Abstract
Background
Heart failure is a common condition with considerable associated costs, morbidity, and mortality. Patients often present to hospital with dyspnea and edema. Inadequate inpatient decongestion is an important contributor to high readmission rates. There is little evidence concerning diuresis to guide clinicians in caring for patients with acute decompensated heart failure. Contemporary diuretic strategies have been defined by expert opinion and older landmark clinical trials.
Objective
To present a narrative review of contemporary recommendations, along with their underlying evidence and pharmacologic rationale, for diuretic strategies in inpatients with acute decompensated heart failure.
Data Sources
PubMed, OVID, and Embase databases were searched from inception to December 22, 2022, with the following search terms: heart failure, acute heart failure, decompensated heart failure, furosemide, bumetanide, ethacrynic acid, hydrochlorothiazide, indapamide, metolazone, chlorthalidone, spironolactone, eplerenone, and acetazolamide.
Study Selection
Randomized controlled trials and systematic reviews involving at least 100 adult patients (> 18 years) were included. Trials involving torsemide, chlorothiazide, and tolvaptan were excluded.
Data Synthesis
Early, aggressive administration of a loop diuretic has been associated with expedited symptom resolution, shorter length of stay, and possibly reduced mortality. Guidelines make recommendations about dose and frequency but do not recommend any particular loop diuretic over another; however, furosemide is most commonly used. Guidelines recommend that the initial furosemide dose (on admission) be 2–2.5 times the patient’s home dose. A satisfactory diuretic response can be defined as spot urine sodium content greater than 50–70 mmol/L at 2 hours; urine output greater than 100–150 mL/h in the first 6 hours or 3–5 L in 24 hours; or a change in weight of 0.5–1.5 kg in 24 hours. If congestion persists after the maximization of loop diuretic therapy over the first 24–48 hours, an adjunctive diuretic such as thiazide or acetazolamide should be added. If decongestion targets are not met, continuous infusion of furosemide may be considered.
Conclusions
Heart failure with congestion can be managed with careful administration of high-dose loop diuretics, supported by thiazides and acetazolamide when necessary. Clinical trials are underway to further evaluate this strategy.
Keywords: heart failure, acute heart failure, decompensated heart failure, furosemide, bumetanide, ethacrynic acid, hydrochlorothiazide, indapamide, metolazone, chlorthalidone, spironolactone, eplerenone, acetazolamide
RÉSUMÉ
Contexte
L’insuffisance cardiaque est une maladie courante entraînant des coûts, une morbidité et une mortalité considérables. Les patients se présentent souvent à l’hôpital avec une dyspnée et un oedème. Une décongestion inadéquate des patients hospitalisés contribue largement aux taux élevés de réadmission. Il existe peu de données probantes concernant la diurèse pour guider les cliniciens dans la prise en charge des patients atteints d’insuffisance cardiaque aiguë décompensée. Les stratégies diurétiques contemporaines ont été définies par l’opinion d’experts et des essais cliniques de référence plus anciens.
Objectif
Présenter une revue narrative des recommandations contemporaines, ainsi que leurs données probantes sous-jacentes et leur justification pharmacologique, pour les stratégies diurétiques chez les patients hospitalisés souffrant d’insuffisance cardiaque aiguë décompensée.
Sources des données
Les bases de données PubMed, OVID et Embase ont été consultées depuis leur création jusqu’au 22 décembre 2022, avec les termes de recherche suivants: insuffisance cardiaque, insuffisance cardiaque aiguë, insuffisance cardiaque décompensée, furosémide, bumétanide, acide éthacrynique, hydrochlorothiazide, indapamide, métolazone, chlorthalidone, spironolactone, éplérénone et acétazolamide.
Choix de l’étude
Les essais contrôlés randomisés et les revues systématiques portant sur au moins 100 patients adultes (plus de 18 ans) ont été inclus. Les essais impliquant le torsémide, le chlorothiazide et le tolvaptan ont été exclus.
Synthèse des données
L’administration précoce et agressive d’un diurétique de l’anse a été associée à une résolution accélérée des symptômes, à une durée de séjour plus courte et éventuellement à une mortalité réduite. Les lignes directrices font des recommandations sur la dose et la fréquence, mais ne recommandent pas un diurétique de l’anse particulier plutôt qu’un autre; cependant, le furosémide est le plus couramment utilisé. Les lignes directrices recommandent que la dose initiale de furosémide à l’admission soit de 2 à 2,5 fois la dose à domicile du patient. Une réponse diurétique satisfaisante peut être définie comme une teneur ponctuelle en sodium dans l’urine supérieure à 50 à 70 mmol/L après 2 heures; débit urinaire supérieur à 100 à 150 mL/h au cours des 6 premières heures ou à 3 à 5 L en 24 heures; ou un changement de poids de 0,5 à 1,5 kg en 24 heures. Si la congestion persiste après la maximisation du traitement par diurétique de l’anse au cours des premières 24 à 48 heures, un diurétique d’appoint tel que le thiazidique ou l’acétazolamide doivent être ajoutés. Si les objectifs de décongestion ne sont pas atteints, une perfusion continue de furosémide peut être envisagée.
Conclusions
L’insuffisance cardiaque accompagnée de congestion peut être gérée par l’administration prudente de diurétiques de l’anse à haute dose, appuyés par des thiazidiques et de l’acétazolamide si nécessaire. Des essais cliniques sont en cours pour évaluer davantage cette stratégie.
Mots-clés: insuffisance cardiaque, insuffisance cardiaque aiguë, insuffisance cardiaque décompensée, furosémide, bumétanide, acide éthacrynique, hydrochlorothiazide, indapamide, métolazone, chlorthalidone, spironolactone, acétazolamide, éplérénone
INTRODUCTION
Heart failure (HF) is a common, costly, and mortal condition. HF currently affects approximately 600 000 Canadians and is a leading cause of hospital admission, which is a major driver of the $2.8 billion dollars per year in direct health care costs spent on this disease.1 Despite important advances in diagnosis and treatment, Canadians hospitalized with HF have readmission rates in excess of 20% at 30 days and mortality rates upward of 30% at 1 year.1 At age 40, the lifetime risk of incident HF is 20%,2 and HF is the most common reason for hospital presentation in individuals over 65 years of age.2 Congestion in HF is defined as signs and symptoms of extracellular fluid accumulation resulting in increased extra-cardiac filling pressures.3 HF with increased neurohormonal activation and sympathetic output leads to increased circulatory volume.3 Symptoms of congestion, which include dyspnea and edema, are the most frequent presenting symptoms among patients presenting with HF.4 Most patients with chronic HF require a maintenance dose of oral loop diuretic to maintain euvolemia and clinical stability.5
Inadequate resolution of symptoms (specifically decongestion) before discharge is an important contributor to high rates of readmission for HF.6 Decongestion is achieved primarily with diuretic therapy, although vasodilators may be used as auxiliary agents.3,7–9 Prompt administration of IV loop diuretics in the emergency department (ED) for patients with acute decompensated HF (ADHF) was associated with reduced inpatient mortality in a prospective multicentre observational cohort.10 Patients for whom complete decongestion is achieved in hospital have improved outcomes. 6,11 Controlled trials with diuretics have shown their effects in increasing urinary sodium excretion and decreasing physical signs of fluid retention, as well as improving symptoms, quality of life, and exercise tolerance.9
Current guidelines recommend IV loop diuretics for patients presenting to the ED with ADHF (Table 1).7–9 Early, aggressive administration of loop diuretics has been associated with accelerated symptom resolution, shorter length of stay,10 and possibly reduced mortality.8 The diuretic response should be evaluated shortly after the start of diuretic therapy. A satisfactory diuretic response can be defined as spot urine sodium content greater than 50–70 mmol/L at 2 hours; urine output greater than 100–150 mL/h during the first 6 hours or 3–5 L in 24 hours; or a change in weight of 0.5–1.5 kg in 24 hours.7,8 If markers of adequate diuresis and natriuresis are not met, a second dose, at double the initial dose, should be administered promptly.7–9 After loop diuretics have been maximized, thiazide diuretics or acetazolamide may be added to augment diuresis.5 If congestion persists, then a continuous infusion of furosemide may be considered.
TABLE 1.
