Abstract
After a stroke, several mechanisms of neural plasticity can be activated, which may lead to significant recovery. Rehabilitation therapies aim to restore surviving tissue over time and reorganize neural connections. With more patients surviving stroke with varying degrees of neurological impairment, new technologies have emerged as a promising option for better functional outcomes. This review explores restorative therapies based on brain-computer interfaces, robot-assisted and virtual reality, brain stimulation, and cell therapies. Brain-computer interfaces allow for the translation of brain signals into motor patterns. Robot-assisted and virtual reality therapies provide interactive interfaces that simulate real-life situations and physical support to compensate for lost motor function. Brain stimulation can modify the electrical activity of neurons in the affected cortex. Cell therapy may promote regeneration in damaged brain tissue. Taken together, these new approaches could substantially benefit specific deficits such as arm-motor control and cognitive impairment after stroke, and even the chronic phase of recovery, where traditional rehabilitation methods may be limited, and the window for repair is narrow.
Keywords: Neuronal plasticity, Biomedical technology, Rehabilitation, Stroke, Stroke rehabilitation
Introduction
In clinical practice, the number of stroke patients is increasing due to the aging of the population and the high prevalence of cardiovascular comorbidities [1]. This is directly correlated with the growing number of stroke survivors who receive acute therapies, such as thrombectomy or pharmacological reperfusion. Early recognition of stroke has led to a larger population of patients who can survive and be discharged from hospitals with less severe neurological impairment. However, a significant proportion of patients do not receive acute therapies due to contraindications or being outside the reperfusion time window, and a significant proportion of patients survive with severe neurological deficits thanks to life-support systems. For these reasons, stroke is a major cause of disability worldwide and the leading neurological cause of lost disability-adjusted life years [2].
Traditionally, rehabilitation for patients with persisting deficits has focused on physical, occupational, and speech-language therapy, as well as the prevention of medical complications [3]. However, new rehabilitation strategies are now part of standard care and include activity-based therapy such as constraint-induced movement therapy, high-dose activity-based rehabilitation, high doses of task-specific training, mirror therapy, and environmental enrichment [4]. Restorative therapies targeting neuroplasticity mechanisms with recent technological advances have emerged as a promising tool for the integral care of patients with special needs. This review addresses the neural basis for stroke recovery and new approaches, including brain-computer interfaces (BCIs), virtual reality, robot-assisted rehabilitation, cell therapies, and brain stimulation. A comprehensive search was conducted in the PUBMED/Medline and SCOPUS databases for articles published in the last 10 years using the terms “robot/virtual reality/brain-computer interface/cell therapy/brain stimulation” combined with the terms “stroke/brain ischemia/rehabilitation/neuroplasticity” in English. Relevant articles and their bibliographic references were included based on the authors’ criteria for clinical practice relevance.
Neuroplasticity after stroke
Neuroplasticity is the ability of the nervous system to modify and regenerate in response to new information or damage. Until recently, it was believed that neuroplasticity was nonexistent in adulthood. However, it has been found that neuroplasticity occurs spontaneously throughout life, although these changes are not sufficient to produce evident recovery after brain damage. Therefore, there are now several strategies available to enhance neuroplasticity, including both pharmacological and non-pharmacological interventions that are essential for post-stroke rehabilitation [5–7].
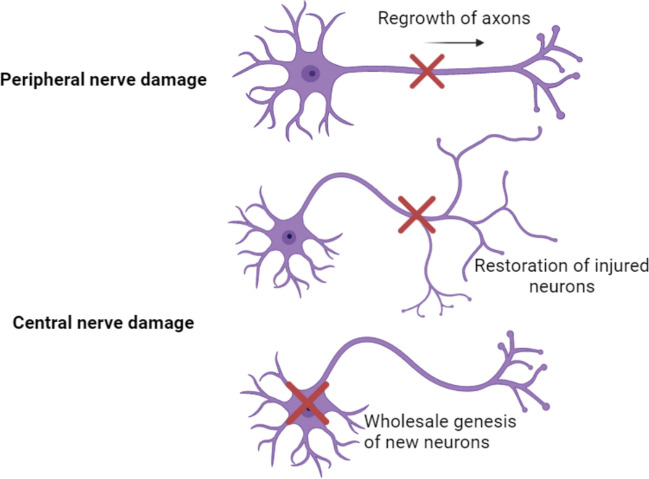
The current understanding of neuroplasticity is based on Hebb’s theory from 1949, which posits that repetitive stimulation of the postsynaptic neuron by the presynaptic neuron is necessary to increase synaptic efficacy [8]. There are three main mechanisms of neuroplasticity in healthy brains. The first is the regrowth of axons after peripheral nerve damage. The second is the restoration of injured central nerve cells through the growth of new dendrites, axons, and synapses from existing cell bodies. The third mechanism is the wholesale generation of new neurons, which occurs in two neurogenic regions: the subventricular zone and the dentate gyrus. In the subventricular zone, neuroblasts are generated and migrate along the rostral migratory path to the olfactory bulb, where they become granular and periglomerular interneurons involved in plastic processes of olfactory learning. In the dentate gyrus, stem cells give rise to neuronal precursors that mature and generate new granular neurons [9, 10] (Fig. 1).
Fig. 1.
