Abstract
Glomerulonephritis (GN) is the most common cause of end-stage renal failure worldwide; in most cases, it cannot be cured and can only delay the progression of the disease. At present, the main treatment methods include symptomatic therapy, immunosuppressive therapy, and renal replacement therapy. However, effective treatment of GN is hindered by issues such as steroid resistance, serious side effects, low bioavailability, and lack of precise targeting. With the widespread application of nanoparticles in medical treatment, novel methods have emerged for the treatment of kidney diseases. Targeted transportation of drugs, nucleic acids, and other substances to kidney tissues and even kidney cells through nanodrug delivery systems can reduce the systemic effects and adverse reactions of drugs and improve treatment effectiveness. The high specificity of nanoparticles enables them to bind to ion channels and block or enhance channel gating, thus improving inflammation. This review briefly introduces the characteristics of GN, describes the treatment status of GN, systematically summarizes the research achievements of nanoparticles in the treatment of primary GN, diabetic nephropathy and lupus nephritis, analyzes recent therapeutic developments, and outlines promising research directions, such as gas signaling molecule nanodrug delivery systems and ultrasmall nanoparticles. The current application of nanoparticles in GN is summarized to provide a reference for better treatment of GN in the future.
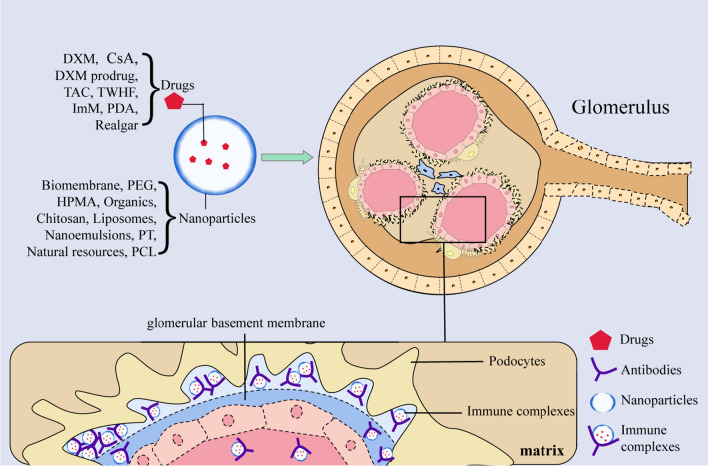
Graphical Abstract
Keywords: Nanotechnology, Nanocarrier, Primary glomerulonephritis, Diabetic nephropathy, Lupus nephritis, Therapy
Introduction
Glomerulonephritis (GN) is a group of inflammatory diseases characterized by glomerular injury and lesions. The causes of GN are relatively complex, and the pathogenesis is not yet clear. It may be caused by autoimmune dysfunction or may be related to multiple factors, such as genetics, infection, drugs, viruses, and the environment. GN can be divided into primary and secondary GN in terms of etiology [1]. Common pathological types of primary GN include acute GN, crescentic GN, membranous GN, membranoproliferative GN, mesangial proliferative GN, focal segmental glomerulosclerosis (FSG), minimal change GN, IgA nephropathy (IgAN) and chronic GN. The most common types of secondary GN include diabetic nephropathy (DN) and lupus nephritis (LN) (Table 1). The symptoms of GN vary widely. Many patients have no special symptoms in the early stage. Some patients present with nephritic syndrome (hematuria, proteinuria, edema and hypertension), and some patients show nephrotic syndrome (hyperlipidemia, severe edema, heavy proteinuria and hypoproteinemia) [2]. Even patients with the same pathological characteristics may exhibit varying degrees of proteinuria [3]. However, some typical clinical manifestations can correspond to specific GN. After 1–4 weeks of streptococcal infection, patients with symptoms such as edema, hypertension, and hematuria can be diagnosed with acute GN [4]. Recurrent episodic microscopic or macroscopic hematuria is a landmark clinical feature of IgAN, which is the most common primary GN in the world [5]. Minimal changes in GN are the most common type in children [2]. Except for LN, the other types of GN are more common in the male population than in the female population [6].
Table 1.
Disease characteristics of common glomerulonephritis
| Disease | Pathogenesis | Symptoms | Treatment | |
|---|---|---|---|---|
| Primary GN | Acute GN | Infection, immune complex deposit circulating or implanted antigens | Abnormal urine, edema, hypertension, abnormal kidney function | Symptomatic treatment |
| Crescentic GN | Immune mechanisms or renal and extrarenal diseases | Symptoms such as proteinuria and hematuria progress rapidly to oliguria and anuria | Corticosteroids, immunosuppressor, plasmapheresis | |
| Membranous GN | Secondary to infections, tumors, systemic immune diseases, or some drugs | Nephrotic syndrome | Spontaneous remission occurs in up to a third of patients, symptomatic treatment, immunosuppressor | |
| Membranoproliferative GN | Immune complex deposit or complement pathway activation | Hematuria, proteinuria, chronic renal failure or nephrotic syndrome | Symptomatic treatment, corticosteroids, immunosuppressor | |
| Mesangial proliferative GN | Immune complex deposit | Hematuria, proteinuria or nephrotic syndrome | Symptomatic treatment, immunosuppressor | |
| FSG and Minimal change GN | Undefined, may be podocyte injury or circulating permeability factor action | Nephrotic syndrome or proteinuria | Symptomatic treatment, immunosuppressor | |
| IgAN | Immune complex deposit or abnormal immunomodulation | Hematuria or proteinuria | Symptomatic treatment, immunosuppressor are offered to patients with a rapidly progressive course of GN | |
| Chronic GN | Caused by the development of acute GN or immune-mediated inflammation | Proteinuria, hematuria, hypertension, edema or chronic renal failure | Symptomatic treatment, corticosteroids, immunosuppressor | |
| Secondary GN | DN | Oxidative Stress, hyperglycemia, inflammation and renin–angiotensin–aldosterone system | Decline in renal function, proteinuria and GFR declined | Symptomatic treatment, control blood glucose |
| LN | Immune complex deposit, chemokine-mediated recruitment of different leukocyte subsets and extrarenal pathogenic mechanisms | Usually asymptomatic or proteinuria | Symptomatic treatment, corticosteroids, immunosuppressor |
GN Glomerulonephritis, FSG Focal segmental glomerulosclerosis, IgAN IgA nephropathy, DN Diabetic nephropathy, LN Lupus nephritis
Renal biopsy is the preferred method of diagnosis for GN and can guide treatment and prognosis [7]. Many patients have no special signs in the early stage, which increases the difficulty of disease diagnosis and treatment. At the same time, increasing evidence shows that immunosuppressive drugs have limited therapeutic effects on IgAN [2]. The 1-year renal survival rate of patients who are double-positive for antineutrophil cytoplasmic antibody (ANCA) and anti-glomerular basement membrane antibody (anti-GBM) with crescentic GN, even after treatment with immunosuppressive drugs and plasma exchange, remains lower than that of patients with either anti-GBM or ANCA alone [8]. GN has a high incidence rate, which usually cannot be completely cured, and may progress to chronic GN and end-stage renal disease (ESRD) [9]. Research has shown that the incidence of ESRD may increase in the future [10]. When GN progresses to ESRD, hemodialysis or kidney transplantation is almost the last option. Dialysis therapy may cause a serious economic burden to patients [11]. In the long run, kidney transplantation may have higher economic value than dialysis, but approximately 40% of kidney transplants fail within ten years, without improving kidney and patient overall survival rates [12].
At present, the primary clinical therapy for GN is symptomatic therapy and immunosuppressive therapy (e.g. glucocorticoid, ciclosporin, etc.). Most patients with GN can only delay the progression of the disease and cannot be completely cured. Some types of patients with GN are prone to relapse and steroid resistance after treatment, or progress to ESRD, which are very difficult to treat. Patients with ESRD are usually treated with replacement therapy, which increases the economic burden of patients and reduces the quality of life. The side effects of some immunosuppressants exceed the therapeutic effect. Therefore, new GN treatment methods are urgently needed [1, 7].
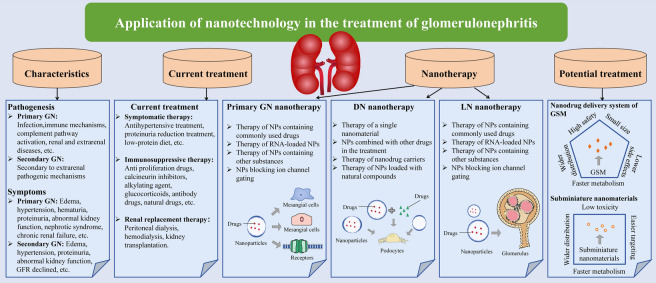
Nanotechnology has flourished in multiple fields, including medicine, such as targeted transportation, in vivo imaging, assisted diagnosis, and disease treatment. Encapsulating drugs with nanoparticles (NPs) can target drug delivery, improve bioavailability, avoid systemic effects, and reduce drug toxicity and adverse reactions [13]. Specific dimensions can be more effectively deposited on the target area and play a targeted role. There have been many attempts to deliver drugs to the kidney by nanodrug loading systems, which mainly deliver drugs to mesangial cells, podocytes and endothelial cells [14]. Although some reviews have summarized the application of nanodrugs in kidney diseases or DN [15, 16], there is no review related to nanodrugs in the treatment of GN. Therefore, this review briefly introduces the concept, pathologic changes and current treatment situation of GN, summarizes the application of nanotechnology in the treatment of primary GN and secondary GN (DN and LN) (Fig. 1), and proposes some treatment methods that have the potential to be applied in the treatment of GN. The focus of this review is on the application and prospects of nanotechnology in the treatment of GN.
Fig. 1.
Schematic diagram of GN treatment. (Symptomatic therapy, renal replacement therapy, immunosuppressive therapy, primary GN nanotherapy, DN nanotherapy, LN nanotherapy). GN glomerulonephritis; DN diabetic nephropathy; LN lupus nephritis; RRT renal replacement therapy; UAE urinary albumin excretionopathy
Changes in the glomerular filtration barrier (GFB) in GN
GFB consists of glomerular endothelial cells, glomerular basement membrane (GBM) and podocytes. Podocytes originate from the renal mesenchyme and reach the outside of the GBM. Structural changes in podocytes may cause kidney diseases, including GN. For example, patients with DN lose and damage podocytes, resulting in GFB damage and kidney lesions.
Physiological and pathophysiological structure of GFB
GFB is a unique structure with selective permeability, serving as both a molecular barrier and a charge barrier, which can prevent the filtration of various plasma albumin and negatively charged macromolecules in the blood. Proteinuria, which occurs when the GFB is damaged, is characteristic of most kidney diseases and is a factor in the rapid progression to renal failure [17].
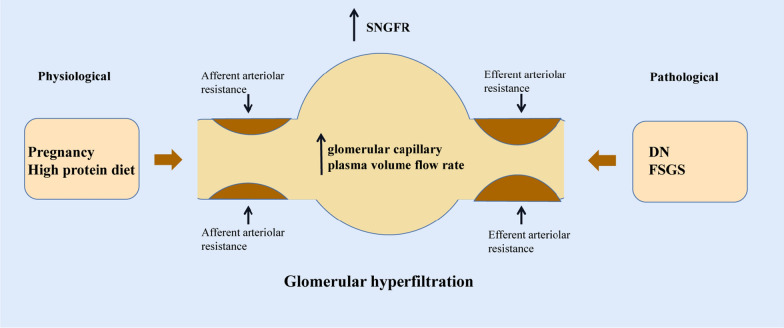
Glomerular hyperfiltration
Glomerular hyperfiltration is an abnormal increase in the glomerular filtration rate (GFR), filtration fraction or nephron filtration. A high-protein diet and pregnancy can cause physiological glomerular hyperfiltration [18, 19]. In DN, changes in renal function successively lead to glomerular hyperfiltration, increased urinary protein and decreased progressive GFR. Glomerular hyperfiltration may be an early manifestation of DN. Hyperglycemia may be a factor in the pathogenesis of glomerular hyperfiltration in patients with diabetes, and changes in renal function related to blood glucose have important significance for the progression of DN. At the same time, glomerular hyperfiltration was also observed in the secondary FSG (Fig. 2).
Fig. 2.
Factors in glomerular hyperfiltration. The factors of glomerular hyperfiltration include physiological glomerular hyperfiltration and pathological glomerular hyperfiltration, among which physiological factors include a high-protein diet and pregnancy, and pathological factors include DN and FSGS. SNGFR single-nephron glomerular filtration rate; DN diabetic nephropathy; FSGS focal segmental glomerulosclerosis
In summary, GFB plays a crucial role in maintaining renal homeostasis. An abnormal increase in GFR and destruction of the GFB are important factors affecting the development and poor prognosis of GN. Repairing and protecting the structure of the GFB and reducing glomerular hyperfiltration are therapeutic strategies for GN.
Current treatment status of GN
GN is the main cause of chronic kidney disease. Therefore, GN patients need symptomatic treatment to delay disease progression. The disease classification of GN lacks complete consensus in histopathology. Different classifications of GN based on pathological or etiological definitions have the same clinical manifestations, and the treatment method is determined based on clinical manifestations [1].
Symptomatic therapy
Symptomatic treatment is applicable to all progressive GN, including antihypertensive treatment, proteinuria reduction treatment and a low-protein diet. ACE inhibitors (ACEis) or angiotensin II receptor blockers (ARBs) are first-line drugs for treating hypertension and proteinuria [7, 20]. Symptomatic treatment can alleviate the symptoms of proteinuria and slow the progression of nonspecific GN. In adult GN patients with nephrotic syndrome, antiedema and anticoagulant therapy also need to be considered. Based on the evaluation of patient status, serum albumin levels below 20–25 g/L may indicate anticoagulation prevention [21]. For edema symptoms, loop diuretics are the first choice, and a low salt diet is needed [7]. For IgAN, because of the typical clinical manifestations, such as massive proteinuria, hypertension and reduced GFR, oral glucocorticoids combined with ACEis or ARBs are the first-line treatment [22]. Immunosuppressive drugs should not be used except for those with rapidly progressing IgAN [2].
Immunosuppressive therapy
In addition to symptomatic treatment applicable to all patients, immunosuppressive treatment methods, including glucocorticoids, calcineurin inhibitors (ciclosporin and tacrolimus), anti-proliferation and anti-metabolism drugs (mycophenolate ester), antibody drugs (baliximab and rituximab), alkylating agents (cyclophosphamide) and natural drugs (tripterygium wilfordii and its derivatives), are applicable to specific GN cases.
Glucocorticoids are first-line drugs for GN that can be applied in almost all types of GN and can effectively reduce urinary protein levels. Some studies have shown that glucocorticoids are beneficial for GN patients with proteinuria exceeding 3 g/day and a GFR below 50 mL/min [23]. Glucocorticoids are often used for minimal change GN and FSG, and FSG requires long-term high-dose treatment [1, 7]. The treatment of proliferative LN (class III and IV) and anti-GBM crescentic GN mostly starts with steroid impulse therapy, followed by oral cyclophosphamide and prednisone [1].
In membranous GN, alkylating agents (such as cyclophosphamide) plus corticosteroids or calcineurin inhibitors are commonly used for treatment. Immunosuppressive drugs are only considered when GFR decreases or severe complications of nephrotic syndrome occur [2]. Rituximab is a relatively new treatment method that has achieved partial or complete remission in membranous GN [24] and may have a direct impact on podocytes, thus having a certain therapeutic effect in minimal change GN [25]. Triptolide can inhibit the proliferation of mesangial cells in IgAN, and its application in membranous GN, LN and DN can reduce proteinuria and play a role in renal protection [26–30].
Renal replacement therapy
Although there are currently treatment and remission programs for GN and researchers are constantly making new attempts, there are still many GN patients whose conditions cannot be controlled and gradually progress to chronic GN and even end-stage renal failure. For these patients, renal replacement therapy is a method of life support and life assurance treatment. Renal replacement therapy includes hemodialysis, peritoneal dialysis and kidney transplantation [31]. Patients use dialysis to remove excess body fluids to restore normal circulation and blood pressure. Different dialysis membranes are used according to the actual situation to remove impurities and fluids. Most patients undergo dialysis three times a week. Patients can maintain a normal life through dialysis treatment, but not all patients can achieve good results. Infection, malnutrition and other factors can all cause complications of hemodialysis, with infection being one of the main causes of death in hemodialysis patients [32].