Summary of Guidelines on Use of Diuretics for Decongestion in Hospitalized Patients with Heart Failure
| Recommendation | Strength of Recommendation | Quality of Evidence |
|---|---|---|
| CCS guidelines for management of heart failure (2017)7 | ||
| IV diuretics should be given as first-line therapy for patients with pulmonary or peripheral congestion | Strong | Low |
| Furosemide should be dosed intermittently (e.g., twice daily) or by continuous infusion | Strong | Moderate |
|
| ||
| AHA/ACC/HFSA guideline for management of heart failure (2022)9 | ||
| Patients with heart failure admitted with evidence of significant fluid overload should be promptly treated with IV loop diuretics to improve symptoms and reduce mortality | Strong | Moderate |
| For patients admitted with heart failure, therapy with diuretics and other guideline-directed medications should be titrated with the goal of resolving clinical evidence of congestion to reduce both symptoms and readmissions | Strong | Moderate |
| For patients admitted with heart failure and for whom diuresis is inadequate to relieve symptoms and signs of congestion, it is reasonable to intensify the diuretic regimen by either using a higher dose of IV loop diuretic or adding a second agent | Moderate | Moderate |
| For patients requiring diuretic treatment during hospitalization for heart failure, the discharge regimen should include a plan for adjustment of diuretics to decrease readmissions | Strong | Moderate |
|
| ||
| ESC guidelines for diagnosis and treatment of acute and chronic heart failure (2021)8 | ||
| IV loop diuretics are recommended for all patients with acute heart failure admitted with signs or symptoms of fluid overload, to improve symptoms | Strong | Low |
| Combination of a loop diuretic with thiazide-type diuretic should be considered for patients with resistant edema and no response to an increase in dose of loop diuretic | Moderate | Moderate |
ACC = American College of Cardiology, AHA = American Heart Association, CCS = Canadian Cardiovascular Society, ESC = European Society of Cardiology, HFSA = Heart Failure Society of America.
There is a paucity of high-quality evidence to inform diuretic practices for inpatients with ADHF. Contemporary diuretic strategies have been developed on the basis of expert opinion3,5 and several older landmark clinical trials.12,13 This dearth of evidence has come under recent scrutiny, and interest in its remedy has been renewed.5,14 The purpose of this study was to review the pharmacology and pharmacokinetics of diuretics and the evidence for diuretic strategies in inpatients with ADHF.
METHODS
The following 4 types of diuretics were considered in this review: loop diuretics, thiazide diuretics, carbonic anhydrase inhibitors, and mineralocorticoid receptor antagonists (MRAs). Only studies involving medications and formulations available in Canada, as determined through Health Canada’s Drug Product Database, were eligible for inclusion. The diuretics reviewed were furosemide, bumetanide, ethacrynic acid, hydrochlorothiazide, indapamide, metolazone, chlorthalidone, spironolactone, eplerenone, and acetazolamide. The PubMed, OVID, and Embase databases were searched from inception to December 22, 2022, with the following search terms: heart failure, acute heart failure, decompensated heart failure, furosemide, bumetanide, ethacrynic acid, hydrochlorothiazide, indapamide, metolazone, chlorthalidone, spironolactone, eplerenone, and acetazolamide. Abstracts and titles were screened, and reference lists were reviewed for additional pertinent articles. Only randomized controlled trials and systematic reviews involving adult patients (> 18 years of age) were eligible for inclusion. Trials with fewer than 100 patients were excluded. Trials involving torsemide and chlorothiazide were also excluded, because these drugs are not available in Canada. Trials evaluating tolvaptan were excluded, because use of this drug should be limited to cases of volume overload with significant hyponatremia when other measures have failed.7 This article presents a narrative review of the literature.
RESULTS
The search yielded 5 randomized controlled trials and 5 systematic reviews that met the inclusion criteria. No randomized controlled trials or systematic reviews for the use of ethacrynic acid in HF met the inclusion criteria. Similarly, no randomized controlled trials compared diuretics with placebo in patients with ADHF.
Loop Diuretics
Loop diuretics block the Na–K–2Cl symporter in the luminal membrane of the thick ascending limb of the loop of Henle.15 Loop diuretics have steep dose–response curves, which means there is little effect until a threshold is reached, beyond which the response rapidly approaches a maximum or ceiling.5,14 Although this pattern is true of natriuretic efficiency, increasing the dose above the ceiling can cause additional natriuresis by increasing the time during which the plasma diuretic concentration exceeds the natriuretic threshold.5,14 Table 2 summarizes the pharmacokinetics of loop diuretics.5,16–19 With more avid sodium retention, as in ADHF, a higher peak drug level may be required, and IV administration may be more effective than oral administration. 5,14 Although gut edema and low duodenal blood flow do not typically affect oral bioavailability (the amount absorbed relative to the amount ingested), they slow absorption, thereby reducing peak plasma levels and contributing to diuretic resistance.5,14 The absorption of furosemide is slower than its elimination half-life, a phenomenon known as “absorption-limited” or “flip-flop” kinetics; the mean bioavailability of this drug is 50%, but absorption is quite variable and may be influenced by food intake.5 Ethacrynic acid and bumetanide have greater oral bioavailability and undergo hepatic metabolism.
TABLE 2.
Pharmacokinetic and Pharmacodynamic Properties of Loop Diuretics
| Property | Furosemide | Ethacrynic Acid | Bumetanide |
|---|---|---|---|
| Relative potency | 1 | 0.7 | 40 |
|
| |||
| Equivalent oral dose (mg)a | 40 | 50 | 1 |
|
| |||
| PO to IV conversion | 2:1 | 1:1 | 1:1 |
|
| |||
| Bioavailability (%) | Range 10–100 (mean 50) | 100 | Range 80–100 |
|
| |||
| Onset (min) | |||
| Oral | 30–60 | 30 | 30–60 |
| IV | 5 | 5 | 5 |
|
| |||
| Time to peak serum concentration after PO administration (h) | 1–2 | 2 | 1–2 |
|
| |||
| Protein binding (%) | 91–99 | 90–95 | 95 |
|
| |||
| Clearance | Urine: 50%–80% Minimal hepatic metabolism | Feces and urine: 30%–60% as unchanged drug Hepatic: 35%–40% | Renal: 81% of total dose, 45% of which is unchanged drug Hepatic: 50% |
|
| |||
| Half-life (h) | 0.5–2 | 2–4 | 1–1.5 |
|
| |||
| Average duration of action (h) | |||
| Oral administration | 6–8 | 12 | 4–6 |
| IV administration | 2 | 2–4 | 2–3 |
|
| |||
| Maximum daily dose (mg) | 600 | 400 | 10 |
Equivalent IV dose: furosemide 40 mg, bumetanide 1 mg (unable to find equivalent dose of ethacrynic acid).
Furosemide and bumetanide are sulfonamide loop-type diuretics, whereas ethacrynic acid is a derivative of aryloxyacetic acid.16 Therefore, ethacrynic acid is an alternative for patients with allergy to furosemide or bumetanide.20,21 The risk of permanent hearing loss or deafness with ethacrynic acid contributes to its limited clinical use.22 Guidelines make recommendations on dose and frequency but do not recommend any particular loop diuretic over another.7–9 However, furosemide is the most commonly administered loop diuretic in patients with HF.5,23 Perhaps its popularity is based upon collective experience and longevity, given that it was the first loop diuretic approved by Health Canada, in 1966.
Loop diuretics are the most potent diuretics, resulting in fractional excretion of sodium (FENA)—a standard measure of diuretic potency—of 20%–25% in healthy volunteers. 15,24 After a period of natriuresis, loop diuretics result in a period of enhanced renal sodium avidity, referred to as rebound sodium reabsorption.15 Depending on the loop diuretic type, dose, and frequency, rebound sodium reabsorption can be of sufficient magnitude to completely offset the natriuresis induced by a given dose of loop diuretic.15,24 To minimize the impact of rebound sodium reabsorption in patients with ADHF, loop diuretics should be administered at least twice daily.5,15
Over repeated doses, loop diuretics lose their natriuretic potency. As a result, the FENA in HF patients receiving long-term loop diuretic therapy decreases from 20%–25% to 10%–15%.24 This sequential loss in potency is referred to as the “braking effect”.15 Over the short term, it is mediated by transient decreases in intravascular volume. Reduced intravascular volume reduces renal perfusion and sodium delivery to the glomerulus and activates the renin–angiotensin–aldosterone and sympathetic nervous systems. 24 This neurohormonal activation increases sodium reabsorption, thereby reducing natriuresis.5 Over the longer term, loss of loop diuretic potency results from distal tubular hypertrophy and increased NaCl symporter expression with subsequent increases in NaCl reabsorption.15
Guidelines rely on the Diuretic Optimization Strategy Evaluation (DOSE) study to direct recommendations on initial loop diuretic dose. The DOSE study12 compared low-versus high-dose IV furosemide strategies in recently admitted patients with ADHF (Table 3). Low-dose therapy was defined as an IV dose equivalent to the patient’s home oral dose (in milligrams), and high-dose therapy was defined as an IV dose equivalent to 2.5 times the patient’s home oral dose (in milligrams).12 For example, patients receiving furosemide 40 mg PO twice daily at home would be given 40 mg IV twice daily or 100 mg IV twice daily in the low- and high-dose groups, respectively. There were no differences in the primary outcome of global assessment of symptoms at 72 hours.12 However, patients in the high-dose group experienced significantly greater urine output, weight loss, and relief of dyspnea without a concomitant decrease in glomerular filtration rate.12 There was no difference between the treatment arms in terms of worsening or persistent HF or treatment failure. On the basis of this study, guidelines recommend initial furosemide dosing on admission at 2 to 2.5 times the previous home dose, with diuretic-naive patients receiving a lower dose (20–40 mg). The DOSE trial had several notable limitations. All of the patients had chronic HF and required approximately 80 to 240 mg/day of furosemide or the equivalent amount of other loop diuretics. As a result, the findings may not be applicable to patients with newly diagnosed HF, and the trial was not powered to detect differences in clinical events. The median time from presentation to randomization was approximately 15 hours, by which time many patients had already received diuretic therapy, and the trial also allowed for adjustments in diuretic dosage 48 hours after randomization. These factors may have influenced the trial outcomes.
