Abstract
Respiratory syncytial virus (RSV) is the leading cause of lower respiratory tract infection in young infants worldwide. Previous studies have reported that the induction of interleukin-8/CXCL8 and RANTES/CCL5 correlates with disease severity in humans. The production of these chemokines is elicited by viral replication and is NF-κB dependent. RSV, a negative-sense single-stranded RNA virus, requires full-length positive-sense RNA for synthesis of new viral RNA. The aim of our studies was to investigate whether active viral replication by RSV could evoke chemokine production through TLR3-mediated signaling pathways. In TLR3-transfected HEK 293 cells, live RSV preferentially activated chemokines in both a time- and dose-dependent manner compared to vector controls. RSV was also shown to upregulate TLR3 in human lung fibroblasts and epithelial cells (MRC-5 and A549). Targeting the expression of TLR3 with small interfering RNA decreased synthesis of IP-10/CXCL10 and CCL5 but did not significantly reduce levels of CXCL8. Blocking the expression of the adapter protein MyD88 established a role for MyD88 in CXCL8 production, whereas CCL5 synthesis was found to be MyD88 independent. Production of CCL5 by RSV was induced directly through TLR3 signaling pathways and did not require interferon (IFN) signaling through the IFN-α/β receptor. TLR3 did not affect viral replication, since equivalent viral loads were recovered from RSV-infected cells despite altered TLR3 expression. Taken together, our studies indicate that TLR3 mediates inflammatory cytokine and chemokine production in RSV-infected epithelial cells.
Respiratory syncytial virus (RSV) is the leading cause of bronchiolitis in children and infects nearly all infants by age 3 (41). While infants are subject to recurrent infections, RSV is also becoming widely recognized as an important pathogen in the elderly and in immunosuppressed adults (7, 9, 15). Severe RSV infection involves the release of inflammatory mediators, epithelial cell necrosis, inflammation, and mucus production (27). The airway epithelium is a major site of RSV infection and likely plays an important role in initiating these inflammatory responses. Previous studies have reported that RSV can induce chemokines and their receptors and thus contribute to the recruitment and activation of cell types involved in severe RSV disease (14, 33, 45). Elevated levels of CXCL8 and CCL5 have been shown to correlate with disease severity in humans. Replicating virus and NF-κB activation are required for the increased production of these two chemokines (5, 10, 13, 20, 36). Although promoter analysis experiments have identified upstream transcription factors and regulatory elements involved in RSV-induced CXCL8 and CCL5 synthesis, the mechanism and signaling pathways for RSV-induced inflammation are still not completely understood.
The airway epithelium serves as the interface between the environment and the host and stands as the first line of host defense. The ability of the airway to detect various pathogens and tailor its response to specific agents is critical for the induction of innate immunity and the establishment of adaptive immune responses. After infection, the innate immune system is able to detect invading pathogens by a variety of pattern recognition receptors called Toll-like receptors (TLRs). Previous studies report the expression of TLRs 1 to 6 in human bronchial epithelium (3). A demonstration of the interaction of RSV with TLR4 was the first report of an involvement between TLRs and viruses (25). Recently, research has shown that RSV induces two peaks of NF-κB activation in vivo. Only the immediate response following inoculation required TLR4 (17). The second peak in NF-κB activation required active viral replication in epithelial cells but was independent of TLR4. Though it remains uncertain which viruses can activate TLRs through intermediates produced during viral replication, TLR3 can be used to detect double-stranded RNA (dsRNA) produced in the course of a viral infection (1, 24). In this study, we demonstrate that induction of chemokines by RSV replication is mediated by TLR3 signaling pathways.
TLR3−/− mice have been reported to have impaired responses to dsRNA and poly(I · C) (1). The cellular responses triggered by dsRNA in TLR3-expressing cells were shown to signal through both a MyD88-dependent and -independent pathway. Production of proinflammatory cytokines induced by dsRNA in TLR3-expressing cells was dependent on MyD88, whereas dendritic cell maturation and activation of NF-κB and mitogen-activated protein kinases was not impaired in MyD88−/− cells (1). It has now become increasingly clear that many of these MyD88-independent responses initiated by dsRNA are channeled through another Toll/interleukin-1 (IL-1) receptor domain containing adaptor protein, identified as TICAM-1/TRIF (37, 47, 48). Recruitment of TICAM-1/TRIF to TLR3 is required for dsRNA to activate both NF-κB and interferon (IFN) regulatory factor 3 (IRF-3) and can subsequently drive production of IFN-β and IFN response genes (IRGs; e.g., CXCL10, CCL12, and CCL5) (47). The induction of IFN-α/β can aid in host defense against many viruses; however, if not tightly regulated, dsRNA may also elevate levels of chemokines implicated in the exacerbation of disease. To identify regulatory mechanisms involved in RSV-induced airway inflammation, we examined the function of TLR3 in RSV-infected cells. In this study, we clearly show that TLR3 plays a critical role in chemokine production but has no effect on viral replication in RSV-infected cells.
MATERIALS AND METHODS
Cell culture and reagents.
Human epithelial kidney HEK 293 cells with stable TLR3 expression and vector control cells were generously provided by Xiaoxia Li (Cleveland Clinic Foundation) and propagated in Dulbecco's modified Eagle medium with 2 mM l-glutamine, 10% fetal bovine serum, and 530 μg of G418 per ml. MRC-5 and A549 cells were obtained from the American Type Culture Collection and maintained in Dulbecco's modified Eagle medium, 2 mM l-glutamine, and 10% fetal bovine serum. Poly(I · C) was purchased from Amersham Biosciences (Piscataway, N.J.). Tumor necrosis factor α was obtained from R&D Systems (Minneapolis, Minn.). Anti-IFN-α/β receptor antibody (MMHAR-2) was purchased from PBL Biomedical Laboratories (Piscataway, N.J.).
