Abstract
Advances in treating and preventing AIDS depend on understanding how human immunodeficiency virus (HIV) is eliminated in vivo and on the manipulation of effective immune responses to HIV. During the development of assays quantifying the elimination of fluorescent autologous cells coated with overlapping 15-mer simian immunodeficiency virus (SIV) or HIV-1 peptides, we made a remarkable observation: the reinfusion of macaque peripheral blood mononuclear cells, or even whole blood, pulsed with SIV and/or HIV peptides generated sharply enhanced SIV- and HIV-1-specific T-cell immunity. Strong, broad CD4+- and CD8+-T-cell responses could be enhanced simultaneously against peptide pools spanning 87% of all SIV- and HIV-1-expressed proteins—highly desirable characteristics of HIV-specific immunity. De novo hepatitis C virus-specific CD4+- and CD8+-T-cell responses were generated in macaques by the same method. This simple technique holds promise for the immunotherapy of HIV and other chronic viral infections.
Control of persistent intracellular pathogens such as human immunodeficiency virus type 1 (HIV-1) requires cell-mediated immunity. CD8+ cytotoxic T lymphocytes (CTL) eliminate virus-infected cells by recognizing and lysing cells presenting viral peptides on cell surface major histocompatibility complex (MHC) class I molecules. HIV-1-specific CD8+-T-cell responses temporally correlate with reduced viremia during acute infection prior to the appearance of virus-specific antibodies (6, 20). Depletion of CD8+ T cells in macaques chronically infected with simian immunodeficiency virus (SIV) results in brisk rises in viremia (31). CD4+-T-helper-cell responses are required to maintain effective virus-specific CD8+-T-cell immunity. The generation of T-cell immune responses correlates with the control of viremia following challenge of macaques with chimeric SIV/HIV-1 (5, 22).
Measuring broad functional T-cell immunity in outbred subjects of unknown MHC genotype generally relies on the detection of antigen-specific gamma interferon (IFN-γ) production from T cells (17). In addition to secreting IFN-γ, an important cytokine mediator of cell-mediated immunity, T cells undergo proliferation and migration and release other effector molecules that facilitate their critical function during a viral infection: clearance of virus-infected cells in vivo. Previous murine studies have utilized fluorescent syngeneic splenocytes pulsed with known T-cell epitopes to sensitively measure virus-specific elimination in vivo (1, 7, 12, 25). No assessment of in vivo T-cell recognition and clearance of SIV/HIV-1 in nonhuman primates has previously been undertaken.
There is an urgent need to induce or enhance HIV-specific T-cell immunity to prevent or control HIV disease. Safely inducing SIV- or HIV-1-specific CD4+ and CD8+ T cells in outbred nonhuman primates or in humans by vaccination generally requires DNA and/or live vaccine vectors that express foreign antigens within cells (2, 5, 19, 33). Peptide or protein approaches to induce CD8+-T-cell responses in primates have previously required ex vivo expansion of sterile antigen-presenting cell fractions such as monocyte-derived dendritic cells (26, 34). Although monocyte-derived dendritic cells loaded with inactivated SIV virions show promise for the control of SIV infection in macaques, such techniques will not be widely applicable since prolonged sterile culture is required (23). More feasible immunotherapy methods are needed.
In this study, we describe a novel in vivo killing (IVK) assay using overlapping 15-mer peptide pools pulsed onto autologous fluorescently labeled peripheral blood mononuclear cells (PBMC) to measure SIV/HIV-1 vaccine-specific T-cell immunity in outbred pigtail macaques. When we analyzed SIV/HIV-1-specific immunity by standard techniques in the weeks following the IVK assays, a marked enhancement of virus-specific CD8+- and CD4+-T-cell immunity was induced.
MATERIALS AND METHODS
Animals.
Colony-bred simian retrovirus type D-negative pigtail macaques (Macaca nemestrina, aged 2 to 4 years) were studied. Animals were anesthetized with ketamine (10 mg/kg of body weight, intramuscularly) prior to all procedures. Conditions and protocols were approved by a University of Melbourne animal ethics committee.
Preimmunizations.
T-cell responses were induced in macaques by administering 1-mg doses of a DNA vaccine expressing HIV-1 or SIV Gag and Pol genes twice followed by a single 5 × 107 PFU fowlpox virus (FPV) vaccine boost expressing similar HIV-1/SIV genes intramuscularly at 4-weekly intervals as previously described (8, 19).
Overlapping peptide pools.
Next, 15-mer peptides (>80% purity) overlapping by 11 amino acids spanning the entire Gag (125 peptides), Pol (263 peptides), and Nef (21 peptides) proteins of SIVmac239; Env (212 peptides) protein of HIV-1MN; and Tat (23 peptides), Rev (21 peptides), and Vpu (19 peptides) proteins of HIV-1 consensus subtype B were pooled for each protein by solubilizing each 1 mg of peptide in 10 to 40 μl of dimethyl sulfoxide (DMSO; Sigma, St. Louis, Mo.). Following that, 18-mer peptides (>80% purity) overlapping by 11 amino acids encoding the entire protein length of hepatitis C virus (HCV) type 1a H77 were solubilized similarly into two pools of structural (peptides 1 through 116, encoding Core, E1, E2, and p7) and nonstructural (peptides 117 through 441, encoding NS2, NS3, NS4a, NS4b, NS5a, and NS5b) proteins. All peptides were kindly provided by the AIDS Research and Reference Reagent Program, Division of AIDS, National Institute of Allergy and Infectious Diseases, National Institutes of Health (NIH).
IVK assays.
PBMC were isolated over Ficoll-Hypaque from 40 to 50 ml of blood. A total of 25 ml of sterile injectable saline was infused into the animals immediately after blood sampling. PBMC were resuspended in phosphate-buffered saline and divided into 0.5 ml containing 1 × 107 to 2 × 107 PBMC two or three times. Cells were pulsed with peptide pools (10 μg/ml) or an equivalent volume of DMSO for 60 to 90 min in a 37°C water bath with regular mixing. Each population was labeled with either 5-(and-6)-carboxyfluorescein diacetate, succinimidyl ester (CFSElo 0.5 μM, CFSEhi 5 μM; Molecular Probes, Eugene, Ore.) or 5-(and-6)-chloromethyl SNARF-1-acetate (SNARF; 5 μM; Molecular Probes). Cells were mixed thoroughly and stained for 10 min at 37°C and washed twice. All peptide-pulsed labeled cells for each animal were pooled in saline and reinfused into the femoral vein on the same day. Prior to infusion, blood was sampled to confirm the absence and/or account for the presence of previously fluorescent-labeled cells in peripheral blood.
