Abstract
Emicizumab is a humanized, bispecific monoclonal antibody that connects active factor IX and X to replace the function of absent factor VIII, restoring hemostasis. It has a long half-life with a subcutaneous route of administration and high bioavailability. Here, we assessed the efficacy of Emicizumab prophylaxis in terms of efficiency, safety, and quality of life of severe hemophilia A (HA) patients with and without inhibitors before and after this treatment.
Methods
In this prospective study, severe HA patients were recruited from January 2022 to June 2023. Inhibitor positive and inhibitor negative patients with annual bleeding rate (ABR) 8 or greater and past histories of bleeding like intra-cranial, intra-abdominal, and pseudo-tumors were included. Emicizumab loading dose was 3 mg/kg in the first 4 weeks, and the maintenance dose was started at week 5 at 6 mg/kg/month. Patients’ detailed bleeding history and demographics were recorded. The five-level EuroQol five-dimensional questionnaire (EQ-5D-5L) was used to evaluate patients’ HRQoL. Furthermore, Hemophilia Joint Health Score (HJHS) and Functional Independence score in Hemophilia (FISH) were applied for the assessment of joints at different time points. Results were analyzed by SPSS version 21.
Results
A total of 36 HA male patients with the mean age of 19.7 ± 14.42 years were recruited in the study; among them, 19 patients were inhibitor positive, while 17 were negative. Patients clinically presented with bleeding symptoms which included: hemarthrosis 95%, GI bleeding 13.8%, and bruises and gums bleeding 13.8%. Significant reduction was observed in the bleeding episodes after the therapeutic intervention, and joints assessment and Euro-Quality-of-life Visual Analog Scale showed a significant improvement in health after treatment. Similarly, there was a remarkable reduction in bleeding episodes and improved quality of life among HA patients. The ABR decreased from 53.6% episodes per year prior to treatment to 2.4% during Emicizumab therapy. Prior to initiating Emicizumab therapy, participants exhibited an average FISH score of 16 and HJHS score of 10, indicating moderate limitations due to joint-related issues. After treatment, the mean FISH score improved to 9 and HJHS score to 4 reflecting a substantial enhancement in participants’ ability to perform daily activities (P < 0.057).
Conclusion
Our results showed that HA patients on prophylactic treatment with Emicizumab were less restricted and had improved quality of life due to marked decrease in bleeding episodes which resulted in improved health and social lives. In addition, it was well tolerated, and no participant discontinued treatment because of adverse events.
Keywords: hemophilia A, bleeding, joint score, health-related quality of life, Hemophilia Joint Health Score, Functional Independence Score in Hemophilia, EQ-5D-5L
Introduction
The clotting factor VIII (FVIII) deficiency is an uncommon bleeding condition known as hemophilia A (HA). 1 It is an X-linked recessive disorder. The plasma levels of factor VIII determine the severity of HA patients. 2 Hemophilic arthropathy, a severe hemophilia complication, manifests as repeated spontaneous bleeding into muscles and joints. 3 In addition, severe hemophilia patients can potentially have life-threatening bleeding, such as intra-cranial hemorrhage, gastro intestinal bleed, joint bleeds, and malena. 4 The treatment of acute bleeding episodes is replacement of FVIII in these patients. 5 FVIII infusion requires regular dosage due to its short plasma half-life. HA patients who need FVIII replacement as prophylaxis get intravenous FVIII treatment at least two to three times per week. However, an important complication of FVIII treatment includes inhibitor development in which FVIII concentrates become ineffective in controlling bleeding symptoms and treatment becomes challenging in these patients requiring bypassing agents. 6
Emicizumab (Hemlibra®) was designed to avoid the problems associated with repeated intravenous administration of FVIII and to provide a standardized treatment option for HA patients with or without inhibitors. 7 The drug Emicizumab is a humanized recombinant bispecific monoclonal antibody that restores missing activated FVIII function regardless of the factor VIII levels. Emicizumab promotes effective hemostasis in HA patients8–10 and is considered to replace the hemostatic function of activated FVIII by combining activated factor IX and factor X (FX) to activate FX, facilitating the coagulation cascade and achieving hemostasis in HA patients. 11 The World Federation of Hemophilia (WFH) is providing humanitarian aid to underdeveloped countries to promote prophylaxis there. Benefits of prophylaxis are well known such as reduced pain and discomfort, reduced frequency of joint bleeding, and improved Qol but due to high cost and unavailability of factor concentrates, this is partially practiced developing countries. Fortunately, Pakistan is receiving factor concentrates and Emicizumab humanitarian aid for hemophilia patients through the Hemophilia Foundation of Pakistan which has registered hemophilia societies in different cities of the country. Hemophilia Welfare Society Karachi has registered congenital bleeding disorder patients (n = 1200) across the province of Sindh where patients receive treatment and rehabilitation through a multidisciplinary team. In the present study, we assessed the efficacy in terms of bleeding, safety, and quality of life of HA patients with and without inhibitors on Emicizumab prophylaxis (donated by the WFH through humanitarian aid to Pakistan) before and after this treatment.
