Abstract
Temperature-induced changes in thermotolerance and protein composition were examined in heat-shocked cells and high-temperature-grown cells of the extremely thermophilic bacterium Rhodothermus obamensis. The survival at temperatures superoptimal for growth (90 and 95°C) was enhanced in both heat-shocked cells and high-temperature-grown cells relative to that of cells grown at optimal temperatures. In a comparison of protein composition using two-dimensional gel electrophoresis, putative heat shock proteins (HSPs) and high-temperature growth-specific proteins (HGPs) were detected. N-terminal amino acid sequence analysis revealed that the putative HSPs were quite similar to the ATP-binding subunits of ABC transporters and the HGPs were proteins corresponding to domains II and III of elongation factor Tu. These results suggested that this extreme thermophile has developed temperature-induced responses that include increased survival under hyperthermal conditions, changes in protein composition, and also the production of novel HSPs.
The heat shock response is a stress response mechanism found in various organisms from Escherichia coli to mammals and involves the increased synthesis of proteins which protect an organism from thermal stress (22, 25). The nature of the heat shock response of extremely thermophilic microorganisms, which grow at temperatures of above 80°C, is intriguing, considering the fact that they thrive at temperatures far above the limits for most cells (9, 36). In some organisms, thermal stress induces not only synthesis of specific proteins but also elevated levels of certain intracellular compatible solutes (8, 15, 23, 33, 40).
Most work to date on heat shock and thermal acclimation in thermophilic microorganisms has focused on members of the domain Archaea. Holden and Baross found enhanced thermotolerance and increased levels of a 98-kDa protein in the hyperthermophilic archaeon ES4 when its growth temperature was shifted from 95 to 102°C (18). Another hyperthermophile, Pyrodictium occultum, produced increased levels of an ATPase complex (i.e., thermosome) when the culture temperature was shifted from 102 to 108°C (28, 29), and a similar ATPase complex has been identified in Thermoplasma acidophilum (39). In the members of the order Sulfolobales, prominent heat shock proteins with subunit molecular masses of approximately 60 kDa have been reported (36). These major heat shock proteins are thermophilic factor 55 and its relatives and are similar to a eukaryotic protein family known as t-complex polypeptide 1 (13, 21, 27, 34, 35). It has also been suggested that these proteins are involved in the heat shock response and thermal acclimation in the thermoacidophilic archaea (11).
Given that the heat shock response and thermal acclimation are ubiquitous mechanisms in the extremely thermophilic microorganisms, the mechanism in extremely thermophilic bacteria represents an interesting model that can be compared with the archaeal mechanism. Rhodothermus obamensis is an extremely thermophilic bacterium, growing at temperatures between 55 and 85°C, that was recently isolated by Sako et al. from a shallow marine hydrothermal vent in Japan (32). In this study, we sought to determine the thermal acclimation of heat-shocked and high-temperature-grown R. obamensis cells. Temperature-induced changes in protein composition were examined, and the possible function of the novel heat shock protein-like proteins is discussed.
Morphological changes and acquired thermotolerance.
The bacterial strain used in this study was R. obamensis OKD7 (JCM 9785), which was isolated by Sako et al. from a shallow marine hydrothermal vent at Tachibana Bay, Nagasaki Prefecture, Japan (32). For the cultivation of R. obamensis, Jx medium was used (32). R. obamensis was grown at 76°C and harvested in the midexponential growth phase (optimal-temperature-grown cells), grown at 76°C until the midexponential growth phase, and then incubated at 88°C for 1 h (heat-shocked cells), or grown at 84°C and harvested in the midexponential growth phase (high-temperature-grown cells). The heat shock at 88°C for 1 h had little effect on cell viability, and the temperature of 84°C was approximately the maximal temperature that permits growth. Cells in the midexponential growth phase at 84°C are smaller than those grown at 76°C (Fig. 1) and have diminished cell pigments (data not shown). It has been reported that mesophilic bacteria such as Vibrio spp. reduce their cell size or volume under starvation stress and oligotrophic conditions, and it has been suggested that the reduced size or volume is involved in the adaptation to the unusual growth conditions (19, 26). It seems likely, therefore, that the morphological changes in high-temperature-grown cells are also involved in thermal acclimation under hyperthermal conditions.