TABLE 3.
Summary of Randomized Double-Blind Trials of Diuretics in Treatment of Acute Heart Failure
| Study | Trial Design | Sample Size | Drug, Dose, and Duration | Primary Outcome | Resultsa | Adverse Events |
|---|---|---|---|---|---|---|
| Felker et al. (2011)12 | R, DB, C | 308 | Furosemide by low-dose strategy (total daily IV furosemide dose equal to patient’s total daily oral dose of loop diuretic) or high-dose strategy (total daily IV furosemide dose 2.5 times patient’s total daily dose of oral loop diuretic in furosemide equivalents); administration either IV q12h or as continuous infusion for 3 days | Patient’s global assessment of symptoms Change in serum creatinine from baseline at 72 h |
Median loop diuretic received over 72 h (in IV furosemide equivalents): Low dose 285 (IQR 200 to 480) mg vs high dose 688 (IQR 429 to 1067) mg, p < 0.0001b IV q12h 518 (IQR 292 to 832) mg vs continuous infusion 406 (IQR 240 to 628) mg, p = 0.008 Global assessment of symptomsc (mean ± SD): Low dose AUC 4171 ± 1436 vs high dose 4430 ± 1401, p = 0.06 Bolus 4236 ± 1440 vs continuous infusion 4373 ± 1404, p = 0.47 Change in serum creatinine (mean ± SD): Low dose 3.5 ± 26.5 μmol/L vs high dose 7.1 ± 26.5 μmol/L, p = 0.21 Bolus 4.4 ± 26.5 μmol/L vs continuous 6.2 ± 26.5 μmol/L, p = 0.45 AUC for dyspnea at 72 h (mean ± SD): Low dose 4478 ± 1550 vs high dose 4668 ± 1496, p = 0.04 Bolus 4456 ± 1468 vs continuous 4699 ± 1573, p = 0.36 Change in weight (mean ± SD): Low dose −2.8 ± 4.3 kg vs high dose −4.0 ± 3.9 kg, p = 0.01 Bolus −3.1 ± 3.6 kg vs continuous −3.7 ± 4.7 kg, p = 0.20 Net fluid loss (mean ± SD): Low dose 3575 ± 2635 mL vs high dose 4899 ± 3479 mL, p = 0.001 |
Hypokalemiad: Low dose 1% vs high dose 1% Bolus 1% vs continuous infusion 1% Renal failured: Low dose 9% vs high dose 4% Bolus 5% vs continuous 8% |
| Butler et al. (2017)25 | R, DB, PC | 360 | Spironolactone 100 mg PO daily for 96 he | Change in log NT-proBNP | Median furosemide equivalent dose: spironolactone 80 (IQR 40 to 200) mg vs usual care 80 (IQR 40 to 240) mg, p = 0.77 Median log NT-proBNP: spironolactone 7.89 (IQR 7.19 to 8.68) vs usual care 7.64 (IQR 6.93 to 8.45), p = 0.57 Median dyspnea score: spironolactone 80 (IQR 65 to 90) vs usual care 83 (IQR 70 to 90), p = 0.61 Median clinical congestion score: spironolactone 4 (IQR 2 to 7) vs usual care 4 (IQR 2 to 6), p = 0.41 Median urine output: spironolactone 6086 mL (IQR 2780 to 8420) vs usual care 5584 mL (IQR 2924 to 8132), p = 0.57 Median weight change: spironolactone −3.3 (−5.9 to −0.9) kg vs usual care −2.8 (−5.1 to −0.8) kg, p = 0.33 Median worsening heart failure in hospital: spironolactone 19% vs usual care 18%, p = 0.76 |
Median change in serum potassium: spironolactone 0.30 (IQR 0.00 to 0.70) mmol/L vs usual care 0.20 (IQR −0.30 to 0.60) mmol/L, p = 0.08 Median change in serum creatinine: spironolactone 8.8 (IQR −4.4 to 23.9) μmol/L vs usual care 8.8 (IQR −1.8 to 29.2) μmol/L, p = 0.77 Median change in estimated GFR: spironolactone −4.34 (IQR −11.06 to 1.74) mL/min vs usual care −5.53 (IQR −13.11 to 0.79) mL/min, p = 0.56 No differences observed between groups in terms of changes in heart rate or blood pressure |
| Trullàs et al. (2023)26 | R, DB, PC | 230 | Hydrochlorothiazide 25– 100 mg PO daily × 5 daysf | Change in body weight Change in patient-reported dyspnea |
Median dose of furosemide in each treatment arm: Day 1, 80 (IQR 80 to 120) mg Day 5, 60 (IQR 40 to 80) mg Change in body weight: hydrochlorothiazide −2.3 kg vs placebo −1.5 kg; adjusted estimated difference −1.14 (95% CI −1.84 to −0.42) kg, p = 0.002 Mean patient-reported dyspneac: hydrochlorothiazide 960 (range 360 to 1620) vs placebo 720 (range 240 to 1455), p = 0.497 24-h urine output: hydrochlorothiazide 1775 mL vs placebo 1400 mL, p = 0.05 90-day mortality: hydrochlorothiazide 19 vs placebo 23; HR 1.26 (95% CI 0.68 to 2.34), p = 0.46 90-day readmission: hydrochlorothiazide 40 vs placebo 43; HR 1.25 (95% CI 0.81 to 1.93), p = 0.32 |
Median increase in serum creatinine at day 5: hydrochlorothiazide 15.9 (IQR 7.1 to 37.1) μmol/L vs placebo 0.00 (IQR 10.6 to 18.6) μmol/L, p < 0.001 Median decrease in serum potassium: hydrochlorothiazide −0.70 (95% CI −0.81 to −0.60) mmol/L vs placebo −0.36 (95% CI −0.46 to −0.26) mmol/Ld Median decrease in serum sodium: hydrochlorothiazide −3.4 (95% CI −4.0 to −2.5) mmol/L vs placebo −2.6 (95% CI −3.5 to −2.0) mmol/L d |
| Mullens et al. (2022)27 | R, DB, PC | 519 | Acetazolamide 500 mg IV daily or placebo for 3 days,g added to standardized IV loop diuretics (at dose equivalent to twice the oral dose), administered twice daily | Successful decongestionh | Median dose of furosemide administered: Days 1 and 2, 120 (IQR 80 to 200) mg in both treatment arms Day 3, acetazolamide arm 80 (IQR 0 to 200) mg vs placebo arm 120 (IQR 80 to 240) mg Successful decongestion: acetazolamide 42.2% vs placebo 30.5%; RR 1.46 (95% CI 1.17 to 1.82), p < 0.001 Death from any cause or readmission for heart failure: acetazolamide 29.7% vs placebo 27.8%; HR 1.07 (95% CI 0.78 to 1.48)d |
Adverse events, as no. of patients affected: Doubling of serum creatinine from baseline: acetazolamide 2 vs placebo 0, p = 0.24 ≥ 50% sustained decrease in estimated GFR: acetazolamide 4 vs placebo 1, p = 0.21 Renal replacement therapy during hospitalization: acetazolamide 4 vs placebo 1, p = 0.21 Hypokalemia: acetazolamide 14 vs placebo 10, p = 0.39 Hypotension: acetazolamide 17 vs placebo 9, p = 0.11 |
| Asakura et al. (2022)28 | R, DB, PC | 300 | Eplerenone 25 mg PO daily for 6 monthsi | Composite of cardiac death or first hospitalization due to any cardiovascular disease | Composite outcome: eplerenone 19.5% vs placebo 17.2%; HR 1.09 (95% CI 0.64 to 1.9)d | Hyperkalemia: eplerenone 4.1% vs placebo 2.7%, p = 0.74 Renal impairment: eplerenone 6.8% vs placebo 6.0%, p = 0.99 Hypotension: eplerenone 2% vs placebo 0%, p = 0.25 |
AUC = area under the curve, C = controlled, CI = confidence interval, DB = double blind, GFR = glomerular filtration rate, HR = hazard ratio, IQR = interquartile range, NT-proBNP = N-terminal prohormone B type natriuretic peptide, PC = placebo-controlled, R = randomized, RR = risk ratio, SD = standard deviation.
