Abstract
Background
Golimumab is a humanized inhibitor of Tumor necrosis factor‐alpha, recently approved by the Food and Drug Administration (FDA) for the treatment of Rheumatoid arthritis (RA).
Objectives
The objective of this systematic review was to compare the efficacy and safety of golimumab (alone or in combination with DMARDs or biologics) to placebo (alone or in combination with DMARDs or biologics) in randomized or quasi‐randomized clinical trials in adults with RA.
Search methods
An expert librarian searched six databases for any clinical trials of golimumab in RA, including the Cochrane Central Register of Controlled Trials (CENTRAL), OVID MEDLINE, CINAHL, EMBASE, Science Citation Index (Web of Science) and Current Controlled Trials databases.
Selection criteria
Studies were included if they used golimumab in adults with RA, were randomized or quasi‐randomized and provided clinical outcomes.
Data collection and analysis
Two review authors (JS, SN) independently reviewed all titles and abstracts, selected appropriate studies for full review and reviewed the full‐text articles for the final selection of included studies. For each study, they independently abstracted study characteristics, safety and efficacy data and performed risk of bias assessment. Disagreements were resolved by consensus. For continuous measures, we calculated mean differences or standardized mean differences and for categorical measures, relative risks. 95% confidence intervals were calculated.
Main results
Four RCTs with 1,231 patients treated with golimumab and 483 patients treated with placebo were included. Of these, 436 were treated with the FDA‐approved dose of golimumab 50 mg every four weeks. Compared to patients treated with placebo+methotrexate, patients treated with the FDA‐approved dose of golimumab+methotrexate were 2.6 times more likely to reach ACR50 (95% confidence interval (CI) 1.3 to 4.9; P=0.005 and NNT= 5, 95% confidence interval 2 to 20), no more likely to have any adverse event (relative risk 1.1, 95% Cl 0.9 to 1.2; P=0.44), and 0.5 times as likely to have overall withdrawals (95% Cl 0.3 to 0.8; P=0.005). Golimumab‐treated patients were significantly more likely to achieve remission, low disease activity and improvement in functional ability compared to placebo (all statistically significant). No significant differences were noted between golimumab and placebo regarding serious adverse events, infections, serious infections, lung infections, tuberculosis, cancer, withdrawals due to adverse events and inefficacy and deaths. No radiographic data were reported.
Authors' conclusions
With an overall high grade of evidence, at the FDA‐approved dose, golimumab is significantly more efficacious than placebo in treatment of patients with active RA , when used in combination with methotrexate. The short‐term safety profile, based on short‐term RCTs, is reasonable with no differences in total adverse events, serious infections, cancer, tuberculosis or deaths. Long‐term surveillance studies are needed for safety assessment.
Plain language summary
Golimumab for rheumatoid arthritis
This summary of a Cochrane review presents what we know from research about the effect of biologics on rheumatoid arthritis (RA). The review shows that in people with rheumatoid arthritis;
‐ Golimumab improves the number of tender or swollen joints and other outcomes such as pain and disability (ACR 50).
‐ Golimumab increases the number of people in remission.
‐ Slightly more people who take Golimumab will have minor side effects (such as a minor infection), but this may be the result of chance. There is no difference in the number of people who will have a serious adverse event, compared to people who took a placebo.
We do not have precise information about side effects and complications. This is particularly true for rare but serious side effects. Possible side effects may include a serious infection or upper respiratory infection. Rare complications may include certain types of cancer.
What is rheumatoid arthritis (RA) and what are is golimumab?
When you have rheumatoid arthritis, your immune system, which normally fights infection, attacks the lining of your joints making them inflamed. This inflammation causes your joints to be hot, swollen, stiff, and painful. The small joints of your hands and feet are usually affected first. If the inflammation goes on without treatment, it can lead to damaged joints. Once the joint is damaged it cannot be repaired, so treating rheumatoid arthritis early is important.
Golimumab is an anti‐tumor necrosis factor (TNF)‐alpha biologic drug. Biologics are a group of medications that suppress the immune system and reduce the inflammation in the joints. Even though suppressing the immune system can make it slightly harder to fight off infections, it also helps to stabilize an overactive immune system. By reducing the inflammation, the aim is to help prevent damage to the joints. Golimumab is an injection given once a month.
Best estimate of what happens to people with rheumatoid arthritis who take golimumab:
ACR 50 (number of tender or swollen joints and other outcomes such as pain and disability)
‐ 23 more people out of 100 experienced improvement in the symptoms of their rheumatoid arthritis after 14‐24 weeks with golimumab (23% absolute improvement).
‐ 38 people out of 100 who took golimumab experienced improvement.
‐ 15 people out of 100 who took a placebo experienced improvement.
Disease Remission
‐ 18 more people out of 100 were considered to be in remission after 14‐24 weeks with golimumab (18% absolute improvement).
‐ 22 people out of 100 were considered to be in remission with golimumab.
‐ 4 people out of 100 were considered to be in remission with placebo.
People who dropped out of the studies due to any reason
‐ 5 fewer people out of 100 who took golimumab dropped out of the studies for any reason. (2% absolute difference)
‐ 5 people out of 100 who took golimumab dropped out of the studies for any reason.
‐ 10 people out of 100 who used a placebo dropped out of the studies for any reason.
Summary of findings
Summary of findings for the main comparison. Golimumab 50 mg every 4 weeks + MTX compared to placebo + MTX for rheumatoid arthritis.
| Golimumab 50 mg every 4 weeks + MTX compared to placebo + MTX for rheumatoid arthritis | ||||||
| Patient or population: patients with rheumatoid arthritis Settings: Intervention: Golimumab 50 mg every 4 weeks + MTX Comparison: placebo + MTX | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of Participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| placebo + MTX | Golimumab 50 mg every 4 weeks + MTX | |||||
| ACR50 Follow‐up: 14‐24 weeks | 149 per 1000 | 383 per 1000 (200 to 736) | RR 2.57 (1.34 to 4.94) | 919 (4 studies) | ⊕⊕⊕⊕ high | Absolute risk difference= 18% (95% CI 8% to 27%). Relative percent change=157% (34% to 394%). NNTB=5 (95% CI 2 to 20). 1 |
| Disease activity (DAS Remission) Follow‐up: 14‐24 weeks | 43 per 1000 | 220 per 1000 (72 to 673) | RR 5.12 (1.67 to 15.66) | 919 (4 studies) | ⊕⊕⊕⊕ high | Absolute risk difference=10% (95% CI 6% to 14%). Relative percent change=412% (67% to1466%). NNTB=6 (95% CI 2 to 35).1 |
| Physical function ‐ HAQ Score points. Scale from: 0 to 3. Follow‐up: mean 14 weeks | The mean physical function ‐ haq score in the control groups was 1.6 points2 | The mean Physical function ‐ HAQ Score in the intervention groups was 0.2 lower (0.25 to 0.15 lower) | 308 (1 study) | ⊕⊕⊕⊕ high | Absolute risk difference= ‐20% (95% CI ‐25% to ‐15%). Relative percent change= ‐11% (95% CI ‐14% to‐8.3%) NNT=3 (95% CI 3 to 4)3,4 | |
| Adverse Events Follow‐up: 14‐24 weeks | 693 per 1000 | 728 per 1000 (644 to 818) | RR 1.05 (0.93 to 1.18) | 918 (4 studies) | ⊕⊕⊕⊕ high | Absolute risk difference= 4% (95% CI ‐5% to 12%). Relative percent change= 5% (95% CI ‐7% to 18%). NNTH=n/a (not statistically significant).4,5 |
| Serious Adverse Events Follow‐up: 14‐24 weeks | 56 per 1000 | 59 per 1000 (35 to 100) | RR 1.05 (0.62 to 1.78) | 918 (4 studies) | ⊕⊕⊕⊕ high | Absolute risk difference= 1% (95% CI ‐2% to 4%). Relative percent change= 5% (95% CI ‐38% to 78%). NNTH=n/a (not statistically significant). 4,5 |
| All Withdrawals Follow‐up: mean 14‐24 weeks | 104 per 1000 | 52 per 1000 (32 to 84) | RR 0.5 (0.31 to 0.81) | 917 (4 studies) | ⊕⊕⊕⊕ high | Absolute risk difference= ‐5% (95% CI ‐10% to 1%). Relative percent change= ‐50% (95% CI ‐69% to‐19%). NNTH= 20 (95% CI 14 to 51). 5 |
| Withdrawals due to Adverse Events Follow‐up: 14‐18 weeks | 50 per 1000 | 28 per 1000 (12 to 64) | RR 0.56 (0.24 to 1.29) | 599 (3 studies) | ⊕⊕⊕⊕ high | Absolute risk difference= ‐2% (95% CI ‐5% to 1%). Relative percent change= ‐44% (95% CI ‐76% to 29%). NNTH=n/a (not statistically significant).4,5 |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: Risk ratio; | ||||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | ||||||
1 NNTB= Number needed to treat to benefit 2 The assumed risk of the control group was taken as the median HAQ score of the placebo group at 14 weeks (Smolen 2009). 3 Calculation of the relative percent change in HAQ score was done by dividing the absolute benefit between treatment and control groups by the baseline of the control group 4 n/a=not applicable 5 NNTH= Number needed to treat to harm
Background
Description of the condition
Rheumatoid arthritis (RA) is a systemic disease characterized by destructive, inflammatory polyarthritis affecting small and large joints (Harris 1990). RA has significant impact on both patient‐reported quality of life and function (HRQoL) (Lubeck 2004) (Kvien 2005) and can sometimes lead to serious work disability (Odegard 2005) (Yelin 2007).
The inflammation in RA is characterized by the activation of many immune cells, including but not limited to T‐cells, B‐cells and macrophages (Weissmann 2006). Such immune cells secrete various cytokines (Choy 2005) (Choy 2001) i.e. tumor necrosis factor‐alpha (TNF‐alpha) and interleukins (Brennan 2008) (Choy 2001), which in turn lead to inflammation, and joint and bone destruction. Lately, much of the focus of treatment of RA has been on inhibition of TNF‐alpha, since it is thought to play a central role in joint inflammation (Connell 2006).
Description of the intervention
The approved dose for treatment of RA with golimumab is 50 mg subcutaneous injection given once a month (FDA 2009b). The pharmacological therapeutic strategies for RA include non‐steroidal anti‐inflammatory drugs (NSAIDs), traditional disease‐modifying anti‐rheumatic drugs (DMARDs) such as methotrexate and newer biologic DMARDs. The newer biologic DMARDs include TNF‐alpha inhibitors such as etanercept, infliximab and adalimumab (Scott 2006) and other non‐TNF biologics. Non‐TNF biologic DMARDs (and respective targets) include anakinra (IL‐1), abatacept (co‐stimulatory molecule, CD28) and rituximab (B‐cell) (Siddiqui 2007). The trade name for golimumab is SIMPONI.
How the intervention might work
Golimumab is a humanized inhibitor of TNF‐alpha (FDA 2009a). It neutralizes TNF‐alpha by binding to it, thus interfering with its binding to the TNF‐alpha receptors on cell surfaces. TNF‐alpha plays a key role in the inflammation and joint destruction, which are the hallmarks of RA (Connell 2006).
Why it is important to do this review
Biologic DMARDs are very effective in treating RA. Golimumab is a recently approved biologic DMARD targeting TNF‐alpha, for which no Cochrane systematic review exists. Cochrane systematic reviews have been completed for other biologics including etanercept (Lethaby 2003), infliximab (Blumenauer 2002), adalimumab (Navarro‐Sarabia 2005), abatacept (Maxwell 2008) and anakinra (Mertens 2009) and systematic review for rituximab is being completed (Lopez‐Olivo 2008). Due to the fact that patients refractory to one anti‐TNF agent may respond to a different anti‐TNF agent (Karlsson 2008; Gomez‐Reino 2006), availability of another anti‐TNF such as golimumab offers an additional option for patients with RA.
Objectives
To compare the efficacy and safety of golimumab in adults with rheumatoid arthritis.
Methods
Criteria for considering studies for this review
Types of studies
All randomized controlled trials (RCTs) or Controlled Clinical Trials (CCTs) (methods of allocating participants to a treatment which are not strictly random, e.g., date of birth, hospital record number or alternation) were considered for inclusion.
Types of participants
Adults 18 years or older, with RA meeting the 1987 American College of Rheumatology Classification criteria for RA (Arnett 1988).
Types of interventions
Interventions compared are golimumab alone or in combination with DMARDs or biologics versus placebo plus methotrexate or golimumab alone or in combination with DMARDs or biologics compared to other DMARDs or biologics. There were no restrictions with regard to dosage or duration of intervention.
Types of outcome measures
Primary outcomes
Major Outcomes:
ACR50 defined as 50% improvement in both tender and swollen joint counts and 50% improvement in three of the five following variables: patient global assessment, physician global assessments, pain scores, Health Assessment Questionnaire (HAQ) score, and acute phase reactants (Erythrocyte Sedimentation Rate (ESR) or C‐Reactive Protein (CRP) (Felson 1995) (Chung 2006).
-
Safety: Safety as assessed by the
Number and type of adverse effects (AEs) and serious adverse events (SAEs)
Withdrawals: (i) Total, (ii) due to lack of efficacy, (iii) due to adverse events
Death
Secondary outcomes
Other Outcomes:
ACR20 and ACR70 defined as 20% and 70% improvement in variables defined above under the primary outcome (Felson 1995).
Changes in either Disease Activity Score (DAS), a composite index of tender and swollen joint counts, patient global assessment and ESR (van der Heijde 1993) or DAS28 score (Prevoo 1995). In an occasional case, where median was presented instead of mean, due to large sample size we used median to substitute for mean and calculated standard deviation from the interquartile range.
Proportion achieving a "good state": (a) good European League Against Rheumatism (EULAR) response (Fransen 2005) (van Gestel 1996)‐ defined by a decrease in DAS or DAS28 of >= 1.2 from baseline with a final DAS < 2.4 (or DAS 28 < 3.2); (b) low disease activity defined by DAS < 2.4 or DAS28 <= 3.2 ; (c) remission defined as DAS < 1.6 or DAS28 < 2.6. We used Good EULAR response based on ESR, where data were presented for both ESR‐ and CRP‐based outcomes.
Quality of Life, measured by Short‐Form‐36 *(SF‐36) (i.e. continuous data, 8 domains; and two summary scores, physical and mental component summary) and function measured by HAQ score or modified HAQ calculated as score changes (Fries 1980) (Pincus 1983) and the proportion achieving minimally clinically important difference on HAQ ≥ 0.22 (Wells 1993).
Radiographic progression, as measured by Larsen/Sharp/modified Sharp scores (Larsen 1977) (Sharp 1971) (van der Heijde 1989).
We realize that randomized controlled trials included in this review are limited in their ability to assess safety. We searched U.S. Food and Drug Administration (FDA) web site for labels and warnings and other similar regulatory agency's web sites to summarize warnings related to golimumab.
Search methods for identification of studies
Electronic searches
We searched the following databases (Appendix 1) for randomized or quasi‐randomized trials:
The Cochrane Central Register of Controlled Trials (CENTRAL), via The Cochrane Library, Wiley InterScience (www.thecochranelibrary.com), issue 2, 2009;
OVID MEDLINE, 1966 to June Week 3, 2009 ;
CINAHL (via EBSCOHost), 1982‐2009;
EMBASE 1980‐2009;
Science Citation Index (Web of Science) 1945‐2009;
Current Controlled Trials.