To quantitate the selective killing of peptide-pulsed cells in vivo, 3 ml of blood was sampled from the opposite femoral vein 5 min following infusion (to confirm equal numbers of recirculating labeled cells) and at 4 h and/or 16 to 20 h afterward. Red blood cells were lysed with fluorescence-activated cell sorter (FACS) lysing solution (Becton Dickinson, San Diego, Calif.), and 106 gated lymphocytes were analyzed by flow cytometry (FACSort, CellQuest software; Becton Dickinson) for the presence of CFSE- or SNARF-labeled circulating cells. Lymph node mononuclear cells were studied following biopsy and isolation of cells 36 h following PBMC infusion. Killing of peptide-pulsed labeled cells was expressed as the proportion or percentage of target versus control-labeled cells relative to events analyzed 5 min following infusion.
For later experiments using peptide-pulsed autologous whole blood to stimulate immune responses, 9 ml of acid citrate dextrose-anticoagulated blood was collected, centrifuged to remove 3 to 4 ml of plasma, and pulsed with overlapping peptide pools (5 μg/peptide/ml) for 1 h at 37°C prior to intravenous reinfusion.
IFN-γ ELISPOT assay.
Enumeration of antigen-specific IFN-γ-secreting cells was assessed using a monkey IFN-γ enzyme-linked immunospot (ELISPOT) commercial kit (U-CyTech, Utrecht, The Netherlands) as previously described (10). All assays were performed in duplicate on fresh PBMC. Briefly, PBMC were stimulated with SIV or HIV-1 peptide pools (solubilized in DMSO at 1 μg/ml) or control wells with DMSO or with whole inactivated SIV or control microvesicles (5 μg/ml; kindly provided by Jeff Lifson, AIDS Vaccine Program, National Cancer Institute, Bethesda, Md.) for 18 h and then transferred to anti-IFN-γ monoclonal antibody-coated flat-bottomed 96-well plates and restimulated with antigen for 5 h. Positive control wells were stimulated with Staphylococcus enterotoxin B (Sigma). Cells were lysed, and wells were incubated with biotinylated anti-IFN-γ polyclonal rabbit antibody, followed by incubation with a gold-labeled antibiotin immunoglobulin G antibody. IFN-γ spots were developed and counted on an automated reader (AID, Strassberg, Germany). Results were normalized to antigen-specific IFN-γ-secreting precursor frequency per 106 PBMC, with positive results identified as >50 spot-forming cells/106 PBMC and three or more times the background (30).
IFN-γ intracellular cytokine staining (ICS).
Phenotyping and quantitation of antigen-specific intracellular IFN-γ expression in CD8+ or CD4+ T cells was assessed by flow cytometry as previously described (10, 24). Briefly, whole blood was stimulated with SIV, HIV-1, or HCV peptides (1 μg/ml) with costimulatory antibodies to human CD28 and CD49d (1 μg/ml; Becton Dickinson) for 7 h. Brefeldin A (10 μg/ml; Sigma) was added during the last 5 h of stimulation. T cells were phenotyped with anti-CD3-phycoerythrin, anti-CD4-fluorescein isothiocyanate, and anti-CD8-PerCP (SP34, L200, and Leu-2a, respectively; Becton Dickinson) by incubation for 20 to 30 min. Red blood cells were lysed, and the remaining cells were permeabilized with FACS Permeabilizing Solution 2 (Becton Dickinson), followed by incubation with anti-human IFN-γ-allophycocyanin antibody (B7; Becton Dickinson). A total of 105 gated lymphocytes were analyzed by flow cytometry. The percentage of antigen-specific gated cells expressing IFN-γ was assessed in both the CD3+ CD4+- and CD3+ CD8+-lymphocyte subsets.
HIV-1/SHIV infection of macaques.
Macaques were infected with either (i) nonpathogenic HIV-1LAI (106 50% tissue culture infective doses [TCID50]) intravenously as previously described (9, 19), (ii) SHIVmn229 intrarectally in two 0.5-ml doses over 2 days (total, 105 TCID50) as previously described (8, 10), or (iii) SHIVSF162P3 atraumatically, intravaginally, in 1 ml twice over 2 days (total, 6 × 103 TCID50). SHIVSF162P3 was obtained from the NIH AIDS Research and Reference Reagent Program and expanded on human PBMC as described previously (15, 16). SHIV viremia was quantified by reverse transcriptase-real-time PCR, and depletion of peripheral CD4+ T cells by flow cytometry was quantified as previously described (10).
In vivo CD8 depletion.
To phenotype cells mediating the in vivo elimination of SHIV peptide-coated cells, the anti-CD8 cM-T807 monoclonal antibody (provided as a generous gift by Keith A. Reimann, Harvard Medical Center, Boston, Mass., obtained from the National Cell Culture Center) was administered subcutaneously (10 mg/kg) on day 0 and intravenously (5 mg/kg) on days 3 and 7 as previously described (31). CD8 T-cell depletion was confirmed and monitored by whole-blood staining with anti-CD8-fluorescein isothiocyanate (DK25; Dako Inc., Carpentaria, Calif.) and anti-CD3-phycoerythrin antibodies as previously described (10). Lack of binding interference between the two CD8 antibodies (cM-T807 and the DK25) was confirmed in vitro on pigtail PBMC (data not shown).
RESULTS
In vivo CTL killing.
Initial experiments studied pigtail macaques (M. nemestrina) vaccinated with HIV-1 Gag-encoding DNA and attenuated FPV and subsequently challenged with nonpathogenic HIV-1LAI. We and others have shown by ex vivo assays that this general immunization and challenge method generates high-level T-cell immunity in macaques (2, 8, 9, 14, 19, 27).
Virus-specific CTL effector activity is generally measured by the standard ex vivo chromium release assay, an insensitive, indirect measurement of the ability of T cells to clear virus in vivo. By infusing fluorescent autologous macaque PBMC pulsed with large pools of SIV and HIV-1 overlapping peptides, we were able to directly measure the clearance of HIV-1- or SHIV-peptide-expressing cells in vivo.
Equivalent numbers of circulating fluorescent cells were detected by flow cytometry 5 min after intravenous infusion (prior to potential in vivo elimination) (Fig. 1A). In an animal previously primed for HIV-1-specific immunity by sequential DNA and FPV vaccination, a 96% elimination of Gag-peptide-pulsed cells, in comparison to non-peptide-pulsed cells, was detected the following day—direct evidence of the elimination of Gag-expressing cells. In six animals not administered DNA and FPV vaccines, there was no significant killing of Gag-pulsed cells (data not shown).
FIG. 1.
