ABSTRACT
A 75-year-old woman with hypertension, dyslipidemia, thalassemia trait, osteoarthritis of the knees, and hyperthyroidism presented with bloating, abdominal discomfort, and change in stool caliber underwent an esophagogastroduodenoscopy. The esophagogastroduodenoscopy revealed varices at the middle of esophagus, which were diagnosed to be downhill esophageal varices (DEVs). DEVs occur mainly from the superior vena cava (SVC) obstruction; however, in the absence of obstruction, angiogenesis and thrombosis could be the etiology. In our patient, computed tomography showed a pulmonary nodule located at periphery of the right upper lung without SVC contact nor obstruction. The nodule was later proven to be an adenocarcinoma. Thus, our patient showed the possibility that adenocarcinoma of the lung might associated with the DEV through a mechanism other than SVC obstruction. Recognition and differentiation of DEV from other benign venous blebs of the esophagus are important, and once detected, further investigation with computed tomography of the chest is recommended.
KEYWORDS: downhill varices, esophageal varices, varix, SVC obstruction, SVC syndrome, lung cancer
INTRODUCTION
The downhill esophageal varices (DEVs) are vertical longitudinal dilatation of the veins in the esophagus, which were different from the more rounded venous dilatations of blebs in the esophagus. The DEVs were similar in appearance to the more common uphill varices seen in cirrhosis, except for the fact that they appeared at the midesophagus and did not extend to the distal esophagus/esophagogastric junction, where uphill esophageal varices arise from. DEVs were first reported by Simchowitz et al in 1932.1 Subsequent reports2 revealed that the most common etiology for downhill varices is the obstruction of the superior vena cava (SVC). An increase in SVC pressure or obstruction of the SVC results in the retrograde blood flowing to the azygos vein, where the pressure transmits to the esophageal venous plexus, respectively. There are several etiologies of DEVs reported in the literature (Table 1), and most of them are caused by the SVC obstruction either from the direct compression of mass or secondary to various conditions. The downhill varices can be treated conservatively while treating the primary cause of the varices or treating the SVC obstruction with dilation of the SVC stricture by SVC stenting or angioplasty. In case of bleeding, which is rare, the treatment includes endoscopic variceal band ligation or systemic embolization through the brachiocephalic vein, injection of sclerosing agents, or balloon tamponade.3,4
Table 1.
The etiologies of DEVs reported in previous cases
| Study | Age | Sex | Comorbidities | Endoscopic findings | Cause | Management |
| Lung cancer | ||||||
| Tanaka et al5 | 68 | Male | Unknown | Four striated DEVs (F1, CB, and RC) | Small-cell lung cancer at upper right lung filed completely obstructed SVC | Concurrent chemoradiotherapy |
| Tanaka et al5 | 55 | Male | Unknown | Three striated downhill varices (F1, CW, and RC) | Squamous cell lung cancer narrowing SVC with good collateral pathways | Concurrent chemoradiotherapy |
| Subramaniam et al6 | 27 | Male | No underlying disease | Marked varicosity of the esophageal veins over almost the entire length (necropsy finding) | Anaplastic bronchogenic carcinoma 2″ × 1½″ at the entire upper lobe, arised from the right upper lobe bronchus with SVC obstruction | Antimitotic drugs |
| Kokubo et al7 | 66 | Male | Unknown | DEVs at the upper esophagus | Recurrence lung cancer | Upper lobectomy |
| Chauvin et al8 | 55 | Male | Stage IIIb non–small-cell carcinoma status post chemotherapy and radiation therapy | Grade III varices 25–30 cm from the incisors with a visible fibrin clot | Stage IIIb non–small-cell carcinoma in the anterior mediastinum encasing brachiocephalic trunk and SVC occlusion | Band ligation |
| Nonlung cancer cause | ||||||
| Berkowitz et al9 | 32 | Female | ESRD MCTD |