Mechanisms of neuroplasticity in healthy individuals
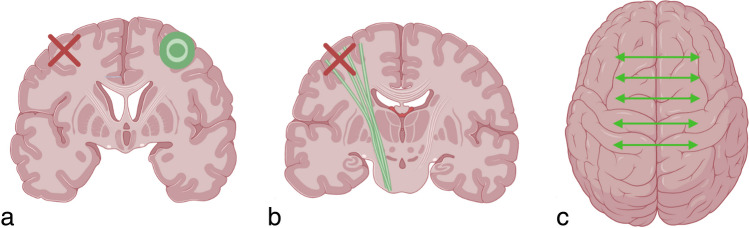
Currently, three major mechanisms have been described for how neuroplasticity works after a stroke. These mechanisms begin in the early stages after the event and continue for at least 3–6 months, leading to the reorganization of neural connections [11, 12]. The first mechanism involves increased functional activity in the somatosensory system on the opposite side of the brain from the infarction, as well as recruitment from distant cortical regions connected to the affected area [13, 14] (Fig. 2a). The second mechanism involves the improvement of the structural integrity of the corticospinal tract on the same side of the brain as the infarction [15] (Fig. 2b). The third mechanism involves the restoration of interhemispheric functional connectivity and the network of the sensorimotor cortex on both sides of the brain [16] (Fig. 2c). As a result, there is a reallocation of functions whose primary representation has been damaged [11].
Fig. 2.
Neuroplasticity after stroke
Brain-computer interfaces
A BCI is a modern tool that allows for the transduction of brain signals into computational commands, enabling the control of technological devices. The positive effects of BCI have been discovered to mainly stimulate neuroplasticity and assist patients with loss of motor function, leading to hopeful clinical implications for post-stroke patients and individuals with other neurological diseases [17–20]. The operation of BCI is based on three main components: signal reception, signal processing, and generation of a specific response in a machine. Multiple methods for developing each of these components have been reported [18, 20–23].
There are two methods for the reception of brain signals, invasive and non-invasive. The invasive method, which may produce a cleaner signal, carries the risks of surgery and is not always the most suitable for clinical use. There are three types of invasive electrodes, including cortical surface microelectrodes (ECoG), cortical penetrating microelectrodes, and deeply penetrating electrodes, which record different characteristics of brain action potentials. Non-invasive approaches, such as electroencephalography (EEG), are more commonly used and have the most scientific evidence to support their effectiveness. However, alternative methods like near-infrared spectroscopy have also been reported [18, 20].
The second step in the BCI process involves filtering the received brain signals using specific techniques to enable interpretation. The commonly used techniques include sensorimotor rhythms, slow cortical potentials, event-related potentials/P300, and visual evoked potentials. The filtered signals are then transformed into voltage/time frequencies using processing techniques such as Fourier, common spatial filter, and wavelet transform. These signals are further analyzed through classification algorithms, which generate a specific command for the machine to execute [18, 20].
The final step in BCI involves generating a response using a device that is programmed to receive the signal and perform a function, such as basic motor patterns to improve rehabilitation and quality of life [18, 20]. In stroke patients, BCI-assisted rehabilitation is an alternative approach that aims to stimulate neuroplasticity by manipulating or self-regulating brain activity, resulting in motor cortical reorganization and changes in motor activation ipsilateral and contralateral to the lesion. BCI has been found to be superior to several types of conventional therapy, as reflected in motor connectivity in fMRI, increased event-related desynchronization activity in the EEG, and increased volitional contraction in electromyographic activity in affected muscles. These, along with other changes not measurable with clinical scales, suggest a promising mechanism for managing these patients [17]. However, the physiological basis of this recovery has not yet been fully elucidated, nor have the factors that influence better outcomes with these therapies, making it an area with great potential for future applications in clinical settings [18, 24, 25].
Robot-assisted rehabilitation and virtual reality
Virtual reality (VR) therapy and robot-assisted therapies (RAT) are emerging technologies that have shown promise as alternative treatments for improving motor function and quality of life [26–29]. VR therapy involves the use of an interface between the computer and the patient, using both hardware and software to simulate interactions with the environment. This allows for the creation of sensory connections that closely mimic reality, with the added benefits of simultaneous task execution and immediate feedback [26, 30]. VR systems are specifically designed to aid in the rehabilitation of patients with neurocognitive impairments, using specialized interfaces that focus on developing functional skills that can be applied in the real world [30, 31].
RAT have shown promise in training lost motor function or compensating for lost skills after stroke. The use of robots in stroke rehabilitation has led to positive trends in motor improvement. For instance, an exoskeleton or a robotic hand can be used to rehabilitate gait and upper limb motor function, respectively. These devices support the patient’s movements in different axes and provide better control and monitoring for specific tasks and patient needs. However, the impact of RAT depends on factors such as the type of support provided to the limb, the patient’s ability to execute the movement, the duration of robot support, and the type of exercise. Additionally, limited research has been conducted on the impact of therapy performed solely with robots, and it is more feasible to use RAT in combination with other techniques such as VR therapy to improve overall motor and cognitive performance [32, 33].
In VR therapy, there are different levels of immersion that can be utilized based on the patient’s needs. Skillful immersion focuses on improving specific motor skills, while strategic immersion aims to improve higher-level cognitive skills such as decision-making and problem-solving. Narrative immersion uses storytelling to provide a context for the exercises and increase engagement, while spatial or total immersion involves creating a completely immersive virtual environment for the patient to interact with. Total immersion is the most immersive type of VR therapy, providing the patient with a fully immersive experience where they are transported to a virtual world or scenario. This can be helpful in providing a distraction from the real world, while still allowing the patient to engage in meaningful activities that improve physical and cognitive function. The use of a virtual scenario with a specific story can also provide emotional and psychological benefits for the patient. Additionally, performance can be monitored and measured in real time, and feedback can be given to the patient to help them improve [26, 27, 30].
