Abstract
Autism spectrum disorder (ASD) has evolved from a narrow and rare childhood-onset disorder to a widely publicized and researched lifelong disease recognized as common and significantly heterogeneous. Researchers have suggested that gastrointestinal symptoms in ASD may be a manifestation of an underlying inflammatory process. However, there is a lack of bibliometric analysis of ASD and gut microbiota in children. Accordingly, this study conducts a bibliometric analysis of ASD and gut microbiota in children from 2000 to 2023, explores the current status and cutting-edge trends in the field of ASD and gut microbiota in children, and identifies new directions for future research. The literature on ASD and gut microbiota in children was screened using the Web of Science Core Collection from 2000 to 2023. Annual publications, countries, institutions, authors, journals, keywords, and references were visualized and analyzed using CiteSpace 5.8. R3 and VOSviewer1.6.18. This study included 1071 publications. Since the beginning of 2011, the overall number of articles shows an upward trend. The most productive country and institution are the United States and the University of California system, respectively. The most frequently cited author is Kang Dae-Wook, with 790 citations, who has contributed significantly to this field. Timothy Dinan is the most prolific author, with 34 articles. The journal with the most published articles on this topic is Nutrients, whereas PLOS One is the most cited journal. The most used keyword is “gut microbiota,” and the reference for the highest outbreak intensity is Hsiao. The research hotspots and trends predicted in this study provide a reference for further in-depth research in this field.
Keywords: autism, autism spectrum disorder, bibliometric analysis, gut microbiota, visual analysis
1. Introduction
Autism spectrum disorder (ASD) refers to a group of neurodevelopmental disorders, including autism, Asperger’s syndrome (AS), and pervasive developmental disorder not otherwise specified (PDD-NOS).[1] ASD is a term used to describe a group of early-appearing social communication deficits and repetitive sensorimotor behaviors associated with a strong genetic component and other causes.[2] The prevalence of ASD in children and adolescents is 0.36% in Asia[3] and 1.85% in Western countries.[4]
Gut microbiota is closely related to an individual’s age. It regulates the host’s physiology and metabolism through different mechanisms.[5] Evidence from animal models and human subjects suggests that microbiota dysbiosis of the gastrointestinal system and its metabolites are implicated in the creation and development of ASD.[6–10] Several recent studies have found significant changes in the composition of gut microbiota in children with ASD,[11,12] suggesting that gastrointestinal symptoms in ASD may be a manifestation of an underlying inflammatory process.[13] As the gut microbiome is related to brain functioning through the gut–brain axis, researchers have hypothesized that an underdeveloped gut microbiome may be related to ASD.[14]
Bibliometric analysis is a statistical and quantitative tool used to study publications.[15] Bibliometrics can specify the most influential countries, institutions, and authors in a particular field by identifying relevant publications, providing researchers with potentially valuable qualitative and quantitative information.[16] CiteSpace and VOSviewer are bibliometric and visualization software packages that play an important role in analyzing the current state of scientific research, exploring the forefront of disciplines, and selecting research directions.[17,18] However, there is limited bibliometric research on pediatric ASD and gut microbiota. Based on bibliometric analysis, this study develops a knowledge map of ASD and gut microbiota, providing insights for researchers to discover new themes and directions. This study has the following objectives: identify the main contributors in the field of ASD and pediatric gut microbiota, including authors, institutions, and countries; explore the current research status and future development trends; and provide new directions for subsequent research on ASD and gut microbiota in children.
2. Methods
We reviewed papers published between January 1, 2000, and August 20, 2023, on the Web of Science Core Collection (WoSCC). The search strategies were as follows: TS = (“autism spectrum disorder” OR “autism” OR “autistic disorder”) AND TS = (“intestinal flora” OR “gut microbiota” OR “intestinal microflora” OR “intestinal microbiota” OR “gut microbiome” OR “gut flora” OR “intestinal bacteria” OR “gut microflora”) AND TS = (“children” OR “childhood” OR “pediatric”); indices = WoSCC—Science Citation Index Expanded (SCIE); language = English; document types included original and review articles; procedure papers, book chapters, data papers, early access papers, and retracted publications were excluded from the search (Fig. 1).
Figure 1.
Flowchart of literature identification and selection.
In total, 1071 relevant articles were identified. The “fully recorded and cited references” of these documents were excerpted in CiteSpace 5.8.R3 and VOSviewer 1.6.18, which adopt a “plain text” format to identify major countries, institutions, authors, journals, keywords, and references.
3. Results
3.1. Annual numbers of publications
From 2000 to 2023, the WoSCC published 1071 publications on ASD and gut microbiota in children, including 593 original articles and 478 review articles. As shown in Figure 2, the number of annual publications on pediatric ASD and gut microbiota has been increasing over the past 23 years, suggesting that the connection between pediatric ASD and gut microbiota is increasingly being confirmed with medical progress. Since 2011, there has been a remarkable increase in the number of articles published, especially from 2017 to 2018, when the annual number of published articles reached 37. From 2019 to 2020, the number of published articles increased to 47. In recent years, the subject’s popularity has remained steady, with 45 articles published between 2021 and 2022. Thus, the relationship between gut microbiota and ASD has received widespread attention in the medical community.