FIG. 1.
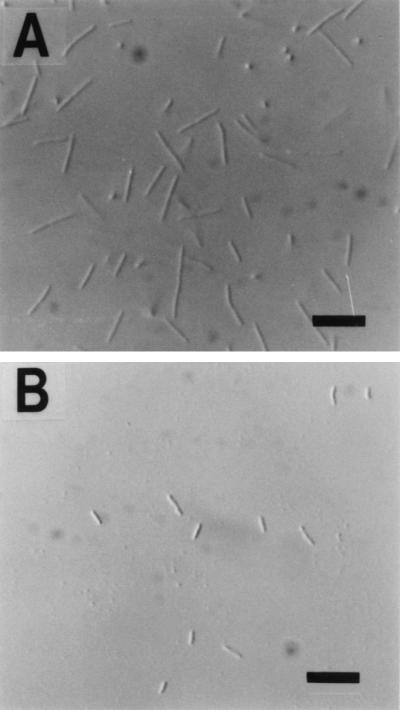
Differential interference micrographs of R. obamensis cells. (A) Cells growing exponentially at the optimum temperature of 76°C. (B) Cells growing exponentially at approximately the maximal temperature of 84°C. Bars, 5 μm.
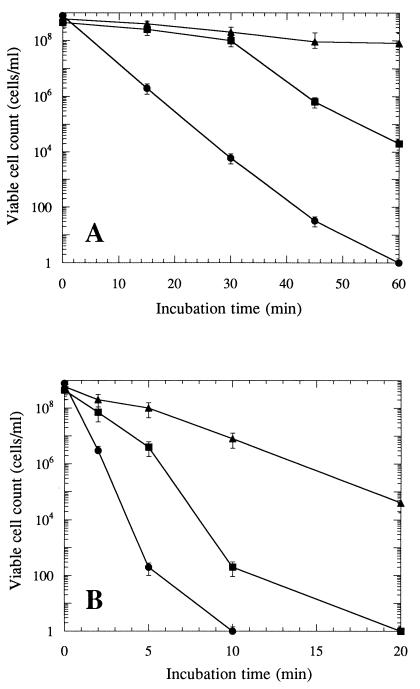
To determine the ability of R. obamensis to withstand hyperthermal conditions, optimum-temperature-grown, heat-shocked, or high-temperature-grown cells were rapidly exposed to lethal temperatures of 90 and 95°C (Fig. 2). The numbers of intact cells and viable cells were estimated by an epifluorescence microscopic method using 4′,6-diamidino-2-phenylindole (30) and by the five-tube most-probable-number technique (2), respectively. Although the total cell counts were constant during the exposure to lethal temperatures, viable cell counts decreased with increasing exposure time. The thermal death curve experiments with optimum-temperature-grown, heat-shocked, and high-temperature-grown cells revealed different levels of thermotolerance. The optimum-temperature-grown cells demonstrated an exponential decrease in viability at both 90 and 95°C, and the heat-shocked cell displayed a significantly lower death rate and transient thermotolerance (Fig. 2). The exponential rate of decline was increased after 30 min at 90°C or after 5 min at 95°C. The cells grown at 84°C were most thermotolerant and were successfully acclimated to hyperthermal conditions. Acquired thermotolerance has been reported in the hyperthermophilic archaeon ES4 and in the extremely thermoacidophilic archaea Sulfolobus shibatae and Metallosphaera sedula (18, 27, 34). In these archaea, thermal acclimation was promoted by prior incubation at temperatures close to the maximum growth temperature for the organisms and was accompanied by the increased synthesis of some proteins. It seems likely that the increased thermotolerance in high-temperature-grown or heat-shocked cells is ubiquitous in the thermophilic microorganisms and represents a possible mechanism for survival under hyperthermal conditions.
FIG. 2.
Thermal-death curves for optimum-temperature-grown cells (•), heat-shocked cells (▪), and high-temperature-grown cells (▴) of R. obamensis exposed to 90°C (A) or 95°C (B). The numbers of viable cells were estimated by the five-tube most-probable-number technique. Error bars represent the 95% confidence interval for each point.
Protein composition variations with temperature changes.
