Summary
Background
Local fistula injection of mesenchymal stromal/stem cells (MSC) is effective for complex perianal Crohn's fistulas but is also expensive and requires specialised facilities for cell revival before administration. Human amnion epithelial cells (hAEC) are non-MSC cells with therapeutic properties. The primary aim of this study was safety of hAEC therapy. Secondary aims included hAEC efficacy, feasibility of the protocol and impact on quality of life.
Methods
A phase I open label study of ten adults with active complex Crohn's perianal fistulas refractory to conventional treatment, including anti-tumour necrosis factor alpha therapy, was undertaken. A single dose of hAEC was injected into the fistula tract(s) after surgical closure of the internal opening(s). Study outcomes were assessed at week 24 with follow up for at least 52 weeks.
Findings
Local injection of hAEC was safe, well tolerated and the injection procedure was feasible. Complete response occurred in 4 patients, and a partial response in an additional 4 patients. There was a mean reduction in the Perianal Disease Activity Index of 6.5 points (95% CI −9.0 to −4.0, p = 0.0002, paired t-test), modified Van Assche MRI Index of 2.3 points (95% CI −3.9 to −0.6, p = 0.012, paired t-test) and a mean improvement of 15.8 points (95% CI 4.9 to 26.8, p = 0.010, paired t-test) in quality of life using the Short IBD-Questionnaire in complete responders.
Interpretation
Local injection of hAEC therapy for refractory complex perianal fistulising Crohn's disease appears safe, well-tolerated, feasible and demonstrated improvement. Quality of life is improved in those who achieve complete fistula healing.
Funding
This study was funded by competitive research grant funding from the Gastroenterological Society of Australia Seed Grant 2018.
Keywords: Crohn's disease, Perianal fistula, Stem cells, Human amnion epithelial cells
Research in context.
Evidence before this study
We undertook a comprehensive literature review of cell therapy for perianal fistulising Crohn's disease including all systematic reviews and individual clinical trials published with a listing on the PubMed database. Search terms included “perianal fistula”, “Crohn's disease”, “stem cells”, “mesenchymal stem cells” and “mesenchymal stromal cells”. We identified hundreds of patients all treated with mesenchymal stromal/stem cells mostly derived from adipose tissue but also bone marrow and umbilical cord which showed efficacy in various stages of clinical trials from phase I studies to phase III, including some longer-term follow up studies. Recent results from meta-analysis by Wang et al. (Stem Cell Research & Therapy, 2023) shows that mesenchymal stromal/stem cells were effective in the short- and long-term for complex perianal Crohn's fistulas and superior to standard therapy (42.35% vs. 18.81% and 55.96% vs. 42.86%, respectively) although the number of randomised controlled studies remains small, the definitions of fistula healing varies in the individual studies as does the dosage of cells. Darvadstrocel is the only commercially available adipose-derived mesenchymal stromal cell. However, it is not available in countries outside of Europe and Japan, requires highly specialised facilities to further prepare the stem cells before use and is extremely expensive.
Added value of this study
This is a human study using human amnion epithelial cells to treat perianal fistulising Crohn's disease. These are non-embryonic, non-mesenchymal stem-like cells derived from the amniotic membrane of the human placenta, which is generally discarded after birth. This early phase study shows that amnion epithelial cells are safe and the treatment is feasible and well tolerated. We reported fistula improvement in a group of patients who were refractory to anti-tumour necrosis factor alpha drugs and those who achieved complete fistula healing reported an improvement in quality of life. For those who responded to a single initial dose of amnion epithelial cells, the improvement persisted at 52 weeks.
Implications of all the available evidence
Human amnion epithelial cells are a more attractive cell therapy compared with mesenchymal stromal/stem cells as cell expansion (performed over weeks) is not required, there is a far greater yield with a single placental isolation and further preparation prior to clinical use is not required—all which significantly reduce manufacturing costs and improve accessibility. Placebo-controlled phase II clinical trials should be undertaken using human amnion epithelial cells in perianal Crohn's fistulas, as it appears safe and may be potentially beneficial in this condition and its therapeutic efficacy needs to be determined.
Introduction
Complex perianal fistulas affect nearly one third of patients with Crohn's disease1 and are particularly difficult to treat. Fistula are associated with negative impacts on quality of life, mental health, and health care utilisation.2 Fistula closure is reported in only 30% of patients3 despite best medical therapy using long-term anti-tumour necrosis factor alpha (anti-TNF) biologic drugs.4 Patients with perianal fistulas in Crohn's disease are also at increased risk of major abdominal surgery, rectal and anal cancer.5 Local fistula injection of (mainly adipose-derived) mesenchymal stromal/stem cells (MSC) has become a recent effective treatment6,7 with a favourable safety profile and persistent long-term results.8 Although the precise mechanisms of perianal fistula pathogenesis and MSC mechanism of action remain unknown,9 the therapeutic effect is likely immunoregulatory, potentially with additional anti-fibrotic and regenerative properties.
The only commercially available allogeneic expanded adipose-derived MSC product, Darvadstrocel (TiGenix, Takeda), is approved by the European Medicines Agency (EMA) for perianal Crohn's fistulas but has significant limitations. These include cost (£54,000 per product alone as per the listing price on the National Institute for Health and Care Excellence, NICE, UK10), MSC expansion to achieve a sufficient yield, and pre-administration revival in specialised facilities. These factors prevent its use in many countries and result in experimental use of impure adipose-derived MSC products including micro-fragmented adipose tissue or stromal vascular fraction (SVF) containing the MSC progenitors which can be autologously harvested during surgery. This requires an additional invasive surgical procedure, produces impure MSC at variable (and unmeasured) quantities and creates significant challenges for regulation. Bone marrow-derived MSC, the main alternative MSC source, are limited by lower isolation yields and donor availability as the MSC quality is reduced by increasing donor age.11
Human amnion epithelial cells (hAEC) are non-MSC, non-embryonic stem-like cells isolated from the amniotic epithelial membrane of the placenta. hAEC display immunomodulatory therapeutic properties and express anti-inflammatory proteins such as IL-10,12 inhibit chemotaxis and reduce lymphocyte proliferation.13 hAEC also have anti-fibrotic properties through down-regulation of transforming growth factor beta (TGF-β) which reduces myofibroblast activation and extracellular matrix deposition.14,15 Similar to MSC, hAEC also demonstrate low immunogenicity with weak expression of major histocompatibility complex (MHC) class I antigens, negligible MHC class II expression16 and do not elicit any immune response in immunocompetent humans17, 18, 19 and thus do not require prior conditioning chemotherapy. hAEC have been studied in several phase I human studies at our institutions including bronchopulmonary dysplasia,20 cirrhosis21 and acute ischaemic stroke22 without any reported safety concerns.