Renal transplantation is the last resort for end-stage renal failure. It can improve the survival rate and quality of life of patients with renal failure. After transplantation, immunosuppressive drugs are required to maintain the status of the transplanted kidney [33]. Although kidney transplantation can be successful, transplant patients are prone to recurrent GN, thus leading to transplant failure [34].
Application of nanomaterials in GN treatment
Nanomaterials are widely used in kidney diseases and can be used for disease diagnosis, detection of renal structure and function, delivery of drugs, and prevention of diseases (such as transplant-induced reactions) [13]. Researchers are committed to developing NPs with both diagnostic and therapeutic functions [35]. NPs are effective carriers that can carry various types of substances and can be targeted for delivery to kidney tissue and cells, achieving better results than conventional therapy and reducing the systemic effects of drugs and adverse reactions [36].
The role of nanomaterials in the treatment of primary GN
In the treatment of primary GN, NPs typically function as a delivery system capable of transporting various drugs and nucleic acids. Additionally, they can modulate ion channel gating to regulate inflammation (Table 2) (Fig. 3).
Table 2.
Application and efficacy of nanomaterials in primary glomerulonephritis
| Disease | Material | Particles | Target | Therapeutic Effects | Ref |
|---|---|---|---|---|---|
| GN | Human serum protein peptide fragments | Peptide fragment of Triptolide (immunosuppressive)—human serum albumin:PF-A1-123,PF-A124-298 and PF-A299-585 | Kidney | Reduce cytotoxicity, relieve symptoms of membranous nephropathic | [41] |
| MSPGN | Albumin | CLT-loaded albumin nanoparticles | MC | Alleviate proteinuria, inflammation, glomerular hypercellularity, and excessive extracellular matrix deposition | [40] |
| MN | Liposomes | 3,5-dipentadecyloxybenzamidine hydrochloride (TRX-20)-modified liposomes | MC | Relieve symptoms of membranous nephropathic, improved proteinuria, serum cholesterol and albumin | [42] |
| GN | Lipid | Lipid based nanocarrier system (Saint-O-Somes) | Podocytes | Targeted drug delivery to podocytes | [43] |
| GN | Micelles | Novel Biomimetic pH Sensitive Nanomicelles Loaded with DXM(MM/HA-DXM) | MC | Improve targeting performance, reduce proteinuria, have anti-inflammatory effect | [46] |
| MsPGN | Polyethylene glycol-polycarbonate | Polyethylene glycol-polycarbonate nanoparticles loaded with DXM | MC | Drugs can accumulate more in the kidney and renal cortex | [47] |
| MGN | Liposomes |
Methylprednisolone bovine serum albumin nanoparticles (ME BSA NPs) |
Mesangium | Reduce the levels of 24 h urinary protein and serum creatinine | [48] |
| GN | Polycarbonate | DXMS and CAP loaded PLGA-IL delivery system (called DXMS/CAP@PLGA-ILs) | MC | Improve the pathological changes in the mesangial area and positive expression of proliferating cell nuclear antigen in glomeruli | [52] |
| GN | L-serine (Ser)-modified polyamidoamine dendrimer (PAMAM) | L-serine modified polyamide dendronized polymer loaded with CAP | Renal cortex | Targeted drug delivery | [53] |
| GN | Polycationic cyclodextrin | Polycationic cyclodextrin nanoparticles containing siRNA (siRNA/CDP-NPs) | Mesangium | Targeted drug delivery to mesangial cells | [58] |
| MsPGN | Chitosan | Chitosan nanoparticles containing xPDGF-Band PDGFR-β | MC | Reduce mesangial cell proliferation and matrix accumulation | [59, 60] |
| IgAN | Liposomes |
Size and Surface charge dependent glomerular targeting liposome nanoparticles containing p38e and Surface csiRNA |
MC and endothelial cell | Alleviate proteinuria, inflammation and excessive extracellular matrix deposition | [61] |
| GN | Cationic liposome | Cationic liposomes loaded with PAI-1R | Glomeruli | Reduce increases in glomerular matrix accumulation and expression of PAI-1R and fibronectin | [62] |
| GN | Au/Liposomes | Gold nanoliposome loaded with DXMS and TGF wiAu-ILs) | Mesangium | Inhibit local inflammation and fibrosis, produce better therapeutic effects | [63] |
| GN | Natural compounds | Fucoidan nanoparticles | Renal cells | Improve the necrosis of renal cells, decreases BUN, creatinine, MDA, IL-6, and TNF-Num > < Disp | [64] |
| NS | Natural compounds | PH sensitive EGCG nanoparticles | Renal cells | Reduce proteinuria and improve renal pathological changes | [65] |
| GN | Antibody | Nanoantibodies of P2X7 | Ion channel | Improve drugs release efficiency and relieve experimental glomerulonephritis | [70] |
GN glomerulonephritis, MSPGN mesangial proliferduringive glomerulonephritis, MC Mesangial cells, MsPGN mesangial proliferative glomerulonephritis, MGN mesangial glomerulonephritis, IgAN IgA nephropathy, NS nephrotic syndrome, MN membranous nephropathy, DXM Dexamethasone, NPs nanoparticles, CAP captopril, PAI plasminogen activator inhibitor
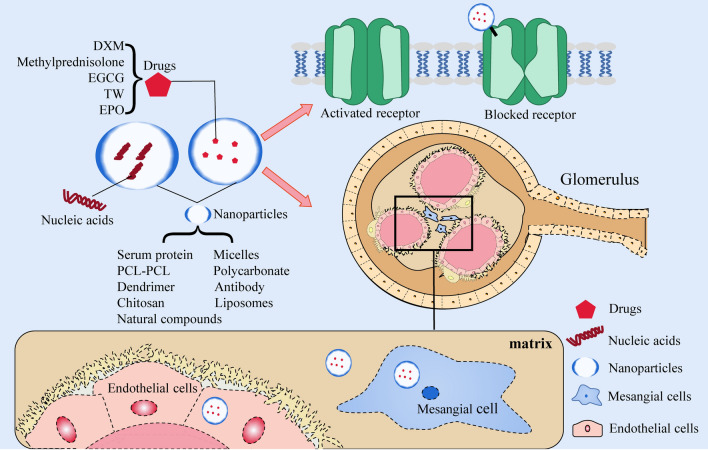
Fig. 3.
Treatment of primary GN with nanomaterials. In the treatment of primary GN, a nanodrug delivery system can deliver various drugs and nucleic acids to mesangial cells, endothelial cells and the mesangium. Nanoparticles can also play a therapeutic role by blocking ion channels. GN glomerulonephritis, PCL-PCL polyethylene, glycol-polycaprolactone, DXM dexamethasone, EGCG epigallocatechi gallate, TW tripterygium wilfordii; EPO erythropoietin
The therapeutic effect of NPs containing commonly used drugs on primary GN
In China, tripterygium wilfordii (TW) and its formulations have been used for the treatment of GN [37]. Human serum albumin (HSA), as the most abundant protein in plasma, has many excellent properties and is widely used in biomedical fields [38]. By cleaving HSA into small molecules that can be filtered by glomeruli, peptide fragments (PFs) of HSA can accumulate in the kidney and be reabsorbed by proximal tubular cells after glomerular filtration. Such PFs can be suitable kidney-targeting carriers [39]. Celastrol (CLT) is an immunosuppressive and anti-inflammatory agent derived from TW. CLT-loaded albumin NPs with a size of 95 nm can target drug delivery to mesangial cells and then achieve maximum accumulation, thus reducing CLT-related systemic toxicity and alleviating the symptoms of proteinuria. It also decreases the mRNA levels of MCP-1 and ICAM-1 to block monocyte infiltration and reduces the levels of IL-6 and IL-1β, thus achieving anti-inflammatory effects [40]. Triptolide (TP), which is also a bioactive diterpenoid epoxide derived from TW, has immunosuppressive and anti-inflammatory functions. TP coupled with PF-A299-585 (PF with amino acid sequence A299-585) can be targeted to the kidney for therapeutic effects, and its cytotoxicity is significantly lower than that of the free drug [41]. Similarly, 3,5-dipentadecyloxybenzamidine hydrochloride-modified liposomes specifically target mesangial cells in the glomerulus. When TP was sealed into liposomes to form drug-loaded nanoliposomes for therapeutic use, it was shown that the nanoliposomes could reverse inflammation in rodent models of nephropathy [42]. Long-term use of sirolimus as a commonly used immunosuppressive drug may lead to severe proteinuria. The lipid-based nanocarrier system can deliver sirolimus to podocytes in a targeted manner, reduce the dosage of application, and thus reduce the risk of serious side effects [43].
Glucocorticoids, which have anti-inflammatory effects, are currently the most commonly used drugs for the treatment of GN. However, the application of glucocorticoids can cause adverse effects on many organ systems, such as osteoporosis, femoral necrosis, hyperglycemia, hypertension, increased risk of infection, Cushing’s syndrome, and gastrointestinal bleeding [44]. Compared with traditional drug delivery modes, nanomaterials as transport carriers for targeted therapy have great superiority. For example, polymeric nanomicelles have the advantages of high structural stability, massive loading of hydrophobic drugs, more effective targeted transport ability and stimulation sensitivity [45]. In one study, pH-sensitive nanomicelles loaded with drugs were evaluated for the treatment of GN. By attaching dexamethasone (DXM) to hyaluronic acid (HA) and then covering the surface with natural macrophage membranes (MM), biomimetic nanomicelles (MM/HA-DXM) were formed. For the active homing action of macrophages to the inflammation site, MM/HA-DXM can be targeted for transport to the inflammation site of glomeruli. In the weakly acidic GN microenvironment, HA and DXM can be targeted for release. DXM has an inhibitory effect on mesangial cell proliferation, HA can remodel the phenotype of macrophages from the proinflammatory M1 phenotype to the anti-inflammatory M2 phenotype, and both have synergistic anti-inflammatory effects. As demonstrated by in vivo experiments, MM/HA-DXM treatment significantly increased albumin and TP levels in the treated group, while blood urea nitrogen (BUN), creatinine, cholesterol and triglyceride levels were significantly reduced, and proteinuria levels decreased up to 2.33-fold compared to the control group [46].
In another study, Li et al. [47] designed glomerulus mesangium-targeted poly(ethylene glycol)-poly(D,L-lactic-co-glycolic acid) NPs loaded with DXM acetate for the treatment of mesangial proliferative GN. The drug delivery system was able to effectively target the drug into the glomerular mesangium membrane, and NPs with a size of 90 nm had maximum accumulation in the renal mesangium membrane. Similarly, the efficacy of methylprednisolone bovine serum albumin NPs in the treatment of mesangial proliferative GN was evaluated. After nanoparticle treatment, rats with cationic bovine serum albumin-induced mesangial proliferative GN showed a significant reduction in 24-h urinary protein volume and serum creatinine, demonstrating the therapeutic effect of this drug system on mesangial proliferative GN [48].
Chronic GN is the end stage of the development of different types of GN, and renal fibrosis inevitably occurs [49]. The use of glucocorticoids does not have an antifibrotic effect, which is detrimental to the prognosis of GN. Therefore, it makes sense to simultaneously perform anti-inflammatory and antifibrotic modulation. ACEis/ARBs are also commonly used in renal diseases due to their anti-inflammatory [50] and antifibrotic functions [51]. Zhou et al. [52] constructed a DXM and captopril (CAP)-loaded PLGA-IL delivery system. The results of in vivo anti-inflammatory/anti-fibrotic treatment showed that all types of cell infiltration and proliferation were effectively controlled in PAS-stained sections in the experimental group, and the expression levels of TNF-α, IL-6, IL-β1, TGF-β, α-SMA and Fn were reduced. In addition, an l-Ser-modified polyamidoamine dendrimer as a targeted drug carrier could successfully transport CAP to the kidney [53]. Hydrophobically modified glycol chitosan (HGC) nanomicelle-delivered olmesartan to the kidney attenuated renal fibrosis in Col4a3-/- mice and did not alter their blood pressure [54].
Therapeutic effect of RNA-loaded NPs on primary GN
RNA interference is a process that can efficiently and specifically silence the expression of almost any gene, including endogenous microRNAs (miRNAs) and synthetic short interfering RNAs (siRNAs). By mediating targeted mRNA degradation or mRNA translation inhibition, the expression of target genes can be knocked down in a sequence-specific manner. siRNA is more advantageous than miRNA in triggering specific gene silencing [55, 56]. siRNA molecules must be delivered to the interior of the target cell to activate the RNA interference pathway, so vectors or chemical modifications are required to enter the interior of the target cell [57].
Zuckerman et al. [58] verified that intravenous injection of polycationic cyclodextrin NPs containing siRNA rapidly accumulated in the glomerular basement membrane. Several studies have shown that chitosan acts as a siRNA delivery system to target siRNA molecules of PDGF-B and PDGFR-β to the kidney. The results showed that silencing the PDGF-B signaling pathway could inhibit the proliferation of mesangial cells and has promise for the treatment of mesangial proliferative GN [59, 60]. In addition, liposome NPs loaded with p38α MAPK and p65 siRNA successfully alleviated proteinuria, inflammation and excessive extracellular matrix deposition in a mouse IgAN model [61]. Targeted application of this nanoliposome in a rat model of nephritis inhibited glomerular TGF-β gene expression, thereby specifically suppressing glomerulosclerosis [62]. In another study, similarly, NPs loaded with DXM and TGFβ1 siRNA effectively reduced the expression levels of cytokines such as TNF-α and TGF-β1, mediating the dual regulation of inflammation and fibrosis [63].
Therapeutic effect of NPs containing other substances on primary GN
Natural compounds often have poor oral availability issues. Nanomodels that can selectively release or target drugs are an effective way to improve drug utilization. Many studies are exploring the treatment of GN, such as administering fucoidan NPs at a dose of 300 mg/kg body weight, which can significantly reduce urea, creatinine, MDA, IL-6, and TNF-α levels, thereby improving the necrosis of renal cells [64]. In a rat model of nephrotic syndrome, epigallocatechin-3-gallate (EGCG) NPs reduced the amount of urine protein. The bioavailability of EGCG NPs was more than 2.4 times higher than that of the EGCG powder group [65].
Erythropoietin (EPO), which is produced by capillary-lined cells around the renal tubules, can modulate the production of red blood cells and is considered a drug that can treat kidney diseases [66]. Zhang et al. [67] established a rat model of IgAN and verified the therapeutic effect of EPO-loaded chitosan-triphosphate NPs for IgAN, with a significant reduction in urea and creatinine levels.
NPs blocking ion channel gating
Ion channels are ideal therapeutic targets, but highly specific targeted drugs are needed. P2X7 is a trimer ion channel gated by extracellular ATP that plays a role in the release of inflammatory molecules, cell proliferation and death, metabolic events and phagocytosis [68]. The activated P2X7 receptor can open pores and allow the passage of organic ions [69]. In one study, the anti-inflammatory effect of nanoantibodies targeting mouse P2X7 was evaluated. The results showed that injection of these nanoantibodies into mice could block (nanoantibody 13A7) or enhance (nanoantibody 14D5) gating of channels, prevent inflammation caused by ATP binding to P2X7 released by injured and dying cells, and improve the clinical parameters of GN. At the same time, the nanoantibody Dano1 was 7 times more effective in inhibiting the release of cellular inflammatory messengers in the human body than small molecule P1X2 antagonists [70].
Application of nanomaterials in the treatment of DN
In DN, hyperglycemia and oxidative stress are two related processes and the focus of research, and many studies are based on these two points. The forms in which NPs play a role in DN include the therapeutic effects of nanomaterials themselves, the combination of nanomaterials with drugs, and nanodrug delivery systems (Table 3) (Fig. 4).
Table 3.