RSV propagation and titer determination.
RSV A strain was derived from a clinical isolate at the University of Michigan and was propagated in Hep2 cells. After viral adsorption, medium was added to the flask, and the infection was allowed to proceed until syncytia were observed. The cells were frozen at −80°C overnight, and the supernatant was harvested, clarified, and aliquoted. To determine viral titers, a plaque assay was performed as previously described (33).
Quantification of chemokines and TLR expression.
Protein levels of cytokines were quantitated by using a double ligand enzyme-linked immunosorbent assay (ELISA) system. Briefly, 96-well plates were coated with capture antibody, blocked with bovine serum albumin, and loaded with samples. Biotinylated detection antibody was then added, followed by incubation with streptavidin-peroxidase chromogen substrate, terminated with H2SO4, and read at 490 nm with an ELISA plate reader. RNA was isolated by using Trizol. Levels of CXCL8, CCL5, and CXCL10 were assessed by using quantitative PCR (QPCR) analysis (Taqman) with predeveloped primers and probe sets from PE Biosystems (Foster City, Calif.). The primer and probe sets used to detect RSV mRNA and TLR3 have been previously described (33, 40). QPCR analysis of MyD88 and IFN-β were performed by using SYBR green I dye. Primer sequences for IFN-β and MyD88 have been previously described (43, 49). Quantifications of the genes of interest were normalized to GAPDH (glyceraldehyde-3-phosphate dehydrogenase) and expressed as the factors of increase over the negative control for each treatment at each time point. Statistical significance was determined by analysis of variance, and significance was determined as P values of <0.05.
RNA interference (RNAi)
Small interfering RNA (siRNA) oligonucleotides for targeting endogenous TLR3 and MyD88 were purchased from Xeragon and were transfected with Lipofectamine 2000 (Invitrogen, Carlsbad, Calif.) for MRC-5 experiments or with Effectene (QIAGEN, Valencia, Calif.) for A549 experiments, per the manufacturers' instructions. The siRNA sequences used in this study were previously described (37). Briefly, 24-well plates were seeded with cells, transfected with siRNA oligonucleotides, and later infected with RSV (multiplicities of infection [MOIs] of 0.5 to 1) or treated with poly(I · C) for the indicated times.
RESULTS
TLR3 evokes chemokine production in RSV-infected cells.
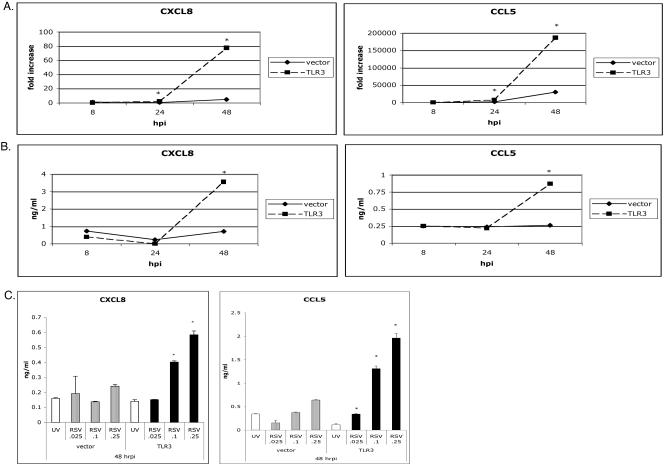
To identify the role of TLR3 in RSV-infected cells, levels of CXCL8 and CCL5 were compared in RSV-infected HEK 293 cells transfected with and stably expressing TLR3 or in an RSV-infected HEK 293 cell line transfected with a vector control. RSV-infected cells expressing TLR3 showed dramatic induction of CXCL8 and CCL5 at 48 h postinfection (hpi) compared to minimal levels detected in vector controls (Fig. 1A and B). As expected, peaks in chemokine synthesis were time dependent and were detected later than chemokines induced by poly(I · C) (data not shown).
FIG. 1.
RSV selectively upregulates chemokines in HEK 293 cells transfected with TLR3. HEK 293 cells stably transfected with TLR3 and vector controls were infected with live RSV or UV-inactivated RSV (MOI of 0.1) or incubated with media alone for 8, 24, and 48 hpi. At each time point, RNA levels were analyzed by QPCR and expressed as the increase (n-fold) relative to media controls (A), and protein levels in the supernatant were determined by ELISA (B and C). HEK 293 cells expressing TLR3 versus vector controls differed significantly in their production of CXCL8 and CCL5 message and protein at 48 hpi following RSV infection. Data are representative of three experiments, with n = 3 per treatment per experiment. Each time point represents the mean ± standard error of the mean. *, P < 0.05.
To determine if this delay was due to a requirement for sufficient amounts of replicating virus to activate TLR3, both cell lines were infected with a UV-inactivated virus and increasing amounts of RSV. As expected, UV-inactivated virus was incapable of driving chemokine production in cells regardless of TLR3 expression, whereas live RSV preferentially activated CXCL8 and CCL5 levels in a dose-dependent manner only in cells transfected with TLR3 (Fig. 1C).
TLR3 plays no role in RSV replication in epithelial cells.
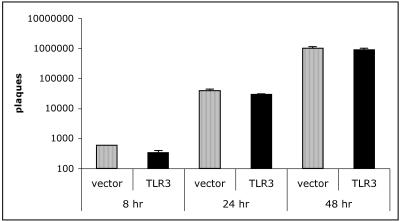
Next, we examined the effect of TLR3 expression on viral replication. Viral load was determined in RSV-infected HEK 293 cells stably expressing TLR3 versus vector controls at 8, 24, and 48 hpi (Fig. 2). RSV kinetics in both cell lines indicate that significant numbers of plaques (new virions) occur rapidly at 48 hpi, similar to peaks in chemokine induction. However, the numbers of plaques recovered between vector and TLR3-expressing cells were similar at peak viral replication. These data suggest that the presence of TLR3 has no effect on RSV replication but is utilized to drive chemokine production in RSV-infected cells.
