Chronic graft-versus-host disease (cGvHD) is an important immunologic complication of allogeneic hematopoietic cell transplantation (HCT) that can affect multiple organs and lead to increased morbidity and mortality.1,2 Several adverse factors for survival in patients with cGvHD have been identified.3,4 Despite significant progress in the classifications of cGvHD, there is a need for improved and practical prognostication systems to address interpatient variability and achieve the goal of personalized treatment.5 In this study, we introduce red cell distribution width (RDW) as a new biomarker candidate that could enhance the development of personalized treatment strategies and risk stratifications for cGvHD.
Red cell distribution width is a measure of the variation of the mean corpuscular volume of red blood cells (RBC) and is routinely reported in most standard complete blood count results.6 Although RDW is affected to some degree by any type of anemia, the etiology of variation in RDW is considered to be multifactorial and is not completely understood.7 Multiple epidemiological and longitudinal observational studies have shown that higher RDW values are independently associated with increased all-cause, cardiovascular and cancer-related mortality.7- 9 Age, inflammation and the presence of clonal hematopoiesis are associated with increased RDW, but the association of RDW with mortality is independent of these factors.6-8,10
Red cell distribution width has not been previously evaluated in patients with cGvHD. The primary objective of this study was to determine if an RDW cut-off value is associated with overall survival (OS) in patients with moderate or severe cGvHD enrolled on an observational, cross-sectional study (NCT00092235) at the National Institutes of Health (NIH),11 and to estimate the 5-year OS probability in patients with RDW values above and below the specified cut-off. Secondary objectives were to determine 5-year relapse-free survival, cumulative incidence of relapse, non-relapse mortality (NRM), and significant associations of RDW with clinical variables and other biomarkers in patients with RDW values above and below the established cut-off. All variables were prespecified in an analysis plan. Selected biomarkers of interest were divided into quartiles and the association of the four groups with OS was determined to establish binary cut-offs. Probabilities of survival were determined by the Kaplan-Meier method, with log-rank tests used to determine the difference in survival between groups. All P values are two-tailed. Statistical analyses were performed using SAS version 9.4 and GraphPad Prism 9.3.
The characteristics of the 404 patients included in this study, who were enrolled from October 2004 to December 2018, are presented in Online Supplementary Table S1. Patients were enrolled at a median of three years after HCT and two years after diagnosis of cGvHD, and underwent a one-week comprehensive multidisciplinary evaluation including laboratory collection at this single time point.11 All patients provided written informed consent to participate in the study, which was approved by the National Cancer Institute Institutional Review Board. The trial was registered at ClinicalTrials.gov (NCT00092235).
The median follow-up of the total cohort, calculated by the reverse Kaplan-Meier method, was 12 years (interquartile range [IQR]: 8.4-14.5 years) but the median OS was not reached (estimated 15-year OS: 54.1% [95% Confidence Interval (CI): 47.7-60.1%]).
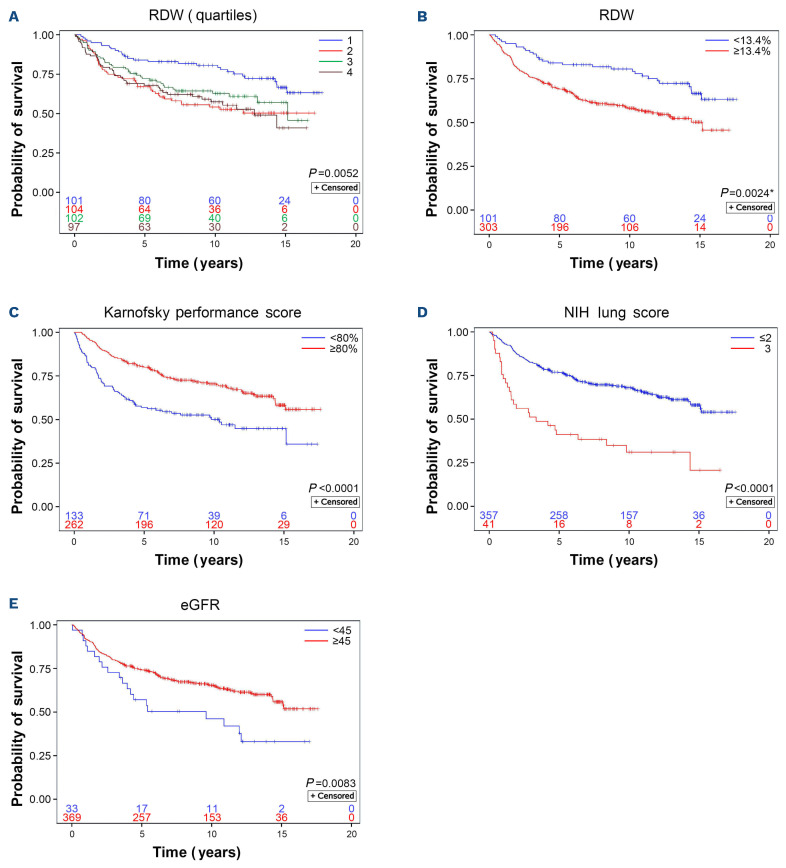
