenous anti-aging and anti-glycating biomolecule [11] is present in muscle, brain tissue and circulation [12, 13]. It is metabolized by carnosinase (a degradative enzyme) and carnosine synthase (a synthesizing enzyme) [14]. Both these enzymes are present in blood, brain and other tissues [14–18]. Further it has been known that there are two forms of carnosinase. One form is called serum carnosinase [17] which is present in serum, cerebrospinal fluid (CSF) and brain, and the other one is tissue carnosinase which is present in liver, spleen and kidney as a non-specific cytosolic dipeptidase [18]. It is also known that serum carnosinase activity is increased and that brain regional carnosine is reduced with aging [19, 20]. In brain and other tissues including plasma, carnosine related compounds (CRCs; e.g. homocarnosine, anserine and carnosine metabolites) have been also found [12]. Carnosine has been considered a mobile organic pH buffer [21] and a metal chelator [12]. It has a potential involvement in gene regulation or signal transduction [22] as it binds with calcium and purines [23]. Mc Farland [24] has shown that carnosine rejuvenates the senescent morphology of cultured human diploid fibroblast cells, maintaining its cellular integrity by its modulatory protein metabolism while retarding its senescence. Advanced glycation end products (AGEs) are an age associated phenomenon produced by the reaction with sugar aldehyde and protein amino groups. Margles [19] has shown that the olfactory bulb contains a high concentration of carnosine, and loss of sense of smell has been thought to be an indicator of Alzheimer’s disease as the carnosine content is reduced in olfactory bulb under this condition. Like different brain regions, the reduction of carnosine content has also been found in muscle tissues [25]. In addition, Margles [19] has shown that carnosine inhibits sugar-induced β-A4-amyloidogenic peptide aggregation. Carnosine and its related compounds have also been shown to be a powerful hydrophilic reactive oxygen species (ROS) scavenger [12] and it (carnosine) plays a role as an endogenous agent to protect against glutamate excitotoxicity which is secondary to ROS generation [26].
Since (a) blood platelet count and platelet serotonin are increased in dementia [27] which may stimulate megakaryocytopoesis [28] and enhancing the risk of platelet aggregation [29], (b) change in platelet MAO activity in neurodegeneration [5–10], (c) carnosine is an endogenous anti-aging and anti-glycating agent [11] and (d) its (carnosine) degradative enzyme serum carnosinase activity is known to be activated with a reduction of brain regional carnosine level during aging or age-induced diseases [19, 20], it is not unreasonable to assume that this molecule (carnosine) may have an effect on blood platelet MAO activity, specifically MAO-A in different ages of rats (4–24 months). The present study deals with the effect of carnosine on blood platelet mitochondrial MAO-A activity, including its kinetic behavior during aging.
Methods
Materials
5-Hydroxytryptamine (5-HT)-HCl, l-Carnosine and Triton X-100 were purchased from Sigma chemicals (St.Louis, MO, USA). All other chemicals, including ethylenediaminetetraacetic acid (EDTA), sodium–potassium tartarate, copper sulfate, sodium hydroxide, sodium carbonate, potassium dihydrogen phosphate, sodium hydrogen phosphate and semicarbazide of analytical grade were purchased from Merck-India (Worli-Mumbai), India.
Animals
Male albino rats of Wistar strain were used as the experimental animal. The rats were maintained in a room having a 12 h light–dark cycle and temperature 28 ± 0.5 °C with a constant relative humidity (80 ± 5 %). Animals were supplemented with a normal standard laboratory diet and water ad libitum. In the present study the guidelines of the animal ethical committee (Department of Biochemistry, University of Calcutta) were followed and all efforts were made to minimize the number of animals used and their suffering.
Experimental procedures
Male albino rats of different ages (4, 18 and 24 months) were housed in different cages. Each of these three different age groups of animals was divided into two subgroups (subgroup 1A, 1B; subgroup 2A, 2B and subgroup 3A, 3B; n = 4–6 animals in each subgroup). The rats of subgroups 1B, 2B and 3B were treated intrathecally (i.t.) with carnosine at various dosages (0.5, 1.0, 2.0 and 2.5 μg/kg/day) for 21 consecutive days. The rats of subgroups 1A, 2A and 3A were considered as control groups of the corresponding above-mentioned experimental groups. These control groups were treated with equivalent amount of vehicle (20 μl saline solution) of carnosine through the same route under similar conditions as mentioned in the corresponding experimental group of animals. The experimental and control groups of animals were sacrificed after 4 h of last administration between 9 and 10 a.m. to avoid the circadian effect, if any.
