Abstract
In spite of the present belief that latent cytomegalovirus (CMV) infection drives CD8+ T-cell differentiation and induces premature immune senescence, no systematic studies have so far been performed to compare phenotypical and functional changes in the CD8+ T-cell repertoire in CMV-infected and noninfected persons of different age groups. In the present study, number, cytokine production, and growth potential of naïve (CD45RA+ CD28+), memory (CD45RA− CD28+), and effector (CD45RA+ CD28− or CD45RA− CD28−) CD8+ T cells were analyzed in young, middle-aged, and elderly clinically healthy persons with a positive or negative CMV antibody serology. Numbers and functional properties of CMVpp65495-503-specific CD8+ T cells were also studied. We demonstrate that aging as well as CMV infection lead to a decrease in the size of the naïve CD8+ T-cell pool but to an increase in the number of CD8+ effector T cells, which produce gamma interferon but lack substantial growth potential. The size of the CD8+ memory T-cell population, which grows well and produces interleukin-2 (IL-2) and IL-4, also increases with aging, but this increase is missing in CMV carriers. Life-long latent CMV infection seems thus to diminish the size of the naïve and the early memory T-cell pool and to drive a Th1 polarization within the immune system. This can lead to a reduced diversity of CD8 responses and to chronic inflammatory processes which may be the basis of severe health problems in elderly persons.
Depending on the geographic area, 60 to 100% of the adult population is infected by cytomegalovirus (CMV), a betaherpesvirus. In immunosuppressed persons, such as patients with human immunodeficiency virus type 1 infection or children following bone marrow transplantation, CMV infection may cause life-threatening diseases of a variety of different organs, such as the lung, the retina, the liver, and the central nervous system (6, 7, 15, 20). In immunocompetent persons, CMV infection remains mostly unnoticed, as it causes no or few relatively nonspecific symptoms. This lack of clinical manifestation is due to the fact that CMV-specific CD8+ T cells, many of which have specificity for the immediate-early protein IE-1 and the structural protein pp65, are capable of controlling the virus by preventing its replication and by killing virus-infected cells (1, 31, 36, 37). This mostly long-lasting attempt of the immune system to defend a host from spreading of CMV leads to characteristic changes in the CD8+ T-cell repertoire, the most striking one being the accumulation of CD8+ effector T cells with a CD28− phenotype (12, 18). CD28− CMV-specific T cells may either have a CD45RA− or a CD45RA+ phenotype (37). CMV-specific CD8+ effector T cells have also been shown to occur as large expanded clones which may dominate the repertoire (16, 18).
Whether the seemingly preferential expansion of CMV-specific clones reduces the space still available for CD8+ T cells of other specificities is not yet fully understood. However, a recent publication demonstrated that the humoral immune response to influenza vaccination is reduced in CMV carriers (35). The effect was observed in all age groups but was most pronounced in elderly persons. This finding may indicate that life-long CMV infection may restrict immunological diversity and compromise immune responsiveness in old age. Indeed, changes in the CD8+ T-cell repertoire reminiscent of those observed in persons with CMV infection have been frequently described in elderly persons but have mostly been interpreted as a consequence of old age per se and not linked with CMV infection (8, 13, 14, 32, 39). Two of these studies have additionally shown that accumulation of CD28− effector T cells is characteristically linked to a decreased humoral immune response to influenza vaccination in elderly persons (13, 32).
In spite of the strong circumstantial evidence that CMV may be a dominant factor to drive CD8+ T-cell differentiation and hereby induce premature immune senescence (29), practically no systematic studies have so far been performed to compare phenotypical and functional changes in the CD8 T-cell repertoire in CMV-infected and noninfected persons of different age groups (25). The present study for the first time demonstrates that aging per se leads to characteristic changes in the T-cell repertoire, some of which may be accelerated but others of which may be prevented by life-long latent CMV infection.
MATERIALS AND METHODS
Volunteers.
Blood was obtained from 74 apparently healthy persons (age 22 to 91 years, mean ± standard error of the mean [SEM], 56 ± 9 years; 33 males and 41 females). All participants gave their written informed consent, and the study was approved of by the local ethical committee. All subjects were in good clinical condition, not suffering from any severe acute or chronic illness, nor taking any medication to interfere with immune function. HLA typing was routinely performed. Serum CMV antibody (immunoglobulin G) titers were determined by enzyme-linked immunosorbent assay (Enzygnost [R] anti-CMV/immunoglobulin G; Dade Behring, Vienna, Austria), according to the specifications given by the manufacturer. On the basis of age and CMV antibody positivity, six subcohorts were formed: young persons (<35 years) with latent CMV infection (n = 8; age, 28 ± 1 years [range, 24 to 34 years]; CMV antibody titer geometric mean [GM], 11,145 [range, 2,300 to 42,000]) or without latent CMV infection (n = 11; age, 27 ± 1 years [range, 22 to 32 years]; no detectable CMV antibodies); middle-aged persons (35 to 60 years) with (n = 7; age, 47 ± 3 years [range, 38 to 56 years]; CMV antibody titer GM, 4,690 [range, 1,900 to 12,000]) or without (n = 9; age, 47 ± 2 years [range, 38 to 56 years]; no detectable CMV antibodies) latent CMV infection; and elderly persons (>65 years) with (n = 27; age, 71 ± 1 years [range, 0 to 85 years]; CMV antibody titer GM, 19,861 [range, 9,500 to 57,000]) or without (n = 12; age, 75 ± 3 years [range, 62 to 91 years]; no detectable CMV antibodies) latent CMV infection.