Median RDW of the total study cohort was 14.5% (IQR: 13.4-16.0%). In univariate Cox regression, RDW (%) displayed an association with survival as a continuous variable (Hazard Ratio [HR]=1.09; 95%CI: 1.03-1.14; P=0.004). However, we identified a clear split in survival curves between the first quartile (<13.4%) and the remaining overlapping quartiles of RDW (Figure 1) distinguishing groups with statistically different OS (P=0.0024, adjusted for 3 cut-points). The corresponding 5-year OS probabilities were 84.1% (95%CI: 75.3-89.9%) and 69.1% (95%CI: 63.6-74.0%) for patients with RDW <13.4% and ≥13.4%, respectively. Information on the cause of death after excluding those with non-malignant reasons for transplant was available in 286 patients (Online Supplementary Figure S1). Patients with RDW ≥13.4% had a higher 5-year NRM of 20.8% (95%CI: 15.3-26.7%) compared to 7.6% (95%CI: 2.8-15.7%) among patients with RDW <13.4% (P=0.0082 for the full curves). The absolute difference in survival between the two RDW groups was similar to Karnofsky performance status (KPS) <80% versus ≥80% and slightly lower than estimated glomerular filtration rate (eGFR) <45 versus ≥45 mL/min/1.73m2 but not comparable to NIH lung score 3 versus 0-2, the latter representing the most serious risk factor for mortality in our cohort (Figure 1).
Patients with an RDW value ≥13.4% were more likely to have received a peripheral blood stem cell graft (PBSC) but there were no between-group differences in age, sex, donor HLA-match or kidney function. Anemia, defined by a hemoglobin (Hb) level below 11 g/dL, was present in 21% (85/402) of patients, and RDW ≥13.4% was strongly associated with lower Hb and higher lactate dehydrogenase (LDH).
Figure 1.
Probability of overall survival according to red cell distribution width and other risk factors. The separation of red cell distribution width (RDW) values into quartiles (A) demonstrates a clear split in the overall survival (OS) probability between the first quartile (RDW <13.4%) and the remaining quartiles, which overlap each other (B) (see also Table 1). Other previously described risk factors of adverse survival such as Karnofsky performance score <80% (C), National Institutes of Health lung score 3 (D), and moderately to severely decreased kidney function (E) defined by estimated glomerular filtration rate (eGFR) <45 mL/min/1.73m2 (calculated according to Chronic Kidney Disease Epidemiology Collaboration 2021 equation) are presented for comparison of impact on OS. *P value adjusted to account for three implicit cut-points that could have been used when dividing the groups into quartiles.
In univariate analyses (Online Supplementary Table S1), RDW ≥13.4% was strongly associated with indicators of increased cGvHD severity and activity such as KPS <80%, cGvHD global score severe, increasing involvement of skin and mouth, higher number of prior lines of GvHD therapy, and higher number of concurrent GvHD treating agents at the time of enrollment. Patients with an RDW value ≥13.4% received steroids more frequently at enrollment than those with RDW <13.4% (70% vs. 47%; P<0.0001). In addition, patients with RDW ≥13.4% receiving steroids had a higher prednisone equivalent dose compared to those with RDW <13.4% (median mg/kg/day: 0.26 [IQR: 0.13-0.5] vs. 0.15 [IQR: 0.07-0.36]; P=0.008). Biomarkers of inflam-mation,12 including high sensitivity CRP, C3, C4, and erythrocyte sedimentation rate were elevated in patients with RDW ≥13.4%, but no differences were found in ferritin or interleukin 6 levels.
The multiple logistic regression established oral cGvHD (binary; Odds Ratio [OR]=1.77), mammalian target of rapamycin (mTOR) inhibitor use (binary; OR=3.49), LDH (continuous; OR=1.01), Hb (continuous; OR=0.68), and absolute neutrophil count (ANC; continuous; OR=1.21) as independent factors associated with RDW ≥13.4% (Online Supplementary Table S1).
In this cohort, there was no statistical difference in OS according to age, sex, stem cell source (bone marrow vs. PBSC; P=0.17) and NIH global score moderate versus severe (P=0.071). The analysis of laboratory biomarkers4,12 revealed notable associations with survival. While platelet count was not associated with OS, we observed important survival differences among patients who were divided into groups based on their albumin, CRP and immunoglobulin G (IgG) levels.
