Abstract
Introduction
Complement 3 glomerulopathy (C3G) is a rare inflammatory kidney disease mediated by dysregulation of the alternative complement pathway. No targeted therapy exists for this aggressive glomerulonephritis. Efficacy, safety, tolerability, pharmacokinetics (PK), and pharmacodynamics (PD) (measured by complement biomarkers) of iptacopan were assessed in patients with C3G.
Methods
In this phase 2, multicenter, open-label, single-arm, nonrandomized study, adults with biopsy-proven, native kidney C3G (native cohort) and kidney transplant recipients with C3G recurrence (recurrent kidney transplant [KT] cohort) received iptacopan twice daily (bid) for 84 days (days 1–21: 10–100 mg; days 22–84: 200 mg). The primary end point was the urine protein-to-creatinine ratio (UPCR; native cohort) and the change in the C3 deposit score of kidney biopsy (recurrent KT cohort). The complement pathway measures included Wieslab assay, soluble C5b9, and serum C3 levels.
Results
A total of 27 patients (16 native cohort and 11 recurrent KT cohort) were enrolled and all completed the study. In the native cohort, UPCR levels decreased by 45% from baseline to week 12 (P = 0.0003). In the recurrent KT cohort, the median C3 deposit score decreased by 2.50 (scale: 0–12) on day 84 versus baseline (P = 0.03). Serum C3 levels were normalized in most patients; complement hyperactivity observed pretreatment was reduced. Severe adverse events (AEs) included post-biopsy hematuria and hyperkalemia. No deaths occurred during the study.
Conclusion
Iptacopan resulted in statistically significant and clinically important reductions in UPCR and normalization of serum C3 levels in the native cohort and reduced C3 deposit scores in the recurrent KT cohort with favorable safety and tolerability. (ClinicalTrials.gov identifier: NCT03832114).
Keywords: complement 3 glomerulopathy, inflammatory kidney disease, iptacopan, kidney transplant, urine protein-to-creatinine ratio
Graphical abstract
C3G, an ultra-rare complement-mediated kidney disease is associated with dysregulation of the complement alternative pathway (AP) in plasma and the glomerular microenvironment, thereby resulting in accumulation of C3 in the glomerulus.1, 2, 3, 4, 5 C3G is caused by acquired and/or genetic abnormalities affecting the complement pathway, such as nephritic factors, or genetic variants in key AP complement genes.1,6, 7, 8, 9 C3G has 2 major subgroups, classified by appearance on electron microscopy, dense deposit disease and C3 glomerulonephritis, that have overlapping clinical and pathological features.6,10 Patients with C3G present with typical features of glomerulonephritis such as proteinuria, hematuria, and hypertension.6,8 Clinical disease is frequently accompanied by low serum C3 levels and/or elevated sC5b9 levels, reflecting activation of the complement cascade.4,6,7
Various registry data suggest an incidence of 1 to 2 cases of C3G per 1 million.11,12 About 30% to 40% of C3G cases present during childhood11, 12, 13, 14; 30% to 50% of adult patients and approximately 70% of pediatric patients with C3G progress to kidney failure within 10 years of diagnosis6,12,15,16; Disease recurrence after kidney transplantation leads to allograft loss in approximately 50% of patients within 10 years of transplant.6,17,18
Currently, there is no approved targeted therapy for C3G and the suggested management options (as recognized by Kidney Disease: Improving Global Outcomes, but not based on controlled studies)19 include the following: (i) supportive care including antiproteinuric therapy with an angiotensin-converting enzyme inhibitor or angiotensin II receptor blocker; (ii) immunosuppression with corticosteroids, mycophenolate mofetil/sodium (MMF/MPS), cyclophosphamide, rituximab, or plasmapheresis; or (iii) terminal complement pathway inhibition through the anti-C5 monoclonal antibody eculizumab.6,8,19, 20, 21, 22, 23 The efficacy of these measures is limited.19,23 There is a high unmet medical need for new therapies to treat C3G. Inhibitors that target the proximal aspect of the complement cascade represent a promising approach.
Iptacopan (LNP023) is a proximal complement inhibitor that specifically binds factor B and inhibits the AP. Inhibition of complement factor B prevents activity of AP-related C3 convertase and the subsequent formation of C5 convertase. Although iptacopan inhibits amplification of the classical and lectin pathways of complement activation, it leaves direct signaling intact.24 By directly targeting the AP, the key pathway involved in the pathogenesis of C3G, iptacopan may prevent both C3 deposition and renal inflammation and is expected to improve both kidney histology and function and prevent further disease progression.24,25 The present study aimed to assess the clinical efficacy, safety, tolerability, PK, and PD of iptacopan in patients with native and recurrent C3G; and to evaluate the effect of different doses of iptacopan on complement biomarkers, in order to inform dose selection for a phase 3 study.
Methods
Study Design and Conduct
In this phase 2, multicenter, nonconfirmatory, open-label, single-arm, nonrandomized study (clinicaltrial.gov identifier: NCT03832114), patients were recruited into 2 cohorts and received an 84-day open-label course of iptacopan (Supplementary File page 4 and Supplementary Figure S1). The study was conducted in compliance with the ethical principles of the Declaration of Helsinki and the International Conference on Harmonization Good Clinical Practice Guidelines.26 Written informed consent was obtained from each patient. The study protocol and all amendments were reviewed and approved by the respective Independent Ethics Committee or Institutional Review Board27 for each center as per local regulations.
Study Population
Adult patients (18 years or older) with biopsy-proven native C3G (native cohort) or recurrent C3G post kidney transplant (recurrent KT cohort), with an estimated glomerular filtration rate (eGFR) ≥30 ml/min per 1.73 m2 were recruited. Patients enrolled in the native cohort were on maximally tolerated angiotensin-converting enzyme inhibitor or angiotensin II receptor blocker therapy and were allowed to receive stable doses of MMF/MPS and/or prednisolone (or equivalent) up to 7.5 mg per day. Patients with reduced serum C3 levels at screening (<0.90 × lower limit of laboratory normal range) and with UPCR ≥100 g/mol in first morning void urine, OR ≥1 g/24h total urinary protein excretion from a 24-hour urine collection during the run-in period were enrolled in the native cohort. Patients with recurrent KT C3G could be enrolled after 90 days following transplantation and were required to be on stable doses of immunosuppression and with no evidence of allograft rejection. Further details on patient eligibility criteria are summarized in Supplementary File page 3 with a list of prohibited medications in Supplementary Table S1.