We have investigated the use of hAEC in pre-clinical murine IBD models and demonstrated that hAEC attenuated acute and chronic colitis and intestinal fibrosis.15,23 In the absence of pre-clinical models for perianal fistulising Crohn's disease, we have undertaken a human phase I study using local fistula injection of hAEC for this condition.
Methods
Ethics
Ethics approval was granted by the Monash Health Human Research Ethics Committee (HREC) to perform this study (HREC/18/MonH/342, 18-0000-225A) under the requirements of the National Health and Medical Research Council (NHMRC) National Statement on Ethical Conduct in Human Research. The investigational product was approved by the Therapeutic Goods Administration (TGA) Clinical Trial Notification scheme. The trial was prospectively registered with the Australian New Zealand Clinical Trials Registry (ANZCTR) prior to commencement (ACTRN 12618001883202) and overseen by an independent Data Safety Monitoring Board (DSMB). Written informed consent was obtained from all participants prior to enrollment in the study.
Study design and participants
We conducted a single centre phase I open label study including ten adult patients with active complex perianal Crohn's fistulas refractory to conventional medical therapy including antibiotics, immunomodulators and/or biologic therapies. Participants were recruited from December 2019 to February 2021 after providing written informed consent. Participants were not included or excluded based on gender or sex assigned at birth, which were determined by self-reporting by the participant. As safety was the primary outcome for this phase I study, a sample population of 10 participants was chosen without randomisation.
Baseline evaluation included clinical assessment, routine blood tests, colonoscopy to assess Crohn's luminal disease activity and MRI pelvis. A preparatory examination under anaesthesia (EUA) was performed 2–4 weeks before hAEC injection to allow for characterisation of the fistula tract(s), debridement and seton placement to adequately drain any collections. Follow up visits included clinical assessments and routine blood tests with the study investigators consisting of a single IBD clinician and colorectal surgeon up until week 104. The study outline is shown in Fig. 1.
Fig. 1.
Phase I study outline.
A perianal fistula was defined as an abnormal communication arising from the intestinal epithelial surface to the epithelial surface in the perianal or perineal skin. The inclusion criteria were as follows: age ≥18 years, complex perianal Crohn's fistula defined by the American Gastroenterological Association (AGA)24 (requiring at least one of the following: high intersphincteric or trans-sphincteric fistulas, supra-sphincteric fistulas, two or more external fistula openings or any associated collections), inactive or mildly active luminal rectal inflammation (on endoscopic assessment based on the absence of ulcers), and active fistulas with a Perianal Disease Activity Index (PDAI) score greater than four.25,26 All participants were required to demonstrate active disease despite conventional treatment. Participants were defined as refractory if they had failed to close the external opening of the perianal fistula(s) without abscess formation despite any of the following: antibiotics for ≥1 month, immunomodulators or anti-TNF induction or maintenance therapy for ≥3 months. Doses of current Crohn's medications were required to be unchanged for 3 months prior to study enrollment until outcomes were assessed at week 24.
Exclusion criteria included: rectal or anal stenosis, moderate or severe proctitis, undrainable abscesses or collections ≥2 cm in diameter, current pregnancy, refusal of contraceptive methods and inability to comply with trial requirements. Prohibited medications during the 24-week trial period included rectal enemas, continued antibiotics for greater than four weeks after hAEC injection and any changes to maintenance immunomodulatory or biologic therapies.
Study outcomes
As this was a phase I study, the primary outcome at week 24 was safety of local hAEC therapy with the causality assessment and severity of adverse events defined using the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE) v4.0. Secondary outcomes at week 24 included: 1) efficacy of hAEC treatment, 2) feasibility of the route of administration and 3) health-related quality of life. Efficacy of local hAEC injection was assessed via history, examination and MRI pelvis. Complete response required the absence of fistula drainage with closure of treated external openings and absence of collection (>2 cm diameter) on MRI. Partial response was defined by closure of ≥50% of all treated external openings or ≥50% reduction of fistula diameter on MRI (if a single fistula), also in the absence of an abscess on MRI. All MRI scans were undertaken using the same Philips Ingenia 3T machine and read by an experienced, non-blinded single radiologist specialising in abdominal MRI for IBD. Validated MRI-based fistula activity assessment scores included the modified van Assche Index (mVAI)27 and the MAGNIFI-CD28 which were evaluated at baseline, week 24 and 52. Feasibility of route of administration was assessed using a purpose-designed questionnaire (Supplementary Information 1) completed by the surgical proceduralist performing hAEC injections for each patient. The short IBD-Questionnaire (sIBDQ) was utilised to assess health-related quality of life changes over the 24-week period. Further follow up was undertaken in these patients up to week 52 from hAEC injection, with week 104 visits nearly completed.
Human amnion epithelial cell isolation
hAEC were isolated from placentas of healthy mothers with normal at-term singleton pregnancies undergoing elective Caesarean section under Good Manufacturing Practice-like conditions in the BioSpherix Unit housed at the Monash Health Translation Precinct. All mothers donating their placentas provided written informed consent. As per the published protocol for hAEC isolation for clinical use,29 hAEC were digested using animal-product free recombinant enzyme solution and resuspended in animal-product free culture medium. Cell purity was ensured using flow cytometry to confirm EpCam + cells with negative MSC markers (CD90, CD105). After screening for adventitious agents (Supplementary Information 2) and ensuring hAEC fulfilled non-disclosed potency criteria, released cells were cryopreserved in locked liquid nitrogen dewars and thawed immediately prior to patient administration in pre-filled sterile syringes using 2% human serum albumin with saline as a diluent to a concentration of 40 million cells per 4.5 ml. Each participant received cells from a single donor without pooling of hAEC.