The protein composition was analyzed by two-dimensional (2-D) electrophoresis in optimal-temperature-grown, heat-shocked, and high-temperature-grown cells. Each culture was collected by centrifugation and washed twice with 50 mM Tris-HCl (pH 8.0). The cells were resuspended in lysis buffer containing 9 M urea, 2% (vol/vol) Triton X-100, 2% (vol/vol) 2-mercaptoethanol, and 0.8% ampholine (pH 3.5 to 9.5; Pharmacia), and ruptured by ultrasonication. The cell lysate was centrifuged at 15,000 × g for 10 min, and the protein was precipitated from the supernatant by 5% (wt/vol) trichloroacetic acid. The protein pellet was resuspended with sample buffer containing 8 M urea, 0.5% (vol/vol) Triton X-100, 2% (vol/vol) 2-mercaptoethanol, 0.8% ampholine (pH 3.5 to 9.5; Pharmacia), and 0.01% bromophenol blue, and equal amounts of proteins (50 μg) were applied to 2-D gels that were processed by using the Multiphor II system (Pharmacia). An Immobiline dry strip (pH 4 to 7) and an ExcelGel sodium dodecyl sulfate gradient (8 to 18%) were used for the first and second dimensions, respectively (Pharmacia). The procedure presented in the manufacturer’s manual was followed. Proteins were visualized after staining with Coomassie brilliant blue R250.
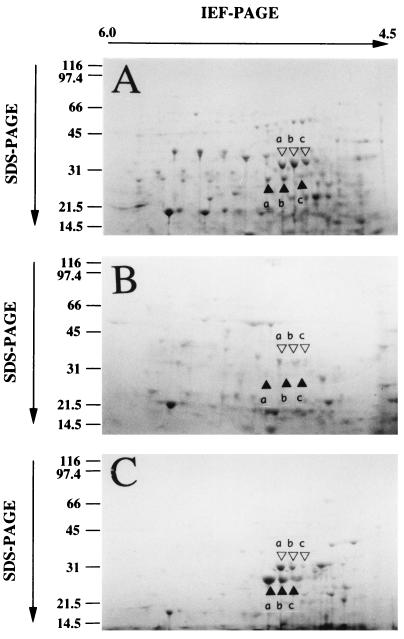
Figure 3 shows the protein compositions of the heat-shocked (A), optimum-temperature-grown (B), and high-temperature-grown cells (C). Although a number of proteins were induced specifically in the heat-shocked or high-temperature-grown cells, multiple proteins of about 35 kDa (open triangles a, b, and c) and protein spots of about 30 kDa (closed triangles a, b, and c) were repeatedly and predominantly recognized in the heat-shocked cells and the high-temperature-grown cells (Fig. 3). Both 35- and 30-kDa proteins were consistently present in the optimum-temperature-grown cells and were increased in both the heat-shocked and the high-temperature-grown cells. When the heat shock incubation period was increased, the expression of 35-kDa heat shock proteins was found to be increased (data not shown). Furthermore, the accumulation of 30-kDa proteins in the high-temperature-grown cells was significantly higher than that in the heat-shocked cells. In accordance with the degree of induction of these proteins, the 35-kDa heat shock proteins and the 30-kDa high-temperature growth-specific proteins were designated the HSP35s and the HGP30s, respectively. On exposure to thermal stress, the accumulation of a 98-kDa protein in the hyperthermophilic archaeon ES4 and of 60-kDa heat shock proteins in members of the order Sulfolobales has been observed (18, 27, 34). However, proteins with molecular masses similar to those of not only such archaeal proteins but also major bacterial heat shock proteins such as those in the Clp family (10), HtpG (4), DnaK (3), GroEL, and GroES (14) were not identified as prominent protein components of R. obamensis cells under hyperthermal conditions. The HSP35s and HGP30s in R. obamensis were significantly different in molecular size from other major heat shock protein families (22, 25).
FIG. 3.
2-D polyacrylamide gel electrophoresis (PAGE) analysis of proteins from heat-shocked cells (A), optimal-temperature-grown cells (B), and high-temperature-grown cells (C). IEF, isoelectric focusing; SDS, sodium dodecyl sulfate.