Esophageal varices in the upper and middle esophagus | SVC syndrome secondary to central venous dialysis catheters | Endoscopic band ligation of a proximal varix |
| Chakinala et al10 | 55 | Male | ESRD Rheumatoid arthritis PAD Esophageal varices |
Upper and middle esophageal varices | Chronic SVC and right brachiocephalic vein occlusion secondary to venous catheters | Esophageal band ligation Failed SVC stenting |
| Loudin et al3 | 22 | Female | ESRD Henoch-Schönlein purpura |
Large varices in the proximal esophagus with positive red wale sign | SVC syndrome secondary to central venous dialysis catheters | Balloon dilation of the stenotic SVC |
| Bédard and Deslauriers11 | 68 | Female | Retrosternal goiter | Upper esophageal varices | Extrinsic compression of the right innominate vein secondary to retrosternal goiter | Thyroidectomy |
| Yaşar B, Kılı,oğlu G.12 | 31 | Male | Behçet disease | Prominent esophageal varices in the upper half of the esophagus with an overlying clot | SVC syndrome secondary to Behçet disease | Conservative management DMARDs Steroids |
| Gholam et al13 | 87 | Female | Cameron ulcers Aortic stenosis Pulmonary hypertension |
Large varices in the upper third of the esophagus and Cameron lesions | Severe pulmonary hypertension secondary to aortic stenosis | Conservative management |
| Harwani et al14 | 55 | Female | Liver cirrhosis Chronic rheumatic heart disease; severe mitral and tricuspid regurgitation Pulmonary hypertension |
Upper and lower esophageal varices | Dilated SVC because of pulmonary hypertension | Variceal banding |
| Maton et al15 | 34 | Female | Idiopathic vasculitis | Upper esophageal varices | Vasculitis | Conservative management |
| Serin et al16 | 60 | Female | None | Upper esophageal varices | Increase blood drainage from the tumor into the esophageal veins | Tumor removal |
| Shirakusa et al17 | 26 | Male | Hepatitis | Upper esophageal varices | Excessive blood flow into the esophageal wall from a giant lymphoma | Thoracotomy |
| Yasar and Abut18 | 45 | Male | Seminoma Pelvic radiotherapy |
Varices in the upper third of the esophagus | Bilateral brachiocephalic truncus stenosis because of mediastinal fibrosis | Conservative management |
| Basar et al19 | 54 | Male | AV block Epicardiac pacemaker |
Upper esophageal varices | Bilateral subclavian veins DVTs secondary to pacemaker | Patient refused treatment |
| Ibis et al20 | 35 | Female | History of subtotal thyroidectomy and multinodular goiter | Upper esophageal varices | Downhill varices secondary to recurrent multinodular goiter | Esophageal band ligation Inferior thyroid artery embolization Repeat subtotal thyroidectomy |
| Van der Veldt et al21 | 77 | Female | COPD Multinodular goiter |
Grade II–III upper esophageal varices | Right internal jugular vein compression secondary to multinodular goiter | Subtotal thyroidectomy |
AV, atrioventricular; CB, blue varices; COPD, chronic obstructive pulmonary disease; CW, white varices; DEVs, downhill esophageal varices; DMARDs, disease-modifying antirheumatic drugs; DVT, deep vein thrombosis; ESRD, end stage renal disease; MCTD, mixed connective tissue disease; PAD, peripheral arterial disease; RC, red color sign; SVC, superior vena cava.
CASE REPORT
A 75-year-old woman with the underlying hypertension, dyslipidemia, thalassemia trait, knee osteoarthritis, and hyperthyroidism presented with bloating, abdominal discomfort, and change in stool caliber for a couple days in the previous few weeks. For her bloating, she denied eating a fatty diet, alcohol consumption, nor frequent intake of the dairy products. She had no respiratory symptoms and no appetite loss nor cachexia. She was a life-long nonsmoker. Physical examination was unremarkable; she was breathing comfortably with normal breath sounds on auscultation; the abdomen was soft without tenderness nor guarding; and no lymphadenopathy, no hepatosplenomegaly, no abdominal mass, and no stigmata of chronic liver disease were found. Her medications were calcium carbonate, calciferol, and atenolol.
