Abstract
Regulated expression of a therapeutic gene is crucial for safe and efficacious gene therapy. Many inducible regulatory systems use a constitutive promoter to express a regulatory protein, such as rtTA in the Tet-On system, which may restrict their use because of cytotoxicity and immunogenicity. Autoregulatory expression of rtTA provides extremely low levels of rtTA when transgene expression is off, with rapid transgene induction upon addition of doxycycline. Lentiviral vectors efficiently transfer genes to dividing and non-dividing cells with long-term gene expression both in vitro and in vivo. We compared regulatory function in a single lentiviral vector where rtTA was either expressed from a constitutive promoter or placed in an autoregulatory loop. Autoregulatory expression of rtTA was superior to constitutive promoter expression, resulting in higher viral titers, undetectable levels of both rtTA and transgene expression in the absence of doxycycline, improved induction kinetics and increased induction levels in all cells tested. We further expanded the utility of the autoregulatory vector by using an improved rtTA variant with an increased sensitivity to doxycycline. This lentiviral vector with doxycycline-regulated transgene expression may be useful for gene therapy applications and in experimental settings where strict temporal expression of a transgene is required.
INTRODUCTION
Lentiviral vectors are efficient vehicles for the delivery of genes to both dividing and non-dividing cells in vitro and in vivo (1–3). Many vectors for gene therapy use a strong constitutive promoter to drive the expression of the transgene, which may not reflect physiological expression levels and can in fact be harmful to the cell (4,5). Progress toward the clinical use of lentiviral vectors for gene therapy will require the ability to regulate the expression of the transgene for improved safety and efficacy (4,5). An ideal gene therapy vector should include a regulatory system that is off in the resting state, exhibit tight regulation and allow for rapid and repeatable induction in response to a clinically approved inducer molecule.
The tetracycline-dependent transcriptional regulatory system (6) is one of the best studied systems with proven efficacy in vitro and in vivo (4,5,7,8). This system is based on the Escherichia coli Tn10 Tetracycline resistance operator consisting of the tetracycline repressor protein (TetR) and a specific DNA-binding site, the tetracycline operator sequence (TetO). In the absence of tetracycline, TetR dimerizes and binds to the TetO. Tetracycline or doxycycline (a tetracycline derivative) can bind and induce a conformational change in the TetR leading to its disassociation from the TetO. A TetR mutant was identified with a reverse phenotype where binding to the TetO was triggered by doxycycline (9). Fusion of the VP16 transactivation domain of the Herpes simplex virus to either TetR or the mutant TetR resulted in a tetracycline responsive transactivator (tTA) (6) and a reverse tTA (rtTA) (9). Mutagenesis and codon optimization of the TetR and reduction of the VP16 activation domain to three repeats of a 12 amino acid minimal activation domain (10) generated an improved rtTA, rtTA2S-S2 (11), with reduced background activity. A tetracycline responsive promoter (TRE) for mammalian expression was constructed by fusing a minimal cytomegalovirus (CMV) promoter to seven TetO repeats (6), which was combined with either tTA to make the Tet-Off or rtTA to make the Tet-On transcriptional regulatory system.
Although the Tet-Off system is more sensitive to doxycycline as opposed to the Tet-On system, there are several features that make the Tet-Off system less suitable for gene therapy applications. Induction with the Tet-Off system depends on the pharmacological elimination of doxycycline and tends to be slower as compared with the Tet-On system (9,12,13). In addition, the Tet-Off system requires persistent administration of doxycycline to suppress gene expression, which may not be ideal when used with a lentiviral vector that provides life long gene expression. These properties make the Tet-On system a better choice for transcriptional regulation in most gene therapy applications. Recent improvements in the rtTA protein (11,14), including reduced background activity and increased doxycycline sensitivity, now make this possible.
Many Tet-On regulated transgene expression systems use two separate constructs, one containing the TRE-regulated transgene and the second containing rtTA expressed by a constitutive promoter. This binary system relies on the co-transduction of cells and requires selection and screening to obtain a homogenously transduced population, which is not possible in vivo. We have combined all the elements required for Tet-On regulation into a single cassette with either a constitutive promoter or an autoregulatory loop for the expression of rtTA. These cassettes were cloned into a third generation lentiviral vector system for the production of self-inactivating vectors (15,16) containing the central polypurine tract (cPPT) and the Hepatitis B post-transcriptional regulatory element (PRE) for enhanced gene expression (17).
A limited number of studies have evaluated the autoregulatory expression of rtTA in a plasmid (18,19) and AAV vector (20) as compared with tTA (21–26). Autoregulatory expression of rtTA allows for extremely low levels of both the transactivator and the transgene product in the absence of doxycycline, with sufficient rtTA molecules for initiating the expression upon addition of doxycycline. Restricted expression of rtTA with an autoregulatory system could expand applicability both in vitro and possibly in vivo. Previous studies of autoregulatory rtTA expression were performed with transient vector systems (18–20) that remain episomal. In contrast, lentiviral vector gene transfer results in stable integration and long-term gene expression, which presents a different context for the evaluation of autoregulatory rtTA expression. Our aim is to compare transcriptional regulatory function between a constitutive promoter and an autoregulatory loop driving expression of rtTA in a single lentiviral vector. Optimization of the lentiviral vector constructs was performed for the production of high titer regulatable lentiviral vectors. We tested this vector system in a panel of human primary and established cell lines of diverse origin. We further compared rtTA protein expression, repeated induction cycles, induction kinetics and doxycycline concentration-dependent expression in single Tet-On lentiviral vectors with a constitutive promoter or an autoregulatory loop for the expression of rtTA.