Figure 2.
Trends of publications in the field of autism spectrum disorder and gut microbiota in children (2000–2023).
3.2. Analysis of countries and institutions
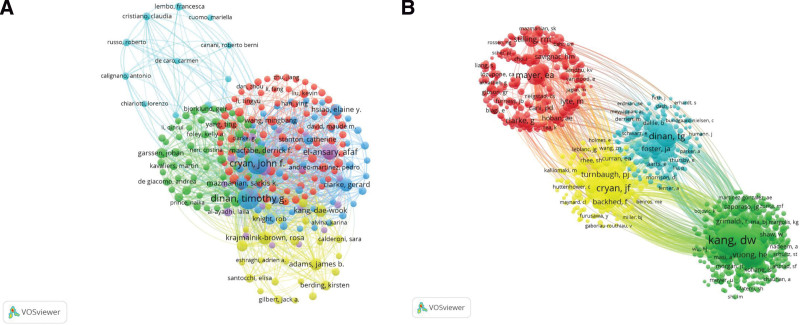
CiteSpace and VOSviewer were used to map the cooperation between countries (Fig. 3A and B) and institutions (Fig. 3C and D). Tables 1 and 2 list the top 5 countries and institutions in the field of ASD and gut microbiota in children, respectively. The US has made the largest contribution (n = 322, 21.9%), followed by China, Italy, Canada, and Ireland. The institutions with the largest number of papers were the University of California system (US), followed by University College Cork (Ireland). As shown in Figure 3A and C, the US institutions, including the University of California system (marked with purple circles), exhibit high centrality, indicating that they promote research in the fields of ASD and gut microbiota in children. In Figure 3B, different colors represent clusters of close relationships. Countries and close cooperation can be divided into 5 and 6 types, respectively. The red part indicates greater cooperation between the US and Italy, China, and Spain. The green part indicates that Saudi Arabia has close relations with Egypt and cooperates with Norway and Russia. The purple part indicates that France often cooperates with Canada and the Netherlands. The yellow part shows that Ireland cooperates more with Finland, Sweden, and Germany. Finally, the blue part indicates that Australia has close relations with England and India. In Figure 3D, the red part shows that Harvard Medical School frequently cooperates with the Chinese Academy of Sciences, Massachusetts General Hospital, and Peking University; the green part shows that University College Cork and the University of California, Los Angeles, cooperate more closely. Further, the navy part shows that the University of California, Davis cooperates closely with the University of Western Ontario; the yellow part shows that the University of California, San Diego is closely related to the University of Toledo. Additionally, the purple part shows that King Saud University closely cooperates with the National Research Center in Egypt; the blue part shows that the University of Oxford and the Medical University of Warsaw cooperate closely with each other.
Figure 3.
(A) Analysis of countries and institutions: CiteSpace network map of countries involved in studying autism spectrum disorder and gut microbiota in children. (B) Analysis of countries and institutions: VOSviewer network map of countries involved in studying autism spectrum disorder and gut microbiota in children. (C). Analysis of countries and institutions: CiteSpace network map of institutions involved in studying autism spectrum disorder and gut microbiota in children. (D) Analysis of countries and institutions: VOSviewer network map of institutions involved in studying autism spectrum disorder and gut microbiota in children.
Table 1.
Top 5 countries with the most publications.
| Rank | Country | Count (%) | Rank | Country | Centrality |
|---|---|---|---|---|---|
| 1 | USA | 322 (21.9%) | 1 | USA | 0.58 |
| 2 | CHINA | 185 (12.6%) | 2 | ENGLAND | 0.26 |
| 3 | ITALY | 108 (7.4%) | 3 | SPAIN | 0.22 |
| 4 | CANADA | 57 (3.9%) | 4 | NORWAY | 0.13 |
| 5 | IRELAND | 54 (3.7%) | 5 | AUSTRALIA | 0.11 |
Table 2.
Top 5 institutions with the most publications.
| Rank | Institution | Count (%) | Rank | Institution | Centrality |
|---|---|---|---|---|---|
| 1 | University of California System | 57 (8.7%) | 1 | University of California System | 0.44 |
| 2 | University College Cork | 41 (6.2%) | 2 | UDICE-French Research Universities | 0.20 |
| 3 | Harvard University | 35 (5.3%) | 3 | Centre National de la Recherche Scientifique (CNRS) | 0.17 |
| 4 | Harvard Medical School | 23 (3.5%) | 4 | Egyptian Knowledge Bank (EKB) | 0.15 |
| 5 | UDICE-French Research Universities | 20 (3.0%) | 5 | INRAE | 0.14 |
3.3. Analysis of authors and cited authors
The top 5 authors in the field of ASD and gut microbiota in children are shown in Table 3. Figure 4A shows the clusters of close cooperation among authors in different colors. Although Cryan, El-Ansary, Krajmalnik-Brown, Dinan, and Bhat are in different clusters, they cooperated with each other. The 5 most frequently cited authors were from the US, Australia, and Ireland (Table 4). The top 3 cited authors, Kang (790), Finegold (760), and Hsiao (504), belong to different cooperative groups and occupy core positions (Fig. 4B).
Table 3.