Material and Methods
A prospective study was conducted at the Hemophilia Welfare Society Karachi, Pakistan (HWSK) from January 2022 to June 2023. This study was approved by the ethics committee of the HWSK/ 16-263/01-2022 in accordance with the declaration of Helsinki. Already diagnosed and registered severe HA male patients with inhibitors and without inhibitors were included with annual bleeding rate (ABR) 8 or greater and history of bleeding like intra-cranial (IC), intra-abdominal (GI), and pseudo-tumors were included in this study (the inclusion criteria set by WFH). The baseline clinical and laboratory data were collected. Emicizumab loading dose was 3 mg/kg in the first 4 weeks and the maintenance dose was started at week 5 at a dose of 6 mg/kg/month. The EuroQol five-dimensional questionnaire (EQ-5D-5L) included mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. These metrics were used to evaluate patients’ HRQoL at baseline and then at 3 monthly intervals through this questionnaire. Furthermore, Hemophilia Joint Health Score (HJHS) and Functional Independence Score in Hemophilia (FISH) were applied for the assessment of joints at different time points (every 3 months).
Statistical Analysis
Statistical analysis was performed using SPSS-21 (Statistical Package for the Social Sciences). It was conducted to systematically examine and interpret the data collected in this study. By utilizing quantitative analysis, we aimed to provide objective and measurable insights into the effectiveness of the treatment. To determine the statistical significance of our findings, we established a value of significance, denoted as α (alpha) at 0.05. This signifies that we are willing to accept a 5% chance of making a type I error, which involves incorrectly rejecting the null hypothesis. A P-value less than α indicates that the observed effects are statistically significant, and we can confidently reject the null hypothesis in favor of the alternative hypothesis. P-values were used for hypothesis testing, suggesting a significant decrease in joint hemorrhages and an associated improvement in patients’ mobility and joint function.
Employing a multi-faceted approach, we conducted qualitative and descriptive analyses to comprehensively assess the impact of the treatment on this cohort. Notably, our statistical analyses yielded significant results with a value of significance (P < 0.05). This statistical significance underscores the credibility of our findings, indicating that the observed effects are unlikely to have occurred by random chance. Furthermore, the implementation of a two-tailed ANOVA allowed us to distinguish meaningful differences before and after treatment, offering valuable insights into the treatment's effectiveness across multiple variables. In addition, a one-sample t-test specifically focused on the FISH scoring, providing a targeted evaluation of this specific aspect.
Results
Thirty-six male severe HA patients with mean ± SD 19.7 ± 14.42 years were enrolled. Among them, patients with inhibitor positive of more than 0.4 Bethesda unit / ml (BU) were 19 (52.77%), under 12 years were 6 (16.6%), and above 12 years were 13 (36.1%). Patients less than 12 years of age without inhibitor with ABR 8 or greater and past history of major bleeding (IC, GI, pseudo-tumors were 5 (13.8%). In addition, patients more than 12 years of age without inhibitors on a case-to-case basis and past histories of major bleeding were 12 (33.3%). Among them, history of consanguinity was present in 30 patients (83.3%) and family history of bleeding in 29 (80.5%). Details of demographic and clinical characteristics before and after the treatment of Emicizumab are shown in Table 1.