Secondary outcomes are reported if they pertain to decongestion parameters, mortality, or readmission for heart failure.
Thiazide diuretic was added during the 72-hour treatment period more frequently in the group with q12h administration than the group with continuous infusion (16% vs 7%, p = 0.02); there was no significant difference in addition of thiazide diuretic between the low-dose and high-dose groups (15% vs 8%, p = 0.06).
Visual analogue scale reported and quantified as AUC of serial assessments.
Statistical significance not reported.
Patients taking spironolactone before admission were randomly assigned to receive 100 mg or 25 mg per day in the usual care arm.
Hydrochlorothiazide dose was adjusted according to GFR, as follows: if GFR > 50 mL/min, dose 25 mg PO daily; if GFR 20–50 mL/min, dose 50 mg PO daily; if GFR < 20 mL/min, dose 100 mg PO daily.
All patients received a maintenance infusion of 500 mL 5% dextrose and 3 g magnesium sulfate over 24 hours until the end of the treatment phase. Use of oral acetazolamide was not recommended after completion of the treatment phase.
Defined as absence of volume overload (no more than trace edema, no residual pleural effusion, no residual ascites).
Dose was increased to 50 mg PO daily after 1 week, provided serum potassium level was not greater than 5 mmol/L.
Continuous infusion of loop diuretics is believed to confer additional benefits over bolus injection, with less variability in peak plasma concentration (which consistently results in persistent urine output) and less risk of electrolyte disturbance. 13,29–32 Continuous infusion has also been suggested to allow more consistent delivery of drug to the nephron, leading to more efficient diuresis, preventing rebound sodium retention and fluid reabsorption, and causing less neurohormonal activation, but it may also be associated with hypotension and acute kidney injury.13,29–32 Five systematic reviews of intermittent bolus versus continuous infusion of furosemide to treat fluid overload in acute HF were analyzed here (Table 4). This literature suggests that continuous infusion led to greater weight loss and urine output, but the clinical significance was small.13,29–32 There were no significant changes in electrolyte disturbances, serum creatinine, or hypotension in either treatment arm. Various limitations of the studies included in these systematic reviews were noted, such as small sample sizes, heterogeneity of patients, crossover design without sufficient wash-out period, differing diuretic dosages and duration, differences in the use of concomitant medications, differences in reporting of adverse events, and differences in primary outcomes.13,29–32 In addition to these systematic reviews of bolus versus continuous infusion, a retrospective analysis of the ASCEND-HF trial, which involved 5738 patients with stable diuretic therapy in the first 24 hours, was conducted.33 This analysis showed that continuous infusion was associated with greater weight loss, greater urine output, greater change in renal function, and worsening HF. However, the continuous infusion arm consisted of a sicker population, for whom higher diuretic dosages were required.
TABLE 4.
Summary of Systematic Reviews of Intermittent Bolus versus Continuous Infusion of Loop Diuretics
| Study | No. of Trials | Total No. of Patients | Primary Outcomes | Resultsa | Adverse Events | Outcome |
|---|---|---|---|---|---|---|
| Salvador et al. (2005)13 | 8 | 254 | Urine output Resolution of heart failure symptoms Length of hospital stay All-cause mortality |
Urine output: WMD 271 mL (95% CI 93.1 to 449), p < 0.01 Resolution of heart failure symptoms: mean 6.6 hours (95% CI −4 to 17), p = 0.56 Length of hospital stay: −3.1 days (95% CI −4.1 to −2), p < 0.01 All-cause mortality: RR 0.52 (95% CI 0.38 to 0.71), p < 0.01 |
Hypokalemia, hypomagnesemia: RR 1.47 (95% CI 0.52 to 4.15), p = 0.50 Increase in serum creatinine: WMD −0.54 (95% CI −0.57 to −0.51), p < 0.01 Tinnitus and hearing loss: RR 0.06 (95% CI 0.01 to 0.44), p < 0.05 |
Greater urine output with continuous infusion Shorter length of hospital stay with continuous infusion Increase in serum creatinine greater with bolus injection Less tinnitus and hearing loss with continuous infusion |
| Wu et al. (2014)30 | 10 | 518 | Urine output Weight loss Increase in serum creatinine Electrolyte abnormalities Length of hospital stay All-cause mortality |
Urine output: WMD 111 (95% CI −338 to 560) mL, p = 0.63b Weight loss: WMD 0.78 (95% CI 0.03 to 1.54) kg, p = 0.04 Length of hospital stay: WMD −1.06 (95% CI −3.88 to 1.76) days, p = 0.46 All-cause mortality: RR 1.13 (95% CI 0.61 to 2.10), p = 0.70 |
Hypokalemia: RR 0.85 (95% CI 0.32 to 2.24), p = 0.74 Hypomagnesemia: RR 0.54 (95% CI 0.12 to 2.40), p = 0.42 Change in creatinine: WMD 0 (95% CI −0.09 to 0.09), p = 0.96 Tinnitus or hearing loss: RR 0.09 (95% CI 0.01 to 1.54)c |
Greater weight loss with continuous infusion |
| Ng and Yap (2018)31 | 8 | 669 | All-cause mortality Length of hospital stay |
All-cause mortality: OR 1.65 (95% CI 0.9 to 2.91), p = 0.08d Length of hospital stay: mean difference 0.27 (95% CI −1.35 to 1.89) day, p = 0.74 Secondary outcomes Change in serum creatinine: mean difference 40.7 (95% CI 22.1 to 103.4) μmol/L, p = 0.20 Change in weight: mean difference 0.70 (95% CI 0.12 to 1.28) kg, p = 0.02 Urine output: mean difference −36.6 (95% CI −663 to 590) mL, p = 0.91 Reduction in BNP: mean difference 400 (95% CI 153 to 646) ng/L, p < 0.01 |
Hypokalemia: mean difference 1.0 (95% CI 0.20 to 5.40) mmol/L, p = 0.97 Change in creatinine: mean difference 103 (95% CI 62 to 168) μmol/L, p = 0.57 |
Greater reduction in weight and BNP with continuous infusion |
| Kuriyama and Urushidani (2019)32 | 12 | 923 | All-cause mortality Length of hospital stay Weight loss |
All-cause mortality: RR 1.19 (95% CI 0.65 to 2.16), p = 0.58 Length of hospital stay: WMD −0.88 (95% CI −2.76 to 1.01) day, p = 0.36 Weight loss: WMD 0.63 (95% CI 0.23 to 1.02) kg, p = 0.002 Secondary outcome Urine output: WMD −37 (95% CI −336 to 387) mL, p = 0.012 |
Hypokalemia: RR 1.41 (95% CI 0.51 to 3.86), p = 0.51 Hyponatremia: RR 1.45 (95% CI 0.75 to 2.80), p = 0.27 Increase in serum creatinine: RR 1.20 (95% CI 0.85 to 1.69), p = 0.30 Hypotension: RR 0.95 (95% CI 0.48 to 1.88), p = 0.88 |
Greater weight loss and small increase in urine output with continuous infusion |
| Chan et al. (2020)29 | 10 | 735 | Urine output Weight loss Length of hospital stay |
Urine output: WMD 444.4 (95% CI 196 to −693) mL, p < 0.001 Weight loss: WMD 0.89 (95% CI 0.04 to 1.75) kg, p = 0.04 Length of hospital stay: WMD 0.95 (95% CI −1.31 to 3.21) day, p = 0.41 |
Potassium WMD 0.03 mmol/L (95% CI −0.25 to 0.31), p = 0.49 Sodium WMD −0.21 mmol/L (95% CI −1.54 to 1.13), p = 0.76 Creatinine WMD 8.8 (95% CI −12.4 to 30.9) μmol/L, p = 0.41 |
Greater urine output and greater weight loss with continuous infusion |
BNP = brain natriuretic peptide, CI = confidence interval, OR = odds ratio, RR = relative risk, WMD = weighted mean difference.
Secondary outcomes reported if they pertain to decongestion parameters.
No significant difference in urine output at 24, 48, or 72 hours.
Four of the included studies reported tinnitus or hearing loss in a total of 5 of 58 patients in the control group and 0 of 53 patients with continuous infusion.
No significant difference in mortality rate with exclusion of patients in the intensive care unit (OR 1.68, 95% CI 0.99 to 3.21, p = 0.05).