Collection of blood and preparation of platelet rich plasma (PRP)
Immediately after sacrifice of both control and experimental rats of different age groups, the blood (with anticoagulant, 1 % EDTA solution) was collected under cold (0–4 °C) conditions. The platelet rich plasma was isolated from the blood by following the method of Collins and Sandler [30], modified by Banerji et al. [31], and this was used as an enzyme source.
Assay of monoamine oxidase-A (MAO-A) activity
Monoamine oxidase-A (MAO-A) was measured using 0.24 mM 5-HT (serotonin) as substrate with 100 μg enzymes according to the method described by Dalal and Poddar [32]. The kinetics study of MAO-A was measured with varying concentrations (0.05–0.4 mM per 100 μg enzyme) of serotonin. The protein content was estimated spectrophotometrically by following the method of Lowry et al. [33] using bovine serum albumin (BSA) as standard.
Statistical analysis
All data are expressed as the mean ± SEM and the number of rats (n) is indicated. The averaged values from the three controls were compared with each of the experimental animals. Statistical analyses of the data were assessed by analysis of variance (ANOVA) with a post hoc Tukey’s test. p < 0.05 was considered as significant.
Results
Changes of platelet MAO-A activity of rats during aging with carnosine
Figure 1 depicts that the platelet MAO-A activity was significantly reduced (48.77–56.79 %, p < 0.001) with increase of age (4–24 months) with respect to the young (4 months) healthy group of rats (Fig. 1a). The maximum inhibition was observed at 24 months of age. Figure 1b represents that carnosine at lower dosage (0.5 μg/kg/day, i.t. for 21 consecutive days) did not significantly affect the platelet MAO-A activity with respect to the corresponding different age-matched (18 and 24 months) vehicle treated rats. Further increase in dosage of carnosine (1.0–2.5 μg/kg/day, i.t.) under similar conditions significantly (p < 0.001) enhanced platelet MAO-A activity in aged rats of both 18 months (44.58–101.20 %) and 24 months (72.86–145.71 %); whereas in young (4 months) rats the MAO-A activity was significantly (p < 0.001) enhanced only at the dosages of 2.0 μg/kg/day, (28.40 %) and 2.5 μg/kg/day, (33.33 %) with respect to the corresponding vehicle treated age-matched control group of rats. It was also observed that carnosine (2.5 μg/kg/day, i.t. for 21 consecutive days) induced increase (33.33 %, p < 0.001) of platelet MAO-A activity in young (4 months) rats was potentiated to 101.20 and 145.71 % with increase of age (from 4 to 18 and 24 months respectively; Fig. 1b). The results of Fig. 1a and b further show that carnosine (2.0 and 2.5 μg/kg/day, i.t.) under similar condition attenuated the age-induced decrease (48.77–56.79 %) of platelet MAO-A activity (Fig. 1a) to 32.21–23.56 and 22.69–20.37 %, respectively.
Fig. 1.
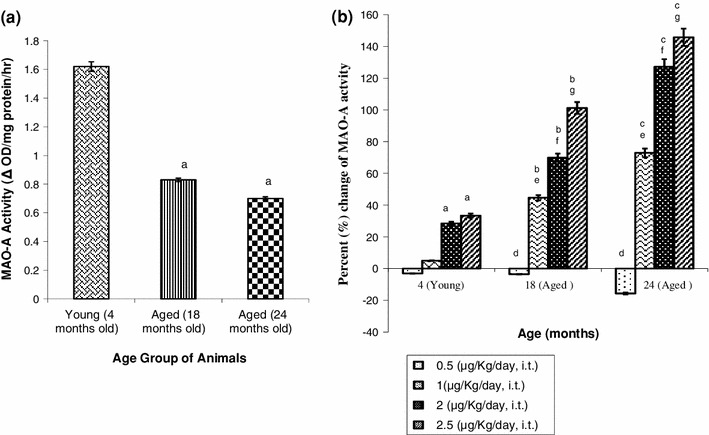
Effect of carnosine on rat blood platelet MAO-A activity during aging of male rats. Results are expressed as mean ± SEM (n = 4–6). Each observation was made from a single rat. Change (%) was calculated with respect to the corresponding age-matched control group of rats (4, 18 and 24 months). a Significantly different from corresponding control were: (i) young (4 months) rats a p < 0.001, (ii) aged (18 months) rats b p < 0.001, (iii) aged (24 months) rats c p < 0.001. b Significantly different from corresponding carnosine treated (dose/day for 21 consecutive days, i.t.) were: young (4 months) rats (i) 0.5 μg/kg d p < 0.001, (ii) 1.0 μg/kg e p < 0.001, (iii) 2.0 μg/kg f p < 0.001, (iv) 2.5 μg/kg g p < 0.001
Changes in kinetic parameters of platelet MAO-A activity in rats during aging with carnosine