Five most-published authors.
| Rank | Author | Publications | Country | Institution |
|---|---|---|---|---|
| 1 | Dinan, Timothy | 34 | Ireland | University College Cork |
| 2 | Cryan, John F | 33 | Ireland | University College Cork |
| 3 | El-Ansary, Afaf | 19 | Saudi Arabia | King Saud University |
| 4 | Bhat, Ramesa Shafi | 14 | Saudi Arabia | King Saud University |
| 5 | Krajmalnik-Brown, Rosa | 13 | USA | Arizona State University |
Figure 4.
(A) Analysis of authors: visualization of authors. (B) Analysis of authors: visualization of cited authors.
Table 4.
Five most-cited authors.
| Rank | Cited author | Cited frequency | Country | Institution |
|---|---|---|---|---|
| 1 | Kang, Dae-Wook | 790 | USA | University of Toledo |
| 2 | Finegold, Sydney M | 760 | USA | VA Medical Center West Los Angeles |
| 3 | Hsiao, Elaine Y | 504 | USA | University of California |
| 4 | Wang, Lv | 440 | Australia | University of South Australia |
| 5 | Cryan, John F | 420 | Ireland | University College Cork |
3.4. Analysis of journals and cited journals
As illustrated in Table 5, Nutrients (impact factor [IF] 5.9) was the journal with the most relevant published articles, with 37 papers. The International Journal of Molecular Sciences ranked second, with 35 papers, and Frontiers in Psychiatry ranked third, with 27 papers. Table 6 illustrates that PLOS One is the most cited journal. In the journal co-occurrence map in Figure 5A, the first node is the largest. This shows that PLOS One is the core journal with the highest number of papers in the field of ASD and gut microbiota in children, indicating that its comprehensive influence is superior to that of other journals. The overlay of graphs representing research in ASD and gut microbiota in children in Figure 5B illustrates the distribution of relationships between journals, with citing journals on the left and cited journals on the right.[19,20] The green path indicates that papers published in the medicine/medical/clinical fields are often cited in molecular/biology/genetics research.
Table 5.
Five most-published journals.
| Rank | Journal | Publications (%) | IF (JCR 2022) | JCR quartile |
|---|---|---|---|---|
| 1 | Nutrients | 37 (8.2%) | 5.9 | Q1 |
| 2 | International Journal of Molecular Sciences | 35 (7.8%) | 5.6 | Q1 |
| 3 | Frontiers in Psychiatry | 27 (6%) | 4.7 | Q2 |
| 4 | Scientific Reports | 21 (4.7%) | 4.6 | Q2 |
| 5 | Brain Behavior and Immunity | 20 (4.4%) | 15.1 | Q1 |
Table 6.
Five most-cited journals.
| Rank | Cited journal | Citations | IF (JCR 2022) | JCR quartile |
|---|---|---|---|---|
| 1 | PLOS One | 3251 | 3.7 | Q2 |
| 2 | Nature | 2502 | 64.8 | Q1 |
| 3 | Cell | 2258 | 64.5 | Q1 |
| 4 | Proceedings of the National Academy of Sciences of the United States of America | 2234 | 11.1 | Q1 |
| 5 | Brain Behavior and Immunity | 2080 | 15.1 | Q1 |
Figure 5.
(A) Analysis of journals: visualization of cited journals. (B) Analysis of journals: overlay of graphs representing research in autism spectrum disorder and gut microbiota in children.
3.5. Analysis of keywords
As illustrated in Table 7, the 10 keywords with the highest frequency in the past 20 years are as follows: “gut microbiota” (492), “children” (326), “autism” (325), “autism spectrum disorder” (312), “microbiota” (212), “brain” (188), “intestinal microbiota” (179), “microbiome” (150), “autism spectrum disorders” (146), and “chain fatty acids” (146). As illustrated in Figure 6A, the keywords can be divided into 5 clusters according to color: red = “autism spectrum disorders,” “intestinal microbiota,” “chain fatty acids,” “microbiome,” “irritable bowel syndrome”; green = “gut microbiota,” “brain,” “behavior,” “oxidative stress,” “risk”; blue = “autism spectrum disorder,” “mouse model,” “brain development”; yellow = “microbiota,” “stress,” “social behavior,” “spectrum disorders,” “anxiety-like behavior”; purple = “children,” “health,” “impact,” “association”; orange = “inflammation,” “obesity,” “probiotics,” “infection,” “blood-brain barrier.” Through burst detection analysis of keywords, 25 co-occurring words were identified. The results are presented in Figure 6B. “Onset of autism” is the keyword related to the earliest outbreak. “Microflora,” “spectrum disorders,” “inflammatory bowel disease,” and “propionic acid” emerged between 2008 and 2013. “Intestinal microbiota,” “anxiety-like behavior,” “irritable bowel syndrome,” “immune system,” “diversity,” “activation,” and “ketogenic diet” emerged from 2014 to 2019. The final keyword is “akkermansia mucinephila.” The keyword with the highest strength is “intestinal microbiota,” with a score of 20.08, followed by “anxiety-like behavior,” with a score of 11.56. We used CiteSpace software to cluster the keywords and draw a timeline after clustering (Fig. 7). In total, 8 clusters were formed: #1 disease, #2 irritable bowel syndrome, #3 maternal immune activation, #4 autism spectrum disorder, #5 autism spectrum disorders, #6 depression, #7 onset of autism, and #8 fecal metabolomics.