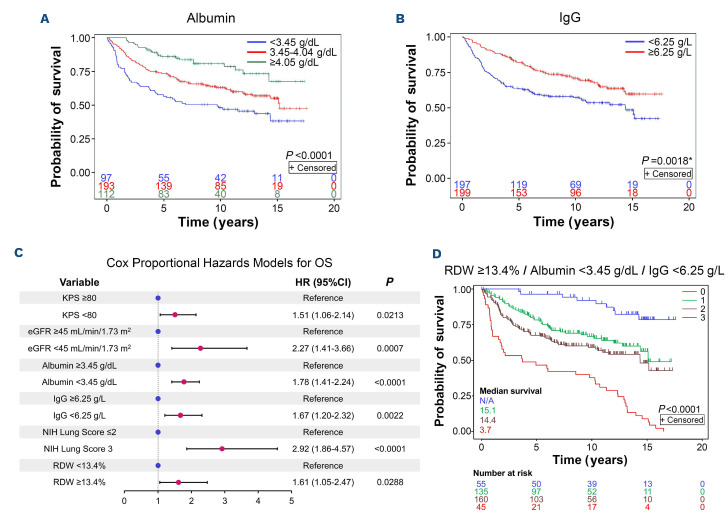
Albumin (g/dL) was strongly associated with longer OS as a continuous variable (HR: 0.42 [univariate Cox regression]; 95%CI: 0.31-0.58; P<0.001) and patients with an albumin level <3.45 g/dL had the poorest survival (Figure 2). Increasing CRP (mg/L) was linked to shorter OS (HR: 1.01; 95%CI: 1.00-1.02; P=0.009) and two groups divided at 7.6 mg/L could be distinguished (Online Supplementary Figure S2). IgG showed a distinctly binary cut-off at 6.25 g/L (Figure 2) and no correlation with OS as a continuous variable (HR: 0.97; 95%CI: 0.93-1.00; P=0.06). Absolute eosinophil count and absolute lymphocyte count displayed a weak association with OS but no significant threshold could be identified. C3 and C4 showed no association with OS.
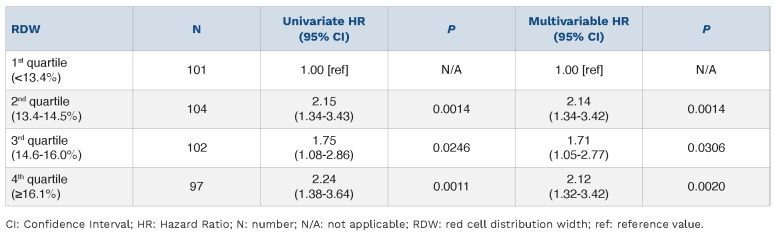
The Cox proportional hazards model confirmed that RDW ≥13.4%, albumin <3.45 mg/dL, IgG <6.25 g/L, KPS <80%, eGFR <45 mL/min/1.73m2 and lung cGvHD NIH score 3 were independent risks factors for increased mortality (Figure 2). Furthermore, there was no linear relationship between RDW quartiles and the risk of death, with similar HR observed for other quartiles (Table 1). Using only lab parameters from the Cox model, RDW ≥13.4% together with albumin <3.45 mg/dL and IgG <6.25 g/L allowed for estimation of different survival probabilities (Figure 2).
Figure 2.
Multivariable Cox model including other laboratory biomarkers. Inspection of the albumin quartiles revealed three distinct groups (A) with different overall survival (OS) probabilities whereas immunoglobulin G (IgG) (B) could be divided into two groups using a binary cut-off. Cox proportional hazards models for OS were built to evaluate the association of red cell distribution width (RDW) on OS, controlling for prespecified variables with P<0.10 by the log-rank test, and with the final model determined by backward selection. The final Cox model for OS (C) shows that RDW ≥13.4%, albumin <3.45 g/dL, and IgG <6.25 g/L are independent adverse risk factors for survival leading to different survival probabilities (D) depending on the number of any of these risk factors identified (i.e., 0, 1, 2, or 3 of these traits). *P value adjusted to account for three implicit cut-points that could have been used when dividing the groups into quartiles. KPS: Karnofsky performance score; eGFR: estimated glomerular filtration rate; NIH: National Institutes of Health.
Table 1.
Cox proportional hazards model for overall survival.
The association of RDW ≥13.4% with acute-phase proteins such as C-reactive protein (CRP) in the univariate analysis, and increased ANC in the multivariable regression analysis, suggests that inflammation may be a contributor to RDW elevation and the associated reduction in Hb. While hypoalbuminemia can also result from malnutrition and renal or hepatic disease, the drop in albumin concentration and association with worse OS is likely due to chronic inflammation from cGvHD, which was independent of moderate or severe renal function impairment. Albumin production by hepatocytes is known to be inhibited by proinflammatory cytokines13 and appears to be a more reliable marker of chronic systemic inflammation in cGvHD patients compared to CRP, which could be more rapidly influenced by transient infections.