There was no language restriction on included studies and non‐English articles were translated. We did not include any abstracts in this review. Full search strategies are in Appendix 1.
Searching other resources
The FDA web site was searched for references to golimumab trials on 09/01/2009 (http://www.fda.gov/). The search was not limited by language, year of publication or type of publication.
Data collection and analysis
Selection of studies
We used our predetermined criteria to identify potential trials for inclusion. Two review authors (JS, SN) independently assessed the methods sections of all identified trials according to the predetermined assessment criteria (see 'Selection criteria'). Disagreements were resolved by consensus. For disagreements not resolved by consensus, the third reviewer (GS) would have served as the referee.
Data extraction and management
Two review authors (JS, SN) independently extracted data from the included trials. The extracted data included information such as the population of study, interventions, number of centers, funding sources, and outcomes and results using standardized data extraction forms. When we needed more information, we contacted the authors' of the studies.
We extracted raw data for outcomes of interest such as the standard deviation and mean for continuous data and number of events for dichotomous data from the published reports in order to evaluate efficacy. When possible, we extracted data based on the intention to treat analysis.
Assessment of risk of bias in included studies
Two review authors (JS, SN) independently assessed the risk of bias of each included trial using the Cochrane Collaboration recommendations for assessment. The main criteria that were applied to measure the risk of bias included: blinding of participant, care provider and outcome assessor in the studies, allocation concealment, random sequence generation, incomplete outcome data, and selective outcome reporting (Higgins 2008). The risk of bias in each study was explicitly judged on each criterion using the following standard: Yes (low risk of bias), No (high risk of bias), or unclear (either lack of information or uncertainty over the potential for bias). Disagreements were resolved by discussion between the two reviewer authors. If needed, we planned to approach the third review author (GS) for disagreements not resolved.
Measures of treatment effect
The results of each RCT were plotted as point estimates, such as risk ratios (RRs) with 95% confidence interval for dichotomous outcomes, and mean and standard deviation for continuous outcomes. We extracted all possibly extractable results from the included trials. When this was impossible, for example if data were reported as median scores only, we presented and described them in the 'Characteristics of Included Studies' table. For continuous measures, we calculated mean differences when possible because results presented in this form are more readily interpreted by clinicians. We planned to used standardized mean differences as measure of treatment effect for outcomes assessing the same construct but using different outcome measures. In this review, we did not find any outcome using different measures for the same construct, therefore standardized mane difference was not used in this review.
Unit of analysis issues
The patient was the unit of analysis. There were no unit of analysis issues as only studies with standard designs were included in this review.
Dealing with missing data
We summarized this in the study characteristics and summarization of the risk of bias tables. No imputation was done.
Assessment of heterogeneity
The factors assessed for clinical homogeneity were: duration of RA, outcomes, and control groups. If studies were clinically heterogeneous, we planned to exclude them from the meta‐analysis and analyze them separately. We performed a χ2 test (chi squared) (P < 0.10) and I‐squared (I2) to quantify heterogeneity. In interpreting the I2 statistic, we complied with the recommendations put forth in the Cochrane Handbook, which determine that a 0% to 40% might not be important; 30% to 60% may represent moderate heterogeneity; 50% to 90% may represent substantial heterogeneity; and 75% to 100% considerable heterogeneity (Deeks 2008).
Assessment of reporting biases
In order to assess reporting biases, we planned to make a funnel plot when doing a meta‐analysis for 10 or more studies. The funnel plot is a scatter plot that takes the sample size on the y‐axis and the treatment effect along the x‐axis. A good indication of reporting bias is observation of asymmetry in the funnel plot. Since only 4 studies met the criteria, a funnel plot could not be made.
Data synthesis
The random‐effects model was the default model for pooling outcomes in the meta‐analysis of studies determined clinically homogeneous. We reported the data from the fixed effect model to evaluate any bias in smaller studies. The number needed to treat to benefit (NNTB) was calculated as the inverse of the absolute risk difference, for outcomes derived from one trial.
The mean difference was used to calculate the benefit (absolute change expressed as both a percentage and in its original units) for continuous outcomes such as pain measured on a Visual Analog Scale. The absolute benefit divided by the baseline mean of the control group was the method of calculation for the relative difference in the change from baseline.
'Summary of findings' table included in RevMan 5 was completed in order to communicate the key outcomes of the review. We used GRADE software to provide an overall grading of the quality of the evidence.
Subgroup analysis and investigation of heterogeneity
The following sub‐group analyses were planned a priori to explore possible effect size differences, if sufficient data were available:
Concomittant methotrexate vs. no methotrexate
Mean RA disease duration ‐ categorized as early RA defined as duration of less than 2 years (Boers 2001) vs. established RA, duration 2 to10 years vs. late RA defined as >10 years (Barlow 1999)
Use in patients who have methotrexate‐failure vs. biologic‐failure
Single biologic DMARD agent vs. combination biologic therapy
DMARD‐naive vs. not naive
Treatment duration with biologic DMARD: Short (6 months), intermediate duration (6 to 12 months) or long‐duration (>1 year)
Sensitivity analysis
In order to assess for any bias attributed to allocation concealment, a sensitivity analysis was performed in the presence of sufficient data following the criteria mentioned in the 'assessment of risk of bias' section above.
Results
Description of studies
Emery 2009 was a phase III, 52‐week, randomized, double blind, placebo controlled trial assessing the efficacy of golimumab in methotrexate‐naive patients (n=637) ("GO‐BEFORE"). Patients were randomized using an interactive voice response system in equal proportions into one of four groups. 160 patients were randomized into the placebo + methotrexate (MTX) group (group 1), 159 patients were randomized into the 100 mg golimumab + placebo (group 2), 159 patients were randomized into the 50mg + MTX (group 3), and 159 patients were randomized into the 100mg golimumab + MTX (group 4). Golimumab was administered via subcutaneous injections at week 0 and every four weeks thereafter. Patients with active RA according to ACR criteria for at least 3 months before administration of the initial study agent and who had not received more than 3 weekly doses of oral MTX as treatment of RA were included. Patients also must have had at least 4 swollen and 4 tender joints and met two of the following criteria at screening or baseline: CRP level of greater than 1.5 mg/dl or ESR of greater than 28mm/hour according to Westergren method, morning stiffness for at least 30 minutes, bone erosion by radiography and/or magnetic resonance imaging prior to the beginning of the treatment, or anti‐cyclic citrulinated peptide antibody positivity or rheumatoid factor positivity. Patients also had have to passed the TB screening criteria and been treated accordingly to their results to be eligible.
The study had a 52‐week, randomized, double blind, placebo controlled phased followed by a 5 year open label extension period. The primary endpoints of the study were the difference in the ACR50 response at 24 weeks between groups 3 and 4 versus group 1 and a pair wise comparison (group 3 or group 4 versus group 1) and a change in the modified Sharp/van der Heijde score at week 52. The secondary outcomes were ACR20, ACR70, ACR90, ACR‐N, DAS28, DAS28 UELAR, DAS28 Remission, HAQ disability index, and changes in hemoglobin levels in patients with baseline anemia. Efficacy and safety results were presented at 24 weeks. Withdrawals due to inefficacy were not extracted because the numbers did not add up from figure 1 of the study.
Kay 2008 was a multicenter, randomized, double‐blind, placebo‐controlled trial, 5‐arm, dose‐ranging study comparing golimumab + methotrexate to placebo + methotrexate (MTX) in 172 patients with active RA among 40 centers. Patients with active RA despite treatment with oral or parenteral MTX were randomly assigned to 1 of 5 treatment groups in approximately equal proportions to receive golimumab plus MTX or placebo plus MTX. Patients were included in the study if they were: (1) adults with RA, as defined by the 1987 revised criteria of the American College of Rheumatology (ACR; formerly, the American Rheumatism Association) (Arnett 1988); and (2) had active RA despite treatment with MTX (dosage of at least 10 mg/week for 3 months and at a stable dosage for 4 weeks), defined as 6 swollen joints and 6 tender joints and at least 2 of the following 3 criteria: C‐reactive protein (CRP) level 1.5 mg/dl, erythrocyte sedimentation rate (ESR) of 28 mm in the first hour, and morning stiffness of 30 minutes, for at least 3 months before screening. Patients were allowed to receive stable doses of oral corticosteroids (<10 mg/day of prednisone equivalent) and nonsteroidal antiinflammatory drugs (NSAIDs). patients were excluded if: (1) hemoglobin <8.5 gm/dl, white blood cells <3,000/l, neutrophils <1,500/l, platelets <100,000/l; serum transaminase levels >1.5 times the upper limit of normal, and serum creatinine > 1.5 mg/dl; (2) they had history/current evidence of latent or active tuberculosis, recent exposure to active tuberculosis, or a positive tuberculin skin test result. All patients received folic acid supplementation.
Patients continued the MTX and were randomly assigned to: placebo (n=35), 50 mg golimumab every 4 weeks (n=35), 50 mg golimumab every 2 weeks (n=34), 100 mg golimumab every 4 weeks (n=34), or 100 mg golimumab every 2 weeks (n=34). The study medication was administered by subcutaneous injection every 2 weeks from week 0 to week 18. Patients in the golimumab every 4 week groups received placebo injections at alternate visits to maintain blinding. At 20‐weeks follow‐up, patients in the placebo group started open‐label treatment with intravenous infliximab at 3 mg/kg, with induction at weeks 20, 22, and 28 followed by maintenance therapy every 8 weeks through week 44, while patients in golimumab groups continued to receive their assigned dose (50 or 100 mg) from week 20‐48 and their dosing frequency was changed to golimumab every 4 weeks for all four treatment arms. The study was sponsored by Centocor, Inc.
Keystone 2009 was a double‐blind, randomized, placebo controlled, phase III, multicenter, trial taking place in 60 sites in 12 countries. 444 patients were randomized into 4 groups: placebo + methotrexate (n=133), placebo + 100 mg golimumab (n=133), 50 mg golimumab + MTX (n=89), or 100 mg golimumab + MTX (n=89). Golimumab was administered via intracutaneous injections every 4 weeks for up to 16 weeks. At 16 weeks, patients who did not show a 20% improvement in swollen and tender joint counts went into early escape in a blinded manner. Those receiving golimumab 50 mg + MTX received 100 mg golimumab + stable dose of MTX, those on placebo + 100 mg golimumab received active MTX instead of placebo at stable dose and continued with 100 mg of golimumab, and those on placebo + MTX continued on their stable dose of MTX but switched from placebo injections to 50 mg of golimumab every 4 weeks. The study lasted 52 weeks in its double‐blind controlled phase and allowed for a 5 year open‐label extension.
Patients were included in the study if they were 1) 18 years of age or older, 2) diagnosed with RA for at least three months before screening according to the 1987 revised criteria of the American College of Rheumatology (ACR; formerly, the American Rheumatism Association) (Arnett 1988), 3) on a stable methotrexate dose of 15 mg/week or greater but 25mg/week or less during the 4‐week period preceding screening, 4) to have tolerated 15 mg a week or greater of methotrexate for at least 3 months before screening, 4) to have active RA defined as four or more out of 66 swollen joints and four or more out of 68 tender joints and 2 of the 4 following criteria: C‐reactive protein (CRP) level 1.5 mg/dl or greater or erythrocyte sedimentation rate (ESR) of 28 mm or greater in the first hour, morning stiffness of at least 30 minutes, bone erosion determined by x ray and/or MRI, or anticyclic citrullinated peptide antibody or rheumatoid factor positive test results, 5) and to have met the TB screening criteria. Patients using NSAIDS or other analgesics for RA had to be taking a stable dose for at least two weeks prior to taking the first dose of the active trial drug.Those taking oral corticosteroids had to have been receiving a stable dose equivalent to 10mg/day or less of prednisone for at least two weeks before the first dose of study agent. Patients were excluded if they had known hypersensitivity to any ingredients of golimumab, had taken an anti‐TNF agent, rituximab, cytotoxic agents, natalizumab, anakinra, DMARDs other than methotrexate, or intravenous, intraarticular, or intramuscular corticosteroids four weeks prior to the first dose of study agent. Patients taking alefacept or efalizumab within three months of the first trial dose were also excluded.
Smolen 2009 was a 24‐week long, randomized, double blind, placebo controlled trial taking place in 82 sites in 10 countries involving 461 patients. Patients were divided into three groups: placebo+DMARD (n=155), golimumab 50mg every 4 weeks+DMARD (n=153) and golimumab 100mg every 4 weeks+DMARD (n=153) in a double blinded fashion. Stable doses of DMARD treatment was allowed but not required. If patients in placebo or 50mg group did not show an ACR20 improvement by week 16, they entered a double blinded rescue therapy phase to receive 50mg or 100 mg golimumab treatment respectively.
Patients were included if they were 1) 18 years of age or older, 2) diagnosed with RA for at least three months before screening according to American College of Rheumatology (ACR; formerly, the American Rheumatism Association) (Arnett 1988), 3) treated with at least one dose of a TNF‐α inhibitor (at least 8 weeks prior to the first dose of study agent for etanercept and adalimumab, and at least 12 weeks before the first dose of the study agent for infliximab). Previous treatment with the TNFα inhibitor could have been discontinued for any reason. Patients were excluded from the trial if they had other inflammatory diseases, had a serious adverse reaction to a previous TNFα inhibitor, had used natalizumab, rituximab, cytotoxic drugs or anakinra less than 4 weeks or efalizumab or alefacept three months before the first dose of study agent. Patients were also excluded if they had a history of latent or active granulomatous infection and a specified list of other diseases. For a full list of exclusion criteria, refer to page 211.
Results of the search
The results of the search are described below and depicted in Figure 1.
1.

Study Selection Flow Chart
The initial search yielded 187 search results. Of these 187, we identified four studies for full review (Kay 2008, Keystone 2009, Oldfield 2009, O'Connell 2008) (Figure 1). Of these four studies, two qualified for inclusion (Kay 2008, Keystone 2009). A search update performed on 8 August, 2009 yielded 29 results (Figure 1). We identified two more studies for full review both of which qualified for inclusion (Emery 2009, Smolen 2009). In sum, 4 studies were included in this systematic review (Figure 1).
Included studies
Overall, there were 1231 patients treated with golimumab and 483 patients treated with placebo included in the studies, which made for 1714 overall patients in all arms. There were 436 patients in the golimumab 50 mg every four weeks + methotrexate treatment arm, 292 patients in the golimumab 100 mg every four weeks plus placebo, 435 patients in the golimumab 100 mg every four weeks plus methotrexate, 34 patients in the golimumab 100 mg every two weeks plus methotrexate and 34 patients in the golimumab 50 mg every two weeks plus methotrexate treatment arm.
Please refer to the characteristics of included studies table for detailed information on each study (Characteristics of included studies).
Patient characteristics of patients randomized to golimumab arm at 50 mg subcutaneous every 4 weeks (FDA‐approved dose) are provided in Table 2.