Application and efficacy of nanomaterials in diabetes nephropathy
| Material | Particles | Loaded substance | Target | Therapeutic Effects | Ref |
|---|---|---|---|---|---|
| Se | SeNPs | Kidney | Reduce rate of urination, urea, creatinine, MDA and glucose | [77] | |
| Zn | ZnONPs | Podocyte | Improve renal functionality; inhibit renal fibrosis, oxidative stress, inflammation and abnormal angiogenesis, and delay the development of podocyte injury | [81] | |
| Zn | ZnONPs | Kidney | Improve uric acid, creatinine, BUN and urinary albumin | [82] | |
| Ag | AgNPs | Anti-inflammatory action, reduce the serum levels of TNF- 、IFN-e、IL-17A、IL-6 and MCP-1 | [85] | ||
| Au | AuNPs | GBM and podocyte | Attenuate hyperglycemia, reduce 24-h urinary albumin excretion rate, glomerular basement membrane thickness, foot process width and renal oxidative stress markers | [87] | |
| Metal organic frameworks | Metal organic framework containing chromium | Reduce HOMA-IR index, blood urea nitrogen, uric acid and malondialdehyde in plasma samples | [89] | ||
| Metal | Nanochelating nanomedicine with iron chelating properties(BCc1) | GBM, podocyte, and tubules | Reduce albumin, malondialdehyde and 8-isoprostane in urine specimen, increased creatinine clearance | [91] | |
| Natural resources | Nanoparticle Curcumin (nCUR) combined with INS | Curcumin/Combined INS | Kidney | Reverse or delay the histological changes of renal injury and delay progress of DN | [93] |
| Chitosan/Se | Metformin and chitosanselenium anoarticles (Ch SeNPs) | Combined metformin | Kidney | Inhibit oxidative stress and restore glucose hemostasis | [94] |
| Se | Combination of selenium nanoparticles and Sildenafil | Combined Sildenafil | Kidney |
Improve renal function and histopathological changes, have protective effect |
[95] |
| Se | Bee venom combined with SeNPs | Combined Bee venom | Kidney | Protective role against the long-term diabetic complications of DN | [96] |
| Se | Rutin combined with SeNPs | Combined | Kidney | Reduce fasting blood glucose, serum creatinine and urea, renoprotective effect against DN | [97] |
| HMSN | HMSN loaded with Metformin | Etformin | Kidney | Increase that accumulation of drugs in the kidney, relieve DN symptoms | [98] |
| Nanobilayer | Nanobilayer loaded with eprosartan eesylate | Eprosartan Mesylate | Decrease in serum creatinine, urea, lactate dehydrogenase, total albumin and malondialdehyde | [100] | |
| Nanocapsules | reactive oxygen species (ROS) responsive nanocapsules (Ad@Gel) | Adropin | Glomerular endothelium | Control blood glucose and lipid levels, improve renal function, inhibit excessive production of ROS, protect mitochondria from damage, improve lipid deposition in renal tissues | [101] |
| PCL-PEI | Polycaprolactone Polyethylenimine (PCL-PEI) core and kidney targeting peptide (KTP) modified lipid layer | Rhein | MC and glomeruli | Reduce BUN, creatinine, fibronectin and collagen, elevate albumin | [75] |
| Engineering polymer | Engineering Polymer Nanoparticles Loaded with DXM | DXM | Podocyte | Repair damaged podocytes | [103] |
| Composite nanomaterials | MicroRNA-Gated polymer Nanocomposite | MicroRNA | Podocyte | Targeted delivery of exogenous miRNA to podocytes | [104] |
| Chitosan | ATRA chitosan/Triphosphoric acid lipid hybrid nanoparticles | ATRA | Kidney | Reduce of creatinine, urea, TNF- lipid hybrid nan and VEGF, elevate AMPK and LKB1 | [105] |
| Protein | KT Targeted sHDL/TO Nanodisk(KT-sHDL/TO) | Liver X receptor agonist | MC | Suppress mesangial cell proliferation, ameliorate dyslipidemia and inflammation | [106] |
| PCL-PEI/natural resources |
Polymer loaded with RH γ- Glutamic acid coated Polycaprolactone Polyethylenimine nanoparticles (RGPP) |
RH | MC and glomeruli | Drugs accumulation in kidney | [109] |
| PCL-PEI | Renal targeted RH lipid nanoparticles with egg yolk shell structure(KLPPR) | RH | Kidney | Reduce the parameters of urea nitrogen, serum creatinine and kidney index, improve the urinary creatinine and the creatinine clearance rate | [110] |
| PEG/natural resources | RH loaded polyethylene glycol co caprolactone co Ethanimine nanoparticles(PPP-RH-NPs) | RH | MC and glomeruli | Targeted drug delivery, decrease the levels of FBG, creatinine, BUN, urine protein and the intensity of oxidative stress | [112] |
| PEG/natural resources | Ultra small polymer nanocarriers for drug delivery | DXM | Podocyte | Repair damaged podocytes | [113] |
| Organics | Deoxycholic acid conjugated nanoparticles (DNPs) | RH | Apical sodium dependent Bile acid transporters in the small intestine | Enhance oral bioavailability | [114] |
| Natural resources/Liposomes | Calycosin loaded nanoliposomes | Calycosin | Kidney cell mitochondria | Restore function of mitochondria, improve diabetic nephropathy | [115] |
| Natural resources/Liposomes | Silymarin loaded nanoliposomes | Silymarin | Podocyte | Reduce body weight/kidney ratio, renal functions and lipid profiles in renal tissues | [117] |
| Natural resources/Liposomes | Sinomenine loaded nanoliposomes | Sinomenine | Kidney | Improve renal function, and have renal protective effect | [116] |
| Natural resources/Liposomes | Nanostructured lipid carriers loaded with ergosterol | Ergosterol | Kidney | Improve oral bioavailability and therapeutic efficacy | [118] |
NPs nanoparticles, RH rhein, GBM glomerular basement membran, INS long-acting insulin, HMSN Hollowmsoporous nano composite, DXM Dexamethasone, PEG polyethylene glycol, PCL-PEI Polycaprolactone-polyethyleneimine, ATRA all-trans retinoic acid
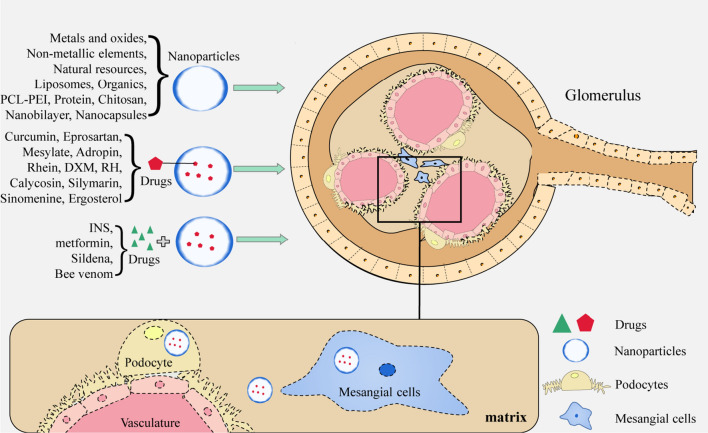
Fig. 4.
Treatment of DN with nanomaterials. In the treatment of DN, the therapeutic forms of nanoparticles include the therapeutic effect of nanoparticles themselves, the combination of nanoparticles and drugs, and the nanodrug delivery system, which can target podocytes, mesangial cells and mesangium. DN diabetic nephropathy, PCL-PEI polycaprolactone-polyethyleneimine, RH rhein; DXM dexamethasone
Therapeutic effect of a single nanomaterial on DN
Many studies are based on hyperglycemia and oxidative stress in DN. Selenium (Se) is an essential trace element for maintaining the basic physiological health of the human body, with functions such as redox regulation, immune response regulation, and control of inflammatory reactions [71, 72]. At the same time, the dosage and form of Se, as well as the lack and excessive intake, can have an impact on human health [73, 74]. Compared with Se and its compounds, SeNPs have smaller volumes, higher bioavailability, lower toxicity, and targeting properties [75, 76]. The results of a previous study showed that SeNPs effectively reduced the levels of BUN, creatinine, albumin, fibronectin, and collagen in DN. DN can be prevented by inhibiting oxidative stress and activating the cell protective protein HSP70 and longevity protein SIRT1 [75]. Similarly, SeNPs can play a renoprotective role in streptozotocin (STZ)-induced diabetes rodent models during pregnancy [77].
ZnONPs are the most common metal oxide nanomaterials, with unique advantages such as improving bioavailability and crossing biological barriers [78, 79]. At present, the medical field has studied the anti-inflammatory, antioxidant stress, antibacterial, and antitumor effects of ZnONPs [80, 81]. In some studies exploring the treatment of STZ-induced DN in rat models with ZnONPs, it has been demonstrated that ZnONPs can improve renal function by reducing the profibrotic cytokine TGF-β1, increasing the expression of MMP-9 and preventing renal fibrosis [81]. ZnONPs can also prevent inflammation by weakening the activation of the NLRP3 inflammasome, inhibiting TXNIP gene expression, and upregulating Nrf2 to suppress oxidative stress [82].
Some studies have shown that precious metal NPs such as AuNPs and AgNPs have anti-inflammatory and antioxidant activities [83, 84]. Among them, AgNPs have unique antibacterial, anticancer, and antiangiogenic properties and are widely used in multiple fields [84]. When inflammation was induced in pregnant mice exposed to AgNPs with STZ, their fetuses exhibited the ability to resist inflammation and kidney damage that persisted until adulthood. After DN was induced in these offspring by STZ, histopathological analysis showed that the damage to renal tubules and glomeruli was significantly reduced compared with that in the control group. This demonstrated the renoprotective effect of AgNPs [85]. AuNPs have been proven to have hypoglycemic effects in STZ-induced diabetes animal models [86]. Alomari et al. [87] confirmed that the use of AuNPs alone could improve podocyte damage and significantly reduce blood sugar and urine protein levels.
Metal organic frameworks have been widely used in various fields, including in the medical field [88]. A study demonstrated that metal organic frameworks effectively reduced the HOMA-IR index, BUN, uric acid and malondialdehyde in plasma samples of experimental DN [89]. At present, research has proven that BCc1, a nanochelate with iron chelating properties, has antitumor effects [90]. Fakharzadeh et al. [91] studied the therapeutic effect in an animal model of DN. The results indicated that the material effectively reduced urinary protein and the albumin/creatinine ratio and alleviated pathological changes in the glomerulus. The above findings suggest that NPs with anti-inflammatory and antioxidant stress properties can improve the renal injury caused by DN and have the potential for renal protection.
NPs combined with other drugs in the treatment of DN
Reducing blood sugar and blood pressure is an important link in the treatment of DN. However, there is also evidence that anti-inflammatory treatment has an effect on DN. Curcumin, as a natural compound, has been proven to have anti-inflammatory effects. Curcumin and its preparations can delay the progression of DN [92]. However, curcumin has low oral availability and is widely metabolized throughout the body, which makes it difficult to completely treat DN. Ganugula et al. [93] used nanocurcumin in combination with long-acting insulin to treat STZ-induced DN in a rodent model. The results showed that although there was no significant synergistic effect between nanocurcumin and insulin, the efficacy of the combination of nanocurcumin and insulin was superior to that of curcumin and insulin alone. SeNPs have been confirmed by many reports to have hypoglycemic effects on rodent models of diabetes. Scholars are not satisfied with only exploring the efficacy of using SeNPs alone, and more studies are focused on the combined application of SeNPs. Metformin is also a first-line drug for the treatment of diabetes. One study proved that the combination of metformin and chitosan SeNPs for 8 weeks was more effective than metformin or chitosan SeNPs alone, which decreased the expression levels of the proinflammatory cytokines TNF-α, IL-6 and IL-1β and restored antioxidant capacity [94]. Sildenafil is widely used to treat angina pectoris and erectile dysfunction and is also used for the experimental treatment of cancer and oxidative stress in mice. In one study, SeNPs in combination with sildenafil were evaluated in the treatment of DN in rats, which reduced oxidative stress and inhibited the expression levels of HMGB1, TNF-α, MCP1, NF-κB and IL1β [95]. Bee venom (BV) has also been reported to have hypoglycemic effects. BV combined with SeNPs can reduce blood glucose, serum BUN, creatinine and C-reactive protein in STZ-induced DN rats and increase serum albumin concentrations [96]. In addition, the combination of rutin and SeNPs can upregulate Nrf-2/HO-1 and downregulate the JAK-2/STAT3 pathway, thereby controlling oxidative damage, inhibiting the increase in IL-6 and TNF-α, and playing a role in renal protection in STZ-induced DN [97]. In short, the combined application of nanotechnology and drug therapy is superior to single therapy, which can not only reduce the blood sugar of a rodent model of DN more effectively but also delay the progression of DN.
Therapeutic effect of nanodrug carriers on DN
Cerium, as a rare earth element, has a unique antioxidant effect and can scavenge excessive free radicals under physiological conditions, thereby inhibiting oxidative stress. Cerium oxide, also known as ceria particles, has the ability to clear reactive oxygen species (ROS). Hollow mesoporous nanocomposite particles perfectly retain the antioxidant capacity of ceria particles and have a high drug loading rate because of their surface area, pore volume, large porosity and stability in solvents. This nanocomposite with an oxidative stress targeting function loaded with metformin has hypoglycemic and antioxidant effects, which can prevent the pathogenesis of ROS-related DN [98]. Angiotensin II receptor antagonists have been proven to delay the progression of renal disease in diabetes patients, possibly because hyperglycemia increases angiotensin II production and oxidative stress, which can lead to diabetic complications, including DN [99]. In one study, the therapeutic potential of eprosartan mesylate-loaded nanobilosomes for treating DN was evaluated. This nanocarrier can reduce oxidative stress and alleviate AT1R, inducible NO synthase (NOS), and TGF-β expression to achieve the goal of renal protection in DN [100]. In addition, adropin encapsulated in ROS-responsive nanocapsules may improve renal injury in DN by controlling blood glucose and lipid levels, inhibiting excessive production of ROS and protecting mitochondria [101].
The main function of podocytes as glomerular cells is glomerular filtration. Proteinuria caused by podocyte injury is an important feature of DN. Many studies have focused on targeted drug delivery to podocytes [87, 102, 103]. Some studies have shown that NPs loaded with DXM can be used for the treatment of primary GN [46, 47, 52]. Similarly, DXM-loaded NPs have been proven to be able to repair podocytes and thus can be used in DN treatment [104]. Chitosan has a hydrophilic adhesive structure and can be loaded with hydrophobic drugs. Loaded all-trans retinoic acid (ATRA) chitosan/triphosphoric acid lipid hybrid NPs have a higher oral absorption rate and better therapeutic effect than ATRA [105]. MiRNA-30a is responsible for podocyte homeostasis. Under DN conditions, miRNA-30a inhibition by the hyperglycemia-induced Notch signaling pathway can lead to podocyte injury and apoptosis. Nanocomplexes target exogenous miRNA-30a to podocytes, which can improve albuminuria and podocyte damage [104]. In DN, dyslipidemia may lead to lipoprotein imbalance and mesangial hyperexpansion. He et al. [106] developed a dual targeted treatment for abnormal cholesterol metabolism and the mesangial inflammatory response. Encapsulating liver X receptor agonists in synthetic high-density lipoprotein nanodiscs can effectively remove excess lipids in mesangial cells, improve inflammation, and restore normal kidney function in DN treatment. In summary, nanocarriers can accurately transport drugs and improve bioavailability so that drugs can have a better therapeutic effect.
Therapeutic effect of NPs loaded with natural compounds on DN
Natural compounds extracted from plants are widely used to treat various diseases. The active ingredients extracted from plants have better safety and fewer side effects [107].