PBMC.
Peripheral blood mononuclear cells (PBMC) were obtained by density gradient centrifugation (Ficoll-Hypaque; Amersham Biosciences, Uppsala, Sweden) from freshly drawn venous blood. After washing, PBMC were resuspended in RPMI 1640 (BioWhittaker, Verviers, Belgium) supplemented with 10% fetal calf serum and 1% penicillin-streptomycin (Gibco, Invitrogen Corporation, Paisley, United Kingdom).
Cell surface staining.
For cell surface staining, 2 × 106 PBMC were incubated with monoclonal antibodies specific for CD8, CD45RA, CD45RO, CD28, CD25, and CD62 ligand (CD62L) and labeled with fluorescein isothiocyanate (FITC), phycoerythrin (PE), peridinin chlorophyll protein (PerCP), or allophycocyanin (APC) (all antibodies were purchased from BD Pharmingen, San Jose, Calif.) for 30 min at 4°C. Subsequently, cells were washed twice with phosphate-buffered saline (PBS) and measured by four-color flow cytometry.
Analysis of CMV-specific cells was performed in HLA-A2-positive donors. As CMVpp65495-503 tetramer binding cells had in pilot studies never been found in CMV Ab-negative donors, the analyses were only performed in CMV Ab-positive donors (young n = 8, middle-aged n = 6, elderly n = 18). A total of 3 × 106 PBMC were incubated with CMVpp65495-503 tetramer (NLVPMVATV) PE labeled (Proimmune, Oxford, United Kingdom) in combination with the monoclonal antibodies described above for 30 min at 4°C. Cells were washed, and the intensity of the bound fluorescent labels was measured using four-color flow cytometry. All analyses were performed using a FACSCalibur flow cytometer utilizing CELLQuest software (BD Pharmingen).
Intracellular staining. (i) Cytokines.
A total of 5 × 106 PBMC from young, middle-aged, and elderly persons were incubated with 30 ng of phorbol myristate acetate (PMA)/ml and 5 μg of ionomycin/ml in the presence of 10 μg of brefeldin A/ml for 4 h at 37°C. After washing with PBS, stimulated PBMC were resuspended in PBS and incubated with either CMVpp65495-503 tetramer-PE in combination with anti-CD8-PerCP (in HLA-A2-positive donors) or, alternatively, with anti-CD8-PerCP in combination with CD45RA-FITC and CD28-APC for 30 min at 4°C. Following two washing steps, cells were resuspended in Cytofix-Cytoperm (BD Pharmingen) according to specifications given by the manufacturer. Cells were again washed twice and resuspended in perm-wash buffer (BD Pharmingen). Anti-gamma interferon (IFN-γ) and anti-interleukin-2 (IL-2), FITC or PE labeled, and anti-IL-4, APC or PE labeled (BD Pharmingen), were added to the cells and incubated for 30 min at 4°C. Intracellular cytokines were also analyzed by four-color flow cytometry. All analyses were performed using a FACSCalibur flow cytometer utilizing CELLQuest software (BD Pharmingen).
(ii) Perforin.
Perforin expression was assessed by intracellular staining using the Cytofix-Cytoperm kit according to the specifications given by the manufacturer (BD Pharmingen). Five microliters of FITC-conjugated antiperforin antibody (BD Pharmingen) was added to permeabilized unstimulated PBMC and incubated for 30 min at 4°C. Cells were washed twice with cytowash buffer (BD Pharmingen) and once with PBS. Perforin detection was performed by flow cytometry.
Purification of CD8 T-cell subpopulations and antigenic stimulation.
Naïve/early memory cells as well as effector cells were isolated from two young and two elderly CMV-positive donors. The naïve/early memory CD8+ T-cell population was defined by the expression of CD28. Two types of effector T cells were purified: CD28− CD45RO+ and CD28− CD45RO− cells. CD8+ cells were preisolated using a magnetic-activated cell sorter (MACS) multisort kit (Miltenyi Biotec, Bergisch Gladbach, Germany) and subsequently stained with anti-CD28-APC antibody for 20 min at 4°C. After washing, anti-APC beads (Miltenyi Biotec) were added according to the manufacturer's instructions, and CD28+ cells were isolated from the CD28− cells by using a MACS separation column (Miltenyi Biotec). CD45RO+ cells were purified from CD28− cells by positive selection using anti-CD45RO beads (Miltenyi Biotec). The purity of each subpopulation was determined by fluorescence-activated cell sorter analysis and was more than 98%. The three subpopulations were stimulated with CMVpp65495-503 peptide (NLVPMVATV) in the presence of IL-2 (20 ng/ml) for 1 week. The three resulting CD8+ T-cell populations were analyzed for the frequency of CMV tetramer binding cells as well as for the expression of CD8, CD28, and CD45RA.
Statistical analysis.
SPSS for Windows version 11.5 (SPSS Inc., Chicago, Ill.) was used for statistical analysis. Comparisons between young, middle-aged, and elderly persons with and without CMV antibody serology were done by analysis of variance followed by post hoc multiple comparisons. Pearson's linear regression analysis was performed. P values of less than 0.05 were considered statistically significant. Data are presented as means ± SEM.
RESULTS
Age and CMV infection induce a decrease in naïve but an increase in effector CD8+ T-cell numbers.
