Abstract
Intermittent arm ischemia before percutaneous coronary intervention induces remote ischemic preconditioning (RIPC) and attenuates myocardial injury in patients with myocardial infarction. Several studies have shown that intermittent arm ischemia increases coronary flow and is related to autonomic nerve system. The aim of this study was to determine whether intermittent arm ischemia induces vasodilatation of other arteries and to assess changes in the autonomic nerve system during intermittent arm ischemia in humans. We measured change in the right brachial artery diameter during intermittent left arm ischemia through three cycles of 5-min inflation (200 mmHg) and 5-min deflation of a blood-pressure cuff using a 10-MHz linear array transducer probe in 20 healthy volunteers. We simultaneously performed power spectral analysis of heart rate. Ischemia-reperfusion of the left arm significantly dilated the right brachial artery time-dependently, resulting in a 3.2 ± 0.4% increase after the 3rd cycle. In the power spectral analysis of heart rate, the high-frequency domain (HF), which is a marker of parasympathetic activity, was significantly higher after the 3rd cycle of ischemia-reperfusion than baseline HF (P = 0.02). Intermittent arm ischemia was accompanied by vasodilatation of another artery and enhancement of parasympathetic activity. Those effects may play an important role in the mechanism of RIPC.
Keywords: Ischemic heart disease, Autonomic nervous system, Ischemia-reperfusion
Introduction
Remote ischemic preconditioning (RIPC) is a powerful innate mechanism by which brief ischemia in one region or organ protects distant tissue or organs from a sustained event of ischemia [1]. In 1993, Przyklenk et al. [2] reported for the first time that brief circumflex artery occlusion could reduce the size of myocardial infarct induced by subsequent sustained occlusion of the left anterior descending artery in dogs. This intramyocardial protection was later extended to non-cardiac organs, with a report that myocardial infarct size could be actually reduced in the animal heart by inducing brief ischemia and reperfusion in either the kidney [3, 4] or the small bowel [5]. In clinical settings, several studies have shown that RIPC by intermittent arm ischemia is useful in protection from myocardial injury after percutaneous coronary intervention [6, 7] and is controversial in myocardial protection after coronary artery bypass graft [8, 9].
Transient limb ischemia reduced coronary resistance and increased coronary blood flow in a porcine model [10]. In human cases, coronary diastolic flow velocity was also increased by 3 cycles of remote intermittent ischemia-reperfusion [11]. However, it is unclear whether intermittent arm ischemia accompanies vasodilatation of distant tissue in human cases. Therefore, we examined whether intermittent arm ischemia induces vasodilatation of another artery, an artery of the contralateral upper limb.
There are several potential mechanisms underlying RIPC in neural and hormonal pathways and systematic response [1, 12]. Loukogeorgakis et al. [13] reported that trimetaphan, an autonomic ganglion blocker, attenuated RIPC by intermittent arm ischemia in humans. The autonomic nerve system may be associated with RIPC by intermittent arm ischemia in humans. Thus, we hypothesized that RIPC by intermittent arm ischemia changes the autonomic balance and induces vasodilatation of the distant organ’s artery. The purpose of this study was to determine the validity of this hypothesis. We evaluated changes in autonomic nerve balance during intermittent arm ischemia using power spectral analysis of the RR interval of the electrocardiography (ECG) and whether vasodilatation of the brachial artery occurred using 2D gray-scale echography.
Methods
Subjects
This study was performed on 20 healthy volunteers (17 men and 3 women; mean age 34 ± 5 years, age range 24–42 years; height 169 ± 2 cm; weight 62 ± 3 kg; right brachial artery diameter 3.78 ± 0.1 mm). The diameter of the brachial artery was measured 3–5 cm above the antecubital space. All of the studies were approved by the Ethics Committee of Okayama University Graduate School of Medicine, Dentistry and Pharmaceutical Sciences, and informed consent was obtained from all subjects before the procedure. The investigation conformed to the principles outlined in the Declaration of Helsinki.