Rhein, the main active ingredient of rhubarb, is an anthraquinone derivative. Rhein has superior characteristics, such as anti-inflammatory, antibacterial, anticancer, antiviral, and antioxidant properties, but its poor water solubility limits its clinical applicability [108]. Nanocarriers loaded with rhein can solve the problems of low bioavailability, reduced distribution in the kidneys, and adverse reactions. NPs with polyethylene glycol (PEL)-polycaprolactone (PCL) as the core can not only be targeted and transported to the kidneys but also exhibit good renal distribution. Rhein-loaded poly-γ-glutamic acid-coated polymeric NPs [109] and kidney-targeted rhein-loaded NPs with sizes in the range of 30–80 nm for DN can be distributed to the kidneys and enhance renal cellular uptake through the glomerular filtration membrane [110]. Similarly, polyethylenimine (PEI) is a widely used carrier that has proton aminable groups and, to some extent, shows the kidney distribution of gene transmission [111]. Polymer NPs synthesized by PEI and PEL-PCL and loaded with rhein can be effectively absorbed by cells and have therapeutic effects [112]. In addition, stable ultrasmall colloidal nanomaterials with PEL (5–30 nm) can penetrate the GFB and release DXM [113]. In another study, the expression of apical sodium-dependent bile acid transporters mediated the absorption of nutrients, and polymers modified with deoxycholic acid exhibited lower cytotoxicity and higher permeability. The synthesized deoxycholic acid-conjugated NPs have relatively high embedding efficiency (90.7 ± 0.73) % and drug loading efficiency (6.5 ± 0.29) %. In vivo experiments demonstrate a significant improvement in oral bioavailability [114].
Nanoliposomes are colloidal structures composed of double-layer membranes that can be used to encapsulate drugs that are not easily soluble in water. Calycosin is widely used to relieve hypertension, inflammation, diabetes and cancer. The calycosin-loaded nanoliposomes have a stable structure and fully utilize their abilities to restore mitochondrial function [115]. The studies of Zhu et al. [116] and Yang et al. [117] have both demonstrated that the liposome delivery system of anti-inflammatory plant extracts (sinomenine/silymarin) can regulate the JAK2/STAT3/SOCS1 and TGF-β/Smad signaling pathways and inhibit inflammation-related proteins, thereby improving renal injury. Similarly, ergosterol-loaded nanostructured lipid carriers have a relative oral bioavailability that is 277.56% higher than that of ergosterol, which more effectively inhibits high glucose-stimulated mesangial cell proliferation and extracellular matrix accumulation [118]. In brief, natural compounds can act on different targets of inflammation and pathological damage. Compared with pure natural compounds, natural compound nanodrugs have the advantages of targeting to the kidney and good kidney distribution, which significantly improves the ability to fight DN.
The role of nanomaterials in the treatment of LN
Systemic lupus erythematosus (SLE) is an autoimmune disease that involves multiple systems. There is an enormous difference between the clinical signs of the disease and the performance of hematology examination, and there is no standardized method to define the response to treatment [119]. LN is one of the most serious complications of SLE. The treatment of LN usually uses hormonal and immunosuppressive therapy, but the effect is not satisfactory and remains an important cause of death in SLE patients [120]. Nanodrug delivery systems can target various drugs to the kidneys (Table 4) (Fig. 5).
Table 4.
Application and efficacy of nanomaterials in lupus nephritis
| Material | Particles | Loaded substance | Therapeutic Effects | Ref |
|---|---|---|---|---|
| Biomembrane | IFN-embrane Effectsicacy of nanomaterials in efficacyectid profiles in renal tissuesati | DXM | Improve current SLE treatment efficacy, minimizie systemic side effects | [124] |
| PEG | Polyethylene glycol-based macromolecular prodrug (ZSJ-0228) | DXM prodrug | Local anti-inflammatory/immunosuppressive effects and improve safety | [125] |
| HPMA | ZSJ-0228 and P-Dex9 (N-(2-hydroxypropyl) methacrylamide copolymer-based dexamethasone prodrug(P-Dex) | DXM prodrug | Reduce proteinuria | [126] |
| Organics | Biodegradable ligand-conjugated nanoparticles(P71Ns-gambogic acid) | CsA | Increase targeted drug delivery and improve bioavailability | [130] |
| Chitosan/Micelles | Ethylene Glycol Chitosan Nanomicelles Loaded with Tacrolimus | TAC | Reduce renal inflammationrenal dysfunction, proteinuria and histological injury | [132] |
| Liposomes | Nanostructured lipid carrier loaded with Tripterygium wilfordii | TWHF | Reduce the collagen content of the renal interstitial cells and remove MCP-1 deposited | [133] |
| Nanoemulsions | Nanoparticle containing ImM | ImM | Reduce potential for toxicities, enhance drugs accumulation in kidney | [135] |
| PT | Npolydopamine (PDA)-based nanocarrier modified with Fe3O4 and Pt nanoparticles (PDA@Pt-Fe3O4) | PDA | Reduce inflammation, potential for photoacoustic/magnetic resonance dual-mode imaging | [138] |
| Natural resources | Nanoparticles loaded with Realgar | Realgar | Reduce anti-dsDNA, IgG, IgM, BUN, Cr, IFN-inand proteinuria | [139] |
| DNA | INH-ODN-DNA nanoflower | INH-ODN DNA | Decrease autoantibodies, reduce cytokine secretion | [140] |
| PCL | Polyethylene glycol cationic liposome related pDNA and siRNA | pDNA and siRNA | Nucleic acids may exacerbate the symptoms in SLE patients who have preexisting anti-nuclear antibodies | [141] |
CsA cyclosporine A, TAC Tacrolimus, PDA Polydopamine, PEG polyethylene glycol, ImM Imatinib mesylate, HPMA P-Dex9 (N-(2-hydroxypropyl) methacrylamide, TWHF Tripterygium wilfordii, PCL polyethylene glycol cationic liposome
Fig. 5.
Treatment of nanomaterials in LN. In the treatment of LN, a nanodrug delivery system can target the kidney and form an immune complex. LN lupus nephritis; CsA cyclosporine A, TAC tacrolimus, PDA polydopamine, PEG polyethylene glycol, HPMA P-Dex9 (N-(2-hydroxypropyl) methacrylamide, TWHF tripterygium wilfordii, ImM imatinib mesylate, PCL polyethylene glycol cationic liposome
The therapeutic effect of nanocarriers loaded with DXM and DXM prodrugs on LN
Prodrugs are inactive compounds in vitro that undergo catalysis or activation in the body before exerting pharmacological effects. They have the abilities to improve pharmacokinetic properties, improve solubility, reduce drug toxicity, and increase the specificity of the site of action [121, 122]. Glucocorticoids are widely used in the clinical management of LN. However, the side effects of long-term use seriously affect its efficacy. Recent studies have shown that prodrug administration in the form of NPs has gradually become a research direction worth exploring [123]. In a study, cancer cell membrane-coated NPs were used to alleviate autoimmune diseases by harnessing the immunosuppressive effects of tumor cells. Researchers prepared IFN-γ-treated MHC class I-deficient cancer membrane-coated NPs containing DXM, a functionally driven, disease-related CD4+ T-cell-targeted drug delivery platform, which could reduce urine protein and serum creatinine levels in LN mice [124]. In another study, a PEL-based macromolecular prodrug (ZSJ-0228) of DXM with the characteristic of self-assembly into micelles in water was applied to female NZB/WF1 mice prone to LN. The results showed that it not only has sustained therapeutic effects but also has no significant adverse reactions [125]. In addition, ZSJ-0228 and the N-(2-hydroxypropyl) methacrylamide copolymer-based DXM prodrug (P-Dex) can both improve LN symptoms in lupus-susceptible NZB/WF1 mice and reduce glucocorticoid side effects. In the MRL/LPR mouse model of LN, P-Dex or ZSJ-0228 was more effective in controlling proteinuria and prolonging survival time than the equivalent dose of Dex. However, adrenal atrophy was observed in P-Dex-treated mice but not in ZSJ-0228-treated mice [126]. In short, prodrug administration in the form of NPs can reduce the side effects of glucocorticoids while ensuring the therapeutic effect and even has a better curative effect. It will be a promising method to treat LN.
Therapeutic effect of nanocarriers loaded with immunosuppressive drugs on LNs
Ciclosporin A (CsA) is a fungal-derived cyclic peptide and a common immunosuppressive drug. CsA can selectively and reversibly inhibit the immune response mediated by T cells by inhibiting the phosphatase activity of calcineurin [127] and weaken the Ca2+-dependent response of leukocytes to proinflammatory stimuli [128]. However, long-term use of CsA treatment may have some side effects, such as newly developed hyperlipidemia, hypertension, and elevated blood creatinine [129]. In one study, a drug delivery system targeting the lymphatic system was designed, and biodegradable ligand-conjugated NPs (P2Ns-gambogic acid (GA)) were synthesized. P2Ns-GA-encapsulated CsAn increased lymphatic drug delivery by 4 to 18 times compared with ligand-free preparations and commercial CsA capsules, respectively. Moreover, the dose of CsA administered was significantly lower than the nephrotoxicity threshold, and only a few treatment-induced renal abnormalities were observed. P2Ns-GA have good transport performance across the gastrointestinal barrier, which may improve transport into the intestinal capillary network or intestinal-related lymphatic tissue, thereby enhancing delivery to the systemic lymphatic circulation and enhancing the therapeutic effect [130]. Other calcium phosphate phosphatase inhibitors, such as tacrolimus, have also been proven to alleviate nephrotoxicity [131]. Weekly use of hydrophobically modified glycol chitosan loaded with tacrolimus nanomicelles can alleviate renal inflammation, proteinuria and tissue damage in LN-positive mice by regulating the TGF-β1/MAPK/NF-κB pathway. Compared to traditional tacrolimus administration, it has the advantages of smaller doses and longer dosing intervals to exert renal protective effects [132]. In addition, the nanostructured lipid carrier loaded with TW extract can effectively reduce the collagen content of renal interstitial cells and remove MCP-1 deposited in the kidney, thus having a therapeutic effect on immune LN mice [133]. In summary, the dosage of nanodrug therapy is smaller than that of traditional drug therapy, which can reduce renal toxicity.
The therapeutic effect of nanocarriers loaded with other substances on LN
Imatinib, a common protein kinase inhibitor, has been developed and approved for the treatment of malignant tumors. Some studies have shown that imatinib also has immunosuppressive and anti-inflammatory effects and can be applied to autoimmune diseases, Alzheimer’s disease, Parkinson's disease and other diseases caused by protein kinase activation mutations [134]. A recent study evaluated the renal deposition status of NPs containing imatinib mesylate in an LN mouse model. The pharmacokinetics of nanoformulations showed changes in pharmacokinetic parameters, indicating a decrease in toxicity potential to imatinib mesylate. Compared to naked drugs, MRL/MpJ-Faslrp mice receiving nanoformulations had a threefold increase in renal deposition of imatinib mesylate after 4 h [135]. Polydopamine (PDA) is rich in phenolic groups, which can eliminate ROS produced in inflammatory reactions [136] and has a strong absorption ability in the near-infrared region. Therefore, it can be used as a photoacoustic imaging contrast agent [137]. In a study, a PDA-based nanocarrier modified with Fe3O4 and Pt NPs was developed and loaded with necrostatin-1, which has dual imaging and therapeutic effects. Necrostatin-1, as an inhibitor of receptor interacting protein 1 kinase, inhibits receptor interacting protein 1 kinase activity and plays an anti-inflammatory role. Pt NPs can catalyze H2O2 to produce oxygen, thereby counteracting the hypoxic microenvironment of LNs. PDA and Fe3O4 act as photographic developers for photoacoustic or magnetic resonance imaging (MRI) [138]. In addition, realgar is widely used as a traditional Chinese medicine for various types of inflammation. Xu et al. [139] prepared NPs loaded with realgar to study the impact on LN. Compared with MRL/lpr control mice, mice treated with realgar NPs showed a significant decrease in the serum levels of anti-dsDNA, IgG, IgM, BUN, creatinine, proteinuria, and the inflammatory cytokine IFN-γ.
Natural DNA is compressed into many spatial folds with long skeletons, and DNA nanopolymers with repetitive target sequences are called nanoflowers. Inhibitive oligodeoxynucleotides have immunomodulatory effects and are dual antagonists of TLR7 and TLR9. Among them, IRS661 and IRS869 nanoflowers can lower the autoantibody levels of mice, reduce the secretion of cytokines, and alleviate LN [140]. In addition, intravenous injection of PEL cationic liposome-related pDNA or siRNA into SLE-susceptible mice forms immune complexes with the previous antinuclear antibody, leading to the occurrence of LN [141]. In summary, nanocarriers not only have excellent targeting properties but also play a role in imaging and auxiliary diagnosis. The application of nanodrug delivery systems can increase the therapeutic efficiency of LN, which has promising application prospects.
Potential treatment strategies for GN
Nanodrug delivery system of gas signaling molecules
Gas signaling molecules (GSM) are signaling molecules that regulate physiological and pathological mechanisms, including NO, CO, H2, and H2S [142]. For a long time, people have defined these gases as harmful substances due to their adverse effects on the human body. However, with the advancement of research, researchers have found that the excellent signal transduction function of GSM can be widely applied in the human body, which has changed people’s perception of GSM [143]. NO and its endogenous producer NOS have been proposed as targets of colorectal cancer carcinogenesis regulation, and inducible NOS inhibitors have application value in the targeted therapy of colorectal cancer [144]. H2S has a role in neuroprotection and vascular smooth muscle relaxation [145]. Carbon monoxide-releasing molecules (CORM), as a prodrug producing carbon monoxide, can release CO to play an anti-inflammatory and cytoprotective role [146]. GSM has the advantage of reducing systemic side effects through local delivery, resulting in the development of stimulus-responsive nanocarriers [143].
A certain concentration of H2 can trigger death receptors on the cell surface, inhibit survival signaling, and downregulate antiapoptotic proteins. Second, H2 can selectively eliminate the most cytotoxic ROS by regulating ROS. In addition, H2 can inhibit ILs and TNFs, thereby regulating inflammation [147, 148]. The byproducts of H2 have almost no side effects and have high safety in clinical applications. In addition, the ultrasmall size of H2 is very suitable for nanomaterial drug delivery systems. Some studies have shown that only excessive amounts of materials can produce therapeutic amounts of H2. This may have limitations on in vivo application and targeted release, but larger nanoscale materials can be used to eliminate it and increase accumulation at the targeted location [149].
The three gases H2S, NO, and CO have been proven to have antioxidant, anti-inflammatory, anti-apoptotic, and anti-proliferative properties [150]. H2S produced by cystathionine β-synthase was proven to have anti-inflammatory activity and energy formation [151]. Some studies have shown that H2S has potential inhibitory effects on inflammation and oxidative stress [152, 153]. The increase in H2S levels in the kidneys can protect the kidneys after ischemia‒reperfusion injury through anti-inflammatory and antioxidant stress [152, 154]. NO and H2S have many similarities, and H2S can increase endothelial NOS activation and promote phosphorylation through intracellular Ca2+ mobilization [155]. Many studies have shown that endogenous and exogenous NO have antifibrotic mechanisms [156, 157]. Adding an NO donor to cultured rat mesangial cells can inhibit the expression of fibrotic genes at the transcriptional level [158]. This has therapeutic potential for scars produced in the end stage of glomerular inflammation.
In comparison to H2S and NO, CO is more stable [159]. In one study, researchers used styrene maleic acid copolymer (SMA) to develop a CO nanodrug delivery system. It forms micelles through self-assembly, which can slow and continuously release CO in the cycle. Mice exposed to CsA for 4 weeks experienced severe kidney damage and decreased renal function. The CO nanodrug delivery system inhibits TGF through the NLRP3 inflammasome-dependent β/Smad signaling pathway and significantly improves inflammatory damage and fibrosis [160]. Similarly, MnO2 NPs loaded with Fla encapsulated in neutrophil membranes can target inflammation sites and release CO to produce anti-inflammatory effects [161]. The above results indicate that nanodelivery systems have good potential for delivering GSM in the treatment of GN. GSM has become a promising molecular target for the treatment of diseases because of its extensive biological effects. In kidney diseases, gasotransmitters can regulate inflammation and improve inflammatory damage and may have the ability to inhibit fibrosis.