Study End Points
The primary end point was comparison of baseline and day 84 UPCR derived from 24-hour urine collection in the native cohort and change from baseline to day 84 in C3 deposit score based on immunofluorescence microscopy of kidney biopsy tissue in the recurrent KT cohort (details of scoring are described in the Supplementary File page 4). Secondary end points were change and evolution of urinary albumin-to-creatinine ratio (UACR, 24-hour urine), UPCR (24-hour urine; only for recurrent KT cohort), eGFR (using Chronic Kidney Disease Epidemiology Collaboration formula), serum creatinine, iptacopan plasma PK assessments, and safety and tolerability outcomes. Biomarker measures included cleaved factor B [Bb], C3, and sC5b9, Wieslab assay, urine sC5b9, and a set of urinary kidney damage/inflammation biomarkers. Further details on end points and assessments are provided in Supplementary File page 4.
Statistical Analysis
Sample size calculation was performed assuming that 15 patients in the native cohort and 12 in the recurrent KT cohort would provide more than 90% power for demonstration of true treatment reduction for the primary end point (log transformed UPCR of 40% [SD 0.6] in the native cohort and reduction in C3 deposit score of 2 [SD 2, using Wilcoxon matched-pairs signed-ranks test] in the recurrent KT cohort) and statistical significance at a 1-sided 10% significance level accounting for a drop-out rate of 20% in the native cohort and 17% in the recurrent KT cohort. Demographics, patient baseline characteristics, biomarker data, and safety data were summarized using descriptive statistics. The PD analysis set included all subjects with available PD data and no protocol deviations with relevant impact on PD data. The changes from baseline in UPCR (native cohort), as well as secondary end points related to proteinuria and kidney function, were analyzed using the mixed model repeated measures analysis of variance model. Change in C3 deposit score by day 84 from baseline in the recurrent KT cohort was analyzed using the Wilcoxon signed rank test to compare the median difference (80% confidence interval). The Hodges-Lehmann estimate is provided. Further details of statistical methods used include handling of the missing values/censoring/discontinuations are described in the Supplementary File page 6.
Results
Patient Disposition
A total of 27 patients (16 native cohort and 11 recurrent KT cohort) were enrolled and treated and all patients completed the study (patient disposition and analysis sets are summarized in Supplementary Table S2). Except for 1 patient from the recurrent KT cohort, all patients who completed the study rolled over into a long-term extension study (further details on trial design are provided in Supplementary File page 4 and Supplementary Figure S1).
Demographics and Baseline
Patient demographics and baseline disease characteristics are presented in Table 1. The majority were male in both cohorts with the average disease duration of >7.5 years in both cohorts.
Table 1.
Demographics and baseline characteristics (Safety analysis set)
| Characteristics, mean (SD) | Native cohort N = 16 |
Recurrent KT cohort N = 11 |
|---|---|---|
| Age (yrs) | 26.1 (10.57) | 34.5 (18.32) |
| Male, n (%) | 10 (63) | 8 (73) |
| Race, n (%) | ||
| Caucasian | 16 (100) | 9 (82) |
| Othera | – | 2 (18) |
| Body mass index (kg/m2) | 21.4 (2.72) | 22.9 (4.02) |
| Disease duration (d), median (range)f | 2183 (267–8988) | 2596 (38–5796) |
| DDD, n (%) | 2 (13) | 3 (27) |
| C3GN, n (%) | 14 (87) | 8 (73) |
| 24-hour UPCR (g/mol), Geo-mean (CV% Geo-Mean) | 401.9 (53.64) | 36.2 (310.78) |
| 24-hour UACR (g/mol), Geo-mean (CV% Geo-Mean) | 338.4 (50.50) | 9.8 (1208.7) |
| 24-hour Urine protein (mg/d), Geo-mean (CV% Geo-Mean) | 4945.2 (54.27) | 283.4 (378.15) |
| 24-hour Urine albumin (mg/d), Geo-mean (CV% Geo-Mean) | 4164.7 (52.39) | 88.1 (1377.81) |
| Proportion of patients with nephrotic range proteinuriai, n (%) | 11 (69) | 2 (18) |
| Serum creatinine (μmol/l) | 134.0 (60.64) | 152.2 (40.52) |
| eGFR (ml/min per 1.73 m2) | 70.1 (35.10) | 52.2 (17.29) |
| C3 deposit scoreb; median (range) (n) | – | 3.0 (0.0–12.0) [10] |
| Plasma sC5b9 (ng/ml)(reference range: 44.8–231)c | 1306 (800.5) | 318.6 (300.1) |
| Urine sC5b9 (pg/ml)(reference range: 43.2–162)e | 584,000 (1,030,000) | 8122 (14200) |
| Serum Wieslab (%) | 22.2 (18.09) | 41.3 (24.32) |
| Serum C3 (g/l)(normal range: 0.9–1.8) | 0.3 (0.23) | 0.6 (0.24) |
| Vaccination prior to iptacopan treatment, n (%) | ||
| Neisseria meningitidis | 16 (100) | 11 (100) |
| Streptococcus pneumoniae | 16 (100) | 11 (100) |
| Haemophilus influenzae | 15 (94)g | 9 (82)g |
| Previous therapies, n (%) | 6 (37.5) | 10 (90.9) |
| ACE inhibitors | 12 (75.0) | 4 (36.4) |
| ARB | 4 (25.0) | 2 (18.2) |
| MMF/MPS | 6 (37.5) | 9 (82) |
| Alemtuzumabd | 0 | 1 (9.1) |
| Eculizumabd | 0 | 1 (9.1) |
| Calcineurin inhibitorsh | – | 11 (100.0) |
| Concomitant therapy, n (%) | ||
| ACE inhibitors | 12 (75) | 4 (36.4) |
| ARB | 4 (25.0) | 2 (18.2) |
| MMF/MPS | 5 (31.3) | 9 (82.0) |
| Glucocorticoids | 3 (18.8) | 9 (82) |
| Calcineurin inhibitors | 0 | 11 (100.0) |
| Azathioprine | 0 | 2 (18.2) |
| Cyclophosphamide | 0 | 1 (9.1) |
ACE, angiotensin-converting enzyme; ARB; angiotensin II receptor blocker; BMI, body mass index; C3GN, complement 3 glomerulonephritis; DDD, dense deposit disease; eGFR, estimated glomerular filtration rate; KT, kidney transplant; MMF/MPS, mycophenolate mofetil/sodium; N, total number of patients; n, number of patients; UACR, urinary albumin-to-creatinine ratio; UPCR, urinary protein-to-creatinine ratio.