Table 7.
Top 20 most frequently used keywords.
| Rank | Keyword | Occurrences |
|---|---|---|
| 1 | Gut Microbiota | 492 |
| 2 | Children | 326 |
| 3 | Autism | 325 |
| 4 | Autism Spectrum Disorder | 312 |
| 5 | Microbiota | 212 |
| 6 | Brain | 188 |
| 7 | Intestinal Microbiota | 179 |
| 8 | Microbiome | 150 |
| 9 | Autism Spectrum Disorders | 146 |
| 10 | Chain Fatty-Acids | 146 |
| 11 | Gut–Brain Axis | 140 |
| 12 | Probiotics | 136 |
| 13 | Behavior | 115 |
| 14 | Inflammation | 110 |
| 15 | Health | 101 |
| 16 | Gastrointestinal Symptoms | 94 |
| 17 | Gut Microbiome | 91 |
| 18 | Irritable-Bowel-Syndrome | 80 |
| 19 | Dysbiosis | 78 |
| 20 | Spectrum Disorders | 78 |
Figure 6.
(A) Cluster analysis and visualization of keywords. (B) Analysis of keywords: top 25 keywords with the strongest citation bursts.
Figure 7.
Timeline view of keywords.
3.6. Analysis of references
Table 8 lists the 10 most frequently cited references. The most frequently cited article was published by Kang et al[21] in Microbiome in 2017, followed by Strati et al[10] in Microbiome in 2017. The results of the top 25 articles based on the burst detection analysis are illustrated in Figure 8. The article with the highest burst strength was published in BMC Gastroenterology by Adams et al[22] in 2011 (26.34), followed by Finegold et al[11] in Anaerobe in 2010 (23.91).
Table 8.
Top 10 references with the highest frequency of occurrence.
| Rank | Citations | Author | Title | Journal | Year | DOI |
|---|---|---|---|---|---|---|
| 1 | 216 | Kang DW | Microbiota Transfer Therapy alters gut ecosystem and improves gastrointestinal and autism symptoms: an open-label study | Microbiome | 2017 | 10.1186/s40168-016-0225-7 |
| 2 | 199 | Strati F | New evidences on the altered gut microbiota in autism spectrum disorders | Microbiome | 2017 | 10.1186/s40168-017-0242-1 |
| 3 | 170 | Sharon G | Human Gut Microbiota from Autism Spectrum Disorder Promote Behavioral Symptoms in Mice | Cell | 2019 | 10.1016/j.cell.2019.05.004 |
| 4 | 145 | Hsiao EY | Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders | Cell | 2013 | 10.1016/j.cell.2013.11.024 |
| 5 | 131 | Kang DW | Long-term benefit of Microbiota Transfer Therapy on autism symptoms and gut microbiota | Scientific Reports | 2019 | 10.1038/s41598-019-42183-0 |
| 6 | 124 | Vuong HE | Emerging Roles for the Gut Microbiome in Autism Spectrum Disorder | Biological Psychiatry | 2017 | 10.1016/j.biopsych.2016.08.024 |
| 7 | 123 | Buffington SA | Microbial Reconstitution Reverses Maternal Diet-Induced Social and Synaptic Deficits in Offspring | Cell | 2016 | 10.1016/j.cell.2016.06.001 |
| 8 | 116 | Tomova A | Gastrointestinal microbiota in children with autism in Slovakia | Physiology & Behavior | 2015 | 10.1016/j.physbeh.2014.10.033 |
| 9 | 106 | Kang DW | Differences in fecal microbial metabolites and microbiota of children with autism spectrum disorders | Anaerobe | 2018 | 10.1016/j.anaerobe.2017.12.007 |
| 10 | 105 | Liu SM | Altered gut microbiota and short chain fatty acids in Chinese children with autism spectrum disorder | Scientific Reports | 2019 | 10.1038/s41598-018-36430-z |
Figure 8.
Top 25 most cited references with the strongest citation bursts.
4. Discussion
A mutual correlation between ASD and alterations in microbiota has been confirmed in many animal and human studies.[23] Srikantha and Mohajeri demonstrated significant changes in the microbial composition of the autistic population in their review.[24] In recent years, research on the mechanism and treatment of ASD has become closely related to studies on gut microbiota, establishing a significant relationship between ASD and gut microbiota. In this study, CiteSpace 5.8.R3 and VOSviewer1.6.18 were used to identify 1017 relevant publication published in the WoSCC database from 2000 to 2023 for statistical analysis and visualization. Subsequently, the current situation and trends related to number of annual publications, countries, institutions, authors, journals, and research in this field were analyzed. Results showed that in the past 2 decades, publications related to ASD and gut microbiota have been on the rise, and the number of publications increased between 2012 and 2020. The number of publications in 2022 was 10 times that of 2013, indicating that this field has attracted increasing attention over time.