To identify the HSP35s and HGP30s in R. obamensis, the N-terminal amino acid sequences of these proteins were determined. The proteins in the 2-D polyacrylamide gels were transferred to polyvinylidene difluoride membranes (Bio-Rad) by using the Multiphor II system, and the protein spots revealed by staining with Coomassie brilliant blue, corresponding to the HSP35s and HGP30s, were analyzed by Edman degradation with a 476A protein sequencer (Applied Biosystems Inc.). The amino acid sequence AVLEIRNLHARVEEKEILKGVNLTVNAG was the same in all three HSP35s, and the sequence of all three HGP30s was NAVDEYIPTPVRDKDKPFLMPIEDV. Multi-isoelectric heat shock proteins were often observed in prokaryotic and eukaryotic cells based on 2-D polyacrylamide gel electrophoresis (13, 20). The amino acid sequences of HSP35s and HGP30s were analyzed by using the basic local alignment search tool (1) to estimate the degree of similarity to other amino acid sequences (Fig. 4). The N-terminal amino acid sequences of HSP35s had significant similarity to the N-terminal sequences of the ATP-binding subunits of bacterial ATP-binding cassette (ABC) transporters (Fig. 4A), while the sequences of HGP30s were highly similar to the internal amino acid sequences of bacterial elongation factor Tu (EF-Tu) (Fig. 4B). The molecular masses of the HSP35s were consistent with those of the ATP-binding subunits of ABC transporters (C subunit of the Fe-hydroximate transporter in E. coli and P subunits of the histidine permeases in E. coli and Salmonella typhimurium) (16, 17). These results implied that the HSP35s were identical polypeptides with different pI values and were possible components of the ABC transporter family in R. obamensis. Likewise, the sequence analysis revealed that the HGP30s were identical proteins having different pI values and were derived from the EF-Tu of this organism. The HGP30s corresponded to the C-terminal half of EF-Tu, resembling domains II and III of the bacterial EF-Tu (6, 24). This is the first report that a high abundance of EF-Tu lacking domain I is accumulated in the cytoplasm under heat shock-like or hyperthermophilic conditions.
FIG. 4.
Similarity analysis of HSP35s (A) and HGP30s (B). N-terminal amino acid sequences of HSP35s and HGP30s were analyzed with the basic local alignment search tool and aligned with closely related amino acid sequences. An asterisk or dot under the alignment indicates identical or similar amino acids, respectively. The amino acid sequences shown have the following GenBank accession numbers: Cyanophora paradoxa chloroplast ABC transporter (chlr.ABC), U30821; Porphyra purpurea chloroplast ABC transporter (chlr.ABC), U38804; E. coli histidine transport ATP-binding protein (HisP), D90861; Methanococcus jannaschii ABC transporter, U67462; S. typhimurium histidine transport ATP-binding protein (HisP), J01805; Agrobacterium tumefaciens nopaline permease ATP-binding protein (NocP), M77785; Thermus thermophilus EF-Tu, X06657; T. aquaticus EF-Tu, X66322; Thermotoga maritima EF-Tu, M27479; E. coli EF-Tu, J01690; Corynebacterium glutamicum EF-Tu, X77034.
In this study, it was demonstrated that the ATP-binding subunits of the ABC transporter family and a polypeptide homologous to domains II and III of EF-Tu (EF-TuII+III) were accumulated in heat-shocked and high-temperature-grown cells of R. obamensis. These HSP35s and HGP30s consisted of multi-isoelectric isoforms. It has been reported that several stress proteins also had isoforms with different isoelectric points, which were due to posttranslational modification such as phosphorylation and adenylylation (13, 20). In addition, the isoform compositions of the HSP35s of optimal-temperature-grown and heat-shocked cells differed. The order of the intensities of HSP35 isoforms was b > a > c in heat-shocked cells, while it was a > b > c in cells grown at optimal temperatures (Fig. 3). The result suggested that posttranslational modification plays an important role in the induction and regulation of HSP35s under the heat shock condition.