1. Characteristics of patients randomized to the golimumab treatment arm at the FDA‐approved dose in included Studies.
| Study Name/year | Intervention group medications | Comparator | % female | Age mean (SD) or median [IQR] |
Prior Mtx failure (yes/no) |
Prior biologic failure (yes/no) |
Mtx in mg/week, Mean (SD) or median [IQR] | HAQ, Baseline, mean (SD) or median [IQR] | Disease Duration in years, mean (SD) or median [IQR] | DAS28, Baseline, mean (SD) or median [IQR] | # DMARDS failed, mean (SD) | |
| Smolen 1999 | Golimumab + Mtx | PL + Mtx | 74% | 55 [46, 63] | Yes‐ 67% | Yes‐ TNF‐inhibitor | ‐‐ | 1.6 [1.1, 2.0] | 9.6 [5.6, 17.2] | 6.3 [5.6, 7.2] | ‐‐ | |
| Kay 2009 | Golimumab + Mtx | PL + Mtx | 86% | 57 [50, 64] | Yes | no | ‐‐ | 1.7 [1.4, 2.0] | 8.2 (4.1, 14.3) | 5.3 [4.5, 6.2]a | ‐‐ | |
| Keystone 2009 | Golimumab + Mtx | PL + Mtx | 81% | 51 [42, 59] | Yes | no | 15.0 [15.0, 20.0] | 1.375 [1.0, 1.9] | 4.50 [2.1 to 9.7] | 5.1 [4.1, 5.6]a | ‐‐ | |
| Emery 2009 | Golimumab + Mtx | PL + Mtx | 85% | 51 (11) | Yes | no | 12.8 (2.2) | 1.5 (0.7) | 3.5 (5.7) | 5.1 (1.0)a | ‐‐ |
aDAS28 using CRP level
IQR, Inter‐quartile range; ‐‐, not provided
Excluded studies
There were two pulled studies that were excluded from the meta analysis (O'Connell 2008, Oldfield 2009). The reasons for their exclusion was that one was a review (Oldfield 2009) and the other was a commentary (O'Connell 2008). Refer to the table of excluded studies (LINK).
Risk of bias in included studies
All studies reported adequate methods of randomization, allocation concealment and blinding. The over all possibility of bias seems to be low. Refer to Figure 2 and Risk of Bias tables for more information on all sections of risk of bias.
2.

Methodological quality summary: review authors' judgements about each methodological quality item for each included study.
Allocation
All four studies reported adequate methods of allocation concealment and randomization. Kay 2008 did not report the method of randomization therefore we contacted the author who reported that they used Interactive Voice Response System [IVRS]. All four studies used the IVRS method of randomization and allocation concealment.
Blinding
All four studies reported methods of blinding. Emery 2009 reported that the study was double‐blind‐ "Active and placebo MTX were supplied as double‐blinded, identical opaque capsules". Kay 2008 reported that the study was double‐blind‐ "Patients and investigators remained blinded during the interim analyses". Keystone 2009 reported that the study was double‐blind‐ "Patients in groups 1, 2 or 3 with less than a 20% improvement from baseline in both tender and swollen joint counts had their study medication adjusted in a double‐blind fashion (i.e., early escape)". Smolen 2009 reported that the study was blinded‐ "Golimumab and placebo were supplied in identical single‐use vials".
Incomplete outcome data
All studies except Keystone 2009 reported adequate methods of handling missing outcome data. Keystone 2009 did not report any statistical methods of accounting for missing outcome data in their article and was therefore contacted. They replied as follows: "For categorical endpoints like ACR20, 50, 70 etc. for primary and major secondary endpoints missing data were considered 'non‐responders.' For continuous endpoints like DAS28 response, remission, ACRn etc. for missing data the LOCF principle was used." Therefore, all studies had adequate methods of handling missing outcome data. We could not use the withdrawal rates due to inefficacy and adverse events from Emery 2009 because they presented withdrawal rates separately for oral and subcutaneous agent in each treatment arm and the total number did not add up.
Selective reporting
Because the protocols available on the Internet for these trials did not provide sufficient information, we did not have an adequate amount of information to make a definitive judgment on the presence of selective reporting in the trials.
Other potential sources of bias
Manufacturers of golimumab played a role either in sponsoring the study drugs, sponsoring the study, providing research grants to authors of the studies, and/or participating in components of the study. There is some evidence that studies with industry support may be more likely to report positive results than those without industry support (Bhandari 2004). However, this and othes biases my also occur in non‐industry‐sponsored studies. All studies included in this review were sponsored by the maker of golimumab.
The baseline characteristics were balanced between groups within each of the four studies included in this review (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009).
Effects of interventions
See: Table 1
Details are presented in the Data and analyses section.
Golimumab 50 mg every four weeks + Methotrexate versus placebo + Methotrexate
The details of these outcomes are in the Data and analyses section, first table. Outcomes presented below were after treatment with golimumab for 14‐24 weeks.
Primary Outcomes:
ACR50: Four studies (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009) provided data. Golimumab treated patients were 2.6 times more likely than placebo to reach ACR50 (95% confidence interval: 1.34 to 4.94), at 14‐24 weeks.
Safety: Safety as assessed by the
-
Number and type of adverse effects (AEs) and serious adverse events (SAEs): All four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009). Golimumab treated patients were 1.1 times more likely (95% confidence interval: 0.93, 1.18) to experience adverse events than placebo treated patients, but this was not statistically significant. There was no significant difference in the number of serious adverse events occurring for golimumab treated patients compared to placebo treated patients, with a relative risk of 1.1 (95% confidence interval: 0.62 to 1.78; P=0.85).
Infections: All four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009).There was no statistically significant difference between the number of infections between the golimumab and placebo groups (P=0.8).
Serious infections: All four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009). There was no statistically significant difference between the number of serious infections between the golimumab and placebo groups (P=0.9).
Tuberculosis: All four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009). There was no statistically significant difference between the number of tuberculosis cases in the golimumab and placebo groups (P=0.5).
Lung Infections: Three studies provided data (Emery 2009, Kay 2008, Smolen 2009). There was no statistically significant difference between the number of lung infections in the golimumab and placebo groups (P=0.9).
Cancer: All four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009). There was no statistically significant difference between the golimumab and placebo groups (P=0.8).
-
Withdrawls: (i) Total, (ii) due to lack of efficacy, (iii) due to adverse events:
Total: All four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009). Patients treated with golimumab were 0.5 times less likely to withdraw compared to placebo (95% confidence interval: 0.31 to 0.81; P=0.005).
Due to lack of efficacy: Three studies provided data (Kay 2008; Keystone 2009; Smolen 2009). There was no statistically significant difference between golimumab and placebo treated patients (P=0.1).
Due to adverse events: There was no statistically significant difference between the number of patients withdrawing due to adverse events in the placebo and treatment groups (P=0.2).
Death: All four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009). Placebo and treatment groups did not statistically significantly differ in number of deaths (P=0.99).
-
Secondary Outcomes:
ACR20, ACR70: Four studies (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009) provided data on ACR20. Golimumab treated patients were 1.5 times more likely to reach ACR20. Four studies (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009) provided data on ACR70. Golimumab treated patients were 2.8 times more likely to reach ACR70.
-
Continuous:
Change in DAS28: One study provided data (Kay 2008). Golimumab treated patients had a significantly greater change in DAS28 scores compared to placebo (P=0.0003).
-
Proportion achieving a good state:
Good EULAR response: Four studies (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009) provided data. Golimumab treated patients were 1.5 times more likely to achieve good EULAR response. We used Good EULAR response based on ESR, where data were presented for both ESR‐ and CRP‐based EULAR responses.
DAS low disease activity: Two studies provided data (Smolen 2009, Kay 2008). Golimumab treated patients were 1.6 times more likely to achieve low disease activity.
DAS remission: Four studies (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009) provided data. Golimumab treated patients were 5.1 times more likely to reach DAS remission.
-
Quality of life:
HAQ scores: One study provided data (Smolen 2009). Golimumab treated patients had statistically significantly lower (better) scores (P < 0.00001).
Change in HAQ scores: One study provided data (Keystone 2009). Golimumab treated patients had statistically significantly more decrease in HAQ scores (P < 0.00001).
HAQ change ≥ 0.22: One study ( Keystone 2009) provided data. Golimumab treated patients were 1.8 times more likely to reach a HAQ change of greater than 0.22.
Radiographic Progression: No studies provided data on this outcome.
Golimumab 100 mg every four weeks + methotrexate versus placebo + methotrexate
The details of these outcomes are in the Data and analyses section, second table. Outcomes presented below were after treatment with golimumab for 14‐24 weeks.
Primary Outcomes:
ACR50: Four studies (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009) provided data. Golimumab treated patients were 2.4 times more likely to reach ACR50 compared to placebo (95% confidence interval: 1.25 to 4.74; P=0.009).
Safety: Safety as assessed by the
-
Number and type of adverse effects (AEs) and serious adverse events (SAEs): All four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009). There was no significant difference between the number of adverse events and serious adverse events occurring for golimumab treated patients compared to placebo treated patients with (P=0.14) and (P=0.9) respectively.
Infections: All four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009).There was no statistically significant difference between the number of infections between the golimumab and placebo groups (P=0.7).
Serious infections: All four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009). There was no statistically significant difference between the number of serious infections between the golimumab and placebo groups (P=0.3).
Tuberculosis: All four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009). There were no patients experiencing tuberculosis in either treatment or placebo groups.
Lung Infections: Two studies provided data (Emery 2009, Smolen 2009). There was no statistically significant difference between the number of lung infections between the golimumab and placebo groups (P=0.1).
Cancer: All four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009). There was no statistically significant difference between the golimumab and placebo groups (P=0.7).
-
Withdrawls: (i) Total, (ii) due to lack of efficacy, (iii) due to adverse events:
Total: All four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009). Patients treated with golimumab were 0.7 times less likely to withdraw compared to placebo.
Due to lack of efficacy: Three studies provided data (Kay 2008; Keystone 2009; Smolen 2009). There was no statistically significant difference between the number of patients withdrawing due to inefficacy in the placebo and treatment groups (P=0.41).
Due to adverse events: Three studies provided data (Kay 2008, Keystone 2009, Smolen 2009). There was no statistically significant difference between the number of patients withdrawing due to adverse events in the placebo and treatment groups (P=0.24).
Death: Four studies provided data (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009). Placebo and treatment groups did not statistically significantly differ in number of deaths (P=0.99).
-
Secondary Outcomes:
ACR20: Four studies (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009) provided data. Golimumab treated patients were 1.6 times more likely to reach ACR20. ACR70: Four studies (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009) provided data. There was no statistically significant difference between golimumab and placebo groups (P=0.11).
-
Continuous
Change in DAS28 scores: One study provided data (Kay 2008). Golimumab treated patients had statistically significantly more decrease in DAS28 scores as compared to placebo (P=0.001).
-
Proportion achieving a good state:
Good EULAR response: Four studies (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009) provided data. Golimumab treated patients were 1.6 times more likely to achieve a good EULAR response.
DAS low disease activity: Two studies provided data (Smolen 2009, Kay 2008). There was no statistically significant difference between the golimumab and the placebo groups (P=0.3).
DAS remission: Four studies (Emery 2009, Kay 2008, Keystone 2009, Smolen 2009) provided data. Golimumab treated patients were 6.3 times more likely to reach DAS remission.
-
Quality of life:
HAQ scores: One study provided data (Smolen 2009). Golimumab treated patients had statistically significantly lower (better) scores (P < 0.00001).
HAQ change ≥ 0.22: One study ( Keystone 2009) provided data. Golimumab treated patients were 1.9 times more likely to reach a HAQ change of at least 0.22.
Change in HAQ scores: One study provided data (Keystone 2009). Golimumab treated patients had statistically significantly more decrease in HAQ scores (P < 0.00001).
Radiographic Progression: No studies provided data on this outcome.
Golimumab 50 mg every two weeks + methotrexate versus placebo + methotrexate
Only Kay 2008 provided data for all outcomes in this comparison group. The details of these outcomes are in the Data and analyses section, third table. Outcomes presented below were after treatment with golimumab for 16 weeks.
Primary Outcomes:
ACR50: Golimumab and placebo groups did not differ statistically significantly from each other (P=0.06).
Safety: Safety as assessed by the
-
Number and type of adverse effects (AEs) and serious adverse events (SAEs):There was no significant difference between the number of adverse events and serious adverse events occurring for golimumab treated patients compared to placebo treated patients with (P=0.3) and (P=0.6) respectively.
Infections: There was no statistically significant difference between the number of infections between the golimumab and placebo groups (P=0.1).
Serious infections: There was no statistically significant difference between the number of serious infections between the golimumab and placebo groups (P=0.97).
Tuberculosis: There were no patients experiencing tuberculosis in either treatment or placebo groups.
Lung Infections: No study provided data on this outcome.
Cancer: There was no statistically significant difference between the golimumab and placebo groups (P=0.5).
-
Withdrawls: (i) Total, (ii) due to lack of efficacy, (iii) due to adverse events:
Total: There was no statistically significant difference between the golimumab and placebo groups (P=0.96).
Due to lack of efficacy: There was no statistically significant difference between the number of patients withdrawing due to inefficacy in the placebo and treatment groups (P=0.3).
Due to adverse events: There was no statistically significant difference between the number of patients withdrawing due to adverse events in the placebo and treatment groups (P=0.97).
Death: No patient deaths were observed in either treatment or placebo arms.
-
Secondary Outcomes:
ACR20; ACR70‐ ACR20: There was no statistically significant difference between the golimumab and placebo groups (P=0.3). ACR70: There was no statistically significant difference between the golimumab and placebo groups (P=0.1).
-
Continuous:
Change in DAS28 scores: One study provided data (Kay 2008). Golimumab treated patients statistically significantly experienced more decrease in DAS28 scores (P=0.005).
-
Proportion achieving a good state:
Good EULAR response: There was no statistically significant difference between the golimumab and placebo groups (P=0.24).
DAS low disease activity: There was no statistically significant difference between the golimumab and the placebo groups (P=0.3).
DAS remission: There was no statistically significant difference between the golimumab and the placebo groups (P=0.13).
-
Quality of Life:
HAQ scores: No data was provided on this outcome.
Change in HAQ scores: No data was provided on this outcome.
HAQ change ≥ 0.22: No data was provided on this outcome.
Radiographic Progression: No studies reported on this outcome.
Golimumab 100 mg every two weeks + methotrexate versus placebo + methotrexate
Only Kay 2008 provided data for all outcomes in this comparison group. The details of these outcomes are in the Data and analyses section, forth table. Outcomes presented below were after treatment with golimumab for 16 weeks.
Primary Outcomes:
ACR50: Golimumab treated patients were 5.7 times more likely to reach ACR50 than placebo (95% confidence interval: 1.35, 23.7; P=0.02 ).
Safety: Safety as assessed by the
-
Number and type of adverse effects (AEs) and serious adverse events (SAEs):There was no significant difference between the number of adverse events and serious adverse events occurring for golimumab treated patients compared to placebo treated patients with (P=0.7) for both outcomes.
Infections: There was no statistically significant difference between the number of infections between the golimumab and placebo groups (P=0.3).
Serious infections: There was no statistically significant difference between the number of serious infections between the golimumab and placebo groups (P=0.5).
Tuberculosis: There were no patients experiencing tuberculosis in either treatment or placebo groups.
Lung Infections: There was no data provided on this outcome.
Cancer: There were no patients experiencing cancer in either treatment or placebo groups.
-
Withdrawls: (i) Total, (ii) due to lack of efficacy, (iii) due to adverse events:
Total: There was no statistically significant difference between placebo and treatment groups (P=0.2).
Due to lack of efficacy: There was no statistically significant difference between the number of patients withdrawing due to inefficacy in the placebo and treatment groups (P=0.3).
Due to adverse events: There was no statistically significant difference between the number of patients withdrawing due to adverse events in the placebo and treatment groups (P=0.3).
Death: No patient deaths were observed.