Local fistula injection of amnion epithelial cells
All EUA for the study were performed by a single experienced colorectal surgeon to prevent variability in surgical technique. A single dose of hAEC was administered at the intervention EUA where immediately prior to hAEC injection, there was final confirmation of the absence of collections >2 cm, removal of all setons (if still present), and curettage to de-epithelialise fistula tract(s). The internal fistula opening was closed with synthetic absorbable sutures and the external opening debrided. Each fistula tract received 40 million hAEC, with half injected around the internal fistula opening and the remaining half injected via submucosal blebs along the tract as described in a published protocol for local adipose-derived MSC therapy.30 Local anaesthesia is routinely administered for all EUA performed at our institution via a pudendal nerve block, however this was avoided during the intervention EUA due to potential direct cell cytotoxicity31 although direct contact of hAEC with anaesthetic agents was unlikely. Any existing biologic therapy was not interrupted after hAEC administration and continued at scheduled intervals.
Statistical analysis
Descriptive statistical analysis was performed to assess frequencies of events. After inspection of the data distribution and confirming normality using the Shapiro-Wilke's test, paired t-tests were used to compare differences in baseline and week 24 scores for PDAI, MRI fistula activity scoring algorithms and the sIBDQ assessments. Statistical analysis software included licensed SPSS software (IBM Corp. Released 2020. IBM SPSS Statistics for Windows, Version 27.0. Armonk, NY: IBM Corp) and figures were prepared using licensed GraphPad Prism software (GraphPad Software for Windows, Version 9.0.0, San Diego, California).
Role of the funders
This study was funded by competitive research grant funding from the Gastroenterological Society of Australia Seed Grant 2018. The funding organisation had no role in any of the study design, collection, analysis or interpretation of the data, writing of the report or decision to submit the paper for publication.
Results
Baseline characteristics
Ten consenting patients who fulfilled exclusion and inclusion criteria were recruited for this phase I study. Most were female (7 patients) with a median age of 37 years (IQR 29–44 years). The median duration of Crohn's disease was 10 years (IQR 4–15 years). Of the ten patients, five had a single complex perianal fistula and five had two complex fistulas with a total of 15 fistulas treated with hAEC during the study. All patients were refractory to anti-TNF biologic therapy (infliximab or adalimumab) and the majority (7 patients) were concurrently on an immunomodulator (azathioprine or 6-mercaptopurine). Nearly all (9 patients) either had therapeutic anti-TNF serum drug levels or were already on an escalated biologic drug dosing regimen at baseline already. Before enrollment in the study, the majority (8 patients) were managed with a long-term seton in situ (for ≥12 months) and 7 patients had previous unsuccessful surgical attempts at internal fistula opening closure. Table 1 depicts the baseline characteristics of the study participants.
Table 1.
Baseline characteristics.
| Variable | n = 10 |
|---|---|
| Age (years) | 37 (29.75–43.75) |
| Sex assigned at birth | 7 female 3 male |
| Montreal classification—age at diagnosis (years) | 1 A1, 8 A2, 1 A3 |
| Montreal classification—location | 1 ileal (L1), 6 colonic (L2), 2 ileocolonic (L3), 1 isolated perianal disease |
| Montreal classification—behaviour | 8 non-stricturing, non-penetrating (B1), 1 penetrating (B3) |
| Crohn's Disease Activity Index (CDAI) at screening | 133 (37–156) |
| Harvey-Bradshaw Index (HBI) at screening | 1 (0–5) |
| Simple Endoscopic Score for Crohn's Disease (SES-CD) at screening | 1 (0–4) |
| Smoking | 3 ex-smokers (nil current) |
| Duration of Crohn's disease (years) | 10 (4.00–15.25) |
| Duration of Crohn's perianal fistula (years) | 7.90 (3.50–9.75) |
| No. of fistula | 5 participants with single fistula, 5 participants with two fistulas |
| Park's classification | 2 intersphincteric, 8 transphincteric |
| PDAI at screening | 9 (6.50–10.25) |
| mVAI at screening (mean, standard deviation) | 9, 3.3 |
| MAGNIFI-CD at screening (mean, standard deviation) | 18.3, 6.8 |
| Previous surgical fistula closure attempts | 5 internal opening closure, 1 mucosal advancement flap, 1 temporary diverting stoma |
| Current biologic therapy | 6 infliximab, 3 adalimumab, 1 ustekinumab |
| Median duration of biologic therapy (months) | 35.00 (27.00–95.50) |
| Concurrent immunomodulator therapy | 6 |
| Long-term seton in situ | 8 |
Categorical variables presented as number and non-parametric and parametric continuous variables expressed as median (interquartile range) and mean (standard deviation), respectively.
Adverse events
The most common related adverse event (AE) to hAEC administration was post-operative anal pain in 7 patients which required oral analgesia after discharge from day surgery. Anal pain completely resolved within a median of 6.5 days (IQR 0.0–10.0) with a maximal pain severity score of a median 7 out of 10 (IQR 6–8 out of 10) on a 10-point pain scale within 48 h of surgery. This was due to surgical closure of the internal fistula opening in the absence of local anaesthesia rather than a direct effect of the hAEC. There were no suspected unexpected serious adverse reactions (SUSAR) reported in the 24 weeks following hAEC therapy. All recorded adverse events and causality assessments are shown in Table 2.
Table 2.
Adverse events within 24 weeks after hAEC injection.
| Causality | CTCAE term | Severity (Grade) | Further details |
|---|---|---|---|
| Probable | |||
| Anal pain | 1 and 2 | ||
| Possible | |||
| Blood bilirubin increased | 1 | Likely pre-existing Gilbert's syndrome | |
| Constipation | 1 | Post-operative likely due to opioid analgesia | |
| Unlikely or unrelated | |||
| Anorectal infection | 2 | Temporally unrelated to hAEC injectiona | |
| Musculoskeletal—other | 1 and 2 | Traumatic wrist sprain Ganglion cyst |
|
| Metabolism and nutrition disorders—other | 2 | Iron deficiency Vitamin D deficiency |
|
| Rash acneiform | 1 | ||
| Hearing impairment | 1 | ||
| Tooth infection | 1 | ||
| Cough | 2 | ||
| Diarrhoea | 1 | Antibiotic adverse event | |
| Increased alanine transaminase | 1 | ||
| Otitis externa | 2 | ||
| Palpitations | 1 | ||
| Neck pain | 1 | ||
| Enterocolitis infectious | 2 | ||
| Soft tissue infection | 1 | ||
| White blood cell decrease | 1 | ||
| Neutrophil count decrease | 1 | ||
| Arthralgia | 1 | ||
| Urticaria | 1 | ||
| Erythema multiforme | 1 | Pseudo-vesicular rash | |
| Myalgia | 1 |
Adverse events terms, severity and causality assessment as defined according to the CTCAE v4.03.