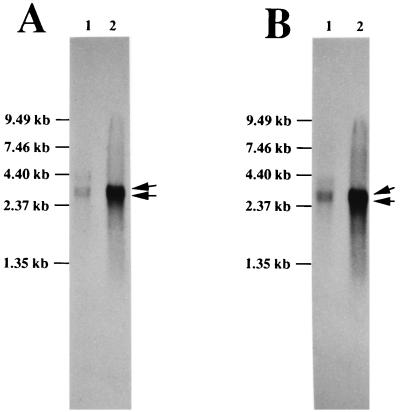
The molecular basis of the induction and regulation of these HSP35s and HGP30s is now under investigation. First, HGP30 accumulation mechanism in high-temperature-grown cells and the relationship of HGP30s to intact EF-Tu should be clarified. To prove that the HGP30s are formed from the intact EF-Tu of R. obamensis, the gene (tuf) for EF-Tu was analyzed by using a PCR. Part of the tuf gene was amplified by a PCR with primers having the sequences 5′-CACGTKGAYCATGGTAAAAC-3′ and 5′-TTATCWCCAGGCATWACCATYTC-3′, corresponding to the highly conserved amino acids in the bacterial EF-Tu sequences reported to date and nucleotide positions 58 to 77 and 1045 to 1067 in the E. coli tufA gene (accession no. J01690), respectively. About 1 kb of the PCR product was directly sequenced on both strands by the dideoxynucleotide chain termination method with a 373As DNA sequencer (Applied Biosystems Inc.). The translated amino acid sequence of the partial tuf gene contained the N-terminal amino acid sequence of HGP30s. This indicated that the HGP30s are constituents of intact R. obamensis EF-Tu. In addition, Southern and Northern analyses were carried out by using RNA probes labeled with digoxigenin (DIG)-11-UTP, corresponding to domain I (not the HGP30 region but nucleotide positions 58 to 550 of the E. coli tufA gene) and domains II and III (HGP30 region, nucleotide positions 580 to 1067 in the E. coli tufA gene) of the partial tuf gene fragment, respectively. A DIG RNA labeling kit (Boehringer Mannheim) was used for labeling as described in the manufacturer’s manual. The Southern analysis indicated that R. obamensis EF-Tu was encoded by two copies of genes (data not shown). In the Northern analysis, both probes hybridized with 3.4- and 3.6-kb RNA fragments in the optimal- and high-temperature-grown cells, indicating that domain I and domains II and III of the tuf genes were transcribed as the same transcription units in both types of cells (Fig. 5). Furthermore, the expression levels of the 3.4- and 3.6-kb transcription units were much higher in the high-temperature-grown cells than in the optimal-temperature-grown cells (Fig. 5). These results suggested that the induction of HGP30s in the high-temperature-grown cells was preceded by induction of intact EF-Tu on the transcriptional level, and then HGP30s were induced and accumulated through posttranslational processes.
FIG. 5.
Northern blot analysis of total RNAs from optimal- and high-temperature-grown cells of R. obamensis. Total RNAs were extracted from optimal- and high-temperature-grown cells with the QuickPrep Total RNA Extraction Kit (Pharmacia). Equal amounts of RNA (1.5 μg; lane 1, from cells grown at an optimal temperature; lane 2, from cells grown at a high temperature) were size fractionated on a formaldehyde-denaturing agarose gel (1.0%), transferred to a positively charged nylon membrane (Boehringer Mannheim), and hybridized with RNA probes corresponding to domain I (A) and domains II and III (B) of the partial tuf gene sequence, respectively. Detection of DIG-labeled RNA hybridized with homologous RNA was carried out with a DIG luminescence detection kit for nucleic acids (Boehringer Mannheim). The arrows on the right indicate the 3.4- and 3.6-kb RNA fragments that hybridized with the probes. The RNA size marker used was from GIBCO BRL, and molecular sizes are indicated on the left.
