Abstract
The global prevalence of chronic obstructive pulmonary disease (COPD) has increased over the last decade and has emerged as the third leading cause of death worldwide. It is characterized by emphysema with prolonged airflow limitation. COPD patients are more susceptible to COVID-19 and increase the disease severity about four times. The most used drugs to treat it show numerous side effects, including immune suppression and infection. This review discusses a narrative opinion and critical review of COPD. We present different aspects of the disease, from cellular and inflammatory responses to cigarette smoking in COPD and signaling pathways. In addition, we highlighted various risk factors for developing COPD apart from smoking, like occupational exposure, pollutants, genetic factors, gender, etc. After the recent elucidation of the underlying inflammatory signaling pathways in COPD, new molecular targeted drug candidates for COPD are signal-transmitting substances. We further summarize recent developments in biomarker discovery for COPD and its implications for disease diagnosis. In addition, we discuss novel drug targets for COPD that could be explored for drug development and subsequent clinical management of cardiovascular disease and COVID-19, commonly associated with COPD. Our extensive analysis of COPD cause, etiology, diagnosis, and therapeutic will provide a better understanding of the disease and the development of effective therapeutic options. In-depth knowledge of the underlying mechanism will offer deeper insights into identifying novel molecular targets for developing potent therapeutics and biomarkers of disease diagnosis.
1. Introduction
Chronic obstructive pulmonary disease (COPD) is a common breathing difficulty caused by the blockage of airflow and other associated breathing problems. It includes chronic bronchitis and emphysema. Airflow limitations are primarily due to remodeling and inflammation of the airways, which is often associated with the destruction of the parenchyma and the development of emphysema. The statistics in 2020 reported COPD as the third leading cause of death worldwide [1].
The interaction between different factors, including environmental and genetic factors, contributes to COPD risk. The most important and most common risk factor for COPD is cigarette smoking. In contrast, other risk factors include occupational/workplace exposure, air pollution, coal dust, asthma, airway hyper-responsiveness, and genetic predispositions [2–4]. Various pulmonary symptoms contribute to a progressive limitation in the airflow of COPD patients, including emphysema and small airway disease (SAD). SAD, called obstructive bronchitis, involves airway inflammation with increased mucus production, peribronchial fibrosis, and remodeling of airway walls [5]. Spirometry is used for the assessment of the degree of airflow limitation. According to global initiative for chronic obstructive lung disease (GOLD) criteria, the severity of COPD is classified spirometrically using the FEV1/FVC ratio [6, 7]. Other grading systems include BODE, where B is the body mass index, O is the airflow obstruction, D is the dyspnoea, and E is the exercise tolerance [8].
Many diseases co-occur with COPD and increase the severity and mortality of the patients, including cardiovascular diseases, malnutrition, osteoporosis, skeletal muscle dysfunction, anaemia, anxiety, depression, and increased gastroesophageal refluxes. The limitation of airflow in patients with COPD increases the likelihood of the patients developing pulmonary cancer over some time. Additionally, patients with COPD belong to the old age group, and the high prevalence of comorbidities requires more extensive medical care. Moreover, the risk of hospitalization and death increases, mainly due to severe airway obstruction [9]. Systemic inflammation in patients with COPD, together with activated inflammatory cells, raises the cytokine levels in the systemic circulation and increases oxidative stress. So, in the light of these researches, it was proposed recently to include the term “chronic systemic inflammatory syndrome” for diagnosing COPD [10].
Biomarkers are characteristics measured as indicators of normal biological processes, pathogenic processes, or responses to an exposure or intervention [11]. Biomarkers can be further categorized into various subtypes based on their relative utilization. It helps define the progression of the disease during pathogenesis. Biomarkers, combined with clinical symptoms, play an essential role in identifying the etiological origin and severity of the disease. Levels of biomarkers can be correlated toward response to therapy intervention, helping us assess the clinical evolution of the disease, and giving us insights into the potential complications that may arise. COPD pathogenesis majorly consists of overexpression of systemic inflammation markers and signaling pathways. Thus, determining these markers is one of the imperative and crucial directions in improving the diagnosis and management of COPD [12].
A multidisciplinary approach is required to optimize the therapeutic management of patients suffering from COPD [13]. Cessation of smoking, oxygen therapy, pharmacological therapy with glucocorticoids and bronchodilators, surgery, and pulmonary rehabilitation are the cornerstone of COPD management, making COPD a treatable and preventable disease [14]. Two types of therapies are used, including the therapy primarily centered in the lung, whereas the latter center shifts to a systemic inflammatory state.
In this review, we discussed the mediators of cellular signaling mechanisms in COPD, the risk factors involved in the progression of the disease, and different types of diagnostic biomarkers used to detect and confirm COPD. Here, we summarized the comorbid diseases with COPD and elaborated on the therapeutic perspectives of approved drugs and ongoing clinical trials. This study provides an extensive literature review followed by an in-depth and critical analysis of the state of the art and identifies challenges for future research.
2. Mediators of Cellular Signaling in COPD
COPD is a group of diseases that causes breathing-related issues and causes blockage of airflow. There are various cellular and inflammatory interactions caused by smoking cigarettes in COPD. Activation of various immunological cells, such as B-cells, T-cells, dendritic cells, macrophages, and neutrophils, and activation of epithelial cells, airway smooth muscle cells, and fibroblast cells results in the release of proteases, chemokines, and cytokines and hence causing COPD [15].
2.1. Role of NF-κB Pathway in COPD
Various canonical and noncanonical pathways play an essential role in the development and pathogenesis of COPD by overexpressing pro-inflammatory factors, causing chronic inflammation in the lungs. Furthermore, NF-κB-regulated genes such as adhesion molecules, cytokines, matrix metalloproteinases (MMPs), antiapoptotic factors, and angiogenic factors are associated with the progression of the disease. Thus, the first line of therapy in lung cancer and COPD is the downregulation of NF-κB activation [16].
2.2. Role of Adaptive Immune Response and Immune Sculpting
The main features of chronic inflammation in COPD patients are the accumulation of CD4+-T, CD8+-T cells, B-cells, dendritic cells, macrophages, neutrophils, and eosinophils in the small airways [5]. Infiltration of these inflammatory immune cells is associated with the severity of COPD disease. The central role of these inflammatory immune cells in COPD patients is the release of granzymes, proteinases, perforins, and oxidants, which destroy the walls and cause the hypersecretion of mucus [17].
2.3. Role of Adhesion Molecules
Integrins (heterodimeric transmembrane receptors) are involved in various cellular functions and lung inflammation. The expression of integrin avb6 (localized in epithelial cells) increases during injury and inflammation of the lungs [16]. Moreover, in COPD patients, there is an upregulation of TGF-β1 protein and mRNA in both the airways and alveolar epithelium cells. TGF-β1 mRNA levels correlate positively with the history of smoking and the degree of obstruction in the small airways, suggesting the proremodeling, profibrogenic, and cell-specific roles of TGF-β in the patients of COPD [16].
2.4. Role of Hypoxia or Angiogenesis
Hypoxia induces lung inflammation either by influencing the expression of pro-inflammatory genes or by activating transcription factors. In patients with COPD, damaged alveolar capillaries and progressive airflow limitations lead to reduced oxygen transport and cause alveolar hypoxia. Furthermore, hypoxia-inducible factor activation induces vascular endothelial growth factor (VEGF) transcription and increases angiogenesis. Thus, clinically, oxygen therapy can be significant in providing temporary relief in hypoxemic COPD patients. Interestingly, chronic oxygen therapy may result in oxidative cellular injury, resulting in aggravation of pulmonary inflammation and cell mortality. VEGF expression is also increased in chronic bronchitis patients [18], suggesting that VEGF has a paradoxical role in the airspaces and bronchi of COPD patients.
2.5. Role of Matrix Metalloproteinases (MMPs)
Emphysema is caused by the shift between the balance of proteinases and antiproteinases (shifts toward proteinases), including MMPs and elastase in the lungs' activated epithelial cells and inflammatory cells. Due to the defective tissue repair and degradation of ER membrane protein complex (EMC) by MMPs, the lungs' structural cells undergo apoptosis and lose their attachment. Additionally, the chemotactic activity of EMC fragments attracts inflammatory cells into the lungs, which results in further progression of emphysema in the mice model [19].
3. Risk Factors of COPD
The interaction between different factors, including environmental and genetic factors, contributes to COPD risk. Comorbid diseases can also affect the interaction between different factors. Risk factors for COPD are summarized in Figure 1.
Figure 1.
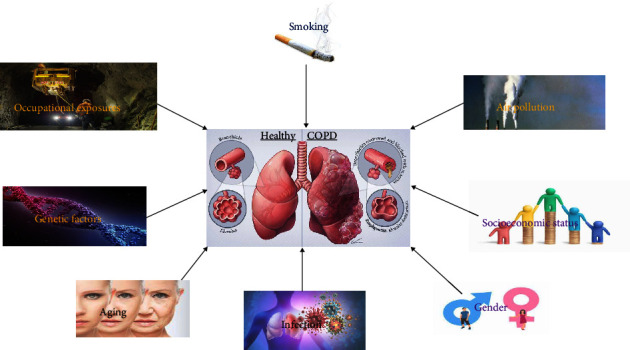
Risk factors of COPD.
3.1. Smoking Tobacco
Smoking tobacco is the leading causative factor of COPD worldwide. According to the estimate of WHO, 73% of COPD mortalities are present in high-income countries, whereas only 40% of COPD mortalities are present in low or middle-income nations [20]. Genes or genetics have a significant role in this relationship, as all smokers do not develop COPD. However, ∼50% of smokers develop COPD in their later life [21]. Smoking during the gestational period can negatively affect fetal lung growth and, therefore, results in the development of various lung diseases [22].
Cessation of tobacco smoking is the most effective intervention in preventing COPD progression, thus reducing morbidity and increasing the survivability of the patients [23]. Apart from tobacco, marijuana smoking has also been linked to various respiratory symptoms, but its direct relation to COPD development is unknown [24].
3.2. Occupational Exposure
Factory workers are exposed to various workplace factors, including dust, mist, fumes, vapors, and various chemicals, which is also a factor in causing COPD in factory workers or associated people [25]. In an estimate, it was shown that in the USA, 19.2% of COPD cases were attributed to exposure to workplace factors, out of which 31.1% cases were the proportion of being never smokers in their life and still develop COPD [26]. Another report showed that people diagnosed with chronic bronchitis or COPD were twice as likely to have previous workplace exposure to fumes, gases, vapors, and dust [27].
Due to less stringent laws in low and middle-income countries, work exposure to fumes and dust is remarkably greater than in high-income countries, where laws are comparatively more stringent. Hence, workplace exposure is assumed to be an important risk factor for COPD.
3.3. Air Pollution
In poorly ventilated homes, cooking or heating exposes biomass fuels such as wood, animal dung, coal, straw, and crop residues. These are considered essential risk factors in COPD development, primarily in rural households. One of the reports from China suggests that the prevalence of COPD is twice or thrice times higher in never-smoker women in rural areas, where women have greater exposure to biomass smoke than urban women without such exposures [28]. According to the estimate of WHO, in low and middle-income countries, 35% of people develop COPD after exposure to biomass fuels and indoor smoke [20]. Furthermore, WHO also suggested that 36% of mortality from lower respiratory diseases is directly related to exposure to indoor smoke [20].
Apart from direct smoke exposure, second-hand smoke inhalation is another form of biomass smoke linked to various respiratory diseases. However, its direct relation to COPD is not known [29].
The risk attributes for COPD development from outdoor air pollutants are remarkably lesser than indoor air pollutants. According to an estimate by WHO, in higher income countries, only 1% of COPD cases are developed by urban air pollution, and only 2% in low and middle-income countries [20]. Outdoor air pollution, or simple air pollution, is linked to acute cardiopulmonary events and various respiratory infections essential in developing and progressing COPD.
3.4. Genetic Factors
The functioning of the lungs in offspring is complexly related to the parental lung functions [30]. Out of all offspring whose both parents lie in the lowest quintile group of lung functioning, 37% of the offspring lie in the lowest quintile group of lung function compared to their peers [30]. On the other hand, out of all offspring whose parents lie in the highest quintile group of lung functioning, 41% of the offspring lie in the highest quintile group of lung functioning compared to their peers [30].
Serine protease α1-antitrypsin deficiency is one of the best-known genetic factors linked to COPD. Serine protease α1-antitrypsin deficiency arises in 1%–3% of patients with COPD [31]. A low concentration of α1-antitrypsin, particularly in amalgam with tobacco smoking and other exposures, increases the probability of pan lobular emphysema [31].
Various studies show that several genes are involved in COPD, such as microsomal epoxide hydrolase 1 [32], tumor necrosis factor (TNF)-α [33], and transforming growth factor-β1 [34]. However, research is still under trial to examine the specific polymorphisms in the abovementioned genes for COPD development. A study by Zhou et al. [35] on the Chinese population shows that the genetic polymorphism in MIR5708 and MIR1208 is related to the susceptibility to COPD.
3.5. Infection
According to a review of The Lancet, most of the exacerbations of COPD are either due to viral infections or bacterial infections [36]. Various infections play an essential role in the development and progression of the disease. Early-life exposure to infections could make the individual susceptible to bronchiectasis and change the airways' responsiveness.
3.6. Aging
The age-related decline in pulmonary function is considered normal. With the increase in age, the prevalence, mortality, and morbidity of COPD also increase. The peak level of pulmonary function reaches young adulthood, which declines in the third or fourth decades of life [36]. However, some researchers have reported that in old age, people with a high level of pulmonary function live comparatively longer than those with lower pulmonary functions [37].