Data are presented as mean (SD) unless stated otherwise.
For all 24-hour urine collection parameters, baseline is defined to be the collection on day −1 to day 1. For serum creatinine and eGFR, baseline is defined to be the last available assessment prior to the first dose of study drug.
Other includes 1 American Indian or Alaska Native and Black or African American.
Baseline kidney biopsy was collected during the run-in period if the most recent biopsy was older than 3 months.
Based on data obtained from first-in human trial of iptacopan (n = 176).
There was a 90-day washout period before starting study treatment.
Based on internal laboratory data (n = 6).
Duration of disease was calculated as the days between the onset of the disease until study day 1.
Three patients did not receive a Hemophilus influenzae vaccination prior to LNP023 treatment; however, a prophylactic antibiotic treatment was in place.
Includes tacrolimus, ciclosporin.
Nephrotic proportion proteinuria is defined as UPCR ≥300 g/mol at baseline.
Efficacy
Primary End Point
In the native cohort, iptacopan 200 mg bid resulted in a 45% reduction in proteinuria (24-hour UPCR) from baseline (geometric mean [GM] = 401.9 g/mol) to day 84 (GM = 220.1 g/mol), with an adjusted GM ratio of 0.55 (80% confidence interval: 0.46, 0.65; 2-sided P = 0.0003) (Figure 1a). In the recurrent KT cohort, the median C3 deposit score in kidney biopsies decreased significantly from baseline (3.0) to day 84 (0.5) with a median difference (80% confidence interval) of −2.5 (−3.75, −0.75; P = 0.03). In Figure 1b, we show individual C3 deposit scores as well as the median.
Figure 1.
Kidney function and C3 deposit score after iptacopan treatment in patients with native and recurrent KT C3G¶. (a) UPCR (adjusted GM [80% CI] of log ratio to baseline) in patients with native C3G; (b) C3 total deposit score (individual plots; n = 9) for patients with recurrent KT C3G. †Baseline is defined to be the 24-hour urine collection on day −1 to day 1. ‡Patients with only baseline values; day 84 biopsy was either not taken or was damaged. §For patient represented by the blue line, the C3G recurrence was confirmed by investigator based on a previous biopsy as per inclusion criteria. ¶A patient with recreational drug overdose was excluded from analysis. C3G, complement 3 glomerulopathy; CI, confidence interval; GM, geometric mean; KT, kidney transplant; UPCR, urine protein-to-creatinine ratio.
Secondary End Points
Total albuminuria (24-hour UACR) decreased by 45% from baseline to day 84 with iptacopan treatment in the native cohort (Supplementary Figure S2A). Proteinuria (UPCR and UACR) levels in the recurrent KT cohort were predominantly normal at baseline (UPCR [GM] = 23.9 g/mol; n = 7 and UACR [GM] = 5.7 g/mol; n = 8) with no significant reduction observed on day 84 (UPCR [GM] = 17.7 g/mol; n = 4; 2-sided P = 0.4766 and UACR [GM] = 4.8 g/mol; n = 4; 2-sided P = 0.3707, respectively; Supplementary Figure S2A and B). Kidney function assessed by eGFR values remained stable during the study in both cohorts (baseline arithmetic mean = 70.1 and 52.6 ml/min per 1.73 m2 respectively; day 84 [arithmetic mean] = 72.7 and 50.6 ml/min per 1.73 m2 respectively; Supplementary Figure S3A). We compared the eGFR measured during the study with eGFR data that was collected from the study participants over the 2 years prior to their entry into the study. Based on the eGFR slope analysis, the mean pretreatment eGFR slope in the native cohort was –16.13 ml/min per 1.73 m2 per year (nominal P = 0.0008) and in the recurrent KT cohort was –3.77 ml/min per 1.73 m2 per year (nominal P = 0.3342). Following treatment with iptacopan for 12 weeks, the eGFR improved by 4.98 ml/min per 1.73 m2 per 3 months in the native cohort (nominal P = 0.0846; Supplementary Figure S3B) and by 1.80 ml/min per 1.73 m2 per 3 months in the recurrent KT cohort (nominal P = 0.4858; Supplementary Figure S3c). The slope analysis showed that eGFR remained stable during the iptacopan treatment.