Anorectal infections were assessed as temporally unrelated to hAEC as one occurred after screening but prior to hAEC injection, the other recurred at week 23 in an anatomically separate pre-existing blind sinus that was not treated with hAEC. These events are described in more detail in the Results section of the manuscript.
Of note, one participant (006) accidently manually self-removed a critical suture at day 12 after hAEC injection, resulting in re-opening of the internal fistula opening. The suture was replaced to re-close the internal fistula opening but this may have reduced fistula healing.
Two participants reported perianal abscesses during the study period, however neither were temporally nor anatomically related to the hAEC injection. Of these, one participant developed an abscess after baseline EUA but prior to hAEC injection in the context of Covid-19 related theatre scheduling delays. Participant 006 also re-accumulated a rectovaginal abscess at week 23, due to enlargement of a collection arising from a pre-existing anterior blind sinus that was anatomically separate from the treated posterior perianal fistulas (that improved and closed) and did not receive hAEC injection.
Fistula healing
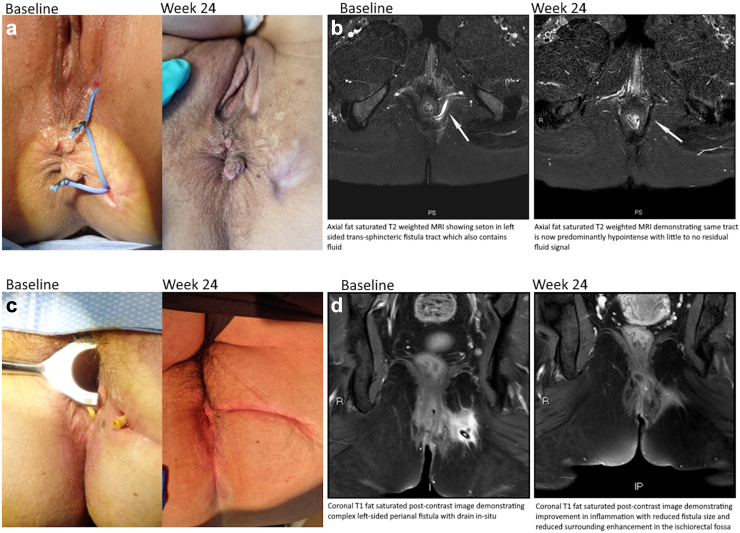
At week 24, 8 fistulas out of the total 15 (53%) belonging to the ten patients, achieved closure of the external opening without any abscess. Complete response was achieved in 4 patients and a partial response occurred in a further 4 patients. Respective baseline and week 24 clinical and MRI images are shown in Fig. 2 of two patients who completely responded.
Fig. 2.
Baseline and week 24 clinical image with corresponding MRI images in selected complete responders. a Participant clinical image. b Participant MRI pelvis. c Participant clinical image. d Participant MRI pelvis.
Both patients who did not fit the study definition of treatment response at week 24 still demonstrated improvement. One participant reported a reduced PDAI score from 7 to 2 with reduced fistula enhancement on MRI compared with baseline, but pin-point non-draining external fistula openings persisted. The other demonstrated a reduction in PDAI score from 8 to 4 with stable fistula activity on MRI.
PDAI score
Overall, there was a mean reduction in week 24 PDAI scores compared with baseline of 6.5 points (95% CI −9.0 to −4.0, p = 0.0002, paired t-test) as shown in Fig. 3. All participants at week 24 reported a PDAI score of ≤4 (accepted as inactive) and 8 remained seton free at that timepoint (setons were re-inserted in two participants at week 23).
Fig. 3.
a Baseline and week 24 PDAI scores. b Baseline and week 52 PDAI scores.
MRI-based fistula activity scores
There was a mean reduction in mVAI and MAGNIFI-CD scores at week 24 by a mean of 2.3 points (95% CI −3.9 to −0.6, p = 0.012, paired t-test) and 1.9 points (95% CI −3.7 to −0.1, p = 0.043, paired t-test), respectively. These scores are shown in Fig. 4.
Fig. 4.
a Baseline and week 24 mVAI score. b Baseline and week 52 mVAI score. c Baseline and week 24 MAGNIFI-CD score. d Baseline and week 52 MAGNIFI-CD score.
Quality of life
There was a mean difference of 6.0 points (95% CI −1.7 to 13.7, p = 0.110, paired t-test) in the sIBDQ scores at baseline and week 24 overall and 7 of the participants reported an improvement in sIBDQ scores. Participants who were complete responders (4 patients) reported a higher improvement in sIBDQ scores than the 6 patients who were non-complete responders by a mean difference of 15.8 points (95% CI 4.9 to 26.8, p = 0.01, paired t-test). Fig. 5 shows baseline and week 24 sIBDQ scores.
Fig. 5.
Baseline and week 24 sIBDQ scores.
Feasibility of study protocol
The thawing and release of the hAEC on the day of surgery did not result in any theatre delays. Minor unanticipated scheduling difficulties were experienced during this study period due to the Covid-19 pandemic resulting in cancellations of elective surgery causing delays from screening to EUA.
The hAEC product delivery and intra-operative preparation were assessed as highly acceptable. High cell viability was noted in the residual syringe after hAEC injection. In two participants, minor improvements were suggested in terms of increasing injectable product volume to accommodate for variable fistula morphology.
The surgical procedure was well tolerated, and the internal fistula opening was easily identifiable in most participants. Injection of the hAEC into the internal opening was assessed with either “none” or only “mild difficulty”, however injection along the fistula tract(s) was more challenging with half of the participants assessed as “moderately” or “severely difficult”.
Avoidance of iodine-based surgical preparations and a pudendal nerve block in all participants did not cause any complications.
Long term outcomes
There were no SUSAR reported at week 52 after hAEC injection. All fistulas with closed external openings (n = 8) remained closed without any abscess at week 52. All complete responders remained in clinical remission with closure of the external fistula opening, absence of significant symptoms and absence of abscess on serial MRI pelvis, with data currently available up to week 104.