-
Secondary Outcomes:
ACR20; ACR70‐ ACR20: Patients in the treatment group were 2.1 times more likely to reach ACR20 than those in placebo group. ACR70: There was no statistically significant difference between the golimumab and placebo groups (P=0.2).
-
Continuous:
Change in DAS28 scores: One study provided data. Golimumab treated patients had statistically significantly more decrease in DAS28 scores as compared to placebo (P < 0.00001).
-
Proportion Achieving a Good State:
Good EULAR response: There was no statistically significant difference between golimumab and placebo treated patients (P=0.06).
DAS low disease activity: Patients in the golimumab group were 1.7 times more likely to reach DAS low disease activity (P=0.05).
DAS remission: There was no statistically significant difference between golimumab treated and placebo treated patients (P=0.13).
-
Quality of Life:
HAQ scores: No data was provided on this outcome.
Change in HAQ scores: No data was provided on this outcome.
HAQ change ≥ 0.22: No data was provided on this outcome.
Radiographic Progression: No studies reported on this outcome.
Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate
The details of these outcomes are in the Data and analyses section, fifth table. Outcomes presented below were after treatment with golimumab for 14‐24 weeks.
Primary Outcomes:
ACR50: Two studies (Emery 2009, Keystone 2009) provided data. There was no statistically significant difference between control and golimumab groups (P=0.24).
Safety: Safety as assessed by the
-
Number and type of adverse effects (AEs) and serious adverse events (SAEs): Two studies (Emery 2009, Keystone 2009) provided data. There was no significant difference between the number of adverse events and serious adverse events occuring for golimumab treated patients compared to control with (P=0.7) and (P=0.7) respectively.
Infections: Two studies (Emery 2009, Keystone 2009) provided data. There was no statistically significant difference between the number of infections between the golimumab and control groups (P=0.3).
Serious infections: Two studies (Emery 2009, Keystone 2009) provided data. There was no statistically significant difference between the number of serious infections between the golimumab and control groups (P=0.7).
Tuberculosis: Two studies (Emery 2009, Keystone 2009) provided data. There were no patients experiencing tuberculosis in either treatment or control groups.
Lung Infections: Only one study provided data (Emery 2009). There was no statistically significant difference between the number of lung infections between the golimumab and control groups (P=0.3).
Cancer: Two studies (Emery 2009, Keystone 2009) provided data. There was no statistically significant difference between the golimumab and control groups (P=0.82).
-
Withdrawls: (i) Total, (ii) due to lack of efficacy, (iii) due to adverse events:
Total: Two studies (Emery 2009, Keystone 2009) provided data. There was no statistically significant difference between the number of total withdrawals in the control and golimumab groups (P=0.5).
Due to lack of efficacy: One study (Keystone 2009) provided data. There were no patients withdrawing due to inefficacy.
Due to adverse events: One study (Keystone 2009) provided data. There was no statistically significant difference between the number of patients withdrawing due to adverse events in the control and golimumab groups (P=0.4).
Death: Two studies (Emery 2009, Keystone 2009) provided data. There was no statistically significant difference between the number of deaths (P=0.5).
-
Secondary Outcomes:
ACR20: Two studies (Emery 2009, Keystone 2009) provided data. There was no statistically significant difference between golimumab and control group (P=0.2). ACR70: Two studies (Emery 2009, Keystone 2009) provided data. There was no statistically significant difference between golimumab and control groups (P=0.7).
-
Continuous:
Change in DAS28 scores: No studies provided data on this outcome
-
Proportion achieving a good state:
Good EULAR response: Two studies (Emery 2009, Keystone 2009) provided data. There was no statistically significant difference between golimumab and control groups (P=0.1).
DAS low disease activity: No studies provided data on this outcome.
DAS remission: Two studies (Emery 2009, Keystone 2009) provided data. There was no statistically significant difference between golimumab and control groups (P=0.2).
-
Quality of Life:
HAQ scores: No studies provided data on this outcome.
HAQ change ≥ 0.22: One study (Keystone 2009) provided data. There was no statistically significant difference between golimumab and control groups (P=0.3).
Change in HAQ scores: One study (Keystone 2009) provided data. There was a a statistically significant decrease in golimumab treated patients compared to placebo (P < 0.00001).
Radiographic Progression: No studies reported on this outcome.
Subgroup Analyses
We had planned the following sub‐group analyses to explore possible effect size differences, if sufficient data were available. Due to lack of more than one study per subgroup for most subgroup comparisons, we were able to perform only the first comparison, i.e., efficacy with and without concomitant methotrexate. For subgroup comparisons 2 to 6 listed below, only zero or one study were available for one group and therefore no formal comparisons were performed.
Concomitant methotrexate versus no methotrexate: Since none of the studies had randomized groups of patients taking or not taking methotrexate at baseline, we were unable to perform this analysis exactly as planned as a direct comparison. Therefore, we analyzed these groups separately.
No Concomitant methotrexate : In two studies (Emery 2009, Keystone 2009), one study arm had golimumab + oral placebo and another, injectable placebo + methotrexate. We analyzed the first study arm to assess efficacy of golimumab without concomitant methotrexate. This comparison has methotrexate as the comparator, so this also provides estimates of efficacy/safety of golimumab vs. methotrexate (Comparison 5).
Concomitant methotrexate : Since all 4 included studies had one or more arm with background methotrexate, we combined all doses of golimumab with concomitant methotrexate to obtain estimates for efficacy and safety of golimumab when used with concomitant methotrexate (Comparison 6). We found that golimumab was statistically significantly more efficacious than control in all efficacy outcomes. For safety outcomes, we found that there was no statistically significant difference between golimumab and placebo, except in withdrawals where there was statistically significantly more total withdrawals in the placebo arm, because of statistically significantly more withdrawals due to inefficacy in the placebo group.
2. Mean RA disease duration (early RA versus established RA vs. late RA): All studies enrolled patients with mean disease duration between 2 and10 years (established RA), therefore this subgroup analysis could not be performed.
3. Use in patients who have methotrexate‐failure versus biologic‐failure: Only one study enrolled patients who had failed biologics (Smolen 2009). A detailed comparison was not done since there was only one study in patients with biologic‐failure.
4. Single biologic DAMRD agent versus combination biologic therapy: None of the studies included combination biologic therapy, therefore this comparison could not be done.
5. DMARD‐naive versus not naive: Only one study (Emery 2009) recruited methotrexate naive patients, therefore this comparison was not done.
6. Treatment duration with biologic DMARD (Short (6 months), intermediate duration (6 to 12 months) or long‐duration (>1 year): All studies were short‐term (before the rescue phase), therefore this comparison could not be done.
FDA web site search for Warnings and adverse events
We searched the FDA web site for warnings and adverse events related to golimumab use on 09/01/2009.
The FDA web site summary review for golimumab states that "the results from five Phase 3 clinical trials in support of the efficacy of Simponi [golimumab] for RA, PA, and AS. These studies clearly establish the efficacy of the 50‐mg dose for RA..." citing three trials for golimumab for RA. The trials are Emery 2009 ("T05"), Keystone 2009 ("T06") and Smolen 2009 ("T11"). According to the FDA, "studies T06 and T11 provided clear evidence of efficacy for Simponi on the primary endpoints and the secondary endpoints were supportive as well. In regard to the absence of a statistically significant treatment effect in Study T05, Dr. Okada notes the following on page 16 of her review: '... previous experience with MTX and other TNF inhibitors are not superior to optimised MTX in this population, so the results of the Study T05 are not unexpected." Furthermore, the FDA notes " The results of the three trials... do not demonstrate additional benefit of a 100‐mg dose of Simponi compared to a 50‐mg dose, and the sponsor is not seeking approval of the higher dose" (FDA 2009b).
The golimumab label warns against the risk of serious infection, stating "serious infections leading to hospitalizations or death including tuberculosis (TB), bacterial sepsis, invasive fungal, and other opportunistic infection have occurred in patients receiving SIMPONI." It further warns of the possibility of Hepatitis B reactivation, the increased risk of malignancies including lymphoma, stating "the incidence of lymphoma was seen more often than in the general U.S. population. Cases of other malignancies have been observed among patients with TNF‐blockers." The FDA web site further states that those with serious infection should not start SIMPONI (the trade name for golimumab). A careful monitoring and discontinuation is advised if a patient develops infection or serious infection. It also warns that the "worsening, or new onset" of heart failure may occur. Furthermore, it states that "demyelinating disease, exacerbation or new onset, may occur." Upper respiratory tract infections, nasopharyngitis are noted as the most common adverse reactions with an incidence rate of more than 5% (FDA 2009a). A letter also found on the FDA web site writes "the safety and efficacy data did not pose unique concerns beyond those applicable to other biologic products in the TNF‐blocker class approved for treatment of rheumatoid arthritis..." (FDA 2009b). The FDA also warns that the simultaneous administration of abatacept, anakinra and live vaccines in conjunction to golimumab lead to an increased risk of serious infection and in the case of live vaccines, should be avoided (FDA 2009a). In a safety alert dated on August 31, 2009, the FDA writes "there is an increased risk of lymphoma and other cancers associated with the use of these drugs in children and adolescents. The FDA has also identified new safety information related to the occurrence of leukemia and new onset psoriasis in patients treated with TNF‐blockers" (FDA 2009c).
Assessment of Heterogeneity
For the primary outcome of ACR50, there was statistically significant heterogeneity in the golimumab 50 mg and 100 mg every four weeks plus methotrexate verus placebo plus methotrexate groups with I2 values of 76% and 77% (P values of 0.005 for each) respectively, denoting a considerable amount of heterogeneity as per the Cochrane review handbook recommendations. This heterogeneity was primarily due to Emery 2009, removing this study decreased the I2 values to 0% and 0%. However, since the direction of these estimates was same across the studies, we determined that it was appropriate to pool these results.
There was only one study that presented data in comparison groups where golimumab was administered every two weeks at doses of 50 mg and 100 mg, and thus no calculations of heterogeneity were necessary. In the comparison group where 100 mg of golimumab was indirectly compared to methotrexate, there was statistically significant heterogeneity with an I2 value of 68% (P value of 0.08) denoting substantial heterogeneity. Lastly, in the sensitivity analysis of all combined doses of golimumab versus placebo plus methotrexate, there was statistically significant heterogeneity with an I2 value of 80% (P value of 0.002) denoting a considerable amount of heterogeneity. This heterogeneity seemed secondary to a very lower risk ratio for Emery 2009.
For the primary outcome of safety, in the golimumab 50 mg every four weeks group, there was no statistically significant heterogeneity except in total adverse events with an I2 of 55% and P value of 0.09, denoting a moderate to substantial amount of heterogeneity. In the golimumab 100 mg every four weeks plus methotrexate versus placebo plus methotrexate group, there was no statistically significant heterogeneity in the safety outcomes except in the outcome of serious adverse events, where a P value of 0.06 and a I2 of 59% indicated substantial heterogeneity. In the comparison group where golimumab was indirectly compared to methotrexate, we found no substantial heterogeneity for any of the safety outcomes. Lastly, in the comparison group where all doses of golimumab were compared to placebo, there was no statistically significant heterogeneity in safety outcomes.
Discussion
Summary of main results
In this review of RCTs of golimumab for treatment of active RA, four RCTs were included with 1,231 patients treated with golimumab. We found that when used with methotrexate for treatment of the RA symptoms, golimumab treatment at FDA‐approved dose of 50 mg every 4 weeks is associated with significantly better efficacy than placebo for achieving ACR20/50/70 and lower DAS28 scores, at 5 to 6 month follow‐up. Remission of RA was achieved by more patients in the golimumab than placebo group. Improvement in physical function as measured by HAQ were significantly more impressive in the golimumab versus placebo group. At the FDA‐approved dose of 50 mg every 4 weeks, golimumab was not associated with any higher risk of total adverse events. In addition, the risk of serious adverse events was similar to placebo. Withdrawals due to any reason and due to inefficacy were significantly lower in golimumab versus placebo group. This may indicate the better efficacy of golimumab compared to placebo, leading to less withdrawals due to inefficacy and less overall withdrawals.
With the caveat that none of the studies were designed with safety as primary outcome, golimumab (50mg every 4‐weeks) was not associated with higher risk of infections, serious infections, tuberculosis, lung infections, cancer or death. Withdrawals due to adverse events did not differ between groups. Similar efficacy and safety were noted for other dosing regimens of golimumab with a few exceptions, where differences that were significant for the FDA‐approved doses were no longer significant for these dosing regimens. It must be noted that the sample sizes for non‐FDA approved doses was much smaller than that for the FDA‐approved dose and therefore liable to type II error, i.e., missing an effect due to lack of power. Similar concern must be kept in mind regarding the safety data, since none of the RCTs had safety as the primary outcome and all RCTs were of short duration. Therefore, adverse events that are uncommon and/or occur with long‐term treatment with golimumab are unknown at this time. Long‐term surveillance studies and RCTs with safety as primary outcome are needed to provide these important data. Another important limitation of evidence is that radiographic data have not yet been reported from these RCTs, an important piece of information for the physicians and patients using this medication.
Many findings in this review deserve further discussion. Our observation of significant impact of golimumab in FDA‐approved dose on both lowering RA disease activity (DAS28) as well as improving RA disease activity (ACR 20/50/70) implies that evidence now exists that golimumab is an effective treatment when used in conjunction with methotrexate in patients with active RA. The ACR20/50/70 rates in golimumab group (with methotrexate) in FDA‐approved dose were 1.5‐2.8 times more likely than the placebo + methotrexate group. The ACR50 rates are similar to those reported in the systematic reviews of other TNF‐blockers including etanercept (Lethaby 2003), infliximab (Blumenauer 2002) and Adalimumab (Navarro‐Sarabia 2005) and to other current approved biologics for the treatment of RA, such as rituximab (Lopez‐Olivo 2008) and abatacept (Maxwell 2008). Thus, in absence of direct comparison studies, based on these data, golimumab seems to have an efficacy similar to the other biologics currently used for the treatment of RA.
Secondly, the improvements in functional limitation as measured by HAQ were significantly greater in golimumab‐treated versus placebo‐treated patients. This is important since one of the goals with effective therapy is not only to reduce inflammation and prevent radiographic destruction, but to improve function and quality of life of the patients. Improvement in HAQ scores in golimumab‐treated patients ranged from a mean difference between golimumab and placebo groups of 0.20 to 0.25 points on a 0 to 3 scale. As well, almost twice as many golimumab‐treated patients achieved the HAQ MCID, compared to the placebo‐treated patients. Thus, at the approved‐dose, golimumab seems to provide clinically meaningful improvements in function in patients with RA. Quality of life data have not been reported in these studies. Typically, those publications lag behind the original publications by a few months, so we hope those data may become available after this review is published.
Thirdly, the short‐term safety for golimumab at the FDA‐approved dose of 50mg every 4 weeks used in combination with methotrexate seems acceptable and similar to other TNF‐inhibitors. Based on the four RCTs included in this review, golimumab was associated with no higher risk of infections, serious infections, lung infections, tuberculosis, cancer or death than placebo. All these RCTs were short‐duration studies powered for efficacy outcomes. Long‐term surveillance studies are needed to assure patients and physicians to its safety and to make them aware of safety risks of golimumab. The FDA web site warns against the risk of serious infections, invasive fungal infections, hepatitis B reactivation, malignancies, heart failure, demyelinating disease and new‐onset psoriasis. In a recently issued safety alert, the FDA also warns against the "increased risk of lymphoma and other malignancies in children and adolescents treated with TNF‐blockers."