Regarding countries and institutions, most research in this field comes from the US, China, and Italy. A central value > 0.1 indicates a relatively important node. As illustrated in Figure 3A and C, the distribution of countries and institutions is scattered, and the centrality of each institution is < 0.1, indicating that international cooperation and academic exchanges are limited. The US has the most publications, with 3 of the top 5 institutions dedicated to this field located in the US. The US is also a seat of multicentered cooperation, and collaborations can be found with Italy, Canada, and Ireland. Although China has the second-largest number of publications, it does not exhibit the highest concentration in terms of country and institution, suggesting limited cooperation with other countries. However, England and Spain, despite having fewer publications, demonstrate high degrees of centrality and cooperation.
To the best of our knowledge, Dinan, from University College Cork, is the first and most prolific author in this field. The second-highest number of publications corresponds to Cryan, who is also from University College Cork and part of the same research team as Dinan. They mainly studied the impact of the brain–gut axis on the mental nervous system,[25–30] including the treatment and mechanism of ASD and gut microbiota in children. Kang is the most frequently cited author (790 citations). He is primarily involved in mechanistic and clinical research on the correlation between ASD and gut microbiota in children.[21,31] Moreover, he worked closely with experts from various cooperative groups and made important contributions to the study of ASD and gut microbiota.
Nutrients (IF 5.9 Q1) is the journal with the most published articles in this field, followed by The International Journal of Molecular Sciences (IF 5.6 Q1). The publication volumes of the top 5 journals are considerably similar, and the IFs and partitions are not different, except for the IF of Brain, Behavior, and Immunity, which is 15.1, indicating that the research direction and field are of high quality. PLOS One (IF 3.7 Q2) is the most frequently cited journal, with 3251 citations. Four of the 5 journals in Table 6 are from Q1, revealing that research on ASD and gut microbiota in children has been highly valued by scholars worldwide.
An analysis of the references demonstrates that Kang published the most frequently cited paper, which revealed the therapeutic significance of gut microbiota in children with ASD.[21] The highest burst strength corresponded to Adams et al, followed by Finegold.[11] Adams et al[22] found a strong correlation between gastrointestinal symptoms and the severity of autism, indicating that children with more severe autism may have more severe gastrointestinal symptoms and vice versa. Symptoms of autism may worsen, even partially, owing to potential gastrointestinal problems.
Through keyword co-occurrence and clustering analysis, we have found a close relationship between gut microbiota and symptoms of ASD in children, such as neurodevelopmental disorders, cognitive impairment, anxiety, depression and gastrointestinal problems. Horvath et al[32] found that most children with autism had varying degrees of gastrointestinal problems, such as diarrhea, constipation, and gas. Although they did not clarify whether gastrointestinal inflammation is a unique multi-organ process or an autoimmune process, they suggested that clinicians can use traditional gastrointestinal treatment regimens to treat most gastrointestinal symptoms in children with autism. Subsequently, research on autism and gastrointestinal problems has increased, and other studies have found that alterations in the gut microbiota composition of children with ASD may contribute to gastrointestinal and central nervous system symptoms.[33,34] At the same time, modern medicine is exploring ASD’s pathogenesis from the perspective of molecular biology and genetics. Specifically, the pathogenesis of ASD in children caused by gut microbiota imbalance is considered to be the key focus of future research. Here, recent research has found that the causes of ASD are complex. Proposing the concept of the “brain-gut axis,” researchers generally believe that gut microbiota may act on the brain through the “gut microbiota-gut-brain axis” in neural, immune, endocrine, and metabolic pathways,[35] which may be an important mechanism affecting the occurrence and development of ASD. The role of gut microbiota in children with ASD is related to intestinal barrier dysfunction, neuroinflammation, and microglial overactivation.[36] In particular, gut microbiota and their metabolites, such as neurotransmitters (dopamine, gamma-aminobutyric, serotonin, norepinephrine, acetylcholine),[37,38] amino acids (phenylalanine, tryptophan), short-chain fatty acids (acetic acid, propionic acid, butyric acid), and phenol compounds, are important contributors.[39–41] Based on the previous studies, we can conclude that significant abnormalities in bacterial strain abundance and changes in bacterial metabolite levels may be markers for disease diagnosis and subsequent treatment in the future.