The persistence of response assessed by the PDAI score was maintained at week 52 with a mean reduction of 6.1 points (95% CI −8.8 to −3.4, p = 0.0007, paired t-test) compared with baseline. The MRI fistula activity indices also maintained their improvement at week 52 compared with baseline with the mVAI demonstrating a mean reduction of 2.5 points (95% CI −4.5 to −0.4, p = 0.023, paired t-test) and the MAGNIFI-CD demonstrating a mean reduction of 2.9 points (95% CI −5.4 to −0.4, p = 0.027, paired t-test). These results are shown in Figs. 3 and 4.
Discussion
In this phase I human study including ten adult patients who underwent local fistula injection of hAEC to treat complex, treatment-refractory perianal Crohn's fistulas, we showed that hAEC are well-tolerated and safe up to 52 weeks using doses of up to 80 million cells per patient. After a single dose of 40 million hAEC per fistula, complete response occurred in four patients with partial response in another four patients which was maintained at week 52. Improvement still occurred in the two participants that did not fulfil response criteria. Quality of life also significantly improved in those who achieved complete response. Our administration protocol appears feasible and well tolerated without major technical issues identified.
The short and long-term safety profile of hAEC is similar when compared with other published human studies using local injection of MSC where the main related adverse event of anal pain has been reported at rates of 13%–64%.6,8,32, 33, 34 At our institution, as the pudendal nerve block is routinely administered at EUA and together with the closure of the internal fistula opening performed, this resulted in 7 of participants reporting increased anal pain greater than 30% baseline compared with previous EUA procedures. This adverse event was mild, temporary and only required oral analgesia.
The main purpose of this phase I study was to assess safety, and subsequently it was not powered to assess efficacy. Further limitations for efficacy assessment include the study design as a before-and-after study with the absence of a placebo-control group, the potential for outcomes to be influenced by temporal change and regression to the mean. However, the main intention of this study was safety. There were 8 patients who demonstrated a response to treatment of which 4 showed a complete response. This is comparable to a meta-analysis reporting an overall fistula healing rate of 57%,7 however there are variations in definitions of fistula healing and response amongst studies. Furthermore, compared with previous early phase locally injected MSC studies that excluded patients on anti-TNF drugs,32,34, 35, 36, 37 our patient cohort appears more treatment refractory as all had inadequate responses to high doses of anti-TNF drugs and a further 7 had previous unsuccessful surgical closure attempts. A prominent finding from the phase III study performed by Panes et al.6 was the importance of surgical technique and attempts at fistula closure with high rates of fistula response with placebo. While we could not standardise to the previous quality of fistula curettage undertaken, the majority of our participants were recruited from tertiary IBD centres with multidisciplinary IBD and expert colorectal surgical experience with complex perianal fistulas. We limited the variability of the current surgical procedure by using a single experienced colorectal surgeon who performed all EUA for all ten participants.
Like other human studies using local injection of MSC therapy, we experienced challenges with the heterogenous morphology of perianal fistulas in Crohn's disease including the presence of cavities, number of internal openings, length and branching and diameter of the tracts. These factors can affect the ease of local stem cell injection as demonstrated by our feasibility study where five participants were rated as “moderately” or “severely difficult” to inject along the fistula tract, the majority of these accompanied by cavities measuring 6–50 mm diameter. In all the reported studies using local MSC injection, it was extremely difficult to standardise the dosage of cells accounting for fistula morphology and only one study based MSC dosage on fistula diameter but only arbitrarily used two different doses.34 The dosage of hAEC administered to our patients at 40 million cells/fistula is difficult to compare with the only phase III randomised placebo-controlled study using allogeneic adipose-derived MSC (Cx601)6 as 120 million cells were used per patient rather than per fistula. Some studies also included repeated administration of cells at various intervals,32,35,36,38,39 or additionally administered glues or plugs32,33,37,38,40, 41, 42 which was not part of this study. As immunomodulation is probably the primary mechanism of action, serial dosing may achieve additional healing rates, although a significant proportion can still achieve remission after a single dose. Further studies are required to determine the optimal interval and number of doses and when repeated treatment is unlikely to achieve fistula healing.
There are very few studies using hAEC in IBD models and none in humans. Our pre-clinical studies found that hAEC-treated mice with chronic dextran sulfate sodium (DSS) colitis demonstrated clinical improvement (based on improved body weight, less colonic shortening and less splenomegaly) and reduced histological inflammation and reduced pro-inflammatory cytokines IL-1β, TNF-α, IL-6 and IFN-γ while promoting anti-inflammatory cytokines IL-10 compared with untreated mice.15 Fibrosis was reduced based on picrosirius red stain area on histology and a quantitative decrease in myofibroblasts and macrophages.15 Another pre-clinical study using licensed hAEC pre-treated with TNF-α and IFN-γ (similar to studies using MSC) identified T cell modulation as a mechanism of action, showing that licensed hAEC improved the Th17 and regulatory T cells balance by increasing regulatory T cells and decreasing other T cell types including Th1, Th2 and Th1743 which is similar to the MSC mechanism of action.44 However, hAEC display favourable advantages over MSC as a cellular therapeutic product. hAEC display greater multipotency with trilineage differentiation potential as hAEC can differentiate into cells derived from endoderm, mesoderm and ectoderm.45 Manufacturing requirements are significantly reduced with hAEC which do not require expansion and can routinely isolate 120 to 200 million highly pure cells per placenta in hours46 compared with MSC expansion over weeks to achieve the same yield. hAEC also do not require re-culture prior to injection and can be cryopreserved for years and thawed immediately prior to use with high viability as demonstrated by this study. Suitable placental donations from healthy women delivering at-term gestation pregnancies are relatively easy to obtain and cells can be pooled to reach required quantities, although in our study it was common for hAEC from a single placenta to be used to treat several patients. Collectively, this translates to a cost-effective, convenient, and accessible therapy with the cost of hAEC per fistula for this study approximately ∼$7000 (AUD) compared with >$100,000 (AUD) for Alofisel®. While MSC therapy appears overall safe in humans with a favourable risk versus benefits ratio, MSC in in vitro studies can display properties that theoretically promote cancer including engraftment, homing to tumours, promoting tumour growth when co-administered in multiple different animal cancer models and differentiation into cells resembling carcinoma-associated fibroblasts under certain conditions.47 While there are no malignancies thought to result directly from MSC in humans, there have been reports of metastatic bowel cancer48 and rectal lymphoma49 in Crohn's patients who have received local injection of MSC to treat their perianal fistulas, although other risk factors for cancer including the IBD and immunomodulator use are far greater contributors. Unlike MSC, hAEC do not express telomerase and have restricted proliferative capacity rendering them non-tumourigenic and theoretically even safer than MSC but further long term data is required.50
Conclusion
In this human study using local fistula injection of allogeneic human amnion epithelial cells to treat perianal Crohn's disease with short and long-term follow up, we have shown hAEC to be safe, well-tolerated and feasible. Although this study was not powered to assess efficacy, there was promising and long-lasting fistula healing with a single hAEC dose resulting in response in 8 patients in this small group of anti-TNF-refractory perianal fistulas. Further investigations into optimal dosing regimens, mechanisms of action and placebo-controlled phase II efficacy studies should be undertaken to develop much needed alternative therapies for this debilitating, treatment-refractory condition.
