Abstract
Background
Stellate ganglion block (SGB) has been shown to reduce perioperative complications in various surgeries. Because laparoscopic techniques and instruments have advanced during the past two decades, laparoscopic liver resection is being increasingly adopted worldwide. Lesser blood loss, fewer postoperative complications, and shorter postoperative hospital stays are the advantages of laparoscopic liver resection, as compared to conventional open surgery. There is an urgent need for an effective intervention to reduce perioperative complications and accelerate postoperative recovery. This study investigated the effect of ultrasound-guided SGB on enhanced recovery after laparoscopic partial hepatectomy.
Methods
We compared patients who received SGB with 0.5% ropivacaine (group S) with those who received SGB with 0.9% saline (group N). A total of 58 patients with partial hepatectomy were enrolled (30 S) and (28 N). Before induction of anesthesia, SGB was performed with 0.5% ropivacaine in group S and 0.9% saline in group N. Main outcome: Comparison of serum inflammatory cytokines concentration at each time point.
Results
Main outcome: When comparing IL-6 and IL-10 concentrations among groups, group S showed less variation over time compared to group N. For comparison between groups, the serum IL-6 concentration in group S was lower than that in group N at 6 and 24 h after operation (P < 0.01), and there was a significant linear relationship between serum IL-6 concentration at 24 h after operation and hospitalization situation.
Conclusions
Ultrasound-guided SGB can stabilize perioperative inflammatory cytokines plays a positive role in the enhanced recovery of patients after laparoscopic partial hepatectomy. The serum IL-6 level within 24 h after surgery may be used as a predictor of hospitalization.
Trial registration
The study was registered at the ClinicalTrials.gov (Registration date: 13/09/2021; Trial ID: NCT05042583).
Keywords: Enhanced recovery after surgery (ERAS), Inflammatory cytokines, Partial hepatectomy, Stellate ganglion, Ultrasound guidance
Introduction
Laparoscopic partial hepatectomy is an increasingly essential surgical therapy for liver, bile duct, and other illnesses, since the benefits of minimally invasive hepatectomy are more validated and its indications are steadily expanded [1–4]. Hepatectomy carries many risks, including a longer operation time, more blood loss during operation, a large amount of fluid transfer, a high risk of hypotension, hepatic portal occlusion, ischemia-reperfusion injury, and more postoperative complications than other abdominal operations, even when performed using minimally invasive techniques. Moreover, patients may have significant liver function impairment due to the effects of anticancer medications, making it even more important to perform careful perioperative treatment.
To create its therapeutic effects, stellate ganglion blockade is known to have a function in controlling the autonomic nerve system, endocrine system, and immunological system, as is common knowledge. All three of these systems—the autonomic nervous system, the endocrine system, and the immune system—play a significant role in a speedier recovery for patients following surgery [5–7]. There are currently only a few numbers of research on the use of stellate ganglion block to improve postoperative recovery.
In this investigation, stellate ganglion block was used because of its ability to control not only the autonomic nervous system (regulating gastrointestinal function [8], sleep disorder [9], hemodynamic stability [10], prevention and treatment of arrhythmias [11]), but also the endocrine and immune systems (regulating the production of acute excessive inflammatory reactions caused by various reasons, such as “cytokine storms” caused by inflammatory cascade reactions [5, 12]). With the modulation of inflammatory cytokines as the primary outcome, we tested it on patients having elective laparoscopic partial hepatectomy and analyzed its effect on postoperative tissue harm and improved postoperative recovery.
Materials and methods
Ethics
The Ethics Committee of Shaoxing People’s Hospital in Shaoxing, China (Chairperson Prof Bao-chun Lu) approved this study (Ethical Committee N°2020-K-Y-185-01) on 24 July 2020. The study was successfully registered in ClinialTrials.gov on 13 September 2021 (ID: NCT05042583), and is continuing in progress, with the informed agreement of patients and their families. The first patient enrolment date was January 1, 2021, and 58 patients who had undergone elective laparoscopic partial hepatectomy at Shaoxing People’s Hospital had been included in the study. The following criteria must be met: (1) patients who underwent elective laparoscopic hepatectomy; (2) Classification of NYHA cardiac function: I or II; (3) ASA-PS ≤ III; (4) Age between 18 and 75 years old. Conditions for exclusion: (1) Non-laparoscopic hepatectomy patients; (2) Patients younger than 18 or older than 75 years old; (3) Classification of NYHA heart function ≥ III; (4) ASA-PS > III; (5) Allergy to local anesthetics; (6) Patients with immune diseases or immunosuppression; (7) Patients with severe mental illness who are unable to cooperate with SGB; (8) Patients with abnormal neck anatomy; (9) Patients refused to participate in the researcher; (10) The coagulation function is obviously abnormal; (11) There are insurmountable difficulties in follow-up and specimen collection.
Study design
This study is a single-center, prospective, randomized controlled, and blinded investigation. Patients were divided into the experimental group (group S) and the control group (group N) on the morning of the surgery. The group assignment was decided by a computer-generated random sequence table, and the random sequences were held in sealed opaque envelopes, which were opened by the observer before the anesthesia. The group assignment and anesthesia were managed by investigators who were not involved in postoperative follow-up, and the surgeon, patient, and investigator responsible for the operation and postoperative follow-up were unaware of the anesthetic approach. In group S, 0.5% ropivacaine 6mL was utilized for stellate ganglion block under ultrasound guidance before general anesthesia. Patients in group N got stellate ganglion block under ultrasound guidance with 0.9% saline 6mL before general anesthesia.
Stellate ganglion block procedure and its successful performance
A high-frequency linear ultrasound probe (12 MHz) is applied while the patient is supine with his head tilted to the opposite side. The probe is positioned at the level of the notch in the cricoid cartilage on the patient’s left side, and its direction is 30–45 degrees off the sagittal plane of the neck. Besides the carotid artery, jugular vein, anterior scalene muscle, and long neck muscle, several nearby structures, like the C6 cone and its distinctive “double-peak” transverse process, can be seen and identified as well. And do local routine disinfection. The needle is put outside the carotid artery and into the loose connective tissue on the longus cervical muscle, avoiding essential blood arteries, nerves, and organs. There is no resistance to pressing the drug or pumping blood and gas back. Diffusion of 6 ml of medicinal liquid blocks stellate ganglions. The common indicators of a successful block are Horner’s syndrome on the operative side, negative sweat test, and noticeable skin temperature increase in the dominating area.