Aging is a natural process, where, at the cellular level, it includes various molecular and systemic mechanisms. In several studies, the role of specific senescence pathways, such as sirtuin family proteins and p-16, has become evident and is implicated in COPD and aging [38]. Various common DNA-level abnormalities, leucocyte response abnormalities, and inflammatory markers were increased both in COPD and aging [38].
In recent years, the prevalence of COPD has increased due to the demographic changes in the world's population, which are attributed to a good nutritional life, and, hence, the reduction and elimination of various infectious diseases in childhood. Therefore, falling in mortality rates are falling due to diseases that kill or eliminate younger people earlier, such as acute infections and cardiac diseases. This leads to a longer life expectancy in most of the world's population and increases the risk for several chronic medical conditions, including COPD [39].
3.7. Socioeconomic Status
Populations with lower socioeconomic status or the population who live in poverty have a higher risk of developing COPD and its related complications compared to their wealthier counterparts [40]. Poverty is considered a surrogate for poor access to health care, poor nutritional status, overcrowding (large family size), more exposure to occupational pollutants (such as, fumes, dust, mists, and chemicals), high rates of tobacco smoking (in low and middle-income countries), and early respiratory infections, and hence, increases the risk of COPD subsequently [40].
In the study by Eisner et al. [41], a consistent and significant inverse relation was observed between the outcomes of socioeconomic status and COPD, which may be used to validate the time and costs needed for the development, research, and implementation of the strategies to improve the health of COPD patients.
3.8. Gender
The role of gender in both the progression and development of COPD is highly controversial and is a topic of a great deal in the scientific community [42]. Earlier in men (due to occupational exposure and related patterns of tobacco smoking), COPD is considered far more frequent than in women of the same age [43]. However, lately, the prevalence of COPD has become equally likely in men and women of higher income countries where the habit of smoking tobacco is similar between the two sexes. Some studies showed that women are more likely to develop COPD than men. By giving equal exposure to smoking and the same environmental factors, researchers support the hypothesis that women are more susceptible to developing COPD than men [44].
4. Diagnostic Biomarkers for COPD
COPD is a diverse disease affecting multiple organs, establishing a systemic infection. Recently, studies have shifted their focus to biomarkers to illustrate the pathogenesis and progression of the disease. A detailed and systematic study on biomarkers can pave a new road to designing novel therapeutic targets for COPD [45]. Biomarkers can be defined fundamentally and straightforwardly as “A defining characteristic that is measured as an indicator of normal biological processes, pathogenic processes, or responses to an exposure or intervention” [11].
Biomarkers can be further categorized into various subtypes based on their relative utilization. Biomarkers help define the progression of the disease during pathogenesis. Biomarkers, combined with clinical symptoms, play a role in identifying the etiological origin and severity of the disease. Levels of biomarkers can be correlated toward response to therapy intervention, helping us assess the clinical evolution of the disease and giving us insights into the potential complications that may arise. Thus, studying these biomarkers will enable us to manage the disease better with better risk stratification. They may also guide us in assessing the effectiveness of clinical trials.
This review will discuss the different types of diagnostic biomarkers used to detect and confirm COPD. These biomarkers help identify the diseased population and aid us in redefining the disease classification. COPD pathogenesis majorly consists of overexpression of systemic inflammation markers and signaling pathways. Thus, determining these markers is one of the imperative and crucial directions in improving the diagnosis and management of COPD [12]. The following are the significant diagnostic biomarkers for the detection and management of COPD (Figure 2).
Figure 2.
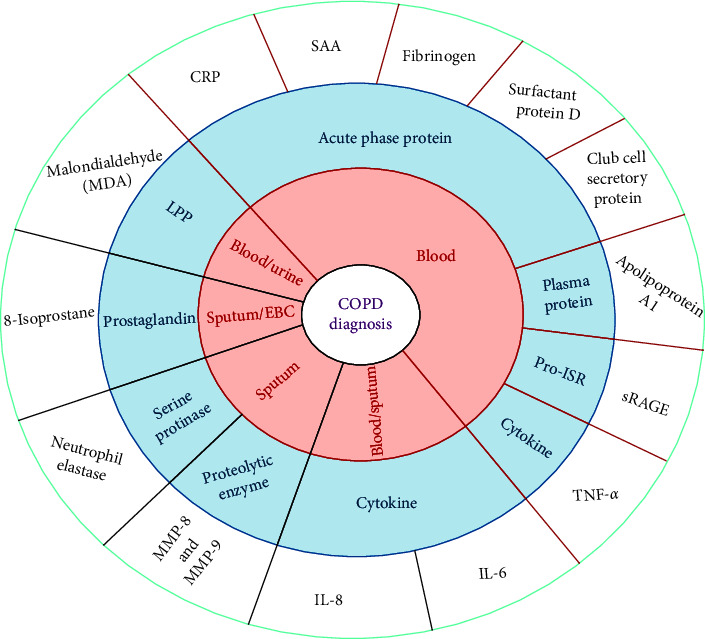
List of biomarkers used in the diagnosis of COPD. ECB, exhaled blood condensate; LPP, lipid peroxidation product; and Pro-ISR, pro-inflammatory signaling receptor.
4.1. C-Reactive Protein
The overexpression of C-reactive protein (CRP) is known at the site of inflammation and infection, as it is an acute inflammatory protein. CRP is the predominant mediator of the acute phase response in an inflammatory event, primarily synthesized by IL-6-dependent hepatic biosynthesis [46]. The serum CRP levels in COPD patients were significantly higher than those of healthy subjects [47]. In COPD patients, the levels of CRP were directly associated with age and inversely related to hemoglobin levels. Studies have found that lung function indices such as dyspnea score, oxygen saturation, and 6-min walking distance (6MWD), potential COPD severity predictors, substantially correlate with CRP levels. Factors such as duration of the disease and BMI in COPD patients show no correlation with CRP levels [48].
Increased serum CRP baseline is more pronounced in COPD mortality than COPD hospitalization cases, consistently marking functional and metabolic damage in advanced COPD [49]. Thus, in the case of COPD exacerbation, CRP is used as a diagnostic biomarker, and for early mortality cases of COPD, it is used as a prognostic biomarker [50, 51]. CRP levels can also be used as an informative biomarker as they can demonstrate low-grade systemic inflammation [52]. Lately, studies have shown the association of serum CRP levels with several different outcomes, including COPD exacerbation [53].
4.2. Interleukin-6
Interleukin-6 (IL-6) plays a vital role in systemic inflammation of COPD patients, as it is a crucial pro-inflammatory cytokine [54]. In the stable and exacerbation phases of COPD patients, the circulatory level of IL-6 was higher than healthy controls [55]. Among the severity of COPD, the circulatory levels of IL-6 were found to be of significant difference, further defining that the level of IL-6 was higher in mild, moderate, severe, and very severe COPD patients [56].
The pro-inflammatory cytokine IL-6 can induce acute-phase proteins like CRP. Thus, the levels of CRP and IL-6 were correlated in COPD patients [57]. COPD exacerbations lead to overexpression of IL-6 in serum, which leads to a surge of plasma fibrinogen. Thus, acute COPD response can prime for stroke or coronary heart disease as comorbidity [58]. A recent finding has established a relation between the plasma level of IL-6 in malnutrition pathophysiology and low-weight COPD patients. The study has also elucidated the sensitivity of the serum level of IL-6 compared to other inflammatory factors in predicting COPD disease development in smokers [59].
IL-6 is the primary regulator in many inflammatory pathways contributing to disease progression. Thus, IL-6 can independently be a predictive power in determining mortality in a basic clinical model [60]. IL-6 serum levels have also been associated with poor physical function in COPD patients irrespective of age, gender, race, and body composition [61]. IL-6-driven inflammation may complicate COPD by contributing extensively to pulmonary hypertension, leading to increased morbidity of the disease [62]. We can summarize the systemic inflammatory process led by IL-6 in COPD patients coerced toward a progressive and persistent disease model with associated mortality and inconsistent physical performance [63].
4.3. Serum Amyloid A
Serum amyloid A (SAA) is a protein of the acute phase having numerous immunological functions. It involves various processes such as inflammatory reactions, lipid metabolism, and granuloma formation. It has an established role in autoimmune lung disease pathogenesis. SAA is used as a biomarker in various lung diseases such as COPD, obstructive sleep apnea (OSA) syndrome, asthma, lung cancer, and cystic fibrosis [64]. SAA has been identified as a novel blood biomarker for acute exacerbation of chronic obstructive pulmonary disease (AECOPD). Changes in SAA levels have shown higher sensitivity and specificity in defining AECOPD severity than other biomarkers like CRP. A significant increase in SAA levels above baseline predicts severe AECOPD [65]. For early detection and management of AECOPD, SAA-level determination is advantageous. It can help categorize the patients with the highest risk of respiratory failure. Biomarkers like IL-6 remain uninformative in severe cases of AECOPD [65].
Patients with AECOPD and COPD have inflammatory reactions and have high blood viscosity, thus showing significantly higher levels of SAA compared to the healthy control group. For the clinical diagnosis and treatment of AECOPD, SAA can be used as an effective index [66]. In the resolution phase of infection, the secretion of SAA becomes self-limiting and protective with a sharp fall. In the AECOPD, there is a steady rise in SAA levels elicited by coinfections [67]. Exhilarated levels of SAA are also associated with cardiovascular diseases, where COPD is a comorbidity. Exacerbation episodes lead to a dramatic increase in mortality related to cardiovascular events in COPD. SAA levels can very well anticipate future cardiovascular events; thus, SAA can be scrutinized as a predictor for frequent exacerbation phenotypes and a marker for comorbid cardiovascular disease [68].
4.4. Tumor Necrosis Factor
TNF is a potent cytokine that mediates inflammation and immune response. It recruits acute phase proteins, transcription factors, cell surface receptors, and cytokines [69]. TNF starts the production of interleukin cascade components when secreted excessively. It can also instigate the secretion of matrix metalloprotease 9, which contributes to lung emphysema, one of COPD's major symptoms [70]. Recently, a meta-analysis study has shown compelling results for a direct relationship between COPD and elevated TNF-α levels. Higher TNF-α level was found in COPD patients in comparison to healthy controls. It may also play a role in the progression and diagnosis of COPD, but its mechanism is still unknown and needs to be further explored [71].
In the pathophysiology of COPD, TNF-α tends to play a central role. Different cells, like alveolar macrophages and T-cells, produce TNF-α in response to various pollutants, including cigarette smoke [72]. Studies with animal models have shown the induction of pathological features of COPD, like emphysema and lung fibrosis, following elevated TNF-α levels [73]. The role of TNF-α during COPD is limited to enhancing inflammatory events and developing systemic inflammation within the respiratory tract, which manifests in cachexia in severe COPD patients. Thus, TNF-α is directly associated with the severity of the disease and its progression [74].
4.5. Fibrinogen
Fibrinogen is a soluble plasma glycoprotein majorly involved in blood coagulation reactions by converting thrombin into fibrin. It can increase significantly during acute phase stimulation in response to increased IL-6 production [75]. A deterioration in lung function can be associated with elevated plasma fibrinogen concentration, which increases the risk of developing COPD [76]. For exacerbation of COPD, elevated fibrinogen can act as an independent risk factor [77].
Fibrinogen is a glycoprotein with biological roles closely associated with cell adhesion, blood coagulation, phagocytosis, extension, proliferation, etc. [78]. The molecular weight of fibrinogen is the largest in available coagulogens, having structure chain-like asymmetric, and can increase the thickening of plasma, contributing to the formation of small pulmonary arterial thrombi and aggravating lung injury [79].
Food and Drug Administration (FDA), USA, and European Medicines Agency (EMA) have qualified plasma fibrinogen as a severity assessment COPD biomarker and qualified it as a drug development tool [80]. As issued by the FDA and EMA, circulating fibrinogen can be suggested as a biomarker for early warning. It can be a potent predictor of future susceptibility to develop COPD and its severity [81].
5. Comorbidities in COPD
Many diseases co-occur with COPD and hence increase the severity and mortality of the patients. The comorbid diseases could also affect the interaction between different environmental factors and their impact on the severity of the disease. Comorbid diseases of COPD are summarized in Figure 3.
Figure 3.
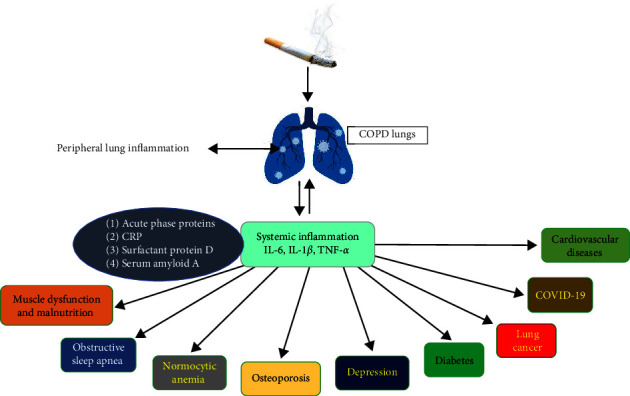
Comorbidities with COPD.
5.1. COVID-19
Patients with preexisting COPD are susceptible to worsening the prognosis and progression of COVID-19 [82]. Meta-analysis studies on COVID-19 from December 2019 to March 2020 demonstrate that the severity of COVID-19 increases fourfold in patients with preexisting COPD [83, 84]. The analysis in China for comorbidities in 1,590 COVID-19-positive patients found that the odds ratio of COPD is about 2.681 (95% CI 1.424–5.048; p=0.002) for ICU patients, ventilators, or death, even after adjusting the smoking and age [84–87]. Furthermore, out of all severe cases, 62.5% of cases had a history of COPD, whereas, in comparison, only 15.3% of cases had a history of COPD in nonsevere cases. Additionally, out of the total fatalities, 25% of cases are COPD patients, whereas, in comparison, only 2.8% of COPD patients have survived.