Induction of intermittent ischemia
Studies were performed in a temperature-controlled laboratory (24–26°C). Intermittent ischemia was induced by inflating a 13.5-cm-wide blood pressure cuff placed around the upper part of the left arm. The cuff was inflated to 200 mmHg for 5 min (ischemia) followed by 5-min deflation. The inflation/deflation cycle was performed three times, as described previously [13].
Assessment of artery response
Artery response was continuously assessed by measuring change in the right brachial artery diameter through three cycles of 5-min inflation (200 mmHg) and 5-min deflation of the blood pressure cuff. Using high-resolution ultrasound with a 10-MHz linear array transducer, the right brachial artery diameter was measured as previously described [14]. Longitudinal images of the artery were recorded at baseline and continuously from the first to third cycles of ischemia-reperfusion. The diameter of the brachial artery was determined semi-automatically using an instrument equipped with software for monitoring brachial artery diameter (Unex Co., Ltd., Nagoya, Japan). Briefly, continuous recording of a 2D gray scale image and A-mode waves of the brachial artery in the longitudinal plane was conducted with a novel stereotactic probe-holding device. A segment with clear anterior (media-adventitia) and posterior (intima-media) interfaces was manually determined. Then these border interfaces were identified automatically on the A-mode waves. The diastolic per-beat diameter of the brachial artery was synchronized with the electrocardiographic R-wave and tracked automatically. The changes in diameter were continuously recorded. Then vasodilatation was examined as percent change in the diameter over the baseline value at maximal vasodilatation after every reperfusion. Both the baseline values and each maximum vasodilatation value came from a single beat. Three-millimeter-wide longitudinal images of the artery in A-mode were obtained, and the images were divided into 20 parts. Mean diameter of the 20 parts was calculated as the brachial artery’s diameter.
Power spectral analysis
To evaluate indices of sympathetic activity and parasympathetic activity, we performed power spectral analysis. ECG was recorded using a two-channel recorder during the protocol (Fukuda Denshi: model Digital walk FM190, Fukuda Denshi Co., Ltd., Tokyo, Japan). The power spectra of the RR interval were calculated using the maximum entropy method with MemCalc software (Suwa Trust Co., Ltd., Tokyo, Japan) [15] at 15-s intervals for 30-s periods. We performed measurements at two time points, baseline and 3rd ischemia. The interpolation was performed by using the linear supplement method after cleaning noise. The interval of resampling is determined as the difference between the mean and minimum values of the RR interval. The high-frequency (HF 0.15–0.40 Hz) domain was used as a marker of parasympathetic activity, and the ratio of low-frequency (LF 0.04–0.15 Hz) domain to HF (LF/HF) was used as an indicator of sympathetic activity [16]. The averaged value during the 10-min period before RIPC was defined as baseline.
Study protocols
After 10-min bed rest in the supine position, the diameter of the right brachial artery was measured at the four times, before and after each of three cycles of 5-min inflation (200 mmHg) and 5-min deflation of the blood pressure cuff in the left upper arm. We obtained the percent change in diameter over the baseline value at maximal vasodilatation in each period (Fig. 1a). Simultaneously, ECG monitoring with a Fukuda Denshi FM-190 (Fukuda Denshi) was performed. In the control study without ischemia-reperfusion of the left upper arm, the diameter of the right brachial artery was measured at four times, before and after each of 3 cycles of 10-min bed rest in the supine position (Fig. 1b).
Fig. 1.