Anesthesia program
The patient next punctured the left radial artery to begin invasive blood pressure monitoring after the block. Nearly all patients in our previous study developed Horner’s syndrome within 10 min after stellate ganglion block, similar to the results reported in previous related studies [13], so we decided to induce anesthesia after 10 min of block with midazolam 0.02 mg/kg, etomidate 0.1–0.3 mg/kg, cis-atracurium 0.3 mg/kg and sufentanil 0.3–0.5 µg/kg, and to use Drager anesthesia machine for mechanical ventilation after tracheal intubation. Intravenous infusion of remifentanil 0.1–0.2 µg/kg/min, cis-atracurium 1 µg/kg/min, propofol and sevoflurane were utilized to maintain the anesthetic depth (BIS 40–60). The administration of cis-atracurium was halted 30 min before the completion of the operation, and all patients were given ondansetron 8 mg to reduce vomiting following the operation. The anesthesiologist judged that all maintenance drugs should be terminated 5 min before the completion of the operation. If the judgment was mistaken, propofol should be given adequately to continue the operation. After the operation, all patients were given patient-controlled intravenous analgesia (PCIA, 100 µg Sufentanil Citrate and 150 mg Flurbiprofen Axetil) and then resuscitated in the post anesthesia care unit (PACU). The indications for transferring out of PACU conform to the guidelines of Miller Anesthesiology, 7th edition.
Outcomes
Main outcome
Inflammatory cytokines: 8 ml blood samples were collected from patients before SGB (T0), 1 h (T1), 3 h (T2), 6 h (T3), 24 h (T4) and 72 h (T5) after operation. All blood samples were centrifuged at 3000 rpm for 10 min, and the separated serum was stored in a refrigerator at -80 °C and thawed until the need for detection. Serum inflammatory cytokines IL-2, IL-6, IL-10, TNF-α and IFN-γ were detected by cytometric bead array (CBA).
Secondary outcome
Related events during and after the operation: record the operation time, blood loss during the operation, blood transfusion cases during the operation, vasoactive drugs used during and after the operation (, extubation time after the end of anesthesia, postoperative complications and Clavien-Dindo Classification, additional tramadol analgesia cases in the ward within 48 h, the first postoperative exhaust time, postoperative fluid intake time, semi-fluid time, the number of liver segments involved in hepatectomy, and postoperative pathology were recorded through the hospital medical record system and postoperative follow-up. Hemodynamics: The hemodynamic changes at baseline before operation, immediately before block, 15 min after block, 35 min after block, and out of PACU were recorded. Transaminase other inflammatory markers: The variations of alanine transaminase (ALT), aspartate transaminase (AST), C-reactive protein (CRP) and white blood cell (WBC) before operation, post operation day 1 (POD1), post operation day 3 (POD3) and post operation day 6 (POD6) were recorded. Post-operation hospitalization: The hospitalization costs and length of stay from the day of operation were recorded.
Statistical analysis
The sample size was determined using the PASS15 program, and information for the independent variable, inflammatory cytokines, was gathered from the published literature [14]. Sample sizes of 24 in group S and 24 in group N were determined using the two independent sample mean test standard, the bilateral = 0.05, and a confidence degree of 90%. Due to the possibility of a sample loss of more than 20% during the experiment, a total of 33 patients meeting the criteria for inclusion in each group were sought out.
The measurement data involved in this study, a single sample K-S normal distribution test. The measurement data conforming to the normal distribution is expressed by the Mean ± SD ( ± s); T-test was used to examine the differences between groups, and repeated measurement ANOVA was used to compare the differences at different time points within groups. Data from non-normal distributions are expressed using the median (interquartile range), and the Wilcoxon rank sum test is used to examine differences across groups; for repeated-measures data, the generalized estimation equation is employed. The percentages from the categorical data are put through a test using either the chi-square or Fisher’s exact statistic. SPSS 25.0 was used for all statistical analysis. P < 0.05 is statistically significant. Carry out an R-value study of correlation using a standard linear regression model. Statistics are mapped using GraphPad Prism.
± s); T-test was used to examine the differences between groups, and repeated measurement ANOVA was used to compare the differences at different time points within groups. Data from non-normal distributions are expressed using the median (interquartile range), and the Wilcoxon rank sum test is used to examine differences across groups; for repeated-measures data, the generalized estimation equation is employed. The percentages from the categorical data are put through a test using either the chi-square or Fisher’s exact statistic. SPSS 25.0 was used for all statistical analysis. P < 0.05 is statistically significant. Carry out an R-value study of correlation using a standard linear regression model. Statistics are mapped using GraphPad Prism.
Results
Study participants
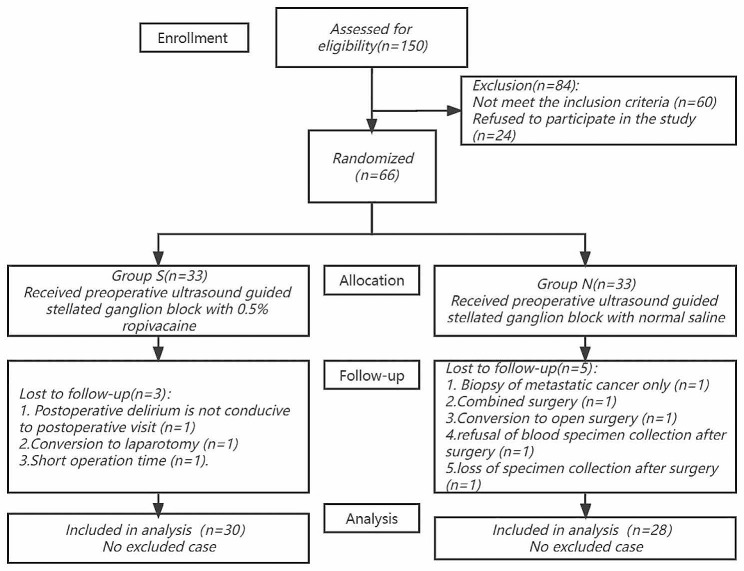
Between January 2021 and June 2022, we evaluated 150 patients who were eligible for laparoscopic partial hepatectomy. In the end, 66 people were randomly allocated after 60 were excluded and 24 declined to take part in the study. There were 66 patients total, evenly split between two groups of 33. In the follow-up, Three patients were eliminated from group S in the follow-up: one had postoperative delirium, making a postoperative visit unfeasible; one was converted to laparotomy during the surgery; and one had a short operation length. Five patients were eliminated from group N: one who only had a biopsy of metastatic cancer during the procedure, one who needed a combination operation, one who converted to laparotomy during the operation, one who declined to collect blood samples after the operation, and one who failed to collect blood samples after the operation. Therefore, 58 patients were assessed for inclusion in the final analysis (Fig. 1). The baseline parameters, operation time, number of liver segments involved in the operation, and postoperative pathology were similar between the two groups, and were comparable between the two groups (Tables 1 and 2).
Fig. 1.
CONSORT flow diagram of participants in the study
Table 1.