To determine the influence of CMV infection on the expression of CD28 and CD45RA within the CD8+ T-cell pool, PBMC from young, middle-aged, and elderly CMV-seronegative and -seropositive persons were analyzed by flow cytometry. After gating CD8++ T cells, four subpopulations were identified according to the expression of CD28 and CD45RA. CD45RA+ CD28+ cells were classified as naïve, CD45RA− CD28+ cells were classified as memory, and CD28− cells were classified as effector T cells. Within the effector T-cell group, two subpopulations were identified: CD45RA− CD28− and CD45RA+ CD28− cells. The size of each population was studied in CMV-negative and CMV-positive persons of each age group (Fig. 1). Age as well as latent CMV infection affected the size of the different CD8+ subpopulations. In the absence of latent CMV infection, the size of the naïve CD8 T-cell pool was unchanged in the middle-aged group but significantly reduced in elderly persons. Latent CMV infection led to a decrease in the number of naïve T cells in each age group compared with uninfected age-matched controls, but the size of the naïve T-cell pool was most severely reduced in elderly persons with latent CMV infection.
FIG. 1.
Aging and CMV infection affect the differentiation of CD8+ T cells. Four CD8+ T-cell subpopulations were defined according to the expression of CD45RA and CD28 within gated CD8++ T cells. (A) The distribution of these subpopulations is demonstrated by showing the example of two young, two middle-aged, and two elderly persons, one without and one with latent CMV infection in each age group. (B) Distribution of naïve (I), memory (II), effector CD45RA− (III), and effector CD45RA+ (IV) CD8 T cells in the different cohorts. □, CMV antibody (Ab) negative; ▨, CMV Ab positive. Data are presented as the mean ± SEM, and statistical comparisons between the groups were performed. Significant differences are indicated as follows: *, CMV Ab- versus CMV Ab+ persons within each age group; †, young versus elderly; #, middle-aged versus elderly within persons of one CMV Ab status.
The number of CD45RA− CD28+ memory CD8+ T cells increased with age in the absence of latent CMV infection, but this increase was not observed in elderly CMV carriers.
In contrast, the numbers of CD28− effector cells increased with age as well as with latent CMV infection, leading to highest effector cell numbers in CMV-infected elderly persons. It was of interest that CMV-infected persons of each age group had increased numbers of CD45RA+ effector T cells compared to uninfected age-matched controls, while CD45RA− effector cells mostly accumulated in CMV-infected elderly persons.
Cytokine production by CD8+ T cells is affected by CMV infection.
In order to analyze whether the CMV status of young, middle-aged, and elderly donors influenced the cytokine production profile of total unpurified CD8 cells, IL-2, IL-4, and IFN-γ production were analyzed in response to stimulation with PMA-ionomycin (Fig. 2). Aging per se led to an increase in the production of all three cytokines upon nonspecific stimulation, as the numbers of IL-2-, IL-4-, and IFN-γ-producing cells were increased in elderly persons in the absence of latent CMV infection (Fig. 2A). The age-related increase in the production of IL-2 and IL-4 was, however, not observed in persons with a positive CMV antibody serology, leading to a pronounced difference in the production profile of these two cytokines between elderly persons with and those without latent CMV infection. In contrast, the age-related increase in the production of IFN-γ was not abolished by CMV infection. IFN-γ production was even higher in middle-aged and elderly persons with a positive CMV antibody serology than in uninfected age-matched controls. In order to define whether the observed changes in the cytokine production profiles were due to the various sizes of the different CD8 T-cell subsets in the six study groups, IL-2-, IL-4-, and IFN-γ-producing cells were gated and analyzed for their expression of CD28 and CD45RA. They were correspondingly classified as naïve, memory, or effector cells, as demonstrated in Fig. 1A. Figure 2B shows that IL-2 was mostly produced by CD28+ naïve and memory T cells in all age groups, while IL-4 was almost exclusively produced by CD28+ memory T cells. Increased IL-2 and IL-4 production in uninfected elderly persons may thus be due to increased memory cell numbers, while decreased IL-2 and IL-4 production by elderly persons with CMV infections could be the result of the smaller size of the CD45RA− CD28+ population and the low naïve T-cell count observed in this cohort. IFN-γ was produced by antigen-experienced CD28+ as well as CD28− cells. The increase in memory and effector CD8+ T cells observed with age and CMV infection may therefore explain the increased production of this cytokine by the total CD8+ T-cell population.
FIG. 2.
Aging and CMV infection lead to an imbalance in the cytokine production profile of the total unpurified CD8+ T-cell population. The production of IL-2, IL-4, and IFN-γ in response to stimulation with PMA-ionomycin was studied. (A) Cytokine production profile of the total gated CD8+ T-cell population in CMV antibody (Ab)-negative and -positive persons of different age groups. Statistical comparisons between the groups were done, and the results are presented as means ± SEM. Significant differences are shown as follows: *, CMV Ab- versus CMV Ab+ persons within each age group; †, young versus elderly; #, middle-aged versus elderly within persons of one CMV Ab status. (B) The example of two young, two middle-aged, and two elderly persons is shown, one without and one with latent CMV infection in each age group. CD8 cells producing either IL-2, IL-4, or IFN-γ were gated and analyzed for their expression of CD45RA and CD28.
Elderly persons have a CD25-expressing IL-2-, IL-4-producing memory T-cell subpopulation, the size of which decreases in the presence of latent CMV infection.