MATERIALS AND METHODS
Cloning of the constructs
First generation vectors were adapted from a conditional replicating HIV-1 vector (27). The viral LTR containing eight copies of the TetO and rtTA was cloned as a partial AflII and EcoRI fragment and cloned into pRRL cPPT PGK eGFP PRE SIN (17) to make LTRCMVAR2. The EMCV IRES fragment was cloned by PCR into the pCR2.1 TOPO vector using the following primers and sequence verified.
IRES S
5′-ATCCGCGGACCGGTACGTACTCGAGGCTGCAGGAATTCCGCCCC-3′
IRES AS
5′-TTGTCCAGTCTAGACATGGTTGTGGCCATATTATCATCGTG-3′
The CMV promoter from LTRConAR2 was removed by XbaI–SacII digestion. An XbaI–SacII digest of pCR2.1 IRES released the internal ribosome entry site (IRES) for ligation into LTRCMVAR2 to make LTRAutoAR2. A BamHI–EcoRI digest of pd2eGFP (28) created a d2eGFP fragment that was cloned into LTRAutoAR2 to make LTRAutoR2. An AgeI–EcoRI digest of pd2eGFP created a d2eGFP fragment that was ligated into LTRConAR2 to make LTRConR2.
pRRL cPPT d2eGFP CMV rtTA2S-S2 LTRTetO8 SIN (LTRCMVR2)
pRRL cPPT d2eGFP IRES rtTA2S-S2 LTRTetO8 SIN (LTRAutoR2)
An AgeI digest of LTRAutoR2 removed d2eGFP and the subsequent ligation created LTRMCSAutoR2. Digestion of pCMV-rtTAS12G F86Y A209T (14) (rtTA3) with XbaI–XmaI created an rtTA3 fragment that was ligated into an XbaI–XmaI digest of LTRMCSAutoR2 to make LTRMCSAutoR3.
pRRL cPPT TRE d2eGFP CMV rtTA2S-S2 PRE LTR SIN (TRECMVR2)
pRRL cPPT TRE d2eGFP IRES rtTA2S-S2 PRE LTR SIN (TREAutoR2)
pRRL cPPT TRE d2eGFP IRES rtTA3 PRE LTR SIN (TREAutoR3)
The CMVR2 and AutoR2 fragments were created by a BamHI–EcoRV digest of LTRCMVR2 and LTRAutoR2, respectively. The pUHV13-3wt4c vector containing CMVmin fused to seven copies of the TetO was also digested with BamHI–EcoRV to remove luciferase. The CMVR2 and AutoR2 fragments were ligated to create TRECMVA and TREAutoA. An XhoI site 5′ of the TRE was replaced by a ClaI site. A ClaI–DraI digest of both TREAutoB and TRECMVB released the complete Tet-On Auto and CMV cassettes, respectively, which were subcloned into a ClaI–EcoRv digested pBSK vector to make pBTREAutoB and pBTRECMVB. The Tet-On Auto and CMV cassettes were released by a ClaI–SpeI digest and ligated into a ClaI–SpeI digested lentiviral vector backbone to make TREAutoR2 and TRECMVR2. An AgeI–EcoRv digest of LTRAutoR3 released the Tet-On Auto fragment with rtTA3. The fragment was cloned into an AgeI–AscI (blunted) digest of TREAutoR2 to create TREAutoR3.
Cell lines and culturing
The following cell lines and primary cells were used in this study: HEK293T, HeLa, HepG2, SJNB-8 (neuroblastoma), human umbilical vein endothelial cells (HUVECs), human fibroblasts and human fetal liver cells (HFLCs). All cells were cultured (with the exception of HUVEC and HFLC) in standard DMEM supplemented with 10% fetal bovine serum (FBS), 100 U/ml penicillin, 100 μg/ml streptomycin and 2 mM glutamine at 37°C in 10% CO2.
HFLC cells were cultured in standard DMEM, above, supplemented with 100 nM dexamethasome and 1× ITS mix (Invitrogen).
HUVEC cells were cultured in M199 with 25 mM HEPES (Gibco) supplemented with 10% FBS, 100 U/ml penicillin, 100 μg/ml streptomycin, 2 mM glutamine, 50 μg/ml heparin and 25 μg/ml endothelial cell growth supplement (Sigma). Cells were cultured on fibronectin coated flasks and plates.
Lentiviral vector preparation
Lentiviral vectors were prepared as reported previously (29). Briefly, HEK 293T cells were transiently transfected by calcium phosphate precipitation with a third generation lentiviral vector system (15,16). Twenty-four hours following transfection, fresh medium was added supplemented with 25 mM HEPES, pH 7.4. Virus containing supernatant was collected 48 h following transfection, filtered through 0.45 μm Millipore filters and frozen at −80°C.
Virus titer determination and cell transduction
Viral titers were determined by serial dilution of virus on HeLa cells transduced for 4 h with 10 μg/ml DEAE Dextran. Doxycycline (Sigma) was dissolved at 10 mg/ml in water and filter-sterilized. Stocks were kept frozen at −20°C. Doxycycline (1000 ng/ml) was added to cells for 72 h and cells were harvested and green fluorescent protein (GFP) expression was measured by flow cytometry. For all experiments, induction was performed with 1000 ng/ml doxycycline unless otherwise specified.