The odds ratio (95% CI) for withdrawals due to adverse events in golimumab‐treated RA patients was 0.80 95% CI (0.26, 2.42), compared to placebo. This is similar to the range reported for withdrawals due to adverse events reported other biologics in the range of odds ratios of 0.82 to 2.2 (Lethaby 2003; Blumenauer 2002; Navarro‐Sarabia 2005; Lopez‐Olivo 2008; Maxwell 2008).
In an analysis comparing golimumab to methotrexate, only two studies (Emery 2009, Keystone 2009) provided the data. We found that golimumab treated patients achieved a significantly lower HAQ score compared to methotrexate‐treated patients. However, the rates of ACR20/50/70, DAS remission, good EULAR response and proportion achieving HAD MCID were no different between these groups. The safety profile of golimumab was similar to that of methotrexate with regards to number of adverse events, serious adverse events, infections, serious infections and cancer. Withdrawals rates overall and due to adverse events were similar. However, these results must be interpreted with great caution due to many reasons. This was a subgroup analysis and therefore, the findings are at best, hypothesis generating. Only two RCTs provide the data, so there is possibility of type II errors, i.e., missing differences when they might exist, due to a small sample size. Additionally, in one study (Keystone 2009) patients had already failed methotrexate before being randomized to methotrexate or golimumab, thereby providing an efficacy advantage to the golimumab group. Emery 2009 enrolled Methotrexate‐naive patients and therefore is less likely to be biased.
Overall completeness and applicability of evidence
We have included all RCTs of golimumab in patients with RA. Thus, this evidence report is current and up to‐date. This evidence synthesis is relevant and applicable, since it combines data across four randomized trials.
Quality of the evidence
The quality of evidence found in the trials included in this review appear to be high because the studies reported adequate methods of allocation concealment, sequence generation using the interactive voice response system and adequate methods of blinding. Only one study lacked in addressing incomplete outcome data adequately (Keystone 2009). The only source of concern for quality of evidence may be that we did not have access to enough information such as complete study protocols to make definitive judgments regarding the risk of selective reporting.
Despite the presence of heterogeneity in ACR50 estimates was carefully noted. However, we doubt that more studies are unlikely to change the overall estimates.
Potential biases in the review process
All titles and abstracts resulting from the electronic search were assessed independently by two review authors. All data were also extracted independently by two review authors to avoid typing errors. We are limited in our ability to comment on outcomes not reported in the published reports, such as radiographic data or summary scores from quality of life instruments such as Short‐Form 36. Typically, these studies are published after the main trial results have been published. We were also limited in analysis of safety data, since we decided to combine the numbers from studies that described number of patients with more than one adverse events with studies that described the total number of adverse events. We made this decision to make the data and results easy to interpret. Furthermore, there may be unpublished studies and data that FDA had access to with regards to safety, resulting in their recently issued safety alert regarding the an increased risk of malignancies and psoriasis with golimumab, to which we had no access. Many risks associated with TNF blockers as a class are also applicable to golimumab.
Agreements and disagreements with other studies or reviews
Oldfield 2009 was a review summarizing the results of trials assessing the efficacy of golimumab for rheumatoid arthritis, ankylosing spondylitis, and psoriatic arthritis without performing a meta analysis. The study summarized the results of the GO‐BEFORE (Emery 2009), GO‐FORWARD (Keystone 2009), and GO‐AFTER (Smolen 2009) studies. Three additional reviews and editorials provided summary data without performing any meta‐analysis (Yazici 2009, Pappas 2009, Scott 2009). We could not make any comparisons since no meta‐analysis were performed by the previously published reviews.
Authors' conclusions
Implications for practice.
The favorable safety and efficacy profile of golimumab used in combination with methotrexate in patients who have failed traditional DMARDs, TNF‐inhibitors or were DMARD‐naive, implies that this is a new option available to RA patients with active disease despite using currently available treatments. As a subcutaneously injected anti‐TNF therapy once a month, it adds to two currently available subcutaneous anti‐TNF biologics i.e. etanercept (twice a week or once a week) and adalimumab (every two weeks). Golimumab provides an option to patients desiring less frequent injections and/or to those that have failed to achieve a good control of their RA with methotrexate or with other anti‐TNF biologics.
Implications for research.
Direct head‐to‐head comparison studies are needed between golimumab and other biologics, including anti‐TNF biologics. Studies comparing methotrexate and golimumab to triple therapy (methotrexate, sulfasalazine and hydroxychloroquine) are needed to compare their safety and efficacy profiles. Longer‐term safety data are needed from RCTs with safety as primary outcome and from phase‐IV post‐marketing surveillance studies.
What's new
| Date | Event | Description |
|---|---|---|
| 11 November 2009 | Amended | CMSG ID: C197‐R |
Notes
Although not published, a proper protocol was done for this review and it went through the Musculoskeletal Group's editorial process. However, the review was completed in a short period of time and also underwent peer review in time for the next submission deadline so the editorial team decided that it was appropriate to publish just the review.
Acknowledgements
We thank Louise Falzon for performing the electronic search, Tamara Rader for helping with the plain language summary and Lara Maxwell for providing editorial comments.
Appendices
Appendix 1. Electronic Searches
Search 1
Ovid MEDLINE(R) In‐Process & Other Non‐Indexed Citations, Ovid MEDLINE(R) Daily and Ovid MEDLINE(R) 1950 to June Week 3, 2009
1. golimum$.af.
2. cnto‐148.af.
3. simoni.tw
4. or/1‐ 3
EMBASE 1980 to 2009 Week 26
1. Golimumab/
2. golimum$.tw
3. cnto‐148.af.
4. simoni.tw
5. or/1‐ 4
The Cochrane Library, Issue 2, 2009
CENTRAL:
#1 golimum*
#2 cnto‐148
#3 cnto next 148
#4 simponi:ti,ab
#5 (#1 OR #2 OR #3 OR #4)
CINAHL
S1 golimum* or cnto‐148 or cnto 148 or simponi
Web of Knowledge
(golimum* or cnto‐148 or cnto 148 or simponi) in Topic
Dissertation Abstracts
(golimum* or cnto‐148 or cnto 148 or simponi) in Citation and abstract
Current Controlled Trials
golimum* or cnto‐148 or cnto 148 or simponi
Data and analyses
Comparison 1. Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 ACR20 (14‐24 wk) | 4 | 919 | Risk Ratio (M‐H, Random, 95% CI) | 1.53 [1.23, 1.91] |
| 2 ACR50 (14‐24 wk) | 4 | 919 | Risk Ratio (M‐H, Random, 95% CI) | 2.57 [1.34, 4.94] |
| 3 ACR70 (14‐24 wk) | 4 | 919 | Risk Ratio (M‐H, Random, 95% CI) | 2.80 [1.31, 5.98] |
| 4 Good EULAR Response (14‐24 wk) | 4 | 919 | Risk Ratio (M‐H, Random, 95% CI) | 1.47 [1.15, 1.89] |
| 5 DAS Low Disease Activity (14‐16 wk) | 2 | 378 | Risk Ratio (M‐H, Random, 95% CI) | 1.64 [1.15, 2.34] |
| 6 DAS Remission (14‐24 wk) | 4 | 919 | Risk Difference (M‐H, Random, 95% CI) | 0.10 [0.06, 0.14] |
| 7 HAQ Change ≥.22 (14 wk) | 1 | 222 | Risk Ratio (M‐H, Random, 95% CI) | 1.79 [1.38, 2.31] |
| 8 Change in HAQ score (14 wk) | 1 | 222 | Mean Difference (IV, Fixed, 95% CI) | ‐0.25 [‐0.29, ‐0.21] |
| 9 HAQ Scores (14 wk) | 1 | 308 | Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐0.25, ‐0.15] |
| 10 Change in DAS28 Scores (16 wk) | 1 | 70 | Mean Difference (IV, Fixed, 95% CI) | ‐1.1 [‐1.69, ‐0.51] |
| 11 Adverse Events (16‐24 wk) | 4 | 918 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.93, 1.18] |
| 12 Serious Adverse Events (16‐24 wk) | 4 | 918 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.62, 1.78] |
| 13 Infections (16‐24 wk) | 4 | 918 | Risk Ratio (M‐H, Random, 95% CI) | 1.03 [0.84, 1.25] |
| 14 Serious Infections (16‐24 wk) | 4 | 918 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.40, 2.86] |
| 15 Tuberculosis (16‐24 wk) | 4 | 918 | Risk Ratio (M‐H, Random, 95% CI) | 3.04 [0.12, 74.01] |
| 16 Lung Infections (16‐24 wk) | 2 | 625 | Risk Ratio (M‐H, Random, 95% CI) | 0.97 [0.55, 1.70] |
| 17 Cancer (16‐24 wk) | 4 | 918 | Risk Ratio (M‐H, Random, 95% CI) | 0.81 [0.16, 4.18] |
| 18 All Withdrawals (14‐24 wk) | 4 | 917 | Risk Ratio (M‐H, Random, 95% CI) | 0.50 [0.31, 0.81] |
| 19 Withdrawals due to Adverse Events (14‐16 wk) | 3 | 599 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.24, 1.29] |
| 20 Withdrawals due to Inefficacy (14‐16 wk) | 3 | 599 | Risk Ratio (M‐H, Random, 95% CI) | 0.43 [0.15, 1.21] |
| 21 Death (24‐52 wk) | 4 | 917 | Risk Ratio (M‐H, Random, 95% CI) | 1.02 [0.11, 9.71] |
1.1. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 1 ACR20 (14‐24 wk).
1.2. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 2 ACR50 (14‐24 wk).
1.3. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 3 ACR70 (14‐24 wk).
1.4. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 4 Good EULAR Response (14‐24 wk).
1.5. Analysis.
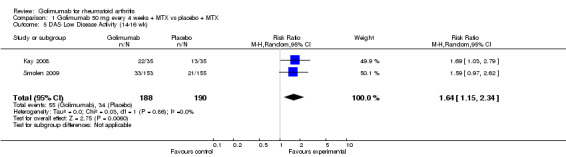
Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 5 DAS Low Disease Activity (14‐16 wk).
1.6. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 6 DAS Remission (14‐24 wk).
1.7. Analysis.
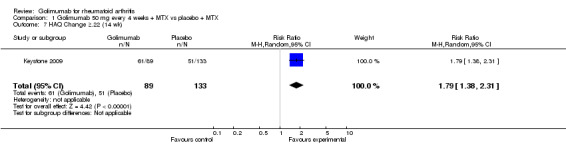
Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 7 HAQ Change ≥.22 (14 wk).
1.8. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 8 Change in HAQ score (14 wk).
1.9. Analysis.
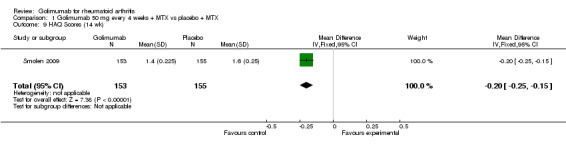
Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 9 HAQ Scores (14 wk).
1.10. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 10 Change in DAS28 Scores (16 wk).
1.11. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 11 Adverse Events (16‐24 wk).
1.12. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 12 Serious Adverse Events (16‐24 wk).
1.13. Analysis.
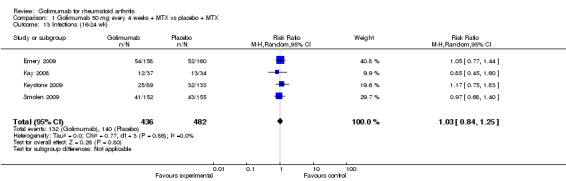
Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 13 Infections (16‐24 wk).
1.14. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 14 Serious Infections (16‐24 wk).
1.15. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 15 Tuberculosis (16‐24 wk).
1.16. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 16 Lung Infections (16‐24 wk).
1.17. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 17 Cancer (16‐24 wk).
1.18. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 18 All Withdrawals (14‐24 wk).
1.19. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 19 Withdrawals due to Adverse Events (14‐16 wk).
1.20. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 20 Withdrawals due to Inefficacy (14‐16 wk).
1.21. Analysis.

Comparison 1 Golimumab 50 mg every 4 weeks + MTX vs placebo + MTX, Outcome 21 Death (24‐52 wk).
Comparison 2. Golimumab 100 mg q4 week + MTX vs placebo + MTX.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 ACR20 (14‐24 wk) | 4 | 918 | Risk Ratio (M‐H, Random, 95% CI) | 1.56 [1.22, 2.01] |
| 2 ACR50 (14‐24 wk) | 4 | 918 | Risk Ratio (M‐H, Random, 95% CI) | 2.43 [1.25, 4.74] |
| 3 ACR70 (14‐24 wk) | 4 | 880 | Risk Ratio (M‐H, Random, 95% CI) | 2.41 [0.81, 7.15] |
| 4 Good EULAR Response (14‐24 wk) | 4 | 918 | Risk Ratio (M‐H, Random, 95% CI) | 1.42 [1.11, 1.82] |
| 5 DAS Low Ds Activity (14‐16 wk) | 2 | 377 | Risk Ratio (M‐H, Random, 95% CI) | 1.61 [0.69, 3.75] |
| 6 DAS Remission (14‐24 wk) | 4 | 918 | Risk Ratio (M‐H, Random, 95% CI) | 6.28 [1.37, 28.78] |
| 7 HAQ Change ≥.22 (14 wk) | 1 | 222 | Risk Ratio (M‐H, Random, 95% CI) | 1.88 [1.46, 2.41] |
| 8 Change in HAQ score (14 wk) | 1 | 222 | Mean Difference (IV, Fixed, 95% CI) | ‐0.25 [‐0.28, ‐0.22] |
| 9 HAQ Scores (14 wk) | 1 | 308 | Mean Difference (IV, Fixed, 95% CI) | ‐0.30 [‐0.36, ‐0.24] |
| 10 Change in DAS28 Scores (16 wk) | 1 | 69 | Mean Difference (IV, Fixed, 95% CI) | ‐1.1 [‐1.78, ‐0.42] |
| 11 Adverse Events (16‐24 wk) | 4 | 915 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.98, 1.15] |
| 12 Serious Adverse Events (16‐24 wk) | 4 | 915 | Risk Ratio (M‐H, Random, 95% CI) | 1.04 [0.40, 2.70] |
| 13 Infections (16‐24 wk) | 4 | 915 | Risk Ratio (M‐H, Random, 95% CI) | 0.96 [0.78, 1.18] |
| 14 Serious Infections (16‐24 wk) | 4 | 915 | Risk Ratio (M‐H, Random, 95% CI) | 1.80 [0.56, 5.80] |
| 15 Tuberculosis (16‐24 wk) | 4 | 915 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 16 Lung Infections (16‐24 wk) | 2 | 626 | Risk Ratio (M‐H, Random, 95% CI) | 1.53 [0.93, 2.54] |
| 17 Cancer (16‐24 wk) | 4 | 915 | Risk Ratio (M‐H, Random, 95% CI) | 1.36 [0.33, 5.56] |
| 18 All Withdrawals (14‐24 wk) | 4 | 917 | Risk Ratio (M‐H, Random, 95% CI) | 0.66 [0.43, 1.02] |
| 19 Withdrawals due to Adverse Events (14‐16 wk) | 3 | 598 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.21, 1.47] |
| 20 Withdrawals due to Inefficacy (14‐16 wk) | 3 | 598 | Risk Ratio (M‐H, Random, 95% CI) | 0.69 [0.29, 1.66] |
| 21 Death (24‐52 wk) | 4 | 917 | Risk Ratio (M‐H, Random, 95% CI) | 1.01 [0.11, 9.68] |
2.1. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 1 ACR20 (14‐24 wk).