We analyzed whether the pronounced difference in the production of IL-2 and IL-4 observed between CD8+ T cells from elderly persons with and without latent CMV infection might be due to the specific decrease in the number of IL-2- and IL-4-producing CD8+ CD28+ memory T cells in elderly persons with CMV infection. An IL-2- IL-4-producing CD8+ T-cell population, which characteristically occurs in 30% of the elderly but not in young persons, has recently been characterized by our laboratory (33). The occurrence of this specific CD8+ T-cell population is associated with a good humoral immune response in old age. Although it does not appear to have a regulatory function, it constitutively expresses CD25 and can be easily identified. We therefore analyzed the frequency of CD25+ cells within the CD8+ population in our study groups. In agreement with our previous results (33), we demonstrated the occurrence of a CD25+-expressing CD28+ CD45RA− subpopulation of substantial size in elderly persons without CMV infection (Fig. 3). The size of this population was significantly reduced in elderly persons with CMV infection, suggesting that the low IL-2 and IL-4 production capacity of the total CD8+ T-cell population of CMV-infected elderly persons (Fig. 2) may be due to the partial loss of a physiologically occurring age-related T-cell subset with specific stimulatory properties. These changes seem to start in mid-life, as a minor (not significant) decrease in the number of CD25+ CD8+ T cells was also observed in CMV-infected persons in the middle-aged group compared with noninfected age-matched controls.
FIG. 3.
(A) A CD25+CD8+ T-cell subpopulation of substantial size is found within the CD45RA− CD28+ memory population in elderly persons without CMV infection. This population is decreased in size in elderly persons with latent CMV infection as well as in younger age groups. A representative example of CD25-expressing cells (black dots) superimposed on a double staining of CD45RA and CD28 in gated CD8+ cells (gray dots) in the different study groups is shown. (B) The frequency of CD25-expressing cells in CD8+ T cells is shown in young, middle-aged, and in elderly persons with (▨) and without (□) latent CMV infection. Bars represent means ± SEM. Significant differences are indicated as follows: *, CMV antibody-negative (Ab−) versus CMV Ab+ persons; †, young versus elderly; #, middle-aged versus elderly persons without latent CMV infection.
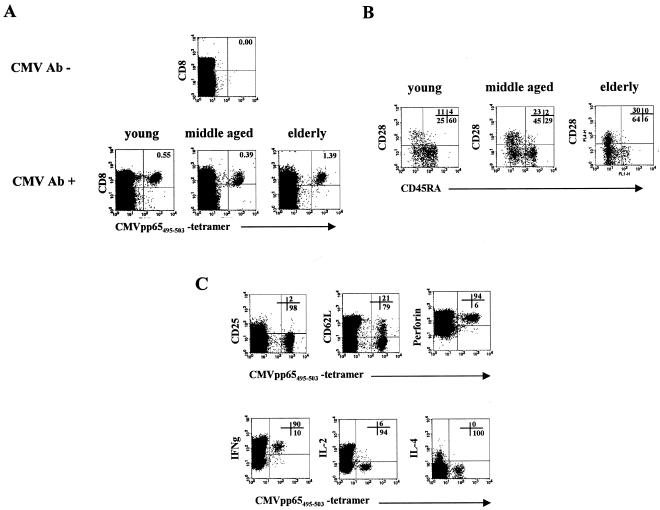
CMVpp65495-503 tetramer binding CD8+ T cells have an effector cell phenotype.
The frequency and the phenotype of CMVpp65495-503 tetramer CD8+ T cells were studied in young, middle-aged, and elderly HLA-A2-positive donors (Fig. 4). None of the CMV-seronegative HLA-A2-positive donors of any age group had CD8+ cells that bound the CMVpp65495-503 tetramer (Fig. 4A). In contrast, CMV-seropositive donors had CD8+ T cells that specifically recognized the pp65495-503 (NLVPMVATV) peptide (Fig. 4A). CMVpp65495-503 tetramer binding cells were analyzed for their expression of CD45RA and CD28 (Fig. 4B). In all three age groups, CMV pp65495-503 tetramer binding CD8 T cells could be found in the CD28+ CD45RA− memory as well as in the CD28− CD45RA− and the CD28− CD45RA+ effector subpopulations. Interestingly, the relative distribution of tetramer binding cells in the different subpopulations varied among the age groups. While in the young group the majority of tetramer binding cells had a CD28− CD45RA+ phenotype (60.0 ± 4.4% [mean ± SEM]; n = 8), tetramer binding cells most frequently had a CD28− CD45RA− phenotype in the middle-aged group (46.1 ± 6.1%; n = 6). This preferential occurrence of CD28− CD45RA− tetramer binding cells was even more pronounced in the elderly population (64.5 ± 6.9%; n = 18). The size of the CD28+ tetramer binding population also tended to increase with age. In all age groups, CMVpp65495-503 tetramer binding cells were CD25−, mostly CD62L−, and expressed perforin (Fig. 4C). Upon stimulation with PMA and ionomycin, CMVpp65495-503 tetramer binding cells produced IFN-γ but very little IL-2 and no IL-4.
FIG. 4.
CMVpp65495-503-specific CD8+ T cells have an effector phenotype and produce IFN-γ. (A) Representative example of CMVpp65495-503 tetramer binding CD8+ T cells in a CMV antibody (Ab)-negative donor and in one young, one middle-aged, and one elderly person with a positive CMV Ab serology. (B) CD28 CD45RA double staining of CMVpp65495-503 tetramer binding cells within the CD8+ gate. A typical example of one young, one middle-aged, and one elderly person is shown. Figures in the diagram represent the percentage of cells of each phenotype within the total CMVpp65495-503 tetramer binding population, which was considered 100%. (C) Representative example showing the expression of CD25, CD62L, and perforin as well as the production of IL-2, IL-4, and IFN-γ in CMVpp65495-503 tetramer binding cells within the CD8+ gate. Cytokines were analyzed following stimulation with PMA and ionomycin. The diagram represents the staining of cells from one elderly person with a positive CMV Ab serology. Similar results were obtained in all other HLA-A2-positive donors with a positive CMV Ab serology from the three age groups.