IVK of HIV-1 Gag-pulsed cells. Equal numbers of autologous PBMC pulsed either with a pool of overlapping HIV-1 Gag peptides (SNARF, red) or without Gag peptides (CFSE, green; control population) were infused intravenously. Circulating labeled cell numbers in the peripheral blood were identified by flow cytometry at various time points thereafter and are expressed as values relative to those of the control population. (A) Similar numbers of circulating Gag-pulsed and control cells are identified 5 min following infusion but selective depletion of the Gag-pulsed population occurs 20 h following infusion in an HIV-1 immune macaque. (B) Lymph node mononuclear cells examined for the loss of Gag-pulsed cells 36 h following infusion in an unimmunized macaque and a preimmune macaque are shown.
The recirculating cells detected in blood the day following PBMC infusion were predominantly lymphocytes (data not shown), leading us to examine whether elimination of HIV-1-peptide-pulsed cells would also be detected in tissues such as lymph nodes, a site of high-level HIV-1 replication in humans. Elimination of Gag-peptide-labeled cells was observed in an inguinal lymph node cell suspension obtained from an HIV-1-vaccinated and -infected macaque, but not in a nonimmune macaque, 36 h following infusion of labeled PBMC (Fig. 1B).
To examine the virus-specific cytotoxicity generated by DNA and FPV vaccines during a virulent lentivirus infection, we studied vaccinated animals subsequently challenged with SHIVmn229, a highly pathogenic chimeric HIV-1IIIB/SIVmac239 virus (8, 10). T-cell immunity was again induced by immunization of animals with DNA and FPV vaccines expressing the shared SIVmac239 gag and pol genes (8). Elimination of both Gag- and Pol-peptide-labeled cells was examined by infusing 3 populations of autologous cells: one labeled with SNARF (5 μM, pulsed with SIV Pol peptides), one labeled with a low concentration of CFSE (0.5 μM, pulsed with SIV Gag peptides), and a control population labeled with a high concentration of CFSE (5 μM, pulsed with either DMSO alone or SIV Nef peptides, an irrelevant peptide pool not expressed by the vaccines).
Immediately following infusion, all three labeled, equivalent-cell populations were distinctly identified (Fig. 2A). In a macaque (macaque H20) previously immunized with DNA and FPV vaccines encoding SIV gag and pol, elimination of Gag-pulsed cells (27%) was detected as early as 4 h after infusion (Fig. 2A). By 16 h, 76% elimination of Gag-pulsed cells was detected in this macaque. No IVK of Pol-labeled targets was observed in vaccinated macaques. No killing of Gag- or Pol-pulsed cells was detected in unvaccinated macaques.
FIG. 2.
In vivo SHIV-specific killing after immunization and pathogenic SHIV infection. Flow cytometric analysis of macaque peripheral blood following infusion of PBMC pulsed with sets of overlapping SIV peptides: Pol (SNARF, red); Gag (CFSElo, green), and Nef or DMSO (CFSEhi, green; control population). Cell numbers are expressed as values relative to those of the control population. (A) Progressive elimination of Gag-pulsed cells in a DNA- and FPV-immunized animal was detected 4 and 16 h following infusion; no Gag or Pol-specific cytotoxicity was detected in an unimmunized naïve animal. (B) At 14 days after SHIV challenge, near-complete elimination of Gag- and Pol-pulsed cells in a previously immunized animal occurred.
We then studied the ability of vaccinated animals to eliminate Gag- or Pol-pulsed cells 2 weeks following acute infection with SHIVmn229, a time of high-level acute viremia and rapid mobilization of SHIV-specific immunity (4, 5, 20, 29, 31, 32). In comparison to killing levels following vaccination alone, a dramatic elevation of killing of both Gag- and Pol-pulsed cells was observed following SHIV challenge (Fig. 2B). Four macaques previously immunized with DNA and FPV vaccines exhibited high degrees of Gag-specific killing (65 to 98%), and two of the four macaques (H20 and H21) further eliminated Pol-labeled cells with >99% efficiency (Table 1). Interestingly, one animal not previously vaccinated with DNA and FPV vaccines but previously studied for IVK responses by using peptide-pulsed cells, macaque E22, was also able to clear 91 and 32% of Gag- and Pol-pulsed PBMC, respectively, 2 weeks after SHIV exposure. The levels of killing of Gag- and Pol-peptide-pulsed cells correlated with the levels of IFN-γ expression by ELISPOT and ICS of CD8+ and CD4+ T cells (Table 1).
TABLE 1.
IVK of Gag- and Pol-pulsed cells after SHIV challenge and correlation with IFN-γ ELISPOT and ICS
| Macaque | % IVKa |
Spot-forming cells/106 PBMC by IFN-γ ELISPOT |
IFN-γ ICS |
|||||
|---|---|---|---|---|---|---|---|---|
| Gag | Pol | Gag | Pol | Gag | Pol | Gag | Pol | |
| E20b | 3.8 | 5.7 | 8 | 0 | 0.0 | 0.0 | 0.0 | 0.0 |
| E22b | 91.3 | 31.9 | 73 | 13 | 1.7 | 1.6 | 3.3 | 0.2 |
| B00c | 64.8 | 22.6 | 73 | 0 | 3.3 | 0.4 | 0.4 | 0.2 |
| H8c | 85.0 | 12.7 | 593 | 8 | 13.7 | 0.2 | 27.0 | 0.2 |
| H20c | 92.3 | 99.8 | 230 | 20 | 3.4 | 2.1 | 5.2 | 3.0 |
| H21c | 98.3 | 99.2 | 183 | 65 | 4.7 | 4.3 | 8.8 | 5.0 |
IVK experiments were performed 2 weeks after SHIVmn229 challenge. IFN-γ ELISPOT and ICS data were from 2 and 3 weeks postchallenge, respectively.
Macaques were not vaccinated with DNA and FPV but previously studied for IVK.
Macaques previously vaccinated with DNA and FPV and studied for IVK.
IVK during CD8 depletion.