2.2. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 2 ACR50 (14‐24 wk).
2.3. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 3 ACR70 (14‐24 wk).
2.4. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 4 Good EULAR Response (14‐24 wk).
2.5. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 5 DAS Low Ds Activity (14‐16 wk).
2.6. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 6 DAS Remission (14‐24 wk).
2.7. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 7 HAQ Change ≥.22 (14 wk).
2.8. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 8 Change in HAQ score (14 wk).
2.9. Analysis.
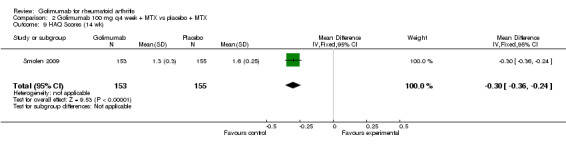
Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 9 HAQ Scores (14 wk).
2.10. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 10 Change in DAS28 Scores (16 wk).
2.11. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 11 Adverse Events (16‐24 wk).
2.12. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 12 Serious Adverse Events (16‐24 wk).
2.13. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 13 Infections (16‐24 wk).
2.14. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 14 Serious Infections (16‐24 wk).
2.15. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 15 Tuberculosis (16‐24 wk).
2.16. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 16 Lung Infections (16‐24 wk).
2.17. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 17 Cancer (16‐24 wk).
2.18. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 18 All Withdrawals (14‐24 wk).
2.19. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 19 Withdrawals due to Adverse Events (14‐16 wk).
2.20. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 20 Withdrawals due to Inefficacy (14‐16 wk).
2.21. Analysis.

Comparison 2 Golimumab 100 mg q4 week + MTX vs placebo + MTX, Outcome 21 Death (24‐52 wk).
Comparison 3. Golimumab 50 mg q2 week + MTX vs placebo + MTX.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 ACR20 (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 1.35 [0.78, 2.33] |
| 2 ACR50 (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 4.12 [0.94, 18.01] |
| 3 ACR70 (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 11.31 [0.65, 197.06] |
| 4 Good EULAR Response (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 2.57 [0.54, 12.38] |
| 5 DAS Low Ds Activity (16 wk) | 1 | 70 | Risk Ratio (M‐H, Random, 95% CI) | 1.31 [0.75, 2.27] |
| 6 DAS Remission (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 9.26 [0.52, 165.65] |
| 7 Change in DAS28 Scores (16 wk) | 1 | 69 | Mean Difference (IV, Fixed, 95% CI) | ‐0.90 [‐1.52, ‐0.28] |
| 8 Adverse Events (20 wk) | 1 | 66 | Risk Ratio (M‐H, Random, 95% CI) | 0.88 [0.69, 1.12] |
| 9 Serious Adverse Events (20 wk) | 1 | 66 | Risk Ratio (M‐H, Random, 95% CI) | 1.59 [0.28, 8.93] |
| 10 Infections (20 wk) | 1 | 66 | Risk Ratio (M‐H, Random, 95% CI) | 0.49 [0.21, 1.13] |
| 11 Serious Infections (20 wk) | 1 | 66 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.07, 16.28] |
| 12 Tuberculosis (20 wk) | 1 | 66 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 13 Cancer (20 wk) | 1 | 66 | Risk Ratio (M‐H, Random, 95% CI) | 3.18 [0.13, 75.38] |
| 14 All Withdrawals (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 1.03 [0.37, 2.88] |
| 15 Withdrawals due to Adverse Events (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 1.03 [0.22, 4.75] |
| 16 Withdrawals due to Inefficacy (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 0.34 [0.04, 3.14] |
| 17 Death (52 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
3.1. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 1 ACR20 (16 wk).
3.2. Analysis.
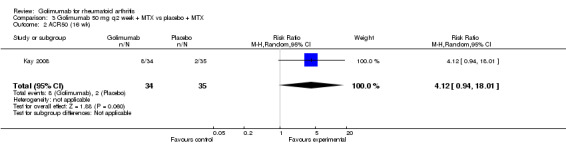
Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 2 ACR50 (16 wk).
3.3. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 3 ACR70 (16 wk).
3.4. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 4 Good EULAR Response (16 wk).
3.5. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 5 DAS Low Ds Activity (16 wk).
3.6. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 6 DAS Remission (16 wk).
3.7. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 7 Change in DAS28 Scores (16 wk).
3.8. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 8 Adverse Events (20 wk).
3.9. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 9 Serious Adverse Events (20 wk).
3.10. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 10 Infections (20 wk).
3.11. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 11 Serious Infections (20 wk).
3.12. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 12 Tuberculosis (20 wk).
3.13. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 13 Cancer (20 wk).
3.14. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 14 All Withdrawals (16 wk).
3.15. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 15 Withdrawals due to Adverse Events (16 wk).
3.16. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 16 Withdrawals due to Inefficacy (16 wk).
3.17. Analysis.

Comparison 3 Golimumab 50 mg q2 week + MTX vs placebo + MTX, Outcome 17 Death (52 wk).
Comparison 4. Golimumab 100 mg q2 week + MTX vs placebo + MTX.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 ACR20 (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 2.14 [1.34, 3.40] |
| 2 ACR50 (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 5.66 [1.35, 23.68] |
| 3 ACR70 (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 7.2 [0.39, 134.36] |
| 4 Good EULAR Response (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 4.12 [0.94, 18.01] |
| 5 DAS Low Ds Activity (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 1.66 [1.00, 2.76] |
| 6 DAS Remission (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 9.26 [0.52, 165.65] |
| 7 Change in DAS28 Scores (16 wk) | 1 | 69 | Mean Difference (IV, Fixed, 95% CI) | ‐1.30 [‐1.84, ‐0.76] |
| 8 Adverse Events (20 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 1.04 [0.86, 1.25] |
| 9 Serious Adverse Events (20 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 1.46 [0.26, 8.19] |
| 10 Infections (20 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.33, 1.36] |
| 11 Serious Infections (20 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 0.32 [0.01, 7.69] |
| 12 Tuberculosis (20 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 13 Cancer (20 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 14 All Withdrawals (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 0.34 [0.07, 1.58] |
| 15 Withdrawals due to Adverse Events (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 0.34 [0.04, 3.14] |
| 16 Withdrawals due to Inefficacy (16 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 0.34 [0.04, 3.14] |
| 17 Death (52 wk) | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
4.1. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 1 ACR20 (16 wk).
4.2. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 2 ACR50 (16 wk).
4.3. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 3 ACR70 (16 wk).
4.4. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 4 Good EULAR Response (16 wk).
4.5. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 5 DAS Low Ds Activity (16 wk).
4.6. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 6 DAS Remission (16 wk).
4.7. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 7 Change in DAS28 Scores (16 wk).
4.8. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 8 Adverse Events (20 wk).
4.9. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 9 Serious Adverse Events (20 wk).
4.10. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 10 Infections (20 wk).
4.11. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 11 Serious Infections (20 wk).
4.12. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 12 Tuberculosis (20 wk).
4.13. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 13 Cancer (20 wk).
4.14. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 14 All Withdrawals (16 wk).
4.15. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 15 Withdrawals due to Adverse Events (16 wk).
4.16. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 16 Withdrawals due to Inefficacy (16 wk).
4.17. Analysis.

Comparison 4 Golimumab 100 mg q2 week + MTX vs placebo + MTX, Outcome 17 Death (52 wk).
Comparison 5. Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 ACR20 (14‐24 wk) | 2 | 585 | Risk Ratio (M‐H, Random, 95% CI) | 1.16 [0.91, 1.47] |
| 2 ACR50 (14‐24 wk) | 2 | 585 | Risk Ratio (M‐H, Random, 95% CI) | 1.44 [0.78, 2.64] |
| 3 ACR70 (14‐24 wk) | 2 | 585 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.55, 2.50] |
| 4 Good EULAR Response (14‐24 wk) | 2 | 585 | Risk Ratio (M‐H, Random, 95% CI) | 1.19 [0.98, 1.44] |
| 5 DAS Remission (14‐24 wk) | 2 | 585 | Risk Ratio (M‐H, Random, 95% CI) | 2.33 [0.62, 8.74] |
| 6 HAQ Change ≥.22 (14 wk) | 1 | 266 | Risk Ratio (M‐H, Random, 95% CI) | 1.18 [0.88, 1.57] |
| 7 Change in HAQ scores (14 wk) | 1 | 266 | Mean Difference (IV, Random, 95% CI) | ‐0.12 [‐0.16, ‐0.08] |
| 8 Adverse Events (16‐24 wk) | 2 | 583 | Risk Ratio (M‐H, Random, 95% CI) | 0.97 [0.87, 1.09] |
| 9 Serious Adverse Events (16‐24 wk) | 2 | 583 | Risk Ratio (M‐H, Random, 95% CI) | 0.80 [0.23, 2.77] |
| 10 Infections (16‐24 wk) | 2 | 583 | Risk Ratio (M‐H, Random, 95% CI) | 1.14 [0.89, 1.45] |
| 11 Serious Infections (16‐24 wk) | 2 | 583 | Risk Ratio (M‐H, Random, 95% CI) | 0.76 [0.17, 3.39] |
| 12 Tuberculosis (16‐24 wk) | 2 | 583 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 13 Lung Infections (24 wk) | 1 | 317 | Risk Ratio (M‐H, Random, 95% CI) | 0.66 [0.29, 1.47] |
| 14 Cancer (16‐24 wk) | 2 | 583 | Risk Ratio (M‐H, Random, 95% CI) | 0.78 [0.08, 7.17] |
| 15 All Withdrawals (14‐24 wk) | 2 | 583 | Risk Ratio (M‐H, Random, 95% CI) | 0.77 [0.37, 1.60] |
| 16 Withdrawals due to Adverse Events (14 wk) | 1 | 266 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.09, 2.68] |
| 17 Withdrawals due to Inefficacy (14 wk) | 1 | 266 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 18 Death (24 wk) | 2 | 583 | Risk Ratio (M‐H, Random, 95% CI) | 3.00 [0.12, 72.98] |
5.1. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 1 ACR20 (14‐24 wk).
5.2. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 2 ACR50 (14‐24 wk).
5.3. Analysis.
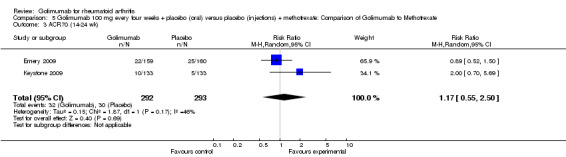
Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 3 ACR70 (14‐24 wk).
5.4. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 4 Good EULAR Response (14‐24 wk).
5.5. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 5 DAS Remission (14‐24 wk).
5.6. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 6 HAQ Change ≥.22 (14 wk).
5.7. Analysis.
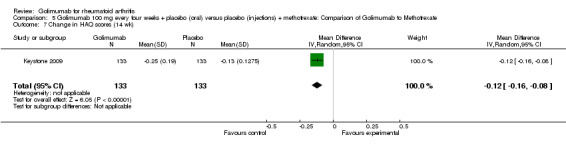
Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 7 Change in HAQ scores (14 wk).
5.8. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 8 Adverse Events (16‐24 wk).
5.9. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 9 Serious Adverse Events (16‐24 wk).
5.10. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 10 Infections (16‐24 wk).
5.11. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 11 Serious Infections (16‐24 wk).
5.12. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 12 Tuberculosis (16‐24 wk).
5.13. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 13 Lung Infections (24 wk).
5.14. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 14 Cancer (16‐24 wk).
5.15. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 15 All Withdrawals (14‐24 wk).
5.16. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 16 Withdrawals due to Adverse Events (14 wk).
5.17. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 17 Withdrawals due to Inefficacy (14 wk).
5.18. Analysis.

Comparison 5 Golimumab 100 mg every four weeks + placebo (oral) versus placebo (injections) + methotrexate: Comparison of Golimumab to Methotrexate, Outcome 18 Death (24 wk).
Comparison 6. Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 ACR20 (14‐24 wk) | 4 | 1422 | Risk Ratio (M‐H, Random, 95% CI) | 1.57 [1.24, 2.00] |
| 2 ACR50 (14‐24 wk) | 4 | 1422 | Risk Ratio (M‐H, Random, 95% CI) | 2.51 [1.30, 4.84] |
| 3 ACR70 (14‐24 wk) | 4 | 1422 | Risk Ratio (M‐H, Random, 95% CI) | 2.66 [1.16, 6.09] |
| 4 Good EULAR Response (14‐24 wk) | 4 | 1422 | Risk Ratio (M‐H, Random, 95% CI) | 1.45 [1.17, 1.81] |
| 5 DAS Low Ds Activity (14 wk) | 2 | 633 | Risk Ratio (M‐H, Random, 95% CI) | 1.71 [1.22, 2.38] |
| 6 DAS Remission (14‐16 wk) | 4 | 1422 | Risk Ratio (M‐H, Random, 95% CI) | 6.00 [1.52, 23.64] |
| 7 HAQ Change ≥.22 (14 wk) | 1 | 311 | Risk Ratio (M‐H, Random, 95% CI) | 1.83 [1.45, 2.32] |
| 8 Change in HAQ score (14 wk) | 1 | 311 | Mean Difference (IV, Fixed, 95% CI) | ‐0.25 [‐0.28, ‐0.22] |
| 9 HAQ Scores (14 wk) | 1 | 461 | Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐0.25, ‐0.15] |
| 10 Change in DAS28 Scores (16 wk) | 1 | 172 | Mean Difference (IV, Fixed, 95% CI) | ‐1.1 [‐1.53, ‐0.67] |
| 11 Adverse Events (16‐24 wk) | 4 | 1418 | Risk Ratio (M‐H, Random, 95% CI) | 1.04 [0.97, 1.12] |
| 12 Serious Adverse Events (16‐24 wk) | 4 | 1418 | Risk Ratio (M‐H, Random, 95% CI) | 1.09 [0.54, 2.18] |
| 13 Infections (16‐24 wk) | 4 | 1418 | Risk Ratio (M‐H, Random, 95% CI) | 0.97 [0.82, 1.16] |
| 14 Serious Infections (16‐24 wk) | 4 | 1418 | Risk Ratio (M‐H, Random, 95% CI) | 1.31 [0.57, 2.97] |
| 15 Tuberculosis (16‐24 wk) | 4 | 1418 | Risk Ratio (M‐H, Random, 95% CI) | 1.52 [0.06, 37.08] |
| 16 Lung Infections (16‐24 wk) | 2 | 936 | Risk Ratio (M‐H, Random, 95% CI) | 1.25 [0.79, 1.99] |
| 17 Cancer (16‐24 wk) | 4 | 1418 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.35, 3.95] |
| 18 All Withdrawals (14‐24 wk) | 4 | 1419 | Risk Ratio (M‐H, Random, 95% CI) | 0.57 [0.40, 0.82] |
| 19 Withdrawals due to Adverse Events (14‐16 wk) | 3 | 942 | Risk Ratio (M‐H, Random, 95% CI) | 0.54 [0.28, 1.07] |
| 20 Withdrawals due to Inefficacy (14‐24 wk) | 3 | 1247 | Risk Ratio (M‐H, Random, 95% CI) | 0.40 [0.16, 0.97] |
| 21 Death (24‐52 wk) | 4 | 1419 | Risk Ratio (M‐H, Random, 95% CI) | 0.69 [0.05, 9.73] |
6.1. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 1 ACR20 (14‐24 wk).