In view of her age, her medical attendance despite her usual habit of medical stoicism, and the previous change in stool caliber, she was investigated with the colonoscopy and esophagogastroduodenoscopy (EGD). Colonoscopy found multiple polyps, which were removed by biopsy forceps and cold snare. These were found to be hyperplastic polyps on pathological examination. EGD found 2 F2 varices and 1 F1 varix between 30 and 35 cm from the incisors, both of which were in the midesophagus and did not extend to the esophagogastric junction (Figure 1). Below the varices, there were a few benign squamous papillomata, and a venous bleb at 39 cm from the incisors just proximal to the esophagogastric junction, but otherwise, the mucosa distal to the varices was normal. No gastric varices were seen. The varices were recognized to be DEVs from their appearance as vertical longitudinal dilatation of the veins in the esophagus, which were different from the more rounded venous dilatations of esophageal blebs, and the fact that they did not extend distally to the esophagogastric junction like the more common uphill varices seen in cirrhosis; thus, chest x-ray and computed tomography (CT) of the chest and neck were performed to evaluate the cause of varices. Although the chest x-ray (Figure 2) showed a barely detectable reticular-ground glass opacity in the right upper zone, the CT results (Figures 3 and 4) revealed a 2.7-cm pulmonary nodule located at periphery of the right upper lung, adjacent to the pleura and far superolateral to the SVC without SVC contact and obstruction, no neck mass, subcentimeter lymph nodes with internal microcalcification at left cervical IIb level, posterior to spinal accessory nerve and internal jugular vein. No thyroid nodules and no liver cirrhosis were found.
Figure 1.
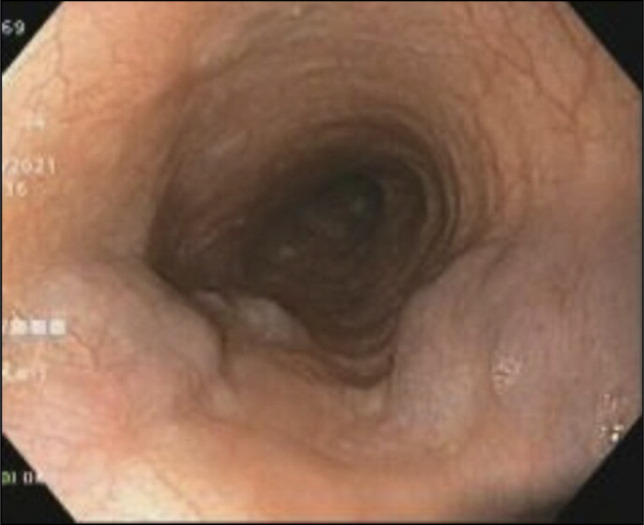
Downhill esophageal varices seen in the patient. Two F2 varices and 1 F1 varix were seen between 30 and 35 cm from the incisors.
Figure 2.
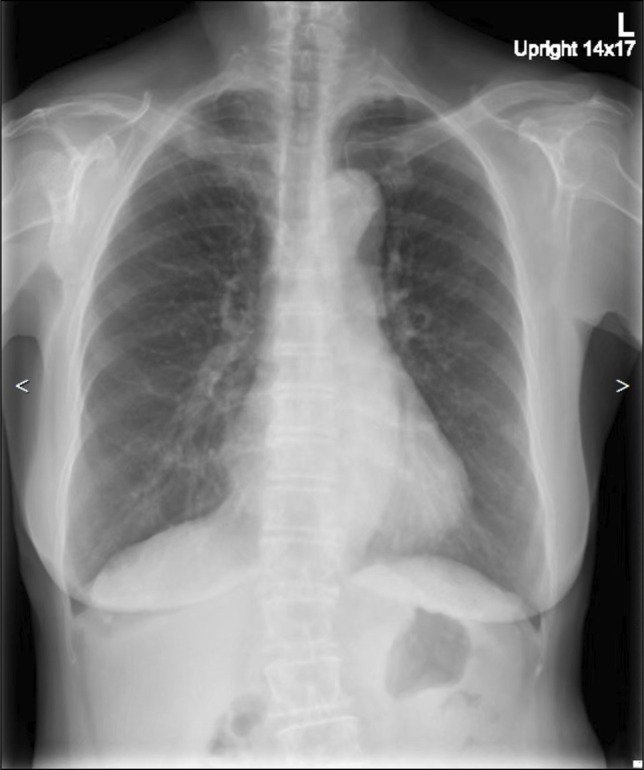
Chest x-ray posteroanterior upright showing a reticular-ground glass at right upper lung zone, which was barely detectable.
Figure 3.