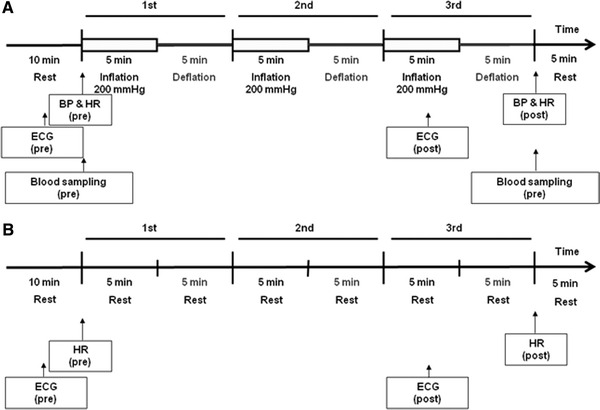
Study protocols with and without ischemia-reperfusion. a Study protocol with ischemia-reperfusion. b Study protocol without ischemia-reperfusion. a After 10-min bed rest in the supine position, the diameter of the right brachial artery was measured four times, before and after each of three cycles of 5-min inflation (200 mmHg) and 5-min deflation of a blood pressure cuff in the left arm. b. In the control study without ischemia-reperfusion of the left arm, the diameter of the right brachial artery was measured four times, before and after 3 cycles of 10-min bed rest in the supine position. BP Blood pressure, HR heart rate, ECG electorcardiogram
Laboratory measurements
Fasting blood samples were drawn before and after 3 cycles of ischemia-reperfusion from 11 of the 20 volunteers who gave informed consent (Fig. 1a). We took both blood samples by needle insertion. To evaluate the relationship between contralateral artery dilatation and nitric oxide (NO), we measured cyclic GMP, a second messenger of NO. To evaluate the relationship between contralateral artery dilatation and sympathetic activity using parameters other than heart rate variability, we measured catecholamine. The plasma levels of cyclic GMP and catecholamine (adrenaline, noradrenalin and dopamine) were measured using the RIA DCC method and HPLC method (SRL, Tokyo, Japan)
Statistical analysis
All data are expressed as mean ± SE unless otherwise stated. Brachial artery diameter was measured in millimeters, and dilatation was expressed as percentage increase from baseline diameter. Data were compared using Student’s paired t test or repeated measures analysis of variance (ANOVA), as appropriate. For assessment of the changes in the power spectral, hemodynamics and laboratory data, Student’s paired t test was applied. For assessment of the changes in artery diameter, analysis of variance (ANOVA) was applied. P value by ANOVA was Bonferroni-adjusted. In all cases, P < 0.05 was considered statistically significant.
Results
Effect of ischemia-reperfusion of the left arm on the right brachial artery
All subjects tolerated the procedures without any complications and any pains. The ischemia-reperfusion protocol had no effect on hemodynamics except for heart rate (Table 1). Ischemia-reperfusion of the left arm gradually enhanced dilatation of the right brachial artery, an artery in the contralateral limb, and this effect was dependent on the number of trials (Figs. 2, 3a). Mean vessel diameters of all subjects were increased significantly [vessel diameter: 3.78 ± 0.1 mm before procedure (control), 3.83 ± 0.1 mm after the 1st cycle of ischemia-reperfusion, 3.86 ± 0.1 mm after the 2nd cycle and 3.89 ± 0.1 mm after the 3rd cycle] (control vs. 1st cycle, P < 0.001; control vs. 2nd cycle, P < 0.001; control vs. 3rd cycle, P < 0.001). Intermittent arm ischemia of the left upper arm increased % changes of vessel diameter in individual subjects. The percent changes were 1.5 ± 0.3, 2.4 ± 0.3 and 3.2 ± 0.4% of the baseline diameter, respectively (control vs. after 1st cycle, P < 0.001; control vs. after 2nd cycle, P < 0.001; control vs. after 3rd cycle, P < 0.001) (Fig. 3a).
Table 1.
Changes in hemodynamics
| Pre | Post | |
|---|---|---|
| Systolic BP (mmHg) | 108 ± 8 | 106 ± 7 |
| Diastolic BP (mmHg) | 66 ± 6 | 66 ± 8 |
| HR (/min) | 64 ± 2 | 59 ± 2* |
Data are mean ± SE of the mean
BP Blood pressure, HR heart rate, Pre before 3 cycles of intermittent arm ischemia-reperfusion, Post after 3 cycles of intermittent arm ischemia-reperfusion
* Significantly different from Pre, P < 0.05
Fig. 2.