Cellular serine protease (TMPRSS2) primes the envelope spike protein of SARS-CoV-2 and hence helps to facilitate the fusion of the SARS-CoV-2 virus with angiotensin-converting enzyme 2 receptor (ACE-2 receptor) in the cells and subsequently facilitates the cell entry [88, 89]. In COPD patients, the expression of ACE-2 was significantly elevated in bronchial epithelial cells compared to the control subjects [90]. However, it is interesting to note that the expression of ACE-2 alone has neither shown an increase in the severity of the disease nor an increase in its susceptibility. Moreover, in patients with predominantly small airway pathologies, ACE-2 expression is relatively low in the bronchial epithelium compared to the nasal epithelium. It has unexplained implications for the susceptibility of the disease [91].
5.2. Lung Cancer
COPD patients are three to four times more susceptible to developing pulmonary cancer than tobacco smokers with normal pulmonary functions [92]. Lung cancer is one of the significant causes of fatality in COPD patients, especially in patients with more severe diseases [93]. An increase in the prevalence of pulmonary cancer in patients with COPD is more likely associated with an increase in oxidative stress and inflammation in COPD patients [94]. NF-κB activation might provide a link between lung cancer and inflammation [95]. By regulating the expression of various detoxifying enzymes, nuclear factor erythroid 2-related factor 2 (Nrf2) plays a vital role in defending against several carcinogens of tobacco inhalation. As COPD patients lack Nrf2, it might contribute to increasing the susceptibility of COPD patients to pulmonary cancers [96].
It is interesting to note that lung cancer is more common among patients with COPD who are never smokers and have never been exposed to tobacco inhalation in their entire life [97]. In females, the risk of getting lung cancer and COPD is greater, probably due to the hormone-stimulated metabolism of carcinogens in smoking tobacco [98].
5.3. Muscle Dysfunction and Malnutrition in COPD
Weakness in the skeletal muscles is one of the important systemic effects of COPD, often accompanied by the loss of fat-free mass (FFM) [99]. In normal males, 40%–50% of total body mass accounts for skeletal muscle. The turnover of proteins in skeletal muscle is a dynamic process involving balancing protein breakdown and protein synthesis. Moreover, in acute illnesses like sepsis and trauma, the loss of muscle mass due to the breakdown of muscle proteins is comparatively more significant. It occurs rapidly, whereas in chronic illnesses like COPD, the loss of muscle mass due to muscle protein breakdown is comparatively slower. Several studies demonstrate that in patients with COPD, the structure and function of skeletal muscles are altered. Several studies on humans indicate that the breakdown of skeletal muscle is evident in patients with COPD and is specific for muscle fiber type IIA/IIx [100].
Muscle wasting has a magical effect on morbidity in severe cases of COPD. Hence, it increases the risk of readmission to the hospital after an exacerbation and increases the need for mechanical ventilation support. Additionally, muscle wasting is considered a significant determinant of mortality in patients with COPD independently of smoking, BMI, and lung functions [101].
5.4. Diabetes
Several population studies indicated an increase in the prevalence of diabetes among patients with COPD, with a relative risk of 1.5–1.8 [9]. Even in patients with mild symptoms of COPD, the prevalence of diabetes is observed [9]. The mechanism behind this association is not precise. Furthermore, the relation between high doses of inhaled corticosteroids and the increased risk of diabetes is also doubtful. Patients with mild disease also have a high risk of diabetes. It is interesting to note here that asthma patients do not have the prevalence of diabetes. However, COPD does have it, suggesting an altogether different mechanism of inflammation in COPD compared to asthma.
Pro-inflammatory cytokines like IL-6 and TNF-α, induce insulin resistance by inhibiting the signals of insulin receptors, thereby increasing the risk of type-2 diabetes [102]. An increase in the concentration of plasma CRP, IL-6, and TNF-α are reported in metabolic syndromes, including cardiovascular diseases and insulin resistance [103]. These metabolic syndromes are also prevalent among patients with COPD, thus leading to the co-occurrence of cardiovascular diseases and diabetes with airway obstruction [103].
5.5. Osteoporosis
Multiple studies have shown that in patients with COPD, there is a very high prevalence of low bone mineral density (BMD) and osteoporosis, even in the milder stages of COPD disease [104]. In a study, 6,000 COPD patients were recruited for the large TORCH trial (toward a Revolution in COPD Health); among these, over half of the COPD patients have osteopenia or osteoporosis, which is determined by Dexa (dual-energy radiograph absorptiometry) [105]. In another study, there is a 75% prevalence of osteoporosis in patients with GOLD stage IV disease, and it has a strong correlation with the decrease in the level of FFM [106, 107].
It is interesting to note that the prevalence of osteoporosis is high in males and even higher in the case of females. At the same time, the incidences of nontraumatic and traumatic fractures are approximately similar in both sexes. Fractures due to vertebral compression are relatively widespread among patients with COPD, and this increase in kyphosis further reduces the functions of the lung [108].
5.6. Normocytic Anemia
Several studies have shown that the prevalence of anemia is very high among patients of COPD, ranging between 15% and 30% of total COPD patients, especially in patients with more severe disease conditions. In contrast, erythrocytosis (polycythemia) is comparatively rare, ranging about 6% [109, 110]. Hemoglobin level is independently and strongly associated with the increase in functional dyspnea, and decrease in exercise capacity, and hence, is an essential contributor to functional capacity and poor quality of life [111]. For chronic inflammation diseases, the anemia is usually of characteristic normochromic normocytic type, which appears to be due to resistance to the erythropoietin, whose concentration is elevated in these patients [112].
In a study, it was shown that the transfusion of blood improves exercise performance in anaemic COPD patients [113]. However, iron supplements show detrimental effects as iron cannot be utilized correctly and may increase systemic oxidative stress.
5.7. Obstructive Sleep Apnea
The condition during sleep where the upper airways collapse repetitively is known as obstructive sleep apnea (OSA). According to an estimate, 20% of OSA patients also have underlined COPD conditions. Of their counterparts, 10% of COPD patients have the severity of the disease independent of OSA [114]. OSA and COPD patients share many common comorbidities, including cardiac failure, endothelial dysfunction, metabolic syndrome, and diabetes [115]. According to some recent research, it is evident that patients with OSA have systemic inflammation, upper airway inflammation, and oxidative stress [116].
5.8. Depression
Due to the physical limitations of COPD patients, they are more often isolated and unable to engage themselves in various social activities. So, it is not surprising to see an increase in the prevalence of depression and anxiety in patients with COPD, and depression and anxiety appear to be more prevalent in COPD than in any other chronic disorder. The symptoms of depression and anxiety are often confused with the symptoms of COPD; hence, they remain undiagnosed and untreated in various clinical practices. Clinically relevant symptoms of depression are estimated to occur in about 10%–18% of all patients. However, among the clinically stable outpatients with COPD, about 19%–42% prevalence is seen for major depression, which requires therapeutical interventions [117, 118]. Depression cannot be diagnosed with any standardized approach in patients with COPD due to underlying differences in the variability and methodology of the screening questionnaires at cut-off points to determine the depression diagnosis. However, various simplified tools can help a clinician screen out depression, and if the case is in doubt, referring to a specialist who specializes in psychiatric disorders can show beneficial effects for the patient.
Much-blooming evidence states that systemic inflammation may result in depression, and IL-6 mainly plays an essential role in human and animal models of depression [119].
5.9. Cardiovascular Diseases
The functional and anatomical relationship between the heart and lungs is very intricate. Any impairment that impacts one of the two organs will likely have consequences on the other. These associations are essential in COPD patients and can be of two types: (A) related pathologies that have similar risk factors, such as COPD and congestive heart failure, or smoking cigarettes and coronary artery disease (CAD), and (B) those diseases that result in the dysfunctioning of the heart to underlining primary lung diseases, such as ventricular dysfunction and secondary pulmonary hypertension due to increased intrathoracic mechanical load. Common cardiovascular diseases which are found in prevalence in COPD patients are described below.
5.10. Coronary Artery Disease
CAD and COPD are very prevalent diseases and share similar risk factors, such as old age, sedentary lifestyle, and smoking cigarettes. Independent of sex, age, and smoking habits, it is evident that patients with any airflow limitations are more prone to death from myocardial infarction [120]. Some results were obtained even when the history of cigarette smoke was included [121]. Some results were obtained even when the history of cigarette smoke is included [121]. Due to respiratory insufficiency, the chances of mortality due to cardiovascular ailments are higher in patients with mild COPD [105]. Clinically, a strong correlation has been found between FEV1 (impaired lung function) and cardiovascular mortality and morbidity. Independent of the status of smoking, COPD patients have a higher prevalence of fatal myocardial infarction [122].
5.11. Heart Failure
The shreds of evidence for the association between left ventricular congestive failure and COPD are comparatively lesser. Although some theories suggest that COPD shares a common inflammatory pathway with left ventricular congestive failure, it is clinically poorly defined how the prevalence of left ventricular function decreases in COPD patients. One study estimates that the prevalence of left ventricular congestive failure is about 20% in patients with COPD who have never had such a diagnosis [123]. The signs and symptoms of heart failure in COPD are very intricate and overlap with one another, making the diagnosis very complicated. The best way to discriminate between COPD and heart failure in COPD is the measurement of N-terminal prohormone brain natriuretic peptide (NT-proBNP) or B-type natriuretic peptide [124]. This measurement of NT-proBNP can be a valuable factor in distinguishing between decompensated heart failure and acute COPD exacerbation [125]. An overexpressed NT-proBNP plasma level correlates with poor physical activity in patients with COPD, suggesting that the defective left ventricular function may contribute to reducing the performance in the patients [126].
5.12. Pulmonary Arterial Hypertension (PAH)
Clinically, pulmonary arterial hypertension is not common among patients with COPD with mild to moderate stages. However, it can develop during exercise. About 50% of patients with COPD who undergo lung transplantation or lung volume reduction surgery have severe to moderate PAH [127]. The ratio between hypoxic vasoconstriction and ventilation-perfusion is higher in less severe cases of COPD than in advanced stages of COPD, where the ratio is less. Various studies suggest that the initial stages of PAH in COPD can be an injury of the endothelium by smoking cigarettes with a simultaneous downregulating of the expression of prostacyclin synthase and endothelial nitric oxide synthase and subsequently the impairment in the function of the endothelium [128].
5.13. Arterial Stiffness and Endothelial Function
Arterial stiffness which occurs in vascular disease is a better marker for cardiovascular events. It can be determined noninvasively by calculating the aorta's radial artery tonometry or pulse wave velocity [129]. In regular smokers and nonsmokers, arterial stiffness increases in COPD patients, and this phenomenon is neither related to the severity of the disease nor to the concentration of circulating CRP [130]. Clinically, this increase in arterial stiffness may result in systemic hypertension and increase the prevalence of cardiovascular diseases in patients with COPD [131].
6. Therapy Approaches in COPD
The usage of bronchodilators for the management of COPD is an important advance. It has been long acting, but the underlying mechanism for these drugs to deal with the inflammatory process is not known [132]. COPD shows heterogeneous clinical phenotypes, which leads to incongruent groups with unstable disease mechanisms or molecular pathways, leading to inconsistent approaches for the development of new therapies [133]. For COPD therapy, most known drug classes are being used in the newly approved drugs. These drugs do not target disease progression processes and mortality but address symptoms and exacerbations. Hence, a humongous requirement exists for developing therapies that target immune dysfunction and underlying inflammation [134]. After the recent elucidation of the underlying inflammatory signaling pathways in COPD, new molecular targeted drug candidates for COPD are signal-transmitting substances. Newer COPD treatment strategies are described in Figure 4 and Table 1.
Figure 4.
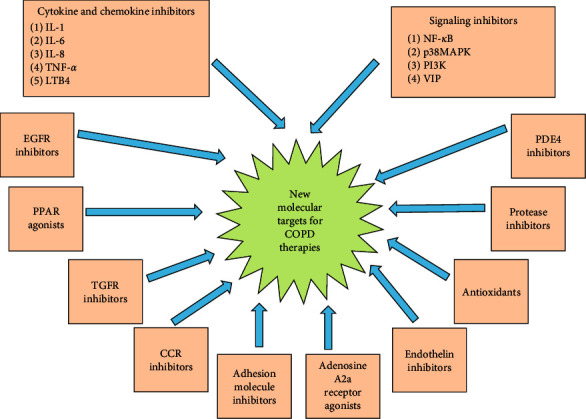
Molecular targets for newer COPD treatment strategies [14].
Table 1.