SDS–PAGE and western blotting
Cells were harvested by washing with phosphate-buffered saline (PBS) and collected by scraping in PBS with 5 mM EDTA. Lysates were made by sonication in PBS with 5 mM EDTA. Protein levels were determined using the Bio-Rad DC Protein Assay. An aliquot of 25 μg of total protein was loaded for each lysate on NuPage gels (10% Bis-Tris with MOPS running buffer; Invitrogen) according to the manufacturer's instructions. Gel and western blotting was performed in an Xcell SureLock Mini-Cell (Invitrogen) following the manufacturer's instructions.
An antibody directed against rtTA was purchased from MoBiTec (TET02) and used at a dilution of 1:2000. A monoclonal antibody directed against eGFP was purchased from Clontech (JL-8) and used at a dilution of 1:5000. A monoclonal antibody directed against actin was purchased from NeoMarkers (Ab-5) and used at a dilution of 1:1000. Goat anti-mouse IgG (H + L)–horseradish peroxidase conjugate (Bio-Rad) was used at a 1:1000 dilution. Lumi-Light western blotting substrate (Roche) was used for the detection of bands and the blot was scanned in using a LumiImager F1 and LumiAnalyst 3.1 software (Roche).
RESULTS
Generation of lentiviral vector constructs
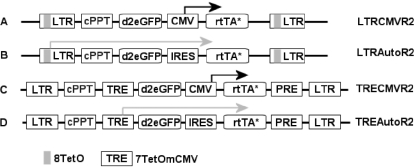
In all our constructs, we used a destabilized form of GFP (d2eGFP, 2 h half-life) (28) specifically designed for use with transcriptional regulatory systems. Our first generation single Tet-On lentiviral vectors, LTRCMVR2 (a constitutive CMV promoter driving rtTA expression) and LTRAutoR2 (autoregulatory rtTA expression), contain the TetO element within the vector LTR (27) (Figure 1A and B). These constructs yielded viral titers ∼100 times lower when compared with lentiviral vectors expressing GFP from a constitutive promoter (Table 1) and were, therefore, not considered for further use. In our second generation vectors TRECMVR2 (a constitutive CMV promoter driving rtTA expression) and TREAutoR2 (autoregulatory rtTA expression) (Figure 1C and D), we introduced two improvements over the first generation. First, the TetO element (TRE) was removed from the LTR and internalized in the vector. Second, to enhance the gene expression, the Hepatitis B post-transcriptional regulatory element was added 3′ of the expression cassette. Only TREAutoR2 and TREAutoR3 yielded viral titers comparable with a lentiviral vector with constitutive expression of GFP (Table 1).
Figure 1.
(A) Schematic representation of the constructed lentiviral vectors. First generation single dox-regulated lentiviral vector with eight repeats of the TetO located within the viral LTR with rtTA expressed from the strong constitutively active CMV promoter LTRCMVR2 (A) or rtTA expressed as a bicistronic mRNA LTRAutoR2 (B). Second generation single dox-regulated lentiviral vector with an internal tetracycline responsive element (TRE) containing a minimal CMV promoter fused to seven copies of the TetO and the HBV PRE with rtTA expressed from the strong constitutively active CMV promoter TRECMVR2 (C) or rtTA expressed as a bicistronic mRNA TREAutoR2 (D). Asterisk: rtTA was either rtTA2S-S2 or rtTA3 (rtTA2S-S2 with three amino acid substitutions S12G, F86Y and A209T).
Table 1.
Viral titers reported as HeLa transducing units (HTU/ml)
| Lentiviral vector | HTU/ml |
|---|---|
| PGKeGFP | 2.5 × 106 ± 3.3 × 105 |
| LTRCMVR2 | 1.6 × 104 ± 1.3 × 104 |
| LTRAutoR2 | 4.2 × 103 ± 8.5 × 102 |
| TRECMVR2 | 2.2 × 105 ± 1.4 × 105 |
| TREAutoR2 | 1.0 × 106 ± 2.5 × 105 |
| TREAutoR3 | 1.3 × 106 ± 3.7 × 105 |
PGKeGFP is a lentiviral vector with constitutive expression of eGFP using the PGK promoter. Mean and standard deviations were determined from at least three independent virus preparations.
Autoregulatory expression of rtTA results in higher induction levels
For a comprehensive analysis of the induction levels of both TRECMVR2 and TREAutoR2, we chose a panel of human primary and established cell lines derived from a variety of tissues. Each cell line was transduced at a multiplicity of infection (MOI) of 1, induced with 1000 ng/ml doxycycline for 72 h and harvested for flow cytometry analysis. Analysis was performed on the whole population of transduced cells to avoid bias from clonal selection. We observed superior gene induction in all cell lines transduced with TREAutoR2 compared with TRECMVR2 (Table 2). Primary human fibroblasts provided the highest induction levels for both TRECMVR2 and TREAutoR2 (Table 2). The primary cells, fibroblasts, HUVEC, and HFLC and the cell line SJNB-8, gave approximately equal percentage of GFP positive cells with TREConR2 and TREAutoR2 (Table 2). In contrast, we observed a higher percentage of GFP positive cells with TREAutoR2 (Table 2) in the 293T, HeLa and HepG2 cell lines. Together these data show that the autoregulatory expression of rtTA improves the induction levels in all cells tested and may lead to an improvement in the transduction efficiency in certain cell types.