Overall, the choice of continuous versus intermittent infusion of furosemide depends on the individual’s clinical condition. It is common to trial furosemide infusions as a secondary strategy if bolus administration fails to achieve the clinical end point of decongestion. A continuous infusion of furosemide may be started at 5 mg/h and titrated up to 40 mg/h as needed.14,16 Thiazide diuretics may be added to the continuous infusion to augment diuresis.14
Thiazide Diuretics
Thiazide diuretics target the NaCl channel in the distal tubule.5 Table 5 summarizes the pharmacokinetics of thiazide diuretics.16,18,19 Hydrochlorothiazide, chlorthalidone, and metolazone have bioavailabilities of approximately 60%–80% and undergo primarily renal excretion. Indapamide has a bioavailability of 93% and undergoes hepatic metabolism. Chlorthalidone has the longest half-life, 40–60 hours, as compared with less than 24 hours for hydrochlorothiazide, indapamide, and metolazone. If congestion persists after careful maximization of loop diuretic therapy over the first 24–48 hours, adjunctive diuretics should be added. Thiazide and thiazide-like diuretics are recommended as second-line agents.3 They are relatively weak diuretics, resulting in a FENA of 5%–8%.5 However, when used in HF patients who are receiving long-term loop diuretic therapy, they cause marked diuresis and natriuresis.34 These patients have compensated nephrons with distal tubular hypertrophy and increased NaCl channel density. Thiazides specifically target this hyperabsorptive segment, causing marked and synergistic diuresis when combined with loop diuretics. The addition of thiazides in this manner has been referred to as “sequential nephronal blockade”.34 Traditionally, thiazides are administered 30–60 minutes before a dose of loop diuretic, but there have been no randomized controlled trials to evaluate this strategy; rather, this practice is based on expert opinion.
TABLE 5.
Pharmacokinetic and Pharmacodynamic Properties of Thiazide Diuretics
| Property | Hydrochlorothiazide | Chlorthalidone | Indapamide | Metolazone |
|---|---|---|---|---|
| Relative potency | 1 | 2 | 20 | 10 |
| Equivalent dose (mg) | 50 | 25 | 2.5 | 5 |
| Bioavailability (%) | 60–80 | 60–70 | 93 | 65 |
| Onset (h) | 2 | 2–3 | 1–2 | 1 |
| Time to peak serum concentration (h) | 4–6 | 2–6 | 2 | 2 |
| Protein binding (%) | About 40 | About 80 | About 75 | About 95 |
| Clearance | Renal: about 50%–70% Minimal hepatic metabolism | Renal: about 30%–75% Minimal hepatic metabolism | Renal: about 70% (5%–7% as unchanged drug) Extensive hepatic metabolism | Renal: about 80% Minimal hepatic metabolism |
| Half-life (h) | 6–15 | 40–60 | 4–22 | 8–14 |
| Average duration of action (h) | 6–12 | 24–72 | Up to 36 | 12–24 |
| Maximum daily dose (mg) | 200 | 200 | 5 | 20 |
Metolazone is a thiazide-like diuretic most often used as an adjunct to loop diuretics when combination therapy is required to meet decongestive targets.5,34 There are no large randomized, controlled trials showing superiority of metolazone over other thiazide diuretics when added to loop diuretics to augment diuresis.35 It has been suggested that metolazone does not decrease glomerular filtration rate,34 and it was widely believed that thiazides were ineffective in patients with glomerular filtration rate less than 30 mL/min. However, studies have demonstrated that combination regimens of furosemide and hydrochlorothiazide are more potent than either agent alone for increasing fractional excretion of sodium and chloride in patients with hypertension and stage 4 or 5 chronic kidney disease.36 Chlorthalidone therapy improved blood pressure control in patients with stage 4 chronic kidney disease and poorly controlled hypertension.37 Diuretic efficacy is a function of drug delivery to the site of action; as such, higher doses are needed for patients with renal dysfunction.
Chlorthalidone,38 indapamide,39 and hydrochlorothiazide40 have also been studied in acute HF. These small, short-duration trials looked at changes in urinary sodium or body weight and did not meet the inclusion criteria for the current review. Salahudin and others39 conducted a randomized, controlled trial in 150 patients who had no response to furosemide 40 mg IV administered every 8 hours. The patients then received either metolazone 5 mg PO daily or indapamide 2.5 mg PO daily in conjunction with the same dosage of IV furosemide. On days 3 and 5, there were no differences in urinary sodium excretion, urine output, or weight loss.39
The CLOROTIC trial was the first large (n = 230) randomized, double-blind, placebo-controlled trial comparing hydrochlorothiazide with placebo, in patients with ADHF who were already receiving IV furosemide (see Table 3).26 The IV furosemide was administered by intermittent bolus every 12 hours, at the same dose as the outpatient oral dose, and the hydrochlorothiazide dosage, determined according to creatinine clearance, ranged from 25 to 100 mg PO daily for 5 days. The primary end points were change in weight and patient-reported dyspnea. Patients who received hydrochlorothiazide achieved greater weight loss at 72 hours (net difference –1.14 kg, 95% confidence interval [CI] −1.84 to −0.42 kg; p = 0.002). There was no significant difference in patient-reported dyspnea. This study had several limitations. It was terminated early because of low enrolment: the authors had originally targeted 304 patients but enrolled only 230. In addition, there were significant differences between the hydrochlorothiazide and placebo groups at baseline, in terms of sex (39.5% vs 56.9% female), systolic blood pressure (121 mm Hg versus 130 mm Hg), body mass index (30 versus 33), and ischemic cardiomyopathy (40.4% vs 25.2%). Many patients did not have severe congestion, with approximately 38% reporting New York Heart Association functional class I or II HF. Moreover, there was no explicit congestion requirement for inclusion. If the study had been limited to patients with greater symptom severity and congestion, a greater reduction in weight might have been observed. Lastly, patients who participated in the trial had chronic HF and were receiving loop diuretics before admission; as such, the findings may not be applicable to patients with newly diagnosed HF.
Carbonic Anhydrase Inhibitors
A single carbonic anhydrase inhibitor was investigated in this review. Acetazolamide is recommended as a third-line diuretic for those with ongoing congestion despite the use of loop diuretics and thiazides.3 This drug interrupts sodium and bicarbonate reabsorption in the proximal tubule,41 which is the segment responsible for approximately 65% of total sodium reabsorption under normal physiologic conditions. 3 Acetazolamide has excellent oral bioavailability, with peak activity at 2 hours, and has a half-life of 4–8 hours in patients with normal renal function.42 Approximately 70%–100% of the drug is excreted unchanged in the urine.16,18 A meta-analysis on the use of acetazolamide in patients with stable or decompensated HF (n = 229) reported dosages ranging from 250 mg to 1.5 g daily.43
Acetazolamide exhibits weak inherent diuretic action, resulting in a FENA of 4%.5 However, like thiazides, it is substantially more potent when used in patients with longstanding exposure to loop diuretics. In addition to compensatory changes in the distal tubule, long-term use of loop diuretics results in hyperabsorptive adaptations of the proximal tubules.3,44 Targeting this adapted segment with acetazolamide results in further synergistic diuresis. Furthermore, through its bicarbonate-wasting mechanism, acetazolamide can improve the metabolic alkalosis that frequently accompanies aggressive diuresis. Metabolic alkalosis reduces respiratory drive, cardiac contractility, and loop diuretic responsiveness, while exacerbating hypokalemia and hypophosphatemia.45
The ADVOR trial (n = 519) showed resolution of decongestion within 3 days of adding acetazolamide 500 mg IV daily to standardized IV loop diuretics at a dose equivalent to twice the patient’s oral maintenance dose (Table 3).27 The total administered dose of IV loop diuretic was similar in the 2 treatment arms. Successful decongestion occurred in 42.2% of patients in the acetazolamide group and 30.5% of those in the placebo group (risk ratio 1.46, 95% CI 1.17 to 1.82; p < 0.001). The study had several limitations. White participants were overrepresented (acetazolamide 99.6% vs placebo 98.5%), which made generalization of results to other racial and ethnic groups questionable. The patients had a history of chronic HF and were receiving outpatient treatment with at least 40 mg equivalent of furosemide; therefore, the results may not be applicable to patients with newly diagnosed HF. Thiazides and amiloride were discontinued on enrolment, and the benefits and risks of these treatments with concomitant acetazolamide are unknown.