FIG. 2.
TLR3 expression has no effect on viral replication in epithelial cells. HEK 293 TLR3 and vector control cells were infected with RSV at an MOI of 0.1. Cells were harvested at various times postinfection, and a plaque assay was performed. Data are representative of three experiments, with n = 3 per cell line per experiment.
TLR3 is essential for chemokine production in human lung cells infected with RSV.
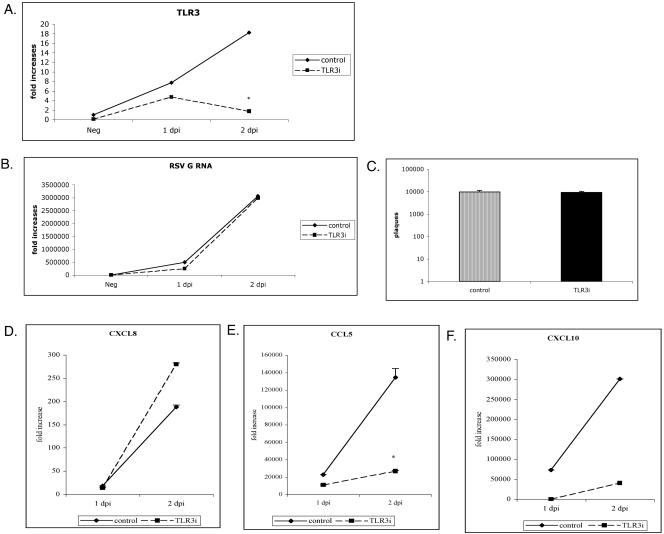
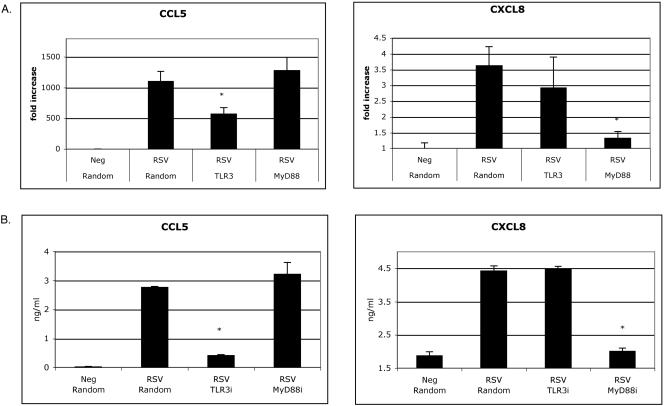
To examine the requirement of TLR3 in the production of chemokines in cells expressing physiologically normal amounts of TLR3, we used RNA interference to silence TLR3 in human lung cells infected with RSV. MRC-5 and A549 are pulmonary-derived human fibroblast and epithelial cells, respectively. They are derived from human lung tissue, express TLR3, and respond to poly(I · C) (32, 37, 44). Recently, Oshiumi et al. used siRNA to show that a reduction in TLR-3 expression in MRC-5 cells resulted in decreased levels of IFN-β when cells were stimulated with poly(I · C) (37). Similarly, when we used siRNA oligonucleotides to specifically target the expression of TLR-3 in MRC-5 cells infected with RSV, we also observed a dramatic decrease in TLR3 expression by 48 hpi (Fig. 3A). After cells were stimulated with poly(I · C) and tumor necrosis factor α, we observed only a decrease in poly(I · C)-mediated chemokine induction in cells with less TLR3, suggesting that we had specifically targeted the expression of TLR3 (data not shown). In cells transfected with random siRNA oligonucleotides and infected with RSV, TLR3 continued to be upregulated in a time-dependent manner. Suppression of TLR3 at 48 hpi had no effect on viral load since the replication curve and number of plaques recovered were similar to results for cells transfected with control siRNA (Fig. 3B and C).
FIG. 3.
TLR3 RNAi impairs CCL5 and CXCL10 production but has no effect on CXCL8 synthesis or viral replication in MRC-5 cells. TLR3 siRNA and random siRNA controls were transfected into MRC-5 cells and infected with RSV (MOIs of 0.5 to 1). Expression levels of TLR3 (A), RSV mRNA (B), and chemokines (D to F) were determined at 24 and 48 hpi. MRC-5 cells transfected with TLR3 siRNA versus random siRNA controls differed significantly in TLR3 expression and production of CCL5 at 48 hpi (*, P < 0.05). At 48 hpi, a plaque assay was performed to determine the viral load in cells with altered TLR3 expression (C). Data are representative of three experiments, with n = 3 per treatment per experiment. Each time point represents the mean ± standard error of the mean. TLR3i, TLR3 siRNA.
Since overexpression of TLR3 in RSV-infected cells induced CXCL8 and CCL5 in HEK 293 cells, we next examined the induction of chemokines in RSV-infected cells with specific RNAi-reduced TLR3 expression. As demonstrated in Fig. 3D to F, the synthesis of chemokines in human lung cells was also time dependent and reached high levels by 48 hpi. Interestingly, silencing TLR3 in MRC-5 cells had no significant effect on RSV-mediated CXCL8 production but drastically inhibited synthesis of CCL5 (Fig. 3D and E). Previous studies have suggested that several IFN-inducible genes (CCL5, CXCL10, and CCL12) are produced in response to activation of TLR3 signaling pathways (11, 47). To further distinguish induction of CXCL8 and CCL5 production in MRC-5 cells, we next assessed levels of CXCL10, another IFN-inducible gene found to be elevated in RSV-infected mice (16). Consistent with the impaired production of CCL5 in TLR3-silenced MRC-5 cells, CXCL10 production was severely inhibited in RSV-infected cells with decreased amounts of TLR3 (Fig. 3F). The expression of both CCL5 and CXCL10 paralleled TLR3 expression at 24 and 48 hpi, suggesting that TLR3 is essential for RSV-mediated CXCL10 and CCL5 production in MRC-5 cells.