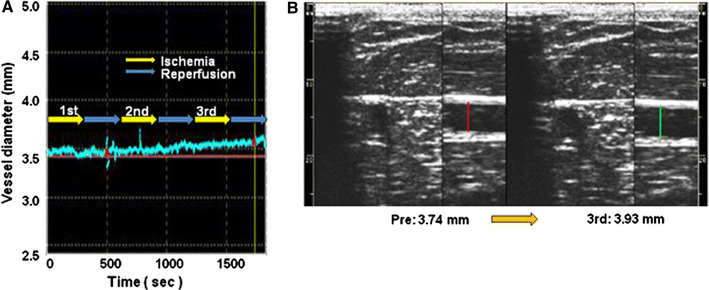
Ischemia-reperfusion of the left arm gradually enhanced right brachial artery dilatation time-dependently. a Representative diameter of a contralateral artery, right brachial artery. b Representative ultrasonographical images of the right brachial artery before (left) and after (right) 3 cycles of intermittent arm ischemia
Fig. 3.
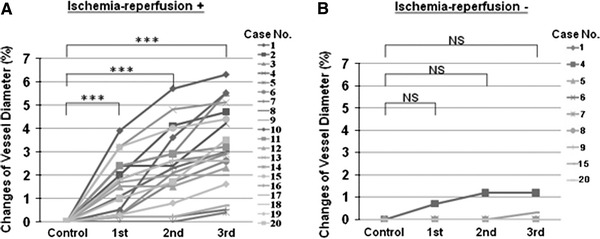
% changes of vessel diameter in individual subjects with or without ischemia-reperfusion. a Effect of ischemia-reperfusion of the left arm on the right brachial artery. ***P < 0.001. b Effect of bed rest without ischemia-reperfusion of the left arm on the right brachial artery
We examined whether the vessel diameter was changed during bed rest without intermittent arm ischemia for the control study in 9 subjects out of the same 20 volunteers with intermittent arm ischemia. Post mean heart rate was significantly lower than pre mean heart rate (pre mean heart rate 65 ± 3/min, post mean heart rate 60 ± 3/min; pre mean heart rate vs. post mean heart rate, P < 0.05). Mean vessel diameters of all subjects were not increased significantly. The vessel diameters were 3.81 ± 0.32, 3.81 ± 0.32, 3.81 ± 0.33 and 3.82 ± 0.33 mm, respectively (control vs. 1st cycle, P = 1.000; control vs. 2nd cycle, P = 1.000; control vs. 3rd cycle, P = 1.000). Bed rest without intermittent arm ischemia did not increase % changes of vessel diameter in individual subjects. The percent changes were 0.1 ± 0.07, 0.2 ± 0.14 and 0.2 ± 0.14%, respectively (control vs. after 1st cycle, P = 1.000; control vs. after 2nd cycle, P = 1.000; control vs. after 3rd cycle, P = 1.000) (Fig. 3b).
Effect of ischemia-reperfusion of the left arm on the autonomic nerve system
The high-frequency domain, a marker of parasympathetic activity, after the 3rd reperfusion was significantly higher than the baseline mean HF domain (425 ± 69 ms2/Hz at baseline vs. 617 ± 108 ms2/Hz after 3rd ischemia, P < 0.05) (Fig. 4a). On the other hand, bed rest without intermittent arm ischemia did not increase the HF domain (370 ± 79 ms2/Hz at baseline vs. 357 ± 59 ms2/Hz after 3rd cycle, P = 0.81) (Fig. 4b).
Fig. 4.
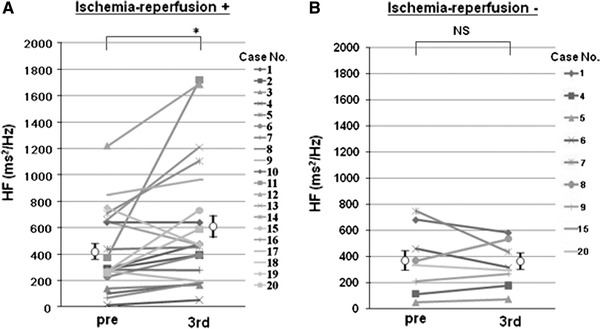
Changes in the high frequency (HF) domain with or without ischemia-reperfusion. a Effect of ischemia-reperfusion of the left arm on HF domain. *P < 0.05. b Effect of bed rest without ischemia-reperfusion of the left arm on HF domain
The ratio of LF to HF, an indicator of sympathetic activity, after the 3rd ischemia was lower than baseline, but not significantly (2.6 ± 0.5 at baseline vs. 2.0 ± 0.3 after the 3rd cycle, P = 0.07). Bed rest without intermittent arm ischemia did not change LF/HF (2.6 ± 0.5 baseline vs. 2.3 ± 0.3 after the 3rd cycle, P = 0.30).