Pro-inflammatory signaling pathway inhibitors for COPD.
| S. no. | Inhibitor/drug | Mechanism/effect | Clinical progress | References |
|---|---|---|---|---|
| 1 | RV568 | P38MAPK pathway inhibitor | No effect (NCT01475292) (NCT01661244) |
[135] |
|
| ||||
| 2 | Nemiralisib (GSK2269557) |
PI3K inhibitor | 28 Days treatment (NCT02294734) 84 Days treatment (NCT02522299) |
[136] |
|
| ||||
| 3 | RV1729 | PI3K inhibitor | Phase I Trial, limited efficacy (NCT02140346) | [136, 137] |
|
| ||||
| 4 | VIP (vasoactive intestinal peptide) | Increases cAMP, adenylate cyclase, and phospholipase C | 3 Months inhaled treatment (NCT00464932) |
[138, 139] |
|
| ||||
| 5 | Adenosine A2A receptor (UK-432097) |
cAMP enhancer | No effect (NCT00430300) |
[140, 141] |
|
| ||||
| 6 | AZD1981 | Prostaglandin D2 receptor inhibitor | No effect (NCT00690482) |
[142] |
|
| ||||
| 7 | Bimosiamose | Selectins inhibitor | 28 Days inhalation treatment (NCT01108913) |
[143] |
6.1. Antioxidants
In the pathogenesis of COPD, oxidative stress plays a vital role. Antioxidants inhibit oxidative stress by scavenging reactive oxygen species (ROS), which reduces inflammation and cellular damage in the lungs [144]. In this aspect, Table 2 provides clinical testing of N-acetyl cysteine and other glutamines have been carried out [154].
Table 2.
Antioxidants and protease inhibitors for COPD.
| S. no. | Inhibitor/drug | Mechanism/effect | Clinical progress | References |
|---|---|---|---|---|
| 1 | N-acetyl cysteine (NAC)/glutamines | Oxidative stress suppressor | Clinical trials are going on. Effective in high-risk patients (NCT01136239) (NCT00184977) | [145, 146] |
|
| ||||
| 2 | SOD/GPx | ROS reducer | Established in animal models. Clinical trials underway | [147] |
|
| ||||
| 3 | Sulforaphane | ROS and RNS reducer | 4 Weeks clinical study, no effect (NCT01335971) | [148] |
|
| ||||
| 4 | Resveratrol | Activator of SIRT1 | 12 Weeks of clinical study done (NCT03819517) | [149] |
|
| ||||
| 5 | SRT1720 | Activator of SIRT1 | Established in animal model | [150] |
|
| ||||
| 6 | AZD1236 | Anti MMP-9 and MMP-12 |
6 Weeks clinical study. Results not statistically significant | [151] |
|
| ||||
| 7 | Sivelestat (ONO-5046) |
Protection from NE-mediated lung damage | Clinically approved in Japan for ALI and ARDS | [152] |
|
| ||||
| 8 | AZD9668 | Protection from NE-mediated lung damage | 12 Weeks clinical study with budesonide. No effect | [153] |
6.2. Protease Inhibitors
In COPD pathophysiology, the lung's antiprotease ratio is an important aspect. Both in vitro and in vivo COPD models have shown efficacy for antiprotease therapy [155]. Selective inhibitors have been designed against matrix Metaloprotein 9 (MMP-9). These inhibitors have shown efficacy in animal models, but their effectiveness is minimal in clinical trials of COPD [156].
6.3. Chemokine and Cytokine Inhibitors
The levels of cytokines and chemokines like IL-1, IL-6, IL-1β, IL-8, and TNF-α increased significantly in COPD patients [157]. Inhibitors have been designed against the IL-8 receptor CXC chemokine receptor 2 (CXCR2). These inhibitors have shown positive results in animal models and clinical trials by blocking neutrophil infiltration [158] (Table 3).
Table 3.
Cytokine and chemokine receptor inhibitors for COPD.
| S. no. | Inhibitor/drug | Mechanism/effect | Clinical progress | References |
|---|---|---|---|---|
| 1 | Tocilizumab | IL-6 inhibitor | Clinical trials need further study | [159] |
|
| ||||
| 2 | Canakinumab | IL-1β inhibitor | Phase I/II RDBPCES | [160] |
|
| ||||
| 3 | Infliximab | TNF-α inhibitor | No effect (NCT00056264) |
[161–163] |
|
| ||||
| 4 | Etanercept | TNF-α inhibitor | 90 Days treatment of COPD (NCT00789997) | [164] |
|
| ||||
| 5 | AZD4818 | CCR1 inhibitor | No effect (NCT00629239) |
[165] |
|
| ||||
| 6 | AZD2423 | CCR1 inhibitor | A study completed statistical analysis not released (NCT012115279) |
[166] |
|
| ||||
| 7 | Navarixin (MK-7123) |
CXCR2 inhibitor | 6 Months of study. Improvement in FEV1 (NCT01006616) (NCT00441701) | [166, 167] |
|
| ||||
| 8 | AZD5069 | CXCR2 inhibitor | 4 Weeks treatment (NCT01233232) |
[166] |
|
| ||||
| 9 | BIIL 284 | LTB4 receptor inhibitor | Clinical study done (NCT02249247) (NCT02249338) |
[168] |
|
| ||||
| 10 | Zileuton | 5-LO inhibitor | No effect in treatment (NCT00493974) |
[169] |
|
| ||||
| 11 | Mepolizumab | IL-5 inhibitor | 26–52 Weeks treatment (NCT02105948, NCT01463644, NCT02105961) |
[170] |
|
| ||||
| 12 | Benralizumab | IL-5Rα inhibitor | No effect (NCT01227278) |
[171] |
|
| ||||
| 13 | Lebrikizumab | IL-13 inhibitor | Decline in COPD exacerbation and lung function (NCT02546700) |
[172, 173] |
6.4. PDE4 Inhibitors
Phosphodiesterase enzymes (PDE) metabolize the intracellular secondary messenger cAMP and cGMP. Inflammatory cells such as T-cells, eosinophils, and neutrophils have shown the expression of PDE4 in asthma and COPD [174]. Thus, inhibiting PDE4 is an effective therapeutic strategy for inflammatory respiratory diseases. PDE4 inhibitors target cAMP hydrolysis, increasing levels and activating downstream phosphorylation cascades. This reduces inflammation and relaxes the airway smooth muscles [175].
The only approved PDE4 inhibitor for treating severe COPD is roflumilast. It has been shown to improve lung function significantly and reduce the exacerbation rate in clinical trials with severe COPD [176] (Table 4).
Table 4.
cAMP and cGMP phosphodiesterase inhibitor for COPD.
| S. no. | Inhibitor/drug | Mechanism/effect | Clinical progress | References |
|---|---|---|---|---|
| 1 | Roflumilast | PDE4 inhibitor | US-FDA approved drug | [177, 178] |
|
| ||||
| 2 | GSK-256066 | PDE4 inhibitor | 4 Weeks inhaled treatment (NCT00549679) |
[179] |
|
| ||||
| 3 | CHF6001 | PDE4 inhibitor | Clinical testing going on (NCT01730404) | [180] |
|
| ||||
| 4 | Tadalafil | PDE5 inhibitor | Approved for pulmonary arterial hypertension 12-week treatment (NCT01197469) | [181] |
|
| ||||
| 5 | RPL554 | PDE3/PDE4 inhibitor | Being investigated as an adjunct (NCT02542254) |
[182] |
6.5. Adhesion Molecule Inhibitors
Adhesion molecules are expressed on a variety of cells. They help intracellular communication via cell–cell adhesion and signal transduction [183]. An antiselectin antibody, EL246, which targets cell adhesion, has been developed. Currently, EL246 is being pursued with acute exacerbation of COPD as a therapeutic drug [184] (Table 5).
Table 5.
Other drugs for COPD.
| S. no. | Inhibitor/drug | Mechanism/effect | Clinical progress | References |
|---|---|---|---|---|
| 1 | Eleuquin (EL246) | Cell adhesion inhibitor | Under predevelopment by LigoCyte | [185] |
|
| ||||
| 2 | BIBW 2948 | EGFR internalization reducer | 4 Weeks treatment (NCT00423137) | [186] |
|
| ||||
| 3 | Bosentan | Endothelin receptor inhibitor | 18 Months treatment (NCT02093195) | [187, 188] |
|
| ||||
| 4 | Solithromycin | Macrolide antibiotic | Early termination of the trial (NCT02628769) | [189] |
|
| ||||
| 5 | Thiazolidinediones | PPARγ agonists | 10 Months treatment (NCT00103922) |
[190, 191] |
6.6. PPAR Agonist
The role of peroxisome proliferator-activated receptor (PPAR) signaling in regulating inflammation has been studied well. Of the various isoforms of PPAR, PPARg plays a crucial role in regulating the expression of genes involved in pathogenic conditions [192]. PPARγ agonists exhibit an anti-inflammatory effect as they suppress the production of pro-inflammatory cytokines [193]. Thiazolidinediones, one of the PPARγ agonists, have reduced lung inflammation in in vivo studies [194]. In cigarette smoke-induced emphysema, thiazolidinediones treatment was shown to reverse the emphysema [195].
7. Conclusions and Prospects
Many comorbidities are present with COPD, which is associated with inaccuracy in death. So, all causes of death must be the prime endpoint for any further studies to evaluate the therapy of COPD. However, the ongoing revolution of COPD health studies, including all causes of mortality, can provide conclusive data for long-acting b2-agonists and inhaled corticosteroids in combination or individually. It is essential to consider COPD as a multicomponent disease [196] with severe comorbidities, such as lung cancer, cardiovascular diseases, and systemic and pulmonary inflammation. Recently, orally administered broad-spectrum anti-inflammatory therapies have been in clinical development, but they appear to have remarkable side effects, so inhaled drugs should be developed for future perspectives. Another approach should be to develop a drug that reverses the resistance of corticosteroids, which is supposed to be a massive barrier to the therapies [197]. In the future, a clear understanding of the molecular mechanism behind corticosteroid resistance may lead to new therapeutic approaches.
Another future area of research should consider COPD and various comorbidities, including osteoporosis, diabetes, and cardiac disease, as a disease of accelerated aging. Molecular pathways behind aging are very well understood and reveal many novel drug targets as interventions, including peroxisome proliferator-activated-γ coactivator 1α and antiaging molecules sirtuin 1 [198]. This review has discussed different types of diagnostic biomarkers used to detect and confirm COPD. These biomarkers help identify the diseased population and aid us in redefining the disease classification. COPD pathogenesis majorly consists of overexpression of systemic inflammation markers and signaling pathways. Thus, determining these markers is one of the imperative and crucial directions in improving the diagnosis and management of COPD.
Acknowledgments
MIH thanks the Council of Scientific and Industrial Research for financial support (Project No. 27(0368)/20/EMR-II). The authors thank the Department of Science and Technology, Government of India, for the FIST support (FIST Program No. SR/FST/LSII/2020/782). This work is supported by the Indian Council of Medical Research, Government of India (ISRM/12(22)/2020). SSS is funded by grants from Clifford Craig Foundation Launceston General Hospital.
Data Availability
The data that support this study's findings are available in this article.
Conflicts of Interest
The authors declared they had no conflicts of interest.
Authors' Contributions
IS, AA, SSS, and MIH contributed to conceptualization and project administration. IS and AA contributed to methodology. SSS and MIH contributed to validation and funding acquisition. AA contributed to formal analysis. IS, AI, AA, and MIH contributed to investigation and writing—original draft preparation. IS, SNK, MA, and MIH contributed to writing—review and editing. MA and MIH contributed to supervision. Ilma Shakeel and Anam Ashraf equally contributed to this work.