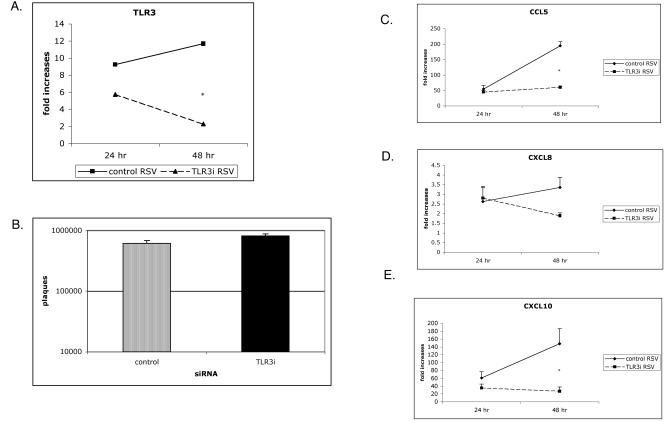
As mentioned above, the respiratory epithelium is a primary target for RSV infection and a major source for chemokine production in the lung. We next wanted to validate and extend our results in MRC-5 cells by using A549 cells. The A549 cell line retains many features of alveolar epithelium and is widely used for studying RSV-induced chemokine production. Upregulation of TLR3 by RSV in A549 cells was comparable to experiments in MRC-5 cells and could also be dramatically inhibited by using RNAi (Fig. 4A). As expected, TLR3 played no role in viral replication since similar numbers of plaques were recovered from A549 cells with altered TLR3 expression (Fig. 4B). Following RSV infection, the chemokine profile and kinetics in A549 cells were similar to those of MRC-5 cells. Also consistent with MRC-5 findings, A549 cells infected with RSV and targeted with TLR3 siRNAs had significantly reduced levels of CXCL10 and CCL5 compared to levels in controls (Fig. 4D and E). Reduction of TLR3 in RSV-infected A549 cells also impaired CXCL8 production, but these differences did not reach statistical significance (Fig. 4C). Altogether, our data indicate that TLR3 plays an essential role in driving CXCL10 and CCL5 in RSV-infected lung cells.
FIG. 4.
TLR3 RNAi impairs CCL5 and CXCL10 production but has no effect on CXCL8 synthesis or viral replication in A549 cells. TLR3 siRNA and random siRNA controls were transfected into MRC-5 cells and infected with RSV (MOI of 1). Levels of TLR3 expression (A), viral replication (B), and chemokines (C to E) were determined at 24 and 48 hpi. A549 cells transfected with TLR3 siRNA versus random siRNA controls differed significantly in TLR3 expression and synthesis of CCL5 and CXCL10 at 48 hpi (*, P < 0.05). Cells were harvested at 48 hpi postinfection, and a plaque assay was performed (B). Data are representative of three experiments, with n = 3 per treatment per experiment. Each time point represents the mean ± standard error of the mean. TLR3i, TLR3 siRNA.
Differential MyD88 requirements for RSV-induced CXCL8 and CCL5 production.
Given that cytokine production in response to poly(I · C) was previously shown to be impaired in MyD88-deficient bone marrow-derived dendritic cells, we next examined whether MyD88 participated in chemokine production in RSV-infected epithelial cells (1, 47). To examine this possibility, we compared levels of CXCL8 and CCL5 in RSV-infected A549 cells in which TLR3 and MyD88 had been targeted with siRNA. TLR3 and MyD88 mRNA levels were suppressed by siRNA up to ∼80 and ∼95%, respectively, by 48 hpi (data not shown). RSV-infected cells transfected with random siRNA induced similar expression levels of TLR3, MyD88, and chemokines compared to levels in cells transfected with reagent alone (data not shown). Our results suggest that TLR3 is critical for the upregulation of CCL5 in a signaling pathway independent of MyD88, since inhibition of MyD88 did not decrease protein or RNA levels of CCL5 (Fig. 5). Of interest, inhibition of MyD88 significantly depressed CXCL8 production independent of TLR3 (Fig. 5). We conclude that RSV triggers CCL5 in a TLR3-dependent, MyD88-independent manner, whereas CXCL8 synthesis is TLR3 independent but MyD88 dependent.
FIG. 5.
MyD88-dependent and -independent signaling linked to RSV-induced chemokine production. A549 cells were transfected with random siRNA or siRNA toward TLR3 and MyD88. Twelve hours after transfection, cells were infected with RSV at an MOI of 1. At 48 hpi, RNA and protein expression levels of chemokines were determined. Data are representative of three experiments, with n = 3 per treatment per experiment. Each time point represents the mean ± standard error of the mean. The asterisk indicates statistically significant (P < 0.05) decreases compared to RSV-infected cells transfected with random siRNA.
IFN-α/β signaling is not essential for RSV-induced chemokine production.
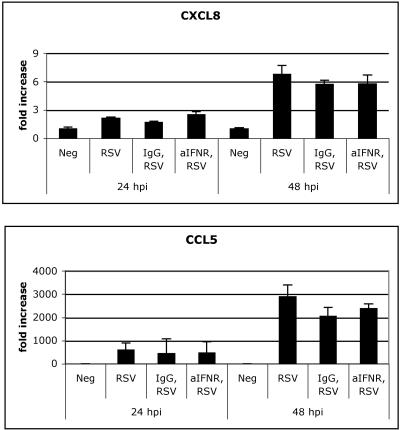
The delay in chemokine production raised the possibility that the activation of chemokines by RSV was secondary to some unidentified soluble mediator. Some IRGs can be generated secondary to the production of IFNs-α/β. RSV may also utilize TLR3 independently of MyD88 to directly activate transcription factors required for CCL5 synthesis (e.g., IRF-3 and NF-κB). A549 cells infected with RSV resulted in a ∼10- to 25-fold upregulation of IFN-β gene expression by 48 hpi, as measured by QPCR analysis (data not shown). To determine whether RSV-induced chemokine production requires IFN-α/β production, we next compared chemokine levels in A549 cells treated with anti-IFN-α/β receptor antibody or immunoglobulin G 1 h prior to RSV infection. Neutralizing the IFN-α/β receptor had no effect on CXCL8 or CCL5 levels at 24 or 48 hpi compared to results with controls (Fig. 6A). These data indicate that the release of IFNs-α/β is not essential for the induction and upregulation of chemokines triggered by RSV.