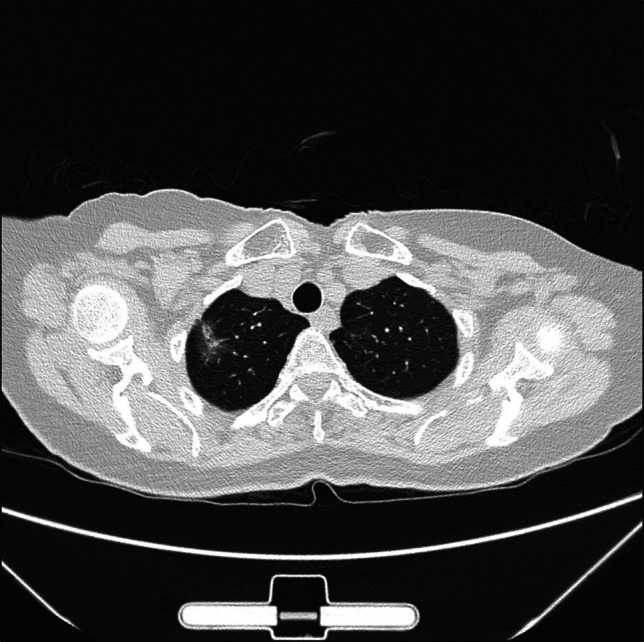
CT of the chest and mediastinum axial plane showing a 2.7-cm nodule at periphery of the right upper lung. CT, computed tomography.
Figure 4.
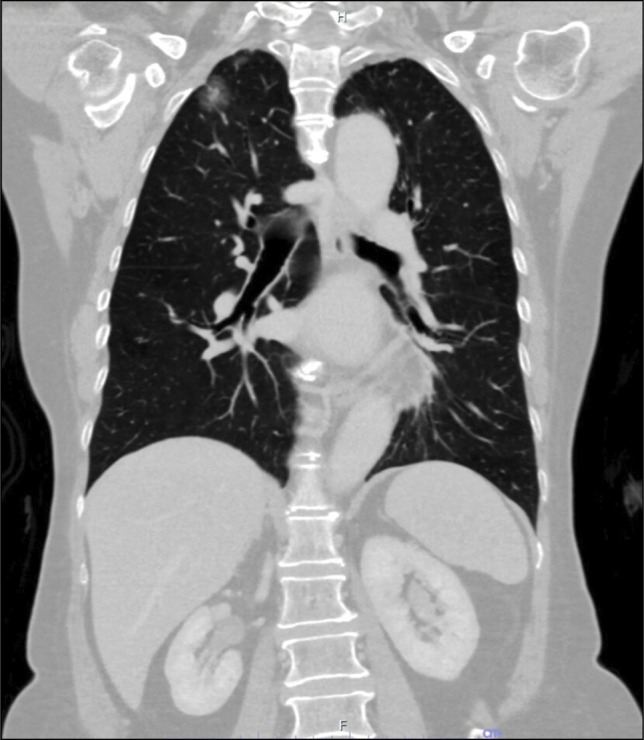
CT of the chest and mediastinum coronal plane showing a 2.7-cm nodule at periphery of the right upper lung. CT, computed tomography.
The patient underwent a lung biopsy, which revealed the lesion to be an adenocarcinoma.
After the pathology results, the patient underwent a video-assisted thoracoscopic surgery with right upper lobe lobectomy and lymph node biopsy. The pathological examination from the surgical specimen (Figure 5) showed a lepidic-predominant invasive adenocarcinoma, the total size of 2.7 cm, with an invasive tumor size of 1.1 cm in the greatest dimension, with acinar pattern at the periphery of the lung and close to pleura (as shown in Figure 5). Visceral pleural invasion was not identified in the additional elastic stain. Lymphovascular invasion and vascular thrombosis were not seen (as shown in Figure 6). The dissection of the lymph nodes of hilar, interlobar, right paratracheal, and subcarinal area showed no evidence of metastatic carcinoma. The vascular abnormalities associated with pulmonary hypertension were not detected. The epidermal growth factor receptor (EGFR) gene mutational analysis in the paraffin block revealed EGFR c.2573T>G (L858R) mutation. Clear resection margins were seen. In conclusion, the tumor staging was T1bN0M0.
Figure 5.
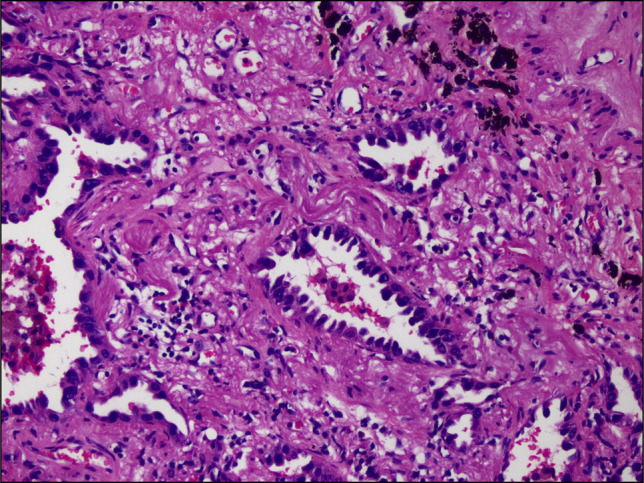
The histopathological specimen showing adenocarcinoma with acinar pattern.