The current study has used keyword burst detection to analyze the cutting-edge research topics in the field of gut microbiota and ASD. Firstly, the “microbiota-gut-brain axis” and therapeutic methods for treating it are advancing research frontiers in this field. Probiotics, one of the most studied methods, has achieved positive results in animal models; probiotic L. reuteri has been proven to effectively improve social behavior in autism mouse models, although clinical evidence is still limited.[42] A recent study proposed that supplementing Bifidobacteria longum can down-regulate the expression of intestinal amino acid transporters and alleviate autism-like behavior in Chd8+/− mouse models.[43] In addition to probiotics, fecal microbiota transplantation has also been proven effective in treating ASD. The clinical trials of Kang et al[21,44,45] have figured out that fecal microbiota transplantation could restore the diversity and richness of intestinal bacteria in ASD participants, and the improvement of ASD core symptoms and gastrointestinal symptoms can still be observed after the end of treatment. Besides, a well tolerance is enrolled. Secondly, researchers have found the potential link between ASD and major depressive disorder. Approximately 26% of patients with ASD suffer from depression, with the risk of depression in patients with ASD being 3 times higher than in the general population.[46,47] The microbiota-gut-brain axis is involved in the pathophysiology of ASD and major depressive disorder, based on neuroinflammatory processes leading to neuronal cell death.[48] A characteristic feature common to both diseases is the presence of more pro-inflammatory bacterial strains and fewer anti-inflammatory bacterial strains.[49] Moreover, the decreased production of short-chain fatty acids plays a significant role in the pathogenesis of both diseases.[50] Therefore, the potential mechanism between depression and ASD deserves further study. The role of Akkermansia muciniphila (A. muciniphila) in pediatric ASD is also a forefront topic of investigation. Newell et al[51] reported that a ketogenic diet alleviated BTRB mouse autism-like behavior by increasing the abundance of A. muciniphila in the gut microbiota, and other evidence suggests that A. muciniphila can maintain gut barrier integrity and regulate inflammation.[52] Studies on humans and rodents have found a decrease in A. muciniphila abundance and increased gut permeability in children with ASD.[53,54] Collectively, these findings suggest that A. muciniphila may be important in the pathophysiology of pediatric and adolescent ASD.
Figure 6B illustrates that the phrase “onset of autism” has remained strong for over a decade but has gradually replaced due to the new definition of ASD in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). Research hotspots in recent years have been the fields of fecal microbiota transplantation, spectrum disorders, and major depressive disorders. Figure 7 illustrates the timeline view of keywords before 2010, demonstrating that research on autism and gut microbiota was mostly lackluster. After 14 years of preparation, the DSM-5 was launched in the US in 2013. With the continuous deepening of research on ASD, focus is being directed to the study of irritable bowel syndrome, maternal immune activation, depression, and fecal metabolomics.
Although this study reveals important findings, it has some limitations. First, we only searched for publications from the WoSCC, which may have been incomplete. Second, the choice of language (English) and literature type (original or review articles) may have led to deviations in the results. Third, software limitations may have led to errors.
5. Conclusion
Using bibliometric methods, this study analyzed papers in the field of ASD and pediatric gut microbiota from 2000 to 2023. Although the number of studies in this field is limited, there is a clear upward trend. In recent years, the number of publications has increased, indicating that more and more scholars are paying attention to ASD and gut microbiota of children. From the results, it is evident that different countries, regions, and organizations need to deepen and strengthen cooperation. Most studies on autism and gut microbiota of children were published in internationally influential journals and cited, indicating that this research has received widespread attention. However, some questions remain unanswered, such as the exact function of gut microbiota and the mechanism of interaction between gut microbiota and autism in children. Therefore, these questions need further research in the future.
In conclusion, the current study provides core literature for scholars on the relationship between autism in children and gut microbiota. Furthermore, it can be used by scholars seeking academic cooperation, provides direction for journal publication, and guidance for new ideas and development in this field for clinicians and researchers.
Acknowledgments
We would like to thank Editage (www.editage.cn) for English language editing.
Author contributions
Conceptualization: Xing-Ruo Gong, Xiao-Rui You, Bing-Xiang Ma.
Data curation: Xing-Ruo Gong, Xiao-Rui You, Xue-Ying Ding.
Software: Xing-Ruo Gong, Mei-Ran Guo, Xue-Ying Ding.
Visualization: Xing-Ruo Gong, Mei-Ran Guo.
Writing – original draft: Xing-Ruo Gong, Xiao-Rui You.
Writing – review & editing: Xiao-Rui You, Bing-Xiang Ma.
Abbreviations:
- AS
- Asperger’s syndrome
- ASD
- autism spectrum disorder
- DSM-5
- Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition
- PDD-NOS
- pervasive developmental disorder not otherwise specified
- WoSCC
- Web of Science Core Collection
This study was funded by Traditional Chinese Medicine, a characteristic backbone discipline in Henan Province (STG-ZYX03-202129).
Ethics approval is not required because individual patient data and privacy were not involved in this study.
The authors have no conflicts of interest to disclose.
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
How to cite this article: Gong X-R, You X-R, Guo M-R, Ding X-Y, Ma B-X. Children autism spectrum disorder and gut microbiota: A bibliometric and visual analysis from 2000 to 2023. Medicine 2023;102:52(e36794).
Contributor Information
Xing-Ruo Gong, Email: gxr0531@126.com.
Xiao-Rui You, Email: yxr1026@163.com.
Mei-Ran Guo, Email: 1432041651@qq.com.
Xue-Ying Ding, Email: 942216145@qq.com.