FIG. 6.
IFN-α/β signaling is not required for RSV-induced chemokine production. A549 cells were pretreated for 1 h before RSV infection with anti-IFN-α/β receptor (aIFNR) antibody (10 μg/ml), immunoglobulin G (IgG; 10 to 20 μg/ml), or medium. The antibody was left in the medium and cells were infected at an MOI of 0.5. At 48 hpi, RNA levels of CXCL8 and CCL5 were analyzed.
DISCUSSION
Previous reports have suggested an important link between RSV-induced chemokines and detrimental pathophysiology in the lung (23, 28, 33, 45). In epithelial cells, RSV activates NF-κB and drives chemokines that subsequently recruit and activate the cell populations responsible for airway inflammation (46). However, the molecular mechanisms involved in RSV-induced NF-κB activation and chemokine production are still poorly understood. In our study, we show that RSV infection triggers the activation of the TLR3 signaling pathways that regulate the expression of chemokines. HEK 293 cells preferentially activated CXCL8 and CCL5 when transfected with TLR3, and CCL5 and CXCL10 are suppressed in lung cells depleted of TLR3. These studies in cells expressing physiological amounts of TLR3 demonstrate that RSV infection initiates TLR3 signaling pathways.
Regulation of CXCL8 by TLR3 in RSV-infected cells was only observed in HEK 293 cells overexpressing TLR3 and not in RSV-infected MRC-5 or A549 cells. We attribute this disparity to differences in cell types, TLR3 expression levels, and the nature in which TLR3 is being expressed. Induction of CXCL8 and CCL5 is cell type and stimulus specific and requires a different combination of transcription factors. Pivotal roles for IRF-3 and NF-κB in CCL5 and IFN-β transcription in RSV-infected epithelial cells have been identified (5, 22). TLR3 signaling elicits the coordinate activation of ATF-2/C-Jun, IRF-3, and NF-κB downstream of TICAM-1/TRIF for the production of IFN-β (30). While there are likely numerous ways to activate NF-κB, we propose that the activation of many of the other cis-regulatory elements required for CCL5 and CXCL10 transcription (e.g., IRF-3) is initiated by TLR3 in RSV-infected cells.
Other reports have shown that the induction of IFN-β and IRGs by dsRNA occurs through an MyD88-independent pathway (37, 47). TLRs can activate distinct cellular pathways in response to various pathogen-associated molecular patterns and through the utilization of different adapter proteins. Our data suggest that RSV triggers the induction of CCL5 independently of MyD88. Surprisingly, reduction of MyD88 levels in RSV-infected cells resulted in a decline in CXCL8 synthesis. The molecular mechanisms involved in TLR3-independent, MyD88-dependent CXCL8 production can be explained by at least three possibilities. First, CXCL8 production could be secondary to a MyD88-dependent cytokine (e.g., IL-1 or IL-18), and, thus, inhibiting MyD88 expression decreased the soluble mediator driving CXCL8 production. Others have reported that the production of IL-1 by RSV triggers the induction of CXCL8 in an autocrine fashion (38). Alternatively, CXCL8 production may be driven by a different TLR that is MyD88 dependent. Along these lines, human TLR8/murine TLR7 and MyD88 facilitate the recognition of single-stranded RNA in some cell types and may be required for CXCL8 production in RSV-infected cells (6, 18). Finally, the redox pathways have been shown to regulate chemokine expression and IRF activation in RSV-infected cells and may be linked to either TLR3 or MyD88 signaling pathways (4, 26, 29).
Engagement of molecular motifs by TLRs can either directly affect the responding cells or indirectly affect cells through the release of soluble mediators. Induction of some IRGs can occur by IFN signaling through the IFN-α/β receptor and/or directly through the activation of TLR3 (19). Our IFN-α/β receptor-blocking experiments argue that the paracrine effects of IFN-α/β are not necessary for the elevation of CCL5 in RSV-infected cells. This suggests that production of CCL5 following RSV infections occurs directly through TLR3 signaling pathways. The triggering of IFNs-α/β by dsRNA suggests an important role for TLR3 in antiviral immunity. However, our data indicate that TLR3 has no effect on viral replication. Peak RSV titers were equivalent in RSV-infected HEK 293, MRC-5, and A549 cells despite altered TLR3 expression. Given that RSV is poorly sensitive to IFNs-α/β (2, 39), this is not surprising. A recent report by Durbin et al. showed that IFNs-α/β only slightly delayed RSV clearance in vivo but had a critical immunomodulatory role in the development of an appropriate Th-1-type response (8). While TLR3 may not directly inhibit RSV viral replication, its participation in viral clearance and overall pathophysiology in vivo remains to be determined.