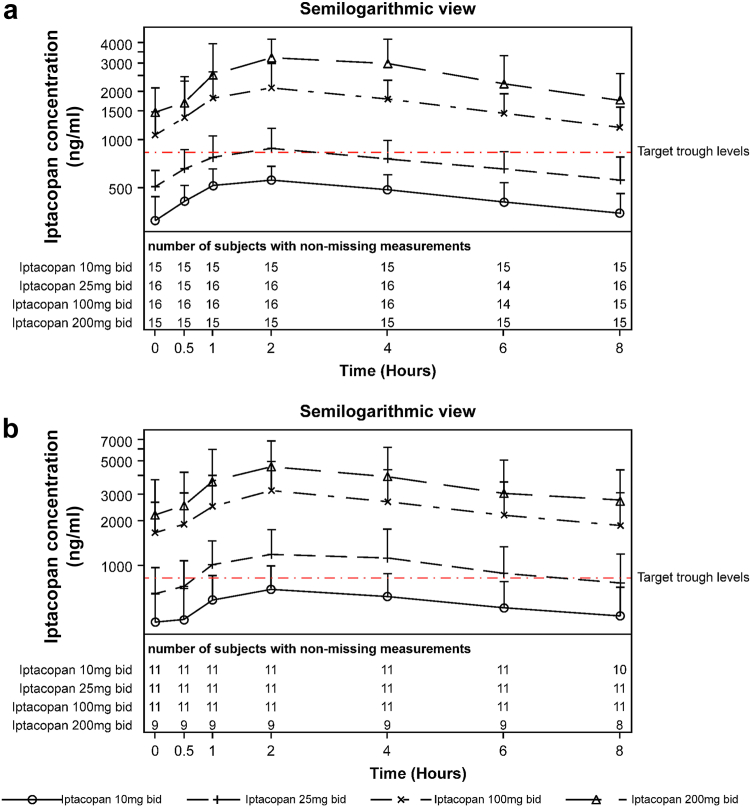
PK Results
Iptacopan was rapidly absorbed after oral administration in both the cohorts with a median Tmax of 2 hours postdose for all doses. In both cohorts, only 100 mg and 200 mg bid doses reached the target trough level of ≥900 ng/ml at steady state that would be expected to provide full target inhibition as per PK/PD modeling predictions (Figure 2a and b, Supplementary Table S3). No relationship was observed between 24-hour UPCR and PK parameters in the native cohort (Supplementary Figure S4A–C).
Figure 2.
Pharmacokinetic profile of iptacopan. Arithmetic mean + SD PK plasma concentration over time (hour) for iptacopan doses in native cohort (a) and in recurrent KT cohort (b). bid, twice daily; KT, Kidney transplant; PK, pharmacokinetic.
PD Analysis: Biomarkers
Sustained normalization of serum C3 levels was seen in most patients from both cohorts (Figure 3a). The serum Wieslab activity indicated dose-dependent inhibition of AP activity with full inhibition (i.e., no quantifiable AP activity) reached with 200 mg bid dosing (Figure 3b). Iptacopan treatment reduced markedly elevated sC5b9 levels in plasma (arithmetic mean, baseline = 1306 ng/ml; day 84 = 398.8 ng/ml) and urine (sC5b9: creatinine ratio; arithmetic mean, baseline = 61.7 pg/nmol; day 84 = 6.5 pg/nmol) in patients with native C3G in a dose-dependent manner (Figure 3c and d). In patients with recurrent KT C3G, plasma sC5b9 levels were comparable to healthy volunteers at baseline and remained as such. sC5b9 levels in urine were low (sC5b9: creatinine ratio) in patients with recurrent KT C3G. Reductions in factor B breakdown (plasma Bb levels) were observed in both cohorts (Figure 3e).
Figure 3.
Effect of iptacopan on biomarkers. Arithmetic mean ± SE, serum C3 (a), serum Wieslab activity (b), plasma sC5b9 (c), creatinine normalized urine sC5b9 (d), plasma Bb (e). Values below LLOQ are imputed as LLOQ/2 and values above ULOQ are imputed as ULOQ for the analysis. Baseline visit is defined to be the baseline 1 visit assessment (day −30). ∗Baseline is defined as day 1 predose assessment. LLOQ, lower limit of quantification; SE, standard error; ULOQ, upper limit of quantification.
The markers of renal tubular dysfunction, kidney injury and inflammation in urine, lipocalin-2, immunoglobulin G, and interleukin 18 were elevated in native cohort patients compared with patients in the recurrent KT cohort at the start of the study; a trend for decrease in these parameters was observed in the native cohort during the study (Supplementary Figure S5). Furthermore, urine cystatin C, a marker of renal tubular dysfunction, was elevated in the native cohort compared to the recurrent KT cohort patients and showed marked reduction on day 84; whereas most patients in the recurrent KT cohort had lower levels of urine cystatin C as compared to the native cohort that did not show relevant change on day 84 (Supplementary Figure S5).
Safety
Iptacopan treatment was well-tolerated, with no unexpected or new safety findings. AEs were mostly mild, except for 6 moderate AEs observed in 3 patients with native cohort and 12 moderate AEs in 6 patients with recurrent KT cohort (Table 2). There were no AEs leading to study discontinuation. AEs by preferred term with an incidence >2 are listed in Table 2. Infections were observed in 5 patients in each cohort and there were no episodes of encapsulated bacterial infection. The native cohort had a severe AE in 1 patient (postbiopsy hematuria in the run-in phase); and the recurrent KT cohort had 2 patients with severe AEs, including hyperkalemia, and recreational drug overdose intoxication during the treatment period, and 1 subject had 2 postprocedural biopsy–related severe AEs during the run-in phase (hematuria and wound infection at the biopsy site). One dose interruption due to a SARS-CoV-2–related AE was also seen in the recurrent KT cohort. No deaths occurred during the study.
Table 2.