Plasma noradrenalin levels after the 3rd cycle of ischemia-reperfusion were significantly lower than those before ischemia-reperfusion. Cyclic GMP, adrenaline and dopamine levels after the 3rd cycle of ischemia-reperfusion tended to be lower than those before ischemia-reperfusion (Table 2).
Table 2.
Peripheral blood chemical analysis
| Pre | Post | |
|---|---|---|
| Cyclic GMP (pmol/ml) | 3.6 ± 0.5 | 3.1 ± 0.4 |
| Adrenaline (pg/ml) | 31.3 ± 5.7 | 25.5 ± 4.8 |
| Noradrenaline (pg/ml) | 302.4 ± 31.3 | 230.0 ± 31.7* |
| Dopamine (pg/ml) | 8.2 ± 1.2 | 6.6 ± 0.8 |
Data are mean ± SE of the mean
GMP Guanosine monophosphate, Pre before 3 cycles of intermittent ischemia-reperfusion, Post after 3 cycles of intermittent ischemia-reperfusion
* Significantly different from Pre, P < 0.05
Discussion
This study showed for the first time in humans that intermittent ischemia in an upper limb was accompanied by vasodilatation in the contralateral limb and enhancement of parasympathetic nerve activity.
There are several possible mechanisms by which vasodilatation in the contralateral limb is induced by intermittent upper limb ischemia. First, several studies have indicated that RIPC is associated with hormonal factors such as adenosine and bradykinin, and other factors such as nitric oxide [12]. Accordingly, contralateral upper limb vasodilatation might be induced by these vasoactive factors. Our results showed that cyclic GMP was not significantly changed, indicating that nitric oxide does not participate in the vasodilatation. Second, in humans, the neural pathway is involved in the RIPC mechanism of the early (short) phase [13] and late phase [17–19], but the afferent-efferent pathway has not been elucidated. Recently, Gourine et al. reported that RIPC might be associated with the nerve reflex, which is the sensory afferent pathway from the peripheral organ (limbs) and parasympathetic vagal efferent outflow to the heart in rats [20]. Previous studies showed that there is evidence of parasympathetic vasodilator in the masseter muscle [21], lower lip [22] and brain vessels [23] in animals. Those findings indicated that vasodilatation is a result of reflex via increasing parasympathetic activity.
It is well known that increased sympathetic activity and reduced parasympathetic activity are induced by cardiovascular diseases. Previous studies showed that physical training could significantly improve exercise capacity and ameliorate the autonomic derangement in chronic heart failure by increasing parasympathetic activity [24–26]. Experimental studies indicated that increased parasympathetic activity may reduce mortality [27] and sudden cardiac death, including death from ventricular fibrillation [28, 29]. Furthermore, Schwartz et al. [30] reported that vagal stimulation improved activities of daily living and several echocardiographic parameters of the left ventricle in humans. Accordingly, parasympathetic stimulation may play a critical role in the care of heart disease. In this study, we demonstrated that enhancement of parasympathetic nerve activity, assessed by the HF domain of heart rate variability, was induced by intermittent arm ischemia.
Limitations of this study
The study has several limitations. First, we did not directly elucidate heart protection by RIPC by intermittent arm ischemia. Second, we did not assess the relevance of the brachial artery’s dilatation and the enhancement of parasympathetic nerve activity by intermittent arm ischemia. Third, we observed vasodilatation of the contralateral upper limb, but the duration of vasodilatation is not clear. Furthermore, we could not directly measure brachial artery flow. Therefore, further studies are needed to clarify these points.