The proteins which accumulated under heat shock-like or hyperthermophilic conditions have not previously been described as heat shock proteins despite the great number of heat shock proteins reported to date (22, 25, 38). No evidence exists that links the enhanced thermotolerance of R. obamensis with induction of its HSP35s and HGP30s. However, it is well understood that the expression and synthesis of bacterial ABC transporters are regulated in response to specific growth conditions and various cellular functions are also regulated by the ABC transporters (16, 17). These points suggest that ABC transporters are involved in the heat shock response in R. obamensis and their presumed function is the transport of molecules into or out of the cell. Furthermore, it has also been pointed out that EF-Tu can participate in multiple cellular activities besides polypeptide chain elongation, such as the formation of RNA replicase (7), interaction with adenylate cyclase (31), and the formation of cytoskeleton-like filament bundles (5). Of these functions, the formation of filament bundles results from polymerization of EF-Tu in the presence of salts and the filaments are formed not only from intact EF-Tu but also from EF-TuII+III (5, 12). Recently, it has been shown that the chaperonins in the extremely thermophilic archaeon S. shibatae, which are the major heat shock proteins and are composed of its two most abundant proteins (thermophilic factors 55-α and -β), form a cytoskeleton-like filament in vitro and in vivo and that the primary function of these heat shock proteins is cytoskeleton formation (37). Therefore, promotion of cytoskeleton formation in high-temperature-grown cells could be a mechanism for survival and adaptation under hyperthermal conditions due to the stabilization of cell structure and immobilization of the cell membrane.
In summary, the significant morphological changes, the enhanced thermotolerance during hyperthermia, and the increased abundance of specific proteins show that a temperature-induced cellular response occurs in the extremely thermophilic bacterium R. obamensis. The induced proteins, factors that could be responsible for thermotolerance, are different from the other heat shock proteins reported to date. The function of the HSP35s and HGP30s and their role in responding to high temperatures are the foci of ongoing research.
Acknowledgments
This work was supported in part by a Grant-in-Aid for Scientific Research (no. 07556048) from the Ministry of Education, Science and Culture of Japan and by JSPS Research Fellowships for Young Scientists (no. 2702).
REFERENCES
- 1.Altschul S F, Gish W, Miller W, Myers E W, Lipman D J. Basic local alignment search tool. J Mol Biol. 1990;215:403–410. doi: 10.1016/S0022-2836(05)80360-2. [DOI] [PubMed] [Google Scholar]
- 2.American Public Health Association, American Water Works Association, and Water Pollution Control Federation. Methods for the examination of water and waste water. 14th ed. Washington, D.C: American Public Health Association; 1972. [Google Scholar]
- 3.Bardwell J C A, Craig E A. Major heat shock gene of Drosophila and Escherichia coli heat-inducible dnaK gene are homologous. Proc Natl Acad Sci USA. 1984;81:848–852. doi: 10.1073/pnas.81.3.848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bardwell J C A, Craig E A. Eukaryotic Mr 83,000 heat shock protein has a homologue in Escherichia coli. Proc Natl Acad Sci USA. 1987;84:5177–5181. doi: 10.1073/pnas.84.15.5177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Beck B D. Polymerization of the bacterial elongation factor for protein synthesis, EF-Tu. Eur J Biochem. 1979;97:495–502. doi: 10.1111/j.1432-1033.1979.tb13137.x. [DOI] [PubMed] [Google Scholar]
- 6.Berchtold H, Reshetnikova L, Reiser C O A, Schirmer N K, Sprinzl M, Hilgenfeld R. Crystal structure of active elongation factor Tu reveals major domain rearrangements. Nature. 1993;365:126–132. doi: 10.1038/365126a0. [DOI] [PubMed] [Google Scholar]