Contributors
CK designed the study, collected data, performed the analysis, and wrote the manuscript. TCN collected data and performed critical revisions of the manuscript. AG collected data and performed critical revisions of the manuscript. RL designed and supervised the study and performed critical revisions of the manuscript. WS performed critical revisions of the manuscript. GM designed and supervised the study, verified the data and analysis and performed critical revisions of the manuscript. CK and GM verified the underlying data. All authors had access to the study data and reviewed and approved the final manuscript.
Data sharing statement
De-identified data may be made available upon request after consultation with a human research ethics committee as it contains human research participant data.
Declaration of interests
A provisional patent (AU2022901083) has been filed in Australia for use of human amnion epithelial cells in perianal fistulising Crohn's disease with RL, CK and GM listed as co-inventors. A company (Exosome Biosciences Pty Ltd 2023) co-owned by the Hudson Institute of Medical Research, Monash University and Monash Health has been established using derivatives of human amnion epithelial cells to treat Crohn's disease. The authors have not received any monetary payments from the company and this company had no role in any part of this study. WS, RL, CK, TCN and GM will participate in a future clinical trial affiliated with Exosome Biosciences. AG has nothing to declare.
Acknowledgements
This study was funded by competitive research grant funding from the Gastroenterological Society of Australia Seed Grant 2018. The funding organisation had no role in any of the study design, collection, analysis or interpretation of the data, writing of the report and decision to submit the paper for publication.
We acknowledge the contribution of the scientists who performed isolation of the hAEC for this study and the members of the Data Safety Monitoring Board for their assistance with the project. The interim results of this project were previously presented at Digestive Diseases Week 2022 and Australian Gastroenterology Week 2022. Use of the Short Inflammatory Bowel Disease Questionnaire, authored by Dr Jan Irvine et al., was made under license from McMaster University, Hamilton, Canada.
Footnotes
Supplementary data related to this article can be found at https://doi.org/10.1016/j.ebiom.2023.104879.
Appendix A. Supplementary data
References
- 1.Schwartz D.A., Loftus E.V., Jr., Tremaine W.J., et al. The natural history of fistulizing Crohn's disease in Olmsted County, Minnesota. Gastroenterology. 2002;122(4):875–880. doi: 10.1053/gast.2002.32362. [DOI] [PubMed] [Google Scholar]
- 2.Lonnfors S., Vermeire S., Greco M., Hommes D., Bell C., Avedano L. IBD and health-related quality of life -- discovering the true impact. J Crohns Colitis. 2014;8(10):1281–1286. doi: 10.1016/j.crohns.2014.03.005. [DOI] [PubMed] [Google Scholar]
- 3.de Groof E.J., Sahami S., Lucas C., Ponsioen C.Y., Bemelman W.A., Buskens C.J. Treatment of perianal fistula in Crohn's disease: a systematic review and meta-analysis comparing seton drainage and anti-tumour necrosis factor treatment. Colorectal Dis. 2016;18(7):667–675. doi: 10.1111/codi.13311. [DOI] [PubMed] [Google Scholar]
- 4.Torres J., Bonovas S., Doherty G., et al. ECCO guidelines on therapeutics in crohn's disease: medical treatment. J Crohns Colitis. 2020;14(1):4–22. doi: 10.1093/ecco-jcc/jjz180. [DOI] [PubMed] [Google Scholar]
- 5.Wewer M.D., Zhao M., Nordholm-Carstensen A., Weimers P., Seidelin J.B., Burisch J. The incidence and disease course of perianal crohn's disease: a Danish Nationwide Cohort Study, 1997-2015. J Crohns Colitis. 2020;15:5. doi: 10.1093/ecco-jcc/jjaa118. [DOI] [PubMed] [Google Scholar]
- 6.Panes J., Garcia-Olmo D., Van Assche G., et al. Expanded allogeneic adipose-derived mesenchymal stem cells (Cx601) for complex perianal fistulas in Crohn's disease: a phase 3 randomised, double-blind controlled trial. Lancet. 2016;388(10051):1281–1290. doi: 10.1016/S0140-6736(16)31203-X. [DOI] [PubMed] [Google Scholar]
- 7.Qiu Y., Li M.Y., Feng T., et al. Systematic review with meta-analysis: the efficacy and safety of stem cell therapy for Crohn's disease. Stem Cell Res Ther. 2017;8(1):136. doi: 10.1186/s13287-017-0570-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Panés J., García-Olmo D., van Assche G., et al. Long-term efficacy and safety of stem cell therapy (Cx601) for complex perianal fistulas in patients with Crohn's disease. Gastroenterology. 2018;154:1–9. doi: 10.1053/j.gastro.2017.12.020. [DOI] [PubMed] [Google Scholar]
- 9.Cheng F., Huang Z., Li Z. Efficacy and safety of mesenchymal stem cells in treatment of complex perianal fistulas: a meta-analysis. Stem Cells Int. 2020;2020 doi: 10.1155/2020/8816737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.NICE . National Institute for Health and Care Excellence; UK: 2019. Darvadstrocel for treating complex perianal fistulas in Crohn's disease. [Google Scholar]
- 11.Barreto-Duran E., Mejia-Cruz C.C., Leal-Garcia E., Perez-Nunez R., Rodriguez-Pardo V.M. Impact of donor characteristics on the quality of bone marrow as a source of mesenchymal stromal cells. Am J Stem Cells. 2018;7(5):114–120. [PMC free article] [PubMed] [Google Scholar]