Figure 6.
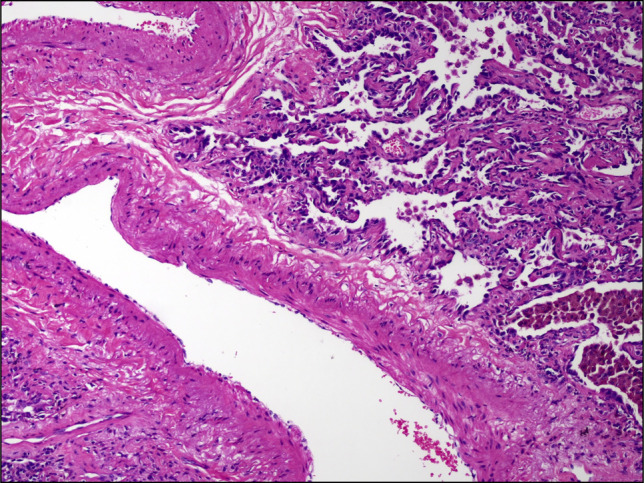
The histopathological specimen showing a lepidic adenocarcinoma, with pulmonary vein but no thrombosis.
The patient recovered postoperatively and remains well at 12 months. No further endoscopy was performed because there has been no clinical indication for a repeat endoscopy.
DISCUSSION
DEVs develop when the esophageal plexus has increased blood flow, and they are usually associated with venous obstruction from the SVC. The level of obstruction affects the formation of varices. Lesions proximal to azygous vein push the drainage through mediastinal collaterals and drain back to the azygous system below the obstruction. Varices that occur from lesions in this location are limited to the upper third of the esophagus. On the other hand, varices that are from obstruction distal to the azygos vein mostly expand along the entire length of the esophagus.
The etiology of downhill varices has been widely reported among case studies, most of which were from SVC syndrome and other vascular occlusions. The literatures describe other causes including mediastinal fibrosis,18 Behçet disease,22 catheter manipulation,3,9,11 retrosternal goiter and other thyroid masses,10,20,21 thymomas,23 bronchial carcinomas,6 metastatic mediastinal tumors,7 pulmonary hypertension,13,14 lymphomas,17 or were idiopathic. The causal pathway for most of these etiologies is through venous obstruction in the paraesophageal/mediastinal area.
Our patient presented with asymptomatic downhill varices diagnosed incidentally on EGD. However, in this case, from the CT, there was no evidence of SVC obstruction. This is unlike most other cases of downhill varices with upper mediastinal/thoracic tumor, which often reported SVC compression from an extrinsic mass such as from small-cell lung carcinoma, lymphoma, or bronchial carcinoma. Downhill varices without SVC obstruction have also been mentioned in the literature and can occur from an increase in blood flow in the esophageal plexuses, eg, from Castleman disease, also known as angiofollicular lymph node hyperplasia, which is known to be hypervascular and cause downhill varices by increasing blood drainage to the esophageal veins.16 Moreover, obstruction in some venous drainage other than SVC has also been reported as a cause of downhill varices. In thyroid disease, obstruction of the inferior thyroid vein can lead to the development of proximal esophageal varices.21
From previous case studies,5–8,24 all lung cancer patients with DEVs were associated with SVC syndrome, by the mechanism of venous obstruction. However, in our case, SVC obstruction was absent on CT, and no vascular invasion nor thrombosis was seen on the histopathological specimen; the varices might be hypothesized to have formed because of increased blood flow in the area as a result of tumor angiogenesis, given that tumor progression depends primarily on vascular supply. Non–small-cell lung cancer, particularly adenocarcinoma, is a highly vascularized tumor,25 suggesting that a consequent increase in the blood flow to the tumor and surrounding vessels is likely.26 In addition, the pathological specimen revealed EGFR gene mutation of the tumor, which have a critical role in tumor angiogenesis through upregulation of vascular endothelial growth factor and other angiogenic factors.27,28 Alternatively, it is possible that thrombosis could have formed in the small vessels in the area because of the hypercoagulable state of cancer, causing a retrograde blood flow into the esophageal plexus and subsequent formation of new varices.