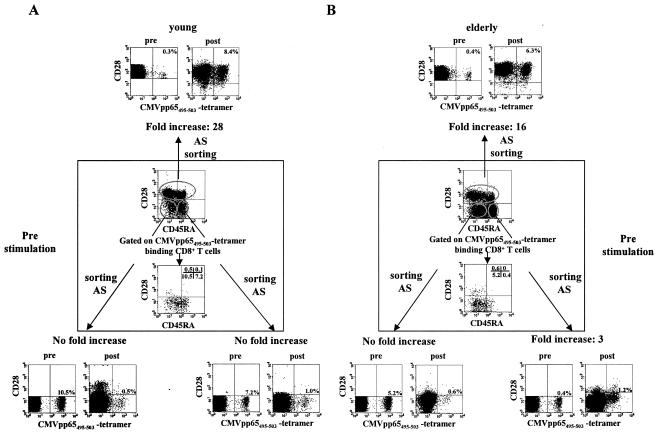
Following antigenic stimulation, CMVpp65495-503-specific CD28+ T cells have a significantly higher growth potential than their CD28− counterparts.
In order to analyze the growth potential of CMVpp65495-503 tetramer binding CD8+ T cells of different phenotypes and from persons of different age groups, we purified three CD8+ T-cell subpopulations from two young and two elderly CMV antibody-positive donors: CD28+ cells, CD28− CD45RA− cells, and CD28− CD45RA+ cells. These cells were then put into culture and stimulated with the CMVpp65495-503 peptide in the presence of IL-2. After 1 week of culture, the proliferation of peptide-specific cells from the different subpopulations was monitored by analyzing the frequency of tetramer binding cells. Figure 5 demonstrates that although the initial percentages of tetramer binding cells had been by far higher in the CD28− subpopulations than in the CD28+ population, the proliferation of peptide-specific cells in response to antigenic stimulation was much more pronounced in the CD28+ than in the CD28− subsets. While a more-than-15-fold increase (young, from 0.3 to 8.4%; elderly, from 0.4 to 6.3%) in the percentage of CMVpp65495-503 tetramer binding CD8+ T cells was observed in the CD28+ population, only unsubstantial proliferation of peptide-specific cells occurred in the CD28− CD45RA+ population. There was practically no growth of peptide-specific cells in the CD28− CD45RA− subset. The relatively few peptide-specific cells which could be captured from the CD28− CD45RA+ T-cell population following antigenic stimulation did not reexpress CD28 following culture (Fig. 5) but reconverted to a CD45RO phenotype (data not shown).
FIG. 5.
Following antigenic stimulation, CMVpp65495-503-specific CD28+ CD8+ T cells have a more pronounced growth potential than their CD28− counterparts. CD28+, CD28− CD45RA−, and CD28− CD45RA+ T cells were isolated, as described in Materials and Methods, and stimulated with CMVpp65495-503 (NLVPMVATV) (2 μg/ml) in the presence of IL-2 (20 ng/ml) for 1 week. Stainings of unpurified cells before subset isolation are shown in the middle of the graph (box): CD28 and CD45RA expression levels in gated CD8 cells as well as in cells gated on the CMVpp65495-503 tetramer binding population are shown. The percentages of cells within each subpopulation that were tetramer positive before in vitro culture are shown outside the box under the heading Pre. Tetramer binding cells in the three subpopulations following 1 week's culture in the presence of CMVpp65495-503 peptide and IL-2 are also shown outside the box. The fold increase in the percentage of peptide-specific cells is indicated for each culture. AS, antigenic stimulation. The experiment was performed in cells from two young and two elderly CMV antibody-positive persons. The graph shows a characteristic example of one young and one elderly person.
DISCUSSION
Our results demonstrate that long-term latent CMV infection leads to pronounced changes in the CD8 T-cell repertoire. In agreement with previous publications (1, 18), we have shown that CMV is accompanied by a dramatic accumulation of CD28− effector cytotoxic T lymphocytes. This is a characteristic feature of all age groups but is most pronounced in elderly persons. Although we cannot presently provide data on absolute cell counts and the exact time point of primary infection in these persons is not known, it seems likely that elderly persons have been infected for many years. Time may thus play a critical role in determining the amount of effector cell accumulation.
It is of interest that CD28− effector T cells in general and CMVpp65495-503 tetramer binding cells in particular frequently have a CD45RA− phenotype in elderly persons with latent CMV infections, while corresponding cells from younger persons are mostly CD28− CD45RA+. CD28− CD45RA− T cells have been reported to represent truly senescent cells, as they resemble CD8 T cells that are driven to the end stage of replicative senescence in cell cultures in response to repeated rounds of antigen-driven proliferation (8, 30). CD57 expression has also been shown to be closely associated with proliferative senescence and might be a helpful parameter in future studies (5). It has also been suggested that CD45RA− CD28− cells have a reduced proliferation and IFN-γ secretion compared with their CD28− CD45RA+ counterparts (27, 38). In agreement with this concept, our results showed that CD28− CD45RA− CMV-specific cells failed to proliferate in vitro in response to antigenic stimulation, even in the presence of IL-2 (Fig. 5). CMV-specific cells with a CD28− CD45RA+ phenotype had a slightly better, but still very moderate, growth potential in vitro, while cells capable of substantial clonal propagation in response to stimulation with CMV peptide had a CD45RA− CD28+ memory phenotype in young as well as in elderly persons. Although failure to proliferate in vitro need not necessarily reflect a general inability to proliferate in vivo, our data suggest that the initially relatively small CD45RA− CD28+ CMV-specific T-cell population seems to be the most important one for the generation of CMV-specific T-cell clones. The facts that the size of this population tends to increase with age and its function is unaffected by aging (Fig. 4 and 5) are in good agreement with the clinical observation that CMV is not reactivated in elderly persons in spite of the frequency and severity of other infections, such as influenza, in old age (10). Staining of markers such as CD27 might have provided additional insight into the exact phenotype of CMV-specific cells, as some memory cells may fail to express CD27 and may still be CD28+ CD45RA+.