- 12.Hao Y., Ma D.H., Hwang D.G., Kim W.S., Zhang F. Identification of antiangiogenic and antiinflammatory proteins in human amniotic membrane. Cornea. 2000;19(3):348–352. doi: 10.1097/00003226-200005000-00018. [DOI] [PubMed] [Google Scholar]
- 13.Li H., Niederkorn J.Y., Neelam S., et al. Immunosuppressive factors secreted by human amniotic epithelial cells. Invest Ophthalmol Vis Sci. 2005;46(3):900–907. doi: 10.1167/iovs.04-0495. [DOI] [PubMed] [Google Scholar]
- 14.Tseng S.C., Li D.Q., Ma X. Suppression of transforming growth factor-beta isoforms, TGF-beta receptor type II, and myofibroblast differentiation in cultured human corneal and limbal fibroblasts by amniotic membrane matrix. J Cell Physiol. 1999;179(3):325–335. doi: 10.1002/(SICI)1097-4652(199906)179:3<325::AID-JCP10>3.0.CO;2-X. [DOI] [PubMed] [Google Scholar]
- 15.Kuk JC N., Alhomrani M., Lim R., Sievert W., Hodge A., Moore G. Human amnion epithelial cells and their conditioned media reduces intestinal inflammation and fibrosis in a murine model of chronic colitis. J Crohns Colitis. 2018;12(Supplement 1):S072. [Google Scholar]
- 16.Sakuragawa N., Enosawa S., Ishii T., et al. Human amniotic epithelial cells are promising transgene carriers for allogeneic cell transplantation into liver. J Hum Genet. 2000;45(3):171–176. doi: 10.1007/s100380050205. [DOI] [PubMed] [Google Scholar]
- 17.Akle C.A., Adinolfi M., Welsh K.I., Leibowitz S., McColl I. Immunogenicity of human amniotic epithelial cells after transplantation into volunteers. Lancet. 1981;2(8254):1003–1005. doi: 10.1016/s0140-6736(81)91212-5. [DOI] [PubMed] [Google Scholar]
- 18.Malhotra A., Lim R., Mockler J.C., Wallace E.M. Two-year outcomes of infants enrolled in the first-in-human study of amnion cells for bronchopulmonary dysplasia. Stem Cells Transl Med. 2020;9(3):289–294. doi: 10.1002/sctm.19-0251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yeager A.M., Singer H.S., Buck J.R., et al. A therapeutic trial of amniotic epithelial cell implantation in patients with lysosomal storage diseases. Am J Med Genet. 1985;22(2):347–355. doi: 10.1002/ajmg.1320220219. [DOI] [PubMed] [Google Scholar]
- 20.Lim R., Malhotra A., Tan J., et al. First-in-human administration of allogeneic amnion cells in premature infants with bronchopulmonary dysplasia: a safety study. Stem Cells Transl Med. 2018;7(9):628–635. doi: 10.1002/sctm.18-0079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lim R., Hodge A., Moore G., Wallace E.M., Sievert W. A pilot study evaluating the safety of intravenously administered human amnion epithelial cells for the treatment of hepatic fibrosis. Front Pharmacol. 2017;8:549. doi: 10.3389/fphar.2017.00549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Phan T.G., Lim R., Chan S.T., et al. Phase I trial outcome of amnion cell therapy in patients with ischemic stroke (I-ACT) Front Neurosci. 2023;17 doi: 10.3389/fnins.2023.1153231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kuk JC N., Alhomrani M., Lim R., Sievert W., Hodge A., Moore G. Human amnion epithelial cells reduce intestinal inflammation in a 2,4,6-trinitrobenzene sulfonic acid induced model of acute colitis. J Crohns Colitis. 2018;12(Supplement 1):S072–S073. [Google Scholar]
- 24.Sandborn W.J., Fazio V.W., Feagan B.G., Hanauer S.B., American Gastroenterological Association Clinical Practice C. AGA technical review on perianal Crohn's disease. Gastroenterology. 2003;125(5):1508–1530. doi: 10.1016/j.gastro.2003.08.025. [DOI] [PubMed] [Google Scholar]
- 25.Irvine E.J. Usual therapy improves perianal Crohn's disease as measured by a new disease activity index. McMaster IBD Study Group. J Clin Gastroenterol. 1995;20(1):27–32. [PubMed] [Google Scholar]
- 26.Losco A., Vigano C., Conte D., Cesana B.M., Basilisco G. Assessing the activity of perianal Crohn's disease: comparison of clinical indices and computer-assisted anal ultrasound. Inflamm Bowel Dis. 2009;15(5):742–749. doi: 10.1002/ibd.20826. [DOI] [PubMed] [Google Scholar]
- 27.Samaan M.A., Puylaert C.A.J., Levesque B.G., et al. The development of a magnetic resonance imaging index for fistulising Crohn's disease. Aliment Pharmacol Ther. 2017;46(5):516–528. doi: 10.1111/apt.14190. [DOI] [PubMed] [Google Scholar]
- 28.Hindryckx P., Jairath V., Zou G., et al. Development and validation of a magnetic resonance index for assessing fistulas in patients with crohn's disease. Gastroenterology. 2019;157(5):1233–12344 e5. doi: 10.1053/j.gastro.2019.07.027. [DOI] [PubMed] [Google Scholar]
- 29.Murphy S., Rosli S., Acharya R., et al. Amnion epithelial cell isolation and characterization for clinical use. Curr Protoc Stem Cell Biol. 2010 doi: 10.1002/9780470151808.sc01e06s13. Chapter 1: Unit 1E 6. [DOI] [PubMed] [Google Scholar]
- 30.Georgiev-Hristov T., Guadalajara H., Herreros M.D., et al. A step-by-step surgical protocol for the treatment of perianal fistula with adipose-derived mesenchymal stem cells. J Gastrointest Surg. 2018;22(11):2003–2012. doi: 10.1007/s11605-018-3895-6. [DOI] [PubMed] [Google Scholar]