It is evident from Table 1 and Fig. 2b that the kinetic parameter, V max of platelet MAO-A activity was significantly reduced with the increase of age to 18 months (45.10 %, p < 0.001) and 24 months (54.90 %, p < 0.001) of rats without any significant effect to their corresponding K m with respect to the corresponding young (4 months) rats. Table 1 also shows that the treatment of carnosine (2.0 μg/kg/day, i.t.) significantly enhanced V max (59.82 and 117.39 %, p < 0.001) of platelet MAO-A activity with an apparent change in corresponding K m in aged [18 months (−13.04 %) and 24 months (−4.55 %)] rats. Unlike aged rats, in young (4 months) rats carnosine at the same dosage significantly increased the V max (28.92 %, p < 0.001) without affecting the K m value of platelet MAO-A activity with respect to the corresponding vehicle treated young rats. It was further observed that in the carnosine treated aged (18 and 24 months) rats the V max values of platelet MAO-A activity still remained reduced (31.94 and 23.95 %, p < 0.001, respectively) but without any significant effect in their corresponding K m value with respect to the corresponding young (4 months) group.
Table 1.
Effect of carnosine on kinetic parameters of blood platelet MAO-A activity during aging of male rats
| Age groups | Age of animals (months) | Kinetic parameters of MAO-A activity | |||
|---|---|---|---|---|---|
| Control (vehicle) | Carnosine (μg/kg/day, i.t. for 21 consecutive days) | ||||
| V max (ΔOD/mg protein/h) | K m (mM) | V max (ΔOD/mg protein/h) | K m (mM) | ||
| Young | 4 | 2.04 ± 0.09 | 0.21 ± 0.009 | 2.63 ± 0.02a | 0.20 ± 0.004 |
| Aged | 18 | 1.12 ± 0.05a | 0.23 ± 0.01 | 1.79 ± 0.03b,d | 0.21 ± 0.005 |
| 24 | 0.92 ± 0.05a | 0.22 ± 0.01 | 2.00 ± 0.06c,d | 0.21 ± 0.003 | |
Results are expressed as mean ± SEM (n = 4–6). V max and K m values were calculated from Line–Weaver Burk plot (Fig. 2b)
Kinetic studies were carried out using varying concentrations (0.05–0.4 mM) of serotonin
Significant differences from (a) corresponding control rats of (i) young (4 months) a p < 0.001, (ii) aged (18 months) b p < 0.001, (iii) aged (24 months) c p < 0.001. Significant differences from (b) corresponding carnosine treated rats (2.0 μg/kg/day, i.t.) (i) young (4 months) d p < 0.001
Fig. 2.
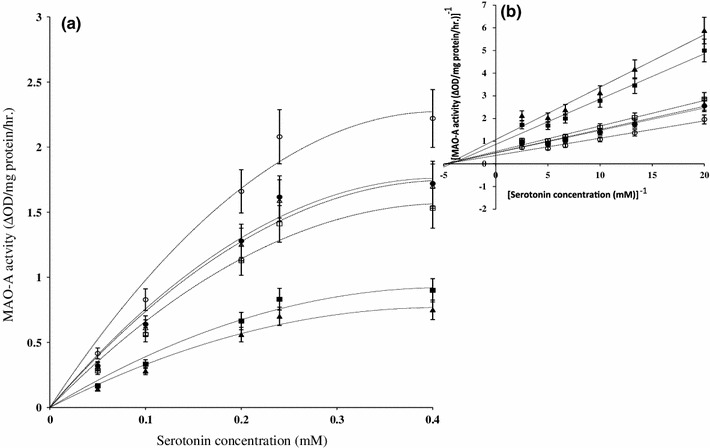
a Effect of serotonin concentration variation (0.05–0.4 mM) on platelet MAO-A activity. Results are expressed as mean ± SEM (n = 4–6). Each observation was made from a single rat. Line (–) represents control, filled circle young (4 months) rats, filled square aged (18 months) rats, filled triangle aged (24 months) rats. Broken line (---) indicates carnosine treated, open circle young (4 months) rats, open square aged (18 months) rats, open triangle aged (24 months) rats. b Line–Weaver Burk plot of blood platelet MAO-A activity in different ages of rats. Line–Weaver Burk plots were drawn from the progress curve with varying substrate concentration as presented in a
Discussion
Monoamine oxidase (MAO) is known to play an important role in the metabolism of biogenic amines in both neuronal and non-neuronal tissues [34]. In non-neuronal tissues it plays a protective influence over the harmful effect of circulating monoamines [35]. This enzyme exists in two isoforms, MAO-A and MAO-B [4], in different tissues including blood platelet mitochondria [1, 3]. Aging is a naturally developing biological process which reduces life span and promotes age pathology. Since MAO-A is serotonin substrate specific [4] and the authors are interested about the involvement of the serotonergic system in relation to aging the present study deals with the effect of carnosine, an endogenous biomolecule having anti-glycating and anti-aging properties [11], on blood platelet mitochondrial MAO-A activity during aging.