Mineralocorticoid Antagonists
An increase in plasma aldosterone levels is associated with progression of myocardial damage during an acute phase of HF and with poor prognosis in patients who have acute HF.46–50 MRAs are an important part of guideline-based HF management and have been associated with reductions in all-cause mortality, death from cardiovascular causes, and HF hospitalizations,48,49,51 but they are ineffective as decongestive agents. Spironolactone and eplerenone have good oral bioavailability, with peak activity in 2–4 hours.16,18 Spironolactone is rapidly and extensively metabolized to several metabolites, including canrenone and the sulfur-containing 7-thiomethylspirolactone, both of which are pharmacologically active. Approximately 25%–30% of spironolactone is converted to canrenone, which has a half-life of 9–23 hours.18 This metabolite is thought to be primarily responsible for the drug’s therapeutic effects. The half-life of 7-thiomethylspirolactone is approximately 14 hours.16,18 Eplerenone also undergoes extensive hepatic metabolism and has a half-life of 3–6 hours.16,18 MRAs are weak diuretics, resulting in a FENA of 2%.3 In the Athena-HF trial, there were no significant changes in any decongestion parameters when spironolactone 100 mg PO daily was added to therapy for patients who presented with acute HF (Table 3).25 Decongestion parameters were secondary outcomes in this study. Approximately 25% of the patients were receiving MRA therapy before enrolment, which may have affected the treatment effect observed. The trial was only 96 hours in duration, which may have influenced the degree of diuresis achieved. The EARLIER trial of eplerenone in acute HF did not report any congestion parameters (Table 3).28 Although they are ineffective as diuretics, MRAs can be used to minimize the hypokalemia associated with aggressive diuretic therapy.
DISCUSSION
Adequate decongestion is often not achieved during hospital admission. Patients with HF for whom complete decongestion is achieved in hospital have improved outcomes relative to those without complete decongestion. It is necessary to review the evidence on the importance of attaining decongestion and to provide potential practical targets for the use of diuretics. In a post hoc analysis of the DOSEAHF and CARESS-HF studies (n = 496), only half of the patients were free from signs of congestion at discharge.52 Peripheral edema, elevated jugular venous pressure, and orthopnea were used as markers of congestion. Patients discharged without congestion had lower rates of death, readmission, and urgent clinic or ED visits (50%) compared with those who had low-grade congestion (52%) or high-grade congestion (68%) (p = 0.038).52 A retrospective analysis of the DOSE study showed that weight loss, fluid loss, and N-terminal prohormone B type natriuretic peptide (NT-proBNP) at 72 hours were poorly correlated with dyspnea relief.11 However, improvements in each of these 3 markers were associated with improved clinical outcomes (death, hospitalization for HF, ED visit for HF) at 60 days. Each 1000-mL increment in net fluid output was associated with a 6% reduction in risk of the combined clinical end point at 60 days. Each 1.8-kg weight loss at 72 hours was associated with a 9% reduction in risk, and each 10% reduction in NT-proBNP from baseline was associated with a 5% reduction in risk. A retrospective analysis of the PROTECT study (n = 1572) found that all-cause mortality at 180 days more than doubled among patients with substantial congestion at day 7 compared with those who had no or mild congestion (hazard ratio [HR] 2.13, 95% CI 1.66 to 2.73). The risk of hospitalization due to HF at day 60 was significantly greater in patients with substantial residual congestion at day 7 (HR 1.88, 95% CI 1.43 to 2.46).6 Earlier administration of furosemide in ED patients with decompensated HF may improve mortality. In a multicentre cohort of 1291 ED patients with acute HF, the 481 patients who received IV furosemide within 90 minutes of arrival in the ED had significantly lower inpatient all-cause mortality than those who received this form of therapy after 90 minutes (2.3% versus 6%, p = 0.002).10
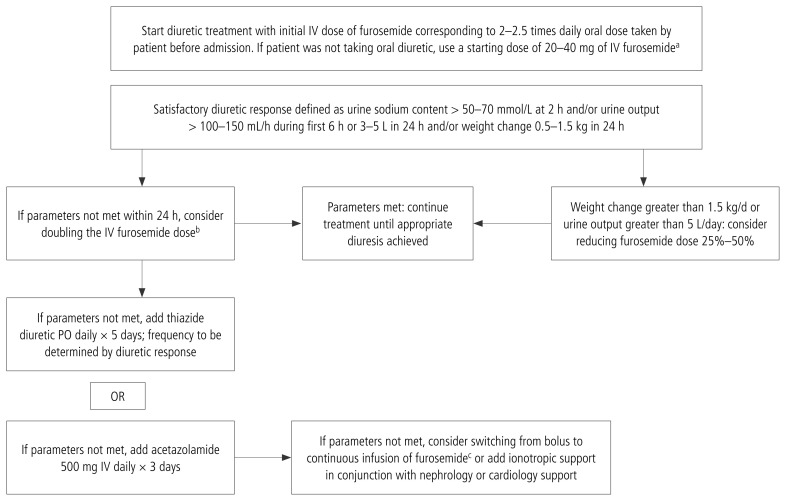
The Canadian, US, and European HF guidelines state that IV loop diuretics are first-line therapy for patients with ADHF who present with pulmonary or peripheral congestion. Loop diuretic therapy should be maximized before other adjunctive diuretic classes are added (Figure 1). Early biochemical targets have been identified to direct the early and rapid escalation of loop diuretic dose. A satisfactory diuretic response can be defined as urine sodium content greater than 50–70 mmol/L at 2 hours; urine output greater than 100–150 mL/h during the first 6 hours or 3–5 L in 24 hours; or weight change of 0.5–1.5 kg in 24 hours.7,8 Data suggest that muted natriuretic responses to early doses of loop diuretics predict diuretic resistance, worsening renal function, readmission, and death in patients with ADHF.53 If this target is not met, it is recommended to double the subsequent dose of loop diuretic dose.3 Frequent reassessments, over the initial 24–48 hours, should trigger thoughtful up-titration of loop diuretic dose in those with ongoing refractory congestion. Therapy for acute HF continues to evolve. The STRONG-HF trial (n = 1078) showed that an intensive treatment strategy, consisting of rapid up-titration of guideline-directed medication and close follow-up after admission for acute HF, improved quality of life and reduced the risk of 180-day all-cause death or HF readmission compared with usual care.54 The EMPULSE trial (n = 530) showed that empagliflozin started during hospitalization for acute HF reduced 90-day all-cause mortality, number of HF events, and time to first HF event, as well as improving quality of life.55 Large randomized controlled trials are needed to evaluate optimal diuretic strategies for patients presenting with ADHF.
FIGURE 1.
Algorithm for diuretic management in patients with acute heart failure and volume overload. aEthacrynic acid 50 mg or 0.5–1 mg/kg per IV dose (maximum single dose 100 mg). bDose of diuretic may be adjusted based on clinician discretion. cFurosemide infusion may be started earlier if hypotension is present or at clinician’s discretion.
The management of congestion due to HF may include restriction of dietary sodium and fluid. The evidence to support these concepts is scarce, and some evidence suggests the opposite of current clinical practice.7 The Canadian Cardiovascular Society suggests that patients with HF should restrict dietary salt intake to between 2 and 3 g/day and should restrict fluid intake to approximately 2 L/day for patients with fluid retention or congestion that is not easily controlled with diuretics.7 In clinical practice, sodium is often restricted to less than 2 g/day and fluid to 1.5–2 L/day. With these targets in place, the goal is 0.5–1.5 kg of weight loss per 24-hour period while a patient with volume overload is undergoing active diuresis.7 High-quality data on this topic are lacking.
The recently published SODIUM-HF study is the largest trial of its type to date, with longer follow-up than previous studies evaluating sodium intake in patients with HF. The trial was an international, open-label, randomized, controlled trial that enrolled 806 patients (about 60% from Canada) who were followed for 12 months.56 A dietary sodium target of less than 100 mmol (1500 mg/day) was prescribed for the low sodium group, whereas the control group received usual care. At 12 months, median sodium intake was 1658 (range 1301–2189) mg/day in the low sodium arm and 2073 (range 1541–2900) mg/day in the control group. There was no difference in terms of reduction in hospital admission or ED visits due to cardiovascular causes or all-cause mortality.56 The lack of benefit may have been due to limited statistical power related to early cessation of the trial when the COVID-19 pandemic reduced hospital admissions.57 The study had a lower-than-expected event rate of 17% at 12 months, whereas the expected event rate was 25%.58 It remains to be seen how HF guidelines will incorporate this study into practice.
CONCLUSION
Diuretics are the primary decongestive agents used in treating HF. As opposed to medications that target neurohormonal compensatory mechanisms, the evidence guiding diuretic therapy in acute HF is sparse. In this context, recent recommendations provide practical support for clinicians managing the care of hospitalized patients with ADHF. Safe and effective diuresis is a dynamic process and requires serial reassessments of data-based clinical and biochemical markers and end points. Careful administration of high-dose loop diuretics, supported by thiazides and acetazolamide when necessary, is an increasingly evidence-based diuretic strategy. Clinical trials are underway to further evaluate this strategy.
Footnotes
Competing interests: None declared.
Funding: None received.