Intraoperative and postoperative procedure-related events
| Event | Group S(n = 30) | Group N(n = 28) | Statistical value | P value |
|---|---|---|---|---|
| Time of operation (min) | 180.00(130.00 to 251.25) | 235.00(171.25 to 320.00) | Z=-1.736 | 0.083 |
| Intraoperative blood loss(ml) | 300.00(142.50 to 450.00) | 300.00(100.00 to 450.00) | Z=-0.165 | 0.8869 |
| Intraoperative blood transfusion | 9(30.0%) | 15(53.6%) | χ2 = 3.317 | 0.069 |
| Plasma only | 4(13.3%) | 7(25%) | ||
| Erythrocyte only | 0(0%) | 0(0%) | ||
| Plasma + Erythrocytes | 5(16.7%) | 8(28.6%) | ||
| Vasoactive drug intervention was required during the operation | 14(43.3%) | 20(71.4%) | χ2 = 4.661 | 0.031 |
| Single dosage | 10(33.3%) | 15(53.6%) | ||
| Pump injection | 3(10%) | 5(17.9%) | ||
| Postoperative vasoactive drug intervention was required | 0 | 0 | ||
| Extubation time after the end of anesthesia (min) | 20.00(10.00 to 32.50) | 15.00(7.00 to 30.00) | Z=-1.528 | 0.127 |
| Postoperative complications Clavien-Dindo Classification | χ2 = 1.809 | 0.179 | ||
| I | 3(10%) | 2(7.1%) | ||
| II | 14(46.7%) | 15(53.6%) | ||
| IIIa | 3(10%) | 6(21.4%) | ||
| IIIb | 0 | 0 | ||
| IVa | 0 | 0 | ||
| IVb | 0 | 0 | ||
| V | 0 | 0 | ||
| Additional cases of tramadol analgesia were required within 48 h | 6(19.4%) | 2(7.1%) | χ2 = 1.077 | 0.299 |
| Use once | 4(13.3%) | 2(7.1%) | ||
| Use twice | 2(7.1%) | 0(0%) | ||
| The first postoperative exhaust time (h) | 44.25(29.00 to 60.00) | 50.00(40.00 to 68.00) | Z=-1.277 | 0.202 |
| Postoperative fluid intake time (h) | 23.50(18.00 to 45.00) | 41.00(22.00 to 44.00) | Z=-1.712 | 0.087 |
| Postoperative semisolid diet intake time (h) | 64.50(42.38 to 69.50) | 67.00(48.00 to 75.00) | Z=-0.981 | 0.326 |
| Number of hepatic segments involved in liver resection | χ2 = 4.481 | 0.236 | ||
| 1 hepatic segment | 15(50%) | 10(35.7%) | ||
| 2 hepatic segments | 10(33.3%) | 7(25%) | ||
| 3 hepatic segments | 3(10%) | 9(32.1%) | ||
| 4 hepatic segments | 2(6.7%) | 2(7.1%) | ||
| Postoperative pathology | ||||
| Benign /Malignant | 18/12 | 13/15 | χ2 = 1.072 | 0.300 |
| Hepatocellular carcinoma | 9(30%) | 10(35.7%) | ||
| Intrahepatic Cholangiocarcinoma | 2(6.7%) | 2(7.1%) | ||
| Benign liver tumors | 7(23.3%) | 1(3.6%) | ||
| Intra-and extrahepatic bile duct stone | 4(13.3%) | 10(35.7%) | ||
| Atrophy of liver cirrhosis | 0(0%) | 1(3.6%) | ||
| Liver metastases | 1(3.3%) | 3(10.7%) | ||
| Hepatic hemangioma | 7(23.3%) | 1(3.6%) | ||
| Pringle maneuvers were used | 23(76.7%) | 22(78.6%) | χ2 = 0.030 | 0.862 |
Values are median (range) or number (proportion)
Table 2.
Baseline characteristics of the trial patients
| Characteristic | Group S(n = 30) | Group N(n = 28) | Statistical value | P value |
|---|---|---|---|---|
| Gender (male/female) | 19/11 | 18/10 | χ2 = 0.006 | 0.940 |
| Age (years old) | 60.23 ± 10.56 | 63.43 ± 10.00 | t=-1.181 | 0.242 |
| BMI | 24.36 ± 2.60 | 23.37 ± 3.08 | t = 1.320 | 0.192 |
| ASA score | χ2 = 0.291 | 0.864 | ||
| 1 | 2(6.7%) | 1(3.6%) | ||
| 2 | 25(83.3%) | 24(85.7%) | ||
| 3 | 3(10.0%) | 3(10.7%) | ||
| 4 | 0 | 0 | ||
| Previous hepatectomy | 1(3.3%) | 1(3.6%) | χ2<0.001 | >0.999 |
| Postoperative interventional therapy was needed | 8(26.7%) | 9(32.1%) | χ2 = 0.210 | 0.647 |
| Postoperative Chemotherapy is Required | 7(23.3%) | 4(14.3%) | χ2 = 0.771 | 0.380 |
Values are mean ± SD or number (proportion)
Main outcome
We detected the levels of five inflammatory cytokines such as IL-2, IL-6, IL-10, TNF-α and IFN-γ in patients before SGB (T0), 1 h (T1), 3 h (T2), 6 h (T3), 24 h (T4) and 72 h (T5) after the start of operation. Figure 2a shows that group S and group N had different IL-6 concentration distributions at each time point (P < 0.001). At T3 and T4, group S [62.82 (26.81 to 173.19), 62.37 (33.50, 117.62)] had lower IL-6 levels than group N [153.00 (82.88 to 610.11), 175.95 (74.05 to 354.83), P < 0.01]. At T5, there was no significant difference between T0 [4.67 (2.9 to 9.66)] and T5 [26.57 (11.36 to 55.21)] in group S (P = 0.652), while there was still significant difference between T0[5.54 (3.06 to 7.53)] and T5 [43.99 (20.16 to 77.87)] in group N (P<0.001). As shown in Fig. 2b, the intra-group comparison of IL-10 concentrations in group S and group N showed substantial differences in inflammatory factor concentrations at each time point (P < 0.001), although there was no statistical difference between groups (P > 0.05). At T5, there was no significant difference between T0 [2.89 (1.36 to 5.46)] and T5[3.36 (2.24 to 7.73)] in group S (P = 0.115), while there was still significant difference between T0 [2.82 (1.87 to 4.98)] and T5 [4.97 (3.18 to 7.63)] in group N (P = 0.002).
Fig. 2.