In our present study the blood platelet mitochondrial MAO-A activity has been found to be reduced with the advancement of age (Fig. 1a), with a significant reduction in V max (45.10–54.90 %) without affecting its corresponding K m (Table 1; Fig. 2). This suggests that there may be a quantitative as well as qualitative modulation by unmasking the substrate binding site and a significant quantitative inhibition of catalytic activity of platelet mitochondrial enzyme with a destabilization of the blood platelet mitochondrial membrane [31] during aging. The present age-induced reduction in MAO-A activity may be supported by others’ reports which have shown that platelet MAO activity reduces [5–10] during age-induced neurodegeneration and neurodegenerative diseases like schizophrenia, ADHD, bipolar depression and dementia. This may be due to the dysfunction of platelet mitochondria at the level of its structure and function [36–38]. The inhibition (Fig. 1a) of age-induced platelet mitochondrial MAO-A activity may be corroborated with the observations of others who have shown that the activity of platelet mitochondrial other enzyme, cytochrome oxidase and the mitochondrial oxydative phosphorylation are reduced with increase of age [39, 40]. In addition, Xu et al. [41] have shown that platelet number and mitochondrial membrane potential (Δψ m) are decreased with aging as an effect of amyloid-beta-induced change in platelet mitochondrial function due to age and the down regulation of mitochondrial biogenesis, which has been shown to be associated with the mitochondrial dysfunction during aging [36–38]. It is well known that stress plays as one of the age-induced factors at the hormonal level and glucocorticoid is one of them [42]. It is also known that stress activates MAO-A activity at the level of its transcription and translation [43] in nucleated tissues like heart [44] and brain [45]. As platelet is an enucleated tissue [42], the role of this stress-induced hormone in platelet mitochondrial MAO-A inhibition does not arise. In addition, the MAO-A gene is present on chromosome X (Xp 11.23) [46].
MAO is known to play in the metabolism of biogenic amines and regulation of neurotransmitter levels as well as intracellular amine stores [47], however in the gastrointestinal tract and circulatory system they (biogenic amines) serve a protective function by regulating monoamine levels with potent vasopressor effects [48]. Therefore, it may be suggested that the reduction in blood platelet MAO-A activity in aged rats (Fig. 1a) may increase the platelet serotonin levels [26] and increase the platelet activating factor content [49] which may stimulate megakaryocytopoesis [27] by stimulating platelet serotonin release [50] and causing platelet aggregation [29] and hence vascular blockage [51] with the advancement of age. Further it is not unlikely to assume that the reduction of platelet MAO-A activity with increase of age (Fig. 1a) may be due to an age-induced structural and functional deterioration of the platelet mitochondrial membrane [36–41] as an effect of depletion in platelet population during aging [41], though this needs further clarification with a platelet mitochondrial preparation under similar conditions.