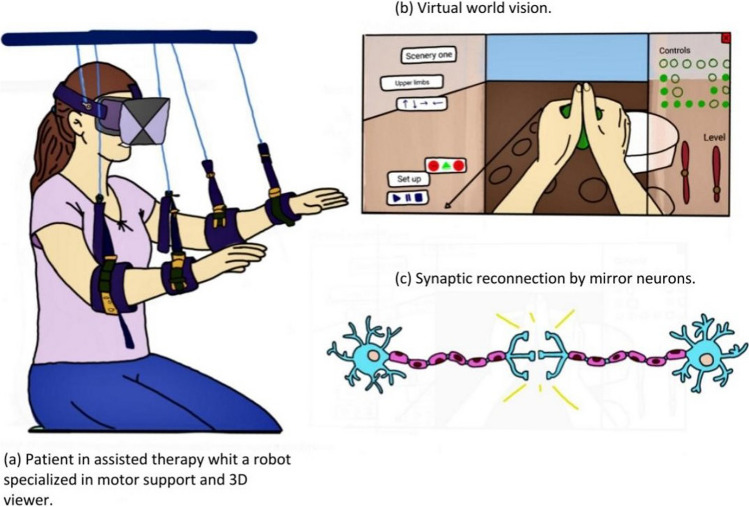
VR therapy is an effective way to provide game-based training tasks to patients with motor deficits. These tasks, such as grasping and releasing a virtual ball, are designed to stimulate and activate mirror neurons. As shown in Fig. 3, patients wear a head-mounted display and use movement controllers to interact with the virtual environment. After 4 weeks of treatment, mirror neuron VR rehabilitation (MNVR-Rehabs) has been shown to promote neuroplasticity in injured brain areas and lead to functional performance improvement. Resting fMRI and Fugl-Meyer assessment support the effectiveness of MNVR-Rehabs in reorganizing brain and motor function [34].
Fig. 3.
Setup and scenario of VR-Rehab system: a the patient can be seen wearing a head-mounted display (HMD) headset and a 3D viewer and monitored by two infrared cameras that track her exact position in the virtual world. The patient also has movement controllers in the form of purple bands on their arms, equipped with sensors that track their gestures and replicate them graphically within the virtual world. b The therapist can monitor the patient’s performance through the PC, which displays the virtual world. The illustration shows a typical exercise in which the patient must move a virtual ball to a virtual basket using their virtual upper limb. c Through immersion in virtual reality and performing these exercises, the patient’s mirror neurons are stimulated, which through repetition contributes to the formation of new synaptic connections and the overall improvement of their physical condition
The results of rehabilitation using VR and robot-assisted therapies can be influenced by differences in intensity, duration, frequency, and personalization. These modalities do not impact overall cognitive function, verbal fluency, or attention compared to the control groups, but they do lead to significant improvements in dynamic balance, executive function, memory, motor function, visuospatial ability, and quality of life [27, 35]. Balance improvement is closely tied to the visual, vestibular, and somatosensory systems, which can be targeted through VR therapy to stimulate and practice postural control in a safe and controlled environment, ultimately reducing fall risk and enhancing cognitive processes [36]. Multidisciplinary rehabilitation that includes immersion therapy using robot assistance can further improve gait control parameters and offer greater control during therapy [26, 28, 32].
Furthermore, the use of robot-assisted therapy has been shown to result in improvements in upper limb motor function, spasticity, and overall functioning, leading to better performance of daily activities. Additionally, patients who received RAT showed better results in cognitive evaluations compared to the control groups. While VR therapy has shown potential for future treatments in stroke rehabilitation, it did not show statistically significant differences in cognitive function compared to the control groups [31].
Cell therapies
The brain’s limited repair capacity has led to the development of new strategies for enhancing brain plasticity, including cell therapy. Stem cells have the potential to differentiate into multiple mature and immature cell types, and therefore have the ability to promote regeneration in damaged brain tissue [37, 38]. Various cell lines have been explored for the treatment and rehabilitation of stroke, including embryonic stem cells (ESCs), neural stem cells (NSCs), bone marrow mononuclear cells (BMMCs), mesenchymal stem cells (MSCs), induced pluripotent stem cells (iPSCs), and others. Among these, MSCs are particularly promising due to their low immunogenicity, easy availability, and positive results in animal models [37, 38]. MSCs are characterized by their adherence to plastic matrix-cultures, fibroblast-like morphology, expression of surface antigens (CD44, CD90, CD29), and ability to differentiate into mesodermal and neural cells [37, 39].
Multiple animal studies have demonstrated that stem cell therapy can improve post-stroke functionality by stimulating various mechanisms, including neuron replacement, trophic stimulus, angiogenesis promotion, remyelination induction, and cell protection [38]. Three main theories have been proposed to explain MSC-mediated brain repair, including “trans”-differentiation, cell fusion, and the paracrine effect, through the release of soluble trophic factors [37, 40]. While there is evidence for all of these phenomena, it is uncertain how much each contributes to rehabilitation and functional improvement [40]. Among these, the release of trophic factors seems to play a significant role in the MSC-mediated brain repair response after stroke. The effects of these factors can be classified as angiogenic, neurogenic, protective, synaptogenic, and prevention of pathological scarring [40, 41]. The immune regulation and tolerance provided by these soluble factors can prevent and regulate the secondary inflammatory response, which can be more harmful than protective. In addition, in vitro studies have shown that MSCs can increase the expression of angiogenic factors such as vascular endothelial growth factor (VEGF) and brain-derived neurotrophic factor (BDNF). Exosomes, which contain DNAs, RNAs, mRNAs, peptides, and other bioactive molecules, have also been found to play a crucial role in MSC-mediated brain repair by stimulating nerve repair, reducing inflammation, and promoting successful remodeling [42]. In fact, the delivery of exosomes may be comparable to MSC transplantation.