Table 2.
Induction levels and percentage of GFP positive cells on indicated cell lines transduced at an MOI of 1 with either TRECMVR2 or TREAutoR2 lentiviral vectors
| Cell type | Virus | Induction | Percentage of GFP positive cells |
|---|---|---|---|
| HUVEC | TRECMVR2 | 11.35 ± 3.91 | 55.02 ± 34.14 |
| TREAutoR2 | 32.97 ± 19.46 | 68.33 ± 33.39 | |
| Fibroblast | TRECMVR2 | 22.32 ± 2.67 | 83.95 ± 7.75 |
| TREAutoR2 | 49.47 ± 6.87 | 93.77 ± 2.89 | |
| HFLC | TRECMVR2 | 6.46 ± 0.85 | 48.62 ± 22.83 |
| TREAutoR2 | 28.89 ± 13.54 | 49.92 ± 20.36 | |
| 293T | TRECMVR2 | 10.44 ± 1.21 | 34.48 ± 5.98 |
| TREAutoR2 | 17.21 ± 1.42 | 71.07 ± 11.95 | |
| HeLa | TRECMVR2 | 13.23 ± 2.40 | 49.60 ± 11.14 |
| TREAutoR2 | 48.84 ± 20.04 | 88.73 ± 3.04 | |
| HepG2 | TRECMVR2 | 3.81 ± 0.86 | 36.23 ± 6.58 |
| TREAutoR2 | 26.94 ± 11.72 | 68.97 ± 13.31 | |
| SJNB-8 | TRECMVR2 | 4.22 ± 0.50 | 31.62 ± 5.33 |
| TREAutoR2 | 14.59 ± 1.65 | 31.80 ± 4.77 |
Induction values were calculated by dividing the mean fluorescence of d2eGFP expression with doxycycline by the mean fluorescence without doxycycline. Primary cell lines are HUVEC, human skin fibroblasts and HFLC. Established cell lines are 293T HEK (human embryonic kidney), HeLa (cervical carcinoma), HepG2 (hepatoma) and SJNB-8 (neuroblastoma). Induction values and percentage of GFP positive cells represent three independent experiments performed in duplicate.
Undetectable basal levels of rtTA with autoregulatory expression
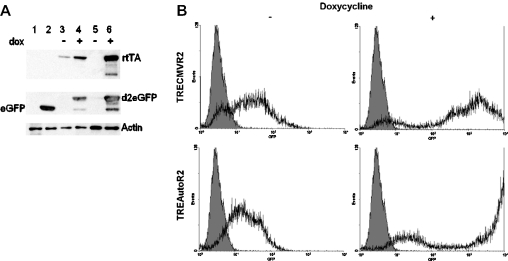
Autoregulatory expression of rtTA was expected to result in a low basal expression of both rtTA and transgene in the absence of doxycycline. To confirm this, primary human fibroblasts were transduced at an MOI of 1 with either TRECMVR2 or TREAutoR2. Following expansion, the cells were cultured with or without doxycycline at 1000 ng/ml for 72 h and then harvested. In the untreated cell lysates of TREAutoR2 transduced cells, we were unable to detect the expression of rtTA and d2eGFP by western blotting (Figure 2A, lane 5), confirming that the basal expression of rtTA was extremely low. Following addition of doxycycline, we observed profound induction of rtTA and d2eGFP (Figure 2A, lane 6). The destabilized GFP, d2eGFP, has a C-terminal fusion from the C-terminus of the mouse ornithine decarboxylase protein (28) resulting in the observed retarded migration (Figure 2A lane 2 versus lanes 4 and 6). TRECMVR2-transduced fibroblasts expressed a significant amount of rtTA protein in the absence of doxycycline (Figure 2A, lane 3). Interestingly, we observed an increase in the rtTA expression with TREConR2 following addition of doxycycline (Figure 2A, lane 4), which is probably a result of transcriptional read-through or promoter interference. Representative flow cytometry histograms of primary human fibroblasts transduced at an MOI of 1 with either TRECMVR2 or TREAutoR2 grown in the absence or presence of doxycycline further demonstrate the superior regulation and induction of TREAutoR2 (Figure 2B). The immunoblotting and flow cytometry results validate that TREAutoR2 allows for the regulated expression of both rtTA2S-S2 and d2eGFP.
Figure 2.
(A) Immunoblots of primary human fibroblasts lysates transduced with the indicated lentiviral vectors: lane 1, non-transduced cells; lane 2, PGKeGFP; lanes 3 and 4, TRECMVR2; lanes 5 and 6, TREAutoR2. Prior to harvesting cells were grown either in the absence (−) or presence (+) of doxycycline (1000 ng/ml) for 72 h. Separate immunoblots were probed with an antibody directed against either rtTA, GFP or actin. (B) Representative flow cytometry histograms of primary human fibroblasts transduced at an MOI of 1 with TRECMVR2 and TREAutoR2 in the absence and presence of doxycycline.