- 7.Blumenthal T, Landers T A, Weber K. Bacteriophage Qβ replicase contains the protein biosynthesis elongation factors EF Tu and EF Ts. Proc Natl Acad Sci USA. 1972;69:1313–1317. doi: 10.1073/pnas.69.5.1313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ciulla R, Clougherty C, Belay N, Krishnan S, Zhou C, Byrd D, Roberts M F. Halotolerance of Methanobacterium thermoautotrophicum ΔH and Marburg. J Bacteriol. 1994;176:3177–3187. doi: 10.1128/jb.176.11.3177-3187.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.De Macario E C, Macario A J L. Heat-shock response in archaea. Trends Biotechnol. 1994;12:512–518. doi: 10.1016/0167-7799(94)90059-0. [DOI] [PubMed] [Google Scholar]
- 10.Gottesman S, Squires C, Pichersky E, Carrington M, Hobbs M, Mattick J S. Conservation of the regulatory subunit for the Clp ATP-dependent protease in prokaryotes and eukaryotes. Proc Natl Acad Sci USA. 1990;87:3513–3517. doi: 10.1073/pnas.87.9.3513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Guagliardi A, Cerchia L, Rossi M. Prevention of in vitro protein thermal aggregation by the Sulfolobus solfataricus chaperonin. J Biol Chem. 1995;270:28126–28132. doi: 10.1074/jbc.270.47.28126. [DOI] [PubMed] [Google Scholar]
- 12.Gulewicz K, Faulhammer H G, Sprinzl M. Properties of native and nicked elongation factor Tu from Thermus thermophilus HB 8. Eur J Biochem. 1981;121:155–162. doi: 10.1111/j.1432-1033.1981.tb06444.x. [DOI] [PubMed] [Google Scholar]
- 13.Han C J, Park S H, Kelly R M. Acquired thermotolerance and stressed-phase growth of the extremely thermoacidophilic archaeon Metallosphaera sedula in continuous culture. Appl Environ Microbiol. 1997;63:2391–2396. doi: 10.1128/aem.63.6.2391-2396.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hendrix R W. Purification and properties of GroE, a host protein involved in bacteriophage assembly. J Mol Biol. 1979;129:375–392. doi: 10.1016/0022-2836(79)90502-3. [DOI] [PubMed] [Google Scholar]
- 15.Hensel R, Konig H. Thermoadaptation of methanogenic bacteria by intracellular ion concentration. FEMS Microbiol Lett. 1988;49:75–79. [Google Scholar]
- 16.Higgins C F. ABC transporters: from microorganisms to man. Annu Rev Cell Biol. 1992;8:67–113. doi: 10.1146/annurev.cb.08.110192.000435. [DOI] [PubMed] [Google Scholar]
- 17.Higgins C F. The ABC of channel regulation. Cell. 1995;82:693–696. doi: 10.1016/0092-8674(95)90465-4. [DOI] [PubMed] [Google Scholar]
- 18.Holden J F, Baross J A. Enhanced thermotolerance and temperature-induced changes in protein composition in the hyperthermophilic archaeon ES4. J Bacteriol. 1993;175:2839–2843. doi: 10.1128/jb.175.10.2839-2843.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Humphrey B A, Marshall K C. The triggering effect of surfaces and surfactants on heat output, oxygen consumption and size reduction of a starving marine Vibrio. Arch Microbiol. 1984;140:166–170. doi: 10.1007/BF00454920. [DOI] [PubMed] [Google Scholar]
- 20.Hynes G, Sutton C W, Sally U, Willison K R. Peptide mass fingerprinting of chaperonin-containing TCP-1 (CCT) and copurifying proteins. FASEB J. 1996;10:137–147. doi: 10.1096/fasebj.10.1.8566534. [DOI] [PubMed] [Google Scholar]
- 21.Jerez C A. The heat shock response in mesophilic and thermoacidophilic chemolithotrophic bacteria. FEMS Microbiol Lett. 1988;56:289–294. [Google Scholar]
- 22.Lindquist S, Craig E A. The heat-shock proteins. Annu Rev Genet. 1988;22:631–677. doi: 10.1146/annurev.ge.22.120188.003215. [DOI] [PubMed] [Google Scholar]
- 23.Martins L O, Santos H. Accumulation of mannosylglycerate and di-myo-inositol-phosphate by Pyrococcus furiosus in response to salinity and temperature. Appl Environ Microbiol. 1995;61:3299–3303. doi: 10.1128/aem.61.9.3299-3303.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Morikawa K, LaCour T F M, Nyborg J, Rasmussen K M, Miller D L, Clark B F C. High resolution X-ray crystallographic analysis of a modified form of the elongation factor Tu: guanosine diphosphate complex. J Mol Biol. 1978;125:325–338. doi: 10.1016/0022-2836(78)90406-0. [DOI] [PubMed] [Google Scholar]