Elderly persons thus seem to be well protected against symptomatic CMV disease. Long-term latent CMV infection may, however, cause other health problems in old age. Firstly, CMV infection accelerates the reduction in the naïve T-cell pool, which characteristically occurs following thymic involution (2, 11). The extremely low number of CD8+ naïve T cells in CMV-infected elderly persons (<15% of CD8+ cells [Fig. 1]) may lead to difficulties in the immunological response to neoantigens in old age. This possibility is supported by the recent observation that vaccination with live yellow fever vaccine led to severe systemic illness in elderly persons (19, 21, 22).
Secondly, latent CMV infection seems to reduce the number of IL-2- and IL-4-producing CD8+ memory T cells in old age (Fig. 2 and 3). These cells, which have recently been first described by our laboratory, specifically occur in healthy elderly persons with a good humoral immune response and seem to represent a reservoir of diversity in the absence of naïve T cells (33). It seems likely that the strong immunostimulatory effect of CMV leads to the preferential expansion of CMV-specific CD8 T-cell clones through which cells of other specificities may be destined to perish.
Chronic CMV infection may affect health in old age in yet another way. CMV-specific CD8+ T cells produce IFN-γ but no IL-4 and very little IL-2 (Fig. 4). The extensive accumulation of these cells in CMV-infected elderly persons leads to an increase in the IFN-γ production of the total CD8+ T-cell population (Fig. 2) and consequently to an increase in the whole-body load of IFN-γ, as indicated by high neopterin serum concentrations during CMV infection (17). A high type 1 combined with a low type 2 cytokine production (Fig. 2) can change the cytokine microenvironment in lymphatic tissues and trigger ubiquitous inflammatory processes in elderly persons (9). Type 2 cytokines act on B cells to induce activation and differentiation, whereas low type 2 cytokine production is likely to hamper B-cell propagation and antibody production following immunization in the elderly. Proinflammatory cytokines are now believed to exacerbate the functional pathology and disease course of age-related disorders such as Alzheimer's disease and atherosclerosis (23, 24, 34). IFN-γ triggers the production of the Alzheimer β-amyloid in combination with tumor necrosis factor alpha in human neural and extraneural cells (3) and increases the production of oxygen radicals by microglial cells. Tumor necrosis factor alpha can also increase the toxicity of β-amyloid (4) as well as stimulate smooth muscle cell proliferation, a key event in the development of atherosclerosis. As CMV is postulated to reside in endothelial cells during latency and has been detected in atherosclerotic plaques (26), the latter mechanism may be of particular relevance in elderly persons with chronic CMV infections.
In conclusion, our data illustrate that long-term latent CMV infection leads to characteristic changes in the CD8+ T-cell repertoire which may drive a Th1 polarization within the immune system. This can have detrimental consequences, which could be prevented right from the beginning by early vaccination against CMV. Immunization strategies against CMV are being discussed (28) but have so far not been considered worth further development. This view could be changed by a greater awareness that the prevention of CMV may slow down immunological aging and the development of conditions closely linked to inflammatory processes in old age.
Acknowledgments
This work has been supported by the Austrian Science Funds (project P 16205-B01) and the Austrian Green Cross Society for Preventive Medicine.