The present study (Fig. 2; Table 1) has shown that the application of higher dosage (2.0 μg/kg/day, i.t.) of carnosine for 21 consecutive days reverses the age-induced inhibition of blood platelet MAO-A activity and restores it towards the activity of MAO-A observed in young adult (4 months; Fig. 1b). The analysis of kinetic parameters of MAO-A activity (Table 1) suggests that the age-induced inhibition in blood platelet MAO-A activity is significantly attenuated with carnosine (2.0 μg/kg/day, i.t. for 21 consecutive days) at the level of its up regulation in only V max (Table 1; Fig 2b). This attenuating effect of carnosine on age-induced inhibition of platelet MAO-A activity may be supported by its enhancing effect on both young and aged animals (Fig. 1a). In fact, carnosine in both young and aged rats enhances only the catalytic activity (V max) of MAO-A enzyme without affecting its substrate binding affinity (1/K m; Table 1), suggesting that carnosine in both ages may act in a dual way with quantitative increases as well as qualitative changes [52, 53] of this platelet mitochondrial enzyme. This present thought may further be strengthened by age-induced potentiation on its (carnosine) enhancing effect on platelet MAO-A activity (Fig. 1b). Also it may be stated that carnosine acts at the level of mitochondrial biogenesis and attenuates the age-induced platelet mitochondrial structural and functional deterioration [36–41] as well as platelet population [41] within the cell and restores its mitochondrial MAO-A activity towards the activity that has been observed in normal young rats (Fig. 1b) by upgrading the vascular system towards a normal condition during aging.
Finally, it may be concluded that: (a) carnosine may have a great role to (i) protect age-induced platelet aggregation by reversing the age-induced reduction of platelet mitochondrial MAO-A activity and hence enhancement of its (platelet) serotonin content, (ii) restore the age-induced depletion in protective function of the circulatory system with the up regulation of reduced platelet MAO-A activity during aging and hence attenuating its (monoamines) potent vasopressor effects; (b) platelet MAO-A activity may be considered as a diagnostic index in the clinical practice of geriatrics.
Acknowledgments
The authors are thankful to Prof. S. Sengupta, Department of Statistics, University of Calcutta, for helping the statistical analyses of the data. The present study was supported by a grant from UGC-UPE, University of Calcutta, Kolkata, India.
Conflict of interest
None.
References
- 1.Tipton KF, Boyce S, O’ Sullivan J, Davey GP, Healy J. Monoamine oxidases: certainties and uncertainties. Curr Med Chem. 2004;11:1965–1982. doi: 10.2174/0929867043364810. [DOI] [PubMed] [Google Scholar]
- 2.Benedetti MS, Keane PE. Differential changes in monoamine oxidase A and B activity in the aging rat brain. J Neurochem. 1980;35:1026–1032. doi: 10.1111/j.1471-4159.1980.tb07856.x. [DOI] [PubMed] [Google Scholar]
- 3.Edmondson DE, Mattevi A, Binda C, Li M, Hubalck F. Structure and mechanism of monoamine oxidase. Curr Med Chem. 2004;11:1983–1993. doi: 10.2174/0929867043364784. [DOI] [PubMed] [Google Scholar]
- 4.Johnston JP. Some observations upon a new inhibitor of monoamine oxidase in brain tissue. Biochem Pharmacol. 1968;17:1285–1297. doi: 10.1016/0006-2952(68)90066-X. [DOI] [PubMed] [Google Scholar]
- 5.Del VM, Maj M, D’Ambrosio A, Kemali D. Low platelet MAO activity in chronic schizophrenics: a long-term effect of neuroleptic treatment? Psychopharmacology. 1983;79:177–179. doi: 10.1007/BF00427807. [DOI] [PubMed] [Google Scholar]
- 6.Baron M, Levitt M, Perlman R. Low platelet monoamine oxidase activity: a possible biochemical correlate of borderline schizophrenia. Psychiatry Res. 1980;3:329–335. doi: 10.1016/0165-1781(80)90063-3. [DOI] [PubMed] [Google Scholar]