Application of subminiature nanomaterials in GN
For NPs, one of the key parameters is size, and different sizes of NPs will have different distributions in the body [162, 163]. If only AuNPs smaller than 10 nm can enter the nucleus, AuNPs larger than 10 nm will remain outside the cell [164]. Ultrasmall AuNPs do not induce ROS toxicity and have higher sensitivity and the ability to be excreted quickly through the kidneys. Therefore, ultrasmall AuNPs can be used in MRI, photoacoustic imaging, positron emission tomography, and X-ray scattering imaging for imaging diagnosis [165]. In addition, the coupling of ultrasmall AuNPs with other drugs can become a treatment platform with dual capabilities, including drugs and targeting agents, which have higher therapeutic effects than free drugs [165]. Inflammation and oxidative stress have a close and complex relationship, and many drugs with the potential to treat acute and chronic inflammation have antioxidant stress effects [166]. ROS in inflammatory reactions may promote local damage, delay healing time, and lead to chronic inflammation [167, 168]. In one study, the ability of ultrasmall Cu5.4O NPs to clear ROS and alleviate inflammation was evaluated. The results showed that ultrasmall Cu5.4O NPs had the characteristics of catalase, superoxide dismutase and glutathione peroxidase simulants. A very small dose of ultrasmall Cu5.4O NPs can clear ROS, effectively improving acute injury and promoting wound healing [169]. Our previous studies confirmed that the microminiature zirconium carbide nanodots used in glioma therapy have excellent anti-inflammatory, ROS clearance and renal clearance abilities [170, 171]. Cerium oxide NPs (CeONPs) have strong antioxidant properties, but formulations containing CeONPs cannot be effectively removed from the body. Ultrasmall CeONPs retain their antioxidant properties, effectively clearing ROS, inhibiting macrophage activation, and minimizing their recruitment and infiltration into inflammatory sites, thereby alleviating acute inflammation. Ultrasmall CeONPs are effectively excreted from the body within 24 h after systemic administration, greatly alleviating toxic side effects [172]. In addition, ultrasmall solid lipid NPs with Dex (SAN-Dex) reduced TNF-α, IL-774 and IL-1 levels in lipopolysaccharide-stimulated J6A12 cells. Through oral administration, SAN-Dex still retains its anti-inflammatory activity, but ordinary solid lipid NPs with Dex lose their anti-inflammatory capacity [173]. Based on the ultrasmall size of nanomaterials, they can effectively utilize their original characteristics, such as the ability to clear ROS, antioxidant stress and anti-inflammatory properties, and have advantages such as low toxicity, easy excretion and targeted localization. It is hoped that more effective drugs or materials can be targeted to the glomeruli for more precise treatment of GN.
Conclusions and future directions
The pathogenesis of GN is complex and has not been thoroughly studied. The treatment of GN is mainly symptomatic treatment, which can rarely cure the disease from the root, mainly slowing down the disease progress and maintaining the status quo. Immunotherapy is used for specific lesions and is accompanied by severe side effects, and steroid resistance and recurrence are also common. When GN progresses to end-stage renal failure, renal replacement therapy is needed. Dialysis treatment needs to be carried out for a long time, which causes a great economic burden to some patients' families. Renal transplant patients are also prone to recurrent GN, leading to transplant failure.
Fortunately, with the widespread development and application of nanodrug delivery systems, targeting NPs to deliver drugs to the kidneys is an emerging and highly promising treatment method. The use of nanomaterial-loaded drugs can accurately target kidney cells, reduce adverse drug reactions, and produce better therapeutic effects. NPs for diagnosis and treatment are constantly being developed, including NPs that integrate diagnosis and treatment at the same time for the purpose of diagnosis, localization and treatment. At present, most of the NPs used for GN are nanoloading platforms, and there are also a few studies on the combination of NPs and first-line drugs for diseases. At the same time, some nanomaterials (mostly metal NPs) have been validated in GN for their inherent anti-inflammatory or antioxidant stress properties.
Except for DN, there are relatively few high-quality studies and experiments on other types of GN, especially primary GN. Meanwhile, any single treatment method has limitations, and combination therapy and multifunctional nanoplatforms can compensate for the shortcomings of a single therapy and achieve better results. In addition, for example, GSM and ultrasmall nanomaterials have the characteristics of wider distribution and faster metabolism due to their small size. We believe that it is possible to explore GSM nanodrug delivery systems and ultrasmall nanomaterials in GN therapy. Current status and future prospects on nanotechnology application in GN therapy were summarized in Fig. 6.
Fig. 6.

Current status and future prospects of nanotechnology in GN therapy. GN glomerulonephritis
Based on the contents discussed above, there is reason to believe that future research on GN therapy should be multifaceted and that more attention should be given to the treatment of primary GN, with more attempts to combine NPs with other treatment methods and develop multifunctional nanoplatforms.
Acknowledgements
The authors would like to thank the Shiyanjia lab (www.shiyanjia.com) for their help in language polishing.
Abbreviations
- GN
Glomerulonephritis
- FSG
Focal segmental glomerulosclerosis
- IgAN
Immunoglobulin A nephropathy
- DN
Diabetic nephropathy
- LN
Lupus nephritis
- ANCA
Antineutrophil cytoplasmic antibody
- anti-GBM
Anti-glomerular basement membrane antibody
- ESRD
End-stage renal disease
- NPs
Nanoparticles
- GFB
Glomerular filtration barrier
- GBM
Glomerular basement membrane
- GFR
Glomerular filtration rate
- ACEis
Angiotensin-converting enzyme inhibitors
- ARBs
Angiotensin II receptor blockers
- TW
Tripterygium wilfordii
- HAS
Human serum albumin
- PFs
Peptide fragments
- CLT
Celastrol
- TP
Triptolide
- PF-A299-585
PF with amino acid sequence A299-585
- DXM
Dexamethasone
- HA
Hyaluronic acid
- MM
Macrophage membranes
- BUM
Blood urea nitrogen
- CAP
Captopril
- HGC
Hydrophobically modified glycol chitosan
- miRNAs
MicroRNAs
- siRNAs
Short interfering RNAs
- EGCG
Epigallocatechin-3-gallate
- EPO
Erythropoietin
- SeNPs
Selenium nanoparticles
- STZ
Streptozotocin
- ZnONPs
Zinc oxide nanoparticles
- AuNPs
Aurum nanoparticles
- AgNPs
Argentum nanoparticles
- BV
Bee venom
- ROS
Reactive oxygen species
- NOS
Inducible nitric oxide synthase
- ATRA
All trans retinoic acid
- PCL-PCL
Polyethylene glycol-polycaprolactone
- PEL
Polyethylenimine
- SLE
Systemic lupus erythematosus
- CsA
Ciclosporin A
- PDA
Polydopamine
- Fe3O4
Ferrosoferric oxide
- H2O2
Hydrogen peroxide
- MRI
Magnetic resonance imaging
- GSM
Gas signaling molecules
- NO
Nitrogen monoxide
- CO
Carbon monoxide
- H2
Hydrogen
- H2S
Hydrogen disulfide
- SMA
Styrene maleic acid copolymer
- MnO2 NPs
Dioxide manganese nanoparticles
- CeONPs
Cerium oxide: nanoparticles
Biographies
He-Qin Zhan
obtained her PhD degree under the guidance of Prof. Xiongzeng Zhu in Shanghai Cancer Center at Fudan University. Now she is an associate professor at the Department of Pathology, School of Basic Medical Sciences, Anhui Medical University. She focuses on the clinical and basic research of kidney diseases and lymphomas. Recently, she has been interested in the application of nanotechnology in the treatment of kidney diseases.
Xiaoxun Zhang
received her bachelor’s degree in clinical medicine from Binzhou Medical University in 2019. Now she is an M.S. student at Anhui Medical University under the supervision of Assoc. Prof. He-Qin Zhan. Her research interest focuses on kidney diseases.
Xu-Lin Chen
is a medical doctor, chief physician and doctoral supervisor. He is working in the Department of Burns, The First Affiliated Hospital of Anhui Medical University. His current research interests are in the treatment of large deep burns, acute and chronic wounds.
Liang Cheng
received his PhD degree from the Institute of Functional Nano & Soft Materials (FUNSOM) at Soochow University in 2012, and then joined Soochow University as a faculty member. He was promoted to an associate professor in 2014 and a full professor in 2018. His research work has been focused on the development of multifunctional nanomaterials for biomedical application.
Xianwen Wang
obtained his PhD degree under the guidance of Prof. Liang Cheng and Prof. Zhuang Liu at the Institute of Functional Nano & Soft Materials (FUNSOM) at Soochow University in 2021. Then, he joined Anhui Medical University as a professor and doctoral supervisor. His current research interest is the development of multifunctional nanomaterials for biomedical applications.
Author contributions
HZ and XZ were responsible for the conception and design of the review. The manuscript was mainly designed by HZ and XW, and written through contributions of all authors. HZ and XZ collected resources. LC, XC and XW revised the manuscript. All authors contributed to the article and approved the final manuscript.
Funding
National Natural Science Foundation of China (52202343, 82172858), Anhui Province Natural Science Foundation (2208085QC81, 1208085MH175), Research Fund of Anhui Institute of Translational Medicine (2022zhyx-C01), Basic and Clinical Cooperative Research and Promotion Program of Anhui Medical University (2021xkjT028).
Data availability
All the data reported in this manuscript is available within the text.
Declarations
Competing interests
The authors declare no conflicts of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
He-Qin Zhan and Xiaoxun Zhang contributed equally to this work.
References
- 1.Chadban SJ, Atkins RC. Glomerulonephritis. Lancet. 2005;365:1797–1806. doi: 10.1016/S0140-6736(05)66583-X. [DOI] [PubMed] [Google Scholar]
- 2.Floege J, Amann K. Primary glomerulonephritides. Lancet. 2016;387:2036–2048. doi: 10.1016/S0140-6736(16)00272-5. [DOI] [PubMed] [Google Scholar]
- 3.Leaf DE, Appel GB, Radhakrishnan J. Glomerular disease: why is there a dearth of high quality clinical trials? Kidney Int. 2010;78:337–342. doi: 10.1038/ki.2010.156. [DOI] [PubMed] [Google Scholar]
- 4.Naicker S, Fabian J, Naidoo S, Wadee S, Paget G, Goetsch S. Infection and glomerulonephritis. Semin Immunopathol. 2007;29:397–414. doi: 10.1007/s00281-007-0088-x. [DOI] [PubMed] [Google Scholar]
- 5.Rajasekaran A, Julian BA, Rizk DV. IgA nephropathy: an interesting autoimmune kidney disease. Am J Med Sci. 2021;361:176–194. doi: 10.1016/j.amjms.2020.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Briganti EM, Dowling J, Finlay M, Hill PA, Jones CL, Kincaid-Smith PS, Sinclair R, McNeil JJ, Atkins RC. The incidence of biopsy-proven glomerulonephritis in Australia. Nephrol Dial Transplant. 2001;16:1364–1367. doi: 10.1093/ndt/16.7.1364. [DOI] [PubMed] [Google Scholar]
- 7.Rovin BH, Adler SG, Barratt J, Bridoux F, Burdge KA, Chan TM, Cook HT, Fervenza FC, Gibson KL, Glassock RJ, et al. Executive summary of the KDIGO 2021 guideline for the management of glomerular diseases. Kidney Int. 2021;100:753–779. doi: 10.1016/j.kint.2021.05.015. [DOI] [PubMed] [Google Scholar]
- 8.Srivastava A, Rao GK, Segal PE, Shah M, Geetha D. Characteristics and outcome of crescentic glomerulonephritis in patients with both antineutrophil cytoplasmic antibody and anti-glomerular basement membrane antibody. Clin Rheumatol. 2013;32:1317–1322. doi: 10.1007/s10067-013-2268-5. [DOI] [PubMed] [Google Scholar]
- 9.AlYousef A, AlSahow A, AlHelal B, Alqallaf A, Abdallah E, Abdellatif M, Nawar H, Elmahalawy R. Glomerulonephritis histopathological pattern change. BMC Nephrol. 2020;21:186. doi: 10.1186/s12882-020-01836-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.McCullough KP, Morgenstern H, Saran R, Herman WH, Robinson BM. Projecting ESRD incidence and prevalence in the United States through 2030. J Am Soc Nephrol. 2019;30:127–135. doi: 10.1681/ASN.2018050531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Held PJ, McCormick F, Ojo A, Roberts JP. A cost-benefit analysis of government compensation of kidney donors. Am J Transplant. 2016;16:877–885. doi: 10.1111/ajt.13490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lai X, Zheng X, Mathew JM, Gallon L, Leventhal JR, Zhang ZJ. Tackling chronic kidney transplant rejection: challenges and promises. Front Immunol. 2021;12:661643. doi: 10.3389/fimmu.2021.661643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Paluszkiewicz P, Martuszewski A, Zaręba N, Wala K, Banasik M, Kepinska M. The Application of Nanoparticles in Diagnosis and Treatment of Kidney Diseases. Int J Mol Sci. 2021;23:131. doi: 10.3390/ijms23010131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tang TT, Wang B, Lv LL, Dong Z, Liu BC. Extracellular vesicles for renal therapeutics: state of the art and future perspective. J Control Release. 2022;349:32–50. doi: 10.1016/j.jconrel.2022.06.049. [DOI] [PubMed] [Google Scholar]
- 15.Desai N, Koppisetti H, Pande S, Shukla H, Sirsat B, Ditani AS, Mallick PP, Kathar U, Kalia K, Tekade RK. Nanomedicine in the treatment of diabetic nephropathy. Future Med Chem. 2021;13:663–686. doi: 10.4155/fmc-2020-0335. [DOI] [PubMed] [Google Scholar]
- 16.Liu C, Wu K, Gao H, Li J, Xu X. Current strategies and potential prospects for nanoparticle-mediated treatment of diabetic nephropathy. Diabetes Metab Syndr Obes. 2022;15:2653–2673. doi: 10.2147/DMSO.S380550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Daehn IS, Duffield JS. The glomerular filtration barrier: a structural target for novel kidney therapies. Nat Rev Drug Discov. 2021;20:770–788. doi: 10.1038/s41573-021-00242-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cortinovis M, Perico N, Ruggenenti P, Remuzzi A, Remuzzi G. Glomerular hyperfiltration. Nat Rev Nephrol. 2022;18:435–451. doi: 10.1038/s41581-022-00559-y. [DOI] [PubMed] [Google Scholar]