6.2. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 2 ACR50 (14‐24 wk).
6.3. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 3 ACR70 (14‐24 wk).
6.4. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 4 Good EULAR Response (14‐24 wk).
6.5. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 5 DAS Low Ds Activity (14 wk).
6.6. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 6 DAS Remission (14‐16 wk).
6.7. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 7 HAQ Change ≥.22 (14 wk).
6.8. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 8 Change in HAQ score (14 wk).
6.9. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 9 HAQ Scores (14 wk).
6.10. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 10 Change in DAS28 Scores (16 wk).
6.11. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 11 Adverse Events (16‐24 wk).
6.12. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 12 Serious Adverse Events (16‐24 wk).
6.13. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 13 Infections (16‐24 wk).
6.14. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 14 Serious Infections (16‐24 wk).
6.15. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 15 Tuberculosis (16‐24 wk).
6.16. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 16 Lung Infections (16‐24 wk).
6.17. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 17 Cancer (16‐24 wk).
6.18. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 18 All Withdrawals (14‐24 wk).
6.19. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 19 Withdrawals due to Adverse Events (14‐16 wk).
6.20. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 20 Withdrawals due to Inefficacy (14‐24 wk).
6.21. Analysis.

Comparison 6 Golimumab (all doses‐ 50 mg q4wks, 50 mg q2wks, 100 mg q4wks, 100 mg q2wks) + MTX vs placebo (injectable) + MTX, Outcome 21 Death (24‐52 wk).
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Emery 2009.
| Methods | Phase III, 52‐week, randomized, double blind, placebo controlled trial for MTX‐naive patients | |
| Participants |
Injectable Placebo+ MTX: N=160; Biologic naive = Yes; % female= 83.8; age, yrs (mean±SD)=48.6±12.91; disease duration, yrs (mean±SD) = 2.9±4.80; HAQ baseline(mean±SD)= 1.5±0.64; DAS28 baseline (mean±SD) = 6.2±1.17 Golimumab 100mg q4wks + placebo capsules: N=159; Biologic naive = Yes; % female= 84.3; age, yrs (mean±SD)=48.2±12.85; disease duration, yrs (mean±SD) = 4.1±5.60; HAQ baseline(mean±SD)= 1.6±0.72; DAS28 baseline (mean±SD) = 6.3±1.19 Golimumab 50 mg q4wks + MTX: N=159; Biologic naive = Yes; % female= 84.9; age, yrs (mean±SD) =50.9±11.32; disease duration, yrs (mean±SD) = 3.5±5.65; HAQ baseline(mean±SD)= 1.5±0.66; DAS28 baseline(mean±SD)= 6.3±1.11 Golimumab 100 mg q4wks + MTX: N=159; Biologic naive = Yes; % female= 78.6; age, yrs (mean±SD)=50.2±11.87; disease duration, yrs (mean±SD) = 3.6±6.09; HAQ baseline (mean±SD)=1.5±0.64; DAS28 baseline(mean±SD)= 6.3±1.11 |
|
| Interventions | Injectable Placebo+ MTX Golimumab 100mg q4wks + placebo capsules Golimumab 50 mg q4wks + MTX Golimumab 100 mg q4wks + MTX |
|
| Outcomes |
Primary: Difference in the ACR50 response at 24 weeks between groups 3 and 4 versus group 1 and a pair wise comparison (group 3 or group 4 versus group 1) a change in the modified Sharp/van der Heijde score at week 52 Secondary: ACR20 ACR70 ACR90 ACR‐N DAS28 DAS28 UELAR DAS28 Remission HAQ disability index changes in haemoglobin levels in patients with baseline anaemia |
|
| Notes | none | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Quote: "An interactive voice response system (IVRS) was used to randomly assign eligible patients to 1 of 4 treatment groups in approximately equal proportions" |
| Allocation concealment? | Low risk | Quote: "An interactive voice response system (IVRS) was used to randomly assign eligible patients to 1 of 4 treatment groups in approximately equal proportions" |
| Blinding? All outcomes | Low risk | Quote: "Golimumab and placebo were supplied as sterile liquid (aqueous medium of histidine, sorbitol, and polysorbate 80 [pH 5.5] with or without adalimumab) for SC injection. Active and placebo MTX were supplied as double‐blinded, identical opaque capsules (filled with microcrystalline cellulose with or without MTX)" |
| Incomplete outcome data addressed? All outcomes | Low risk | Full account of all withdrawals and reasons for withdrawals ITT analysis performed |
| Free of selective reporting? | Unclear risk | Insufficient information to make a judgment |
| Free of other bias? | Unclear risk | Quote: "All study agents were provided by Centocor
(Malvern, PA)" Comment: A manufacturer of golimumab |
Kay 2008.
| Methods | multicenter, randomized, double‐blind, placebo‐controlled trial, 5‐arm, dose‐ranging study comparing golimumab + methotrexate to placebo + methotrexate (MTX) in 172 patients (40 centers) Double blinded treatment for 20 weeks, and at 20 weeks open‐label treatment commences |
|
| Participants |
Placebo + MTX: N=35; Biologic naive = No; % female= 74.3; age, yrs (median)=52; disease duration, yrs (median) = 5.6; HAQ baseline= 1.3; DAS28 baseline= 5.3 Golimumab 50 mg q4 wks + MTX: N=35; Biologic naive = No; % female= 85.7; age, yrs (median)=57; disease duration, yrs (median) = 8.2; HAQ baseline= 1.7; DAS28 baseline= 5.3 Golimumab 50 mg q2 wks + MTX: N=34; Biologic naive = No; % female= 67.7; age, yrs (median)=48; disease duration, yrs (median) = 8.2; HAQ baseline= 1.6; DAS28 baseline= 4.8 Golimumab 100 mg q4 wks + MTX: N=34; Biologic naive = No; % female= 76.5; age, yrs (median)=57.5; disease duration, yrs (median) = 6.3; HAQ baseline= 1.8; DAS28 baseline= 5.4 Golimumab 100 mg q2 wks + MTX: N=34; Biologic naive = No; % female= 79.4; age, yrs (median)=53.5; disease duration, yrs (median) = 9; HAQ baseline= 1.3; DAS28 baseline= 5.1 |
|
| Interventions | Placebo + MTX Golimumab 50 mg q4 wks + MTX Golimumab 50 mg q2 wks + MTX Golimumab 100 mg q4 wks + MTX Golimumab 100 mg q2 wks + MTX |
|
| Outcomes |
Primary: ACR20% at 16 wks Secondary: ACR50% at wk 16 ACR70% at wk 16 DAS28 improvement from baseline at wk 16 ACR‐N (numeric index of ACR response) at wk 16 ACR20 over time through wk 52 ACR50 over time through wk 52 ACR70 over time through wk 52 |
|
| Notes | none | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Quote: "Patients were randomly assigned to 1 of 5 treatment groups in approximately equal proportions... randomization was stratified by investigational site." Comment: No mention of method of randomization sequence generation; first author contacted who responded: "Computer‐generated randomization was performed at an independent central location, using IVRS." |
| Allocation concealment? | Low risk | Comment: No mention of method of allocation concealment; first author contacted who responded: "Computer‐generated randomization was performed at an independent central location, using IVRS." |
| Blinding? All outcomes | Low risk | Quote: "Study medication was administered by SC injection every 2 weeks from week 0 to week 18. Patients who received golimumab every 4 weeks received placebo injections at the alternate visits to maintain blinding. Patients and investigators remained blinded during the interim analyses." |
| Incomplete outcome data addressed? All outcomes | Low risk | Quote: "For primary analysis, a last observation carried forward procedure was used for patients who did not return for an evaluation or for whom we had insufficient data to determine ACR20 response... pAtients who initiated treatment with oral corticosteroids or disease‐modifying anti‐rheumatic drugs (other than MTX but including biologics), increase MTX or oral corticosteroids dosages above baseline levels, or discontinued the study agent because the lack of efficacy before week 16 were considered to have not achieved the primary end point at week 16." Comment: Adequate. |
| Free of selective reporting? | Unclear risk | Insufficient information to make a judgment |
| Free of other bias? | Unclear risk | Quote: "Supported by Centocor Research and Development, Inc... Dr. Kay has received consulting fees, speaking fees, and/or honoraria (less than $10,000 each) from Abbott, Amgen, Array, BioPharma, Centocor, Novartis, Roche, and Wyeth. Dr. Matteson has Principal Investigator from Centocor. Dr. Dasgupta has received consulting fees, speaking fees, and/or honoraria (less than $10,000 each) from merck, Sharp, and Dohme, Servier, Roche, and Wyeth. Dr. Hsia owns stock options in Johnson & Johnson, and Drs. Han, Wagner, Xu, Visvanathan, and Rahman own stock in Johnson & Johnson, of which Centocor is a subsidiary." Comment: Possible involvement |
Keystone 2009.
| Methods | double‐blind, randomized, placebo controlled, phase III, multicenter, trial in 60 sites (12 countries), 4 arm | |
| Participants |
Placebo injections + oral MTX: N=133; Biologic naive = No; % female= 82; age, yrs (median)=52; disease duration, yrs (median) = 6.5; HAQ baseline= 1.250; DAS28 baseline= 4.86 Go 100 mg q4 wks + oral PL: N=133; Biologic naive = No; % female= 78.9; age, yrs (median)=51; disease duration, yrs (median) = 5.9; HAQ baseline= 1.375; DAS28 baseline= 4.803 Go 50 mg q4 wks + oral MTX: N=89; Biologic naive = No; % female= 80.9; age, yrs (median)=52; disease duration, yrs (median) = 4.5; HAQ baseline= 1.375; DAS28 baseline= 5.1 Go 100 mg q4 wks + oral MTX: N=89; Biologic naive = No; % female= 80.9; age, yrs (median)=52; disease duration, yrs (median) = 6.7; HAQ baseline= 1.375; DAS28 baseline= 4.902 |
|
| Interventions | Placebo injections + oral MTX Go 100 mg q4 wks + oral PL Go 50 mg q4 wks + oral MTX Go 100 mg q4 wks + oral MTX |
|
| Outcomes | ACR20 at 14 and 24 wks ACR50 at wk 14 and 24 ACR70 at wk 14 and 24 DAS28 improvement from baseline at wk 14 and 24 ACR‐N (numeric index of ACR response) at wk 14 and 24 HAQ‐DI scores at wk 14 and 24 Fatigue score (FACIT‐F) DAS28 response according to EULAR DAS28 remission |
|
| Notes | none | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Quote: "Patients who still met the study criteria were randomly assigned in a 3:3:2:2 ratio... Randomization was stratified by investigational site and was conducted using a telephone interactive voice response system." Comment: Sufficient method of randomization |
| Allocation concealment? | Low risk | Quote: "Randomization was conducted using a telephone interactive voice response system." Comment: Sufficient method of allocation concealment |
| Blinding? All outcomes | Low risk | Quote: "Patients in groups 1, 2 or 3 with less than a 20% improvement from baseline in both tender and swollen joint counts had their study medication adjusted in a double‐blind fashion (i.e., early escape)... Patients in group 2 who met the criteria for early escape had their sham methotrexate capsules replaced with active methotrexate capsules at the same stable dose of active methotrexate... Placebo injections contained the same solution as active golimumab but did not contain the monoclonal antibody. Active methotrexate and placebo methotrexate were supplied as identical opaque capsules." Comment: Adequate blinding methods |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Clear account of all withdrawals; no mention of ITT or LOCF |
| Free of selective reporting? | Unclear risk | Insufficient information to make a judgment |
| Free of other bias? | Unclear risk | " Centocor Research and Development, Inc supplied all study medication. This study was funded by Centocor Research and Development, Inc a wholly owned subsidiary of Johnson&Johnson, Inc and Schering Plough Research Institute. Competing interests: declared. ECK, MCG, LK, STG, JP, SCB, and WP (or their institutions) have received research grants from Centocor and/or Schering‐Plough. LK and ECk have received consulting fees from Centocor and/or Schering ‐Plough. ECH, JZ, SV, and MUR are employees of Centocor Research and Development, Inc and own stock and/or stock options in Johnson&Johnson, Inc. MW is an employee of and owns stock in Schering Plough Research Institute, Inc." Comment: Possible sources of bias |
Smolen 2009.
| Methods | 24‐week long, randomized, double blind, placebo controlled trial in 82 sites (10 countries) | |
| Participants |
Placebo + DMARD: N=155; Biologic naive = No; % female= 85; age, yrs (median)=54; disease duration, yrs (median) = 9.8; HAQ baseline= 1.8; DAS28 baseline= 6.3 Go 50 mg q4wks + DMARD: N=153; Biologic naive = No; % female= 74; age, yrs (median)=55; disease duration, yrs (median) = 9.6; HAQ baseline= 1.6; DAS28 baseline= 6.3 Go 100 mg q4wks + DMARD: N=153; Biologic naive = No; % female= 80; age, yrs (median)=55; disease duration, yrs (median) = 8.7; HAQ baseline= 1.5; DAS28 baseline=6.1 |
|
| Interventions | Placebo + DMARD (methotrexate, sulfasalazine or hydroxychloroquine in stable doses) Go 50 mg q4wks + DMARD Go 100 mg q4wks + DMARD |
|
| Outcomes |
Primary: ACR20 at wk 14 Secondary: ACR50% at wk 14 ACR70% at wk 14 DAS28 improvement from baseline at wk 14 and 24 ACR‐N (numeric index of ACR response) at wk 14 and 24 HAQ‐DI scores at wk 14 and 24 Fatigue score (FACIT‐F) DAS28 response according to EULAR DAS28 remission |
|
| Notes | "Concomitant disease‐modifying anti‐rheumatic drug (DMARD) treatment with methotrexate, sulfasalazine, and hydroxychloroquine (alone or in combination) was permitted but not required. Patients receiving such drugs must have tolerated the dose for at least 12 weeks, and the dose must have been stable for 4 weeks before the first dose of study drug. Patients who were receiving methotrexate, sulfasalazine, or hydroxychloroquine at baseline were allowed to discontinue these drugs before starting the study." | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Quote: "patients were randomly assigned in a 1:1:1 ratio... [Interactive Voice Response System, (IVRS)] supplied a code number that corresponded to a box that contained the appropriate treatment at the study site. The packaging for every box was identical except for the code number." Comment: Sufficient method of randomization |
| Allocation concealment? | Low risk | Quote: "IRVS supplied a code number that corresponded to a box that contained the appropriate treatment at the study site. The packaging for every box was identical except for the code number." Comment: Sufficient method of allocation concealment |
| Blinding? All outcomes | Low risk | Quote: "Golimumab and placebo were supplied in identical single‐use vials. Every patient received a .5 mL and a 1 mL injection every 4 weeks. Patients in the 50mg group received golimumab in the .5 mL syringe and placebo in the 1 mL syringe..." Comment: Adequate. |
| Incomplete outcome data addressed? All outcomes | Low risk | Quote: "Patients were included in the statistical analysis if they discontinued the study drug for reasons unrelated to lack of effectiveness and returned for assessment, but they were regarded as non‐responders if they met any of the failure criteria above. All efficacy data were analysed by intention to treat. All safety data were analysed according to the study drug that the patient received; patients who were randomized but never treated were not included. For patients who received rescue therapy, efficacy data from week 16 were carried forward for analysis at week 24 to ensure that the results were not biased by the increased dose the patient received." Comment: Sufficient statistical analysis of missing outcome data. |
| Free of selective reporting? | Unclear risk | Insufficient information to make a judgment |
| Free of other bias? | Unclear risk | "Centocor employees participated in study design, data analysis, data interpretation, and writing of the report. Schering‐Plough employees participated in data interpretation. Neither sponsor participated in data collection. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication." |
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| O'Connell 2008 | Commentary |
| Oldfield 2009 | Review |
Differences between protocol and review
None
Contributions of authors
JS‐ Title Registation
JS‐ Protocol development
JS, SN, GS‐ Protocol review and editing
JS, SN‐ Data Extraction and Analysis
JS, SN‐ Results and draft of the review
JS, SN, GS‐ Editing the review
Sources of support
Internal sources
-
Minneapolis VA Medical Support, USA.