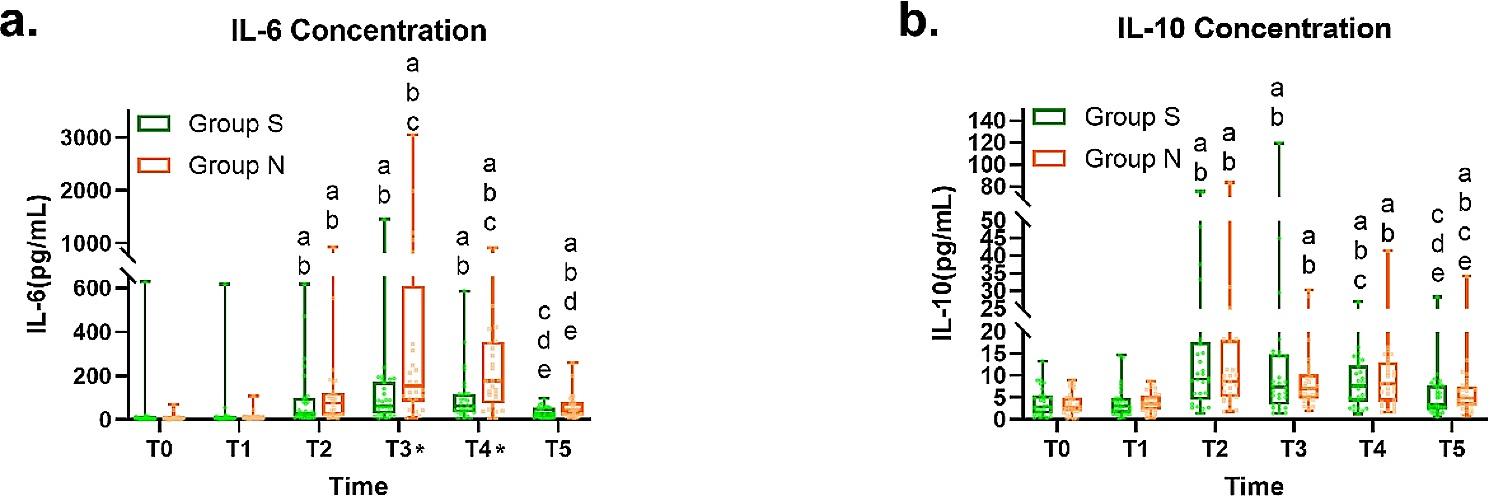
(a) Box line scatter diagram of the concentration change of IL-6 in the group S and group N at each time point; (b) Box line scatter diagram of the concentration change of IL-10 in group S and group N at each time point
(The box is the quartile separation, the ends of the extension line are the maximum and minimum values, and the scatter points are the distribution of the data)
a: There is statistical difference with before block; b: There was a statistical difference with 1 h after the operation; c: There was a statistical difference with 3 h after the operation; d: There was a statistical difference with 6 h after the operation; e: There was a statistical difference with 24 h after the operation started; *: There is a statistical difference between group S and group N (P < 0.05)
Secondary outcome
Intraoperative situation
The two groups had similar bleeding and blood transfusions, but vasoactive medication use was statistically different. Group S [14 (43.3%)] had fewer vasoactive drug instances than group N [20 (71.4%)] (P = 0.031) (Table 1). At the same time, we recorded some hemodynamic data (Fig. 3). Although there were significant differences in intra-group comparison at each time point (P < 0.001), the variance of group S (F = 19.443) was smaller than that of group N (F = 24.318) in terms of the fluctuation of MAP, and the dispersion of data was smaller and the fluctuation of blood pressure was smaller.
Fig. 3.
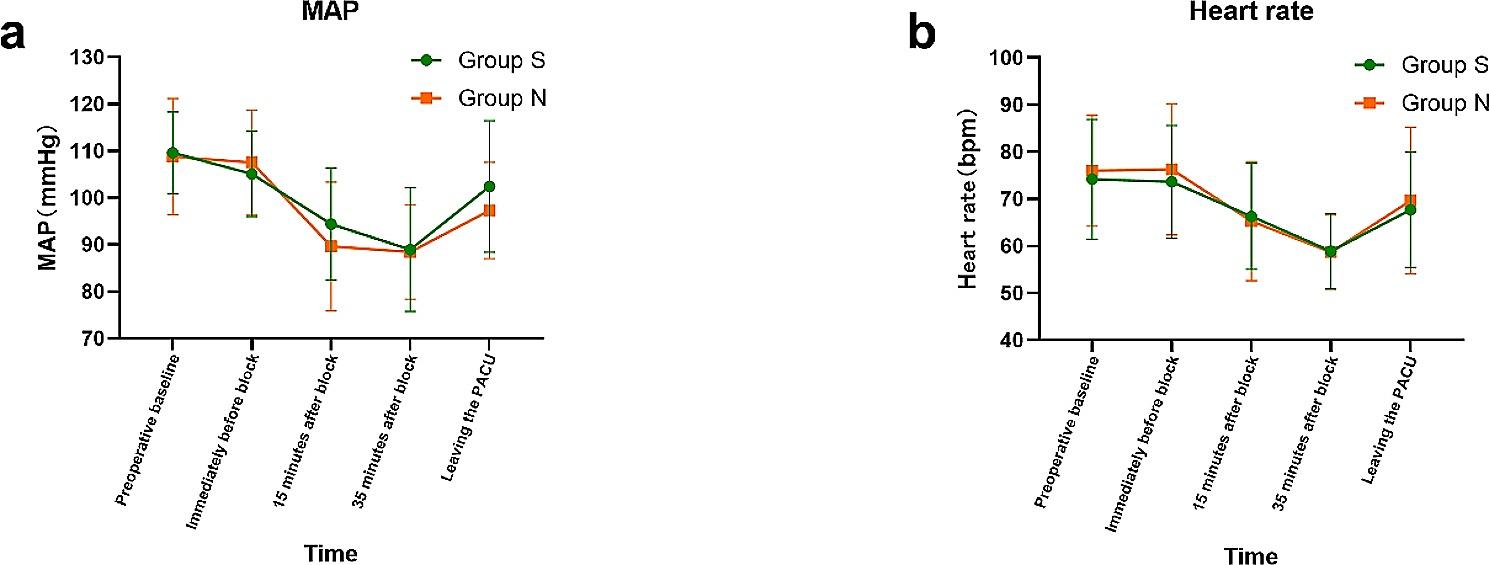
(a) Trend plot of mean arterial pressure (MAP) in group S and group N at each time points; (b) Trend plot of heart rate in group S and group N at each time points
(The center point is the mean, and the two ends of the extension line are standard deviations)
Complications 30 days after the operation
Both groups had similar Clavien-Dindo Classification of Postoperative Complications scores (Table 1). Only the number of instances with greater drainage fluid in the abdominal cavity in Group S was lower than in Group N (P = 0.047), while the rest of the problems were identical (Table 3). We detected no ultrasound-guided stellate ganglion block complications.
Table 3.