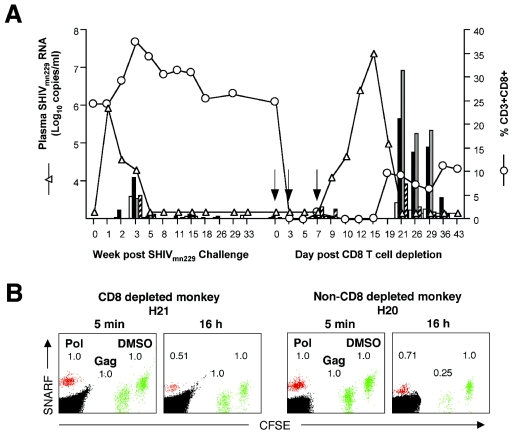
The phenotype of cells mediating in vivo cytotoxicity was addressed by analyzing the killing of Gag- and Pol-peptide-pulsed cells 9 days following parenteral administration of a CD8-depleting monoclonal antibody to a DNA- and FPV-vaccinated macaque (H21) with controlled SHIV viremia (Fig. 3A). CD8 depletion was successful, and a surge in SHIV viremia ensued. CD8 depletion eliminated all killing of Gag-expressing cells. In a similarly immunized and infected macaque (H20) studied concurrently without CD8 depletion, Gag-specific killing activity was maintained (Fig. 3B). Interestingly, CD8 depletion did not eliminate killing of Pol-labeled cells, suggesting either that any remaining tissue CD8+ cells were still functionally effective or that CD4-mediated cytotoxicity may have contributed to the clearance of Pol-labeled cells (3, 18, 29).
FIG. 3.
IVK mediated by CD8+ cells. (A) Depletion of CD8+ T cells (○) following administration of a CD8 depleting monoclonal antibody over 7 days (arrows) in immunized and SHIVmn229-challenged macaque H21 led to a brisk rise in SHIV viremia (▵). Repletion of CD8+ T cells correlated temporally with recontrol of viremia, in association with strong T-cell immunity as measured by ICS (bars) to Gag (CD4+ [□] and CD8+ [▪]) and Pol (CD4+ [░⃞] and CD8+ [hatched]). No Pol responses were assayed between weeks 18 and 33 post-SHIV challenge, and no Gag or Pol responses were assayed at week 8 post-SHIV challenge and between days 10 and 19 post-CD8+-T-cell depletion. (B) Flow cytometric analysis of IVK of infused PBMC pulsed with SIV peptides: Pol (SNARF, red); Gag (CFSElo, green), and DMSO (CFSEhi, green; control population) was performed on macaque H21 on day 9 of CD8 depletion. A similarly immunized and SHIVmn229-challenged but non-CD8-depleted macaque, H20, was assayed concurrently. Cell numbers are expressed as values relative to those of the control population.
When Gag- and Pol-specific T-cell immunity was measured by IFN-γ ICS during recontrol of SHIV viremia, a very large increase of both CD4+- and CD8+-specific immunity was detected (Fig. 3A). Up to 35% of all CD4+ T cells and 29% of the restoring CD8+ T cells were specific for Pol and Gag during recontrol of SHIV viremia.
Immunogenicity of peptide-pulsed autologous cells.
We have previously shown that our DNA and FPV vaccines express Pol at low levels and do not induce the strong Pol-specific immune responses measured by ELISPOT and ICS (8), which correlated with the lack of Pol-specific killing observed by IVK following vaccination alone shown above. Interestingly, the three animals that generated very high levels of Pol-specific killing (H20, H21, and E22) early after SHIV challenge were the only animals that received an infusion of Pol-pulsed PBMC twice previously. We reasoned that immune recognition of the infused peptide-pulsed PBMC could stimulate or boost T-cell immunity. SHIV-specific immunity was therefore studied before and after cell infusion, first by measuring SHIV-specific IFN-γ secretion by ELISPOT assays (Fig. 4A).
FIG. 4.
T-cell immunity measured by IFN-γ ELISPOT. (A) IFN-γ spot-forming cells responding to SIV Gag, Pol, and DMSO (control antigen) antigens in each well from naïve and SHIV-immunized macaques before and after infusion of Gag- and Pol-peptide-pulsed autologous PBMC. Numbers of positive spots in actual ELISPOT wells are shown. (B) T cells secreting IFN-γ in response to either SIV Gag peptides or whole inactivated SIV prior to (□) and 1 week following (▪) infusion of peptide-pulsed autologous cells in two unimmunized macaques (E20 and E22) and four macaques previously immunized with DNA and FPV vaccines (B00, H8, H20, and H21).
In macaques previously primed with DNA and FPV vaccinations, a mean fourfold increase in IFN-γ-secreting cells to the SIV Gag peptide pool was detected 1 week following the infusion of Gag-peptide-pulsed cells (Fig. 4B). This result was confirmed using a separate antigen, whole inactivated SIV (8.5-fold mean increase), demonstrating that induced responses could recognize processed whole virions. The animals that received Pol-peptide-pulsed cells at week 10 of vaccination (vaccinated animals H20 and H21 and unvaccinated animal E22) previously had no significant (<20 spots/106 PBMC) responses to Pol but generated 55 to 158 Pol-specific IFN-γ-secreting cells/106 PBMC following infusion. Six macaques immunized concurrently with the same DNA and FPV vaccines but not receiving peptide-pulsed cell infusion had mean 44, 40, and 9% decreases in IFN-γ-secreting cells specific for Gag, Pol, and whole inactivated SIV, respectively, at similar time points (8). These results demonstrate that the infusion of Gag- and Pol-peptide-pulsed autologous cells was immunogenic in the animals previously primed for SIV Gag and Pol responses.
To our surprise, T-cell immune responses to SIV Pol were also detected in a naïve animal (E22) not previously exposed to any vaccinations or SHIV infection following infusions of peptide-pulsed PBMC. This suggested that priming of responses can also be induced with this method.
Induction of CD4+- and CD8+-T-cell immunity.
We next studied whether the infusion of peptide-pulsed autologous cells could enhance both CD4+- and CD8+-T-cell immune responses by ICS analysis in SHIV-infected macaques. Two macaques with vaccine-induced partial control of SHIVmn229 37 weeks following challenge were restudied. Nonfluorescent PBMC pulsed with Gag and Pol peptides were infused, and SHIV-specific immunity was subsequently analyzed by ICS (Fig. 5A). A brisk rise in both CD4+ and CD8+ SHIV-specific T lymphocytes was observed following infusion with a mean peak increase of 8.5% Gag-specific and 1.3% Pol-specific CD4+ T lymphocytes (Fig. 5B). A mean peak increase of 1.1% Gag-specific and 0.27% Pol-specific CD8+ T lymphocytes was also observed following infusion.
FIG. 5.
Induction of CD4+- and CD8+-T-cell immunity. SHIV-specific T-cell responses measured by IFN-γ ICS after infusion of SIV Gag- and Pol-peptide-pulsed PBMC and whole blood is shown. (A) Flow cytometric analysis of increased CD8+ and CD8− (CD4+) SIV Gag-specific T cells before (day 0) and after (day 17) infusion of peptide-pulsed PBMC in a previously immunized and SHIV-infected macaque. Percents CD3+ CD8+ and CD3+ CD4+ SIV Gag-specific lymphocytes are indicated in upper right and left quadrants, respectively. (B) Kinetic analysis of CD4+- and CD8+-T-cell responses to SIV Gag and Pol following infusion of Gag- and Pol-peptide-pulsed PBMC into two macaques previously immunized and challenged with SHIVmn229 (H8 [▪] and H20 [♦]). (C) Analysis of T-cell responses following infusion of Gag- and Pol-peptide-pulsed whole blood in three macaques previously infected with SHIVsf162P3 (9E [▪], 23E [♦], and 24E [•]).