A constitutive promoter expression of rtTA results in a loss of transduced cells following repeating induction cycles
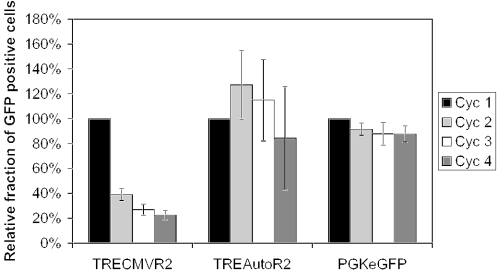
Repeated induction cycles may be required for many gene therapy and gene function applications. A constitutive promoter driving expression of rtTA may be detrimental, particularly when transgene expression is not induced owing to cytotoxic, anti-proliferative and immunogenic properties of rtTA. We, therefore, performed repeated induction cycles in HeLa cells transduced with either TRECMVR2 or TREAutoR2 to evaluate the relative effects on cell proliferation and toxicity. HeLa cells were transduced at a low MOI of 0.1 with either TRECMVR2, TREAutoR2 or a control lentiviral vector PGKeGFP expressing GFP from the PGK promoter. In one induction cycle, transduced cells were grown in the presence of doxycycline for 72 h, passaged and grown without doxycycline for 96 h. We set the percentage of GFP positive cells following the first induction cycle to 100% and subsequent induction cycles are presented relative to this value. The percentage of GFP positive cells transduced with the control lentiviral vector remained unchanged for the duration of the study (Figure 3). The percentage of GFP positive cells transduced with TREConR2 decreased by more than 50% after a single induction cycle and fell to 20% of the starting value after repeated cycles. In contrast, the percentage of TREAutoR2-transduced cells remained unchanged even after four induction cycles (Figure 3). Only when TREAutoR2-transduced HeLa cells were grown continuously in the presence of doxycycline, we observed a similar reduction in the relative amount of GFP positive cells (data not shown). These results indicate that high levels of rtTA protein obtained with a strong constitutive promoter driving rtTA expression or a continuously activated autoregulatory loop may decrease cell proliferation or is cytotoxic.
Figure 3.
Cyclic induction with serial passaging of HeLa cells transduced at an MOI of 0.1 with PGKeGFP, TRECMVR2 or TREAutoR2. The number of GFP positive cells detected upon first induction was set to 100% with all subsequent measurements normalized to the first induction levels. Cells were cultured for 3 days with doxycycline and the percentage of GFP positive cells was determined by flow cytometry. Subsequently, the cells were passaged and cultured for 4 days without doxycycline. A new induction cycle was initiated by passaging the cells and the addition of doxycycline. These data are from three independent experiments performed in duplicate with mean and standard deviations.
Rapid induction with the autoregulated rtTA lentiviral vector
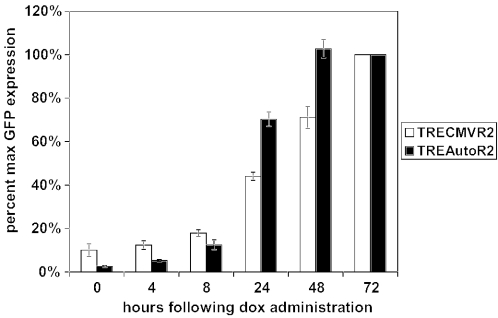
An effective transcriptional regulatory system should be rapidly turned on following the administration of the inducer molecule. The low basal expression of rtTA2S-S2 from TREAutoR2 in the absence of doxycycline could increase the time required for maximal activation as compared with TRECMVR2. To determine the time for maximal induction, we transduced HeLa cells at an MOI of 1 with either TRECMVR2 or TREAutoR2 and added doxycycline at fixed time points. We set 72 h as the time required for maximal expression because we observed no further increase in GFP expression at later time points (D.M. Markusic, unpublished data). Maximum gene expression of d2eGFP was obtained with TREAutoR2 at 48 h as compared with TREConR2 at 72 h (Figure 4). Thus, in addition to higher induction levels, TREAutoR2 also triggers a more rapid gene induction than TRECMVR2.
Figure 4.
Induction kinetics of HeLa cells transduced at an MOI of 1 with TRECMVR2 or TREAutoR2. Percentage of maximum expression at indicated time points following doxycycline administration. Values represent the mean and standard deviation of three independent experiments performed in duplicate.
The autoregulated lentiviral vector is greatly improved with a more sensitive rtTA variant
The development of a lentiviral vector with regulated transgene expression for use in gene therapy requires complete gene control at physiological obtainable concentrations of doxycycline. We compared the level of d2eGFP expression of TRECMVR2 and TREAutoR2 on primary fibroblasts over a range of doxycycline concentrations (0–1000 ng/ml). Low doxycycline concentrations did not activate d2eGFP expression with TREAutoR2 (Figure 5A and B). The very low basal expression of rtTA2S-S2 and low sensitivity to doxycycline probably prevents the successful initiation of the autoregulatory loop. To increase doxycycline sensitivity, we replaced rtTA2S-S2 with the recently described improved rtTA3 variant with improved activity and doxycycline sensitivity (14). Substitution of rtTA3 into TREAutoR2 to make TREAutoR3 resulted in viral titers similar to TREAutoR2 (Table 1) and activation of the autoregulatory loop and gene expression at a lower concentration of doxycycline (Figure 5A and B). Furthermore, at 100 ng/ml doxycycline TREAutoR3 reached expression levels 4-fold higher than TREConR2 and 7-fold higher than TREAutoR2, and at 1000 ng/ml doxycycline both TREAutoR2 and TREAutoR3 reached expression levels higher than TRECMVR2.
Figure 5.