Complications within 30 days after the operation
| Event | Group S(n = 30) | Group N(n = 28) | χ2 value | P value |
|---|---|---|---|---|
| Incision infection | 1(3.3%) | 0(0%) | <0.001 | >0.99 |
| Fever | 7(23.3%) | 11(39.3%) | 1.722 | 0.189 |
| Nausea | 1(3.3%) | 1(3.6%) | <0.001 | >0.99 |
| NRS ≥ 4 for pain | 5(16.7%) | 2(7.1%) | 0.503 | 0.478 |
| Bile leakage (no treatment required) | 0(0%) | 3(10.7%) | 1.557 | 0.212 |
| Acute Kidney Injury (Transient) | 1(3.3%) | 2(7.1%) | 0.004 | 0.951 |
| Chronic Pain | 1(3.3%) | 0(0%) | <0.001 | >0.99 |
| Atrial fibrillation | 1(3.3%) | 0(0%) | <0.001 | >0.99 |
| Gastrointestinal dysfunction | 13(43.3%) | 8(28.6%) | 1.366 | 0.242 |
| More drainage fluid in the abdominal cavity | 4(13.3%) | 10(35.7%) | 3.962 | 0.047 |
| Culture of drainage fluid suggested abdominal infection | 3(10.0%) | 6(21.4%) | 0.703 | 0.402 |
| Pulmonary infection | 1(3.3%) | 2(7.1%) | 0.004 | 0.951 |
| Bile leakage (treatment required) | 1(3.3%) | 0(0%) | <0.001 | >0.99 |
| Pleural effusion (moderate) | 3(10.0%) | 6(21.4%) | 0.703 | 0.402 |
Values are number (proportion)
Hospitalization situation
Group S had a shorter length of stay (calculated from the day of operation) (8.2 ± 2.1, 10.2 ± 3.8, P = 0.020) and lower hospitalization costs (calculated from the day of operation) (38622.35 (31676.04 to 44861.15), P = 0.015) than group N. (Fig. 4). A simple linear regression analysis was made between IL-6 concentrations at the time points of the primary outcome at which there was a between-group difference and the hospitalization situation (Fig. 5). The length of stay (calculated from the day of operation) was moderately significantly correlated with the concentration of IL-6 at T4 (r = 0.3917; P = 0.0024; n = 58; Fig. 5a), as were the hospitalization expenses (calculated from the day of operation) that were moderately significantly correlated with the concentration of IL-6 at T4 (r = 0.4170; P = 0.0011; n = 58; Fig. 5b). However, the length of stay (calculated from the day of operation) had little correlation with the concentration of IL-6 at T3 (r = 0.2761; P = 0.0358), and hospitalization expenses (calculated from the day of operation) had no correlation with the concentration of IL-6 at T3 (r = 0.1938; P = 0.1450).
Fig. 4.
(a) Histogram of comparison of length of stay (calculated from the day of operation) between Group S and Group N (expressed as average and standard deviation); (b) Box line scatter diagram of comparison of hospitalization costs (calculated from the day of operation) between Group S and Group N
(The box is the quartile separation, the ends of the extension line are the maximum and minimum values, and the scatter points are the distribution of the data)
Fig. 5.
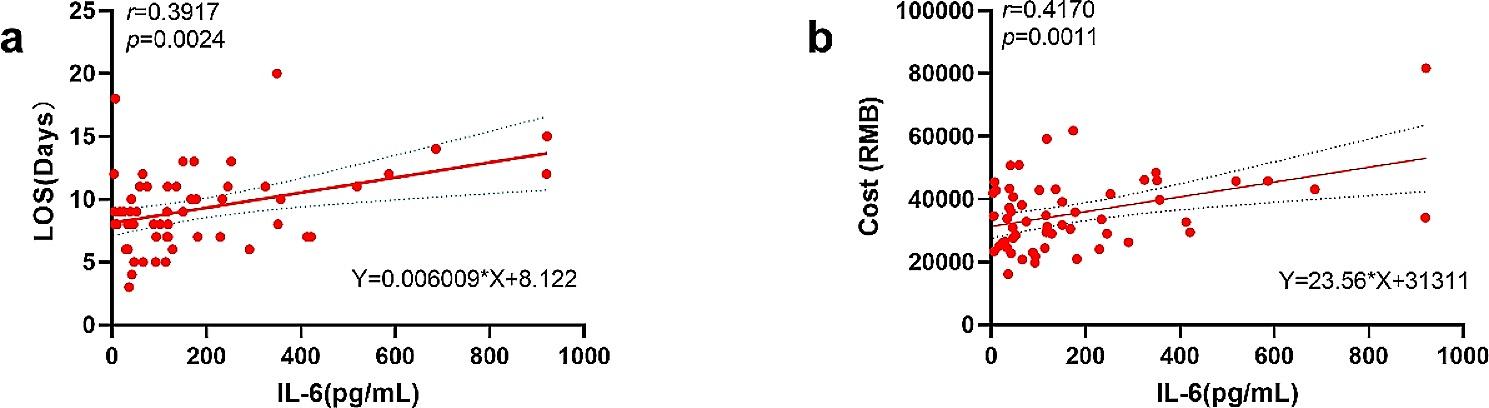
(a) Correlation between length of stay (calculated from the day of operation) and IL-6 concentration at T4; (b) Correlation between hospitalization expenses (calculated from the day of operation) and IL-6 concentration at T4
(n = 58, dotted line is 95% confidence interval)
Liver function
There was no significant difference in the effect on liver function between the two groups (Table 4).
Table 4.
Comparison of transaminases
| Event | Preoperative | POD1 | POD3 | POD6 | P value | |
|---|---|---|---|---|---|---|
| ALT (U/L) | Group S(n = 30) | 23.6(16.08 to 40.9) | 178.2(123.68 to 353.85) a | 137.15(79.53 to 210.3) a | 64.9(46.53 to 108.3) abc | <0.01 |
| Group N(n = 28) | 28.75(14.95 to 39.95) | 169.75(110.58 to 302.05) a | 127.7(74.68 to 242.5) a | 61(38.63 to 95.85) bc | <0.01 | |
| Z value | -0.047 | -0.467 | -0.319 | -0.576 | ||
| P value | 0.963 | 0.641 | 0.75 | 0.565 | ||
| AST(U/L) | Group S(n = 30) | 23.2(19.8 to 30.85) | 165.05(114.28 to 330.2) a | 66.15(31.55 to 112.75) ab | 25.9(17.8 to 51.33) bc | <0.01 |
| Group N(n = 28) | 30.1(20.1 to 40.58) | 201.8(121.25 to 353.45) a | 62.1(40.45 to 134.38) ab | 30(22.13 to 46.48) bc | <0.01 | |
| Z value | -1.611 | -0.42 | -0.288 | -0.607 | ||
| P value | 0.107 | 0.674 | 0.773 | 0.544 |
Values are median (range)
a: There is a statistical difference with before operation, b: There is a statistical difference with POD1, c: There is a statistical difference with POD3.
CRP and WBC
The level of CRP in group S on day 6 after surgery was found to be significantly lower compared to that in group N [29.97 (21.94 to 63.33), 46.23(35.27 to 65.22), P = 0.038] (Table 5). There was no significant difference in the effect on WBC between the two groups (Table 5).
Table 5.