References
- [1].Sharma SR, Gonda X, Tarazi FI. Autism spectrum disorder: classification, diagnosis and therapy. Pharmacol Ther. 2018;190:91–104. [DOI] [PubMed] [Google Scholar]
- [2].Lord C, Elsabbagh M, Baird G, et al. Autism spectrum disorder. Lancet. 2018;392:508–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Qiu S, Lu Y, Li Y, et al. Prevalence of autism spectrum disorder in Asia: a systematic review and meta-analysis. Psychiatry Res. 2020;284:112679. [DOI] [PubMed] [Google Scholar]
- [4].Knopf A. Autism prevalence increases from 1 in 60 to 1 in 54: CDC. Brown Univ Child Adolesc Behav Lett. 2020;36:4. [Google Scholar]
- [5].Fattorusso A, Di Genova L, Dell’Isola GB, et al. Autism spectrum disorders and the gut microbiota. Nutrients. 2019;11:521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Hsiao EY, McBride SW, Hsien S, et al. Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell. 2013;155:1451–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Li Q, Zhou JM. The microbiota-gut-brain axis and its potential therapeutic role in autism spectrum disorder. Neuroscience. 2016;324:131–9. [DOI] [PubMed] [Google Scholar]
- [8].Li N, Yang J, Zhang J, et al. Correlation of gut microbiome between ASD children and mothers and potential biomarkers for risk assessment. Genomics Proteomics Bioinformatics. 2019;17:26–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Larroya-Garcia A, Navas-Carrillo D, Orenes-Pinero E. Impact of gut microbiota on neurological diseases: diet composition and novel treatments. Crit Rev Food Sci Nutr. 2019;59:3102–16. [DOI] [PubMed] [Google Scholar]
- [10].Strati F, Cavalieri D, Albanese D, et al. New evidences on the altered gut microbiota in autism spectrum disorders. Microbiome. 2017;5:24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Finegold SM, Dowd SE, Gontcharova V, et al. Pyrosequencing study of fecal microflora of autistic and control children. Anaerobe. 2010;16:444–53. [DOI] [PubMed] [Google Scholar]
- [12].De Angelis M, Piccolo M, Vannini L, et al. Fecal microbiota and metabolome of children with autism and pervasive developmental disorder not otherwise specified. PLoS One. 2013;8:e76993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Navarro F, Liu Y, Rhoads JM. Can probiotics benefit children with autism spectrum disorders? World J Gastroenterol. 2016;22:10093–102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Wan Y, Zuo T, Xu Z, et al. Underdevelopment of the gut microbiota and bacteria species as non-invasive markers of prediction in children with autism spectrum disorder. Gut. 2022;71:910–8. [DOI] [PubMed] [Google Scholar]
- [15].Luo F, Li Y, Zhang Y, et al. Bibliometric analysis of IgA vasculitis nephritis in children from 2000 to 2022. Front Public Health. 2022;10:1020231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Ai Y, Xing Y, Yan L, et al. Atrial fibrillation and depression: a bibliometric analysis from 2001 to 2021. Front Cardiovasc Med. 2022;9:775329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].van Eck NJ, Waltman L. Software survey: VOSviewer, a computer program for bibliometric mapping. Scientometrics. 2010;84:523–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Chen C, Song M. Visualizing a field of research: a methodology of systematic scientometric reviews. PLoS One. 2019;14:e0223994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Wang Z, Bai C, Hu T, et al. Emerging trends and hotspot in gut-lung axis research from 2011 to 2021: a bibliometrics analysis. Biomed Eng Online. 2022;21:27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Ma D, Yang B, Guan B, et al. A bibliometric analysis of pyroptosis from 2001 to 2021. Front Immunol. 2021;12:731933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Kang DW, Adams JB, Gregory AC, et al. Microbiota transfer therapy alters gut ecosystem and improves gastrointestinal and autism symptoms: an open-label study. Microbiome. 2017;5:10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Adams JB, Johansen LJ, Powell LD, et al. Gastrointestinal flora and gastrointestinal status in children with autism--comparisons to typical children and correlation with autism severity. BMC Gastroenterol. 2011;11:22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Inoue R, Sakaue Y, Sawai C, et al. A preliminary investigation on the relationship between gut microbiota and gene expressions in peripheral mononuclear cells of infants with autism spectrum disorders. Biosci Biotechnol Biochem. 2016;80:2450–8. [DOI] [PubMed] [Google Scholar]
- [24].Srikantha P, Mohajeri MH. The possible role of the microbiota-gut-brain-axis in autism spectrum disorder. Int J Mol Sci. 2019;20:2115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Cryan JF, O’Riordan KJ, Sandhu K, et al. The gut microbiome in neurological disorders. Lancet Neurol. 2020;19:179–94. [DOI] [PubMed] [Google Scholar]
- [26].Cryan JF, O’Riordan KJ, Cowan C, et al. The microbiota-gut-brain axis. Physiol Rev. 2019;99:1877–2013. [DOI] [PubMed] [Google Scholar]
- [27].Berding K, Vlckova K, Marx W, et al. Diet and the microbiota-gut-brain axis: sowing the seeds of good mental health. Adv Nutr. 2021;12:1239–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Morkl S, Butler MI, Holl A, et al. Probiotics and the microbiota-gut-brain axis: focus on psychiatry. Curr Nutr Rep. 2020;9:171–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Sherwin E, Bordenstein SR, Quinn JL, et al. Microbiota and the social brain. Science. 2019;366:eaar2016. [DOI] [PubMed] [Google Scholar]