Safety results for native and recurrent KT cohort
| Adverse event | Run-in phase | Dose-escalation phasea | Iptacopan 200 mg bid |
Overall |
|---|---|---|---|---|
| Native cohortN = 16, n (%) | ||||
| Patients with ≥1 AE | 7 (43.8) | 6 (37.5) | 8 (50.0) | 14 (87.5) |
| AEs of mild intensity | 6 (37.5) | 6 (37.5) | 8 (50.0) | 14 (87.5) |
| AEs of moderate intensity | 2 (12.5) | 1 (6.3) | 1 (6.3) | 3 (18.8) |
| AEs of severe intensity | 1 (6.3) | 0 | 0 | 1 (6.3) |
| SAEs | 1 (6.3) | 0 | 0 | 1 (6.3) |
| Study drug–related AEs | 0 | 0 | 2 (12.5)b | 2 (12.5) |
| PTc | ||||
| Abdominal pain upper | 0 | 1 (6.3) | 1 (6.3) | 2 (12.5) |
| Anemia | 1 (6.3) | 1 (6.3) | 0 | 2 (12.5) |
| Blood creatine phosphokinase increased | 1 (6.3) | 1 (6.3) | 1 (6.3) | 2 (12.5) |
| Nausea | 2 (12.5) | 0 | 0 | 2 (12.5) |
| Upper respiratory tract infection | 2 (12.5) | 0 | 0 | 2 (12.5) |
| Recurrent KT CohortN = 11, n (%) | ||||
| Patients with at least one AE | 2 (18.2) | 5 (45.5) | 6 (54.5) | 8 (72.7) |
| AEs of mild intensity | 1 (9.1) | 4 (36.4) | 6 (54.5) | 7 (63.6) |
| AEs of moderate intensity | 2 (18.2) | 1 (9.1) | 4 (36.4) | 6 (54.5) |
| AEs of severe intensity | 1 (9.1) | 0 | 0 | 1 (9.1) |
| SAEs | 1 (9.1) | 0 | 2 (18.2) | 3 (27.3) |
| Study drug–related AEs | 0 | 1 (9.1) | 2 (18.2) | 3 (27.3)d |
| AEs leading to study drug interruption | 0 | 0 | 1 (9.1) | 1 (9.1) |
| PTc | ||||
| Headache | 0 | 2 (18.2) | 0 | 2 (18.2) |
| Hyperkalemia | 0 | 0 | 2 (18.2) | 2 (18.2) |
AE, adverse event; bid, twice a day; KT, kidney transplant; N, total number of patients; PT, preferred term; SAE, serious AE.
Iptacopan 10 mg, 25 mg, and 100 mg bid.
1 patient had mild skin discoloration and second had mild normochromic normocytic anemia.
PTs with incidence of >2 events in the overall column are reported in this table.
1 patient had lipase increased, second had syncope and urinary tract infection, and third had worsening of nausea.
Discussion
This study has demonstrated the clinical efficacy, safety, and tolerability of iptacopan, an oral complement factor B inhibitor in patients with both native and recurrent KT C3G. Iptacopan showed a statistically significant and clinically relevant reduction in UPCR by 45% following 12 weeks of treatment in patients with native kidney C3G. A statistically significant reduction in C3 deposit score was observed in patients with recurrent KT C3G. Iptacopan was well-tolerated in patients with both native and recurrent KT C3G with no new safety signals observed; overall safety profile was consistent with previous data.28
Prior prospective investigations of the terminal pathway complement inhibitor eculizumab in C3G have shown a variable clinical response.20,29 Similarly, a prospective study of the selective C5a receptor inhibitor, avacopan, currently published as abstract only, reported a lower progression of C3G Histological Index of disease chronicity score as compared to placebo,30 suggesting at least a partial beneficial effect of avacopan.30
In contrast, a recent observational study suggests that treatment with MMF and corticosteroids is associated with higher rates of remission and lower rates of kidney failure compared with other combinations of immunosuppressive therapies.31 In this study, treatment with MMF and corticosteroids resulted in 36% of patients developing complete remission (proteinuria <0.5 g/24h) and 43% partial remission (>50% reduction in proteinuria).31 However, the effect of this treatment approach on complement activity was not assessed.
The approach of inhibition of AP in the current study differs from previous studies because it targets the main mechanism of disease in C3G, rather than simply inhibiting the terminal complement pathway. Of note, 6 of the 16 patients in the native C3G cohort (and 9 of 11 in the recurrent KT cohort) had prior treatment with MMF/MPS, highlighting the need for more beneficial therapeutics. The potential benefits of blockade of the proximal complement cascade are supported by data from another clinical trial [NCT03453619] describing a 50.9% reduction in proteinuria (as measured by 24-h UPCR) in 7 patients with native C3G receiving pegcetacoplan.32 Furthermore, our study showed stabilization of eGFR during the period of iptacopan treatment with both native and recurrent KT cohorts compared to the 2 years prior to enrolment into the study and suggests a short-term benefit.
For the first time in published literature, AP inhibition has been shown to lead to parallel changes in biomarkers that are associated with complement dysregulation in patients with C3G. Patients with native C3G receiving iptacopan doses of 100 mg and 200 mg bid reached the target trough level of ≥900 ng/ml, a level expected to provide full target inhibition as per PK/PD modeling predictions. A sustained normalization of serum C3 levels was shown in the majority of patients with native C3G, with the reduction of sC5b9 levels (both plasma and urinary), plasma Bb levels, and Wieslab activity, all consistent with inhibition of the AP. This is the first study demonstrating a statistically significant reduction in C3 deposit scores among patients with recurrent KT C3G following kidney transplantation. These patients achieved similar improvements in serum C3 levels as the native cohort. Although the study does not have direct proof in the native cohort, because repeat biopsies were not part of the protocol in these patients, given the similarities in improvement in complement biomarker profile in the 2 cohorts, it is possible that a similar reduction in kidney C3 deposition could be occurring in patients with native C3G, which is consistent with the observed reduction in urinary markers of inflammation and the reduction in proteinuria. The low serum and urine sC5b9 in the recurrent KT C3G cohort detected at baseline and on treatment require further investigation. It is noteworthy that the urine biomarkers of glomerular inflammation were also low in the recurrent KT C3G cohort possibly due to early stage of the disease in this cohort (Supplementary Figure S5). All patients (except for one) who completed this study continued to receive iptacopan upon entry into the open-label extension study (NCT03955445) evaluating the longer-term efficacy and safety of iptacopan in patients with C3.