References
- 1. Virani SS, Bains M, Code J, Ducharme A, Harkness K, Howlett JG, et al. The need for heart failure advocacy in Canada. Can J Cardiol. 2017;33(11):1450–4. doi: 10.1016/j.cjca.2017.08.024. [DOI] [PubMed] [Google Scholar]
- 2. Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Borden WB, et al. Heart disease and stroke statistics–2013 update: a report from the American Heart Association. Circulation. 2013;127(1):e6–e245. doi: 10.1161/CIR.0b013e31828124ad. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Mullens W, Damman K, Harjola VP, Mebazaa A, Brunner-La Rocca HP, Martens P, et al. The use of diuretics in heart failure with congestion - a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2019;21(2):137–55. doi: 10.1002/ejhf.1369. [DOI] [PubMed] [Google Scholar]
- 4. Gheorghiade M, Pang PS. Acute heart failure syndromes. J Am Coll Cardiol. 2009;53(7):557–73. doi: 10.1016/j.jacc.2008.10.041. [DOI] [PubMed] [Google Scholar]
- 5. Felker GM, Ellison DH, Mullens W, Cox ZL, Testani JM. Diuretic therapy for patients with heart failure: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75(10):1178–95. doi: 10.1016/j.jacc.2019.12.059. [DOI] [PubMed] [Google Scholar]
- 6. Rubio-Gracia J, Demissei BG, Ter Maaten JM, Cleland JG, O’Connor CM, Metra M, et al. Prevalence, predictors and clinical outcome of residual congestion in acute decompensated heart failure. Int J Cardiol. 2018;258:185–91. doi: 10.1016/j.ijcard.2018.01.067. [DOI] [PubMed] [Google Scholar]
- 7. Ezekowitz JA, O’Meara E, McDonald MA, Abrams H, Chan M, Ducharme A, et al. 2017 comprehensive update of the Canadian Cardiovascular Society guidelines for the management of heart failure. Can J Cardiol. 2017;33(11):1342–433. doi: 10.1016/j.cjca.2017.08.022. [DOI] [PubMed] [Google Scholar]
- 8. McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, et al. 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42(36):3599–726. doi: 10.1093/eurheartj/ehab368. [DOI] [PubMed] [Google Scholar]
- 9. Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, et al. 2022 AHA/ACC/HFSA guideline for the management of heart failure: executive summary: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2022;79(17):1757–80. doi: 10.1016/j.jacc.2021.12.011. [DOI] [PubMed] [Google Scholar]
- 10. Matsue Y, Damman K, Voors AA, Kagiyama N, Yamaguchi T, Kuroda S, et al. Time-to-furosemide treatment and mortality in patients hospitalized with acute heart failure. J Am Coll Cardiol. 2017;69(25):3043–51. doi: 10.1016/j.jacc.2017.04.042. [DOI] [PubMed] [Google Scholar]
- 11. Kociol RD, McNulty SE, Hernandez AF, Lee KL, Redfield MM, Tracy RP, et al. Markers of decongestion, dyspnea relief, and clinical outcomes among patients hospitalized with acute heart failure. Circ Heart Fail. 2013;6(2):240–5. doi: 10.1161/CIRCHEARTFAILURE.112.969246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Felker GM, Lee KL, Bull DA, Redfield MM, Stevenson LW, Goldsmith SR, et al. Diuretic strategies in patients with acute decompensated heart failure. N Engl J Med. 2011;364(9):797–805. doi: 10.1056/NEJMoa1005419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Salvador DRK, Punzalan FE, Rey NR, Bernado MR, Nablo MD. Continuous infusion versus bolus injection of loop diuretics in congestive heart failure. Cochrane Database Syst Rev. 2005;2005(3):CD003178. doi: 10.1002/14651858.CD003178.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Ellison DH, Felker GM. Diuretic treatment in heart failure. N Engl J Med. 2017;377(20):1964–75. doi: 10.1056/NEJMra1703100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Shankar SS, Brater DC. Loop diuretics: from the Na-K-2Cl transporter to clinical use. Am J Physiol Renal Physiol. 2003;284(1):F11–21. doi: 10.1152/ajprenal.00119.2002. [DOI] [PubMed] [Google Scholar]
- 16.Lexicomp online [database on Internet] Lexi-Comp Inc; 2022. [cited 2022 Dec 11]. Available from: https://online.lexi.com (subscription required to access content) [Google Scholar]
- 17.Jackson EK. Drugs affecting renal excretory function. In: Brunton LL, Hilal-Dandan R, Knollmann BC, editors. Goodman & Gilman’s: the pharmacological basis of therapeutics. 13th ed. McGraw-Hill Education; 2018. [cited 2022 Dec 12]. Available from: https://accessmedicine.mhmedical.com (subscription required to access content) [Google Scholar]
- 18.Drugdex [database on Internet] IBM Corporation; [cited 2022 Dec 12]. Available from: https://www.micromedexsolutions.com(subscription required to access content) [Google Scholar]
- 19.CPS. Canadian Pharmacists Association; c2022. [cited 2022 Dec 12]. Available from: https://www.e-therapeutics.ca (subscription required to access content) [Google Scholar]
- 20. Wall GC, Bigner D, Craig S. Ethacrynic acid and the sulfa-sensitive patient. Arch Intern Med. 2003;163(1):116–7. doi: 10.1001/archinte.163.1.116. [DOI] [PubMed] [Google Scholar]
- 21. Juang P, Page RL, 2nd, Zolty R. Probable loop diuretic-induced pancreatitis in a sulfonamide-allergic patient. Ann Pharmacother. 2006;140(1):128–34. doi: 10.1345/aph.1G314. [DOI] [PubMed] [Google Scholar]
- 22. Molnar J, Somberg JC. The clinical pharmacology of ethacrynic acid. Am J Ther. 2009;16(1):86–92. doi: 10.1097/MJT.0b013e318195e460. [DOI] [PubMed] [Google Scholar]
- 23. Bikdeli B, Strait KM, Dharmarajan K, Chohreh Partovian C, Coca SG, Kim N, et al. Dominance of furosemide for loop diuretic therapy in heart failure: time to revisit the alternatives? J Am Coll Cardiol. 2013;61(14):1549–50. doi: 10.1016/j.jacc.2012.12.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Brater DC. Pharmacokinetics of loop diuretics in congestive heart failure. Br Heart J. 1994;72(2 Suppl):S40–S43. doi: 10.1136/hrt.72.2_Suppl.S40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Butler J, Anstrom KJ, Felker GM, Givertz MM, Kalogeropoulos AP, Konstam MA, et al. Efficacy and safety of spironolactone in acute heart failure: the ATHENA-HF randomized clinical trial. JAMA Cardiol. 2017;2(9):950–8. doi: 10.1001/jamacardio.2017.2198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Trullàs JC, Morales-Rull JL, Casado J, Carrera-Izquierdo M, Sánchez-Marteles M, Conde-Martel A, et al. Combining loop with thiazide diuretics for decompensated heart failure: the CLOROTIC trial. Eur Heart J. 2023;44(5):411–21. doi: 10.1093/eurheartj/ehac689. [DOI] [PubMed] [Google Scholar]
- 27. Mullens W, Dauw J, Martens P, Verbrugge FH, Nijst P, Meekers E, et al. Acetazolamide in acute decompensated heart failure with volume overload. N Engl J Med. 2022;387(13):1185–95. doi: 10.1056/NEJMoa2203094. [DOI] [PubMed] [Google Scholar]
- 28. Asakura M, Ito S, Yamada T, Saito Y, Kimura K, Yamashina A, et al. Efficacy and safety of early initiation of eplerenone treatment in patients with acute heart failure (EARLIER trial): a multicenter, randomized, double blind, placebo-controlled trial. Eur Heart J Cardiovasc Pharmacother. 2022;8(2):108–17. doi: 10.1093/ehjcvp/pvaa132. [DOI] [PubMed] [Google Scholar]
- 29. Chan JSK, Kot TKM, Ng M, Harky A. Continuous infusion versus intermittent boluses of furosemide in acute heart failure: a systematic review and meta-analysis. J Card Fail. 2020;26(9):786–93. doi: 10.1016/j.cardfail.2019.11.013. [DOI] [PubMed] [Google Scholar]
- 30. Wu M, Chang N, Su C, Hsu Y, Chen T, Lin Y, et al. Loop diuretic strategies in patients with acute decompensated heart failure: a meta-analysis of randomized controlled trials. J Crit Care. 2014;29(1):2–9. doi: 10.1016/j.jcrc.2013.10.009. [DOI] [PubMed] [Google Scholar]
- 31. Ng KT, Yap JLL. Continuous infusion vs. intermittent bolus injection of furosemide in acute decompensated heart failure: systematic review and meta-analysis of randomised controlled trials. Anaesthesia. 2018;73(2):238–47. doi: 10.1111/anae.14038. [DOI] [PubMed] [Google Scholar]