- 7.Adolfsson R, Gottfries CG, Oreland L, Wiberg A, Winblad B. Increased activity of brain and platelet monoamine oxidase in dementia of Alzheimer type. Life Sci. 1980;27:1029–1034. doi: 10.1016/0024-3205(80)90025-9. [DOI] [PubMed] [Google Scholar]
- 8.Alexopoulos GS, Liberman KW, Yang RC. Platelet MAO activity in primary degenerative dementia. Am J Psychiatry. 1984;141:97–99. doi: 10.1176/ajp.141.1.97. [DOI] [PubMed] [Google Scholar]
- 9.Bongioanni P, Mondino C, Boccardi B, Borgna M, Castagna M. Monoamine oxidase molecular activity in platelets of Parkinsonian and demented patients. Neurodegeneration. 1996;5:351–357. doi: 10.1006/neur.1996.0047. [DOI] [PubMed] [Google Scholar]
- 10.Parnetti L, Reboldi GP, Santucci C, Santucci A, Gaiti A, Brunetti M, Cecchetti R, Senin U. Platelet MAO-B activity as a marker of behavioral characteristics in dementia disorders. Aging Milano. 1994;6:201–207. doi: 10.1007/BF03324240. [DOI] [PubMed] [Google Scholar]
- 11.Holiday R, McFarland GA. A role for carnosine in cellular maintenance. Biochemistry (Mosc) 2000;65:843–848. [PubMed] [Google Scholar]
- 12.Kohen R, Yammamoto Y, Cundy KC, Ames BN. Antioxidant activity of carnosine, homocarnosine and anserine present in muscle and brain. Proc Natl Acad Sci USA. 1988;85:3175–3179. doi: 10.1073/pnas.85.9.3175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Seely JE, Marshall FD. Carnosine levels in blood. In. Experientia. 1981;37:1256–1257. doi: 10.1007/BF01948344. [DOI] [PubMed] [Google Scholar]
- 14.Wim D, Inge E, Sam B, Audrey B. Muscle carnosine metabolism and β-alanine supplementation in relation to exercise and training. Sports Med. 2010;40:247–263. doi: 10.2165/11530310-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 15.Ng RH, Marshall FD. Subcellular distribution and some properties of homocarnosine-carnosine synthetase from chick red blood cells. Comp Biochem Physiol B. 1976;54:523–525. doi: 10.1016/0305-0491(76)90133-4. [DOI] [PubMed] [Google Scholar]
- 16.Lunde HA, Lenney JF, Halvorsen S, Gjessing L. Serum carnosine in blood and homocarnosine in CSF of patients with Folling’s disease (PKU) J Oslo City Hosp. 1987;37:29–33. [PubMed] [Google Scholar]
- 17.Jackson MC, Kucera CM, Lenney JF. Purification and properties of human serum carnosinase. Clin Chim Acta. 1991;196:193–205. doi: 10.1016/0009-8981(91)90073-L. [DOI] [PubMed] [Google Scholar]
- 18.Peppers SC, Lenney JF. Bestatin inhibition of human tissue carnosinase, a non-specific cytosolic dipeptidase. Biol Chem Hoppe Seyler. 1988;369:1281–1286. doi: 10.1515/bchm3.1988.369.2.1281. [DOI] [PubMed] [Google Scholar]
- 19.Margles FL. Carnosine in the primary olfactory pathway. Science. 1994;184:909–911. doi: 10.1126/science.184.4139.909. [DOI] [PubMed] [Google Scholar]
- 20.Bellia F, Calabrese V, Guarino F, Cavallaro M, Cornelius C, De Pinto V, Rizzarelli E. Carnosinase levels in aging brain: redox state induction and cellular stress response. Antioxid Redox Signal. 2009;11:2759–2775. doi: 10.1089/ars.2009.2738. [DOI] [PubMed] [Google Scholar]
- 21.Boldyrev AA, Kurella EG, Stovoliusky SL. Biological role of carnosine metabolism in excitable tissues: speculations and facts (a commentary) Pathophysiology. 1994;1:215–219. doi: 10.1016/0928-4680(94)90040-X. [DOI] [Google Scholar]
- 22.Quinn PJ, Boldyrev AA, Formazuyk VE. Carnosine, its properties, functions and potential therapeutic applications. Mol Asp Med. 1992;13:379–444. doi: 10.1016/0098-2997(92)90006-L. [DOI] [PubMed] [Google Scholar]
- 23.Neurhro KJ, Mantiesh HH. Complete formation of carnosine with purine nucleotides in aqueous solution. Z Naturforsch C. 1979;35:557–561. doi: 10.1515/znc-1980-7-806. [DOI] [PubMed] [Google Scholar]