We then addressed whether the simpler procedure of infusing peptide-pulsed fresh anticoagulated whole blood (rather than Ficoll-prepared PBMC) could similarly boost SHIV-specific CD4+- and CD8+-T-cell immunity. For this experiment, we studied three nonvaccinated macaques infected intravaginally with SHIVsf162P3 who all subsequently had SHIV plasma RNA controlled to undetectable levels by 6 months following infection. Peptide-pulsed whole blood was reinfused 56 weeks postinfection. All three animals had sustained rises in Gag- and Pol-specific CD4+-T-cell responses (means, 1.1 and 0.1%, respectively) and more transient rises in Gag- and Pol-specific CD8+-T-cell responses (means, 1.0 and 0.3%, respectively) following a single infusion of peptide-pulsed whole blood (Fig. 5C).
Multiple infusions of peptide-pulsed whole blood in SHIV viremic animals.
These initial results suggest that multiple infusions of overlapping peptide-pulsed autologous cells might contribute to a greater rise in T-cell immunity. Further, since both Gag- and Pol-specific responses could be induced, we postulated that broad responses to additional structural and regulatory proteins could also be simultaneously elicited.
We then infused an animal chronically infected with SHIVmn229 for 47 weeks with sustained but low-level viremia (3.9 log10 RNA copies/ml) with fresh autologous SHIV-peptide-pulsed whole blood three times. Separate 9-ml aliquots of blood were pulsed with 15-mer peptide pools spanning (i) SIV Gag, (ii) SIV Pol, and (iii) HIV-1 Env, Tat, Rev, Vpu, and SIV Nef proteins (covering >87% of all SHIV proteins). A sequential boosting of both CD4+- and CD8+-T-cell immunity to all of the peptide pools infused was detected by ICS (Fig. 6A), with higher SHIV-specific responses consistently detected in the CD4+-T-cell subset. The immunogenicity was greatest for Gag, Pol, and Env proteins (each ≥125 peptides/pool) and less for the regulatory proteins Rev, Tat, Vpu, and Nef (84 peptides), which may reflect the smaller number of epitopes in this pool recognized by this animal.
FIG. 6.
T-cell immunity to induced SHIV and HCV. Multiple infusions of SHIV- or HCV-peptide-pulsed whole blood were administered to chronically SHIVmn229-infected macaques. (A) Increased SIV Gag, Pol, HIV-1 Env, and HIV-1 Tat-Rev-Vpu-SIV-Nef-specific CD4+ (•)- and CD8+ (□)-T-cell responses by ICS following three monthly infusions of SHIV peptide-pulsed whole blood (arrows) in a macaque with partial control of SHIVmn229 viremia. (B) Correlation between baseline plasma SHIV viral load (prior to infusion) and induction of total SHIV-specific CD4+-T-cell responses following SHIV-peptide-pulsed whole-blood infusion in five macaques previously infected with SHIVmn229. (C) Induction of CD4+ (•)- and CD8+ (□)-T-cell responses to both structural and nonstructural HCV proteins following three monthly infusions of HCV-peptide-pulsed whole blood (arrows) in a macaque with controlled SHIVmn229 viremia.
We studied this procedure in four additional animals chronically infected with SHIVmn229 with higher levels of baseline viremia (4.6 to 6.2 log10 RNA copies/ml). Although immune responses could be generated in four of the five animals studied, total SHIV-specific immunity was significantly inversely proportional to the level of viremia present (r2 = 1.0, P < 0.001 by the Spearman nonparametric rank test) (Fig. 6B). The macaque with the highest SHIV plasma RNA level (6.2 log10 copies/ml) had no increase in SHIV-specific CD4+ T cells. Macaques with 4.6, 5.1, and 5.5 log10 copies of baseline SHIV viral load/ml had 2.43, 0.83, and 0.44% increases in SHIV-specific CD4+ T cells. The animal with the lowest baseline HIV viral load (3.9 log10 copies/ml) had the greatest rise in SHIV-specific CD4+ T cells (6.87%). These results suggest it will be more difficult to generate effective immunity in the presence of high levels of viremia.
Induction of HCV-specific T-cell immunity with peptide-pulsed whole blood.
Last, we asked whether de novo T-cell immunity to HCV, another important chronic viral pathogen, could be generated utilizing peptide-pulsed whole blood. A macaque chronically infected with SHIVmn229 with controlled SHIV viremia prior to infusion (<3.2 log10 RNA copies/ml) was administered three doses of fresh autologous whole blood pulsed with two HCV 18-mer peptide pools spanning (i) structural proteins Core, E1, E2, and p7 (116 total peptides) and (ii) nonstructural proteins NS2-5b (325 total peptides). Induction of new CD4+- and CD8+-T-cell responses to both structural and nonstructural HCV proteins was detected (Fig. 6C). HCV-specific responses were highest in the CD4+-T-cell subset and were greater for the structural HCV peptide pool than those for the larger nonstructural peptide pool. This may be due to a greater number of epitopes present in the structural pool or the potential for antigenic competition within the very large nonstructural peptide pool. These results should be interpreted cautiously, as only one macaque with a low SHIV viral load and thought likely to respond to the peptide-pulsed cell immunizations was available to study.
DISCUSSION
This series of macaque studies had three important findings. First, we found that an IVK assay can be utilized in outbred macaques to directly measure CTL responses by clearance of HIV-1- or SIV-peptide-pulsed fluorescent autologous cells. Soon after SHIV challenge, near-complete clearance of Gag- and Pol-labeled cells was demonstrated in previously vaccinated macaques. Interestingly, despite such very high levels of virus elimination, latent lentiviral infection is still established in macaques challenged with SHIV. Taken together with the observed recrudescence of viremia following CD8 depletion, these findings illustrate the potential difficulties of the long-term control of primate lentiviral infection by T-cell responses alone.
Second, we demonstrated that infusion of autologous PBMC or whole blood pulsed for 1 h with large sets of overlapping SHIV 15-mer peptides boosts broadly reactive SHIV-specific immunity in outbred macaques primed for SHIV-specific immunity by vaccination or SHIV infection. Both CD4+ and CD8+ SHIV-specific T-cell immune responses were highly elevated following the infusion of peptide-pulsed cells, even in SHIV-infected animals. These findings were repeated in four separate macaque experiments involving 16 animals. Not surprisingly, SHIV viremia levels inversely correlated with the ability to boost SHIV-specific T-cell responses, providing parameters with which to approach human studies.