- 31.Gray A., Marrero-Berrios I., Weinberg J., et al. The effect of local anesthetic on pro-inflammatory macrophage modulation by mesenchymal stromal cells. Int Immunopharmacol. 2016;33:48–54. doi: 10.1016/j.intimp.2016.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lee W.Y., Park K.J., Cho Y.B., et al. Autologous adipose tissue-derived stem cells treatment demonstrated favorable and sustainable therapeutic effect for Crohn's fistula. Stem Cell. 2013;31(11):2575–2581. doi: 10.1002/stem.1357. [DOI] [PubMed] [Google Scholar]
- 33.Herreros M.D., Garcia-Olmo D., Guadalajara H., Georgiev-Hristov T., Brandariz L., Garcia-Arranz M. Stem cell therapy: a compassionate use program in perianal fistula. Stem Cells Int. 2019;2019 doi: 10.1155/2019/6132340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhou C., Li M., Zhang Y., et al. Autologous adipose-derived stem cells for the treatment of Crohn's fistula-in-ano: an open-label, controlled trial. Stem Cell Res Ther. 2020;11(1):124. doi: 10.1186/s13287-020-01636-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.de la Portilla F., Alba F., Garcia-Olmo D., Herrerias J.M., Gonzalez F.X., Galindo A. Expanded allogeneic adipose-derived stem cells (eASCs) for the treatment of complex perianal fistula in Crohn's disease: results from a multicenter phase I/IIa clinical trial. Int J Colorectal Dis. 2013;28(3):313–323. doi: 10.1007/s00384-012-1581-9. [DOI] [PubMed] [Google Scholar]
- 36.Garcia-Arranz M., Herreros M.D., Gonzalez-Gomez C., et al. Treatment of crohn's-related rectovaginal fistula with allogeneic expanded-adipose derived stem cells: a phase I-IIa clinical trial. Stem Cells Transl Med. 2016;5(11):1441–1446. doi: 10.5966/sctm.2015-0356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Park K.J., Ryoo S.B., Kim J.S., et al. Allogeneic adipose-derived stem cells for the treatment of perianal fistula in Crohn's disease: a pilot clinical trial. Colorectal Dis. 2016;18(5):468–476. doi: 10.1111/codi.13223. [DOI] [PubMed] [Google Scholar]
- 38.Garcia-Olmo D., Herreros D., Pascual I., et al. Expanded adipose-derived stem cells for the treatment of complex perianal fistula: a phase II clinical trial. Dis Colon Rectum. 2009;52(1):79–86. doi: 10.1007/DCR.0b013e3181973487. [DOI] [PubMed] [Google Scholar]
- 39.Ciccocioppo R., Bernardo M.E., Sgarella A., et al. Autologous bone marrow-derived mesenchymal stromal cells in the treatment of fistulising Crohn's disease. Gut. 2011;60(6):788–798. doi: 10.1136/gut.2010.214841. [DOI] [PubMed] [Google Scholar]
- 40.Cho Y.B., Lee W.Y., Park K.J., Kim M., Yoo H.W., Yu C.S. Autologous adipose tissue-derived stem cells for the treatment of Crohn's fistula: a phase I clinical study. Cell Transplant. 2013;22(2):279–285. doi: 10.3727/096368912X656045. [DOI] [PubMed] [Google Scholar]
- 41.Garcia-Olmo D., Garcia-Arranz M., Herreros D., Pascual I., Peiro C., Rodriguez-Montes J.A. A phase I clinical trial of the treatment of Crohn's fistula by adipose mesenchymal stem cell transplantation. Dis Colon Rectum. 2005;48(7):1416–1423. doi: 10.1007/s10350-005-0052-6. [DOI] [PubMed] [Google Scholar]
- 42.Garcia-Olmo D., Guadalajara H., Rubio-Perez I., Herreros M.D., de-la-Quintana P., Garcia-Arranz M. Recurrent anal fistulae: limited surgery supported by stem cells. World J Gastroenterol. 2015;21(11):3330–3336. doi: 10.3748/wjg.v21.i11.3330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wang S., Ruan P., Peng L., Wang J. Cytokine-stimulated human amniotic epithelial cells alleviate DSS-induced colitis in mice through anti-inflammation and regulating Th17/Treg balance. Int Immunopharmacol. 2023;120 doi: 10.1016/j.intimp.2023.110265. [DOI] [PubMed] [Google Scholar]
- 44.Carvello M., Lightner A., Yamamoto T., Kotze P.G., Spinelli A. Mesenchymal stem cells for perianal crohn's disease. Cells. 2019;8(7) doi: 10.3390/cells8070764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.ArefNezhad R., Motedayyen H., Mohammadi A. Therapeutic aspects of mesenchymal stem cell-based cell therapy with a focus on human amniotic epithelial cells in multiple sclerosis: a mechanistic review. Int J Stem Cells. 2021;14(3):241–251. doi: 10.15283/ijsc21032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Motedayyen H., Esmaeil N., Tajik N., et al. Method and key points for isolation of human amniotic epithelial cells with high yield, viability and purity. BMC Res Notes. 2017;10(1):552. doi: 10.1186/s13104-017-2880-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Musial-Wysocka A., Kot M., Majka M. The pros and cons of mesenchymal stem cell-based therapies. Cell Transplant. 2019;28(7):801–812. doi: 10.1177/0963689719837897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Molendijk I., Bonsing B.A., Roelofs H., et al. Allogeneic bone marrow-derived mesenchymal stromal cells promote healing of refractory perianal fistulas in patients with crohn's disease. Gastroenterology. 2015;149(4):918–927.e6. doi: 10.1053/j.gastro.2015.06.014. [DOI] [PubMed] [Google Scholar]
- 49.Barnhoorn M.C., Van Halteren A.G.S., Van Pel M., et al. Lymphoproliferative disease in the rectum 4 Years after local mesenchymal stromal cell therapy for refractory perianal crohn's fistulas: a case report. J Crohns Colitis. 2019;13(6):807–811. doi: 10.1093/ecco-jcc/jjy220. [DOI] [PubMed] [Google Scholar]
- 50.Miki T., Lehmann T., Cai H., Stolz D.B., Strom S.C. Stem cell characteristics of amniotic epithelial cells. Stem Cell. 2005;23(10):1549–1559. doi: 10.1634/stemcells.2004-0357. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.