Table 1.
Demographic Characteristics of HA Patients Before and After Treatment of Emicizumab.
| S No. | Demographic Characteristics | Before Treatment | After Treatment | |
|---|---|---|---|---|
| Age (years) | 19.7 ± 14.42 | 20.7 ± 15.42 | ||
| 1. | Sex | Male | Male | |
| 2. | Total no. of hospitalization | N = 47 | N = 2 | |
| 3. | Absenteeism(days) | Mean = 69 | Mean = 5 | |
| 4. | Major bleeding |
|
|
|
| 5. | Minor bleeding |
|
|
|
| 6. | ABR | n = 110.6 | n = 2.6 | |
| 7. | Inhibitor | Positive (30.5%) | Positive (<1%) | |
| 8. | Prophylaxis patients to on demand therapy | (89%) | (10%) | |
| 9. | No of transfusions |
|
|
|
| 10. | Adverse reactions to therapy |
|
|
|
| 11. | Any surgery performed |
|
|
|
| 12. | FISH scoring | Average scoring; (16) | Average scoring; (9) | P-value; 0.057 |
| 13. | HJHS | Average scoring;(10) | Average scoring;(4) | |
HA, hemophilia; ABR, annual bleeding rate; FISH, Functional Independence score in Hemophilia; HJHS, Hemophilia Joint Health Score.
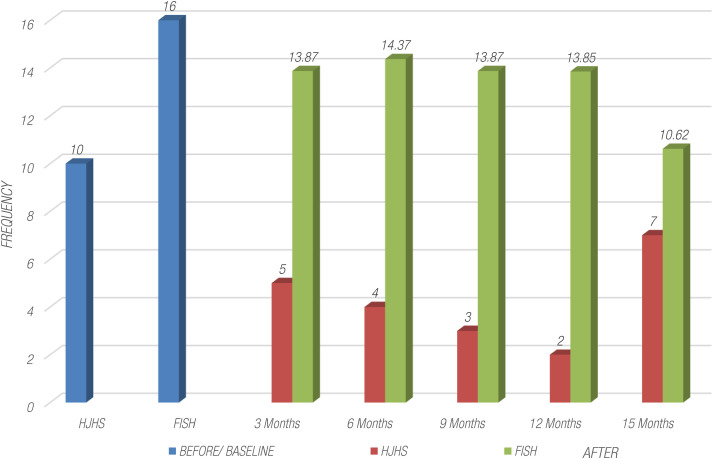
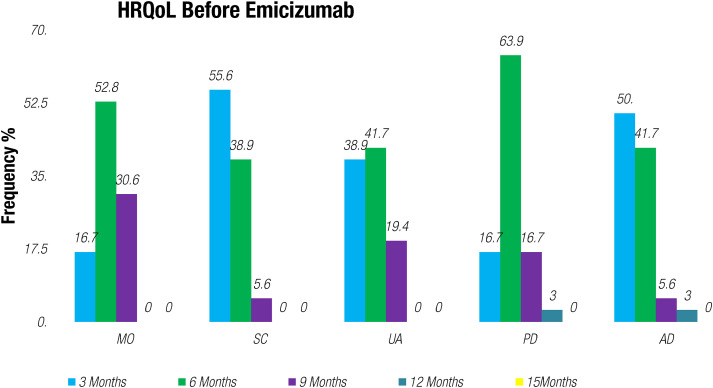
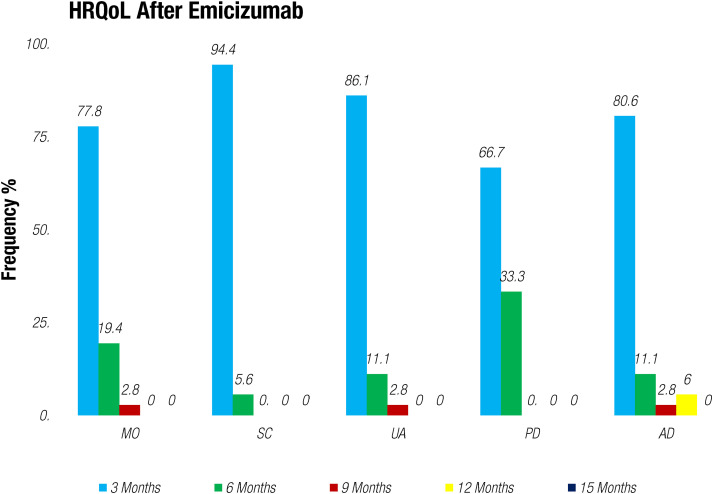
Upon initiating treatment with Emicizumab, a marked and statistically significant improvement was observed in patients’ weight as average mean weight increased from 41 kg to 64.62 kg (n = 36), severity of major and minor bleedings decreased to <1%, and percentage of ABR reduced to 2.4%. When evaluating the HJHS and FISH scoring before and after treatment with Emicizumab, we evaluated the level of joint damage. Specific joint evaluations revealed that knee joints had the highest mean HJHS score (mean pre-treatment score = 10), followed by FISH scores by functional domain indicated that mobility had the lowest mean score (mean pre-treatment score = 16((16.1 ± 8.5)). Following the administration of Emicizumab, there was a notable reduction in the mean HJHS and FISH scores, and this decrease was statistically