Conclusions
Intermittent arm ischemia was accompanied by vasodilatation of another artery and enhancement of parasympathetic activity. It may be associated with the mechanism of RIPC.
Acknowledgments
This work was supported by the Department of Cardiovascular Medicine, Okayama University Graduate School of Medicine, Dentistry, and Pharmaceutical Sciences. We thank K.A., A.O., H.O., K.O., Y.K., T.S., H.S., M.S., Y.S., D.M., A.M., M.F. and M.O. for their excellent technical assistance.
Conflict of interest
We have no financial relationship with the organization that sponsored the research. We declare that they have no conflict of interest.
Footnotes
K. Enko and K. Nakamura contributed equally to this work.
Contributor Information
Kenki Enko, Email: en9422@yahoo.co.jp.
Kazufumi Nakamura, Email: ichibun@cc.okayama-u.ac.jp.
References
- 1.Kharbanda RK, Nielsen TT, Redington AN. Translation of remote ischaemic preconditioning into clinical practice. Lancet. 2009;374:1557–1565. doi: 10.1016/S0140-6736(09)61421-5. [DOI] [PubMed] [Google Scholar]
- 2.Przyklenk K, Bauer B, Ovize M, Kloner RA, Whittaker P. Regional ischemic ‘preconditioning’ protects remote virgin myocardium from subsequent sustained coronary occlusion. Circulation. 1993;87:893–899. doi: 10.1161/01.cir.87.3.893. [DOI] [PubMed] [Google Scholar]
- 3.Takaoka A, Nakae I, Mitsunami K, Yabe T, Morikawa S, Inubushi T, Kinoshita M. Renal ischemia/reperfusion remotely improves myocardial energy metabolism during myocardial ischemia via adenosine receptors in rabbits: effects of “remote preconditioning”. J Am Coll Cardiol. 1999;33:556–564. doi: 10.1016/S0735-1097(98)00559-2. [DOI] [PubMed] [Google Scholar]
- 4.Lang SC, Elsasser A, Scheler C, Vetter S, Tiefenbacher CP, Kubler W, Katus HA, Vogt AM. Myocardial preconditioning and remote renal preconditioning—identifying a protective factor using proteomic methods? Basic Res Cardiol. 2006;101:149–158. doi: 10.1007/s00395-005-0565-0. [DOI] [PubMed] [Google Scholar]
- 5.Gho BC, Schoemaker RG, van den Doel MA, Duncker DJ, Verdouw PD. Myocardial protection by brief ischemia in noncardiac tissue. Circulation. 1996;94:2193–2200. doi: 10.1161/01.cir.94.9.2193. [DOI] [PubMed] [Google Scholar]
- 6.Botker HE, Kharbanda R, Schmidt MR, Bottcher M, Kaltoft AK, Terkelsen CJ, Munk K, Andersen NH, Hansen TM, Trautner S, Lassen JF, Christiansen EH, Krusell LR, Kristensen SD, Thuesen L, Nielsen SS, Rehling M, Sorensen HT, Redington AN, Nielsen TT. Remote ischaemic conditioning before hospital admission, as a complement to angioplasty, and effect on myocardial salvage in patients with acute myocardial infarction: a randomised trial. Lancet. 2010;375:727–734. doi: 10.1016/S0140-6736(09)62001-8. [DOI] [PubMed] [Google Scholar]
- 7.Hoole SP, Heck PM, Sharples L, Khan SN, Duehmke R, Densem CG, Clarke SC, Shapiro LM, Schofield PM, O’Sullivan M, Dutka DP. Cardiac Remote Ischemic Preconditioning in Coronary Stenting (CRISP Stent) Study: a prospective, randomized control trial. Circulation. 2009;119:820–827. doi: 10.1161/CIRCULATIONAHA.108.809723. [DOI] [PubMed] [Google Scholar]