- [30].Cruz-Pereira JS, Rea K, Nolan YM, et al. Depression’s unholy trinity: dysregulated stress, immunity, and the microbiome. Annu Rev Psychol. 2020;71:49–78. [DOI] [PubMed] [Google Scholar]
- [31].Sharon G, Cruz NJ, Kang DW, et al. Human gut microbiota from autism spectrum disorder promote behavioral symptoms in mice. Cell. 2019;177:1600–1618.e17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Horvath K, Perman JA. Autistic disorder and gastrointestinal disease. Curr Opin Pediatr. 2002;14:583–7. [DOI] [PubMed] [Google Scholar]
- [33].Diaz HR, Wang S, Anuar F, et al. Normal gut microbiota modulates brain development and behavior. Proc Natl Acad Sci U S A. 2011;108:3047–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Wang Y, Kasper LH. The role of microbiome in central nervous system disorders. Brain Behav Immun. 2014;38:1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].De Sales-Millán A, Aguirre-Garrido JF, González-Cervantes RM, et al. Microbiome-gut-mucosal-immune-brain axis and Autism Spectrum Disorder (ASD): a novel proposal of the role of the gut microbiome in ASD Aetiology. Behav Sci (Basel). 2023;13:548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Wang Q, Yang Q, Liu X. The microbiota-gut-brain axis and neurodevelopmental disorders. Protein Cell. 2023;14:762–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Wang L, Christophersen CT, Sorich MJ, et al. Elevated fecal short chain fatty acid and ammonia concentrations in children with autism spectrum disorder. Dig Dis Sci. 2012;57:2096–102. [DOI] [PubMed] [Google Scholar]
- [38].Chen Y, Xu J, Chen Y. Regulation of neurotransmitters by the gut microbiota and effects on cognition in neurological disorders. Nutrients. 2021;13:2099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Teskey G, Anagnostou E, Mankad D, et al. Intestinal permeability correlates with behavioural severity in very young children with ASD: a preliminary study. J Neuroimmunol. 2021;357:577607. [DOI] [PubMed] [Google Scholar]
- [40].Davies C, Mishra D, Eshraghi RS, et al. Altering the gut microbiome to potentially modulate behavioral manifestations in autism spectrum disorders: a systematic review. Neurosci Biobehav Rev. 2021;128:549–57. [DOI] [PubMed] [Google Scholar]
- [41].Zheng Y, Prince N, van Hattem C, et al. The interaction between intestinal bacterial metabolites and phosphatase and tensin homolog in autism spectrum disorder. Mol Cell Neurosci. 2023;124:103805. [DOI] [PubMed] [Google Scholar]
- [42].Kong XJ, Liu J, Li J, et al. Probiotics and oxytocin nasal spray as neuro-social-behavioral interventions for patients with autism spectrum disorders: a pilot randomized controlled trial protocol. Pilot Feasibility Stud. 2020;6:20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [43].Yu Y, Zhang B, Ji P, et al. Changes to gut amino acid transporters and microbiome associated with increased E/I ratio in Chd8+/- mouse model of ASD-like behavior. Nat Commun. 2022;13:1151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Kang DW, Adams JB, Vargason T, et al. Distinct fecal and plasma metabolites in children with autism spectrum disorders and their modulation after microbiota transfer therapy. mSphere. 2020;5:e00314–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Kang DW, Adams JB, Coleman DM, et al. Long-term benefit of Microbiota transfer therapy on autism symptoms and gut microbiota. Sci Rep. 2019;9:5821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Hossain MM, Khan N, Sultana A, et al. Prevalence of comorbid psychiatric disorders among people with autism spectrum disorder: an umbrella review of systematic reviews and meta-analyses. Psychiatry Res. 2020;287:112922. [DOI] [PubMed] [Google Scholar]
- [47].Hudson CC, Hall L, Harkness KL. Prevalence of depressive disorders in individuals with autism spectrum disorder: a meta-analysis. J Abnorm Child Psychol. 2019;47:165–75. [DOI] [PubMed] [Google Scholar]
- [48].Eicher TP, Mohajeri MH. Overlapping mechanisms of action of brain-active bacteria and bacterial metabolites in the pathogenesis of common brain diseases. Nutrients. 2022;14:2661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [49].Góralczyk-Bińkowska A, Szmajda-Krygier D, Kozłowska E. The microbiota-gut-brain axis in psychiatric disorders. Int J Mol Sci. 2022;23:11245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Soltysova M, Tomova A, Ostatnikova D. Gut microbiota profiles in children and adolescents with psychiatric disorders. Microorganisms. 2022;10:2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [51].Newell C, Bomhof MR, Reimer RA, et al. Ketogenic diet modifies the gut microbiota in a murine model of autism spectrum disorder. Mol Autism. 2016;7:37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [52].de Vos WM. Microbe profile: Akkermansia muciniphila: a conserved intestinal symbiont that acts as the gatekeeper of our mucosa. Microbiology (Reading). 2017;163:646–8. [DOI] [PubMed] [Google Scholar]
- [53].Alamoudi MU, Hosie S, Shindler AE, et al. Comparing the gut microbiome in autism and preclinical models: a systematic review. Front Cell Infect Microbiol. 2022;12:905841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [54].Wang L, Christophersen CT, Sorich MJ, et al. Low relative abundances of the mucolytic bacterium Akkermansia muciniphila and Bifidobacterium spp in feces of children with autism. Appl Environ Microbiol. 2011;77:6718–21. [DOI] [PMC free article] [PubMed] [Google Scholar]