There have been human studies on stem cell therapy [43]. One study published in 2019 focused on the safety of intravenous doses of allogeneic ischemia-tolerant mesenchymal stem cells and the potential behavioral changes after therapy. The study concluded that intravenous transfusion of allogeneic ischemia-tolerant mesenchymal stem cells was safe for patients with chronic stroke and significant functional deficits, and suggested possible behavioral improvements. However, the results should be evaluated further with a randomized, placebo-controlled study [44]. Another study, called the PISCES-2 study, used direct intracerebral implantation of neural stem cells by stereotaxic injection to the putamen ipsilateral to the cerebral infarct. The study aimed to observe upper limb movement improvement, but the clinical enhancement was only seen in those with residual upper limb movement at baseline [45]. The RECOVER stroke trial used internal carotid infusion of autologous bone marrow-derived aldehyde dehydrogenase-bright stem cell and found no allergic reactions or adverse events related to the cell therapy, but there was no significant difference between the intervention and placebo groups in the modified Rankin scale (mRS) at 3 months [45, 46]. Another randomized clinical trial, called the ISIS-HERMES study, evaluated the safety, efficacy, and feasibility of intravenous autologous bone marrow-derived MSCs infusion in subacute stroke patients (< 2 weeks after diagnosis). The study found no differences in the Barthel Index, NIHSS, and mRS, but there were significant improvements in motor recovery evidenced with motor-NIHSS and motor-Fugl-Meyer score. The study also observed that intravenous MSCs treatment was safe [47]. These studies indicate that we are getting closer to identifying a consistent, secure, and efficient method for using cell therapy in stroke recovery and rehabilitation, particularly with MSCs. However, there is still a need for further research to demonstrate a strong effect in actual clinical practice.
Brain stimulation
Non-invasive brain stimulation (NIBS) is a widely used technique in many fields, particularly in stroke rehabilitation [48]. Both transcranial magnetic stimulation (TMS) and transcranial direct current stimulation (TDCS) have demonstrated improved functional outcomes and responses in individuals who have experienced a stroke. These techniques have been observed to induce changes in long-term neuroplasticity, modulating local and distant networks that underlie various clinical symptoms resulting from stroke [49–52].
TMS is a technique that uses electromagnetic induction to create an electric current which can modulate the electrophysiological activity of cells, leading to neuron depolarization and action potential. The frequency of the pulses can alter cortical activity by reducing the permeability of the neurovascular complex, thereby improving cerebral perfusion and angiogenesis [53]. TMS can also modulate cortical excitability (CE), with low-frequency TMS (<1 Hz) decreasing excitability (commonly used in the contralateral hemisphere to inhibit activity) and high-frequency TMS (>1 Hz) increasing excitability (used in the affected area to stimulate activity) [54, 55]. The primary motor cortex, parietal cortex, and Broca-Wernicke areas are commonly treated with TMS [56–59]. There is a subtype of repetitive TMS called theta burst stimulation (TBS), which can enhance in intermittent mode (iTBS) or depress in continuous mode (cTBS) and has been used in stroke recovery; beyond its original description in primary motor areas, there are publications on TBS in the cerebellum, given its parietal-frontal connections, showing improvement in motor learning [60, 61].
TDCS generates a weak direct electrical constant current (1–2 mA) that modulates CE, stimulates neuroplasticity, improves local blood flow, and has an effect over nearby areas and connectivity; the anode depolarizes the resting potential (potentiating CE), while the cathode hyperpolarizes it (inhibiting CE) [62–65]. In studies using TDCS, current intensities of 1, 1.5, and 2 mA have been applied for a duration of 20 min. Anodic TDCS stimulates the affected primary motor cortex, while cathodic TDCS stimulates the contralateral primary motor cortex; high intensity and long duration TDCS have been found to yield better results [66].
TMS has been studied for its potential to aid in post-stroke rehabilitation in both acute and subacute cases. Various testing protocols for motor function, neglect, and aphasia have shown better recovery, although the results have been contradictory because the symptoms have not been compared between them or at the same time [67–69]. In contrast, TDCS has had minimal impact on post-stroke rehabilitation in the acute and subacute periods, possibly due to a lack of standardization in trials. However, in chronic scenarios, TDCS has been found to improve motor function as an adjuvant to other classical therapies [70, 71].
TMS is generally considered safe, but adverse events such as seizures, headache, cervicalgia, dizziness, and local discomfort have been reported in a low proportion of cases. Seizures are rare but more common with high-frequency TMS [72–74]. On the other hand, TDCS is better tolerated, with fewer effects such as skin redness, slight tingling, dizziness, and fatigue, depending on the current dose used [75]. In conclusion, NIBS holds promise as an adjuvant therapy for future research and possible translational applications in clinical scenarios [76], particularly for post-stroke rehabilitation [77].
Future and perspectives
Traditionally, the phases of recovery after stroke have been divided into acute (first month), functional or subacute (6 months), and chronic or plateau (first year and thereafter) [78]. However, new approaches to stroke rehabilitation could be incorporated into these different phases and improve the outcomes of standard therapies. For example, cell therapy is a promising intervention for the acute and subacute phases, and may eventually become part of reperfusion therapies. Brain-computer interfaces, transcranial brain stimulation, robot-assisted therapy, and virtual reality can benefit not only the subacute phase but also the chronic phase of recovery, where classical rehabilitation may be limited and the time window for repair is narrow. However, these new approaches should be coupled with training and other restorative therapies for the best results.
The availability of rehabilitation services varies across countries, and many stroke patients suffer from delays and poor quality of rehabilitation services due to limited options and other public health barriers. Although the new technologies described in this paper hold great promise for stroke rehabilitation, they are still in the clinical phase of research and their availability is limited due to their high cost and complexity. However, given the high impact of stroke disability and associated costs, it is important to continue developing and implementing new strategies for stroke recovery.
Conclusions
Stroke disability is an area of increasing interest, as recovery after stroke involves various neural mechanisms of plasticity that can be potentiated with both traditional and new rehabilitation therapies. The emerging approaches for stroke rehabilitation include brain-computer interfaces, robot-assisted therapy coupled with virtual reality, brain stimulation, and cell therapy. Although ongoing research is still exploring the impact of these modalities on stroke recovery, promising results suggest that these new therapies could lead to better functional outcomes for stroke patients.
Funding
Open Access funding provided by Colombia Consortium Own of the authors.
Declarations
Ethical approval
This manuscript is a review article and does not involve a research protocol requiring approval by the relevant institutional review board or ethics committee.