Our studies suggest that RSV not only utilizes TLR3 to promote chemokine production but further upregulates its expression in RSV-infected cells. Following RSV infection, MyD88 was also upregulated but to a lesser extent than TLR3 levels (data not shown). It is interesting to consider how RSV may alter TLR expression in the lung during infection. Recently, RSV was shown to upregulate TLR4 and the responsiveness to lipopolysaccharide in human epithelial cells (34). Elevated levels of TLR4 expression were also detected in monocytes from infants with respiratory syncytial virus bronchiolitis (12). While it is likely that both A549 and MRC-5 cells express a repertoire of various TLRs and adapters, expression of TLR4 has only been detected in A549 cells (32, 34). Previous reports have demonstrated that mice with various genetic backgrounds respond to RSV with a spectrum of symptoms (21, 42). The idea that differences in the genes involved in chemokine production or the inflammatory response dictate the severity of RSV disease is appealing. In light of our studies, it would be particularly interesting to determine if TLR3 expression in the lung correlates with disease severity. The other pulmonary cell population that abundantly expresses TLR3 is the dendritic cell (31, 35). The participation of TLR3 in detrimental pathophysiology or viral immunity may rely on which cell population expresses the protein.
In summary, our studies indicate that TLR3 may contribute to the exacerbation of RSV in infected epithelial cells. TLR3 does not alter viral replication but is essential for induction of MyD88-independent chemokines that can lead to pathophysiological responses in pulmonary RSV infections. We speculate that the TLR3 signaling pathway may be an attractive therapeutic target for modulating RSV-induced lung inflammation.
Acknowledgments
This work was supported in part by National Institutes of Health grant AI36302.
We thank Mike Newstead (University of Michigan) for outstanding technical assistance.
REFERENCES
- 1.Alexopoulou, L., A. C. Holt, R. Medzhitov, and R. A. Flavell. 2001. Recognition of double-stranded RNA and activation of NF-κB by Toll-like receptor 3. Nature 413:732-738. [DOI] [PubMed] [Google Scholar]
- 2.Atreya, P. L., and S. Kulkarni. 1999. Respiratory syncytial virus strain A2 is resistant to the antiviral effects of type I interferons and human MxA. Virology 261:227-241. [DOI] [PubMed] [Google Scholar]
- 3.Becker, M. N., G. Diamond, M. W. Verghese, and S. H. Randell. 2000. CD14-dependent lipopolysaccharide-induced beta-defensin-2 expression in human tracheobronchial epithelium. J. Biol. Chem. 275:29731-29736. [DOI] [PubMed] [Google Scholar]
- 4.Casola, A., N. Burger, T. Liu, M. Jamaluddin, A. R. Brasier, and R. P. Garofalo. 2001. Oxidant tone regulates RANTES gene expression in airway epithelial cells infected with respiratory syncytial virus. Role in viral-induced interferon regulatory factor activation. J. Biol. Chem. 276:19715-19722. [DOI] [PubMed] [Google Scholar]
- 5.Casola, A., R. P. Garofalo, H. Haeberle, T. F. Elliott, R. Lin, M. Jamaluddin, and A. R. Brasier. 2001. Multiple cis regulatory elements control RANTES promoter activity in alveolar epithelial cells infected with respiratory syncytial virus. J. Virol. 75:6428-6439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Diebold, S. S., T. Kaisho, H. Hemmi, S. Akira, and E. S. C. Reis. 2004. Innate antiviral responses by means of TLR7-mediated recognition of single-stranded RNA. Science 303:1529-1531. [DOI] [PubMed] [Google Scholar]
- 7.Dowell, S. F., L. J. Anderson, H. E. Gary, Jr., D. D. Erdman, J. F. Plouffe, T. M. File, Jr., B. J. Marston, and R. F. Breiman. 1996. Respiratory syncytial virus is an important cause of community-acquired lower respiratory infection among hospitalized adults. J. Infect. Dis. 174:456-462. [DOI] [PubMed] [Google Scholar]
- 8.Durbin, J. E., T. R. Johnson, R. K. Durbin, S. E. Mertz, R. A. Morotti, R. S. Peebles, and B. S. Graham. 2002. The role of IFN in respiratory syncytial virus pathogenesis. J. Immunol. 168:2944-2952. [DOI] [PubMed] [Google Scholar]
- 9.Falsey, A. R., R. M. McCann, W. J. Hall, M. M. Criddle, M. A. Formica, D. Wycoff, and J. E. Kolassa. 1997. The “common cold” in frail older persons: impact of rhinovirus and coronavirus in a senior daycare center. J. Am. Geriatr. Soc. 45:706-711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fiedler, M. A., K. Wernke-Dollries, and J. M. Stark. 1996. Inhibition of viral replication reverses respiratory syncytial virus-induced NF-κB activation and interleukin-8 gene expression in A549 cells. J. Virol. 70:9079-9082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fitzgerald, K. A., S. M. McWhirter, K. L. Faia, D. C. Rowe, E. Latz, D. T. Golenbock, A. J. Coyle, S. M. Liao, and T. Maniatis. 2003. IKKε and TBK1 are essential components of the IRF3 signaling pathway. Nat. Immunol. 4:491-496. [DOI] [PubMed] [Google Scholar]