References
- 1.Eapen M. S., Myers S., Walters E. H., Sohal S. S. Airway inflammation in chronic obstructive pulmonary disease (COPD): a true paradox. Expert Review of Respiratory Medicine . 2017;11(10):827–839. doi: 10.1080/17476348.2017.1360769. [DOI] [PubMed] [Google Scholar]
- 2.Eisner M. D., Balmes J., Katz P. P., Trupin L., Yelin E. H., Blanc P. D. Lifetime environmental tobacco smoke exposure and the risk of chronic obstructive pulmonary disease. Environmental Health . 2005;4(1) doi: 10.1186/1476-069X-4-7.7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hersh C. P., DeMeo D. L., Silverman E. K. National Emphysema Treatment Trial state of the art: genetics of emphysema. Proceedings of the American Thoracic Society . 2008;5(4):486–493. doi: 10.1513/pats.200706-078ET. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Blanc P. D., Iribarren C., Trupin L., et al. Occupational exposures and the risk of COPD: dusty trades revisited. Thorax . 2009;64(1):6–12. doi: 10.1136/thx.2008.099390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hogg J. C., Chu F., Utokaparch S., et al. The nature of small-airway obstruction in chronic obstructive pulmonary disease. New England Journal of Medicine . 2004;350(26):2645–2653. doi: 10.1056/NEJMoa032158. [DOI] [PubMed] [Google Scholar]
- 6.Rabe K. F., Hurd S., Anzueto A., et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. American Journal of Respiratory and Critical Care Medicine . 2007;176(6):532–555. doi: 10.1164/rccm.200703-456SO. [DOI] [PubMed] [Google Scholar]
- 7.Venkatesan P. GOLD COPD report: 2023 update. The Lancet Respiratory Medicine . 2023;11(1) doi: 10.1016/S2213-2600(22)00494-5.18 [DOI] [PubMed] [Google Scholar]
- 8.Lamb D., McLean A., Gillooly M., Warren P. M., Gould G. A., MacNee W. Relation between distal airspace size, bronchiolar attachments, and lung function. Thorax . 1993;48(10):1012–1017. doi: 10.1136/thx.48.10.1012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mannino D. M., Thorn D., Swensen A., Holguin F. Prevalence and outcomes of diabetes, hypertension and cardiovascular disease in COPD. European Respiratory Journal . 2008;32(4):962–969. doi: 10.1183/09031936.00012408. [DOI] [PubMed] [Google Scholar]
- 10.Fabbri L. M., Rabe K. F. From COPD to chronic systemic inflammatory syndrome? The Lancet . 2007;370(9589):797–799. doi: 10.1016/S0140-6736(07)61383-X. [DOI] [PubMed] [Google Scholar]
- 11.FDA-NIH Biomarker Working Group. BEST (biomarkers, endpoints, and other tools) resource. 2016. [PubMed]
- 12.Wang C., Zhou J., Wang J., et al. Progress in the mechanism and targeted drug therapy for COPD. Signal Transduction and Targeted Therapy . 2020;5(1) doi: 10.1038/s41392-020-00345-x.248 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lin C.-H., Cheng S.-L., Chen C.-Z., Chen C.-H., Lin S.-H., Wang H.-C. Current progress of COPD early detection: key points and novel strategies. International Journal of Chronic Obstructive Pulmonary Disease . 2023;18:1511–1524. doi: 10.2147/COPD.S413969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Barnes P. J. Future treatments for chronic obstructive pulmonary disease and its comorbidities. Proceedings of the American Thoracic Society . 2008;5(8):857–864. doi: 10.1513/pats.200807-069TH. [DOI] [PubMed] [Google Scholar]
- 15.Chung K. F., Adcock I. M. Multifaceted mechanisms in COPD: inflammation, immunity, and tissue repair and destruction. European Respiratory Journal . 2008;31(6):1334–1356. doi: 10.1183/09031936.00018908. [DOI] [PubMed] [Google Scholar]
- 16.Yao H., Rahman I. Current concepts on the role of inflammation in COPD and lung cancer. Current Opinion in Pharmacology . 2009;9(4):375–383. doi: 10.1016/j.coph.2009.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hogg J. C., Timens W. The pathology of chronic obstructive pulmonary disease. Annual Review of Pathology: Mechanisms of Disease . 2009;4:435–459. doi: 10.1146/annurev.pathol.4.110807.092145. [DOI] [PubMed] [Google Scholar]
- 18.Siafakas N. M., Antoniou K. M., Tzortzaki E. G. Role of angiogenesis and vascular remodeling in chronic obstructive pulmonary disease. International Journal of Chronic Obstructive Pulmonary Disease . 2007;2(4):453–462. [PMC free article] [PubMed] [Google Scholar]
- 19.Houghton A. M., Quintero P. A., Perkins D. L., et al. Elastin fragments drive disease progression in a murine model of emphysema. The Journal of Clinical Investigation . 2006;116(3):753–759. doi: 10.1172/JCI25617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mathers C. D., Lopez A. D., Murray C., Ezzati M., Jamison D. Global Burden of Disease and Risk Factors . Washington, DC, USA: World Bank; 2013. [PubMed] [Google Scholar]
- 21.Lundbäck B., Lindberg A., Lindström M., et al. Not 15 but 50% of smokers develop COPD?—Report from the obstructive lung disease in Northern Sweden studies. Respiratory Medicine . 2003;97(2):115–122. doi: 10.1053/rmed.2003.1446. [DOI] [PubMed] [Google Scholar]
- 22.Gilliland F. D., Li Y.-F., Dubeau L., et al. Effects of glutathione S-transferase M1, maternal smoking during pregnancy, and environmental tobacco smoke on asthma and wheezing in children. American Journal of Respiratory and Critical Care Medicine . 2002;166(4):457–463. doi: 10.1164/rccm.2112064. [DOI] [PubMed] [Google Scholar]
- 23.Tønnesen P. Smoking cessation and COPD. European Respiratory Review . 2013;22(127):37–43. doi: 10.1183/09059180.00007212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tashkin D. P. Smoked marijuana as a cause of lung injury. Monaldi Archives for Chest Disease . 2005;63(2) doi: 10.4081/monaldi.2005.645. [DOI] [PubMed] [Google Scholar]
- 25.Murgia N., Gambelunghe A. Occupational COPD—the most under-recognized occupational lung disease? Respirology . 2022;27(6):399–410. doi: 10.1111/resp.14272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hnizdo E., Sullivan P. A., Bang K. M., Wagner G. Association between chronic obstructive pulmonary disease and employment by industry and occupation in the US population: a study of data from the Third National Health and Nutrition Examination Survey. American Journal of Epidemiology . 2002;156(8):738–746. doi: 10.1093/aje/kwf105. [DOI] [PubMed] [Google Scholar]
- 27.Trupin L., Earnest G., San Pedro M., et al. The occupational burden of chronic obstructive pulmonary disease. European Respiratory Journal . 2003;22(3):462–469. doi: 10.1183/09031936.03.00094203. [DOI] [PubMed] [Google Scholar]
- 28.Ran P.-X., Wang C., Yao W.-Z., et al. The risk factors for chronic obstructive pulmonary disease in females in Chinese rural areas. Zhonghua Nei Ke Za Zhi . 2006;45(12):974–979. [PubMed] [Google Scholar]
- 29.A Report of the Surgeon General. The Health Consequences of Involuntary Exposure to Tobacco Smoke: A Report of the Surgeon General . Atlanta, GA, USA: US Department of Health and Human Services, Centers for Disease; 2006. [Google Scholar]
- 30.Mannino D. M., Watt G., Hole D., et al. The natural history of chronic obstructive pulmonary disease. European Respiratory Journal . 2006;27(3):627–643. doi: 10.1183/09031936.06.00024605. [DOI] [PubMed] [Google Scholar]
- 31.Stoller J. K., Aboussouan L. S. α1-Antitrypsin deficiency. The Lancet . 2005;365(9478):2225–2236. doi: 10.1016/S0140-6736(05)66781-5. [DOI] [PubMed] [Google Scholar]
- 32.Cheng S. L., Yu C. J., Chen C. J., Yang P. C. Genetic polymorphism of epoxide hydrolase and glutathione S-transferase in COPD. European Respiratory Journal . 2004;23(6):818–824. doi: 10.1183/09031936.04.00104904. [DOI] [PubMed] [Google Scholar]
- 33.Keatings V. M., Cave S. J., Henry M. J., et al. A polymorphism in the tumor necrosis factor-α gene promoter region may predispose to a poor prognosis in COPD. Chest . 2000;118(4):971–975. doi: 10.1378/chest.118.4.971. [DOI] [PubMed] [Google Scholar]
- 34.Celedón J. C., Lange C., Raby B. A., et al. The transforming growth factor-β1 (TGFB1) gene is associated with chronic obstructive pulmonary disease (COPD) Human Molecular Genetics . 2004;13(15):1649–1656. doi: 10.1093/hmg/ddh171. [DOI] [PubMed] [Google Scholar]
- 35.Zhou Y., Bai F., Li X., et al. Genetic polymorphisms in MIR1208 and MIR5708 are associated with susceptibility to COPD in the Chinese population. Pulmonology . 2023;29(1):6–12. doi: 10.1016/j.pulmoe.2021.07.004. [DOI] [PubMed] [Google Scholar]
- 36.Wedzicha J. A., Seemungal T. A. R. COPD exacerbations: defining their cause and prevention. The Lancet . 2007;370(9589):786–796. doi: 10.1016/S0140-6736(07)61382-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mannino D. M., Davis K. J. Lung function decline and outcomes in an elderly population. Thorax . 2006;61(6):472–477. doi: 10.1136/thx.2005.052449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kakkera K., Atchley W. T., Kodali M., Bartter T. Ageing and chronic obstructive pulmonary disease: interrelationships. Current Opinion in Pulmonary Medicine . 2023;29(2):90–95. doi: 10.1097/MCP.0000000000000943. [DOI] [PubMed] [Google Scholar]
- 39.Jemal A., Ward E., Hao Y., Thun M. Trends in the leading causes of death in the United States, 1970–2002. JAMA . 2005;294(10):1255–1259. doi: 10.1001/jama.294.10.1255. [DOI] [PubMed] [Google Scholar]
- 40.Shohaimi S., Welch A., Bingham S., et al. Area deprivation predicts lung function independently of education and social class. European Respiratory Journal . 2004;24(1):157–161. doi: 10.1183/09031936.04.00088303. [DOI] [PubMed] [Google Scholar]
- 41.Eisner M. D., Blanc P. D., Omachi T. A., et al. Socioeconomic status, race and COPD health outcomes. Journal of Epidemiology and Community Health . 2010;65(1):26–34. doi: 10.1136/jech.2009.089722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.de Torres J. P., Casanova C., Hernández C., Abreu J., Aguirre-Jaime A., Celli B. R. Gender and COPD in patients attending a pulmonary clinic. Chest . 2005;128(4):2012–2016. doi: 10.1378/chest.128.4.2012. [DOI] [PubMed] [Google Scholar]
- 43.Silverman E. K., Weiss S. T., Drazen J. M., et al. Gender-related differences in severe, early-onset chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine . 2000;162(6):2152–2158. doi: 10.1164/ajrccm.162.6.2003112. [DOI] [PubMed] [Google Scholar]
- 44.Watson L., Vonk J. M., Löfdahl C. G., et al. Predictors of lung function and its decline in mild to moderate COPD in association with gender: results from the Euroscop study. Respiratory Medicine . 2006;100(4):746–753. doi: 10.1016/j.rmed.2005.08.004. [DOI] [PubMed] [Google Scholar]
- 45.Agustí A. Systemic effects of chronic obstructive pulmonary disease: what we know and what we don’t know (but should) Proceedings of the American Thoracic Society . 2007;4(7):522–525. doi: 10.1513/pats.200701-004FM. [DOI] [PubMed] [Google Scholar]
- 46.Sproston N. R., Ashworth J. J. Role of C-reactive protein at sites of inflammation and infection. Frontiers in Immunology . 2018;9 doi: 10.3389/fimmu.2018.00754.754 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Higashimoto Y., Yamagata Y., Taya S., et al. Systemic inflammation in chronic obstructive pulmonary disease and asthma: similarities and differences. Respirology . 2008;13(1):128–133. doi: 10.1111/j.1440-1843.2007.01170.x. [DOI] [PubMed] [Google Scholar]
- 48.Aksu F., Capan N., Aksu K., et al. C-reactive protein levels are raised in stable chronic obstructive pulmonary disease patients independent of smoking behavior and biomass exposure. Journal of Thoracic Disease . 2013;5(4):414–421. doi: 10.3978/j.issn.2072-1439.2013.06.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Broekhuizen R., Wouters E. F., Creutzberg E. C., Schols A. M. Raised CRP levels mark metabolic and functional impairment in advanced COPD. Thorax . 2005;61(1):17–22. doi: 10.1136/thx.2005.041996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Leuzzi G., Galeone C., Taverna F., Suatoni P., Morelli D., Pastorino U. C-reactive protein level predicts mortality in COPD: a systematic review and meta-analysis. European Respiratory Review . 2017;26(143) doi: 10.1183/16000617.0070-2016.160070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Dahl M., Vestbo J., Lange P., Bojesen S. E., Tybjærg-Hansen A., Nordestgaard B. G. C-reactive protein as a predictor of prognosis in chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine . 2007;175(3):250–255. doi: 10.1164/rccm.200605-713OC. [DOI] [PubMed] [Google Scholar]