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.GBD Stroke Collaborators. Global, regional, and national burden of stroke and its risk factors, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 2021;20(10):795–820. doi: 10.1016/S1474-4422(21)00252-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Johnston SC, Claiborne Johnston S, Hauser SL. Neurological disease on the global agenda. Ann Neurol. 2008;64(1):A11–A12. doi: 10.1002/ana.21477. [DOI] [PubMed] [Google Scholar]
- 3.Winstein CJ, Stein J, Arena R, Bates B, Cherney LR, Cramer SC, et al. Guidelines for adult stroke rehabilitation and recovery: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2016;47(6):e98–e169. doi: 10.1161/STR.0000000000000098. [DOI] [PubMed] [Google Scholar]
- 4.Cramer SC. Recovery after stroke. Continuum (Minneap Minn) 2020;26(2):415–434. doi: 10.1212/CON.0000000000000838. [DOI] [PubMed] [Google Scholar]
- 5.Mensen A, Pigorini A, Facchin L, Schöne C, D’Ambrosio S, Jendoubi J, et al. Sleep as a model to understand neuroplasticity and recovery after stroke: observational, perturbational and interventional approaches. J Neurosci Methods. 2019;313:37–43. doi: 10.1016/j.jneumeth.2018.12.011. [DOI] [PubMed] [Google Scholar]
- 6.Rahayu UB, Wibowo S, Setyopranoto I, Hibatullah RM. Effectiveness of physiotherapy interventions in brain plasticity, balance and functional ability in stroke survivors: a randomized controlled trial. NeuroRehabilitation. 2020;47(4):463–470. doi: 10.3233/NRE-203210. [DOI] [PubMed] [Google Scholar]
- 7.Khan F, Amatya B, Galea MP, Gonzenbach R, Kesselring J. Neurorehabilitation: applied neuroplasticity. J Neurol. 2017;264(3):603–615. doi: 10.1007/s00415-016-8307-9. [DOI] [PubMed] [Google Scholar]
- 8.Brown RE, Donald O. Hebb and the organization of behavior: 17 years in the writing. Mol Brain. 2020;13(1):55. doi: 10.1186/s13041-020-00567-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kumar A, Pareek V, Faiq MA, Ghosh SK, Kumari C. Adult neurogenesis in humans: a review of basic concepts, history, current research, and clinical implications. Innov Clin Neurosci. 2019;16(5-6):30–37. [PMC free article] [PubMed] [Google Scholar]
- 10.Ribeiro FF, Xapelli S. An overview of adult neurogenesis. Adv Exp Med Biol. 2021;1331:77–94. doi: 10.1007/978-3-030-74046-7_7. [DOI] [PubMed] [Google Scholar]
- 11.Dąbrowski J, Czajka A, Zielińska-Turek J, Jaroszyński J, Furtak-Niczyporuk M, Mela A, et al. Brain functional reserve in the context of neuroplasticity after stroke. Neural Plast. 2019;2019:9708905. doi: 10.1155/2019/9708905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hara Y. Brain plasticity and rehabilitation in stroke patients. J Nippon Med Sch. 2015;82(1):4–13. doi: 10.1272/jnms.82.4. [DOI] [PubMed] [Google Scholar]
- 13.Xing Y, Bai Y. A review of exercise-induced neuroplasticity in ischemic stroke: pathology and mechanisms. Mol Neurobiol. 2020;57(10):4218–4231. doi: 10.1007/s12035-020-02021-1. [DOI] [PubMed] [Google Scholar]
- 14.Cichon N, Wlodarczyk L, Saluk-Bijak J, Bijak M, Redlicka J, Gorniak L, et al. Novel advances to post-stroke aphasia pharmacology and rehabilitation. J Clin Med. 2021;10(17):3778. doi: 10.3390/jcm10173778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sandvig I, Augestad IL, Håberg AK, Sandvig A. Neuroplasticity in stroke recovery. The role of microglia in engaging and modifying synapses and networks. Eur J Neurosci. 2018;47(12):1414–1428. doi: 10.1111/ejn.13959. [DOI] [PubMed] [Google Scholar]
- 16.Guggisberg AG, Koch PJ, Hummel FC, Buetefisch CM. Brain networks and their relevance for stroke rehabilitation. Clin Neurophysiol. 2019;130(7):1098–1124. doi: 10.1016/j.clinph.2019.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ushiba J, Soekadar SR. Brain-machine interfaces for rehabilitation of poststroke hemiplegia. Prog Brain Res. 2016;228:163–183. doi: 10.1016/bs.pbr.2016.04.020. [DOI] [PubMed] [Google Scholar]
- 18.Cervera MA, Soekadar SR, Ushiba J, Millán JDR, Liu M, Birbaumer N, et al. Brain-computer interfaces for post-stroke motor rehabilitation: a meta-analysis. Ann Clin Transl Neurol. 2018;5(5):651–663. doi: 10.1002/acn3.544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Remsik A, Young B, Vermilyea R, Kiekhoefer L, Abrams J, Evander Elmore S, et al. A review of the progression and future implications of brain-computer interface therapies for restoration of distal upper extremity motor function after stroke. Expert Rev Med Devices. 2016;13(5):445–454. doi: 10.1080/17434440.2016.1174572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Burns A, Adeli H, Buford JA. Brain-computer interface after nervous system injury. Neuroscientist. 2014;20(6):639–651. doi: 10.1177/1073858414549015. [DOI] [PubMed] [Google Scholar]
- 21.Liu M, Ushiba J. Brain-machine interface (BMI)-based neurorehabilitation for post-stroke upper limb paralysis. Keio J Med. 2022;71(4):82–92. doi: 10.2302/kjm.2022-0002-OA. [DOI] [PubMed] [Google Scholar]