- 19.Helal I, Fick-Brosnahan GM, Reed-Gitomer B, Schrier RW. Glomerular hyperfiltration: definitions, mechanisms and clinical implications. Nat Rev Nephrol. 2012;8:293–300. doi: 10.1038/nrneph.2012.19. [DOI] [PubMed] [Google Scholar]
- 20.Ruggenenti P, Perticucci E, Cravedi P, Gambara V, Costantini M, Sharma SK, Perna A, Remuzzi G. Role of remission clinics in the longitudinal treatment of CKD. J Am Soc Nephrol. 2008;19:1213–1224. doi: 10.1681/ASN.2007090970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kodner C. Diagnosis and management of nephrotic syndrome in adults. Am Fam Physician. 2016;93:479–485. [PubMed] [Google Scholar]
- 22.Pozzi C, Bolasco PG, Fogazzi GB, Andrulli S, Altieri P, Ponticelli C, Locatelli F. Corticosteroids in IgA nephropathy: a randomised controlled trial. Lancet. 1999;353:883–887. doi: 10.1016/S0140-6736(98)03563-6. [DOI] [PubMed] [Google Scholar]
- 23.Tesar V, Troyanov S, Bellur S, Verhave JC, Cook HT, Feehally J, Roberts IS, Cattran D, Coppo R. Corticosteroids in IgA nephropathy: a retrospective analysis from the VALIGA study. J Am Soc Nephrol. 2015;26:2248–2258. doi: 10.1681/ASN.2014070697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gauckler P, Shin JI, Alberici F, Audard V, Bruchfeld A, Busch M, Cheung CK, Crnogorac M, Delbarba E, Eller K, et al. Rituximab in membranous nephropathy. Kidney Int Rep. 2021;6:881–893. doi: 10.1016/j.ekir.2020.12.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fornoni A, Sageshima J, Wei C, Merscher-Gomez S, Aguillon-Prada R, Jauregui AN, Li J, Mattiazzi A, Ciancio G, Chen L, et al. Rituximab targets podocytes in recurrent focal segmental glomerulosclerosis. Sci Transl Med. 2011;3:85ra46. doi: 10.1126/scitranslmed.3002231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhao L, Lan Z, Peng L, Wan L, Liu D, Tan X, Tang C, Chen G, Liu H. Triptolide promotes autophagy to inhibit mesangial cell proliferation in IgA nephropathy via the CARD9/p38 MAPK pathway. Cell Prolif. 2022;55:e13278. doi: 10.1111/cpr.13278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang PN, Tang JY, Yang KZ, Zheng QY, Dong ZC, Geng YL, Liu YN, Liu WJ. Integrated network pharmacology analysis and experimental validation to investigate the molecular mechanism of triptolide in the treatment of membranous nephropathy. Drug Des Devel Ther. 2022;16:4061–4076. doi: 10.2147/DDDT.S386031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhang LY, Li H, Wu YW, Cheng L, Yan YX, Yang XQ, Zhu FH, He SJ, Tang W, Zuo JP. (5R)-5-hydroxytriptolide ameliorates lupus nephritis in MRL/lpr mice by preventing infiltration of immune cells. Am J Physiol Renal Physiol. 2017;312:F769–f777. doi: 10.1152/ajprenal.00649.2016. [DOI] [PubMed] [Google Scholar]
- 29.Qi Q, Li H, Lin ZM, Yang XQ, Zhu FH, Liu YT, Shao MJ, Zhang LY, Xu YS, Yan YX, et al. (5R)-5-hydroxytriptolide ameliorates anti-glomerular basement membrane glomerulonephritis in NZW mice by regulating Fcγ receptor signaling. Acta Pharmacol Sin. 2018;39:107–116. doi: 10.1038/aps.2017.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chen ZH, Qin WS, Zeng CH, Zheng CX, Hong YM, Lu YZ, Li LS, Liu ZH. Triptolide reduces proteinuria in experimental membranous nephropathy and protects against C5b-9-induced podocyte injury in vitro. Kidney Int. 2010;77:974–988. doi: 10.1038/ki.2010.41. [DOI] [PubMed] [Google Scholar]
- 31.Fleming GM. Renal replacement therapy review: past, present and future. Organogenesis. 2011;7:2–12. doi: 10.4161/org.7.1.13997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mallick NP, Gokal R. Haemodialysis. Lancet. 1999;353:737–742. doi: 10.1016/S0140-6736(97)09411-7. [DOI] [PubMed] [Google Scholar]
- 33.Augustine J. Kidney transplant: new opportunities and challenges. Cleve Clin J Med. 2018;85:138–144. doi: 10.3949/ccjm.85gr.18001. [DOI] [PubMed] [Google Scholar]
- 34.Lim WH, Shingde M, Wong G. Recurrent and de novo glomerulonephritis after kidney transplantation. Front Immunol. 1944;2019:10. doi: 10.3389/fimmu.2019.01944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Theek B, Rizzo LY, Ehling J, Kiessling F, Lammers T. The theranostic path to personalized nanomedicine. Clin Transl Imaging. 2014;2:66–76. doi: 10.1007/s40336-014-0051-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sabiu G, Kasinath V, Jung S, Li X, Tsokos GC, Abdi R. Targeted nanotherapy for kidney diseases: a comprehensive review. Nephrol Dial Transplant. 2023;38:1385–1396. doi: 10.1093/ndt/gfac233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shui G, Wan Y, Jiang C, Zhang H, Chen P, Wang C, Yao J. Progress in tripterygium wilfordiiand its bioactive components in the field of pharmacodynamics and pharmacology. Zhongguo Zhong Yao Za Zhi. 2010;35:515–520. doi: 10.4268/cjcmm20100425. [DOI] [PubMed] [Google Scholar]
- 38.Fanali G, di Masi A, Trezza V, Marino M, Fasano M, Ascenzi P. Human serum albumin: from bench to bedside. Mol Aspects Med. 2012;33:209–290. doi: 10.1016/j.mam.2011.12.002. [DOI] [PubMed] [Google Scholar]
- 39.Yuan ZX, He XK, Wu XJ, Gao Y, Fan M, Song LQ, Xu CQ. Peptide fragments of human serum albumin as novel renal targeting carriers. Int J Pharm. 2014;460:196–204. doi: 10.1016/j.ijpharm.2013.10.041. [DOI] [PubMed] [Google Scholar]
- 40.Guo L, Luo S, Du Z, Zhou M, Li P, Fu Y, Sun X, Huang Y, Zhang Z. Targeted delivery of celastrol to mesangial cells is effective against mesangioproliferative glomerulonephritis. Nat Commun. 2017;8:878. doi: 10.1038/s41467-017-00834-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yuan ZX, Wu XJ, Mo J, Wang YL, Xu CQ, Lim LY. Renal targeted delivery of triptolide by conjugation to the fragment peptide of human serum albumin. Eur J Pharm Biopharm. 2015;94:363–371. doi: 10.1016/j.ejpb.2015.06.012. [DOI] [PubMed] [Google Scholar]
- 42.Yuan ZX, Jia L, Lim LY, Lin JC, Shu G, Zhao L, Ye G, Liang XX, Ji H, Fu HL. Renal-targeted delivery of triptolide by entrapment in pegylated TRX-20-modified liposomes. Int J Nanomed. 2017;12:5673–5686. doi: 10.2147/IJN.S141095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Visweswaran GR, Gholizadeh S, Ruiters MH, Molema G, Kok RJ, Kamps JA. Targeting rapamycin to podocytes using a vascular cell adhesion molecule-1 (VCAM-1)-harnessed SAINT-based lipid carrier system. PLoS ONE. 2015;10:e0138870. doi: 10.1371/journal.pone.0138870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Schäcke H, Döcke WD, Asadullah K. Mechanisms involved in the side effects of glucocorticoids. Pharmacol Ther. 2002;96:23–43. doi: 10.1016/S0163-7258(02)00297-8. [DOI] [PubMed] [Google Scholar]
- 45.Li L, Zeng Y, Chen M, Liu G. Application of nanomicelles in enhancing bioavailability and biological efficacy of bioactive nutrients. Polymers. 2022;14:3278. doi: 10.3390/polym14163278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zhang H, He Q, Wang J, Wang Y, Xuan X, Sui M, Zhang Z, Hou L. Biomimetic micelles to accurately regulate the inflammatory microenvironment for glomerulonephritis treatment. Pharmacol Res. 2022;181:106263. doi: 10.1016/j.phrs.2022.106263. [DOI] [PubMed] [Google Scholar]
- 47.Li S, Zeng YC, Peng K, Liu C, Zhang ZR, Zhang L. Design and evaluation of glomerulus mesangium-targeted PEG-PLGA nanoparticles loaded with dexamethasone acetate. Acta Pharmacol Sin. 2019;40:143–150. doi: 10.1038/s41401-018-0052-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gai X, Jiang Z, Liu M, Li Q, Wang S, Li T, Pan W, Yang X. Therapeutic effect of a novel nano-drug delivery system on membranous glomerulonephritis rat model induced by cationic bovine serum. AAPS PharmSciTech. 2018;19:2195–2202. doi: 10.1208/s12249-018-1034-z. [DOI] [PubMed] [Google Scholar]
- 49.Djudjaj S, Boor P. Cellular and molecular mechanisms of kidney fibrosis. Mol Aspects Med. 2019;65:16–36. doi: 10.1016/j.mam.2018.06.002. [DOI] [PubMed] [Google Scholar]
- 50.Barra S, Vitagliano A, Cuomo V, Vitagliano G, Gaeta G. Vascular and metabolic effects of angiotensin II receptor blockers. Expert Opin Pharmacother. 2009;10:173–189. doi: 10.1517/14656560802653180. [DOI] [PubMed] [Google Scholar]
- 51.Suh SH, Choi HS, Kim CS, Kim IJ, Ma SK, Scholey JW, Kim SW, Bae EH. Olmesartan attenuates kidney fibrosis in a murine model of alport syndrome by suppressing tubular expression of TGFβ. Int J Mol Sci. 2019;20:3843. doi: 10.3390/ijms20153843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhou L, Ye Z, Zhang E, Chen L, Hou Y, Lin J, Huang F, Yuan Z. Co-delivery of dexamethasone and captopril by α8 integrin antibodies modified liposome-PLGA nanoparticle hybrids for targeted anti-inflammatory/anti-fibrosis therapy of glomerulonephritis. Int J Nanomed. 2022;17:1531–1547. doi: 10.2147/IJN.S347164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Matsuura S, Katsumi H, Suzuki H, Hirai N, Hayashi H, Koshino K, Higuchi T, Yagi Y, Kimura H, Sakane T, Yamamoto A. l-Serine-modified polyamidoamine dendrimer as a highly potent renal targeting drug carrier. Proc Natl Acad Sci U S A. 2018;115:10511–10516. doi: 10.1073/pnas.1808168115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Suh SH, Mathew AP, Choi HS, Vasukutty A, Kim CS, Kim IJ, Ma SK, Kim SW, Park IK, Bae EH. Kidney-accumulating olmesartan-loaded nanomicelles ameliorate the organ damage in a murine model of Alport syndrome. Int J Pharm. 2021;600:120497. doi: 10.1016/j.ijpharm.2021.120497. [DOI] [PubMed] [Google Scholar]
- 55.Kanasty R, Dorkin JR, Vegas A, Anderson D. Delivery materials for siRNA therapeutics. Nat Mater. 2013;12:967–977. doi: 10.1038/nmat3765. [DOI] [PubMed] [Google Scholar]
- 56.Hu B, Zhong L, Weng Y, Peng L, Huang Y, Zhao Y, Liang XJ. Therapeutic siRNA: state of the art. Signal Transduct Target Ther. 2020;5:101. doi: 10.1038/s41392-020-0207-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kulkarni JA, Witzigmann D, Chen S, Cullis PR, van der Meel R. Lipid nanoparticle technology for clinical translation of siRNA therapeutics. Acc Chem Res. 2019;52:2435–2444. doi: 10.1021/acs.accounts.9b00368. [DOI] [PubMed] [Google Scholar]
- 58.Zuckerman JE, Gale A, Wu P, Ma R, Davis ME. siRNA delivery to the glomerular mesangium using polycationic cyclodextrin nanoparticles containing siRNA. Nucleic Acid Ther. 2015;25:53–64. doi: 10.1089/nat.2014.0505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Salva E, Turan S, Akbuğa J. Inhibition of glomerular mesangial cell proliferation by siPDGF-B- and siPDGFR-β-containing chitosan nanoplexes. AAPS PharmSciTech. 2017;18:1031–1042. doi: 10.1208/s12249-016-0687-8. [DOI] [PubMed] [Google Scholar]
- 60.Alan S, Şalva E, Yılmaz İ, Turan S, Akbuğa J. The effectiveness of chitosan-mediated silencing of PDGF-B and PDGFR-β in the mesangial proliferative glomerulonephritis therapy. Exp Mol Pathol. 2019;110:104280. doi: 10.1016/j.yexmp.2019.104280. [DOI] [PubMed] [Google Scholar]
- 61.Wang Y, Wu Q, Wang J, Li L, Sun X, Zhang Z, Zhang L. Co-delivery of p38α MAPK and p65 siRNA by novel liposomal glomerulus-targeting nano carriers for effective immunoglobulin a nephropathy treatment. J Control Release. 2020;320:457–468. doi: 10.1016/j.jconrel.2020.01.024. [DOI] [PubMed] [Google Scholar]
- 62.Liu X, Zhang J, Tang A, Xu L, Huang Y. A novel peptide ligand-coated nano-siRNA-lipoplex technology for kidney targeted gene therapy. Am J Transl Res. 2022;14:7362–7377. [PMC free article] [PubMed] [Google Scholar]
- 63.Fang P, Han L, Liu C, Deng S, Zhang E, Gong P, Ren Y, Gu J, He L, Yuan ZX. Dual-regulated functionalized liposome-nanoparticle hybrids loaded with dexamethasone/TGFβ1-siRNA for targeted therapy of glomerulonephritis. ACS Appl Mater Interfaces. 2022;14:307–323. doi: 10.1021/acsami.1c20053. [DOI] [PubMed] [Google Scholar]
- 64.Wardani G, Nugraha J, Mustafa MR, Sudjarwo SA. Antioxidative stress and anti-inflammatory activity of fucoidan nanoparticles against nephropathy of streptozotocin-induced diabetes in rats. Evid Based Complement Alternat Med. 2022;2022:3405871. doi: 10.1155/2022/3405871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Zhang G, Zhang J. Enhanced oral bioavailability of EGCG using pH-sensitive polymeric nanoparticles: characterization and in vivo investigation on nephrotic syndrome rats. Drug Des Devel Ther. 2018;12:2509–2518. doi: 10.2147/DDDT.S172919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Jelkmann W. Regulation of erythropoietin production. J Physiol. 2011;589:1251–1258. doi: 10.1113/jphysiol.2010.195057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zhang X, Wu Y, Sun K, Tan J. Effect of erythropoietin loading chitosan-tripolyphosphate nanoparticles on an IgA nephropathy rat model. Exp Ther Med. 2014;7:1659–1662. doi: 10.3892/etm.2014.1643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Sluyter R. The P2X7 receptor. Adv Exp Med Biol. 2017;1051:17–53. doi: 10.1007/5584_2017_59. [DOI] [PubMed] [Google Scholar]
- 69.Alves LA, de Melo Reis RA, de Souza CA, de Freitas MS, Teixeira PC, Moreira Ferreira DN, Xavier RF. The P2X7 receptor: shifting from a low- to a high-conductance channel - an enigmatic phenomenon? Biochim Biophys Acta. 2014;1838:2578–2587. doi: 10.1016/j.bbamem.2014.05.015. [DOI] [PubMed] [Google Scholar]
- 70.Danquah W, Meyer-Schwesinger C, Rissiek B, Pinto C, Serracant-Prat A, Amadi M, Iacenda D, Knop J-H, Hammel A, Bergmann P, et al. Nanobodies that block gating of the P2X7 ion channel ameliorate inflammation. Sci Transl Med. 2016;8:366ra162–366ra162. doi: 10.1126/scitranslmed.aaf8463. [DOI] [PubMed] [Google Scholar]