This study has several limitations. Although reduction in proteinuria was shown in native C3G, the impact on long-term kidney outcome of this finding is not certain. A recent study suggested that a >50% reduction in proteinuria may be predictive of a lower risk of kidney failure and provides evidence that the proteinuria reduction in the native cohort is likely to be beneficial.33 Evidence suggests that this reduction in proteinuria may be directly attributed to the effects of targeting the AP, the underlying disease mechanism in C3G. The study design limits the ability to directly correlate the reduction in urine protein (native cohort) with changes in glomerular histology (recurrent KT cohort). Whether the parallel improvement in complement biomarkers and kidney deposits in the recurrent KT cohort suggests a similar change in kidney injury in the native cohort remains speculative. Regardless, when extrapolating the effects of iptacopan to C3 deposit score from patients with recurrent KT disease to native disease, the differences in baseline proteinuria and complement biomarkers are likely to reflect differing level of disease activity in the 2 cohorts at time of study. Finally, inherent study design–related limitations likely exist given that this was an open-label study. Having demonstrated the ability to target the underlying mechanism of disease in patients with C3G with iptacopan 200 mg bid, it was possible to proceed to a phase 3 study (ongoing, NCT04817618). This study is designed to evaluate the clinical benefit of iptacopan in a double-blinded randomized placebo-controlled trial in patients with native C3G.34
In summary, the present study demonstrates the potential for iptacopan to prevent the progression of kidney disease and transplant graft loss in a kidney disease for which there are currently no approved treatments. Iptacopan was well-tolerated and demonstrated a dose-dependent, sustained inhibition of the complement AP in both kidney disease cohorts. At 12 weeks, iptacopan treatment led to a statistically significant reduction in urinary protein excretion (UPCR) in patients with native C3G and a significantly reduced histologic C3 deposit score in patients with recurrent KT C3G.
It provides unprecedented evidence as follows: that (i) factor B activity and complement AP activation drive C3G markers of severity and pathogenesis and (ii) therapeutic inhibition of specific complement proteins can modify complement-dependent inflammation and kidney impairment in complement-mediated kidney diseases. Despite being a small phase 2 trial, this is a landmark study that has provided insights that may alter the research and management of glomerular diseases.
Disclosure
EW is or was recently a speaker and/or advisor for and/or has received research funding and/or leadership role in Apellis, Biocryst, Novartis, Alexion, and UK Kidney Association MPGN C3G Rare Disease Group-Chair. CN has received research support and/or grants from Biocryst, Achillion, Novartis, Apellis, and Silence Therapeutics; and participated on data safety monitoring board of Kira. TC has no conflicts of interest to declare. AK has received consulting fees and/or payment for lectures from Otsuka, Vifor, GSK, and AstraZeneca. MLQ has no conflicts of interest to declare. LL is or was recently a speaker and/or advisor for and/or has received research funding and/or received consulting fees and/or payment for lectures from Alexion, Novartis, GSK; and participated on data safety monitoring board of Alexion, AstraZeneca, Biogen Idec, BMS, Kezar, Pfizer, Novartis, and GSK; and/or leadership role in Kidney Research UK and Deputy Chair ISN Western EU Regional board, and member of the International Society of Nephrology Executive Council. UE has received payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing, or educational events from Novartis, Biotest, Astellas, and Chiesi. MJS has received grants and/or payment and/or participated on data safety monitoring board of Instituto Carlos III, Marató TV3, Novo Nordisk, Jansen, Boehringer, Mundipharma, AstraZeneca, Ingelheim Lilly, Vifor, ICU Medical, Bayer, GE Healthcare, and Travere Therapeutics; and leadership role as Cardiorenal Group Spanish Society of Cardiology (SEC) group member, Big Data Spanish Society of Nephrology (SEN) Board member, Ex European Renal Association (ERA) Board member, and Western Europe representative of International Society of Nephrology. DK has received grants and/or consulting fees from Wellcome Trust, MRC, Macular society, Gyroscope Therapeutics, Kidney Research UK, Alexion, Silence Therapeutics, Sarepta, and Novartis; and has patent filed Methods of Producing Recombinant Complement Proteins, Vectors and Therapeutic Uses U.S. Patent Application No. 17/928,798 and leadership role as Director of the National Renal Complement Therapeutics Centre. ED has no conflicts of interest to declare. MP has received payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing or educational events from Novartis, Otsuka, Alexion, Vifor, and Travere; and participated on data safety monitoring board of Travere, Otsuka, Alexion, Vifor, and Sanofi. NRM-T has received consulting fees from Silence Therapeutics and participated on data safety monitoring board of Novartis. AG is or was recently a speaker and/or has received grants from Novartis, Alexion, AstraZeneca, Sanofi, Roche, Otsuka, Vera Therapeutics, Chiesi, Ablynx, and Biomerieux; and participated on advisory board of Novartis, Alexion, AstraZeneca, Sanofi, Ablynx, and Travere. CG-C has received travel and congress fees support from AstraZeneca, Esteve, NovoNordisk, Boehringer Ingelheim Lilly, Astellas, Otsuka, Novartis, Astellas, and Baxter; and has given scientific lectures and participated in advisory boards organized by AstraZeneca, Boehringer Ingelheim Lilly, Mundipharma, Esteve, and NovoNordisk; and has leadership role on Clinical Kidney Journal editorial board and on Kidney News editorial board. AB, FC, NJAW, and GJ are employees of Novartis AG, Basel, Switzerland. KK is an employee of Novartis Institutes for BioMedical Research, Cambridge, Massachusetts, USA. JM was an employee of Novartis until time of manuscript submission. PKN is an employee of Novartis Healthcare Pvt. Ltd., Hyderabad, India. GR has received consulting fees from Alexion Pharmaceuticals, BioCryst Pharmaceuticals, and Silence Therapeutics
Acknowledgments
The authors would like to thank all patients, investigators, and the participants sites involved in this study. The authors gratefully acknowledge Drs Agnes Fogo, Terry Cook and Sanjeev Sethi for their immense support in developing the score, for scoring the histopathological data and for the review of the manuscript. The authors would also like to thank Megha Bansal and Anuja Shah for providing medical writing support and editorial assistance, and Sushant Thakur for his technical support in submission of paper (all Novartis Healthcare Pvt. Ltd., Hyderabad, India) and Ronda Tawfik and Angelo Trapani for the help with the historical data. All under the direction of the authors and in accordance with Good Publication Practice guidelines (www.ismpp.org/gpp-2022). Writing support was provided by Novartis Pharma AG, Basel, Switzerland.
Funding
The study was funded by Novartis Pharma AG, Basel, Switzerland.