- 22.Cantillo-Negrete J, Carino-Escobar RI, Carrillo-Mora P, Rodriguez-Barragan MA, Hernandez-Arenas C, Quinzaños-Fresnedo J, et al. Brain-computer interface coupled to a robotic hand orthosis for stroke patients’ neurorehabilitation: a crossover feasibility study. Front Hum Neurosci. 2021;15:656975. doi: 10.3389/fnhum.2021.656975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Behboodi A, Lee WA, Hinchberger VS, Damiano DL. Determining optimal mobile neurofeedback methods for motor neurorehabilitation in children and adults with non-progressive neurological disorders: a scoping review. J Neuroeng Rehabil. 2022;19(1):104. doi: 10.1186/s12984-022-01081-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Carelli L, Solca F, Faini A, Meriggi P, Sangalli D, Cipresso P, et al. Brain-computer interface for clinical purposes: cognitive assessment and rehabilitation. Biomed Res Int. 2017;2017:1695290. doi: 10.1155/2017/1695290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jeunet C, Glize B, McGonigal A, Batail JM, Micoulaud-Franchi JA. Using EEG-based brain computer interface and neurofeedback targeting sensorimotor rhythms to improve motor skills: theoretical background, applications and prospects. Neurophysiol Clin. 2019;49(2):125–136. doi: 10.1016/j.neucli.2018.10.068. [DOI] [PubMed] [Google Scholar]
- 26.Zhang Q, Fu Y, Lu Y, Zhang Y, Huang Q, Yang Y, et al. Impact of virtual reality-based therapies on cognition and mental health of stroke patients: systematic review and meta-analysis. J Med Internet Res. 2021;23(11):e31007. doi: 10.2196/31007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Aramaki AL, Sampaio RF, Reis ACS, Cavalcanti A, Dutra FCMSE. Virtual reality in the rehabilitation of patients with stroke: an integrative review. Arq Neuropsiquiatr. 2019;77(4):268–278. doi: 10.1590/0004-282X20190025. [DOI] [PubMed] [Google Scholar]
- 28.Li Z, Han XG, Sheng J, Ma SJ. Virtual reality for improving balance in patients after stroke: a systematic review and meta-analysis. Clin Rehabil. 2016;30(5):432–440. doi: 10.1177/0269215515593611. [DOI] [PubMed] [Google Scholar]
- 29.Szczepańska-Gieracha J, Cieślik B, Rutkowski S, Kiper P, Turolla A. What can virtual reality offer to stroke patients? A narrative review of the literature. NeuroRehabilitation. 2020;47(2):109–120. doi: 10.3233/NRE-203209. [DOI] [PubMed] [Google Scholar]
- 30.Hao J, Xie H, Harp K, Chen Z, Siu KC. Effects of virtual reality intervention on neural plasticity in stroke rehabilitation: a systematic review. Arch Phys Med Rehabil. 2022;103(3):523–541. doi: 10.1016/j.apmr.2021.06.024. [DOI] [PubMed] [Google Scholar]
- 31.Maier M, Rubio Ballester B, Duff A, Duarte Oller E, Verschure PFMJ. Effect of specific over nonspecific VR-based rehabilitation on poststroke motor recovery: a systematic meta-analysis. Neurorehabil Neural Repair. 2019;33(2):112–129. doi: 10.1177/1545968318820169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Huang X, Naghdy F, Naghdy G, Du H. Clinical effectiveness of combined virtual reality and robot assisted fine hand motion rehabilitation in subacute stroke patients. IEEE Int Conf Rehabil Robot. 2017;2017:511–515. doi: 10.1109/ICORR.2017.8009299. [DOI] [PubMed] [Google Scholar]
- 33.Veerbeek JM, Langbroek-Amersfoort AC, van Wegen EEH, Meskers CGM, Kwakkel G. Effects of robot-assisted therapy for the upper limb after stroke. Neurorehabil Neural Repair. 2017;31(2):107–121. doi: 10.1177/1545968316666957. [DOI] [PubMed] [Google Scholar]
- 34.Mekbib DB, Zhao Z, Wang J, Xu B, Zhang L, Cheng R, et al. Proactive motor functional recovery following immersive virtual reality-based limb mirroring therapy in patients with subacute stroke. Neurotherapeutics. 2020;17(4):1919–1930. doi: 10.1007/s13311-020-00882-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Iruthayarajah J, McIntyre A, Cotoi A, Macaluso S, Teasell R. The use of virtual reality for balance among individuals with chronic stroke: a systematic review and meta-analysis. Top Stroke Rehabil. 2017;24(1):68–79. doi: 10.1080/10749357.2016.1192361. [DOI] [PubMed] [Google Scholar]
- 36.Cho KH, Lee KJ, Song CH. Virtual-reality balance training with a video-game system improves dynamic balance in chronic stroke patients. Tohoku J Exp Med. 2012;228(1):69–74. doi: 10.1620/tjem.228.69. [DOI] [PubMed] [Google Scholar]
- 37.Gutiérrez-Fernández M, Rodríguez-Frutos B, Ramos-Cejudo J, Otero-Ortega L, Fuentes B, Díez-Tejedor E. Stem cells for brain repair and recovery after stroke. Expert Opin Biol Ther. 2013;13(11):1479–1483. doi: 10.1517/14712598.2013.824420. [DOI] [PubMed] [Google Scholar]
- 38.Lindvall O, Kokaia Z. Stem cell research in stroke: how far from the clinic? Stroke. 2011;42(8):2369–2375. doi: 10.1161/STROKEAHA.110.599654. [DOI] [PubMed] [Google Scholar]
- 39.Richards LG, Cramer SC. Advances in stroke: therapies targeting stroke recovery. Stroke. 2021;52(1):348–350. doi: 10.1161/STROKEAHA.120.033231. [DOI] [PubMed] [Google Scholar]
- 40.Niv F, Keiner S, Krishna K, Witte OW, Lie DC, Redecker C. Aberrant neurogenesis after stroke: a retroviral cell labeling study. Stroke. 2012;43(9):2468–2475. doi: 10.1161/STROKEAHA.112.660977. [DOI] [PubMed] [Google Scholar]
- 41.Maltman DJ, Hardy SA, Przyborski SA. Role of mesenchymal stem cells in neurogenesis and nervous system repair. Neurochem Int. 2011;59(3):347–356. doi: 10.1016/j.neuint.2011.06.008. [DOI] [PubMed] [Google Scholar]