significant (P < 0.057) indicated an improvement in joint health as shown in Figure 1. Additionally included in the observed average EQ-5D-5L index score, mobility showed an impressive improvement of 63.3% (P=0.000) indicated a substantial increase in patients’ ability to move and engage in physical activities. Self-care with an improvement rate of 66.6% (P=0.009) demonstrated enhanced independence in self-care tasks and regular activities, and a significant improvement rate of 70% (P=0.000) pointed to a significant positive shift in patients’ ability to carry out their usual daily activities without pain/discomfort. Notably, the improvement in this domain was 52.6% (P=0.000) highlighting a reduction in pain and discomfort experienced by the patients. Similarly, anxiety/depression also improved 58.6% (P=0.000). Our results demonstrated a remarkable progress in overall joint health scores, showing a significant 45% (P=0.057) increase after every 3 months of therapy. This improvement indicated a significant reduction in joint hemorrhages and an associated increase in patients’ joint mobility as demonstrated in Figures 2 and 3. Importantly, this outcome was statistically significant (P < 0.05), reinforcing the therapeutic benefits of Emicizumab on joints health. The positive impact on both Quality-Of-Life domains and joints health underscores the significance of this therapeutic intervention for enhancing the well-being and clinical outcomes of patients with HA.
Figure 1.
Evaluation of HJHS and FISH scoring before and after treatment of Emicizumab. FISH, Functional Independence score in Hemophilia, HJHS, Hemophilia Joint Health Score.
Figure 2.
HRQoL Pre-treatment with Emicizumab. MO: mobility SC: self-care UA: usual activities PD: pain/discomfort AD: anxiety/depression.
Figure 3.
HRQoL Post-treatment with Emicizumab. MO: mobility SC: self-care UA: usual activities PD: pain/discomfort AD: anxiety/depression.