Comparison of CRP and WBC
| Event | Preoperative | POD1 | POD3 | POD6 | P value | |
|---|---|---|---|---|---|---|
| CRP (M (P25, P75), mg/L) | Group S(n = 30) | 1.27(0.50 to 4.10) | 32.97(20.86 to 66.90) a | 86.80(47.21 to 130.22) ab | 29.97(21.94 to 63.33) ac | <0.01 |
| Group N(n = 28) | 1.79(0.80 to 15.83) | 31.99(20.30 to 59.19) a | 105.15(83.52 to 173.55) ab | 46.23(35.27 to 65.22) ac | <0.01 | |
| Z value | -1.408 | -0.436 | -1.572 | -2.07 | ||
| P value | 0.159 | 0.663 | 0.116 | 0.038 | ||
| WBC (M (P25, P75),10^9/L) | Group S(n = 30) | 5.25(4.40 to 6.94) | 11.77(9.05 to 14.56) a | 9.03(6.18 to 10.97) a | 6.64(5.42 to 8.06) bc | <0.01 |
| Group N(n = 28) | 5.20(4.49 to 6.30 | 11.50(9.25 to 14.35) a | 9.35(7.90 to 11.93) a | 7.08(5.86 to 8.78) bc | <0.01 | |
| Z value | -0.233 | -0.062 | -0.996 | -1.089 | ||
| P value | 0.815 | 0.95 | 0.319 | 0.276 |
Values are median (range)
a: There is a statistical difference with before operation, b: There is a statistical difference with POD1, c: There is a statistical difference with POD3
Discussion
Ultrasound-guided stellate ganglion block is becoming increasingly popular, and perioperative investigations are increasing [8, 15, 16]. This prospective randomized controlled trial found that ultrasound-guided stellate ganglion block (SGB) plays a role in the management of inflammatory cytokines after elective laparoscopic partial hepatectomy. Group S had much lower levels of the pro-inflammatory cytokine IL-6 at 6 and 24 h after surgery, and it recovered faster. At 72 h, there was no significant difference from before the block in group S, but the level in group N was still significantly higher than before the block, and the level of IL-6 in group S was significantly more stable than that in group N from rise to recovery. Although there was no significant difference in the anti-inflammatory cytokine IL-10 between the two groups at each time point, 72 h after the operation, IL-10 in group S was no different from that before the block, and IL-10 in group N was still much greater. As a result, SGB reduces pro-inflammatory cytokines following laparoscopic partial hepatectomy, minimizing tissue damage.SGB reduces the release of pro-inflammatory cytokines, which may explain the early recovery of anti-inflammatory cytokines. The lack of substantial variations in anti-inflammatory cytokines between groups may be due to the hepatic portal block’s mild ischemia-reperfusion damage and low proinflammatory cytokine production. After all, anti-inflammatory cytokines play a role by inhibiting excessive inflammatory responses, thus limiting the adverse effects induced by excessive inflammation [17]. At the same time, CRP, another significant indicator of inflammation dependent on IL-6 expression, is associated with inflammatory liver damage and liver cancer progression. CRP in group S was significantly lower than that in group N on the sixth day after surgery, and there was no statistical difference when IL-6 reached its peak, which may be related to factors such as the longer delay difference in CRP change and the effect of liver resection on liver release of CRP. In terms of the inflammatory response, Ying Li et al. found that stellate ganglion block could increase CD4 + T cell activity and decreased IL-2, IL-4, and TNF- stimulated TIPE2 production, hence increasing rat survival after severe hemorrhagic shock [6]. In 2020, Eugene Lipov et al. put forward a hypothesis and gave some evidence to show that stellate ganglion block may regulate immune inflammation by regulating sympathetic innervation of primary (thymus, bone marrow) and secondary immune organs (such as the spleen, lymph nodes, and mucosa-associated lymphoid tissues), and it finally inhibits the production of pro-inflammatory cytokines (like IL-1, IL-6, TNF-α, etc.) while increasing the production of anti-inflammatory cytokines (such as IL-4, IL-10, IL-13, etc.) [18]. The findings of these other researches are consistent with ours. Although blocking the stellate ganglion does decrease pro-inflammatory cytokine production and increase anti-inflammatory cytokine production, the mechanism by which this occurs has not been investigated in this work.
Secondly, we chose multiple time points (preoperative baseline, immediately before the block, 15 min after the block, 35 min after the block, and out of the resuscitation room) to monitor the hemodynamics for safety reasons because early stellate ganglion block is a type of cervical sympathetic nerve block and there are not many studies on its application in general anesthesia before. The 15 min after the block was chosen mainly because of the large hemodynamic fluctuations during induction. The 35 min after the block was chosen primarily to observe the hemodynamic effects of stellate ganglion block in the presence of a long waiting period for the start of the operation. According to our findings, the application of SGB in laparoscopic partial hepatectomy will not only have no negative impact on hemodynamics due to blocking sympathetic nerves, but also stabilize the fluctuation of blood pressure during the operation. This may be one of the reasons why vasoactive drugs are used less in group S, where blood pressure is relatively stable, so the number of cases requiring vasoactive drugs is naturally small. This is consistent with the findings of Yong-Quan Chen et al., who discovered that stellate ganglion block stabilizes hemodynamics in elderly patients undergoing carbon dioxide pneumoperitoneum [13].
Furthermore, with regard to the guidance of both Diagnosis Related Groups (DRGs) and The Chinese Expert Consensus on Enhanced Recovery after Hepatectomy (2017 edition), our study statistics from the day of surgery shows that patients in group S exhibited a reduced length of hospital stay and incurred lower hospital expenses compared to those in group N. Although it has been reported that stellate ganglion block can reduce the surgical inflammatory response, reduce surgical stress [8], promote postoperative recovery of gastrointestinal function [19], relieve postoperative pain and reduce arrhythmia [7], the effect on hospital stays has been reported on very seldom. Since IL-6 levels at T3 and T4 varied across groups, we used a simple linear regression analysis to determine whether or not there was a link between the length of stay and hospital costs of the two groups of patients on the day of the surgery and the primary result. According to our findings, stellate ganglion block has a significant impact on the length of stay (calculated from the day of operation) and hospitalization costs (calculated from the day of operation) of patients by influencing the change in IL-6 concentration at T4, and there is a certain linear relationship. This might provide a theoretical study for selecting the concentration of IL-6 24 h after the operation as a visual evaluation index of the prognosis after SGB. After all, according to the existing literature on SGB and inflammatory response, SGB can improve postoperative cognitive dysfunction, and its mechanism may be through the regulation of silent information regular 1 (SIRT1) -mediated neuroinflammation after SGB and correcting white matter damage to improve postoperative cognitive dysfunction [20–22]. SGB can improve postoperative atrial fibrillation. The mechanism may be that it can regulate the balance of autonomic nervous system and reduce systemic and local inflammatory response represented by IL-6, and finally improve postoperative atrial fibrillation [23–25]. SGB improves postoperative gastrointestinal function, and its mechanism may be to regulate the excessive release of systemic and local pro-inflammatory factors to improve postoperative gastrointestinal function [20, 26]. SGB can relieve postoperative inflammatory pain. In addition, inflammatory cytokines have been shown in the literature to influence the occurrence and recovery of postoperative delirium [27], perioperative hemodynamics [28], postoperative liver disease prognosis [28], postoperative wound prognosis and revascularization [29], and other important indicators of surgical recovery. As we all know, TNF-α and IL-6 are the main triggers of inflammatory factor storms within a few minutes after the beginning of hepatectomy, and most liver operations require hepatic portal occlusion [30], which will cause a certain degree of ischemia-reperfusion injury, and ischemia-reperfusion injury will produce a large number of inflammatory cytokines [17, 31]. Another interesting point is that IL-6 plays a pivotal role in facilitating the mitosis and survival of hepatocytes; while maintaining the homeostasis of hepatocytes, and aberrant release of IL-6 can trigger inflammatory damage of the liver [32, 33]. The involvement of the IL-6 trans-signaling is indispensable for liver regeneration after partial hepatectomy. Previous studies suggested that high levels of IL-6 could potentially promote the liver regeneration pathway. However, current research indicates that low levels of IL-6 accompanied by elevated soluble IL-6 receptors can actually facilitate the IL-6 trans-signaling. It is important to note that excessive levels of IL-6 may lead to abnormal hepatocyte proliferation and exacerbate tissue damage through the classic signaling [34]. Therefore, it is still necessary to control the excessive increase of IL-6 level after surgery. In our study, it is of certain significance to select the concentration of IL-6 24 h after operation as the visual evaluation index of the prognosis of SGB after this operation. If this condition is established, it will create more favorable factors for us to apply the stellate ganglion block guided by ultrasound to a laparoscopic partial hepatectomy. After all, this is a clinically easy indicator to detect.