- 71.Hariharan S, Dharmaraj S. Selenium and selenoproteins: it’s role in regulation of inflammation. Inflammopharmacology. 2020;28:667–695. doi: 10.1007/s10787-020-00690-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Chen G, Yang F, Fan S, Jin H, Liao K, Li X, Liu GB, Liang J, Zhang J, Xu JF, Pi J. Immunomodulatory roles of selenium nanoparticles: novel arts for potential immunotherapy strategy development. Front Immunol. 2022;13:956181. doi: 10.3389/fimmu.2022.956181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Rayman MP. Selenium intake, status, and health: a complex relationship. Hormones. 2020;19:9–14. doi: 10.1007/s42000-019-00125-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Skalickova S, Milosavljevic V, Cihalova K, Horky P, Richtera L, Adam V. Selenium nanoparticles as a nutritional supplement. Nutrition. 2017;33:83–90. doi: 10.1016/j.nut.2016.05.001. [DOI] [PubMed] [Google Scholar]
- 75.Kumar GS, Kulkarni A, Khurana A, Kaur J, Tikoo K. Selenium nanoparticles involve HSP-70 and SIRT1 in preventing the progression of type 1 diabetic nephropathy. Chem Biol Interact. 2014;223:125–133. doi: 10.1016/j.cbi.2014.09.017. [DOI] [PubMed] [Google Scholar]
- 76.Ferro C, Florindo HF, Santos HA. Selenium nanoparticles for biomedical applications: from development and characterization to therapeutics. Adv Healthc Mater. 2021;10:e2100598. doi: 10.1002/adhm.202100598. [DOI] [PubMed] [Google Scholar]
- 77.Alhazza IM, Ebaid H, Omar MS, Hassan I, Habila MA, Al-Tamimi J, Sheikh M. Supplementation with selenium nanoparticles alleviates diabetic nephropathy during pregnancy in the diabetic female rats. Environ Sci Pollut Res Int. 2022;29:5517–5525. doi: 10.1007/s11356-021-15905-z. [DOI] [PubMed] [Google Scholar]
- 78.Rizzo LY, Theek B, Storm G, Kiessling F, Lammers T. Recent progress in nanomedicine: therapeutic, diagnostic and theranostic applications. Curr Opin Biotechnol. 2013;24:1159–1166. doi: 10.1016/j.copbio.2013.02.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Smijs TG, Pavel S. Titanium dioxide and zinc oxide nanoparticles in sunscreens: focus on their safety and effectiveness. Nanotechnol Sci Appl. 2011;4:95–112. doi: 10.2147/NSA.S19419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Jiang J, Pi J, Cai J. The advancing of zinc oxide nanoparticles for biomedical applications. Bioinorg Chem Appl. 2018;2018:1062562. doi: 10.1155/2018/1062562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Alomari G, Al-Trad B, Hamdan S, Aljabali AAA, Al Zoubi MS, Al-Batanyeh K, Qar J, Eaton GJ, Alkaraki AK, Alshaer W, et al. Alleviation of diabetic nephropathy by zinc oxide nanoparticles in streptozotocin-induced type 1 diabetes in rats. IET Nanobiotechnol. 2021;15:473–483. doi: 10.1049/nbt2.12026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Abd El-Khalik SR, Nasif E, Arakeep HM, Rabah H. The prospective ameliorative role of zinc oxide nanoparticles in STZ-induced diabetic nephropathy in rats: mechanistic targeting of autophagy and regulating Nrf2/TXNIP/NLRP3 inflammasome signaling. Biol Trace Elem Res. 2022;200:1677–1687. doi: 10.1007/s12011-021-02773-4. [DOI] [PubMed] [Google Scholar]
- 83.Barathmanikanth S, Kalishwaralal K, Sriram M, Pandian SR, Youn HS, Eom S, Gurunathan S. Anti-oxidant effect of gold nanoparticles restrains hyperglycemic conditions in diabetic mice. J Nanobiotechnology. 2010;8:16. doi: 10.1186/1477-3155-8-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Boyer TD, Rouff SL. Acetaminophen-induced hepatic necrosis and renal failure. JAMA. 1971;218:440–441. doi: 10.1001/jama.1971.03190160088021. [DOI] [PubMed] [Google Scholar]
- 85.Tiwari R, Singh RD, Singh S, Singh D, Srivastav AK, Kumar M, Srivastava V. Gestational exposure to silver nanoparticles enhances immune adaptation and protection against streptozotocin-induced diabetic nephropathy in mice offspring. Nanotoxicology. 2022;16:450–471. doi: 10.1080/17435390.2022.2098863. [DOI] [PubMed] [Google Scholar]
- 86.Karthick V, Kumar VG, Dhas TS, Singaravelu G, Sadiq AM, Govindaraju K. Effect of biologically synthesized gold nanoparticles on alloxan-induced diabetic rats-an in vivo approach. Colloids Surf B Biointerfaces. 2014;122:505–511. doi: 10.1016/j.colsurfb.2014.07.022. [DOI] [PubMed] [Google Scholar]
- 87.Alomari G, Al-Trad B, Hamdan S, Aljabali A, Al-Zoubi M, Bataineh N, Qar J, Tambuwala MM. Gold nanoparticles attenuate albuminuria by inhibiting podocyte injury in a rat model of diabetic nephropathy. Drug Deliv Transl Res. 2020;10:216–226. doi: 10.1007/s13346-019-00675-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Cai W, Chu CC, Liu G, Wáng YX. Metal-organic framework-based nanomedicine platforms for drug delivery and molecular imaging. Small. 2015;11:4806–4822. doi: 10.1002/smll.201500802. [DOI] [PubMed] [Google Scholar]
- 89.Fakharzadeh S, Kalanaky S, Argani H, Dadashzadeh S, Torbati PM, Nazaran MH, Basiri A. Ameliorative effect of a nano chromium metal-organic framework on experimental diabetic chronic kidney disease. Drug Dev Res. 2021;82:393–403. doi: 10.1002/ddr.21759. [DOI] [PubMed] [Google Scholar]
- 90.Savage S, Estacio RO, Jeffers B, Schrier RW. Urinary albumin excretion as a predictor of diabetic retinopathy, neuropathy, and cardiovascular disease in NIDDM. Diabetes Care. 1996;19:1243–1248. doi: 10.2337/diacare.19.11.1243. [DOI] [PubMed] [Google Scholar]
- 91.Fakharzadeh S, Argani H, Dadashzadeh S, Kalanaky S, Mohammadi Torbati P, Nazaran MH, Basiri A. BCc1 nanomedicine therapeutic effects in streptozotocin and high-fat diet induced diabetic kidney disease. Diabetes Metab Syndr Obes. 2020;13:1179–1188. doi: 10.2147/DMSO.S240757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Tu Q, Li Y, Jin J, Jiang X, Ren Y, He Q. Curcumin alleviates diabetic nephropathy via inhibiting podocyte mesenchymal transdifferentiation and inducing autophagy in rats and MPC5 cells. Pharm Biol. 2019;57:778–786. doi: 10.1080/13880209.2019.1688843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Ganugula R, Nuthalapati NK, Dwivedi S, Zou D, Arora M, Friend R, Sheikh-Hamad D, Basu R, Kumar M. Nanocurcumin combined with insulin alleviates diabetic kidney disease through P38/P53 signaling axis. J Control Release. 2023;353:621–633. doi: 10.1016/j.jconrel.2022.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Khater SI, Mohamed AA, Arisha AH, Ebraheim LLM, El-Mandrawy SAM, Nassan MA, Mohammed AT, Abdo SA. Stabilized-chitosan selenium nanoparticles efficiently reduce renal tissue injury and regulate the expression pattern of aldose reductase in the diabetic-nephropathy rat model. Life Sci. 2021;279:119674. doi: 10.1016/j.lfs.2021.119674. [DOI] [PubMed] [Google Scholar]
- 95.El-Azab MF, Al-Karmalawy AA, Antar SA, Hanna PA, Tawfik KM, Hazem RM. A novel role of Nano selenium and sildenafil on streptozotocin-induced diabetic nephropathy in rats by modulation of inflammatory, oxidative, and apoptotic pathways. Life Sci. 2022;303:120691. doi: 10.1016/j.lfs.2022.120691. [DOI] [PubMed] [Google Scholar]
- 96.Lotfy MM, Dowidar MF, Ali HA, Ghonimi WAM, Al-Farga A, Ahmed AI. Effect of selenium nanoparticles and/or bee venom against STZ-induced diabetic cardiomyopathy and nephropathy. Metabolites. 2023;13:400. doi: 10.3390/metabo13030400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Zaghloul RA, Abdelghany AM, Samra YA. Rutin and selenium nanoparticles protected against STZ-induced diabetic nephropathy in rats through downregulating Jak-2/Stat3 pathway and upregulating Nrf-2/HO-1 pathway. Eur J Pharmacol. 2022;933:175289. doi: 10.1016/j.ejphar.2022.175289. [DOI] [PubMed] [Google Scholar]
- 98.Tong Y, Zhang L, Gong R, Shi J, Zhong L, Duan X, Zhu Y. A ROS-scavenging multifunctional nanoparticle for combinational therapy of diabetic nephropathy. Nanoscale. 2020;12:23607–23619. doi: 10.1039/D0NR06098D. [DOI] [PubMed] [Google Scholar]
- 99.Nakamura T, Fujiwara N, Sato E, Ueda Y, Sugaya T, Koide H. Renoprotective effects of various angiotensin II receptor blockers in patients with early-stage diabetic nephropathy. Kidney Blood Press Res. 2010;33:213–220. doi: 10.1159/000316707. [DOI] [PubMed] [Google Scholar]
- 100.Ahad A, Raish M, Ahmad A, Al-Jenoobi FI, Al-Mohizea AM. Eprosartan mesylate loaded bilosomes as potential nano-carriers against diabetic nephropathy in streptozotocin-induced diabetic rats. Eur J Pharm Sci. 2018;111:409–417. doi: 10.1016/j.ejps.2017.10.012. [DOI] [PubMed] [Google Scholar]
- 101.Yu M, Wang D, Zhong D, Xie W, Luo J. Adropin carried by reactive oxygen species-responsive nanocapsules ameliorates renal lipid toxicity in diabetic mice. ACS Appl Mater Interfaces. 2022;14:37330–37344. doi: 10.1021/acsami.2c06957. [DOI] [PubMed] [Google Scholar]
- 102.Barutta F, Bellini S, Kimura S, Hase K, Corbetta B, Corbelli A, Fiordaliso F, Bruno S, Biancone L, Barreca A, et al. Protective effect of the tunneling nanotube-TNFAIP2/M-sec system on podocyte autophagy in diabetic nephropathy. Autophagy. 2023;19:505–524. doi: 10.1080/15548627.2022.2080382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Colombo C, Li M, Watanabe S, Messa P, Edefonti A, Montini G, Moscatelli D, Rastaldi MP, Cellesi F. Polymer nanoparticle engineering for podocyte repair: from in vitro models to new nanotherapeutics in kidney diseases. ACS Omega. 2017;2:599–610. doi: 10.1021/acsomega.6b00423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Raval N, Gondaliya P, Tambe V, Kalia K, Tekade RK. Engineered nanoplex mediated targeted miRNA delivery to rescue dying podocytes in diabetic nephropathy. Int J Pharm. 2021;605:120842. doi: 10.1016/j.ijpharm.2021.120842. [DOI] [PubMed] [Google Scholar]
- 105.Asfour MH, Salama AAA, Mohsen AM. Fabrication of all-trans retinoic acid loaded chitosan/tripolyphosphate lipid hybrid nanoparticles as a novel oral delivery approach for management of diabetic nephropathy in rats. J Pharm Sci. 2021;110:3208–3220. doi: 10.1016/j.xphs.2021.05.007. [DOI] [PubMed] [Google Scholar]
- 106.He H, Halseth TA, Mei L, Shen C, Liu L, Schwendeman A. Nanodisc delivery of liver X receptor agonist for the treatment of diabetic nephropathy. J Control Release. 2022;348:1016–1027. doi: 10.1016/j.jconrel.2022.06.029. [DOI] [PubMed] [Google Scholar]
- 107.Mohanram A, Zhang Z, Shahinfar S, Keane WF, Brenner BM, Toto RD. Anemia and end-stage renal disease in patients with type 2 diabetes and nephropathy. Kidney Int. 2004;66:1131–1138. doi: 10.1111/j.1523-1755.2004.00863.x. [DOI] [PubMed] [Google Scholar]
- 108.Wang J, Qian Y, Qian C, Shi F, Yao J, Bi X, Chen Z. A novel β-cyclodextrin-rhein conjugate for improving the water solubility and bioavailability of rhein. Carbohydr Res. 2020;490:107958. doi: 10.1016/j.carres.2020.107958. [DOI] [PubMed] [Google Scholar]
- 109.Wang Q, Han S, Zhu Y, Wang G, Chen D. Poly-γ-glutamic acid coating polymeric nanoparticles enhance renal drug distribution and cellular uptake for diabetic nephropathy therapy. J Drug Target. 2023;31:89–99. doi: 10.1080/1061186X.2022.2106488. [DOI] [PubMed] [Google Scholar]
- 110.Wang G, Li Q, Chen D, Wu B, Wu Y, Tong W, Huang P. Kidney-targeted rhein-loaded liponanoparticles for diabetic nephropathy therapy via size control and enhancement of renal cellular uptake. Theranostics. 2019;9:6191–6208. doi: 10.7150/thno.37538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Boletta A, Benigni A, Lutz J, Remuzzi G, Soria MR, Monaco L. Nonviral gene delivery to the rat kidney with polyethylenimine. Hum Gene Ther. 1997;8:1243–1251. doi: 10.1089/hum.1997.8.10-1243. [DOI] [PubMed] [Google Scholar]
- 112.Chen D, Han S, Zhu Y, Hu F, Wei Y, Wang G. Kidney-targeted drug delivery via rhein-loaded polyethyleneglycol-co-polycaprolactone-co-polyethylenimine nanoparticles for diabetic nephropathy therapy. Int J Nanomedicine. 2018;13:3507–3527. doi: 10.2147/IJN.S166445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Bruni R, Possenti P, Bordignon C, Li M, Ordanini S, Messa P, Rastaldi MP, Cellesi F. Ultrasmall polymeric nanocarriers for drug delivery to podocytes in kidney glomerulus. J Control Release. 2017;255:94–107. doi: 10.1016/j.jconrel.2017.04.005. [DOI] [PubMed] [Google Scholar]
- 114.Yao W, Xu Z, Sun J, Luo J, Wei Y, Zou J. Deoxycholic acid-functionalised nanoparticles for oral delivery of rhein. Eur J Pharm Sci. 2021;159:105713. doi: 10.1016/j.ejps.2021.105713. [DOI] [PubMed] [Google Scholar]
- 115.Huang C, Xue LF, Hu B, Liu HH, Huang SB, Khan S, Meng Y. Calycosin-loaded nanoliposomes as potential nanoplatforms for treatment of diabetic nephropathy through regulation of mitochondrial respiratory function. J Nanobiotechnology. 2021;19:178. doi: 10.1186/s12951-021-00917-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Zhu M, Wang H, Chen J, Zhu H. Sinomenine improve diabetic nephropathy by inhibiting fibrosis and regulating the JAK2/STAT3/SOCS1 pathway in streptozotocin-induced diabetic rats. Life Sci. 2021;265:118855. doi: 10.1016/j.lfs.2020.118855. [DOI] [PubMed] [Google Scholar]
- 117.Chen Y, Chen L, Yang T. Silymarin nanoliposomes attenuate renal injury on diabetic nephropathy rats via co-suppressing TGF-β/Smad and JAK2/STAT3/SOCS1 pathway. Life Sci. 2021;271:119197. doi: 10.1016/j.lfs.2021.119197. [DOI] [PubMed] [Google Scholar]
- 118.Dong Z, Iqbal S, Zhao Z. Preparation of ergosterol-loaded nanostructured lipid carriers for enhancing oral bioavailability and antidiabetic nephropathy effects. AAPS PharmSciTech. 2020;21:64. doi: 10.1208/s12249-019-1597-3. [DOI] [PubMed] [Google Scholar]
- 119.Luijten KM, Tekstra J, Bijlsma JW, Bijl M. The systemic lupus erythematosus responder index (SRI); a new SLE disease activity assessment. Autoimmun Rev. 2012;11:326–329. doi: 10.1016/j.autrev.2011.06.011. [DOI] [PubMed] [Google Scholar]
- 120.Anders HJ, Saxena R, Zhao MH, Parodis I, Salmon JE, Mohan C. Lupus nephritis. Nat Rev Dis Primers. 2020;6:7. doi: 10.1038/s41572-019-0141-9. [DOI] [PubMed] [Google Scholar]
- 121.Chen KJ, Plaunt AJ, Leifer FG, Kang JY, Cipolla D. Recent advances in prodrug-based nanoparticle therapeutics. Eur J Pharm Biopharm. 2021;165:219–243. doi: 10.1016/j.ejpb.2021.04.025. [DOI] [PubMed] [Google Scholar]