- 42.Li W, Shi L, Hu B, Hong Y, Zhang H, Li X, et al. Mesenchymal stem cell-based therapy for stroke: current understanding and challenges. Front Cell Neurosci. 2021;15:628940. doi: 10.3389/fncel.2021.628940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Satani N, Cai C, Giridhar K, McGhiey D, George S, Parsha K, et al. World-wide efficacy of bone marrow derived mesenchymal stromal cells in preclinical ischemic stroke models: systematic review and meta-analysis. Front Neurol. 2019;10:405. doi: 10.3389/fneur.2019.00405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Levy ML, Crawford JR, Dib N, Verkh L, Tankovich N, Cramer SC. Phase I/II study of safety and preliminary efficacy of intravenous allogeneic mesenchymal stem cells in chronic stroke. Stroke. 2019;50(10):2835–2841. doi: 10.1161/STROKEAHA.119.026318. [DOI] [PubMed] [Google Scholar]
- 45.Muir KW, Bulters D, Willmot M, Sprigg N, Dixit A, Ward N, et al. Intracerebral implantation of human neural stem cells and motor recovery after stroke: multicentre prospective single-arm study (PISCES-2) J Neurol Neurosurg Psychiatry. 2020;91(4):396–401. doi: 10.1136/jnnp-2019-322515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Savitz SI, Yavagal D, Rappard G, Likosky W, Rutledge N, Graffagnino C, et al. A phase 2 randomized, sham-controlled trial of internal carotid artery infusion of autologous bone marrow–derived ALD-401 cells in patients with recent stable ischemic stroke (RECOVER-Stroke) Circulation. 2019;139(2):192–205. doi: 10.1161/CIRCULATIONAHA.117.030659. [DOI] [PubMed] [Google Scholar]
- 47.Jaillard A, Hommel M, Moisan A, Zeffiro TA, Favre-Wiki IM, Barbieux-Guillot M, et al. Autologous mesenchymal stem cells improve motor recovery in subacute ischemic stroke: a randomized clinical trial. Transl Stroke Res. 2020;11(5):910–923. doi: 10.1007/s12975-020-00787-z. [DOI] [PubMed] [Google Scholar]
- 48.Richards LG, Cramer SC. Advances in stroke recovery therapeutics. Stroke. 2022;52(1):348–350. doi: 10.1161/STROKEAHA.120.033231. [DOI] [PubMed] [Google Scholar]
- 49.Shen QR, Hu MT, Feng W, Li KP, Wang W. Narrative review of noninvasive brain stimulation in stroke rehabilitation. Med Sci Monit. 2022;28:e938298. doi: 10.12659/MSM.938298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Badoiu A, Mitran SI, Catalin B, Balseanu TA, Popa-Wagner A, Gherghina FL, et al. From molecule to patient rehabilitation: the impact of transcranial direct current stimulation and magnetic stimulation on stroke—a narrative review. Neural Plast. 2023;2023:5044065. doi: 10.1155/2023/5044065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Veldema J, Gharabaghi A. Non-invasive brain stimulation for improving gait, balance, and lower limbs motor function in stroke. J Neuroeng Rehabil. 2022;19(1):84. doi: 10.1186/s12984-022-01062-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Di Lorenzo F, Koch G. Integrated methods of neuromodulation for guiding recovery following stroke. In: Petrosini L, editor. Neurobiological and psychological aspects of brain recovery. Cham: Springer International Publishing; 2017. pp. 183–191. [Google Scholar]
- 53.Guo Z, Jin Y, Bai X, Jiang B, He L, McClure MA, et al. Distinction of high- and low-frequency repetitive transcranial magnetic stimulation on the functional reorganization of the motor network in stroke patients. Neural Plast. 2021;2021:8873221. doi: 10.1155/2021/8873221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.de Jesus DR, de Souza Favalli GP, Hoppenbrouwers SS, Barr MS, Chen R, Fitzgerald PB, et al. Determining optimal rTMS parameters through changes in cortical inhibition. Clin Neurophysiol. 2014;125(4):755–762. doi: 10.1016/j.clinph.2013.09.011. [DOI] [PubMed] [Google Scholar]
- 55.Starosta M, Cichoń N, Saluk-Bijak J, Miller E. Benefits from repetitive transcranial magnetic stimulation in post-stroke rehabilitation. J Clin Med. 2022;11(8):2149. doi: 10.3390/jcm11082149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sebastianelli L, Versace V, Martignago S, Brigo F, Trinka E, Saltuari L, et al. Low-frequency rTMS of the unaffected hemisphere in stroke patients: a systematic review. Acta Neurol Scand. 2017;136(6):585–605. doi: 10.1111/ane.12773. [DOI] [PubMed] [Google Scholar]
- 57.Kondo T, Kakuda W, Yamada N, Shimizu M, Hagino H, Abo M. Effect of low-frequency rTMS on motor neuron excitability after stroke. Acta Neurol Scand. 2013;127(1):26–30. doi: 10.1111/j.1600-0404.2012.01669.x. [DOI] [PubMed] [Google Scholar]
- 58.Hamaguchi T, Abo M, Tanaka T, Murata K, Suzuki M, Nakaya N, et al. Predicting recovery of upper extremity motor function after stroke according to the novel intervention using repetitive transcranial magnetic stimulation and occupational therapy: NEURO. Front Neurol. 2020;11:581186. doi: 10.3389/fneur.2020.581186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Nazarova MA, Novikov PA, Nikulin VV, Ivanova GE. Diagnostic capabilities of transcranial magnetic stimulation to predict motor recovery after a stroke. Neuromuscul Dis. 2020;10(1):64–74. doi: 10.17650/2222-8721-2020-10-1-64-74. [DOI] [Google Scholar]