Regarding SGB, there is no observed impact on postoperative complications; however, a disparity in hospital stay duration exists. This discrepancy may be attributed to individual cases with prolonged hospitalization due to specific complications, although our analysis solely accounted for the number of complications. Additionally, it is plausible that the sample size of postoperative complications as a secondary outcome might have limitations. In terms of liver function comparison, no significant findings were obtained possibly due to ethical considerations influenced by the administration of liver protective drugs after surgery.
Limitations
First, our selected patients with laparoscopic partial hepatectomy belong to the middle-aged group, and their age span is relatively large, which may have some influence on the results of postoperative inflammatory cytokines and complications. Secondly, we did not further refine the choice of laparoscopic partial hepatectomy. We included all the patients who had undergone partial hepatectomy in our hospital and met the inclusion criteria. Although this is consistent with our initial desire to study the use of ultrasound-guided stellate ganglion block in patients undergoing major surgical procedures such as laparoscopic partial hepatectomy, I believe that if we can refine the study of a small class of partial hepatectomy, we should be able to get more positive results. The third potential confounding factor in our research is that Horner syndrome will manifest itself after SGB, making it possible for patients and anesthesiologists who follow up on anesthetic work to be aware of the grouping condition.
Conclusion
Ultrasound-guided stellate ganglion block plays a positive role in the enhanced recovery of patients after laparoscopic partial hepatectomy, which mainly shows that it can make the level of inflammatory cytokines more stable, the perioperative hemodynamics more stable, and reduce the length of stay and hospitalization expenses. The serum IL-6 level within 24 h after surgery may be used as a predictor of hospitalization.
Acknowledgements
We thank our colleagues in the Department of hepatobiliary surgery for their help.
Author contributions
Substantial contribution to conception and design: WL, SS, ZC. Patient recruitment: WL, SS, LH, SY, MG, HD, ZC, GZ. Study conduct: WL, SS, SY, MG, HD, ZC, GZ. Data collection: WL, SS, LH, ZC, GZ. Data analysis: WL, SY, MG, HD. Drafting the article: WL, SS. Revising it critically for important intellectual content: WL, LH, SY, MG, HD. Approval of final version: WL, SS, LH, SY, MG, HD, GZ, ZC.
Funding
Our study was supported by grants from the Zhejiang Medical and Health Science and Technology Program (2021KY361) and the Clinical Research Fund Project of the Zhejiang Rehabilitation Medical Association (2021KF016).
Data Availability
Data and materials related to this study can be obtained from the corresponding authors.
Declarations
Ethics approval and consent to participate
The Ethics Committee of Shaoxing People’s Hospital in Shaoxing, China (Chairperson Prof Bao-chun Lu) approved this study (Ethical Committee N°2020-K-Y-185-01) on 24 July 2020. The study protocol adheres to the ethical requirements of the 1975 Declaration of Helsinki (6th reversion, 2008) as shown in a priori approval by the institution’s human research committee. All participants gave written informed consent and completed the questionnaires.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Wei-long Lao and Shuang Sang are co-first authors.
References
- 1.Gorgec B, Benedetti Cacciaguerra A, Lanari J, et al. Assessment of Textbook Outcome in Laparoscopic and Open Liver Surgery. JAMA Surg Aug. 2021;1(8):e212064. doi: 10.1001/jamasurg.2021.2064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kawaguchi Y, Fuks D, Kokudo N, et al. Difficulty of laparoscopic liver resection: proposal for a new classification. Ann Surg Jan. 2018;267(1):13–7. doi: 10.1097/SLA.0000000000002176. [DOI] [PubMed] [Google Scholar]
- 3.Moris D, Vernadakis S. Laparoscopic hepatectomy for Hepatocellular Carcinoma: the opportunities, the challenges, and the limitations. Ann Surg Jul. 2018;268(1):e16. doi: 10.1097/SLA.0000000000002458. [DOI] [PubMed] [Google Scholar]
- 4.Zhu P, Liao W, Zhang WG et al. A prospective study using propensity score matching to compare long-term survival outcomes after Robotic-assisted, laparoscopic or open liver resection for patients with BCLC stage 0-A Hepatocellular Carcinoma. Ann Surg Jan 25 2022. [DOI] [PubMed]
- 5.Liu LD, Duricka DL. Stellate ganglion block reduces symptoms of long COVID: a case series. J Neuroimmunol Jan. 2022;15:577784. doi: 10.1016/j.jneuroim.2021.577784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li Y, Du HB, Jiang LN, et al. Stellate ganglion Block improves the proliferation and function of Splenic CD4 + T cells through inhibition of posthemorrhagic shock mesenteric lymph-mediated autophagy. Inflamm Dec. 2021;44(6):2543–53. doi: 10.1007/s10753-021-01523-x. [DOI] [PubMed] [Google Scholar]
- 7.Wen S, Chen L, Wang TH, et al. The efficacy of ultrasound-guided stellate ganglion block in alleviating postoperative pain and ventricular arrhythmias and its application prospects. Neurol Sci Aug. 2021;42(8):3121–33. doi: 10.1007/s10072-021-05300-4. [DOI] [PubMed] [Google Scholar]
- 8.Zhu G, Kang Z, Chen Y, et al. Ultrasound-guided stellate ganglion block alleviates stress responses and promotes recovery of gastrointestinal function in patients. Dig Liver Dis May. 2021;53(5):581–6. doi: 10.1016/j.dld.2020.11.028. [DOI] [PubMed] [Google Scholar]
- 9.Jin F, Li XQ, Tan WF, et al. Effects of ultrasound-guided stellate-ganglion block on sleep and regional cerebral oxygen saturation in patients undergoing Breast cancer surg ery: a randomized, controlled, double-blinded trial. J Clin Monit Comput Oct. 2018;32(5):855–62. doi: 10.1007/s10877-017-0074-3. [DOI] [PubMed] [Google Scholar]
- 10.Xie A, Zhang X, Ju F, et al. Effects of the Ultrasound-guided stellate ganglion block on hemodynamics, stress response, and gastrointestinal function in postoperative patients with Colorectal Cancer. Comput Intell Neurosci. 2022;2022:2056969. doi: 10.1155/2022/2056969. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 11.Savastano S, Dusi V, Baldi E, et al. Anatomical-based percutaneous left stellate ganglion block in patients with drug-refractory electrical Storm and structural Heart Disease: a single-centre case series. Europ ace Apr. 2021;6(4):581–6. doi: 10.1093/europace/euaa319. [DOI] [PubMed] [Google Scholar]