- 122.Mehellou Y, Rattan HS, Balzarini J. The protide prodrug technology: from the concept to the clinic. J Med Chem. 2018;61:2211–2226. doi: 10.1021/acs.jmedchem.7b00734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Tessier B, Tsapis N, Fattal E, Moine L. Emerging nanoparticle platforms to improve the administration of glucocorticoids. J Control Release. 2023;358:273–292. doi: 10.1016/j.jconrel.2023.04.039. [DOI] [PubMed] [Google Scholar]
- 124.Guo Q, Chen C, Wu Z, Zhang W, Wang L, Yu J, Li L, Zhang J, Duan Y. Engineered PD-1/TIGIT dual-activating cell-membrane nanoparticles with dexamethasone act synergistically to shape the effector T cell/Treg balance and alleviate systemic lupus erythematosus. Biomaterials. 2022;285:121517. doi: 10.1016/j.biomaterials.2022.121517. [DOI] [PubMed] [Google Scholar]
- 125.Jia Z, Wang X, Wei X, Zhao G, Foster KW, Qiu F, Gao Y, Yuan F, Yu F, Thiele GM, et al. Micelle-forming dexamethasone prodrug attenuates nephritis in lupus-prone mice without apparent glucocorticoid side effects. ACS Nano. 2018;12:7663–7681. doi: 10.1021/acsnano.8b01249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Zhao Z, Jiang H, Xu X, Jia Z, Ren R, Foster KW, Wei X, Chen N, Goldring SR, Crow MK, Wang D. Polymeric dexamethasone prodrugs attenuate lupus nephritis in MRL/lpr mice with reduced glucocorticoid toxicity. Nanomedicine. 2022;44:102579. doi: 10.1016/j.nano.2022.102579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Mok CC. Pro: the use of calcineurin inhibitors in the treatment of lupus nephritis. Nephrol Dial Transplant. 2016;31:1561–1566. doi: 10.1093/ndt/gfw289. [DOI] [PubMed] [Google Scholar]
- 128.Ina K, Kusugami K, Shimada M, Tsuzuki T, Nishio Y, Binion DG, Imada A, Ando T. Suppressive effects of cyclosporine A on neutrophils and T cells may be related to therapeutic benefits in patients with steroid-resistant ulcerative colitis. Inflamm Bowel Dis. 2002;8:1–9. doi: 10.1097/00054725-200201000-00001. [DOI] [PubMed] [Google Scholar]
- 129.Sumethkul K, Kitumnuaypong T, Angthararak S, Pichaiwong W. Low-dose cyclosporine for active lupus nephritis: a dose titration approach. Clin Rheumatol. 2019;38:2151–2159. doi: 10.1007/s10067-019-04469-6. [DOI] [PubMed] [Google Scholar]
- 130.Ganugula R, Arora M, Zou D, Agarwal SK, Mohan C, Kumar M. A highly potent lymphatic system-targeting nanoparticle cyclosporine prevents glomerulonephritis in mouse model of lupus. Sci Adv. 2020;6:eabb3900. doi: 10.1126/sciadv.abb3900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Naesens M, Kuypers DR, Sarwal M. Calcineurin inhibitor nephrotoxicity. Clin J Am Soc Nephrol. 2009;4:481–508. doi: 10.2215/CJN.04800908. [DOI] [PubMed] [Google Scholar]
- 132.Kim CS, Mathew AP, Vasukutty A, Uthaman S, Joo SY, Bae EH, Ma SK, Park IK, Kim SW. Glycol chitosan-based tacrolimus-loaded nanomicelle therapy ameliorates lupus nephritis. J Nanobiotechnology. 2021;19:109. doi: 10.1186/s12951-021-00857-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Li Y, Qi W. Experimental study of nanostructured lipid carrier in the treatment of immune lupus nephritis. J Nanosci Nanotechnol. 2021;21:1099–1106. doi: 10.1166/jnn.2021.18671. [DOI] [PubMed] [Google Scholar]
- 134.Cohen P, Cross D, Jänne PA. Kinase drug discovery 20 years after imatinib: progress and future directions. Nat Rev Drug Discov. 2021;20:551–569. doi: 10.1038/s41573-021-00195-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Fogueri U, Charkoftaki G, Roda G, Tuey S, Ibrahim M, Persaud I, Wempe MF, Brown JM, Thurman JM, Anchordoquy TJ, Joy MS. An evaluation of a novel nanoformulation of imatinib mesylate in a mouse model of lupus nephritis. Drug Deliv Transl Res. 2022;12:1445–1454. doi: 10.1007/s13346-021-01022-4. [DOI] [PubMed] [Google Scholar]
- 136.Chen Y, Wang Y, Chen Z, Cai J, Li K, Huang H, Song F, Gao M, Yang Y, Zheng L, Zhao J. NIR-driven polydopamine-based nanoenzymes as ROS scavengers to suppress osteoarthritis progression. Materials Today Nano. 2022;19:100240. doi: 10.1016/j.mtnano.2022.100240. [DOI] [Google Scholar]
- 137.Xie L, Pang X, Yan X, Dai Q, Lin H, Ye J, Cheng Y, Zhao Q, Ma X, Zhang X, et al. Photoacoustic imaging-trackable magnetic microswimmers for pathogenic bacterial infection treatment. ACS Nano. 2020;14:2880–2893. doi: 10.1021/acsnano.9b06731. [DOI] [PubMed] [Google Scholar]
- 138.Li M, Wang Y, Han X, Liu Y, Ma M, Zhang L. Multifunctional polydopamine-based nanoparticles for dual-mode imaging guided targeted therapy of lupus nephritis. Pharmaceutics. 1988;2022:14. doi: 10.3390/pharmaceutics14101988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Xu W, Chen Z, Shen X, Pi C. Reno-protective effect of realgar nanoparticles on lupus nephritis of MRL/Lpr mice through STAT1. Iran J Immunol. 2019;16:170–181. doi: 10.22034/IJI.2019.80260. [DOI] [PubMed] [Google Scholar]
- 140.Wang J, Gan M. DNA nanoflowers' amelioration of lupus symptoms in mice via blockade of TLR7/9's signal. Int J Mol Sci. 2022;23:16030. doi: 10.3390/ijms232416030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Takata H, Shimizu T, Kawaguchi Y, Ueda H, Elsadek NE, Ando H, Ishima Y, Ishida T. Nucleic acids delivered by PEGylated cationic liposomes in systemic lupus erythematosus-prone mice: a possible exacerbation of lupus nephritis in the presence of pre-existing anti-nucleic acid antibodies. Int J Pharm. 2021;601:120529. doi: 10.1016/j.ijpharm.2021.120529. [DOI] [PubMed] [Google Scholar]
- 142.Wang L, Xie X, Ke B, Huang W, Jiang X, He G. Recent advances on endogenous gasotransmitters in inflammatory dermatological disorders. J Adv Res. 2022;38:261–274. doi: 10.1016/j.jare.2021.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Fang Y, Cheng J, Shen Z, You T, Ding S, Hu J. Ultrasound-mediated release of gaseous signaling molecules for biomedical applications. Macromol Rapid Commun. 2022;43:e2100814. doi: 10.1002/marc.202100814. [DOI] [PubMed] [Google Scholar]
- 144.Wang H, Wang L, Xie Z, Zhou S, Li Y, Zhou Y, Sun M. Nitric oxide (NO) and NO synthases (NOS)-based targeted therapy for colon cancer. Cancers. 2020;12:1881. doi: 10.3390/cancers12071881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Wallace JL, Vaughan D, Dicay M, MacNaughton WK, de Nucci G. Hydrogen sulfide-releasing therapeutics: translation to the clinic. Antioxid Redox Signal. 2018;28:1533–1540. doi: 10.1089/ars.2017.7068. [DOI] [PubMed] [Google Scholar]
- 146.Pathak V, Roemhild K, Schipper S, Groß-Weege N, Nolte T, Ruetten S, Buhl EM, El Shafei A, Weiler M, Martin L, et al. Theranostic trigger-responsive carbon monoxide-generating microbubbles. Small. 2022;18:e2200924. doi: 10.1002/smll.202200924. [DOI] [PubMed] [Google Scholar]
- 147.Li S, Liao R, Sheng X, Luo X, Zhang X, Wen X, Zhou J, Peng K. Hydrogen gas in cancer treatment. Front Oncol. 2019;9:696. doi: 10.3389/fonc.2019.00696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Sun R, Liu X, Li G, Wang H, Luo Y, Huang G, Wang X, Zeng G, Liu Z, Wu S. Photoactivated H(2) nanogenerator for enhanced chemotherapy of bladder cancer. ACS Nano. 2020;14:8135–8148. doi: 10.1021/acsnano.0c01300. [DOI] [PubMed] [Google Scholar]
- 149.Wu Y, Yuan M, Song J, Chen X, Yang H. Hydrogen gas from inflammation treatment to cancer therapy. ACS Nano. 2019;13:8505–8511. doi: 10.1021/acsnano.9b05124. [DOI] [PubMed] [Google Scholar]
- 150.Hendriks KD, Maassen H, van Dijk PR, Henning RH, van Goor H, Hillebrands JL. Gasotransmitters in health and disease: a mitochondria-centered view. Curr Opin Pharmacol. 2019;45:87–93. doi: 10.1016/j.coph.2019.07.001. [DOI] [PubMed] [Google Scholar]
- 151.Kimura H. Hydrogen sulfide (H(2)S) and polysulfide (H(2)S(n)) signaling: the first 25 years. Biomolecules. 2021;11:896. doi: 10.3390/biom11060896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Shibuya N, Koike S, Tanaka M, Ishigami-Yuasa M, Kimura Y, Ogasawara Y, Fukui K, Nagahara N, Kimura H. A novel pathway for the production of hydrogen sulfide from D-cysteine in mammalian cells. Nat Commun. 2013;4:1366. doi: 10.1038/ncomms2371. [DOI] [PubMed] [Google Scholar]
- 153.Han SJ, Kim JI, Park JW, Park KM. Hydrogen sulfide accelerates the recovery of kidney tubules after renal ischemia/reperfusion injury. Nephrol Dial Transplant. 2015;30:1497–1506. doi: 10.1093/ndt/gfv226. [DOI] [PubMed] [Google Scholar]
- 154.Ahmad A, Olah G, Szczesny B, Wood ME, Whiteman M, Szabo C. AP39, a mitochondrially targeted hydrogen sulfide donor, exerts protective effects in renal epithelial cells subjected to oxidative stress in vitro and in acute renal injury in vivo. Shock. 2016;45:88–97. doi: 10.1097/SHK.0000000000000478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Altaany Z, Yang G, Wang R. Crosstalk between hydrogen sulfide and nitric oxide in endothelial cells. J Cell Mol Med. 2013;17:879–888. doi: 10.1111/jcmm.12077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Svegliati-Baroni G, Saccomanno S, van Goor H, Jansen P, Benedetti A, Moshage H. Involvement of reactive oxygen species and nitric oxide radicals in activation and proliferation of rat hepatic stellate cells. Liver. 2001;21:1–12. doi: 10.1034/j.1600-0676.2001.210101.x. [DOI] [PubMed] [Google Scholar]
- 157.Pini A, Viappiani S, Bolla M, Masini E, Bani D. Prevention of bleomycin-induced lung fibrosis in mice by a novel approach of parallel inhibition of cyclooxygenase and nitric-oxide donation using NCX 466, a prototype cyclooxygenase inhibitor and nitric-oxide donor. J Pharmacol Exp Ther. 2012;341:493–499. doi: 10.1124/jpet.111.190660. [DOI] [PubMed] [Google Scholar]
- 158.Wani J, Carl M, Henger A, Nelson PJ, Rupprecht H. Nitric oxide modulates expression of extracellular matrix genes linked to fibrosis in kidney mesangial cells. Biol Chem. 2007;388:497–506. doi: 10.1515/BC.2007.056. [DOI] [PubMed] [Google Scholar]
- 159.Boczkowski J, Poderoso JJ, Motterlini R. CO-metal interaction: vital signaling from a lethal gas. Trends Biochem Sci. 2006;31:614–621. doi: 10.1016/j.tibs.2006.09.001. [DOI] [PubMed] [Google Scholar]
- 160.Xia Z, Zhang C, Guo C, Song B, Hu W, Cui Y, Xue Y, Xia M, Xu D, Zhang S, Fang J. Nanoformulation of a carbon monoxide releasing molecule protects against cyclosporin A-induced nephrotoxicity and renal fibrosis via the suppression of the NLRP3 inflammasome mediated TGF-β/Smad pathway. Acta Biomater. 2022;144:42–53. doi: 10.1016/j.actbio.2022.03.024. [DOI] [PubMed] [Google Scholar]
- 161.Liu C, Du Z, Ma M, Sun Y, Ren J, Qu X. Carbon monoxide controllable targeted gas therapy for synergistic anti-inflammation. iScience. 2020;23:101483. doi: 10.1016/j.isci.2020.101483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Huang K, Ma H, Liu J, Huo S, Kumar A, Wei T, Zhang X, Jin S, Gan Y, Wang PC, et al. Size-dependent localization and penetration of ultrasmall gold nanoparticles in cancer cells, multicellular spheroids, and tumors in vivo. ACS Nano. 2012;6:4483–4493. doi: 10.1021/nn301282m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Zhang XD, Wu D, Shen X, Chen J, Sun YM, Liu PX, Liang XJ. Size-dependent radiosensitization of PEG-coated gold nanoparticles for cancer radiation therapy. Biomaterials. 2012;33:6408–6419. doi: 10.1016/j.biomaterials.2012.05.047. [DOI] [PubMed] [Google Scholar]
- 164.Huo S, Jin S, Ma X, Xue X, Yang K, Kumar A, Wang PC, Zhang J, Hu Z, Liang XJ. Ultrasmall gold nanoparticles as carriers for nucleus-based gene therapy due to size-dependent nuclear entry. ACS Nano. 2014;8:5852–5862. doi: 10.1021/nn5008572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Fan M, Han Y, Gao S, Yan H, Cao L, Li Z, Liang XJ, Zhang J. Ultrasmall gold nanoparticles in cancer diagnosis and therapy. Theranostics. 2020;10:4944–4957. doi: 10.7150/thno.42471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Mittal M, Siddiqui MR, Tran K, Reddy SP, Malik AB. Reactive oxygen species in inflammation and tissue injury. Antioxid Redox Signal. 2014;20:1126–1167. doi: 10.1089/ars.2012.5149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Rimessi A, Previati M, Nigro F, Wieckowski MR, Pinton P. Mitochondrial reactive oxygen species and inflammation: Molecular mechanisms, diseases and promising therapies. Int J Biochem Cell Biol. 2016;81:281–293. doi: 10.1016/j.biocel.2016.06.015. [DOI] [PubMed] [Google Scholar]
- 168.Abraham NG, Kappas A. Pharmacological and clinical aspects of heme oxygenase. Pharmacol Rev. 2008;60:79–127. doi: 10.1124/pr.107.07104. [DOI] [PubMed] [Google Scholar]
- 169.Liu T, Xiao B, Xiang F, Tan J, Chen Z, Zhang X, Wu C, Mao Z, Luo G, Chen X, Deng J. Ultrasmall copper-based nanoparticles for reactive oxygen species scavenging and alleviation of inflammation related diseases. Nat Commun. 2020;11:2788. doi: 10.1038/s41467-020-16544-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Liu D, Dai X, Zhang W, Zhu X, Zha Z, Qian H, Cheng L, Wang X. Liquid exfoliation of ultrasmall zirconium carbide nanodots as a noninflammatory photothermal agent in the treatment of glioma. Biomaterials. 2023;292:121917. doi: 10.1016/j.biomaterials.2022.121917. [DOI] [PubMed] [Google Scholar]
- 171.Yin M, Chen X, Guo Q, Xiao L, Gao P, Zang D, Dong J, Zha Z, Dai X, Wang X. Ultrasmall zirconium carbide nanodots for synergistic photothermal-radiotherapy of glioma. Nanoscale. 2022;14:14935–14949. doi: 10.1039/D2NR04239H. [DOI] [PubMed] [Google Scholar]
- 172.Kim J, Hong G, Mazaleuskaya L, Hsu JC, Rosario-Berrios DN, Grosser T, Cho-Park PF, Cormode DP. Ultrasmall antioxidant cerium oxide nanoparticles for regulation of acute inflammation. ACS Appl Mater Interfaces. 2021;13:60852–60864. doi: 10.1021/acsami.1c16126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Higa LH, Jerez HE, de Farias MA, Portugal RV, Romero EL, Morilla MJ. Ultra-small solid archaeolipid nanoparticles for active targeting to macrophages of the inflamed mucosa. Nanomedicine. 2017;12:1165–1175. doi: 10.2217/nnm-2016-0437. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All the data reported in this manuscript is available within the text.