- 12.Gagro, A., M. Tominac, V. Krsulovic-Hresic, A. Bace, M. Matic, V. Drazenovic, G. Mlinaric-Galinovic, E. Kosor, K. Gotovac, I. Bolanca, S. Batinica, and S. Rabatic. 2004. Increased Toll-like receptor 4 expression in infants with respiratory syncytial virus bronchiolitis. Clin. Exp. Immunol. 135:267-272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Garofalo, R., M. Sabry, M. Jamaluddin, R. K. Yu, A. Casola, P. L. Ogra, and A. R. Brasier. 1996. Transcriptional activation of the interleukin-8 gene by respiratory syncytial virus infection in alveolar epithelial cells: nuclear translocation of the RelA transcription factor as a mechanism producing airway mucosal inflammation. J. Virol. 70:8773-8781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Garofalo, R. P., J. Patti, K. A. Hintz, V. Hill, P. L. Ogra, and R. C. Welliver. 2001. Macrophage inflammatory protein-1α (not T helper type 2 cytokines) is associated with severe forms of respiratory syncytial virus bronchiolitis. J. Infect. Dis. 184:393-399. [DOI] [PubMed] [Google Scholar]
- 15.Glezen, W. P., S. B. Greenberg, R. L. Atmar, P. A. Piedra, and R. B. Couch. 2000. Impact of respiratory virus infections on persons with chronic underlying conditions. JAMA 283:499-505. [DOI] [PubMed] [Google Scholar]
- 16.Haeberle, H. A., W. A. Kuziel, H. J. Dieterich, A. Casola, Z. Gatalica, and R. P. Garofalo. 2001. Inducible expression of inflammatory chemokines in respiratory syncytial virus-infected mice: role of MIP-1α in lung pathology. J. Virol. 75:878-890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Haeberle, H. A., R. Takizawa, A. Casola, A. R. Brasier, H. J. Dieterich, N. Van Rooijen, Z. Gatalica, and R. P. Garofalo. 2002. Respiratory syncytial virus-induced activation of nuclear factor-κB in the lung involves alveolar macrophages and toll-like receptor 4-dependent pathways. J. Infect. Dis. 186:1199-1206. [DOI] [PubMed] [Google Scholar]
- 18.Heil, F., H. Hemmi, H. Hochrein, F. Ampenberger, C. Kirschning, S. Akira, G. Lipford, H. Wagner, and S. Bauer. 2004. Species-specific recognition of single-stranded RNA via Toll-like receptor 7 and 8. Science 303:1526-1529. [DOI] [PubMed] [Google Scholar]
- 19.Hertzog, P. J., L. A. O'Neill, and J. A. Hamilton. 2003. The interferon in TLR signaling: more than just antiviral. Trends Immunol. 24:534-539. [DOI] [PubMed] [Google Scholar]
- 20.Hornsleth, A., L. Loland, and L. B. Larsen. 2001. Cytokines and chemokines in respiratory secretion and severity of disease in infants with respiratory syncytial virus (RSV) infection. J. Clin. Virol. 21:163-170. [DOI] [PubMed] [Google Scholar]
- 21.Hussell, T., A. Georgiou, T. E. Sparer, S. Matthews, P. Pala, and P. J. Openshaw. 1998. Host genetic determinants of vaccine-induced eosinophilia during respiratory syncytial virus infection. J. Immunol. 161:6215-6222. [PubMed] [Google Scholar]
- 22.Jamaluddin, M., S. Wang, R. P. Garofalo, T. Elliott, A. Casola, S. Baron, and A. R. Brasier. 2001. IFN-beta mediates coordinate expression of antigen-processing genes in RSV-infected pulmonary epithelial cells. Am. J. Physiol. Lung Cell Mol. Physiol. 280:L248-L257. [DOI] [PubMed] [Google Scholar]
- 23.John, A. E., A. A. Berlin, and N. W. Lukacs. 2003. Respiratory syncytial virus-induced CCL5/RANTES contributes to exacerbation of allergic airway inflammation. Eur. J. Immunol. 33:1677-1685. [DOI] [PubMed] [Google Scholar]
- 24.Kariko, K., H. Ni, J. Capodici, M. Lamphier, and D. Weissman. 2004. mRNA is an endogenous ligand for Toll-like receptor 3. J. Biol. Chem. 279:12542-12550. [DOI] [PubMed] [Google Scholar]
- 25.Kurt-Jones, E. A., L. Popova, L. Kwinn, L. M. Haynes, L. P. Jones, R. A. Tripp, E. E. Walsh, M. W. Freeman, D. T. Golenbock, L. J. Anderson, and R. W. Finberg. 2000. Pattern recognition receptors TLR4 and CD14 mediate response to respiratory syncytial virus. Nat. Immunol. 1:398-401. [DOI] [PubMed] [Google Scholar]
- 26.Liu, T., S. Castro, A. R. Brasier, M. Jamaluddin, R. P. Garofalo, and A. Casola. 2004. Reactive oxygen species mediate virus-induced STAT activation: role of tyrosine phosphatases. J. Biol. Chem. 279:2461-2469. [DOI] [PubMed] [Google Scholar]
- 27.Lugo, R. A., and M. C. Nahata. 1993. Pathogenesis and treatment of bronchiolitis. Clin. Pharm. 12:95-116. [PubMed] [Google Scholar]
- 28.Lukacs, N. W., K. K. Tekkanat, A. Berlin, C. M. Hogaboam, A. Miller, H. Evanoff, P. Lincoln, and H. Maassab. 2001. Respiratory syncytial virus predisposes mice to augmented allergic airway responses via IL-13-mediated mechanisms. J. Immunol. 167:1060-1065. [DOI] [PubMed] [Google Scholar]
- 29.Mastronarde, J. G., M. M. Monick, and G. W. Hunninghake. 1995. Oxidant tone regulates IL-8 production in epithelium infected with respiratory syncytial virus. Am. J. Respir. Cell Mol. Biol. 13:237-244. [DOI] [PubMed] [Google Scholar]
- 30.Matsumoto, M., K. Funami, H. Oshiumi, and T. Seya. 2004. Toll-like receptor 3: a link between Toll-like receptor, interferon and viruses. Microbiol. Immunol. 48:147-154. [DOI] [PubMed] [Google Scholar]
- 31.Matsumoto, M., K. Funami, M. Tanabe, H. Oshiumi, M. Shingai, Y. Seto, A. Yamamoto, and T. Seya. 2003. Subcellular localization of Toll-like receptor 3 in human dendritic cells. J. Immunol. 171:3154-3162. [DOI] [PubMed] [Google Scholar]
