Abstract
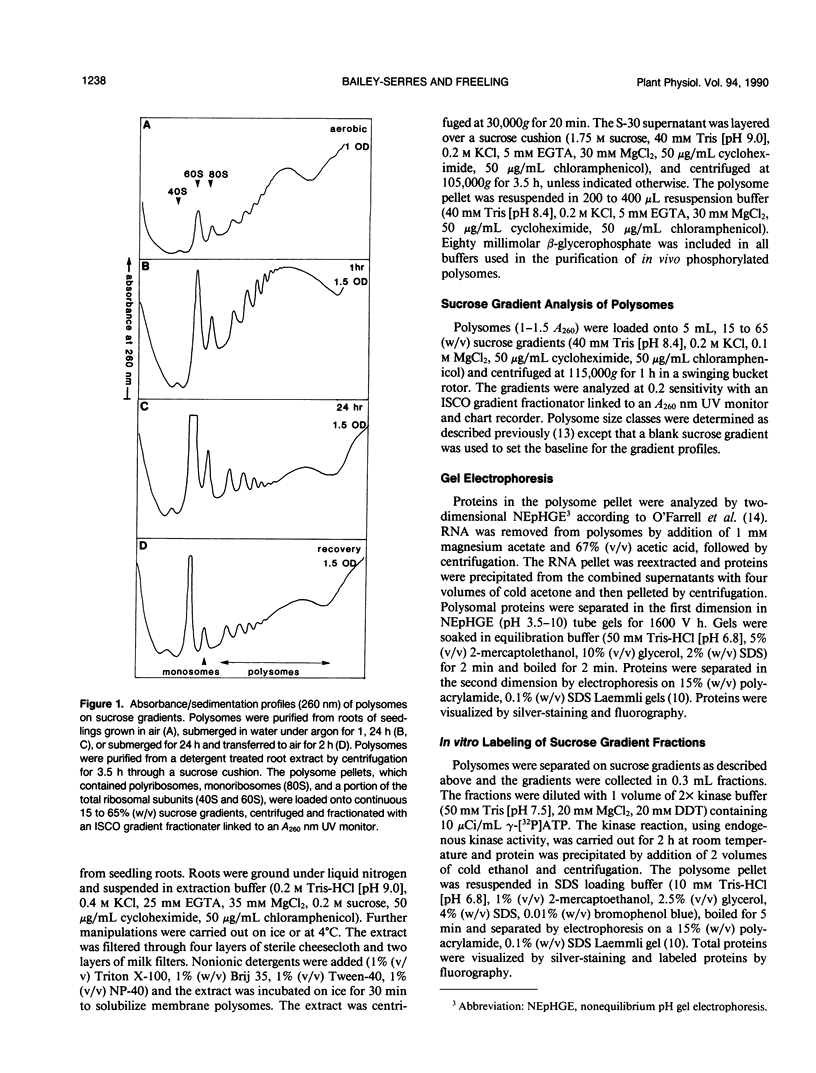
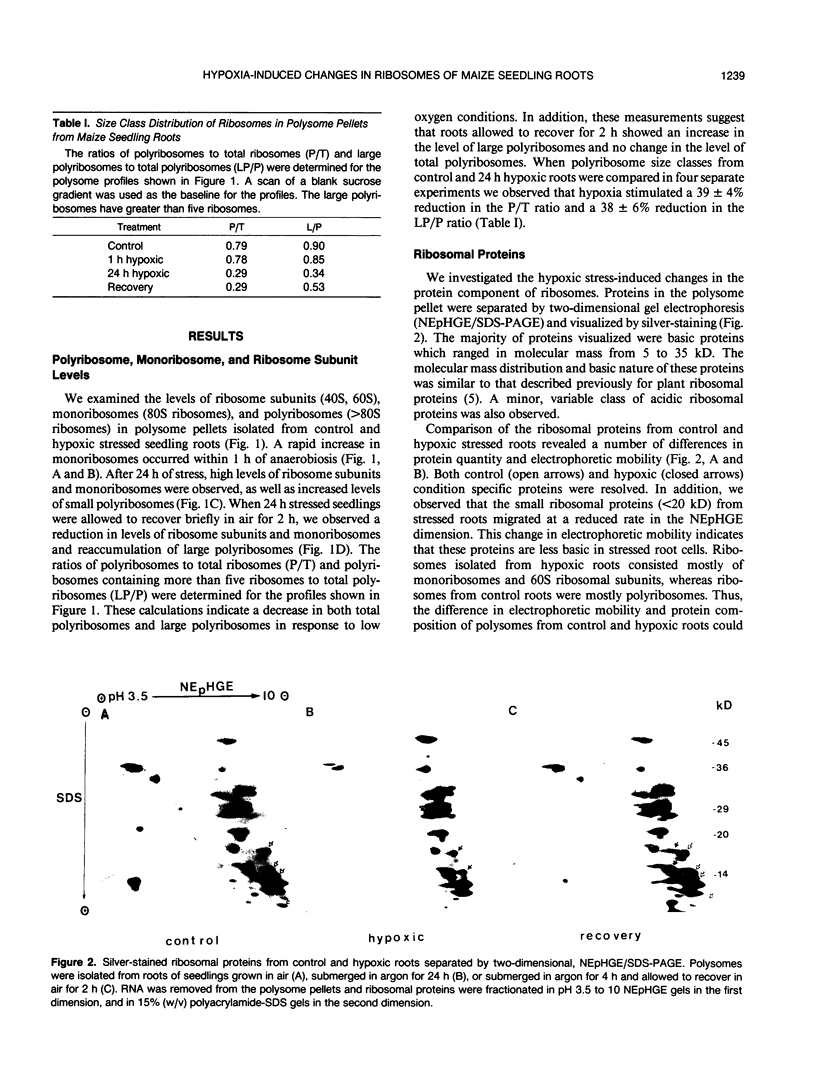
The hypoxic stress response of Zea mays L. seedling roots involves regulation of gene expression at transcriptional and posttranscriptional levels. We investigated the effect of hypoxia on the translational machinery of seedling roots. The levels of monoribosomes and ribosomal subunits increased dramatically within 1 hour of stress. Prolonged hypoxia resulted in continued accumulation of nontranslating ribosomes, as well as increased levels of small polyribosomes. The return of seedlings to normal aerobic conditions resulted in recovery of normal polyribosome levels. Comparison of ribosomal proteins from control and hypoxic roots revealed differences in quantity and electrophoretic mobility. In vivo labeling of roots with [35S]methionine revealed variations in newly synthesized ribosomal proteins. In vivo labeling of roots with [32P]orthophosphate revealed a major reduction in the phosphorylation of a 31 kilodalton ribosomal protein in hypoxic stressed roots. In vitro phosphorylation of ribosomal proteins by endogenous kinases was used to probe for differences in ribosome structure and composition. The patterns of in vitro kinased phosphoproteins of ribosomes from control and hypoxic roots were not identical. Variation in phosphoproteins of polyribosomes from control and hypoxic roots, as well as among polyribosomes from hypoxic roots were observed. These results indicate that modification of the translational machinery occurs in response to hypoxic stress.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ashburner M., Bonner J. J. The induction of gene activity in drosophilia by heat shock. Cell. 1979 Jun;17(2):241–254. doi: 10.1016/0092-8674(79)90150-8. [DOI] [PubMed] [Google Scholar]
- Browning K. S., Yan T. F., Lauer S. J., Aquino L. A., Tao M., Ravel J. M. Phosphorylation of wheat germ initiation factors and ribosomal proteins. Plant Physiol. 1985 Feb;77(2):370–373. doi: 10.1104/pp.77.2.370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glover C. V. Heat shock induces rapid dephosphorylation of a ribosomal protein in Drosophila. Proc Natl Acad Sci U S A. 1982 Mar;79(6):1781–1785. doi: 10.1073/pnas.79.6.1781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gualerzi C., Janda H. G., Passow H., Stöffler G. Studies on the protein moiety of plant ribosomes. Enumeration of the proteins of the ribosomal subunits and determination of the degree of evolutionary conservation by electrophoretic and immunochemical methods. J Biol Chem. 1974 Jun 10;249(11):3347–3355. [PubMed] [Google Scholar]