Downhill varices occur mainly from SVC obstruction, which could be caused by malignant or benign processes. The presence of DEVs should lead to a suspicion of lesions compressing venous drainage, including lung cancer, especially non–small-cell lung cancer at apex or the upper lung. However, without obstruction, aberrant blood flow, angiogenesis, or thrombosis may also be the etiology for DEVs. Recognition of downhill varices at endoscopy and its differentiation from other benign venous blebs of the esophagus is important, and once detected, further investigation with CT of the chest is recommended.
DISCLOSURES
Author contributions: N. Kitkarncharoensin, S. Catithammanit, and N. Chavanachinda: draft, edited the article, and reviewed the literature. B. Phruttinarakorn supplied, interpreted the pathology images, and edited the article. P. Cherntanomwong edited the article. T. Kitiyakara edited the article and is the article guarantor. All authors approved the final version of the manuscript.
Financial disclosure: None to report.
Informed consent was obtained for this case report.
Contributor Information
Nicharee Kitkarncharoensin, Email: nicharee.kit@student.mahidol.edu.
Sarin Catithammanit, Email: sarin.cat@student.mahidol.edu.
Bantita Phruttinarakorn, Email: Bantita.phr@mahidol.ac.th.
Piya Cherntanomwong, Email: piya.che@mahidol.ac.th.
Taya Kitiyakara, Email: tayakiti@gmail.com.
REFERENCES
- 1.Israelski M, Simchowitz H. 1. Roentgendiagnostic der esophagus varizen. Laryngol Rhinol Otol. 1932;22:208–18. [Google Scholar]
- 2.Ali H, Pamarthy R, Bolick NL, Ali E, Paleti S, Kapuria D. Downhill esophageal varices: A systematic review of the case reports. Explor Med. 2022;3(4):317–30. [Google Scholar]
- 3.Loudin M, Anderson S, Schlansky B. Bleeding ‘downhill’ esophageal varices associated with benign superior vena cava obstruction: Case report and literature review. BMC Gastroenterol. 2016;16(1):134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Savoy AD, Wolfsen HC, Paz-Fumagalli R, Raimondo M. Endoscopic therapy for bleeding proximal esophageal varices: A case report. Gastrointest Endosc. 2004;59(2):310–3. [DOI] [PubMed] [Google Scholar]
- 5.Tanaka H, Nakahara K, Goto K. Two cases of downhill esophageal varices associated with superior vena cava syndrome due to lung cancer [in Japanese]. Nihon Kyobu Shikkan Gakkai Zasshi. 1991;29(11):1484–8. [PubMed] [Google Scholar]
- 6.Subramaniam R, Madanagopalan N, Krishnan KT, Padmanabhan C. A case of anaplastic bronchogenic carcinoma with “downhill varices” of the esophagus. Dis Chest. 1967;51(5):545–9. [DOI] [PubMed] [Google Scholar]
- 7.Kokubo M, Sasaki H, Sakai S, Murakawa S, Mori Y, Hirose H. Downhill esophageal varices due to superior vena cava syndrome [in Japanese]. Nihon Kyobu Shikkan Gakkai Zasshi. 1991;29(7):854–7. [PubMed] [Google Scholar]
- 8.Chauvin RN, Arbour GT, Hagood L, Borhan F. Downhill varices due to chemotherapy and radiation therapy in non-small cell carcinoma of the lung: 74. Am J Coll Gastroenterol. 2006;101:S67. [Google Scholar]
- 9.Berkowitz JC, Bhusal S, Desai D, Cerulli MA, Inamdar S. Downhill esophageal varices associated with central venous catheter-related thrombosis managed with endoscopic and surgical therapy. ACG Case Rep J. 2016;3(4):e102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chakinala RC, Kumar A, Barsa JE, et al. Downhill esophageal varices: A therapeutic dilemma. Ann Transl Med. 2018;6(23):463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bédard EL, Deslauriers J. Bleeding “downhill” varices: A rare complication of intrathoracic goiter. Ann Thorac Surg. 2006;81(1):358–60. [DOI] [PubMed] [Google Scholar]