- 24.Mc Farland GA, Holiday R. Retardation of senescence of cultured human fibroblasts carnosine. Exp Cell Res. 1994;212:167–175. doi: 10.1006/excr.1994.1132. [DOI] [PubMed] [Google Scholar]
- 25.Stuerenburg HJ, Kunze K. Concentrations of free carnosine (a putative membrane-protective antioxidant) in human muscle biopsies and rat muscle. Arch Gerentol Geriatr. 1999;29:107–113. doi: 10.1016/S0167-4943(99)00020-5. [DOI] [PubMed] [Google Scholar]
- 26.Boldyrev A, Song R, Lowrence D, Carpenter DO. Carnosine protects against excitotoxicity cell death independently of effects on reactive oxygen species. Neuroscience. 1999;94:571–577. doi: 10.1016/S0306-4522(99)00273-0. [DOI] [PubMed] [Google Scholar]
- 27.Meszaros Z, Borcsiozky D, Mate M, Tarcali J, Szombathy T, Mekes K, Magyar K. Platelet MAO-B activity and serotonin content in patients with dementia: effect of age, medication and disease. Neurochem Res. 1998;23:863–868. doi: 10.1023/A:1022458928442. [DOI] [PubMed] [Google Scholar]
- 28.Yang M, Srikiatkhachorn A, Anthony M, Chong BH. Serotonin stimulates megakaryocytopoesis via the 5HT-2 receptor. Blood Coagul Fibrinolysis. 1996;7:127–133. doi: 10.1097/00001721-199603000-00004. [DOI] [PubMed] [Google Scholar]
- 29.Lopez-Vilchez I, Diaz-Ricart M, White JG, Escolar G, Galan AM. Serotonin enhances platelet procoagulant properties and their activation induced during platelet tissue factor uptake. Cardiovascular Res. 2009;84:309–316. doi: 10.1093/cvr/cvp205. [DOI] [PubMed] [Google Scholar]
- 30.Collins GGS, Sandler M. Human blood platelet MAO. Biochem Pharmacol. 1971;20:289–297. doi: 10.1016/0006-2952(71)90063-3. [DOI] [PubMed] [Google Scholar]
- 31.Banerji A, Poddar MK, Ghosh JJ. Action of Δ9-tetrahydrocannabinol on membrane-bound monoamine oxidase activity. Toxicol Appl Pharmacol. 1977;40:347–354. doi: 10.1016/0041-008X(77)90106-5. [DOI] [PubMed] [Google Scholar]
- 32.Dalal A, Poddar MK. Involvement of high plasma corticosteron status and activation of brain regional serotonin metabolism in long-term erythrosine induced rearing motor hyperactivity in young adult male rats. Toxicol Mech Methods. 2010;20:287–297. doi: 10.3109/15376516.2010.483070. [DOI] [PubMed] [Google Scholar]
- 33.Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with folin-phenol reagent. J Biol Chem. 1951;193:265–275. [PubMed] [Google Scholar]
- 34.Saura J, Richards JG, Mahy N. Age-related changes on MAO in B1/C57 mouse tissues: a quantitative radiographic study. J Neural Transm Suppl. 1994;41:89–94. doi: 10.1007/978-3-7091-9324-2_11. [DOI] [PubMed] [Google Scholar]
- 35.Holschneider DP, Shih JC (2000) Monoamine oxidase: basic and clinical perspectives. Neuropsychopharmacology: the fifth generation of progress. http://www.acnp.org/g4/gn401000046/ch046.html
- 36.Jon V, Keith DC, Richard C, Marwan NS, Geoffrey LA, Leslie B, Gene A, Douglas GW, Eric MR. Impaired platelet mitochondrial activity in Alzheimer’s disease and mild cognitive impairment. Mitochondrion. 2006;6:323–330. doi: 10.1016/j.mito.2006.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chun S, Kaihua G, David TY, Zhibin Y, Lucy Forster E, Huaqiao W, Jie Xu. Effects of aging and Alzheimer’s disease on mitochondrial function of human platelets. Exp Gerentol. 2008;43:589–594. doi: 10.1016/j.exger.2008.02.004. [DOI] [PubMed] [Google Scholar]
- 38.Lenaz G, D’Aurelio M, Merlo Pich M, Genova ML, Ventura C, Bovina C, Formiggini G, Parenti Castelli G. Mitochondrial bioenergetics in aging. Biochem Biophys Acta. 2000;1459:397–404. doi: 10.1016/S0005-2728(00)00177-8. [DOI] [PubMed] [Google Scholar]
- 39.Parker WD, Jr, Mahr NJ, Filley CM, Parks JK, Hughes D, Young DA, Cullum CA. Reduced platelet cytochrome oxidase activity in Alzheimer’s disease. Neurology. 1994;44:1086–1090. doi: 10.1212/WNL.44.6.1086. [DOI] [PubMed] [Google Scholar]