- 8.Hausenloy DJ, Mwamure PK, Venugopal V, Harris J, Barnard M, Grundy E, Ashley E, Vichare S, Di Salvo C, Kolvekar S, Hayward M, Keogh B, MacAllister RJ, Yellon DM. Effect of remote ischaemic preconditioning on myocardial injury in patients undergoing coronary artery bypass graft surgery: a randomised controlled trial. Lancet. 2007;370:575–579. doi: 10.1016/S0140-6736(07)61296-3. [DOI] [PubMed] [Google Scholar]
- 9.Rahman IA, Mascaro JG, Steeds RP, Frenneaux MP, Nightingale P, Gosling P, Townsend P, Townend JN, Green D, Bonser RS. Remote ischemic preconditioning in human coronary artery bypass surgery: from promise to disappointment? Circulation. 2010;122:S53–S59. doi: 10.1161/CIRCULATIONAHA.109.926667. [DOI] [PubMed] [Google Scholar]
- 10.Shimizu M, Konstantinov IE, Kharbanda RK, Cheung MH, Redington AN. Effects of intermittent lower limb ischaemia on coronary blood flow and coronary resistance in pigs. Acta Physiol (Oxf) 2007;190:103–109. doi: 10.1111/j.1748-1716.2007.01667.x. [DOI] [PubMed] [Google Scholar]
- 11.Zhou K, Yang B, Zhou XM, Tan CM, Zhao Y, Huang C, Liao XB, Xiao HB. Effects of remote ischemic preconditioning on the flow pattern of the left anterior descending coronary artery in normal subjects. Int J Cardiol. 2007;122:250–251. doi: 10.1016/j.ijcard.2006.11.079. [DOI] [PubMed] [Google Scholar]
- 12.Hausenloy DJ, Yellon DM. Remote ischaemic preconditioning: underlying mechanisms and clinical application. Cardiovasc Res. 2008;79:377–386. doi: 10.1093/cvr/cvn114. [DOI] [PubMed] [Google Scholar]
- 13.Loukogeorgakis SP, Panagiotidou AT, Broadhead MW, Donald A, Deanfield JE, MacAllister RJ. Remote ischemic preconditioning provides early and late protection against endothelial ischemia-reperfusion injury in humans: role of the autonomic nervous system. J Am Coll Cardiol. 2005;46:450–456. doi: 10.1016/j.jacc.2005.04.044. [DOI] [PubMed] [Google Scholar]
- 14.Tomiyama H, Matsumoto C, Yamada J, Teramoto T, Abe K, Ohta H, Kiso Y, Kawauchi T, Yamashina A. The relationships of cardiovascular disease risk factors to flow-mediated dilatation in Japanese subjects free of cardiovascular disease. Hypertens Res. 2008;31:2019–2025. doi: 10.1291/hypres.31.2019. [DOI] [PubMed] [Google Scholar]
- 15.Ito K, Hirooka Y, Kimura Y, Sagara Y, Sunagawa K. Ovariectomy augments hypertension through rho-kinase activation in the brain stem in female spontaneously hypertensive rats. Hypertension. 2006;48:651–657. doi: 10.1161/01.HYP.0000238125.21656.9e. [DOI] [PubMed] [Google Scholar]
- 16.Morita H, Yamanari H, Ohe T. Evaluation of autonomic nervous activity in patients with congenital long QT syndrome by an analysis of RR variability. Jpn Circ J. 1996;60:742–748. doi: 10.1253/jcj.60.742. [DOI] [PubMed] [Google Scholar]
- 17.Wolfrum S, Schneider K, Heidbreder M, Nienstedt J, Dominiak P, Dendorfer A. Remote preconditioning protects the heart by activating myocardial PKCepsilon-isoform. Cardiovasc Res. 2002;55:583–589. doi: 10.1016/S0008-6363(02)00408-X. [DOI] [PubMed] [Google Scholar]
- 18.Schoemaker RG, van Heijningen CL. Bradykinin mediates cardiac preconditioning at a distance. Am J Physiol Heart Circ Physiol. 2000;278:H1571–H1576. doi: 10.1152/ajpheart.2000.278.5.H1571. [DOI] [PubMed] [Google Scholar]