- 52.Munuswamy R., De Brandt J., Burtin C., et al. Monomeric CRP is elevated in patients with COPD compared to non-COPD control persons. Journal of Inflammation Research . 2021;14:4503–4507. doi: 10.2147/JIR.S320659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tangedal S., Aanerud M., Persson L. J. P., Brokstad K. A., Bakke P. S., Eagan T. M. Comparison of inflammatory markers in induced and spontaneous sputum in a cohort of COPD patients. Respiratory Research . 2014;15(1):1–8. doi: 10.1186/s12931-014-0138-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wei J., Xiong X.-F., Lin Y.-H., Zheng B.-X., Cheng D.-Y. Association between serum interleukin-6 concentrations and chronic obstructive pulmonary disease: a systematic review and meta-analysis. PeerJ . 2015;3 doi: 10.7717/peerj.1199.e1199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Karadag F., Karul A. B., Cildag O., Yilmaz M., Ozcan H. Biomarkers of systemic inflammation in stable and exacerbation phases of COPD. Lung . 2008;186(6):403–409. doi: 10.1007/s00408-008-9106-6. [DOI] [PubMed] [Google Scholar]
- 56.Singh S., Verma S. K., Kumar S., et al. Correlation of severity of chronic obstructive pulmonary disease with potential biomarkers. Immunology Letters . 2018;196:1–10. doi: 10.1016/j.imlet.2018.01.004. [DOI] [PubMed] [Google Scholar]
- 57.Kolsum U., Roy K., Starkey C., et al. The repeatability of interleukin-6, tumor necrosis factor-α, and C-reactive protein in COPD patients over one year. International Journal of Chronic Obstructive Pulmonary Disease . 2009;4:149–156. doi: 10.2147/copd.s5018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wedzicha J., Seemungal T., MacCallum P., et al. Acute exacerbations of chronic obstructive pulmonary disease are accompanied by elevations of plasma fibrinogen and serum IL-6 levels. Thrombosis and Haemostasis . 2017;84(8):210–215. doi: 10.1055/s-0037-1613998. [DOI] [PubMed] [Google Scholar]
- 59.Ardestani M. E., Zaerin O. Role of serum interleukin 6, albumin and C-reactive protein in COPD patients. Tanaffos . 2015;14(2):134–140. [PMC free article] [PubMed] [Google Scholar]
- 60.Celli B. R., Locantore N., Yates J., et al. Inflammatory biomarkers improve clinical prediction of mortality in chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine . 2012;185(10):1065–1072. doi: 10.1164/rccm.201110-1792OC. [DOI] [PubMed] [Google Scholar]
- 61.Brinkley T. E., Leng X., Miller M. E., et al. Chronic inflammation is associated with low physical function in older adults across multiple comorbidities. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences . 2009;64A(4):455–461. doi: 10.1093/gerona/gln038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Chaouat A., Savale L., Chouaid C., et al. Role for interleukin-6 in COPD-related pulmonary hypertension. Chest . 2009;136(3):678–687. doi: 10.1378/chest.08-2420. [DOI] [PubMed] [Google Scholar]
- 63.Godoy I., Corrêa C. R., Corrêa C., Caram L. M., Tanni S. E., Ferrari R. Three-year follow-up of Interleukin 6 and C-reactive protein in chronic obstructive pulmonary disease. Respiratory Research . 2013;14(1) doi: 10.1186/1465-9921-14-24.24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Vietri L., Fui A., Bergantini L., et al. Serum amyloid A: a potential biomarker of lung disorders. Respiratory Investigation . 2020;58(1):21–27. doi: 10.1016/j.resinv.2019.09.005. [DOI] [PubMed] [Google Scholar]
- 65.Bozinovski S., Hutchinson A., Thompson M., et al. Serum amyloid a is a biomarker of acute exacerbations of chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine . 2008;177(3):269–278. doi: 10.1164/rccm.200705-678OC. [DOI] [PubMed] [Google Scholar]
- 66.Lin T. L., Chen W. W., Ding Z. R., Wei S. C., Huang M. L., Li C. H. Correlations between serum amyloid A, C-reactive protein and clinical indices of patients with acutely exacerbated chronic obstructive pulmonary disease. Journal of Clinical Laboratory Analysis . 2019;33(4) doi: 10.1002/jcla.22831.e22831 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Wang H., Anthony D., Selemidis S., Vlahos R., Bozinovski S. Resolving viral-induced secondary bacterial infection in COPD: a concise review. Frontiers in Immunology . 2018;9 doi: 10.3389/fimmu.2018.02345.2345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Zhao D., Abbasi A., Rossiter H. B., et al. Serum amyloid A in stable COPD patients is associated with the frequent exacerbator phenotype. International Journal of Chronic Obstructive Pulmonary Disease . 2020;15:2379–2388. doi: 10.2147/COPD.S266844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ferrarotti I., Zorzetto M., Beccaria M., et al. Tumour necrosis factor family genes in a phenotype of COPD associated with emphysema. European Respiratory Journal . 2003;21(3):444–449. doi: 10.1183/09031936.03.00051303. [DOI] [PubMed] [Google Scholar]
- 70.Danilko K., Korytyna G., Akhmadishina L., Yanbaeva D., Zagidullin S. Z., Victorova T. Association of polymorphisms of cytokine genes (IL1B, IL8, and IL10) with chronic obstructive pulmonary disease. Molecular Biology . 2007;41(1):26–36. [PubMed] [Google Scholar]
- 71.Yao Y., Zhou J., Diao X., Wang S. Association between tumor necrosis factor-α and chronic obstructive pulmonary disease: a systematic review and meta-analysis. Therapeutic Advances in Respiratory Disease . 2019;13 doi: 10.1177/1753466619866096.1753466619866096 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Churg A., Wang R. D., Tai H., Wang X., Xie C., Wright J. L. Tumor necrosis factor-α drives 70% of cigarette smoke-induced emphysema in the mouse. American Journal of Respiratory and Critical Care Medicine . 2004;170(5):492–498. doi: 10.1164/rccm.200404-511OC. [DOI] [PubMed] [Google Scholar]
- 73.Matera M. G., Calzetta L., Cazzola M. TNF-α inhibitors in asthma and COPD: we must not throw the baby out with the bath water. Pulmonary Pharmacology & Therapeutics . 2010;23(2):121–128. doi: 10.1016/j.pupt.2009.10.007. [DOI] [PubMed] [Google Scholar]
- 74.Kubysheva N., Boldina M., Eliseeva T., et al. Relationship of serum levels of IL-17, IL-18, TNF-α, and lung function parameters in patients with COPD, asthma-COPD overlap, and bronchial asthma. Mediators of Inflammation . 2020;2020:11. doi: 10.1155/2020/4652898.4652898 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Duvoix A., Dickens J., Haq I., et al. Blood fibrinogen as a biomarker of chronic obstructive pulmonary disease. Thorax . 2013;68(7):670–676. doi: 10.1136/thoraxjnl-2012-201871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Dahl M., Tybjœrg-Hansen A., Vestbo J., Lange P., Nordestgaard B. G. Elevated plasma fibrinogen associated with reduced pulmonary function and increased risk of chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine . 2001;164(6):1008–1011. doi: 10.1164/ajrccm.164.6.2010067. [DOI] [PubMed] [Google Scholar]
- 77.Groenewegen K. H., Postma D. S., Hop W. C. J., Wielders P. L. M. L., Schlösser N. J. J., Wouters E. F. M. Increased systemic inflammation is a risk factor for COPD exacerbations. Chest . 2008;133(2):350–357. doi: 10.1378/chest.07-1342. [DOI] [PubMed] [Google Scholar]
- 78.Li M., Wu Y., Han H., Yuan Y. Clinical significance of plasma D-dimer and fibrinogen detection in patients with AECOPD with hypoxemia. Journal of Chinese Physician . 2020;12:1464–1467. [Google Scholar]
- 79.Hu X., Xu J., Li P., Zheng H. Correlation of serum clara cell secretory protein 16, plasma fibrinogen and serum amyloid A with the severity of acute exacerbated COPD and their combination in prognosis assessment. International Journal of Chronic Obstructive Pulmonary Disease . 2023;18:1949–1957. doi: 10.2147/COPD.S410917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Miller B. E., Tal-Singer R., Rennard S. I., et al. Plasma fibrinogen qualification as a drug development tool in chronic obstructive pulmonary disease. Perspective of the chronic obstructive pulmonary disease biomarker qualification consortium. American Journal of Respiratory and Critical Care Medicine . 2016;193(6):607–613. doi: 10.1164/rccm.201509-1722PP. [DOI] [PubMed] [Google Scholar]
- 81.Zhou B., Liu S., He D., et al. Fibrinogen is a promising biomarker for chronic obstructive pulmonary disease: evidence from a meta-analysis. Bioscience Reports . 2020;40(7) doi: 10.1042/BSR20193542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Zhao Q., Meng M., Kumar R., et al. The impact of COPD and smoking history on the severity of COVID-19: a systemic review and meta-analysis. Journal of Medical Virology . 2020;92(10):1915–1921. doi: 10.1002/jmv.25889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Alqahtani J. S., Oyelade T., Aldhahir A. M., et al. Prevalence, severity and mortality associated with COPD and smoking in patients with COVID-19: a rapid systematic review and meta-analysis. PLoS One . 2020;15(5) doi: 10.1371/journal.pone.0233147.e0233147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Naqvi A. A. T., Fatima K., Mohammad T., et al. Insights into SARS-CoV-2 genome, structure, evolution, pathogenesis and therapies: structural genomics approach. Biochimica et Biophysica Acta Molecular Basis of Disease . 2010;1866(10) doi: 10.1016/j.bbadis.2020.165878.165878 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Guan W.-J., Liang W.-H., Zhao Y., et al. Comorbidity and its impact on 1590 patients with COVID-19 in China: a nationwide analysis. European Respiratory Journal . 2020;55(5) doi: 10.1183/13993003.00547-2020.2000547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Asrani P., Eapen M. S., Hassan M. I., Sohal S. S. Implications of the second wave of COVID-19 in India. The Lancet Respiratory Medicine . 2021;9(9):e93–e94. doi: 10.1016/S2213-2600(21)00312-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Asrani P., Hassan M. I. SARS-CoV-2 mediated lung inflammatory responses in host: targeting the cytokine storm for therapeutic interventions. Molecular and Cellular Biochemistry . 2021;476(2):675–687. doi: 10.1007/s11010-020-03935-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Hoffmann M., Kleine-Weber H., Schroeder S., et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell . 2020;181(2):271–280.e8. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Walls A. C., Park Y.-J., Tortorici M. A., Wall A., McGuire A. T., Veesler D. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell . 2020;181(2):281–292.e6. doi: 10.1016/j.cell.2020.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Leung J. M., Yang C. X., Tam A., et al. ACE-2 expression in the small airway epithelia of smokers and COPD patients: implications for COVID-19. European Respiratory Journal . 2020;55(5) doi: 10.1183/13993003.00688-2020.2000688 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.HCA Lung Biological Network, Sungnak W., Huang N., et al. SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes. Nature Medicine . 2020;26(5):681–687. doi: 10.1038/s41591-020-0868-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Tockman M. S., Anthonisen N. R., Wright E. C., Donithan M. G. Airways obstruction and the risk for lung cancer. Annals of Internal Medicine . 1987;106(4):512–518. doi: 10.7326/0003-4819-106-4-512. [DOI] [PubMed] [Google Scholar]
- 93.Sin D. D., Anthonisen N. R., Soriano J. B., Agusti A. G. Mortality in COPD: role of comorbidities. European Respiratory Journal . 2006;28(6):1245–1257. doi: 10.1183/09031936.00133805. [DOI] [PubMed] [Google Scholar]
- 94.Zhang H. Molecular signaling and genetic pathways of senescence: its role in tumorigenesis and aging. Journal of Cellular Physiology . 2007;210(3):567–574. doi: 10.1002/jcp.20919. [DOI] [PubMed] [Google Scholar]
- 95.Lin W.-W., Karin M. A cytokine-mediated link between innate immunity, inflammation, and cancer. Journal of Clinical Investigation . 2007;117(5):1175–1183. doi: 10.1172/JCI31537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Cho H.-Y., Reddy S. P., Kleeberger S. R. Nrf2 defends the lung from oxidative stress. Antioxidants & Redox Signaling . 2006;8(1-2):76–87. doi: 10.1089/ars.2006.8.76. [DOI] [PubMed] [Google Scholar]
- 97.Turner M. C., Chen Y., Krewski D., Calle E. E., Thun M. J. Chronic obstructive pulmonary disease is associated with lung cancer mortality in a prospective study of never smokers. American Journal of Respiratory and Critical Care Medicine . 2007;176(3):285–290. doi: 10.1164/rccm.200612-1792OC. [DOI] [PubMed] [Google Scholar]
- 98.Cohen S. B.-Z., Paré P. D., Man S. F. P., Sin D. D. The growing burden of chronic obstructive pulmonary disease and lung cancer in women: examining sex differences in cigarette smoke metabolism. American Journal of Respiratory and Critical Care Medicine . 2007;176(2):113–120. doi: 10.1164/rccm.200611-1655PP. [DOI] [PubMed] [Google Scholar]
- 99.Agusti A., Soriano J. B. COPD as a systemic disease. COPD: Journal of Chronic Obstructive Pulmonary Disease . 2009;5(2):133–138. doi: 10.1080/15412550801941349. [DOI] [PubMed] [Google Scholar]
- 100.Gosker H. R., Kubat B., Schaart G., van der Vusse G. J., Wouters E. F. M., Schols A. M. W. J. Myopathological features in skeletal muscle of patients with chronic obstructive pulmonary disease. European Respiratory Journal . 2003;22(2):280–285. doi: 10.1183/09031936.03.00012803. [DOI] [PubMed] [Google Scholar]