Author Contributions
CN, AB, MQ, UE, PKN, NW, JM, and GJ performed the conception and design of the study. EW, TC, AK, MQ, LL, UE, DK, ED, MP, NM, AG, CG-C, AB, FC, PKN, NW, JM, and GJ participated in execution of the study and acquisition of the data. EW, CN, LL, UE, MJS, DK, ED, MP, AG, AB, FC, PKN, NW, JM, KK, GJ, and GR participated in analysis and interpretation of the study data. PKN performed the statistical analysis of the study and all authors participated in writing and reviewing the manuscript. All authors edited the manuscript for intellectual content, provided guidance during manuscript development and approved the final version submitted for publication.
Data Statement
Anonymized patient-level data from clinical trials may be shared by Novartis in a consortium called ClinicalStudyDataRequest.com (CSDR) in accordance with Novartis’ policy for sharing clinical trial data (https://www.clinicalstudydatarequest.com/Study-Sponsors/Study-Sponsors-Novartis.aspx).
Footnotes
Supplementary Methods.
Figure S1. Study design.
Figure S2. Proteinuria function after iptacopan treatment native cohort and recurrent KT cohort.
Figure S3. Kidney function as assessed by eGFR after iptacopan treatment in patients with native kidney or recurrent C3G after tansplant.
Figure S4. PK parameters and UPCR (24 hour) in patients with native C3G.
Figure S5. Effect of iptacopan on urine biomarkers.
Table S1. Prohibited medications.
Table S2. Patient disposition and analysis sets.
Table S3. Summary statistics of LNP023 PK parameters after multiple dose administration of LNP023 10, 25, 100, or 200 mg to subjects in the native cohort and recurrent KT cohorts (PK analysis set).
CONSORT Checklist.
Supplementary Materials
Supplementary Methods.
Figure S1. Study design.
Figure S2. Proteinuria after iptacopan treatment native cohort and recurrent KT cohort.
Figure S3. Kidney function as assessed by eGFR after iptacopan treatment in patients with native kidney or recurrent C3G after tansplant.
Figure S4. PK parameters and UPCR (24 hour) in patients with native C3G.
Figure S5. Effect of iptacopan on urine biomarkers.
Table S1. Prohibited medications.
Table S2. Patient disposition and analysis sets.
Table S3. Summary statistics of LNP023 PK parameters after multiple dose administration of LNP023 10, 25, 100, or 200 mg to subjects in the native cohort and recurrent KT cohorts (PK analysis set).
CONSORT Checklist.
References
- 1.Caravaca-Fontán F., Lucientes L., Cavero T., Praga M. Update on C3 glomerulopathy: a complement-mediated disease. Nephron. 2020;144:272–280. doi: 10.1159/000507254. [DOI] [PubMed] [Google Scholar]
- 2.Heiderscheit A.K., Hauer J.J., Smith R.J.H. C3 glomerulopathy: understanding an ultra-rare complement-mediated renal disease. Am J Med Genet C Semin Med Genet. 2022;190:344–357. doi: 10.1002/ajmg.c.31986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sethi S., Vrana J.A., Fervenza F.C., et al. Characterization of C3 in C3 glomerulopathy. Nephrol Dial Transplant. 2017;32:459–465. doi: 10.1093/ndt/gfw290. [DOI] [PubMed] [Google Scholar]
- 4.Angioi A., Fervenza F.C., Sethi S., et al. Diagnosis of complement alternative pathway disorders. Kidney Int. 2016;89:278–288. doi: 10.1016/j.kint.2015.12.003. [DOI] [PubMed] [Google Scholar]
- 5.Sethi S., Haas M., Markowitz G.S., et al. Mayo Clinic/renal pathology society consensus report on pathologic classification, diagnosis, and reporting of GN. J Am Soc Nephrol. 2016;27:1278–1287. doi: 10.1681/ASN.2015060612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Smith R.J.H., Appel G.B., Blom A.M., et al. C3 glomerulopathy - understanding a rare complement-driven renal disease. Nat Rev Nephrol. 2019;15:129–143. doi: 10.1038/s41581-018-0107-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ahmad S.B., Bomback A.S. C3 glomerulopathy: pathogenesis and treatment. Adv Chronic Kidney Dis. 2020;27:104–110. doi: 10.1053/j.ackd.2019.12.003. [DOI] [PubMed] [Google Scholar]
- 8.Ravindran A., Fervenza F.C., Smith R.J.H., De Vriese A.S., Sethi S. C3 glomerulopathy: ten years’ experience at Mayo Clinic. Mayo Clin Proc. 2018;93:991–1008. doi: 10.1016/j.mayocp.2018.05.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhang Y., Meyer N.C., Fervenza F.C., et al. C4 nephritic factors in C3 glomerulopathy: a case series. Am J Kidney Dis. 2017;70:834–843. doi: 10.1053/j.ajkd.2017.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sethi S., Fervenza F.C. Pathology of renal diseases associated with dysfunction of the alternative pathway of complement: C3 glomerulopathy and atypical hemolytic uremic syndrome (aHUS) Semin Thromb Hemost. 2014;40:416–421. doi: 10.1055/s-0034-1375701. [DOI] [PubMed] [Google Scholar]
- 11.Bomback A.S., Santoriello D., Avasare R.S., et al. C3 glomerulonephritis and dense deposit disease share a similar disease course in a large United States cohort of patients with C3 glomerulopathy. Kidney Int. 2018;93:977–985. doi: 10.1016/j.kint.2017.10.022. [DOI] [PubMed] [Google Scholar]
- 12.Medjeral-Thomas N.R., O’Shaughnessy M.M., O’Regan J.A., et al. C3 glomerulopathy: clinicopathologic features and predictors of outcome. Clin J Am Soc Nephrol. 2014;9:46–53. doi: 10.2215/CJN.04700513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Servais A., Noël L.-H., Roumenina L.T., et al. Acquired and genetic complement abnormalities play a critical role in dense deposit disease and other C3 glomerulopathies. Kidney Int. 2012;82:454–464. doi: 10.1038/ki.2012.63. [DOI] [PubMed] [Google Scholar]