- 12.Fischer L, Barop H, Ludin SM, et al. Regulation of acute reflectory hyperinflammation in viral and other Diseases by means of stellate ganglion block. A conceptual view with a focus on Covid-19. Auton Neurosci Jan. 2022;237:102903. doi: 10.1016/j.autneu.2021.102903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chen YQ, Xie YY, Wang B, et al. Effect of stellate ganglion block on hemodynamics and stress responses during CO (2)-pneumoperitoneum in elderly patients. J Clin Anesth Feb. 2017;37:149–53. doi: 10.1016/j.jclinane.2016.12.003. [DOI] [PubMed] [Google Scholar]
- 14.Liu MH, Tian J, Su YP, et al. Cervical sympathetic block regulates early systemic inflammatory response in severe trauma patients. Med Sci Monit Mar. 2013;15:194–201. doi: 10.12659/MSM.883833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhang J, Nie Y, Pang Q, et al. Effects of stellate ganglion block on early brain injury in patients with subarachnoid Hemorrhage: a randomised control trial. BMC Anesthesiol Jan. 2021;20(1):23. doi: 10.1186/s12871-020-01215-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wu CN, Wu XH, Yu DN, et al. A single-dose of stellate ganglion block for the prevention of postoperative dysrhythmias in patients undergoing thoracoscopic Surgery for canc er: a randomised controlled double-blind trial. Eur J Anaesthesiol Apr. 2020;37(4):323–31. doi: 10.1097/EJA.0000000000001137. [DOI] [PubMed] [Google Scholar]
- 17.Hwang W, Lee J. Pathophysiologic implications of cytokines secretion during liver transplantation Surgery. Int J Med Sci. 2018;15(14):1737–45. doi: 10.7150/ijms.28382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lipov E, Gluncic V, Lukic IK, et al. How does stellate ganglion block alleviate immunologically-linked disorders? Med Hypotheses Nov. 2020;144:110000. doi: 10.1016/j.mehy.2020.110000. [DOI] [PubMed] [Google Scholar]
- 19.Peng K, Zhang J, Chen WR, et al. Ultrasound-guided stellate ganglion Block improves gastrointestinal function after Thoracolumbar spinal Surgery. Clin Ther Nov. 2017;39(11):2322–30. doi: 10.1016/j.clinthera.2017.09.008. [DOI] [PubMed] [Google Scholar]
- 20.Chen W, Chen B, Wang F, et al. Clinical study of Stellate Ganglion Block combined with General Anesthesia on Hemodynamics, cognitive function, and gastrointestinal function in Elderly patients undergoing partial Hepatectomy. Evid Based Complement Alternat Med. 2021;2021:1426753. doi: 10.1155/2021/1426753. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 21.Li TT, Wan Q, Zhang X, et al. Stellate ganglion block reduces inflammation and improves neurological function in diabetic rats during ischemic Stroke. Neural Regen Res Sep. 2022;17(9):1991–7. doi: 10.4103/1673-5374.335162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang J, Liu Y, Li H, et al. Stellate ganglion Block improves postoperative cognitive dysfunction in aged rats by SIRT1-mediated White Matter Lesion Repair. Neurochem Res Dec. 2022;47(12):3838–53. doi: 10.1007/s11064-022-03800-z. [DOI] [PubMed] [Google Scholar]
- 23.Zafeiropoulos S, Doundoulakis I, Farmakis IT, et al. Autonomic neuromodulation for Atrial Fibrillation following cardiac Surgery: JACC Review topic of the Week. J Am Coll Cardiol Feb. 2022;22(7):682–94. doi: 10.1016/j.jacc.2021.12.010. [DOI] [PubMed] [Google Scholar]
- 24.Zhang D, Hu W, Tu H, et al. Macrophage depletion in stellate ganglia alleviates cardiac sympathetic overactivation and ventricular arrhythmogenesis by attenuating neuroinflammation in Heart Failure. Basic Res Cardiol Apr. 2021;21(1):28. doi: 10.1007/s00395-021-00871-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ajijola OA, Hoover DB, Simerly TM et al. Inflammation, oxidative stress, and glial cell activation characterize stellate ganglia from humans with electrical Storm. JCI Insight Sep 21 2017;2(18). [DOI] [PMC free article] [PubMed]
- 26.Raut MS, Maheshwari A. Stellate ganglion block: important Weapon in the anesthesiologists’ Armamentarium. J Cardiothorac Vasc Anesth Apr. 2018;32(2):e36–7. doi: 10.1053/j.jvca.2017.03.005. [DOI] [PubMed] [Google Scholar]
- 27.Noah AM, Almghairbi D, Evley R, et al. Preoperative inflammatory mediators and postoperative delirium: systematic review and meta-analysis. Br J Anaesth Sep. 2021;127(3):424–34. doi: 10.1016/j.bja.2021.04.033. [DOI] [PubMed] [Google Scholar]
- 28.Bezinover D, Kadry Z, McCullough P, et al. Release of cytokines and hemodynamic instability during the reperfusion of a liver graft. Liver Transpl Mar. 2011;17(3):324–30. doi: 10.1002/lt.22227. [DOI] [PubMed] [Google Scholar]
- 29.Sapienza P, Mingoli A, Borrelli V, et al. Different inflammatory cytokines release after open and endovascular reconstructions influences wound healing. Int Wound J Aug. 2019;16(4):1034–44. doi: 10.1111/iwj.13154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Murtha-Lemekhova A, Fuchs J, Ghamarnejad O, et al. Influence of cytokines, circulating markers and growth factors on liver regeneration and post-hepatectomy Liver Failure: a systematic review and meta-analysis. Sci Rep Jul. 2021;2(1):13739. doi: 10.1038/s41598-021-92888-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sosa RA, Zarrinpar A, Rossetti M, et al. Early cytokine signatures of ischemia/reperfusion injury in human orthotopic liver transplantation. JCI Insight Dec. 2016;8(20):e89679. doi: 10.1172/jci.insight.89679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Schmidt-Arras D, Rose-John S. IL-6 pathway in the liver: from physiopathology to therapy. J Hepatol. 2016;64(6):1403–15. doi: 10.1016/j.jhep.2016.02.004. [DOI] [PubMed] [Google Scholar]
- 33.Wang J, Zhang X, Han J, et al. MicroRNA-124 expression in Kupffer cells modulates liver injury by targeting IL-6/STAT3 signaling. Antiviral Res. 2023;211:105510. doi: 10.1016/j.antiviral.2022.105510. [DOI] [PubMed] [Google Scholar]
- 34.Fazel Modares N, Polz R, Haghighi F, et al. IL-6 trans-signaling Controls Liver Regeneration after partial Hepatectomy. Hepatology. 2019;70(6):2075–91. doi: 10.1002/hep.30774. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data and materials related to this study can be obtained from the corresponding authors.