- 32. Kuriyama A, Urushidani S. Continuous versus intermittent administration of furosemide in acute decompensated heart failure. Heart Fail Rev. 2019;24(1):31–9. doi: 10.1007/s10741-018-9727-7. [DOI] [PubMed] [Google Scholar]
- 33. Fudim M, Spates T, Sun JL, Kittipibul Y, Testani JM, Starling RC, et al. Early diuretic strategies and the association with in-hospital and post discharge outcomes in acute heart failure. Am Heart J. 2021;239:110–9. doi: 10.1016/j.ahj.2021.05.011. [DOI] [PubMed] [Google Scholar]
- 34. Sica DA, Gehr TWB. Diuretic combinations in refractory oedema states. Pharmacokinetic-pharmacodynamic relationships. Clin Pharmacokinet. 1996;30(3):229–49. doi: 10.2165/00003088-199630030-00004. [DOI] [PubMed] [Google Scholar]
- 35. Jentzer JC, DeWald TA, Hernandez AF. Combination loop diuretics with thiazide-type diuretics in heart failure. J Am Coll Cardiol. 2010;56(19):1527–34. doi: 10.1016/j.jacc.2010.06.034. [DOI] [PubMed] [Google Scholar]
- 36. Dussol B, Moussi-Frances J, Morange S, Somma-Delpero C, Mundler O, Berland Y. A pilot study comparing furosemide and hydrochlorothiazide in patients with hypertension and stage 4 or 5 chronic kidney disease. J Clin Hypertens (Greenwich) 2012;14(1):32–7. doi: 10.1111/j.1751-7176.2011.00564.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Agarwal R, Sinha AD, Cramer AE, Balmes-Fenwick M, Dickinson JH, Ouyang F, et al. Chlorthalidone for hypertension in advanced chronic kidney disease. N Engl J Med. 2021;385(27):2507–19. doi: 10.1056/NEJMoa2110730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Liacer P, Nunez J, Garcia M, Ruiz R, Lopez G, Fabregate M, et al. Comparison of chlorthalidone and spironolactone as additional diuretic therapy in patients with acute heart failure and reserved ejection fraction. Eur Heart J Acute Cardiovasc Care. 2022;11(4):350–5. doi: 10.1093/ehjacc/zuac006. [DOI] [PubMed] [Google Scholar]
- 39. Salahudin M, Shah H, Jan MU, Altaf A. Comparing the sodium excreting efficacy of furosemide and indapamide combination against furosemide and metolazone combination in congestive heart failure patients: a randomized controlled trial. J Pak Med Assoc. 2019;69(12):1794–9. doi: 10.5455/JPMA.3401. [DOI] [PubMed] [Google Scholar]
- 40. Piardi DS, Butzke M, Mazzuca ACM, Gomes BS, Alves SG, Kotzian BJ, et al. Effect of adding hydrochlorothiazide to usual treatment of patients with acute decompensated heart failure: a randomized clinical trial. Sci Rep. 2021;11(1):164–74. doi: 10.1038/s41598-021-96002-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Imiela T, Budaj A. Acetazolamide as add-on diuretic therapy in exacerbations of chronic heart failure: a pilot study. Clin Drug Investig. 2017;37(12):1175–81. doi: 10.1007/s40261-017-0577-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Kassamali R, Sica DA. Acetazolamide: a forgotten diuretic agent. Cardiol Rev. 2011;19(6):276–8. doi: 10.1097/CRD.0b013e31822b4939. [DOI] [PubMed] [Google Scholar]
- 43. Wongboonsin J, Thongprayoon C, Bathini T, Ungprasert P, Aeddula NR, Mao MA, et al. Acetazolamide therapy in patients with heart failure: a meta-analysis. J Clin Med. 2019;8(3):349. doi: 10.3390/jcm8030349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Mullens W, Verbrugge FJ, Nijst P, Martens P, Tartaglia K, Theunissen E, et al. Rationale and design of the ADVOR (acetazolamide in decompensated heart failure with volume overload) trial. Eur J Heart Fail. 2018;20(11):1591–600. doi: 10.1002/ejhf.1307. [DOI] [PubMed] [Google Scholar]
- 45. Mazur JE, Devlin JW, Peters MJ, Jankowski MA, Iannuzzi MC, Zarowitz BJ, et al. Single versus multiple doses of acetazolamide in critically ill medical patients: a randomized, double-blind trial. Crit Care Med. 1999;27(7):1257–61. doi: 10.1097/00003246-199907000-00004. [DOI] [PubMed] [Google Scholar]
- 46. Girerd N, Pang PS, Swedberg K, Fought A, Kwasny MJ, Subacius H, et al. Serum aldosterone is associated with mortality and re-hospitalization in patients with reduced ejection fraction hospitalized for acute heart failure: analysis from the EVEREST trial. Eur J Heart Fail. 2013;15(11):1228–35. doi: 10.1093/eurjhf/hft100. [DOI] [PubMed] [Google Scholar]
- 47. Gheorghiade M, Pang PS, Ambrosy AP, Lan G, Schmidt P, Filippatos G, et al. A comprehensive, longitudinal description of the in-hospital and post-discharge clinical, laboratory, and neurohormonal course of patients with heart failure who die or are re-hospitalized within 90 days: analysis from the EVEREST trial. Heart Fail Rev. 2012;17(3):485–509. doi: 10.1007/s10741-011-9280-0. [DOI] [PubMed] [Google Scholar]
- 48. Pitt B, Zannad F, Remme WJ, Cody R, Castaigne A, Perez A, et al. Randomized Aldactone Evaluation Study Investigators. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. N Engl J Med. 1999;341(10):709–17. doi: 10.1056/NEJM199909023411001. [DOI] [PubMed] [Google Scholar]
- 49. Pitt B, Remme W, Zannad F, Neaton J, Martinez F, Roniker B, et al. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med. 2003;348(14):1309–21. doi: 10.1056/NEJMoa030207. [DOI] [PubMed] [Google Scholar]
- 50. McMurray JVJ. CONSENSUS to EMPHASIS: the overwhelming evidence which makes blockade of the renin-angiotensin-aldosterone system the cornerstone of therapy for systolic heart failure. Eur J Heart Fail. 2011;13(9):929–36. doi: 10.1093/eurjhf/hfr093. [DOI] [PubMed] [Google Scholar]
- 51. Zannad F, McMurray JJ, Krum H, van Veldhuisen DJ, Swedberg K, Shi H, et al. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med. 2011;364(1):11–21. doi: 10.1056/NEJMoa1009492. [DOI] [PubMed] [Google Scholar]
- 52. Lala A, McNulty SE, Mentz RJ, Dunlay SM, Vader JM, AbouEzzeddine OF, et al. Relief and recurrence of congestion during and after hospitalization for acute heart failure: insights from diuretic optimization strategy evaluation in acute decompensated heart failure (DOSE-AHF) and cardiorenal rescue study in acute decompensated heart failure (CARESS-HF) Circ Heart Fail. 2015;8(4):741–8. doi: 10.1161/CIRCHEARTFAILURE.114.001957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Tersalvi G, Dauw J, Gasperetti A, Winterton D, Cioffi GM, Scopigni F, et al. The value of urinary sodium assessment in acute heart failure. Eur Heart J Acute Cardiovasc Care. 2021;10(2):216–23. doi: 10.1093/ehjacc/zuaa006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Mebazaa A, Davison B, Chioncel O, Cohen-Solal A, Diaz R, Filippatos G, et al. Safety, tolerability and efficacy of up-titration of guideline-directed medical therapies for acute heart failure (STRONG-HF): a multinational, open-label, randomised, trial. Lancet. 2022;400(10367):1938–52. doi: 10.1016/S0140-6736(22)02076-1. [DOI] [PubMed] [Google Scholar]
- 55. Voors AA, Angermann CE, Teerlink JR, Collins SP, Kosiborod M, Biegus J, et al. The SGLT2 inhibitor empagliflozin in patients hospitalized for acute heart failure: a multinational randomized trial. Nat Med. 2022;28(3):568–74. doi: 10.1038/s41591-021-01659-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Ezekowitz JA, Colin-Ramirez E, Ross H, Escobedo J, Macdonald P, Troughton R, et al. Reduction of dietary sodium to less than 100 mmol in heart failure (SODIUM-HF): an international, open-label, randomised, controlled trial. Lancet. 2022;399(10333):1391–400. doi: 10.1016/S0140-6736(22)00369-5. [DOI] [PubMed] [Google Scholar]
- 57. Alvarado F, Borzak S. Reduced dietary sodium did not reduce clinical events in heart failure. Ann Intern Med. 2022;175(8):JC88. doi: 10.7326/J22-0058. [DOI] [PubMed] [Google Scholar]
- 58. Beck-da-Silva L, Rohde LE. Reduction of dietary sodium for heart failure: a step forward. Lancet. 2022;399(10333):1361–3. doi: 10.1016/S0140-6736(22)00457-3. [DOI] [PubMed] [Google Scholar]