- 101.Marquis K., Debigaré R., Lacasse Y., et al. Midthigh muscle cross-sectional area is a better predictor of mortality than body mass index in patients with chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine . 2002;166(6):809–813. doi: 10.1164/rccm.2107031. [DOI] [PubMed] [Google Scholar]
- 102.Spranger J., Kroke A., Möhlig M., et al. Inflammatory cytokines and the risk to develop type 2 diabetes: results of the prospective population-based European Prospective Investigation into Cancer and Nutrition (EPIC)—Potsdam study. Diabetes . 2003;52(3):812–817. doi: 10.2337/diabetes.52.3.812. [DOI] [PubMed] [Google Scholar]
- 103.Poulain M., Doucet M., Drapeau V., et al. Metabolic and inflammatory profile in obese patients with chronic obstructive pulmonary disease. Chronic Respiratory Disease . 2008;5(1):35–41. doi: 10.1177/1479972307087205. [DOI] [PubMed] [Google Scholar]
- 104.Jørgensen N. R., Schwarz P. Osteoporosis in chronic obstructive pulmonary disease patients. Current Opinion in Pulmonary Medicine . 2008;14(2):122–127. doi: 10.1097/MCP.0b013e3282f4efb6. [DOI] [PubMed] [Google Scholar]
- 105.Calverley P. M. A., Anderson J. A., Celli B., et al. Salmeterol and fluticasone propionate and survival in chronic obstructive pulmonary disease. New England Journal of Medicine . 2007;356(8):775–789. doi: 10.1056/NEJMoa063070. [DOI] [PubMed] [Google Scholar]
- 106.Vrieze A., de Greef M. H. G., Wýkstra P. J., Wempe J. B. Low bone mineral density in COPD patients related to worse lung function, low weight and decreased fat-free mass. Osteoporosis International . 2007;18(9):1197–1202. doi: 10.1007/s00198-007-0355-7. [DOI] [PubMed] [Google Scholar]
- 107.Jørgensen N. R., Schwarz P., Holme I., Henriksen B. M., Petersen L. J., Backer V. The prevalence of osteoporosis in patients with chronic obstructive pulmonary disease—a cross sectional study. Respiratory Medicine . 2007;101(1):177–185. doi: 10.1016/j.rmed.2006.03.029. [DOI] [PubMed] [Google Scholar]
- 108.Carter J. D., Patel S., Sultan F. L., et al. The recognition and treatment of vertebral fractures in males with chronic obstructive pulmonary disease. Respiratory Medicine . 2008;102(8):1165–1172. doi: 10.1016/j.rmed.2008.03.003. [DOI] [PubMed] [Google Scholar]
- 109.John M., Lange A., Hoernig S., Witt C., Anker S. D. Prevalence of anemia in chronic obstructive pulmonary disease: comparison to other chronic diseases. International Journal of Cardiology . 2006;111(3):365–370. doi: 10.1016/j.ijcard.2005.07.043. [DOI] [PubMed] [Google Scholar]
- 110.Shorr A. F., Doyle J., Stern L., Dolgitser M., Zilberberg M. D. Anemia in chronic obstructive pulmonary disease: epidemiology and economic implications. Current Medical Research and Opinion . 2008;24(4):1123–1130. doi: 10.1185/030079908X280699. [DOI] [PubMed] [Google Scholar]
- 111.Cote C., Zilberberg M. D., Mody S. H., Dordelly L. J., Celli B. Haemoglobin level and its clinical impact in a cohort of patients with COPD. European Respiratory Journal . 2007;29(5):923–929. doi: 10.1183/09031936.00137106. [DOI] [PubMed] [Google Scholar]
- 112.John M., Hoernig S., Doehner W., Okonko D. D., Witt C., Anker S. D. Anemia and inflammation in COPD. Chest . 2005;127(3):825–829. doi: 10.1378/chest.127.3.825. [DOI] [PubMed] [Google Scholar]
- 113.Schonhofer B., Wenzel M., Geibel M., Kohler D. Blood transfusion and lung function in chronically anemic patients with severe chronic obstructive pulmonary disease. Critical Care Medicine . 1998;26(11):1824–1828. doi: 10.1097/00003246-199811000-00022. [DOI] [PubMed] [Google Scholar]
- 114.Fletcher E. C. Chronic lung disease in the sleep apnea syndrome. Lung . 1990;168(S1):751–761. doi: 10.1007/BF02718204. [DOI] [PubMed] [Google Scholar]
- 115.Alam I., Lewis K., Stephens J. W., Baxter J. N. Obesity, metabolic syndrome and sleep apnoea: all pro-inflammatory states. Obesity Reviews . 2007;8(2):119–127. doi: 10.1111/j.1467-789X.2006.00269.x. [DOI] [PubMed] [Google Scholar]
- 116.Jelic S., Padeletti M., Kawut S. M., et al. Inflammation, oxidative stress, and repair capacity of the vascular endothelium in obstructive sleep apnea. Circulation . 2008;117(17):2270–2278. doi: 10.1161/CIRCULATIONAHA.107.741512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Yohannes A. M., Baldwin R. C., Connolly M. J. Depression and Anxiety in Elderly Patients with Chronic Obstructive Pulmonary Disease . New York: Oxford University Press; 2006. [DOI] [PubMed] [Google Scholar]
- 118.Hill K., Geist R., Goldstein R. S., Lacasse Y. Anxiety and depression in end-stage COPD. European Respiratory Journal . 2008;31(3):667–677. doi: 10.1183/09031936.00125707. [DOI] [PubMed] [Google Scholar]
- 119.Anisman H., Merali Z., Hayley S. Neurotransmitter, peptide and cytokine processes in relation to depressive disorder: comorbidity between depression and neurodegenerative disorders. Progress in Neurobiology . 2008;85(1):1–74. doi: 10.1016/j.pneurobio.2008.01.004. [DOI] [PubMed] [Google Scholar]
- 120.Sin D. D., Wu L. L., Man S. F. P. The relationship between reduced lung function and cardiovascular mortality: a population-based study and a systematic review of the literature. Chest . 2005;127(6):1952–1959. doi: 10.1378/chest.127.6.1952. [DOI] [PubMed] [Google Scholar]
- 121.Anthonisen N. R., Skeans M. A., Wise R. A., et al. The effects of a smoking cessation intervention on 14.5-year mortality: a randomized clinical trial. Annals of Internal Medicine . 2005;142(4):233–239. doi: 10.7326/0003-4819-142-4-200502150-00005. [DOI] [PubMed] [Google Scholar]
- 122.Barger G., Dale H. H. Chemical structure and sympathomimetic action of amines. The Journal of Physiology . 1910;41(1-2):19–59. doi: 10.1113/jphysiol.1910.sp001392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Rutten F. H., Cramer M. J. M., Lammers J. W. J., Grobbee D. E., Hoes A. W. Heart failure and chronic obstructive pulmonary disease: an ignored combination? European Journal of Heart Failure . 2006;8(7):706–711. doi: 10.1016/j.ejheart.2006.01.010. [DOI] [PubMed] [Google Scholar]
- 124.Rutten F. H., Cramer M.-J. M., Zuithoff N. P. A., et al. Comparison of B-type natriuretic peptide assays for identifying heart failure in stable elderly patients with a clinical diagnosis of chronic obstructive pulmonary disease. European Journal of Heart Failure . 2007;9(6-7):651–659. doi: 10.1016/j.ejheart.2007.01.010. [DOI] [PubMed] [Google Scholar]
- 125.Padeletti M., Jelic S., LeJemtel T. H. Coexistent chronic obstructive pulmonary disease and heart failure in the elderly. International Journal of Cardiology . 2008;125(2):209–215. doi: 10.1016/j.ijcard.2007.12.001. [DOI] [PubMed] [Google Scholar]
- 126.Watz H., Waschki B., Boehme C., Claussen M., Meyer T., Magnussen H. Extrapulmonary effects of chronic obstructive pulmonary disease on physical activity: a cross-sectional study. American Journal of Respiratory and Critical Care Medicine . 2008;177(7):743–751. doi: 10.1164/rccm.200707-1011OC. [DOI] [PubMed] [Google Scholar]
- 127.Thabut G., Dauriat G. B., Stern J. B., et al. Pulmonary hemodynamics in advanced COPD candidates for lung volume reduction surgery or lung transplantation. Chest . 2005;127(5):1531–1536. doi: 10.1378/chest.127.5.1531. [DOI] [PubMed] [Google Scholar]
- 128.Giaid A., Saleh D. Reduced expression of endothelial nitric oxide synthase in the lungs of patients with pulmonary hypertension. New England Journal of Medicine . 1995;333(4):214–221. doi: 10.1056/NEJM199507273330403. [DOI] [PubMed] [Google Scholar]
- 129.Mattace-Raso F. U. S., van der Cammen T. J. M., Hofman A., et al. Arterial stiffness and risk of coronary heart disease and stroke: the Rotterdam study. Circulation . 2006;113(5):657–663. doi: 10.1161/CIRCULATIONAHA.105.555235. [DOI] [PubMed] [Google Scholar]
- 130.McAllister D. A., Maclay J. D., Mills N. L., et al. Arterial stiffness is independently associated with emphysema severity in patients with chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine . 2007;176(12):1208–1214. doi: 10.1164/rccm.200707-1080OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Mills N. L., Miller J. J., Anand A., et al. Increased arterial stiffness in patients with chronic obstructive pulmonary disease: a mechanism for increased cardiovascular risk. Thorax . 2008;63(4):306–311. doi: 10.1136/thx.2007.083493. [DOI] [PubMed] [Google Scholar]
- 132.Barnes P. J., Stockley R. COPD: current therapeutic interventions and future approaches. European Respiratory Journal . 2005;25(6):1084–1106. doi: 10.1183/09031936.05.00139104. [DOI] [PubMed] [Google Scholar]
- 133.Barnes P. J. Endo-phenotyping of COPD patients. Expert Review of Respiratory Medicine . 2021;15(1):27–37. doi: 10.1080/17476348.2020.1804364. [DOI] [PubMed] [Google Scholar]
- 134.Barnes P. J. COPD 2020: New Directions Needed . Bethesda, MD, USA: American Physiological Society; 2020. [Google Scholar]
- 135.Norman P. Investigational p38 inhibitors for the treatment of chronic obstructive pulmonary disease. Expert Opinion on Investigational Drugs . 2014;24(3):383–392. doi: 10.1517/13543784.2015.1006358. [DOI] [PubMed] [Google Scholar]
- 136.Fung-Leung W.-P. Phosphoinositide 3-kinase delta (PI3Kδ) in leukocyte signaling and function. Cellular Signalling . 2011;23(4):603–608. doi: 10.1016/j.cellsig.2010.10.002. [DOI] [PubMed] [Google Scholar]
- 137.Down K., Amour A., Baldwin I. R., et al. Optimization of novel indazoles as highly potent and selective inhibitors of phosphoinositide 3-kinase δ for the treatment of respiratory disease. Journal of Medicinal Chemistry . 2015;58(18):7381–7399. doi: 10.1021/acs.jmedchem.5b00767. [DOI] [PubMed] [Google Scholar]
- 138.Lakshmi S. P., Reddy A. T., Reddy R. C. Emerging pharmaceutical therapies for COPD. International Journal of Chronic Obstructive Pulmonary Disease . 2017;12:2141–2156. doi: 10.2147/COPD.S121416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Mathioudakis A. G., Chatzimavridou-Grigoriadou V., Evangelopoulou E., Mathioudakis G. Vasoactive intestinal peptide inhaled agonists: potential role in respiratory therapeutics. Hippokratia . 2013;17(1):12–16. [PMC free article] [PubMed] [Google Scholar]
- 140.Suzuki M. Present status and tasks for future of point of care testing. Rinsho Byori. The Japanese Journal of Clinical Pathology . 2002;50(10):953–957. [PubMed] [Google Scholar]
- 141.Goto K., Chiba Y., Misawa M. IL-13 induces translocation of NF-κB in cultured human bronchial smooth muscle cells. Cytokine . 2009;46(1):96–99. doi: 10.1016/j.cyto.2008.12.021. [DOI] [PubMed] [Google Scholar]
- 142.Snell N., Foster M., Vestbo J. Efficacy and safety of AZD1981, a CRTH2 receptor antagonist, in patients with moderate to severe COPD. Respiratory Medicine . 2013;107(11):1722–1730. doi: 10.1016/j.rmed.2013.06.006. [DOI] [PubMed] [Google Scholar]
- 143.Watz H., Bock D., Meyer M., et al. Inhaled pan-selectin antagonist bimosiamose attenuates airway inflammation in COPD. Pulmonary Pharmacology & Therapeutics . 2013;26(2):265–270. doi: 10.1016/j.pupt.2012.12.003. [DOI] [PubMed] [Google Scholar]
- 144.Barnes P. J. Oxidative stress-based therapeutics in COPD. Redox Biology . 2020;33 doi: 10.1016/j.redox.2020.101544.101544 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Zheng J.-P., Wen F.-Q., Bai C.-X., et al. Twice daily N-acetylcysteine 600 mg for exacerbations of chronic obstructive pulmonary disease (PANTHEON): a randomised, double-blind placebo-controlled trial. The Lancet Respiratory Medicine . 2014;2(3):187–194. doi: 10.1016/S2213-2600(13)70286-8. [DOI] [PubMed] [Google Scholar]
- 146.Tse H. N., Raiteri L., Wong K. Y., Ng L. Y., Yee K. S., Tseng C. Z. S. Benefits of high-dose N-acetylcysteine to exacerbation-prone patients with COPD. Chest . 2014;146(3):611–623. doi: 10.1378/chest.13-2784. [DOI] [PubMed] [Google Scholar]
- 147.Khabour O. F., Alzoubi K. H., Al-Sawalha N., Ahmad M. B., Shihadeh A., Eissenberg T. The effect of chronic exposure to waterpipe tobacco smoke on airway inflammation in mice. Life Sciences . 2018;200:110–114. doi: 10.1016/j.lfs.2018.03.034. [DOI] [PubMed] [Google Scholar]
- 148.Wise R. A., Holbrook J. T., Criner G., et al. Lack of effect of oral sulforaphane administration on Nrf2 expression in COPD: a randomized, double-blind, placebo controlled trial. PLoS One . 2016;11(11) doi: 10.1371/journal.pone.0163716.e0163716 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Beijers R. J., Gosker H. R., Sanders K. J. C., et al. Resveratrol and metabolic health in COPD: a proof-of-concept randomized controlled trial. Clinical Nutrition . 2020;39(10):2989–2997. doi: 10.1016/j.clnu.2020.01.002. [DOI] [PubMed] [Google Scholar]
- 150.Gu C., Li Y., Xu W.-L., et al. Sirtuin 1 activator SRT1720 protects against lung injury via reduction of type II alveolar epithelial cells apoptosis in emphysema. COPD: Journal of Chronic Obstructive Pulmonary Disease . 2014;12(4):444–452. doi: 10.3109/15412555.2014.974740. [DOI] [PubMed] [Google Scholar]