- 19.Liem DA, Verdouw PD, Ploeg H, Kazim S, Duncker DJ. Sites of action of adenosine in interorgan preconditioning of the heart. Am J Physiol Heart Circ Physiol. 2002;283:H29–H37. doi: 10.1152/ajpheart.01031.2001. [DOI] [PubMed] [Google Scholar]
- 20.Gourine A, Mastitskaya S, Gilbey MP, Ackland GL, Gourine AV. Remote preconditioning reflex—a neural pathway of cardioprotection during myocardial ischaemia and reperfusion induced by remote ischaemic preconditioning. Eur Heart J. 2010;31(Suppl 1):319. [Google Scholar]
- 21.Ishii H, Niioka T, Sudo E, Izumi H. Evidence for parasympathetic vasodilator fibres in the rat masseter muscle. J Physiol. 2005;569:617–629. doi: 10.1113/jphysiol.2005.087643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Izumi H. Reflex parasympathetic vasodilatation in facial skin. Gen Pharmacol. 1995;26:237–244. doi: 10.1016/0306-3623(94)00155-G. [DOI] [PubMed] [Google Scholar]
- 23.Boysen NC, Dragon DN, Talman WT. Parasympathetic tonic dilatory influences on cerebral vessels. Auton Neurosci. 2009;147:101–104. doi: 10.1016/j.autneu.2009.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Adamopoulos S, Ponikowski P, Cerquetani E, Piepoli M, Rosano G, Sleight P, Coats AJ. Circadian pattern of heart rate variability in chronic heart failure patients. Effects of physical training. Eur Heart J. 1995;16:1380–1386. doi: 10.1093/oxfordjournals.eurheartj.a060746. [DOI] [PubMed] [Google Scholar]
- 25.Coats AJ, Adamopoulos S, Radaelli A, McCance A, Meyer TE, Bernardi L, Solda PL, Davey P, Ormerod O, Forfar C, et al. Controlled trial of physical training in chronic heart failure. Exercise performance, hemodynamics, ventilation, and autonomic function. Circulation. 1992;85:2119–2131. doi: 10.1161/01.cir.85.6.2119. [DOI] [PubMed] [Google Scholar]
- 26.Kiilavuori K, Toivonen L, Naveri H, Leinonen H. Reversal of autonomic derangements by physical training in chronic heart failure assessed by heart rate variability. Eur Heart J. 1995;16:490–495. doi: 10.1093/oxfordjournals.eurheartj.a060941. [DOI] [PubMed] [Google Scholar]
- 27.Li M, Zheng C, Sato T, Kawada T, Sugimachi M, Sunagawa K. Vagal nerve stimulation markedly improves long-term survival after chronic heart failure in rats. Circulation. 2004;109:120–124. doi: 10.1161/01.CIR.0000105721.71640.DA. [DOI] [PubMed] [Google Scholar]
- 28.Vanoli E, De Ferrari GM, Stramba-Badiale M, Hull SS, Jr, Foreman RD, Schwartz PJ. Vagal stimulation and prevention of sudden death in conscious dogs with a healed myocardial infarction. Circ Res. 1991;68:1471–1481. doi: 10.1161/01.res.68.5.1471. [DOI] [PubMed] [Google Scholar]
- 29.Billman GE, Schwartz PJ, Stone HL. The effects of daily exercise on susceptibility to sudden cardiac death. Circulation. 1984;69:1182–1189. doi: 10.1161/01.CIR.69.6.1182. [DOI] [PubMed] [Google Scholar]
- 30.Schwartz PJ, De Ferrari GM, Sanzo A, Landolina M, Rordorf R, Raineri C, Campana C, Revera M, Ajmone-Marsan N, Tavazzi L, Odero A. Long term vagal stimulation in patients with advanced heart failure: first experience in man. Eur J Heart Fail. 2008;10:884–891. doi: 10.1016/j.ejheart.2008.07.016. [DOI] [PubMed] [Google Scholar]