- 14.Zahir Z., Wani A.S., Gupta A., Agrawal V. Pediatric C3 glomerulopathy: a 12-year single-center experience. Pediatr Nephrol. 2021;36:601–610. doi: 10.1007/s00467-020-04768-0. [DOI] [PubMed] [Google Scholar]
- 15.Smith R.J., Alexander J., Barlow P.N., et al. New approaches to the treatment of dense deposit disease. J Am Soc Nephrol. 2007;18:2447–2456. doi: 10.1681/ASN.2007030356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rabasco C., Cavero T., Román E., et al. Effectiveness of mycophenolate mofetil in C3 glomerulonephritis. Kidney Int. 2015;88:1153–1160. doi: 10.1038/ki.2015.227. [DOI] [PubMed] [Google Scholar]
- 17.Angelo J.R., Bell C.S., Braun M.C. Allograft failure in kidney transplant recipients with membranoproliferative glomerulonephritis. Am J Kidney Dis. 2011;57:291–299. doi: 10.1053/j.ajkd.2010.09.021. [DOI] [PubMed] [Google Scholar]
- 18.Zand L., Lorenz E.C., Cosio F.G., et al. Clinical findings, pathology, and outcomes of C3GN after kidney transplantation. J Am Soc Nephrol. 2014;25:1110–1117. doi: 10.1681/ASN.2013070715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kidney Disease: Improving Global Outcomes (KDIGO) Glomerular Diseases Work Group KDIGO 2021 clinical practice guideline for the management of glomerular diseases. Kidney Int. 2021;100:S1–S276. doi: 10.1016/j.kint.2021.05.021. [DOI] [PubMed] [Google Scholar]
- 20.Bomback A.S., Smith R.J., Barile G.R., et al. Eculizumab for dense deposit disease and C3 glomerulonephritis. Clin J Am Soc Nephrol. 2012;7:748–756. doi: 10.2215/CJN.12901211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gurkan S., Fyfe B., Weiss L., Xiao X., Zhang Y., Smith R.J. Eculizumab and recurrent C3 glomerulonephritis. Pediatr Nephrol. 2013;28:1975–1981. doi: 10.1007/s00467-013-2503-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Le Quintrec M., Lapeyraque A.L., Lionet A., et al. Patterns of clinical response to eculizumab in patients with C3 glomerulopathy. Am J Kidney Dis. 2018;72:84–92. doi: 10.1053/j.ajkd.2017.11.019. [DOI] [PubMed] [Google Scholar]
- 23.Wong E.K.S., Marchbank K.J., Lomax-Browne H., et al. C3 glomerulopathy and related disorders in children: etiology-phenotype correlation and outcomes. Clin J Am Soc Nephrol. 2021;16:1639–1651. doi: 10.2215/CJN.00320121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Schubart A., Anderson K., Mainolfi N., et al. Small-molecule factor B inhibitor for the treatment of complement-mediated diseases. Proc Natl Acad Sci. 2019;116:7926–7931. doi: 10.1073/pnas.1820892116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kościelska-Kasprzak K., Bartoszek D., Myszka M., Zabińska M., Klinger M. The complement cascade and renal disease. Arch Immunol Ther Exp (Warsz) 2014;62:47–57. doi: 10.1007/s00005-013-0254-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.World Medical Association World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310:2191–2194. doi: 10.1001/jama.2013.281053. [DOI] [PubMed] [Google Scholar]
- 27.Singh J., Singh J. International conference on harmonization of technical requirements for registration of pharmaceuticals for human use. J Pharmacol Pharmacother. 2015;6:185–187. doi: 10.4103/0976-500X.162004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Risitano A.M., Röth A., Soret J., et al. Addition of iptacopan, an oral factor B inhibitor, to eculizumab in patients with paroxysmal nocturnal haemoglobinuria and active haemolysis: an open-label, single-arm, phase 2, proof-of-concept trial. Lancet Haematol. 2021;8:e344–e354. doi: 10.1016/S2352-3026(21)00028-4. [DOI] [PubMed] [Google Scholar]
- 29.Ruggenenti P., Daina E., Gennarini A., et al. C5 convertase blockade in membranoproliferative glomerulonephritis: a single-arm clinical trial. Am J Kidney Dis. 2019;74:224–238. doi: 10.1053/j.ajkd.2018.12.046. [DOI] [PubMed] [Google Scholar]
- 30.Bomback A., Herlitz L.C., Yue H., Kedia P.P., Schall T.J., Bekker P. POS-112 effect of avacopan, a selective C5a receptor inhibitor, on complement 3 glomerulopathy histologic index of disease chronicity. Kidney Int Rep. 2022;7(suppl 2):S47–S48. doi: 10.1016/j.ekir.2022.01.124. [DOI] [Google Scholar]
- 31.Caravaca-Fontán F., Díaz-Encarnación M.M., Lucientes L., et al. Mycophenolate mofetil in C3 glomerulopathy and pathogenic drivers of the disease. Clin J Am Soc Nephrol. 2020;15:1287–1298. doi: 10.2215/CJN.15241219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dixon B.P., Greenbaum L.A., Huang L., et al. Clinical safety and efficacy of pegcetacoplan in a phase 2 study of patients with C3 glomerulopathy and other complement-mediated glomerular diseases. Kidney Int Rep. 2023 doi: 10.1016/j.ekir.2023.08.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Caravaca-Fontán F., Díaz-Encarnación M., Cabello V., et al. Longitudinal change in proteinuria and kidney outcomes in C3 glomerulopathy. Nephrol Dial Transplant. 2022;37:1270–1280. doi: 10.1093/ndt/gfab075. [DOI] [PubMed] [Google Scholar]
- 34.Bomback A.S., Kavanagh D., Vivarelli M., et al. Alternative complement pathway inhibition with iptacopan for the treatment of C3 glomerulopathy-study design of the APPEAR-C3G Trial. Kidney Int Rep. 2022;7:2150–2159. doi: 10.1016/j.ekir.2022.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.