Doxycycline dose–response on primary human fibroblasts transduced at an MOI 0.1 with TRECMVR2, TREAutoR2 or TREAutoR3. (A) Representative flow cytometry histograms of primary human fibroblasts transduced with indicated virus and treated with either 0, 100 or 1000 ng/ml doxycycline. (B) Mean fluorescent intensity of fibroblasts treated with 0, 25, 50, 100, 500 or 1000 ng/ml doxycycline transduced with indicated inducible lentiviral vector. Closed triangles represent TRECMVR2; open squares represent TREAutoR2; and closed diamonds represent TREAutoR3. Values represent the mean and standard deviation of two independent experiments performed in duplicate.
DISCUSSION
We report a single Tet-On lentiviral vector with autoregulated rtTA expression for regulated expression of a transgene. Previous studies using autoregulation of tTA or rtTA tested the addition of a second TRE (18,24) or a bi-directional TRE (19,20,25). When the strong TRE promoter was used to drive the expression of tTA or rtTA, cytotoxicity (19,24) and reduced cell proliferation (30) were reported. To reduce the efficiency of rtTA expression, we used a bicistronic mRNA that expresses both the transgene and rtTA via an IRES (31) (Figure 1D). Previous studies using a bicistronic mRNA for autoregulatory expression were performed in a single cell line with the Tet-Off system (tTA) in a retroviral vector backbone (21,22). These studies used either drug selection or cell sorting to isolate cell clones that exhibit optimal induction and regulation, which leads to bias and overestimation of regulatory function as compared with the evaluation on a population of transduced cells. Our Tet-On lentiviral vector with autoregulated rtTA expression further allows for efficient gene transfer to non-dividing cells, does not require persistent doxycycline administration to switch off the gene expression, and is better suited for gene therapy and a variety of gene function applications.
We compared lentiviral vectors in which rtTA is expressed from a constitutive promoter with vectors in which rtTA is placed in an autoregulatory loop. Autoregulatory expression of rtTA (TREAutoR2 and TREAutoR3) resulted in higher lentiviral vector titers as compared with the constitutive promoter expression vector (TRECMVR2) (Table 1). The autoregulatory vector TREAutoR2 also provided superior induction levels on a panel of human primary and established cells lines (Table 2), which is consistent with previous findings with the Tet-Off system (24). The variable induction levels observed with the different cell lines can be attributed to relative differences in transduction efficiencies or perhaps relative sensitivity to the cytotoxic effects of rtTA. With two established cell lines commonly used in our laboratory, Caco-2 (a coloncarcinoma cell line) and CC-LP-1 (32) (a cholangiocarcinoma cell line), we observed cytotoxicity when cells were transduced with TREConR2 in the absence or presence of doxycycline and with TREAutoR2 only when doxycycline was present (data not shown). These findings further demonstrate the very low basal expression of rtTA in the absence of doxycycline obtained with TREAutoR2 and suggest that rtTA may not be compatible with all cell types in vitro.
We demonstrated with immunoblotting that there was no detectable level of rtTA expression with TREAutoR2 in the absence of doxycycline, and transgene expression was rapidly induced following addition of doxycycline (Figure 2A and B). Repeated cycles of gene induction and expansion of HeLa cells transduced with TRECMVR2 resulted in a loss of GFP expressing cells over time, suggesting that a strong constitutive promoter driving expression of rtTA may result in decreased cell proliferation or cytotoxicity (Figure 3). This was not observed when HeLa cells were transduced with TREAutoR2 or a control lentiviral vector expressing GFP from the PGK promoter. We, therefore, demonstrated that TREAutoR2 is superior over TRECMVR2 for long-term culturing with repeated induction cycles. The TREAutoR2 lentiviral vector resulted in superior induction kinetics over TRECMVR2 (Figure 4). With TREAutoR2, rtTA is able to activate its own promoter upon doxycycline administration resulting in a rapid increase in the level of rtTA protein, which leads to the strong and rapid induction in transgene expression. One disadvantage of TREAutoR2 was the requirement for high concentrations of doxycycline to turn on gene expression, which may not be possible to obtain in vivo. By substituting rtTA2s-S2 with a greatly improved rtTA variant (14), rtTA3, we were able to significantly reduce the concentration of doxycycline required for activation (Figure 5A and B).
This is the first comprehensive study comparing gene regulation with constitutive or autoregulatory expression of rtTA in the context of a single lentiviral vector system. Single lentiviral vectors with doxycycline regulated gene expression have been described with tTA (33–35) and rtTA (36). All of these vectors used a strong constitutive promoter to express either tTA or rtTA. Our results indicate that the autoregulatory expression of rtTA creates an improved transcriptional regulatory system with expanded applications in vitro and may be more effective in vivo as compared with the constitutive expression of rtTA. The Tet-On system with autoregulatory rtTA expression in a single lentiviral vector is well suited for a variety of applications, including gene therapy, generation of transgenic animals, conditional immortalization and in research applications where strict temporal gene expression is required.
Acknowledgments
This research was made possible by a grant from NWO, 016.026.012 (to J.S.). Funding to pay the Open Access publication charges for this article was provided by the AMC Liver center.
Conflict of interest statement. None declared.