REFERENCES
- 1.Appay, V., P. R. Dunbar, M. Callan, P. Klenerman, G. M. Gillespie, L. Papagno, G. S. Ogg, A. King, F. Lechner, C. A. Spina, S. Little, D. V. Havlir, D. D. Richman, N. Gruener, G. Pape, A. Waters, P. Easterbrook, M. Salio, V. Cerundolo, A. J. McMichael, and S. L. Rowland-Jones. 2002. Memory CD8+ T cells vary in differentiation phenotype in different persistent virus infections. Nat. Med. 8:379-385. [DOI] [PubMed] [Google Scholar]
- 2.Aspinall, R., and D. Andrew. 2000. Thymic involution in aging. J. Clin. Immunol. 20:250-256. [DOI] [PubMed] [Google Scholar]
- 3.Blasko, I., F. Marx, E. Steiner, T. Hartmann, and B. Grubeck-Loebenstein. 1999. TNF-α plus IFN-γ induce the production of Alzheimer β-amyloid peptides and decrease the secretion of APPs. FASEB J. 13:63-68. [DOI] [PubMed] [Google Scholar]
- 4.Blasko, I., T. L. Schmitt, E. Steiner, K. Trieb, and B. Grubeck-Loebenstein. 1997. Tumor necrosis factor alpha augments amyloid beta protein (25-35) induced apoptosis in human cells. Neurosci. Lett. 238:17-20. [DOI] [PubMed] [Google Scholar]
- 5.Brenchley, J. M., N. J. Karandikar, M. R. Betts, D. R. Ambrozak, B. J. Hill, L. E. Crotty, J. P. Casazza, J. Kuruppu, S. A. Migueles, M. Connors, M. Roederer, D. C. Douek, and R. A. Koup. 2003. Expression of CD57 defines replicative senescence and antigen-induced apoptotic death of CD8+ T cells. Blood 101:2711-2720. [DOI] [PubMed] [Google Scholar]
- 6.Castagnola, E., B. Cappelli, D. Erba, A. Rabagliati, E. Lanino, and G. Dini. 2004. Cytomegalovirus infection after bone marrow transplantation in children. Hum. Immunol. 65:416-422. [DOI] [PubMed] [Google Scholar]
- 7.Drew, W. L. 1998. Cytomegalovirus infection in patients with AIDS. J. Infect. Dis. 158:449-456. [DOI] [PubMed] [Google Scholar]
- 8.Effros, R. B. 2004. T cell replicative senescence: pleiotropic effects on human aging. Ann. N. Y. Acad. Sci. 1019:123-126. [DOI] [PubMed] [Google Scholar]
- 9.Franceschi, C., M. Bonafe, S. Valensin, F. Olivieri, M. De Luca, E. Ottaviani, and G. De Benedictis. 2000. Inflamm-aging. An evolutionary perspective on immunosenescence. Ann. N. Y. Acad. Sci. 908:244-254. [DOI] [PubMed] [Google Scholar]
- 10.Gavazzi, G., and K. H. Krause. 2002. Ageing and infection. Lancet Infect. Dis. 2:659-666. [DOI] [PubMed] [Google Scholar]
- 11.George, A. J., and M. A. Ritter. 1996. Thymic involution with ageing: obsolescence or good housekeeping? Immunol. Today 17:267-272. [DOI] [PubMed] [Google Scholar]
- 12.Gillespie, G. M., M. R. Wills, V. Appay, C. O'Callaghan, M. Murphy, N. Smith, N., P. Sissons, S. Rowland-Jones, J. I. Bell, and P. A. Moss. 2000. Functional heterogeneity and high frequencies of cytomegalovirus-specific CD8+ T lymphocytes in healthy seropositive donors. J. Virol. 74:8140-8150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Goronzy, J. J., J. W. Fulbright, C. S. Crowson, G. A. Poland, W. M. O'Fallon, and C. M. Weyand. 2001. Value of immunological markers in predicting responsiveness to influenza vaccination in elderly individuals. J. Virol. 75:12182-12187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gupta, S., R. Bi, K. Su, L. Yel, S. Chiplunkar, and S. Gollapudi. 2004. Characterization of naive, memory and effector CD8+ T cells: effect of age. Exp. Gerontol. 39:545-550. [DOI] [PubMed] [Google Scholar]
- 15.Jacobson, M. A. 1998. AIDS-related cytomegalovirus retinitis. Drugs Today (Barcelona) 34:409-413. [DOI] [PubMed] [Google Scholar]
- 16.Jin, X., M. A. Demoitie, S. M. Donahoe, G. S. Ogg, S. Bonhoeffer, W. M. Kakimoto, G. Gillespie, P. A. Moss, W. Dyer, M. G: Kurilla, S. R. Riddell, J. Downie, J. S. Sullivan, A. J. McMichael, C. Workman, and D. F. Nixon. 2000. High frequency of cytomegalovirus-specific cytotoxic T-effector cells in HLA-A*0201-positive subjects during multiple viral coinfections. J. Infect. Dis. 181:165-175. [DOI] [PubMed] [Google Scholar]
- 17.Jungraithmayr, T. C., M. Reschke, S. O. Grebe, H. Lange, K. Radsak, and T. F. Mueller. 2001. Assessment of cytomegalovirus infections using neopterin and a new immunoblot. Clin. Chim. Acta 310:63-69. [DOI] [PubMed] [Google Scholar]
- 18.Khan, N., N. Shariff, M. Cobbold, R. Bruton, J. A. Ainsworth, A. J. Sinclair, L. Nayak, and P. A. Moss. 2002. Cytomegalovirus seropositivity drives the CD8 T cell repertoire toward greater clonality in healthy elderly individuals. J. Immunol. 169:1984-1992. [DOI] [PubMed] [Google Scholar]
- 19.Kitchener, S. 2004. Viscerotropic and neurotropic disease following vaccination with the 17D yellow fever vaccine, ARILVAX. Vaccine 22:2103-2105. [DOI] [PubMed] [Google Scholar]