- 60.Zong X, Li Y, Liu C, Qi W, Han D, Tucker L, et al. Theta-burst transcranial magnetic stimulation promotes stroke recovery by vascular protection and neovascularization. Theranostics. 2020;10(26):12090–12110. doi: 10.7150/thno.51573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Koch G, Esposito R, Motta C, et al. Improving visuo-motor learning with cerebellar theta burst stimulation: behavioral and neurophysiological evidence. NeuroImage. 2020;208:116424. doi: 10.1016/j.neuroimage.2019.116424. [DOI] [PubMed] [Google Scholar]
- 62.Jackson MP, Rahman A, Lafon B, Kronberg G, Ling D, Parra LC, et al. Animal models of transcranial direct current stimulation: methods and mechanisms. Clin Neurophysiol. 2016;127(11):3425–3454. doi: 10.1016/j.clinph.2016.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Woods AJ, Antal A, Bikson M, Boggio PS, Brunoni AR, Celnik P, et al. A technical guide to tDCS, and related non-invasive brain stimulation tools. Clin Neurophysiol. 2016;127(2):1031–1048. doi: 10.1016/j.clinph.2015.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kunze T, Hunold A, Haueisen J, Jirsa V, Spiegler A. Transcranial direct current stimulation changes resting state functional connectivity: a large-scale brain network modeling study. Neuroimage. 2016;140:174–187. doi: 10.1016/j.neuroimage.2016.02.015. [DOI] [PubMed] [Google Scholar]
- 65.Jamil A, Batsikadze G, Kuo HI, Labruna L, Hasan A, Paulus W, et al. Systematic evaluation of the impact of stimulation intensity on neuroplastic after-effects induced by transcranial direct current stimulation. J Physiol. 2017;595(4):1273–1288. doi: 10.1113/JP272738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Halakoo S, Ehsani F, Hosnian M, Kheirkhahan A, Samaei A, Emadi A. The comparative effects of anodal and cathodal trans-cranial direct current stimulation on balance and posture: a systematic review of literature and meta-analysis. J Clin Neurosci. 2023;107:68–76. doi: 10.1016/j.jocn.2022.12.001. [DOI] [PubMed] [Google Scholar]
- 67.Sasaki N, Abo M, Hara T, Yamada N, Niimi M, Kakuda W. High-frequency rTMS on leg motor area in the early phase of stroke. Acta Neurol Belg. 2017;117(1):189–194. doi: 10.1007/s13760-016-0687-1. [DOI] [PubMed] [Google Scholar]
- 68.Gong Y, Long XM, Xu Y, Cai XY, Ye M. Effects of repetitive transcranial magnetic stimulation combined with transcranial direct current stimulation on motor function and cortex excitability in subacute stroke patients: a randomized controlled trial. Clin Rehabil. 2021;35(5):718–727. doi: 10.1177/0269215520972940. [DOI] [PubMed] [Google Scholar]
- 69.Cha B, Kim J, Kim JM, Choi JW, Choi J, Kim K, et al. Therapeutic effect of repetitive transcranial magnetic stimulation for post-stroke vascular cognitive impairment: a prospective pilot study. Front Neurol. 2022;13:813597. doi: 10.3389/fneur.2022.813597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Yang CL, Gad A, Creath RA, Magder L, Rogers MW, Waller SM. Effects of transcranial direct current stimulation (tDCS) on posture, movement planning, and execution during standing voluntary reach following stroke. J Neuroeng Rehabil. 2021;18(1):5. doi: 10.1186/s12984-020-00799-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Elsner B, Kugler J, Pohl M, Mehrholz J. Transcranial direct current stimulation (tDCS) for improving aphasia in adults with aphasia after stroke. Cochrane Database Syst Rev. 2019;5(5):CD009760. doi: 10.1002/14651858.CD009760.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Chou YH, Ton That V, Chen AYC, Sundman M, Huang YZ. TMS-induced seizure cases stratified by population, stimulation protocol, and stimulation site: a systematic literature search. Clin Neurophysiol. 2020;131(5):1019–1020. doi: 10.1016/j.clinph.2020.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Rossi S, Antal A, Bestmann S, Bikson M, Brewer C, Brockmöller J, et al. Safety and recommendations for TMS use in healthy subjects and patient populations, with updates on training, ethical and regulatory issues: expert guidelines. Clin Neurophysiol. 2021;132(1):269–306. doi: 10.1016/j.clinph.2020.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Tendler A, Roth Y, Zangen A. Rate of inadvertently induced seizures with deep repetitive transcranial magnetic stimulation. Brain Stimul. 2018;11(6):1410–1414. doi: 10.1016/j.brs.2018.09.001. [DOI] [PubMed] [Google Scholar]
- 75.Nikolin S, Huggins C, Martin D, Alonzo A, Loo CK. Safety of repeated sessions of transcranial direct current stimulation: a systematic review. Brain Stimul. 2018;11(2):278–288. doi: 10.1016/j.brs.2017.10.020. [DOI] [PubMed] [Google Scholar]
- 76.Dionísio A, Carvalho S, Castelhano J. TMS application in both health and disease. Front Hum Neurosci. 2023;16:1110274. doi: 10.3389/fnhum.2022.1110274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Lefaucheur JP, Aleman A, Baeken C, Benninger DH, Brunelin J, Di Lazzaro V, et al. Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS): An update (2014–2018) Clin Neurophysiol. 2020;131(2):474–528. doi: 10.1016/j.clinph.2019.11.002. [DOI] [PubMed] [Google Scholar]
- 78.Bernhardt J, Hayward KS, Kwakkel G, Ward NS, Wolf SL, Borschmann K, et al. Agreed definitions and a shared vision for new standards in stroke recovery research: The Stroke Recovery and Rehabilitation Roundtable taskforce. Int J Stroke. 2017;12(5):444–450. doi: 10.1177/1747493017711816. [DOI] [PubMed] [Google Scholar]