REFERENCES
- 1.Ailles L.E., Naldini L. HIV-1-derived lentiviral vectors. Curr. Top. Microbiol. Immunol. 2002;261:31–52. doi: 10.1007/978-3-642-56114-6_2. [DOI] [PubMed] [Google Scholar]
- 2.Trono D. Lentiviral vectors: turning a deadly foe into a therapeutic agent. Gene Ther. 2000;7:20–23. doi: 10.1038/sj.gt.3301105. [DOI] [PubMed] [Google Scholar]
- 3.Galimi F., Verma I.M. Opportunities for the use of lentiviral vectors in human gene therapy. Curr. Top. Microbiol. Immunol. 2002;261:245–254. doi: 10.1007/978-3-642-56114-6_13. [DOI] [PubMed] [Google Scholar]
- 4.Clackson T. Regulated gene expression systems. Gene Ther. 2000;7:120–125. doi: 10.1038/sj.gt.3301120. [DOI] [PubMed] [Google Scholar]
- 5.Toniatti C., Bujard H., Cortese R., Ciliberto G. Gene therapy progress and prospects: transcription regulatory systems. Gene Ther. 2004;11:649–657. doi: 10.1038/sj.gt.3302251. [DOI] [PubMed] [Google Scholar]
- 6.Gossen M., Bujard H. Tight control of gene expression in mammalian cells by tetracycline-responsive promoters. Proc. Natl Acad. Sci. USA. 1992;89:5547–5551. doi: 10.1073/pnas.89.12.5547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Agha-Mohammadi S., Lotze M.T. Regulatable systems: applications in gene therapy and replicating viruses. J. Clin. Invest. 2000;105:1177–1183. doi: 10.1172/JCI10027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Corbel S.Y., Rossi F.M. Latest developments and in vivo use of the Tet system: ex vivo and in vivo delivery of tetracycline-regulated genes. Curr. Opin. Biotechnol. 2002;13:448–452. doi: 10.1016/s0958-1669(02)00361-0. [DOI] [PubMed] [Google Scholar]
- 9.Gossen M., Freundlieb S., Bender G., Muller G., Hillen W., Bujard H. Transcriptional activation by tetracyclines in mammalian cells. Science. 1995;268:1766–1769. doi: 10.1126/science.7792603. [DOI] [PubMed] [Google Scholar]
- 10.Baron U., Gossen M., Bujard H. Tetracycline-controlled transcription in eukaryotes: novel transactivators with graded transactivation potential. Nucleic Acids Res. 1997;25:2723–2729. doi: 10.1093/nar/25.14.2723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Urlinger S., Baron U., Thellmann M., Hasan M.T., Bujard H., Hillen W. Exploring the sequence space for tetracycline-dependent transcriptional activators: novel mutations yield expanded range and sensitivity. Proc. Natl Acad. Sci. USA. 2000;97:7963–7968. doi: 10.1073/pnas.130192197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kistner A., Gossen M., Zimmermann F., Jerecic J., Ullmer C., Lubbert H., Bujard H. Doxycycline-mediated quantitative and tissue-specific control of gene expression in transgenic mice. Proc. Natl Acad. Sci. USA. 1996;93:10933–10938. doi: 10.1073/pnas.93.20.10933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mohammadi S., Alvarez-Vallina L., Ashworth L.J., Hawkins R.E. Delay in resumption of the activity of tetracycline-regulatable promoter following removal of tetracycline analogues. Gene Ther. 1997;4:993–997. doi: 10.1038/sj.gt.3300491. [DOI] [PubMed] [Google Scholar]
- 14.Das A.T., Zhou X., Vink M., Klaver B., Verhoef K., Marzio G., Berkhout B. Viral evolution as a tool to improve the tetracycline-regulated gene expression system. J. Biol. Chem. 2004;279:18776–18782. doi: 10.1074/jbc.M313895200. [DOI] [PubMed] [Google Scholar]
- 15.Dull T., Zufferey R., Kelly M., Mandel R.J., Nguyen M., Trono D., Naldini L. A third-generation lentivirus vector with a conditional packaging system. J. Virol. 1998;72:8463–8471. doi: 10.1128/jvi.72.11.8463-8471.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zufferey R., Dull T., Mandel R.J., Bukovsky A., Quiroz D., Naldini L., Trono D. Self-inactivating lentivirus vector for safe and efficient in vivo gene delivery. J. Virol. 1998;72:9873–9880. doi: 10.1128/jvi.72.12.9873-9880.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Seppen J., Rijnberg M., Cooreman M.P., Oude Elferink R.P. Lentiviral vectors for efficient transduction of isolated primary quiescent hepatocytes. J. Hepatol. 2002;36:459–465. doi: 10.1016/s0168-8278(01)00308-7. [DOI] [PubMed] [Google Scholar]
- 18.Gould D.J., Berenstein M., Dreja H., Ledda F., Podhajcer O.L., Chernajovsky Y. A novel doxycycline inducible autoregulatory plasmid which displays ‘on’/‘off’ regulation suited to gene therapy applications. Gene Ther. 2000;7:2061–2070. doi: 10.1038/sj.gt.3301354. [DOI] [PubMed] [Google Scholar]
- 19.Strathdee C.A., McLeod M.R., Hall J.R. Efficient control of tetracycline-responsive gene expression from an autoregulated bi-directional expression vector. Gene. 1999;229:21–29. doi: 10.1016/s0378-1119(99)00045-1. [DOI] [PubMed] [Google Scholar]