Third, we demonstrated that autologous PBMC or whole blood pulsed with either 15-mer SHIV or 18-mer HCV peptides can, at least in the small numbers of animals studied in this report, prime broad de novo CD4+- and CD8+-T-cell responses in immunocompetent macaques. Taken together, these studies suggest that the use of a relatively simple and safe procedure of pulsing whole blood for 1 h with pools of overlapping peptides spanning whole proteins or indeed the entire protein sequence of a pathogen, is a useful immunotherapy technology. Large numbers of fresh antigen-loaded cells are delivered widely to immune compartments. Minimal or no antigen processing is required, circumventing many virus-specific strategies to avoid effective antigen presentation. The short incubation period, lack of requirement to isolate cell subpopulations and/or extensive ex vivo culture (23), and lack of requirement to define the MHC genotypes of the host or epitope specificity of the pathogen are significant, practical advantages of this immunotherapy approach. We have termed this immunotherapy technology overlapping peptide-pulsed autologous cells.
Although HIV-1- or HCV-specific CD4+- and CD8+-T-cell responses assist the control of these chronic virus infections, assessing the utility of manipulating T-cell immunity by infusing overlapping-peptide-pulsed cells for controlling viral replication requires larger nonhuman primate or human studies. Many of the animals studied to date have had low or undetectable viral loads, with no power to detect significant reduction in viremia. The animals studied with high viral loads have had poor induction of immunity, reflecting their immunodeficiency and consistency with other vaccine studies in SIV/HIV-infected monkeys or HIV-1-infected humans (13). It was of interest that early, complete control of SHIV viremia was observed in one DNA- and FPV-vaccinated animal (H21) boosted twice with Gag- and Pol-labeled cells. The control of viremia in this animal was mediated primarily by CD8+ T cells, rather than neutralizing antibodies (2), since high-level SHIV viremia reemerged promptly following CD8 depletion. Recontrol of viremia was associated with CD8+-T-cell repletion and dramatically high levels of both CD4+ and CD8+ SHIV-specific T-cell immunity.
Several additional studies are immediately suggested by these results. First, we are endeavoring to adapt the IVK assay to measure the clearance of SHIV-infected, rather than SHIV-peptide-pulsed, cells. Although technically challenging, such studies could dissect additional important roles of non-CD4 and -CD8 T-cell components, such as natural killer cells, natural killer T cells, neutrophils, and antibody in the clearance of SHIV-infected cells. Second, it was of interest that a higher proportion of CD4+ T cells than that of CD8+ T cells was stimulated by this immunotherapy technique using 15- to 18-mer overlapping peptides. The preponderance of CD4 over CD8 T-cell responses was consistent across multiple antigens and time points in all seven animals studied by ICS in this report. Although potentially highly desirable in HIV-1- and HCV-infected subjects where virus-specific CD4+ T cells are commonly absent or dysfunctional (11), other pathogens may require a more balanced or predominant CD8+-T-cell response. Utilizing smaller peptides or pulsing for longer time periods may facilitate more efficient binding with MHC class I molecules and CD8+-T-cell stimulation. Third, we are planning studies to compare the administration of peptide-pulsed PBMC, peptide-pulsed whole blood, and peptide-pulsed-expanded-dendritic-cell populations for their ability to assist the clearance of acute SIV viremia in macaques. The very high levels of antigen-specific responses expanded by the use of PBMC or whole blood (up to 13.1 and 2.3% of all CD4+ and CD8+ T cells, respectively) suggest the responses induced could be at least equivalent to employing more complicated and less accessible modalities of expanding antigen-presenting cell populations extensively ex vivo.
Last, immunotherapy clinical trials for HIV and HCV by using autologous whole blood pulsed with overlapping viral peptides are suggested by these studies. Since T-cell responses are associated with the control of HIV-1 and clearance (and cure) of HCV (21), the ability to induce and expand CD4+- and CD8+-T-cell populations across most or all virus proteins could be highly advantageous. Our results, consistently demonstrating sharply enhanced T-cell immunity in macaques with controlled SHIV viremia, suggest the testable hypothesis that the enhancement of T-cell immunity in humans with HIV-1 viremia controlled by antiretroviral therapy could facilitate improved control upon withdrawal of drug therapy (28).
Acknowledgments
We thank J. Medveczky, I. Stratov, C. Fernandez, M. Smith, and S. Grueninger for technical assistance; R. Sydenham, A. Sydenham, L. Protyniak, and K. Szalnowski for animal care; K. Reimann for providing the depleting CD8 antibody (produced under NIH award R24 RR16001); M. Law for statistical analyses; and F. Carbone, S. Mueller, R. Coles, J. McCluskey, K. Walsh, W. Kimpton, and R. Cahill for helpful discussions.