- 20.Long, C. M., L. Drew, R. Miner, D. Jekic-McMullen, C. Impraim, and S. Y. Kao. 1998. Detection of cytomegalovirus in plasma and cerebrospinal fluid specimens from human immunodeficiency virus-infected patients by the AMPLICOR CMV test. J. Clin. Microbiol. 36:2434-2438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Martin, M., L. H. Weld, T. F. Tsai, G. T. Mootrey, R. T. Chen, M. Niu, and M. S. Cetron. 2001. Advanced age a risk factor for illness temporally associated with yellow fever vaccination. Emerg. Infect. Dis. 7:945-951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Martin, M., T. F. Tsai, B. Cropp, G. J. Chang, D. A. Holmes, J. Tseng, W. Shieh, S. R. Zaki, I. Al-Sanouri, A. F. Cutrona, G. Ray, L. H. Weld, and M. S. Cetron. 2001. Fever and multisystem organ failure associated with 17D-204 yellow fever vaccination: a report of four cases. Lancet 358:98-104. [DOI] [PubMed] [Google Scholar]
- 23.McGeer, P. L., and E. G. McGeer. 1996. Anti-inflammatory drugs in the fight against Alzheimer's disease. Ann. N. Y. Acad. Sci. 777:213-220. [DOI] [PubMed] [Google Scholar]
- 24.McGeer, P. L., and E. G. McGeer. 1995. The inflammatory response system of brain: implications for therapy of Alzheimer and other neurodegenerative diseases. Brain Res. Rev. 21:195-218. [DOI] [PubMed] [Google Scholar]
- 25.Moss, P., and N. Khan. 2004. CD8+ T-cell immunity to cytomegalovirus. Hum. Immunol. 65:456-464. [DOI] [PubMed] [Google Scholar]
- 26.Nerheim, P. L., J. L. Meier, M. A. Vasef, W. G. Li, L. Hu, J. B. Rice, D. Gavrila, W. E. Richenbacher, and N. L. Weintraub. 2004. Enhanced cytomegalovirus infection in atherosclerotic human blood vessels. Am. J. Pathol. 164:589-600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ouyang, Q., W. M. Wagner, W. Zheng, A. Wikby, E. J. Remarque, and G. Pawelec. 2004. Dysfunctional CMV-specific CD8+ T cells accumulate in the elderly. Exp. Gerontol. 39:607-613. [DOI] [PubMed] [Google Scholar]
- 28.Paston, S. J., I. A. Dodi, and J. A. Madrigal. 2004. Progress made towards the development of a CMV peptide vaccine. Hum. Immunol. 65:544-549. [DOI] [PubMed] [Google Scholar]
- 29.Pawelec, G., A. Akbar, C. Caruso, R. Effros, B. Grubeck-Loebenstein, and A. Wikby. 2004. Is immunosenescence infectious? Trends Immunol. 25:406-410. [DOI] [PubMed] [Google Scholar]
- 30.Pawelec, G., A. Rehbein, K. Haehnel, A. Merl, and M. Adibzadeh. 1997. Human T-cell clones in long-term culture as a model of immunosenescence. Immunol. Rev. 160:31-42. [DOI] [PubMed] [Google Scholar]
- 31.Reddehase, M. J. 2000. The immunogenicity of human and murine cytomegaloviruses. Curr. Opin. Immunol. 12:390-396. [DOI] [PubMed] [Google Scholar]
- 32.Saurwein-Teissl, M., T. L. Lung, F. Marx, C. Gschösser, E. Asch, I. Blasko, W. Parson, G. Bock, D. Schönitzer, E. Trannoy, and B. Grubeck-Loebenstein. 2002. Lack of antibody production following immunization in old age: association with CD8+ CD28− T cell clonal expansions and an imbalance in the production of Th1 and Th2 cytokines. J. Immunol. 168:5893-5899. [DOI] [PubMed] [Google Scholar]
- 33.Schwaiger, S., A. M. Wolf, P. Robatscher, B. Jenewein, and B. Grubeck-Loebenstein. 2003. IL-4-producing CD8+ T cells with a CD62L++(bright) phenotype accumulate in a subgroup of older adults and associated with the maintenance of intact humoral immunity in old age. J. Immunol. 170:613-619. [DOI] [PubMed] [Google Scholar]
- 34.Shoenfeld, Y., Y. Sherer, and D. Harats. 2001. Artherosclerosis as an infectious, inflammatory and autoimmune disease. Trends Immunol. 22:293-295. [DOI] [PubMed] [Google Scholar]
- 35.Trzonkowski, P., J. Mysliwska, E. Szmit, J. Wieckiewicz, K. Lukaszuk, L. B. Brydak, M. Machala, and A. Mysliwski. 2003. Association between cytomegalovirus infection, enhanced proinflammatory response and low level of anti-hemagglutinins during the anti-influenza vaccination—an impact of immunosenescence. Vaccine 21:3826-3836. [DOI] [PubMed] [Google Scholar]
- 36.Weekes, M. P., M. R. Wills, K. Mynard, A. J. Carmichael, and J. G. Sissons. 1999. The memory cytotoxic T-lymphocyte (CTL) response to human cytomegalovirus infection contains individual peptide-specific CTL clones that have undergone extensive expansion in vivo. J. Virol. 73:2099-2108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wills, M. R., A. J. Carmichael, M. P. Weekes, K. Mynard, G. Okecha, R. Hicks, and J. G. Sissons. 1999. Human virus-specific CD8+ CTL clones revert from CD45ROhigh to CD45RAhigh in vivo: CD45RAhigh CD8+ T cells comprise both naive and memory cells. J. Immunol. 162:7080-7087. [PubMed] [Google Scholar]
- 38.Wills, M. R., G. Okecha, M. P. Weekes, M. K. Gandhi, P. J. G. Sissons, and A. J. Carmichael. 2002. Identification of naïve or antigen-experienced human CD8+ T cells by expression of costimulation and chemokine receptors: analysis of the human cytomegalovirus-specific CD8+ T cell response. J. Immunol. 168:5455-5464. [DOI] [PubMed] [Google Scholar]
- 39.Zanni, F., R. Vescovili, C. Biasimi, F. Fagnoni, L. Zanlari, A. Telera, P. Di Pede, G. Passeri, M. Pedrazzoni, M. Passeri, C. Franceschi, and P. Sansoni. 2003. Marked increase with age of type 1 cytokines within memory and effector/cytotoxic CD8+ T cells in humans: a contribution to understand the relationship between inflammation and immunosenescence. Exp. Gerontol. 38:981-987. [DOI] [PubMed] [Google Scholar]