- 40.Aurelio Marilena D’, Milena MP, Lucia C, Gian LS, Carla B, Gabriella F, Giovanna PC, Harold B, Sante T, Giorgio L. Decreased Pasteur effect in platelets of aged individuals. Mech Ageing Dev. 2001;122:823–833. doi: 10.1016/S0047-6374(01)00239-1. [DOI] [PubMed] [Google Scholar]
- 41.Jie X, Shi C, Li Q, Lam WP, Wai MSM, Yew DT. Effects of β-amyloid peptide and estrogen on platelet mitochondrial function of Sprague-Dawley rats. Platelets. 2007;18:460–468. doi: 10.1080/09537100701206808. [DOI] [PubMed] [Google Scholar]
- 42.Fumagalli F, Jones SR, Caron MG, Seidler FJ, Slotkin TA. Expression of mRNA coding for the serotonin transporter in aged vs. young rat brain: differential effects of glucocorticoids. Brain Res. 1996;719:225–228. doi: 10.1016/0006-8993(96)00119-9. [DOI] [PubMed] [Google Scholar]
- 43.Xm Ou, Chen K, Shih JC. Glucocorticoid and androgen activation of monoamine oxidase A is regulated differently by R1 and SP1. J Biol Chem. 2006;281:21512–21525. doi: 10.1074/jbc.M600250200. [DOI] [PubMed] [Google Scholar]
- 44.Maurel A, Hernandez C, Kunduzova O, Bompart G, Cambon C, Parini A, Frances B. Age dependent increase in hydrogen peroxide production by cardiac monoamine oxidase A in rats. Am J Physiol Heart Circ Physiol. 2003;284:H1460–H1467. doi: 10.1152/ajpheart.00700.2002. [DOI] [PubMed] [Google Scholar]
- 45.Kabuto H, Yokoi I, Mori A, Murakami M, Sawada S. Neurochemical changes related to ageing in the senescence-accelerated mouse brain and the effects of chronic administration of nimodipine. Mech Ageing Dev. 1995;80:1–9. doi: 10.1016/0047-6374(94)01542-T. [DOI] [PubMed] [Google Scholar]
- 46.Zeirmans T, Dumontheil I, Roggeman C, Peyrard-Janvid M, Matsson H, Kere J, Klingberg T. Working memory, brain activity and capacity link MAO-A polymorphism to aggressive behavior during development. Transl Psychi. 2012;2:E85. doi: 10.1038/tp.2012.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Nunes SF, Figueiredo IV, Pereira JS, De Lemos ET, Reis F, Teixeira F, Caramona MM. Monoamine oxidase and semicarbazide-sensitive amine oxidase kinetic analysis in mesenteric arteries of patients with type 2 diabetes. Physiol Res. 2011;60:309–315. doi: 10.33549/physiolres.931982. [DOI] [PubMed] [Google Scholar]
- 48.Herriz T, Chaparro C. Analysis of monoamine oxidase enzymatic activity by reversed phase high performance liquid chromatography and inhibition by beta-carboline alkaloids occurring in foods and plants. J Chromatogr A. 2006;1120:237–243. doi: 10.1016/j.chroma.2005.12.009. [DOI] [PubMed] [Google Scholar]
- 49.Zhang X, Yuan CL, Zhang HZ, Huang RX. Age-related increase of plasma platelet-activating factor concentrations in Chinese. Clin Chim Acta. 2003;337:157–162. doi: 10.1016/j.cccn.2003.08.010. [DOI] [PubMed] [Google Scholar]
- 50.Hidetaka I, Yoshimi T, Kozo K, Makoto S, Yasutomi N. A role of calcium-activated, phospholipid-dependent protein kinase in platelet-activating factor-induced serotonin release from rabbit platelets. Biochem Biophys Res Commun. 1982;108:1701–1708. doi: 10.1016/S0006-291X(82)80107-1. [DOI] [PubMed] [Google Scholar]
- 51.Moore S. Platelet aggregation secondary to coronary obstruction. Circulation. 1976;53:166–169. [PubMed] [Google Scholar]
- 52.Gerhard M. Determination of Michaelis constants and inhibitor constants. In: Bergmeyer HU, editor. Methods of enzymatic analysis, vol 1, 2nd English edn. New York-San Francisco-London: Academic Press Inc; 1974. pp. 146–151. [Google Scholar]
- 53.Masciandaro GBC, Rouchi V. Kinetic parameters of dehydrogenase in the assessment of the response of soil to vermicompost and inorganic fertilizers. Biol Fert Soils. 2000;32:479–483. doi: 10.1007/s003740000280. [DOI] [Google Scholar]