REFERENCES
- 1.Aichele, P., K. Brduscha-Riem, S. Oehen, B. Odermatt, R. M. Zinkernagel, H. Hengartner, and H. Pircher. 1997. Peptide antigen treatment of naive and virus-immune mice: antigen-specific tolerance versus immunopathology. Immunity 6:519-529. [DOI] [PubMed] [Google Scholar]
- 2.Amara, R. R., F. Villinger, J. D. Altman, S. L. Lydy, S. P. O'Neil, S. I. Staprans, D. C. Montefiori, Y. Xu, J. G. Herndon, L. S. Wyatt, M. A. Candido, N. L. Kozyr, P. L. Earl, J. M. Smith, H. L. Ma, B. D. Grimm, M. L. Hulsey, J. Miller, H. M. McClure, J. M. McNicholl, B. Moss, and H. L. Robinson. 2001. Control of a mucosal challenge and prevention of AIDS by a multiprotein DNA/MVA vaccine. Science 292:69-74. [DOI] [PubMed] [Google Scholar]
- 3.Appay, V., J. J. Zaunders, L. Papagno, J. Sutton, A. Jaramillo, A. Waters, P. Easterbrook, P. Grey, D. Smith, A. J. McMichael, D. A. Cooper, S. L. Rowland-Jones, and A. D. Kelleher. 2002. Characterization of CD4(+) CTLs ex vivo. J. Immunol. 168:5954-5958. [DOI] [PubMed] [Google Scholar]
- 4.Barouch, D. H., T. M. Fu, D. C. Montefiori, M. G. Lewis, J. W. Shiver, and N. L. Letvin. 2001. Vaccine-elicited immune responses prevent clinical AIDS in SHIV(89.6P)- infected rhesus monkeys. Immunol. Lett. 79:57-61. [DOI] [PubMed] [Google Scholar]
- 5.Barouch, D. H., S. Santra, J. E. Schmitz, M. J. Kuroda, T. M. Fu, W. Wagner, M. Bilska, A. Craiu, X. X. Zheng, G. R. Krivulka, K. Beaudry, M. A. Lifton, C. E. Nickerson, W. L. Trigona, K. Punt, D. C. Freed, L. Guan, S. Dubey, D. Casimiro, A. Simon, M. E. Davies, M. Chastain, T. B. Strom, R. S. Gelman, D. C. Montefiori, M. G. Lewis, E. A. Emini, J. W. Shiver, and N. L. Letvin. 2000. Control of viremia and prevention of clinical AIDS in rhesus monkeys by cytokine-augmented DNA vaccination. Science 290:486-492. [DOI] [PubMed] [Google Scholar]
- 6.Borrow, P., H. Lewicki, B. H. Hahn, G. M. Shaw, and M. B. Oldstone. 1994. Virus-specific CD8+ cytotoxic T-lymphocyte activity associated with control of viremia in primary human immunodeficiency virus type 1 infection. J. Virol. 68:6103-6110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Coles, R. M., C. M. Jones, A. G. Brooks, P. U. Cameron, W. R. Heath, and F. R. Carbone. 2003. Virus infection expands a biased subset of T cells that bind tetrameric class I peptide complexes. Eur. J. Immunol. 33:1557-1567. [DOI] [PubMed] [Google Scholar]
- 8.Dale, C. J., R. De Rose, I. Stratov, S. Chea, D. C. Montefiori, S. Thomson, I. A. Ramshaw, B. E. H. Coupar, D. B. Boyle, M. Law, and S. J. Kent. 2004. Efficacy of DNA and fowlpox virus priming/boosting vaccines for simian/human immunodeficiency virus. J. Virol. 78:13819-13828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dale, C. J., R. De Rose, K. Wilson, H. Croom, S. A. Thomson, B. E. Coupar, A. J. Ramsay, D. F. Purcell, R. Ffrench, M. Law, S. Emery, D. A. Cooper, I. A. Ramshaw, D. B. Boyle, and S. J. Kent. 2004. Evaluation in macaques of HIV-1 DNA vaccines containing primate CpG motifs and fowlpoxvirus vaccines co-expressing IFNγ or IL-12. Vaccine 23:188-197. [DOI] [PubMed] [Google Scholar]
- 10.Dale, C. J., X. S. Liu, R. De Rose, D. F. Purcell, J. Anderson, Y. Xu, G. R. Leggatt, I. H. Frazer, and S. J. Kent. 2002. Chimeric human papilloma virus-simian/human immunodeficiency virus virus-like-particle vaccines: immunogenicity and protective efficacy in macaques. Virology 301:176-187. [DOI] [PubMed] [Google Scholar]
- 11.Douek, D. C., J. M. Brenchley, M. R. Betts, D. R. Ambrozak, B. J. Hill, Y. Okamoto, J. P. Casazza, J. Kuruppu, K. Kunstman, S. Wolinsky, Z. Grossman, M. Dybul, A. Oxenius, D. A. Price, M. Connors, and R. A. Koup. 2002. HIV preferentially infects HIV-specific CD4+ T cells. Nature 417:95-98. [DOI] [PubMed] [Google Scholar]
- 12.Estcourt, M. J., A. J. Ramsay, A. Brooks, S. A. Thomson, C. J. Medveckzy, and I. A. Ramshaw. 2002. Prime-boost immunization generates a high frequency, high-avidity CD8(+) cytotoxic T lymphocyte population. Int. Immunol. 14:31-37. [DOI] [PubMed] [Google Scholar]
- 13.Fuller, J. D., D. E. Craven, K. A. Steger, N. Cox, T. C. Heeren, and D. Chernoff. 1999. Influenza vaccination of human immunodeficiency virus (HIV)-infected adults: impact on plasma levels of HIV type 1 RNA and determinants of antibody response. Clin. Infect. Dis. 28:541-547. [DOI] [PubMed] [Google Scholar]
- 14.Hanke, T., R. V. Samuel, T. J. Blanchard, V. C. Neumann, T. M. Allen, J. E. Boyson, S. A. Sharpe, N. Cook, G. L. Smith, D. I. Watkins, M. P. Cranage, and A. J. McMichael. 1999. Effective induction of simian immunodeficiency virus-specific cytotoxic T lymphocytes in macaques by using a multiepitope gene and DNA prime-modified vaccinia virus Ankara boost vaccination regimen. J. Virol. 73:7524-7532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Harouse, J. M., A. Gettie, T. Eshetu, R. C. H. Tan, R. Bohm, J. Blanchard, G. Baskin, and C. Cheng-Mayer. 2001. Mucosal transmission and induction of simian AIDS by CCR5-specific simian/human immunodeficiency virus SHIVSF162P3. J. Virol. 75:1990-1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Harouse, J. M., A. Gettie, R. C. Tan, J. Blanchard, and C. Cheng-Mayer. 1999. Distinct pathogenic sequela in rhesus macaques infected with CCR5 or CXCR4 utilizing SHIVs. Science 284:816-819. [DOI] [PubMed] [Google Scholar]
- 17.Karlsson, A. C., J. N. Martin, S. R. Younger, B. M. Bredt, L. Epling, R. Ronquillo, A. Varma, S. G. Deeks, J. M. McCune, D. F. Nixon, and E. Sinclair. 2003. Comparison of the ELISPOT and cytokine flow cytometry assays for the enumeration of antigen-specific T cells. J. Immunol. Methods 283:141-153. [DOI] [PubMed] [Google Scholar]
- 18.Kent, S. J., P. D. Greenberg, M. C. Hoffman, R. E. Akridge, and M. J. McElrath. 1997. Antagonism of vaccine-induced HIV-1-specific CD4+ T cells by primary HIV-1 infection: potential mechanism of vaccine failure. J. Immunol. 158:807-815. [PubMed] [Google Scholar]