- 20.Chtarto A., Bender H.U., Hanemann C.O., Kemp T., Lehtonen E., Levivier M., Brotchi J., Velu T., Tenenbaum L. Tetracycline-inducible transgene expression mediated by a single AAV vector. Gene Ther. 2003;10:84–94. doi: 10.1038/sj.gt.3301838. [DOI] [PubMed] [Google Scholar]
- 21.Hofmann A., Nolan G.P., Blau H.M. Rapid retroviral delivery of tetracycline-inducible genes in a single autoregulatory cassette. Proc. Natl Acad. Sci. USA. 1996;93:5185–5190. doi: 10.1073/pnas.93.11.5185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kuhnel F., Fritsch C., Krause S., Mundt B., Wirth T., Paul Y., Malek N.P., Zender L., Manns M.P., Kubicka S. Doxycyline regulation in a single retroviral vector by an autoregulatory loop facilitates controlled gene expression in liver cells. Nucleic Acids Res. 2004;32:e30. doi: 10.1093/nar/gnh034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mohammadi S., Hawkins R.E. Efficient transgene regulation from a single tetracycline-controlled positive feedback regulatory system. Gene Ther. 1998;5:76–84. doi: 10.1038/sj.gt.3300557. [DOI] [PubMed] [Google Scholar]
- 24.Shockett P., Difilippantonio M., Hellman N., Schatz D.G. A modified tetracycline-regulated system provides autoregulatory, inducible gene expression in cultured cells and transgenic mice. Proc. Natl Acad. Sci. USA. 1995;92:6522–6526. doi: 10.1073/pnas.92.14.6522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Unsinger J., Kroger A., Hauser H., Wirth D. Retroviral vectors for the transduction of autoregulated, bidirectional expression cassettes. Mol. Ther. 2001;4:484–489. doi: 10.1006/mthe.2001.0480. [DOI] [PubMed] [Google Scholar]
- 26.Unsinger J., Lindenmaier W., May T., Hauser H., Wirth D. Stable and strictly controlled expression of LTR-flanked autoregulated expression cassettes upon adenoviral transfer. Biochem. Biophys. Res. Commun. 2004;319:879–887. doi: 10.1016/j.bbrc.2004.05.067. [DOI] [PubMed] [Google Scholar]
- 27.Verhoef K., Marzio G., Hillen W., Bujard H., Berkhout B. Strict control of human immunodeficiency virus type 1 replication by a genetic switch: Tet for Tat. J. Virol. 2001;75:979–987. doi: 10.1128/JVI.75.2.979-987.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Li X., Zhao X., Fang Y., Jiang X., Duong T., Fan C., Huang C.C., Kain S.R. Generation of destabilized green fluorescent protein as a transcription reporter. J. Biol. Chem. 1998;273:34970–34975. doi: 10.1074/jbc.273.52.34970. [DOI] [PubMed] [Google Scholar]
- 29.Seppen J., Barry S.C., Klinkspoor J.H., Katen L.J., Lee S.P., Garcia J.V., Osborne W.R. Apical gene transfer into quiescent human and canine polarized intestinal epithelial cells by lentivirus vectors. J. Virol. 2000;74:7642–7645. doi: 10.1128/jvi.74.16.7642-7645.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gallia G.L., Khalili K. Evaluation of an autoregulatory tetracycline regulated system. Oncogene. 1998;16:1879–1884. doi: 10.1038/sj.onc.1201706. [DOI] [PubMed] [Google Scholar]
- 31.Mizuguchi H., Xu Z., Ishii-Watabe A., Uchida E., Hayakawa T. IRES-dependent second gene expression is significantly lower than cap-dependent first gene expression in a bicistronic vector. Mol. Ther. 2000;1:376–382. doi: 10.1006/mthe.2000.0050. [DOI] [PubMed] [Google Scholar]
- 32.Shimizu Y., Demetris A.J., Gollin S.M., Storto P.D., Bedford H.M., Altarac S., Iwatsuki S., Herberman R.B., Whiteside T.L. Two new human cholangiocarcinoma cell lines and their cytogenetics and responses to growth factors, hormones, cytokines or immunologic effector cells. Int. J. Cancer. 1992;52:252–260. doi: 10.1002/ijc.2910520217. [DOI] [PubMed] [Google Scholar]
- 33.Kafri T., van Praag H., Gage F.H., Verma I.M. Lentiviral vectors: regulated gene expression. Mol. Ther. 2000;1:516–521. doi: 10.1006/mthe.2000.0083. [DOI] [PubMed] [Google Scholar]
- 34.Vigna E., Cavalieri S., Ailles L., Geuna M., Loew R., Bujard H., Naldini L. Robust and efficient regulation of transgene expression in vivo by improved tetracycline-dependent lentiviral vectors. Mol. Ther. 2002;5:252–261. doi: 10.1006/mthe.2002.0542. [DOI] [PubMed] [Google Scholar]
- 35.Haack K., Cockrell A.S., Ma H., Israeli D., Ho S.N., McCown T.J., Kafri T. Transactivator and structurally optimized inducible lentiviral vectors. Mol. Ther. 2004;10:585–596. doi: 10.1016/j.ymthe.2004.06.109. [DOI] [PubMed] [Google Scholar]
- 36.Vogel R., Amar L., Thi A.D., Saillour P., Mallet J. A single lentivirus vector mediates doxycycline-regulated expression of transgenes in the brain. Hum. Gene Ther. 2004;15:157–165. doi: 10.1089/104303404772679968. [DOI] [PubMed] [Google Scholar]