- 12.Yaşar B, Kılıçoğlu G. Behçet's disease-related superior vena cava syndrome and bleeding downhill varices: A rare complication. Ulus Travma Acil Cerrahi Derg. 2017;23(2):170–2. [DOI] [PubMed] [Google Scholar]
- 13.Gholam S, Ghazala S, Pokhrel B, Desai AP. A rare case of downhill esophageal varices in the absence of superior vena cava obstruction. Am J Gastroenterol. 2017;112(3):413. [DOI] [PubMed] [Google Scholar]
- 14.Harwani YP, Kumar A, Chaudhary A, et al. Combined uphill and downhill varices as a consequence of rheumatic heart disease: A unique presentation. J Clin Exp Hepatol. 2014;4(1):63–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Maton PN, Allison DJ, Chadwick VS. “Downhill” esophageal varices and occlusion of superior and inferior vena cavas due to a systemic venulitis. J Clin Gastroenterol. 1985;7(4):331–7. [DOI] [PubMed] [Google Scholar]
- 16.Serin E, Ozer B, Gümürdülü Y, Yildirim T, Barutçu O, Boyacioglu S. A case of Castleman's disease with “downhill” varices in the absence of superior vena cava obstruction. Endoscopy. 2002;34(2):160–2. [DOI] [PubMed] [Google Scholar]
- 17.Shirakusa T, Iwasaki A, Okazaki M. Downhill esophageal varices caused by benign giant lymphoma. Case report and review of downhill varices cases in Japan. Scand J Thorac Cardiovasc Surg. 1988;22(2):135–8. [DOI] [PubMed] [Google Scholar]
- 18.Yasar B, Abut E. A case of mediastinal fibrosis due to radiotherapy and ‘downhill’ esophageal varices: A rare cause of upper gastrointestinal bleeding. Clin J Gastroenterol. 2015;8(2):73–6. [DOI] [PubMed] [Google Scholar]
- 19.Basar N, Cagli K, Basar O, et al. Upper-extremity deep vein thrombosis and downhill esophageal varices caused by long-term pacemaker implantation. Tex Heart Inst J. 2010;37(6):714–6. [PMC free article] [PubMed] [Google Scholar]
- 20.Ibis M, Ucar E, Ertugrul I, et al. Inferior thyroid artery embolization for downhill varices caused by a goiter. Gastrointest Endosc. 2007;65(3):543–5. [DOI] [PubMed] [Google Scholar]
- 21.van der Veldt AA, Hadithi M, Paul MA, van den Berg FG, Mulder CJ, Craanen ME. An unusual cause of hematemesis: Goiter. World J Gastroenterol. 2006;12(33):5412–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Orikasa H, Ejiri Y, Suzuki S, et al. A case of Behçet's disease with occlusion of both caval veins and “downhill” esophageal varices. J Gastroenterol. 1994;29(4):506–10. [DOI] [PubMed] [Google Scholar]
- 23.Inoue Y, Sakai S, Aoki T. Downhill oesophageal varices resulting from superior vena cava graft occlusion after resection of a thymoma. Interact Cardiovasc Thorac Surg. 2013;17(3):598–600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mikkelsen WJ. Varices of the upper esophagus in superior vena caval obstruction. Radiology. 1963;81:945–8. [DOI] [PubMed] [Google Scholar]
- 25.Yuan A, Yang PC, Yu CJ, et al. Tumor angiogenesis correlates with histologic type and metastasis in non-small-cell lung cancer. Am J Respir Crit Care Med. 1995;152(6 Pt 1):2157–62. [DOI] [PubMed] [Google Scholar]
- 26.Daum S, Hagen H, Naismith E, Wolf D, Pircher A. The role of anti-angiogenesis in the treatment landscape of non-small cell lung cancer: New combinational approaches and strategies of neovessel inhibition. Front Cell Dev Biol. 2020;8:610903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hung MS, Chen IC, Lin PY, et al. Epidermal growth factor receptor mutation enhances expression of vascular endothelial growth factor in lung cancer. Oncol Lett. 2016;12(6):4598–604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Imoto H, Osaki T, Taga S, Ohgami A, Ichiyoshi Y, Yasumoto K. Vascular endothelial growth factor expression in non-small-cell lung cancer: Prognostic significance in squamous cell carcinoma. J Thorac Cardiovasc Surg. 1998;115(5):1007–14. [DOI] [PubMed] [Google Scholar]