The association of RDW ≥13.4% with mTOR inhibitor therapy cannot be explained by a direct side-effect of this class of drugs. Contrary to expectations, a placebo-controlled randomized trial involving 25 healthy older adults (age 70-95 years) observed a decrease in RDW during sirolimus treatment.14 This suggests that the association may be attributed to higher cGvHD activity, necessitating treatment with these agents.
The association of RDW ≥13.4% with elevated LDH in multiple regression analysis is unclear. LDH elevation at this stage after HCT is rarely explained by relapsing disease. Possible causes include inflammation, disturbed erythropoiesis, tissue damage from cGvHD, drugs, and subclinical transplant-associated thrombotic microangiopathy or hemolytic anemia.15 Patients in this population infrequently require transfusions but no systematic information regarding transfusion support was available.
The cross-sectional design of the study does not allow us to assess changes in the measured variables over time. Survivorship bias might have affected the biomarker cutoff values because patients with extreme values could have died before enrollment in this trial. The identified biomarker thresholds should be interpreted in the context of the severely affected cGvHD population included in this study. Further studies, ideally clinical trials, are required to replicate these observations and confirm the prognostic validity of the RDW. These findings, however, strongly support the conclusion that RDW values beyond the normal range are associated with an unfavorable outcome.
Supplementary Material
Acknowledgments
Part of this work was presented at the 64th Annual Meeting of the American Society of Hematology 2022.
Funding Statement
Funding: This work was supported by funding from the Intramural Research Program, National Institutes of Health, National Cancer Institute, Center for Cancer Research.
References
- 1.Holtzman NG, Pavletic SZ. The clinical landscape of chronic graft-versus-host disease management in 2021. Br J Haematol. 2022;196(4):830-848. [DOI] [PubMed] [Google Scholar]
- 2.Palmer J, Chai X, Pidala J, et al. Predictors of survival, nonrelapse mortality, and failure-free survival in patients treated for chronic graft-versus-host disease. Blood. 2016;127(1):160-166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Arora M, Klein JP, Weisdorf DJ, et al. Chronic GVHD risk score: a Center for International Blood and Marrow Transplant Research analysis. Blood. 2011;117(24):6714-6720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ayuk F, Veit R, Zabelina T, et al. Prognostic factors for survival of patients with newly diagnosed chronic GVHD according to NIH criteria. Ann Hematol. 2015;94(10):1727-1732. [DOI] [PubMed] [Google Scholar]
- 5.Buxbaum NP, Socie G, Hill GR, et al. Chronic GvHD NIH Consensus Project Biology Task Force: evolving path to personalized treatment of chronic GvHD. Blood Adv. 2023;7(17):4886-4902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hoffmann JJ, Nabbe KC, van den Broek NM. Effect of age and gender on reference intervals of red blood cell distribution width (RDW) and mean red cell volume (MCV). Clin Chem Lab Med. 2015;53(12):2015-2019. [DOI] [PubMed] [Google Scholar]
- 7.Horne BD, Muhlestein JB, Bennett ST, et al. Extreme erythrocyte macrocytic and microcytic percentages are highly predictive of morbidity and mortality. JCI Insight. 2018;3(14):e120183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Perlstein TS, Weuve J, Pfeffer MA, Beckman JA. Red blood cell distribution width and mortality risk in a community-based prospective cohort. Arch Intern Med. 2009;169(6):588-594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Riedl J, Posch F, Konigsbrugge O, et al. Red cell distribution width and other red blood cell parameters in patients with cancer: association with risk of venous thromboembolism and mortality. PLoS One. 2014;9(10):e111440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jaiswal S, Fontanillas P, Flannick J, et al. Age-related clonal hematopoiesis associated with adverse outcomes. N Engl J Med. 2014;371(26):2488-2498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Goklemez S, Im AP, Cao L, et al. Clinical characteristics and cytokine biomarkers in patients with chronic graft-vs-host disease persisting seven or more years after diagnosis. Am J Hematol. 2020;95(4):387-394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Grkovic L, Baird K, Steinberg SM, et al. Clinical laboratory markers of inflammation as determinants of chronic graft-versus-host disease activity and NIH global severity. Leukemia. 2012;26(4):633-643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mantovani A, Garlanda C. Humoral innate immunity and acute-phase proteins. N Engl J Med. 2023;388(5):439-452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kraig E, Linehan LA, Liang H, et al. A randomized control trial to establish the feasibility and safety of rapamycin treatment in an older human cohort: immunological, physical performance, and cognitive effects. Exp Gerontol. 2018;105:53-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Young JA, Pallas CR, Knovich MA. Transplant-associated thrombotic microangiopathy: theoretical considerations and a practical approach to an unrefined diagnosis. Bone Marrow Transplant. 2021;56(8):1805-1817. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.