Office space and Computer access
External sources
-
NIH CTSA KL2 Award (Mayo Clinic), USA.
75% time protection for research
Declarations of interest
JS‐ Speaker honoraria from Abbott; research and travel grants from Takeda, Savient, Wyeth and Amgen; consultant fees from Savient and URL pharmaceuticals
SN‐ None
GS‐ Honoraria/consultant/research grants from Centocor, Abbott, AMGEN, Johnson and Johnson
Edited (no change to conclusions)
References
References to studies included in this review
Emery 2009 {published data only}
- Emery P, Fleischmann RM, Moreland LW, Hsia EC, Strusberg I, Durez P, et al. Golimumab, a human anti‐tumor necrosis factor alpha monoclonal antibody, injected subcutaneously every four weeks in methotrexate‐naive patients with active rheumatoid arthritis: Twenty‐four‐week results of a phase III, multicenter, randomized, double‐blind, placebo‐controlled study of golimumab before methotrexate as first‐line therapy for early‐onset rheumatoid arthritis. Arthritis Rheum 2009;60(8):2272‐83. [DOI] [PubMed] [Google Scholar]
Kay 2008 {published data only}
- Kay J, Matteson EL, Dasgupta B, Nash P, Durez P, Hall S, et al. Golimumab in patients with active rheumatoid arthritis despite treatment with methotrexate: a randomized, double‐blind, placebo‐controlled, dose‐ranging study. Arthritis Rheum 2008;58:964‐75. [DOI] [PubMed] [Google Scholar]
Keystone 2009 {published data only}
- Keystone EC, Genovese MC, Klareskog L, Hsia EC, Hall ST, Miranda PC, et al. Golimumab, a human antibody to tumour necrosis factor {alpha} given by monthly subcutaneous injections, in active rheumatoid arthritis despite methotrexate therapy: the GO‐FORWARD Study. Ann Rheum Dis 2009;68:789‐96. [DOI] [PMC free article] [PubMed] [Google Scholar]
Smolen 2009 {published data only}
- Smolen JS, Kay J, Doyle MK, Landewe R, Matteson EL, Wollenhaupt J, et al. Golimumab in patients with active rheumatoid arthritis after treatment with tumour necrosis factor alpha inhibitors (GO‐AFTER study): a multicentre, randomised, double‐blind, placebo‐controlled, phase III trial. Lancet 2009;374(9685):210‐21. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
O'Connell 2008 {published data only}
- O'Connell N. New data shows golimumab significantly improves signs and symptoms of rheumatoid arthritis. Geriatric Medicine 2008;38:374‐375. [Google Scholar]
Oldfield 2009 {published data only}
- Oldfield V, Plosker GL. Golimumab: in the treatment of rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis. Bio Drugs 2009;23:125‐35. [DOI] [PubMed] [Google Scholar]
Additional references
Arnett 1988
- Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, et al. The American Rheumatism Association revised criteria for the classification of rheumatoid arthritis. Arthritis and Rheumatism 1988;31:315‐24. [DOI] [PubMed] [Google Scholar]
Barlow 1999
- Barlow JH, Cullen LA, Rowe IF. Comparison of knowledge and psychological well‐being between patients with a short disease duration (< or = 1 year) and patients with more established rheumatoid arthritis (> or = 10 years duration). Patient Educ Couns 1999;38(3):195‐203. [DOI] [PubMed] [Google Scholar]
Bhandari 2004
- Bhandari M, Busse JW, Jackowski D, Montori VM, Schunemann H, Sprague S, et al. Association between industry funding and statistically significant pro‐industry findings in medical and surgical randomized trials. CMAJ 2004;170(4):477‐80. [PMC free article] [PubMed] [Google Scholar]
Blumenauer 2002
- Blumenauer BBTB, Judd M, Wells GA, Burls A, Cranney A, Hochberg MC, et al. Infliximab for the treatment of rheumatoid arthritis. Cochrane Database of Systematic Reviews 2002, Issue 3. [DOI: 10.1002/14651858.CD003785] [DOI] [PMC free article] [PubMed] [Google Scholar]
Boers 2001
- Boers M. Rheumatoid arthritis. Treatment of early disease. Rheum Dis Clin North Am 2001;27(2):405‐14. [DOI] [PubMed] [Google Scholar]
Brennan 2008
- Brennan FM, McInnes IB. Evidence that cytokines play a role in rheumatoid arthritis. J Clin Invest 2008;118(11):3537‐45. [DOI] [PMC free article] [PubMed] [Google Scholar]
Choy 2001
- Choy EH, Panayi GS. Cytokine pathways and joint inflammation in rheumatoid arthritis. N Engl J Med 2001;344(12):907‐16. [DOI] [PubMed] [Google Scholar]
Choy 2005
- Choy EHS, Smith C, Dore CJ, Scott DL. A meta‐analysis of the efficacy and toxicity of combining disease‐modifying anti‐rheumatic drugs in rheumatoid arthritis based on patient withdrawal. Rheumatology 2005;44(11):1414‐21. [DOI] [PubMed] [Google Scholar]
Chung 2006
- Chung CP, Thompson JL, Koch GG, Amara I, Strand V, Pincus T. Are American College of Rheumatology 50% response criteria superior to 20% criteria in distinguishing active aggressive treatment in rheumatoid arthritis clinical trials reported since 1997? A meta‐analysis of discriminant capacities. Ann Rheum Dis 2006;65(12):1602‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Connell 2006
- Connell L, McInnes IB. New cytokine targets in inflammatory rheumatic diseases. Best Pract Res Clin Rheumatol 2006;20(5):865‐78. [DOI] [PubMed] [Google Scholar]
Deeks 2008
- Deeks J, Higgins J, Altman D. Chapter 9: Analysing data and undertaking meta‐analyses, Cochrane Handbook for Systematic Reviews of Interventions Version 5.0.0 (updated February 2008). The Cochrane Collaboration. Available from: www.cochrane‐handbook.org. 2008.
FDA 2009a
- FDA. SIMPONI® (Golimumab) Physician Packet Insert: Highlights of Prescribing Information. http://www.accessdata.fda.gov/drugsatdfa_docs/label/2009/125289s000lbl.pdf 2009.
FDA 2009b
- FDA. SIMPONI® (Golimumab) Summary Review. FDA Center for Drug Evaluation and Research http://www.accessdata.fda.gov/drugsatdfa_docs/nda/2009/125289s000_SumR.pdf April 4, 2009 (accessed September 20, 2009).
FDA 2009c
- FDA. Tumor Necrosis Factor (TNF) Blockers (marketed as Remicade, Enbrel, Humira, Cimzia, and Simponi) August 2009. http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm175843.htm August 31, 2009.
Felson 1995
- Felson DT, Anderson JJ, Boers M, Bombardier C, Furst D, Goldsmith C, et al. American college of rheumatology preliminary definition of improvement in rheumatoid arthritis. Arthritis & Rheumatism 1995;38(6):727‐35. [DOI] [PubMed] [Google Scholar]
Fransen 2005
- Fransen J, Riel PLCM. The disease activity score and the EULAR response criteria. Clinical and Experimental Rheumatology 2005;23(S39):S93‐9. [PubMed] [Google Scholar]
Fries 1980
- Fries JF, Spitz PW, Kraines RG, Holman HR. Measurement of patient outcome in arthritis. Arthritis & Rheumatism 1980;23:137‐45. [DOI] [PubMed] [Google Scholar]
Gomez‐Reino 2006
- Gomez‐Reino JJ, Carmona L. Switching TNF antagonists in patients with chronic arthritis: an observational study of 488 patients over a four‐year period. Arthritis Res Ther 2006;8(1):R29. [DOI] [PMC free article] [PubMed] [Google Scholar]
Harris 1990
- Harris E.D. Jr. Rheumatoid arthritis. Pathophysiology and implications for therapy. N Engl J Med 1990;322(18):1277‐89. [DOI] [PubMed] [Google Scholar]
Higgins 2008
- Higgins JP, Altman DG. Chapter 8: Assessing risk of bias in included studies. . In: Higgins JP, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions Version 5.0.0 [updated February 2008]. www.cochrane‐handbook.org. The Cochrane Collaboration, 2008. [Google Scholar]
Karlsson 2008
- Karlsson JA, Kristensen LE, Kapetanovic MC, Gulfe A, Saxne T, Geborek P. Treatment response to a second or third TNF‐inhibitor in RA: results from the South Swedish Arthritis Treatment Group Register. Rheumatology (Oxford) 2008;47(4):507‐13. [DOI] [PubMed] [Google Scholar]
Kvien 2005
- Kvien TK, Uhlig T. Quality of life in rheumatoid arthritis. Scand J Rheumatol 2005;34(5):333‐41. [DOI] [PubMed] [Google Scholar]
Larsen 1977
- Larsen A, Dale K, Eek M. Radiographic evaluation of rheumatoid arthritis and related conditions by standard reference films. Acta Radiologic Diagnosis (Stockholm) 1977;18(4):481–91. [DOI] [PubMed] [Google Scholar]
Lethaby 2003
- Lethaby A, Lopez‐Olivo MA, Blumenauer BBTB, Cranney A, Burls A, Coyle D, et al. Etanercept for the treatment of rheumatoid arthritis. Cochrane Database of Systematic Reviews 2003, Issue 4. [DOI: 10.1002/14651858.CD004525] [DOI] [PubMed] [Google Scholar]
Lopez‐Olivo 2008
- Lopez‐Olivo MA, Amezaga M, McGahan L, Suarez‐Almazor ME. Rituximab for rheumatoid arthritis. Cochrane Database of Systematic Reviews 2008, Issue 4. [DOI: 10.1002/14651858.CD007356] [DOI] [PMC free article] [PubMed] [Google Scholar]
Lubeck 2004
- Lubeck DP. Patient‐reported outcomes and their role in the assessment of rheumatoid arthritis. Pharmacoeconomics 2004;22(2 Suppl 1):27‐38. [DOI] [PubMed] [Google Scholar]
Maxwell 2008
- Maxwell L, Singh JA. Abatacept for rheumatoid arthritis. Cochrane Database of Systematic Reviews 2008, Issue 3. [DOI: 10.1002/14651858.CD007277] [DOI] [PMC free article] [PubMed] [Google Scholar]
Mertens 2009
- Mertens M, Singh JA. Anakinra for rheumatoid arthritis. Cochrane Database of Systematic Reviews 2009, Issue 1. [10.1002/14651858. CD005121] [DOI] [PMC free article] [PubMed] [Google Scholar]
Navarro‐Sarabia 2005
- Navarro‐Sarabia F, Ariza‐Ariza R, Hernandez‐Cruz B, Villanueva I. Adalimumab for treating rheumatoid arthritis. Cochrane Database of Systematic Reviews 2005, Issue 3. [DOI: 10.1002/14651858.CD005113.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Odegard 2005
- Odegard S, Finset A, Kvien Tk, Mowinckel P, Uhlig T. Work disability in rheumatoid arthritis is predicted by physical and psychological health status: a 7‐year study from the Oslo RA register.. Scand J Rheumatol 2005;34(6):441‐7. [DOI] [PubMed] [Google Scholar]
Pappas 2009
- Pappas DA, Bathon JM, Hanicq D, Yasothan U, Kirkpatrick P. Golimumab. Nat Rev Drug Discov 2009;8:695‐696. [DOI] [PubMed] [Google Scholar]
Pincus 1983
- Pincus T, Summey JA, Soraci SA, Jr, Wallston KA, Hummon NP. Assessment of patient satisfaction in activities of daily living using a modified Stanford Health Assessment Questionnaire. Arthritis & Rheumatism 1983;26:1346‐53. [DOI] [PubMed] [Google Scholar]
Prevoo 1995
- Prevoo MLL, Van't Hof MA, Kuper HH, Leeuwen MA, Putte LBA, Riel PLCM. Modified disease activity scores that include twenty‐eight‐joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis & Rheumatism 1995;38:44‐8. [DOI] [PubMed] [Google Scholar]
Scott 2006
- Scott DL, Kingsley GH. Tumor necrosis factor inhibitors for rheumatoid arthritis. N Engl J Med 2006;355(7):704‐12. [DOI] [PubMed] [Google Scholar]
Scott 2009
- Scott DL, Cope A. New tumour necrosis factor inhibitors for rheumatoid arthritis: are there benefits from extending choice?. Ann Rheum Dis 2009;68:767‐769. [DOI] [PubMed] [Google Scholar]
Sharp 1971
- Sharp JT, Lidsky MD, Collins LC, Moreland J. Methods of scoring the progression of radiologic changes in rheumatoid arthritis. Correlation of radiologic, clinical and laboratory abnormalities. Arthritis & Rheumatism 1971;14(6):706–20. [DOI] [PubMed] [Google Scholar]
Siddiqui 2007
- Siddiqui MA. The efficacy and tolerability of newer biologics in rheumatoid arthritis: best current evidence. Curr Opin Rheumatol 2007;19(3):308‐13. [DOI] [PubMed] [Google Scholar]
van der Heijde 1989
- Heijde DMFM, Riel PL, Nuver‐Zwart IH, Gribnau FW, Puttte LB. Effects of hydroxychloroquine and sulfasalazine on progression of joint damage in rheumatoid arthritis. Lancet 1989;1:1036‐8. [DOI] [PubMed] [Google Scholar]
van der Heijde 1993
- Heijde DM, 't Hof M, Riel PL, Putte LB. Development of a disease activity score based on judgment in clinical practice by rheumatologists. Journal of Rheumatology 1993;20(3):579‐81. [PubMed] [Google Scholar]
van Gestel 1996
- Gestel A, Riel P. American College of Rheumatology preliminary definition of improvement in rheumatoid arthritis: comment on the article by Felson et al. Arthritis Rheum 1996;39(3):535‐7. [DOI] [PubMed] [Google Scholar]
Weissmann 2006
- Weissmann G. The pathogenesis of rheumatoid arthritis. Bull NYU Hosp Jt Dis 2006;64(1‐2):12‐5. [PubMed] [Google Scholar]
Wells 1993
- Wells GA, Tugwell P, Kraag GR, Baker PR, Groh J, Redelmeier DA. Minimum important difference between patients with rheumatoid arthritis: the patient's perspective. Journal of Rheumatology 1993;20:557. [PubMed] [Google Scholar]
Yazici 2009
- Yazici Y. Treatment of rheumatoid arthritis: we are getting there. Lancet 2009;374:178‐180. [DOI] [PubMed] [Google Scholar]
Yelin 2007
- Yelin E. Work disability in rheumatic diseases. Curr Opin Rheumatol 2007;19(2):91‐6. [DOI] [PubMed] [Google Scholar]