- 25.Neidhardt F C, VanBogelen R A. Heat shock response. In: Neidhardt F C, Ingraham J L, Low K B, Magasanik B, Schaechter M, Umbarger H E, editors. Escherichia coli and Salmonella typhimurium: cellular and molecular biology. Washington, D.C: American Society for Microbiology; 1987. pp. 1334–1345. [Google Scholar]
- 26.Novitsky J A, Morita R Y. Morphological characterization of small cells resulting from nutrient starvation of a psychrophilic marine vibrio. Appl Environ Microbiol. 1976;32:617–622. doi: 10.1128/aem.32.4.617-622.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Peeples T L, Kelly R M. Bioenergetic response of the extreme thermoacidophile Metallosphaera sedula to thermal and nutritional stress. Appl Environ Microbiol. 1995;61:2314–2321. doi: 10.1128/aem.61.6.2314-2321.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Phipps B M, Hoffmann A, Stetter K O, Baumeister W. A novel ATPase complex selectively accumulated upon heat shock is a major cellular component of thermophilic archaebacteria. EMBO J. 1991;10:1711–1722. doi: 10.1002/j.1460-2075.1991.tb07695.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Phipps B M, Typke D, Hegerl R, Volker S, Hoffmann A, Stetter K O, Baumeister W. Structure of a molecular chaperone from a thermophilic archaebacterium. Nature. 1993;361:475–477. [Google Scholar]
- 30.Porter K G, Feig Y S. The use of DAPI for identifying and counting microflora. Limnol Oceanogr. 1980;25:943–948. [Google Scholar]
- 31.Reddy P, Miller D, Peterkofsky A. Simulation of Escherichia coli adenylate cyclase activity by elongation factor Tu, a GTP-binding protein essential for protein synthesis. J Biol Chem. 1986;261:11448–11451. [PubMed] [Google Scholar]
- 32.Sako Y, Takai K, Ishida I, Uchida A, Katayama Y. Rhodothermus obamensis sp. nov., a modern lineage of extremely thermophilic marine bacteria. Int J Syst Bacteriol. 1996;46:1099–1104. doi: 10.1099/00207713-46-4-1099. [DOI] [PubMed] [Google Scholar]
- 33.Scholz S, Sonnenbichler J, Schäfer W, Hensel R. Di-myo-inositol-1,1′-phosphate: a new inositol phosphate isolated from Pyrococcus woesei. FEBS Lett. 1992;306:239–242. doi: 10.1016/0014-5793(92)81008-a. [DOI] [PubMed] [Google Scholar]
- 34.Trent J D, Osipiuk J, Pinkau T. Acquired thermotolerance and heat shock in the extremely thermophilic archaebacterium Sulfolobus sp. strain B12. J Bacteriol. 1990;172:1478–1484. doi: 10.1128/jb.172.3.1478-1484.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Trent J D, Nimmesgern E, Wall J S, Hartl F-U, Horwich A. A molecular chaperone from a thermophilic archaebacterium is related to the eukaryotic protein t-complex polypeptide-1. Nature. 1991;354:490–493. doi: 10.1038/354490a0. [DOI] [PubMed] [Google Scholar]
- 36.Trent J D. A review of acquired thermotolerance, heat-shock proteins, and molecular chaperones in archaea. FEMS Microbiol Rev. 1996;18:249–258. [Google Scholar]
- 37.Trent J D, Kagawa H K, Yaoi T, Olle E, Zaluzec N J. Chaperonin filaments: the archaeal cytoskeleton? Proc Natl Acad Sci USA. 1997;94:5383–5388. doi: 10.1073/pnas.94.10.5383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Vanbogelen R A, Neidhardt F C. Ribosomes as sensors of heat and cold shock in Escherichia coli. Proc Natl Acad Sci USA. 1990;87:5589–5593. doi: 10.1073/pnas.87.15.5589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Waldmann T, Nimmesgern E, Nitsch M, Peters J, Pfeifer G, Müller S, Kellermann J, Engel A, Hartl F-U, Baumeister W. The thermosome of Thermoplasma acidophilum and its relationship to the eukaryotic chaperonin TriC. Eur J Biochem. 1995;227:848–856. doi: 10.1111/j.1432-1033.1995.tb20210.x. [DOI] [PubMed] [Google Scholar]
- 40.Zellner G, Kneifel H. Caldopentamine and caldohexamine in cells of Thermotoga species, a possible adaptation to the growth at high temperatures. Arch Microbiol. 1993;159:472–476. [Google Scholar]