- Hake S., Kelley P. M., Taylor W. C., Freeling M. Coordinate induction of alcohol dehydrogenase 1, aldolase, and other anaerobic RNAs in maize. J Biol Chem. 1985 Apr 25;260(8):5050–5054. [PubMed] [Google Scholar]
- Kruppa J., Clemens M. J. Differential kinetics of changes in the state of phosphorylation of ribosomal protein S6 and in the rate of protein synthesis in MPC 11 cells during tonicity shifts. EMBO J. 1984 Jan;3(1):95–100. doi: 10.1002/j.1460-2075.1984.tb01767.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lin C. Y., Key J. L. Dissocation and reassembly of polyribosomes in relation to protein synthesis in the soybean root. J Mol Biol. 1967 Jun 14;26(2):237–247. doi: 10.1016/0022-2836(67)90294-x. [DOI] [PubMed] [Google Scholar]
- Mason H. S., Mullet J. E., Boyer J. S. Polysomes, Messenger RNA, and Growth in Soybean Stems during Development and Water Deficit. Plant Physiol. 1988 Mar;86(3):725–733. doi: 10.1104/pp.86.3.725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Farrell P. Z., Goodman H. M., O'Farrell P. H. High resolution two-dimensional electrophoresis of basic as well as acidic proteins. Cell. 1977 Dec;12(4):1133–1141. doi: 10.1016/0092-8674(77)90176-3. [DOI] [PubMed] [Google Scholar]
- Russell D. A., Sachs M. M. Differential expression and sequence analysis of the maize glyceraldehyde-3-phosphate dehydrogenase gene family. Plant Cell. 1989 Aug;1(8):793–803. doi: 10.1105/tpc.1.8.793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell D. A., Wong D. M., Sachs M. M. The anaerobic response of soybean. Plant Physiol. 1990 Feb;92(2):401–407. doi: 10.1104/pp.92.2.401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sachs M. M., Freeling M., Okimoto R. The anaerobic proteins of maize. Cell. 1980 Jul;20(3):761–767. doi: 10.1016/0092-8674(80)90322-0. [DOI] [PubMed] [Google Scholar]
- Scharf K. D., Nover L. Heat-shock-induced alterations of ribosomal protein phosphorylation in plant cell cultures. Cell. 1982 Sep;30(2):427–437. doi: 10.1016/0092-8674(82)90240-9. [DOI] [PubMed] [Google Scholar]
- Traugh J. A., Pendergast A. M. Regulation of protein synthesis by phosphorylation of ribosomal protein S6 and aminoacyl-tRNA synthetases. Prog Nucleic Acid Res Mol Biol. 1986;33:195–230. doi: 10.1016/s0079-6603(08)60024-0. [DOI] [PubMed] [Google Scholar]
- Walker J. C., Howard E. A., Dennis E. S., Peacock W. J. DNA sequences required for anaerobic expression of the maize alcohol dehydrogenase 1 gene. Proc Natl Acad Sci U S A. 1987 Oct;84(19):6624–6628. doi: 10.1073/pnas.84.19.6624. [DOI] [PMC free article] [PubMed] [Google Scholar]