- 151.Ostridge K., Williams N., Kim V., et al. Relationship between pulmonary matrix metalloproteinases and quantitative CT markers of small airways disease and emphysema in COPD. Thorax . 2016;71(2):126–132. doi: 10.1136/thoraxjnl-2015-207428. [DOI] [PubMed] [Google Scholar]
- 152.Groutas W. C., Dou D., Alliston K. R. Neutrophil elastase inhibitors. Expert Opinion on Therapeutic Patents . 2011;21(3):339–354. doi: 10.1517/13543776.2011.551115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Kuna P., Jenkins M., O’Brien C. D., Fahy W. A. AZD9668, a neutrophil elastase inhibitor, plus ongoing budesonide/formoterol in patients with COPD. Respiratory Medicine . 2012;106(4):531–539. doi: 10.1016/j.rmed.2011.10.020. [DOI] [PubMed] [Google Scholar]
- 154.Moitra S. N-acetylcysteine (NAC) in COPD: benefits often lost in trials. QJM: An International Journal of Medicine . 2019;112(5):387–388. doi: 10.1093/qjmed/hcy166. [DOI] [PubMed] [Google Scholar]
- 155.Dey T., Kalita J., Weldon Séad, Taggart C. Proteases and their inhibitors in chronic obstructive pulmonary disease. Journal of Clinical Medicine . 2018;7(9) doi: 10.3390/jcm7090244.244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Dahl R., Titlestad I., Lindqvist A., et al. Effects of an oral MMP-9 and-12 inhibitor, AZD1236, on biomarkers in moderate/severe COPD: a randomised controlled trial. Pulmonary Pharmacology & Therapeutics . 2012;25(2):169–177. doi: 10.1016/j.pupt.2011.12.011. [DOI] [PubMed] [Google Scholar]
- 157.Adcock I. M., Caramori G., Barnes P. J. Chronic obstructive pulmonary disease and lung cancer: new molecular insights. Respiration . 2011;81(4):265–284. doi: 10.1159/000324601. [DOI] [PubMed] [Google Scholar]
- 158.Chapman R. W., Phillips J. E., Hipkin R. W., Curran A. K., Lundell D., Fine J. S. CXCR2 antagonists for the treatment of pulmonary disease. Pharmacology & Therapeutics . 2009;121(1):55–68. doi: 10.1016/j.pharmthera.2008.10.005. [DOI] [PubMed] [Google Scholar]
- 159.Wendling D., Vidon C., Godfrin-Valnet M., Rival G., Guillot X., Prati C. Exacerbation of combined pulmonary fibrosis and emphysema syndrome during tocilizumab therapy for rheumatoid arthritis. Joint Bone Spine . 2013;80(6):670–671. doi: 10.1016/j.jbspin.2013.03.009. [DOI] [PubMed] [Google Scholar]
- 160.Botelho F. M., Bauer C. M. T., Finch D., et al. IL-1α/IL-1R1 expression in chronic obstructive pulmonary disease and mechanistic relevance to smoke-induced neutrophilia in mice. PLoS One . 2011;6(12) doi: 10.1371/journal.pone.0028457.e28457 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Loza M. J., Watt R., Baribaud F., Barnathan E. S., Rennard S. I. Systemic inflammatory profile and response to anti-tumor necrosis factor therapy in chronic obstructive pulmonary disease. Respiratory Research . 2012;13(1):1–12. doi: 10.1186/1465-9921-13-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Rennard S. I., Flavin S. K., Agarwal P. K., Lo K. H., Barnathan E. S. Long-term safety study of infliximab in moderate-to-severe chronic obstructive pulmonary disease. Respiratory Medicine . 2013;107(3):424–432. doi: 10.1016/j.rmed.2012.11.008. [DOI] [PubMed] [Google Scholar]
- 163.Aaron S. D., Vandemheen K. L., Maltais F., et al. TNFα antagonists for acute exacerbations of COPD: a randomised double-blind controlled trial. Thorax . 2013;68(2):142–148. doi: 10.1136/thoraxjnl-2012-202432. [DOI] [PubMed] [Google Scholar]
- 164.Chung K. F. Inflammatory mediators in chronic obstructive pulmonary disease. Current Drug Targets-Inflammation & Allergy . 2005;4(6):619–625. doi: 10.2174/156801005774912806. [DOI] [PubMed] [Google Scholar]
- 165.Kerstjens H. A., Bjermer L., Eriksson L., Dahlström K., Vestbo J. Tolerability and efficacy of inhaled AZD4818, a CCR1 antagonist, in moderate to severe COPD patients. Respiratory Medicine . 2010;104(9):1297–1303. doi: 10.1016/j.rmed.2010.04.010. [DOI] [PubMed] [Google Scholar]
- 166.Kirsten A. M., Förster K., Radeczky E., et al. The safety and tolerability of oral AZD5069, a selective CXCR2 antagonist, in patients with moderate-to-severe COPD. Pulmonary Pharmacology & Therapeutics . 2015;31:36–41. doi: 10.1016/j.pupt.2015.02.001. [DOI] [PubMed] [Google Scholar]
- 167.Rennard S. I., Dale D. C., Donohue J. F., et al. CXCR2 antagonist MK-7123. A phase 2 proof-of-concept trial for chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine . 2015;191(9):1001–1011. doi: 10.1164/rccm.201405-0992OC. [DOI] [PubMed] [Google Scholar]
- 168.Marian E., Baraldo S., Visentin A., et al. Up-regulated membrane and nuclear leukotriene B4 receptors in COPD. Chest . 2006;129(6):1523–1530. doi: 10.1378/chest.129.6.1523. [DOI] [PubMed] [Google Scholar]
- 169.Woodruff P. G., Albert R. K., Bailey W. C., et al. Randomized trial of zileuton for treatment of COPD exacerbations requiring hospitalization. COPD: Journal of Chronic Obstructive Pulmonary Disease . 2011;8(1):21–29. doi: 10.3109/15412555.2010.540273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Vedel-Krogh S., Nielsen S. F., Lange P., Vestbo J., Nordestgaard B. G. Blood eosinophils and exacerbations in chronic obstructive pulmonary disease. The Copenhagen General Population Study. American Journal of Respiratory and Critical Care Medicine . 2016;193(9):965–974. doi: 10.1164/rccm.201509-1869OC. [DOI] [PubMed] [Google Scholar]
- 171.Rogliani P., Calzetta L., Ora J., Matera M. G. Canakinumab for the treatment of chronic obstructive pulmonary disease. Pulmonary Pharmacology & Therapeutics . 2015;31:15–27. doi: 10.1016/j.pupt.2015.01.005. [DOI] [PubMed] [Google Scholar]
- 172.Grubek-Jaworska H., Paplińska M., Hermanowicz-Salamon J., et al. IL-6 and IL-13 in induced sputum of COPD and asthma patients: correlation with respiratory tests. Respiration . 2012;84(2):101–107. doi: 10.1159/000334900. [DOI] [PubMed] [Google Scholar]
- 173.Tanabe T., Kanoh S., Tsushima K., Yamazaki Y., Kubo K., Rubin B. K. Clarithromycin inhibits interleukin-13-induced goblet cell hyperplasia in human airway cells. American Journal of Respiratory Cell and Molecular Biology . 2011;45(5):1075–1083. doi: 10.1165/rcmb.2010-0327OC. [DOI] [PubMed] [Google Scholar]
- 174.Lederer D. J., Bartels M. N., Schluger N. W., et al. Sildenafil for chronic obstructive pulmonary disease: a randomized crossover trial. COPD: Journal of Chronic Obstructive Pulmonary Disease . 2012;9(3):268–275. doi: 10.3109/15412555.2011.651180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Phillips J. E. Inhaled phosphodiesterase 4 (PDE4) inhibitors for inflammatory respiratory diseases. Frontiers in Pharmacology . 2020;11 doi: 10.3389/fphar.2020.00259.259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Chan J. F.-W., Yuan S., Kok K.-H., et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. The Lancet . 2020;395(10223):514–523. doi: 10.1016/S0140-6736(20)30154-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Rennard S. I., Calverley P. M. A., Goehring U. M., Bredenbröker D., Martinez F. J. Reduction of exacerbations by the PDE4 inhibitor roflumilast—the importance of defining different subsets of patients with COPD. Respiratory Research . 2011;12(1):1–10. doi: 10.1186/1465-9921-12-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Rennard S. I., Sun S. X., Tourkodimitris S., et al. Roflumilast and dyspnea in patients with moderate to very severe chronic obstructive pulmonary disease: a pooled analysis of four clinical trials. International Journal of Chronic Obstructive Pulmonary Disease . 2014;9:657–673. doi: 10.2147/COPD.S55738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Watz H., Mistry S. J., Lazaar A. L. Safety and tolerability of the inhaled phosphodiesterase 4 inhibitor GSK256066 in moderate COPD. Pulmonary Pharmacology & Therapeutics . 2013;26(5):588–595. doi: 10.1016/j.pupt.2013.05.004. [DOI] [PubMed] [Google Scholar]
- 180.Moretto N., Caruso P., Bosco R., et al. CHF6001 I: a novel highly potent and selective phosphodiesterase 4 inhibitor with robust anti-inflammatory activity and suitable for topical pulmonary administration. Journal of Pharmacology and Experimental Therapeutics . 2015;352(3):559–567. doi: 10.1124/jpet.114.220541. [DOI] [PubMed] [Google Scholar]
- 181.Goudie A. R., Lipworth B. J., Hopkinson P. J., Wei L., Struthers A. D. Tadalafil in patients with chronic obstructive pulmonary disease: a randomised, double-blind, parallel-group, placebo-controlled trial. The Lancet Respiratory Medicine . 2014;2(4):293–300. doi: 10.1016/S2213-2600(14)70013-X. [DOI] [PubMed] [Google Scholar]
- 182.Abbott-Banner K. H., Page C. P. Dual PDE 3/4 and PDE 4 inhibitors: novel treatments for COPD and other inflammatory airway diseases. Basic & Clinical Pharmacology & Toxicology . 2014;114(5):365–376. doi: 10.1111/bcpt.12209. [DOI] [PubMed] [Google Scholar]
- 183.Atto B., Eapen M. S., Sharma P., et al. New therapeutic targets for the prevention of infectious acute exacerbations of COPD: role of epithelial adhesion molecules and inflammatory pathways. Clinical Science . 2019;133(14):1663–1703. doi: 10.1042/CS20181009. [DOI] [PubMed] [Google Scholar]
- 184.Woodside D. G., Vanderslice P. Cell adhesion antagonists. BioDrugs . 2008;22(2):85–100. doi: 10.2165/00063030-200822020-00002. [DOI] [PubMed] [Google Scholar]
- 185.Bedard P. W., Kaila N. Selectin inhibitors: a patent review. Expert Opinion on Therapeutic Patents . 2010;20(6):781–793. doi: 10.1517/13543771003767468. [DOI] [PubMed] [Google Scholar]
- 186.Woodruff P. G., Wolff M., Hohlfeld J. M., et al. Safety and efficacy of an inhaled epidermal growth factor receptor inhibitor (BIBW 2948 BS) in chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine . 2010;181(5):438–445. doi: 10.1164/rccm.200909-1415OC. [DOI] [PubMed] [Google Scholar]
- 187.Valerio G., Bracciale P., D’Agostino A. G. Effect of bosentan upon pulmonary hypertension in chronic obstructive pulmonary disease. Therapeutic Advances in Respiratory Disease . 2009;3(1):15–21. doi: 10.1177/1753465808103499. [DOI] [PubMed] [Google Scholar]
- 188.Stolz D., Rasch H., Linka A., et al. A randomised, controlled trial of bosentan in severe COPD. European Respiratory Journal . 2008;32(3):619–628. doi: 10.1183/09031936.00011308. [DOI] [PubMed] [Google Scholar]
- 189.Kobayashi Y., Wada H., Rossios C., et al. A novel macrolide/fluoroketolide, solithromycin (CEM-101), reverses corticosteroid insensitivity via phosphoinositide 3-kinase pathway inhibition. British Journal of Pharmacology . 2013;169(5):1024–1034. doi: 10.1111/bph.12187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Rinne S. T., Liu C.-F., Feemster L. C., et al. Thiazolidinediones are associated with a reduced risk of COPD exacerbations. International Journal of Chronic Obstructive Pulmonary Disease . 2015;10:1591–1597. doi: 10.2147/COPD.S82643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Lakshmi S. P., Reddy A. T., Zhang Y., et al. Down-regulated peroxisome proliferator-activated receptor γ (PPARγ) in lung epithelial cells promotes a PPARγ agonist-reversible proinflammatory phenotype in chronic obstructive pulmonary disease (COPD) Journal of Biological Chemistry . 2014;289(10):6383–6393. doi: 10.1074/jbc.M113.536805. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 192.Tontonoz P., Spiegelman B. M. Fat and beyond: the diverse biology of PPARγ. Annual Review of Biochemistry . 2008;77(1):289–312. doi: 10.1146/annurev.biochem.77.061307.091829. [DOI] [PubMed] [Google Scholar]
- 193.Boo H.-J., Park S. J., Noh M., Min H.-Y., Jeong L. S., Lee H.-Y. LJ-529, a partial peroxisome proliferator-activated receptor gamma (PPARγ) agonist and adenosine A3 receptor agonist, ameliorates elastase-induced pulmonary emphysema in mice. Archives of Pharmacal Research . 2020;43(5):540–552. doi: 10.1007/s12272-020-01235-0. [DOI] [PubMed] [Google Scholar]
- 194.Lea S., Plumb J., Metcalfe H., et al. The effect of peroxisome proliferator-activated receptor-γ ligands on in vitro and in vivo models of COPD. European Respiratory Journal . 2014;43(2):409–420. doi: 10.1183/09031936.00187812. [DOI] [PubMed] [Google Scholar]
- 195.Shan M., You R., Yuan X., et al. Agonistic induction of PPARγ reverses cigarette smoke-induced emphysema. The Journal of Clinical Investigation . 2014;124(3):1371–1381. doi: 10.1172/JCI70587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Agusti A. G. N. COPD, a multicomponent disease: implications for management. Respiratory Medicine . 2005;99(6):670–682. doi: 10.1016/j.rmed.2004.11.006. [DOI] [PubMed] [Google Scholar]
- 197.Barnes P. J. Reduced histone deacetylase in COPD: clinical implications. Chest . 2006;129(1):151–155. doi: 10.1378/chest.129.1.151. [DOI] [PubMed] [Google Scholar]
- 198.Ito K., Barnes P. J. COPD as a disease of accelerated lung aging. Chest . 2009;135(1):173–180. doi: 10.1378/chest.08-1419. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support this study's findings are available in this article.