- 19.Kent, S. J., A. Zhao, S. J. Best, J. D. Chandler, D. B. Boyle, and I. A. Ramshaw. 1998. Enhanced T-cell immunogenicity and protective efficacy of a human immunodeficiency virus type 1 vaccine regimen consisting of consecutive priming with DNA and boosting with recombinant fowlpox virus. J. Virol. 72:10180-10188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Koup, R. A., J. T. Safrit, Y. Cao, C. A. Andrews, G. McLeod, W. Borkowsky, C. Farthing, and D. D. Ho. 1994. Temporal association of cellular immune responses with the initial control of viremia in primary human immunodeficiency virus type 1 syndrome. J. Virol. 68:4650-4655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lechner, F., D. K. Wong, P. R. Dunbar, R. Chapman, R. T. Chung, P. Dohrenwend, G. Robbins, R. Phillips, P. Klenerman, and B. D. Walker. 2000. Analysis of successful immune responses in persons infected with hepatitis C virus. J. Exp. Med. 191:1499-1512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Letvin, N. L., J. E. Schmitz, H. L. Jordan, A. Seth, V. M. Hirsch, K. A. Reimann, and M. J. Kuroda. 1999. Cytotoxic T lymphocytes specific for the simian immunodeficiency virus. Immunol. Rev. 170:127-134. [DOI] [PubMed] [Google Scholar]
- 23.Lu, W., X. Wu, Y. Lu, W. Guo, and J. M. Andrieu. 2003. Therapeutic dendritic-cell vaccine for simian AIDS. Nat. Med. 9:27-32. [DOI] [PubMed] [Google Scholar]
- 24.Maecker, H. T., H. S. Dunn, M. A. Suni, E. Khatamzas, C. J. Pitcher, T. Bunde, N. Persaud, W. Trigona, T. M. Fu, E. Sinclair, B. M. Bredt, J. M. McCune, V. C. Maino, F. Kern, and L. J. Picker. 2001. Use of overlapping peptide mixtures as antigens for cytokine flow cytometry. J. Immunol. Methods 255:27-40. [DOI] [PubMed] [Google Scholar]
- 25.Oehen, S., and K. Brduscha-Riem. 1998. Differentiation of naive CTL to effector and memory CTL: correlation of effector function with phenotype and cell division. J. Immunol. 161:5338-5346. [PubMed] [Google Scholar]
- 26.Oosterwijk-Wakka, J. C., D. M. Tiemessen, I. Bleumer, I. J. de Vries, W. Jongmans, G. J. Adema, F. M. Debruyne, P. H. de Mulder, E. Oosterwijk, and P. F. Mulders. 2002. Vaccination of patients with metastatic renal cell carcinoma with autologous dendritic cells pulsed with autologous tumor antigens in combination with interleukin-2: a phase 1 study. J. Immunother. 25:500-508. [DOI] [PubMed] [Google Scholar]
- 27.Robinson, H. L., D. C. Montefiori, R. P. Johnson, K. H. Manson, M. L. Kalish, J. D. Lifson, T. A. Rizvi, S. Lu, S. L. Hu, G. P. Mazzara, D. L. Panicali, J. G. Herndon, R. Glickman, M. A. Candido, S. L. Lydy, M. S. Wyand, and H. M. McClure. 1999. Neutralizing antibody-independent containment of immunodeficiency virus challenges by DNA priming and recombinant pox virus booster immunizations. Nat. Med. 5:526-534. [DOI] [PubMed] [Google Scholar]
- 28.Rosenberg, E. S., M. Altfeld, S. H. Poon, M. N. Phillips, B. M. Wilkes, R. L. Eldridge, G. K. Robbins, R. T. D'Aquila, P. J. Goulder, and B. D. Walker. 2000. Immune control of HIV-1 after early treatment of acute infection. Nature 407:523-526. [DOI] [PubMed] [Google Scholar]
- 29.Rosenberg, E. S., J. M. Billingsley, A. M. Caliendo, S. L. Boswell, P. E. Sax, S. A. Kalams, and B. D. Walker. 1997. Vigorous HIV-1-specific CD4+ T cell responses associated with control of viremia. Science 278:1447-1450. [DOI] [PubMed] [Google Scholar]
- 30.Russell, N. D., M. G. Hudgens, R. Ha, C. Havenar-Daughton, and M. J. McElrath. 2003. Moving to human immunodeficiency virus type 1 vaccine efficacy trials: defining T cell responses as potential correlates of immunity. J Infect. Dis. 187:226-242. [DOI] [PubMed] [Google Scholar]
- 31.Schmitz, J. E., M. J. Kuroda, S. Santra, V. G. Sasseville, M. A. Simon, M. A. Lifton, P. Racz, K. Tenner-Racz, M. Dalesandro, B. J. Scallon, J. Ghrayeb, M. A. Forman, D. C. Montefiori, E. P. Rieber, N. L. Letvin, and K. A. Reimann. 1999. Control of viremia in simian immunodeficiency virus infection by CD8+ lymphocytes. Science 283:857-860. [DOI] [PubMed] [Google Scholar]
- 32.Seth, A., I. Ourmanov, J. E. Schmitz, M. J. Kuroda, M. A. Lifton, C. E. Nickerson, L. Wyatt, M. Carroll, B. Moss, D. Venzon, N. L. Letvin, and V. M. Hirsch. 2000. Immunization with a modified vaccinia virus expressing simian immunodeficiency virus (SIV) Gag-Pol primes for an anamnestic Gag- specific cytotoxic T-lymphocyte response and is associated with reduction of viremia after SIV challenge. J. Virol. 74:2502-2509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shiver, J. W., T. M. Fu, L. Chen, D. R. Casimiro, M. E. Davies, R. K. Evans, Z. Q. Zhang, A. J. Simon, W. L. Trigona, S. A. Dubey, L. Huang, V. A. Harris, R. S. Long, X. Liang, L. Handt, W. A. Schleif, L. Zhu, D. C. Freed, N. V. Persaud, L. Guan, K. S. Punt, A. Tang, M. Chen, K. A. Wilson, K. B. Collins, G. J. Heidecker, V. R. Fernandez, H. C. Perry, J. G. Joyce, K. M. Grimm, J. C. Cook, P. M. Keller, D. S. Kresock, H. Mach, R. D. Troutman, L. A. Isopi, D. M. Williams, Z. Xu, K. E. Bohannon, D. B. Volkin, D. C. Montefiori, A. Miura, G. R. Krivulka, M. A. Lifton, M. J. Kuroda, J. E. Schmitz, N. L. Letvin, M. J. Caulfield, A. J. Bett, R. Youil, D. C. Kaslow, and E. A. Emini. 2002. Replication-incompetent adenoviral vaccine vector elicits effective anti-immunodeficiency-virus immunity. Nature 415:331-335. [DOI] [PubMed] [Google Scholar]
- 34.Smithers, M., K. O'Connell, S. MacFadyen, M. Chambers, K. Greenwood, A. Boyce, I. Abdul-Jabbar, K. Barker, K. Grimmett, E. Walpole, and R. Thomas. 2003. Clinical response after intradermal immature dendritic cell vaccination in metastatic melanoma is associated with immune response to particulate antigen. Cancer Immunol. Immunother. 52:41-52. [DOI] [PMC free article] [PubMed] [Google Scholar]