Discussion
This study was conducted on severe HA patients receiving Emicizumab prophylaxis highlighting the efficacy of its treatment in terms of bleeding, safety, and quality of life with and without inhibitors and its analysis shows that it was well tolerated with improvement in the joint scores in different intervals of time. Moreover, efficacy data were consistent among those with or without FVIII inhibitors and a significant reduction in rate of bleeding was observed after the treatment with ABR (2.4%) highlighting the safety and better clinical outcomes with the administration of Emicizumab in HA patients which was consistent with the findings of previously reported studies.12–15 In addition, further benefits of Emicizumab were evaluated and significantly reduced rate of bleeding was observed in patients with major and minor bleeding episodes. More than 90% of the patients receiving prophylaxis experienced reduced joint bleed (2.4%) after receiving Emicizumab suggesting its long-term efficacy which was also observed by the HAVEN program. 16
The HRQoL is a five-level questionnaire that assesses on mobility, self-care, usual activities, pain/discomfort, and anxiety/depression, and there was a notable reduction in the mean HJHS and FISH scores, and this decrease was statistically significant (P < 0.057), indicating an improvement in joint health. (Figure 1) The significant improvement in this domain was 52.6% (P=0.000) highlighting a reduction in pain and discomfort experienced by the patients. Similarly, anxiety/depression improved 58.6% (P=0.000) which was also observed in previous studies.17–19 Additionally, the HJHS and FISH scores were also evaluated for the assessment of joint damage and significant reduced scores were observed after the treatment of Emicizumab (P < 0.057) indicating better response of Emicizumab for the joint damage as the improvement was also observed in young hemophilic patients in Haven 3 study without the factor VIII inhibitors. 20
The baseline parameters like age and occurrence of target joint are important predictors for the improvement of HRQoL, HJHS, and FISH scores that emphasizes for further multi-center studies with large number of sample size from different countries. This was the first single-center prospective study conducted to assess the efficacy in terms of bleeding, safety, and quality of life of HA patients with and without inhibitors on Emicizumab prophylaxis before and after treatment. This emphasizes on the use on prophylaxis especially in the severe hemophilic patients for better outcomes in children and adolescences to promote zero bleeds in these patients.
Conclusion
Significant and important improvements were observed in terms of bleeding, HRQoL, HJHS, and FISH scores in severe HA patients treated with Emicizumab prophylaxis. These strong efficacy and safety data, together with a clinically significant improvement suggest that Emicizumab improves patient care by reducing the burden of treatment, which in turn allows effective adherence to prophylaxis and reduces secondary complications in these patients. In conclusion, this study's findings show a lot of possibilities for solving serious issues in terms of joint bleeding and deformities, QoL and cost-effective therapy as compared to on-demand treatment. By recognizing the revolutionary potential of this research, we open the door for creative solutions that will enable developing countries to rise above hardship and create more promising and resilient futures.
Acknowledgements
Authors acknowledge Madiha Abid and Sidra Zafar for helping in data analysis and Maliha Umair for physiotherapy assessment.
Footnotes
Authors Contributions: M. Borhany conceived the idea of this study and wrote the manuscript. A. Arshad helped with writing. Rakshanda did data collection and physiotherapy assessment. H. Qureshi did data analysis. R. Ahmed helped in patient assessment and counseling along with M. Borhany. All authors have read and approved the manuscript.
Consent: Written informed consent was obtained from the participants of the study for their anonymized information to be published.
Data Availability: The Dataset used and/or analyzed during the current study is available from the corresponding author on reasonable request.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval: A prospective study was conducted at the Hemophilia Welfare Society Karachi, Pakistan, (HWSK) from January 2022 to June 2023.This study was approved by the ethics committee of the HWSK/ 16-263/01-2022 in accordance with the declaration of Helsinki. Confidentiality of participants and privacy was maintained during the data collection process.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received a grant from the WFH (World Federation of Hemophilia).
Future Directions: Conducting multi-center studies with a greater number of patients with hemophilia may help to understand the effects Emicizumab prophylaxis on the outcome, especially in our population.