- 32.Matsumoto, M., S. Kikkawa, M. Kohase, K. Miyake, and T. Seya. 2002. Establishment of a monoclonal antibody against human Toll-like receptor 3 that blocks double-stranded RNA-mediated signaling. Biochem. Biophys. Res. Commun. 293:1364-1369. [DOI] [PubMed] [Google Scholar]
- 33.Miller, A. L., R. M. Strieter, A. D. Gruber, S. B. Ho, and N. W. Lukacs. 2003. CXCR2 regulates respiratory syncytial virus-induced airway hyperreactivity and mucus overproduction. J. Immunol. 170:3348-3356. [DOI] [PubMed] [Google Scholar]
- 34.Monick, M. M., T. O. Yarovinsky, L. S. Powers, N. S. Butler, A. B. Carter, G. Gudmundsson, and G. W. Hunninghake. 2003. Respiratory syncytial virus up-regulates TLR4 and sensitizes airway epithelial cells to endotoxin. J. Biol. Chem. 278:53035-53044. [DOI] [PubMed] [Google Scholar]
- 35.Muzio, M., D. Bosisio, N. Polentarutti, G. D'Amico, A. Stoppacciaro, R. Mancinelli, C. van't Veer, G. Penton-Rol, L. P. Ruco, P. Allavena, and A. Mantovani. 2000. Differential expression and regulation of Toll-like receptors (TLR) in human leukocytes: selective expression of TLR3 in dendritic cells. J. Immunol. 164:5998-6004. [DOI] [PubMed] [Google Scholar]
- 36.Noah, T. L., S. S. Ivins, P. Murphy, I. Kazachkova, B. Moats-Staats, and F. W. Henderson. 2002. Chemokines and inflammation in the nasal passages of infants with respiratory syncytial virus bronchiolitis. Clin. Immunol. 104:86-95. [DOI] [PubMed] [Google Scholar]
- 37.Oshiumi, H., M. Matsumoto, K. Funami, T. Akazawa, and T. Seya. 2003. TICAM-1, an adaptor molecule that participates in Toll-like receptor 3-mediated interferon-beta induction. Nat. Immunol. 4:161-167. [DOI] [PubMed] [Google Scholar]
- 38.Patel, J. A., Z. Jiang, N. Nakajima, and M. Kunimoto. 1998. Autocrine regulation of interleukin-8 by interleukin-1alpha in respiratory syncytial virus-infected pulmonary epithelial cells in vitro. Immunology 95:501-506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Roberts, N. J., Jr., J. Hiscott, and D. J. Signs. 1992. The limited role of the human interferon system response to respiratory syncytial virus challenge: analysis and comparison to influenza virus challenge. Microb. Pathog. 12:409-414. [DOI] [PubMed] [Google Scholar]
- 40.Schmidt, K. N., B. Leung, M. Kwong, K. A. Zarember, S. Satyal, T. A. Navas, F. Wang, and P. J. Godowski. 2004. APC-independent activation of NK cells by the Toll-like receptor 3 agonist double-stranded RNA. J. Immunol. 172:138-143. [DOI] [PubMed] [Google Scholar]
- 41.Shay, D. K., R. C. Holman, G. E. Roosevelt, M. J. Clarke, and L. J. Anderson. 2001. Bronchiolitis-associated mortality and estimates of respiratory syncytial virus-associated deaths among US children, 1979-1997. J. Infect. Dis. 183:16-22. [DOI] [PubMed] [Google Scholar]
- 42.Srikiatkhachorn, A., W. Chang, and T. J. Braciale. 1999. Induction of Th-1 and Th-2 responses by respiratory syncytial virus attachment glycoprotein is epitope and major histocompatibility complex independent. J. Virol. 73:6590-6597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Strahle, L., D. Garcin, P. Le Mercier, J. F. Schlaak, and D. Kolakofsky. 2003. Sendai virus targets inflammatory responses, as well as the interferon-induced antiviral state, in a multifaceted manner. J. Virol. 77:7903-7913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tanabe, M., M. Kurita-Taniguchi, K. Takeuchi, M. Takeda, M. Ayata, H. Ogura, M. Matsumoto, and T. Seya. 2003. Mechanism of up-regulation of human Toll-like receptor 3 secondary to infection of measles virus-attenuated strains. Biochem. Biophys. Res. Commun. 311:39-48. [DOI] [PubMed] [Google Scholar]
- 45.Tekkanat, K. K., H. Maassab, A. Miller, A. A. Berlin, S. L. Kunkel, and N. W. Lukacs. 2002. RANTES (CCL5) production during primary respiratory syncytial virus infection exacerbates airway disease. Eur. J. Immunol. 32:3276-3284. [DOI] [PubMed] [Google Scholar]
- 46.Tian, B., Y. Zhang, B. A. Luxon, R. P. Garofalo, A. Casola, M. Sinha, and A. R. Brasier. 2002. Identification of NF-κB-dependent gene networks in respiratory syncytial virus-infected cells. J. Virol. 76:6800-6814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yamamoto, M., S. Sato, H. Hemmi, K. Hoshino, T. Kaisho, H. Sanjo, O. Takeuchi, M. Sugiyama, M. Okabe, K. Takeda, and S. Akira. 2003. Role of adaptor TRIF in the MyD88-independent Toll-like receptor signaling pathway. Science 301:640-643. [DOI] [PubMed] [Google Scholar]
- 48.Yamamoto, M., S. Sato, K. Mori, K. Hoshino, O. Takeuchi, K. Takeda, and S. Akira. 2002. Cutting edge: a novel Toll/IL-1 receptor domain-containing adapter that preferentially activates the IFN-β promoter in the Toll-like receptor signaling. J. Immunol. 169:6668-6672. [DOI] [PubMed] [Google Scholar]
- 49.Zarember, K. A., and P. J. Godowski. 2002. Tissue expression of human Toll-like receptors and differential regulation of Toll-like receptor mRNAs in leukocytes in response to microbes, their products, and cytokines. J. Immunol. 168:554-561. [DOI] [PubMed] [Google Scholar]