ORCID iD: Munira Borhany https://orcid.org/0000-0001-7867-1275
References
- 1.Srivastava A, Santagostino E, Dougall A, et al. WFH guidelines for the management of hemophilia, 3rd edition. Haemophilia. 2020;27(4). doi: 10.1111/hae.14046 [DOI] [PubMed] [Google Scholar]
- 2.Blanchette V, Key N, Ljung L, Manco-Johnson M, Van Den Berg H, Srivastava A. Definitions in hemophilia: Communication from the SSC of the ISTH. J Thromb Haemostasis. 2014;12(11):1935-1939. [DOI] [PubMed] [Google Scholar]
- 3.Kizilocak H, Young G. Diagnosis and treatment of hemophilia. Clin Adv Hematol Oncol. 2019;17(6):344-351. [PubMed] [Google Scholar]
- 4.Witmer C, Presley R, Kulkarni R, Michael Soucie J, Manno CS, Raffini L. Associations between intracranial haemorrhage and prescribed prophylaxis in a large cohort of haemophilia patients in the United States. Br J Haematol. 2011;152(2):211-216. [DOI] [PubMed] [Google Scholar]
- 5.Bauer KA. Current challenges in the management of hemophilia. Am J Manag Care. 2015;21(6 Suppl):S112–-S122. [PubMed] [Google Scholar]
- 6.Haya S. Prophylactic treatment in hemophilic patients with inhibitors. Blood Coagul Fibrinolysis. 2019;30(1S Suppl 1):S14–-S18. [DOI] [PubMed] [Google Scholar]
- 7.Oldenburg J, Mahlangu JN, Bujan W, et al. The effect of emicizumab prophylaxis on health-related outcomes in persons with haemophilia A with inhibitors: HAVEN 1 study. Haemophilia. 2019;25(1):33-44. [DOI] [PubMed] [Google Scholar]
- 8.Barg AA, Avishai E, Budnik I, et al. Emicizumab prophylaxis among infants and toddlers with severe hemophilia A and inhibitors—a single-center cohort. Pediatr Blood Cancer. 2019;66(11):e27886. [DOI] [PubMed] [Google Scholar]
- 9.Blair HA. Emicizumab: A review in haemophilia A. Drugs. 2019;79(15):1697-1707. [DOI] [PubMed] [Google Scholar]
- 10.Knoebl P, Thaler J, Jilma P, Quehenberger P, Gleixner K, Sperr WR. Emicizumab for the treatment of acquired hemophilia A. Blood. J Am Soc Hematol. 2021;137(3):410-419. [DOI] [PubMed] [Google Scholar]
- 11.Gelbenegger G, Schoergenhofer C, Knoebl P, Jilma B. Bridging the missing link with emicizumab: A bispecific antibody for treatment of hemophilia A. Thromb Haemostasis. 2020;120(10):1357-1370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Oldenburg J, Mahlangu JN, Kim B, et al. Emicizumab prophylaxis in hemophilia A with inhibitors. N Engl J Med. 2017;377(9):809-818. [DOI] [PubMed] [Google Scholar]
- 13.Mahlangu J, Oldenburg J, Paz-Priel I, et al. Emicizumab prophylaxis in patients who have hemophilia A without inhibitors. N Engl J Med. 2018;379(9):811-822. [DOI] [PubMed] [Google Scholar]
- 14.Young G, Liesner R, Chang T, et al. A multicenter, open-label phase 3 study of emicizumab prophylaxis in children with hemophilia A with inhibitors. Blood. 2019;134(24):2127-2138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shima M, Nogami K, Nagami S, et al. A multicentre, open-label study of emicizumab given every 2 or 4 weeks in children with severe haemophilia A without inhibitors. Haemophilia. 2019;25(6):979-987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Callaghan MU, Negrier C, Paz-Priel I, et al. Long-term outcomes with emicizumab prophylaxis for hemophilia A with or without FVIII inhibitors from the HAVEN 1-4 studies. Blood. 2021;137(16):2231-2242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Antunes S, Tangada S, Stasyshyn O, et al. Randomized comparison of prophylaxis and on-demand regimens with FEIBA NF in the treatment of haemophilia A and B with inhibitors. Haemophilia. 2014;20(1):65-72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wyrwich K, Krishnan S, Poon J, et al. Interpreting important health-related quality of life change using the Haem-A-QoL. Haemophilia. 2015;21(5):578-584. [DOI] [PubMed] [Google Scholar]
- 19.Skinner MW, Négrier C, Paz-Priel I, et al. The effect of emicizumab prophylaxis on long-term, self-reported physical health in persons with haemophilia A without factor VIII inhibitors in the HAVEN 3 and HAVEN 4 studies. Haemophilia. 2021;27(5):854-865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kiialainen A, Niggli M, Kempton CL, et al. Effect of emicizumab prophylaxis on bone and joint health markers in people with haemophilia A without factor VIII inhibitors in the HAVEN 3 study. Haemophilia. 2022;28(6):1033-1043. [DOI] [PMC free article] [PubMed] [Google Scholar]