Abstract
Background: We aimed to analyse the effectiveness, efficiency, and safety of initial treatment with biological therapies in rheumatoid arthritis (RA). Methods: Qualitative study. A group of RA experts was selected. A scoping review in Medline was conducted to analyse the evidence of initial RA treatment with biological therapies. Randomised clinical trials were selected. Two reviewers analysed the articles and compiled the data, whose quality was assessed using the Jadad scale. The experts discussed the review’s findings and generated a series of general principles: Results: Seventeen studies were included. Most of the included patients were middle-aged women with early RA (1–7 months) and multiple poor prognostic factors. Initial treatment with TNF-alpha inhibitors combined with methotrexate (MTX) and an IL6R inhibitor (either in mono or combination therapy) is effective (activity, function, radiographic damage, quality of life), safe, and superior to MTX monotherapy in the short and medium term. In the long term, patients who received initial treatment with biologicals presented better results than those whose initial therapy was with MTX. Conclusions: Initial treatment of RA with biological therapies is effective, efficient, and safe in the short, medium, and long term, particularly for patients with poor prognostic factors.
Keywords: rheumatoid arthritis, biologic therapy, initial treatment, narrative review, experts’ opinion
1. Introduction
Rheumatoid arthritis (RA) is a chronic inflammatory joint disease of autoimmune origin, with an estimated prevalence of around 1% [1]. The disease is associated with a major impact on the patient, their environment, and the healthcare system [2,3,4].
For many years, the pharmacological treatment of RA was essentially based on the use of corticosteroids and classical synthetic disease-modifying anti-rheumatic drugs (csDMARD) like methotrexate (MTX) [5]. However, the arrival of biologic DMARD (bDMARD) significantly changed treatment paradigms for RA patients [6].
Pivotal studies on bDMARD showed their efficacy and safety for patients with RA refractory to csDMARD [5,7], which led regulatory agencies to approve the use of these drugs for this group of RA patients. Subsequent publications have presented the results of post hoc analyses and studies specifically designed to analyse bDMARD as the initial treatment for RA (csDMARD-naïve patients) [8,9]. This is why the summaries of products for some bDMARD contemplate the drug’s use in patients with severe, active, progressive RA not previously treated with MTX or other csDMARD. However, the guidelines of national and international scientific societies still recommend using csDMARD, particularly MTX, as the initial treatment for RA [6,10]. These guidelines highlight the high cost of many bDMARDs as a limitation to their wider use [11,12].
On the other hand, the approval of biosimilar medicines has ushered in more competitive pricing, bringing a considerable reduction in bDMARD costs [13]. A report by BioSim (the Spanish Association of Biosimilar Medicines) estimates savings derived from the introduction of biosimilars for the 2009–2019 period to be 2306 million euros in Spain [14]. It also highlights adalimumab (ADA) as the drug that generates the most savings [14]. There are currently over 60 biosimilar medicines corresponding to 17 drugs, 4 of which are for immune-mediated diseases: infliximab (IFX), ADA, etanercept (ETN), tocilizumab (recently approved by the European Medicines Agency), and rituximab, which are widely used [15]. The guidelines of scientific societies acknowledge that biosimilar medicines have contributed to a substantial reduction in the cost of medicines, following a policy of rational prescribing based on the principle that if two medicines are equally effective and safe for a specific patient, the least expensive should be used [6].
In light of this, we can consider the use of bDMARD as the initial treatment for RA, at least for some subgroups of patients. The design of this project aimed to address this topic, with the objectives of analysing the existing evidence on initial treatment with bDMARD and issuing a series of positions for better stratification of RA patients. Bearing in mind that many countries recommend biosimilar TNF-alpha inhibitors as the first choice for bDMARD and, among patients with intolerance to/contraindications for MTX, an interleukin 6 receptor (IL6R) inhibitor, this document focuses on these pharmacological groups. Abatacept and rituximab are currently reserved for special situations, and in clinical practice, Janus kinase inhibitors are used in later lines of treatment.
We believe that this document will help rheumatologists in their therapeutic decision-making for RA patients.
2. Materials and Methods
This qualitative study is based on a scoping review of the literature and on expert opinion. The project was conducted in full compliance with the principles established in the Declaration of Helsinki for medical research involving human subjects in its latest version [16] and in accordance with applicable regulations on Good Clinical Practice.
Participant selection and the first nominal group meeting. A group of three rheumatologists with extensive experience and knowledge of RA management were selected. In a first meeting, they analysed the current status of RA management and defined the objectives, scope, and literature review to be undertaken.
Scoping review of the literature. The objective was to assess the efficacy, effectiveness, and safety of the initial treatment of RA with bDMARD.
With the help of an expert documentalist, different search strategies were designed for Medline (up to January 2023) that combined both MeSH and free-text terms. Examples of terms used are “rheumatoid arthritis” or “disease-modifying anti-rheumatic drugs”. Searches were also made using PubMed’s Clinical Queries tool (see Supplementary Table S1).
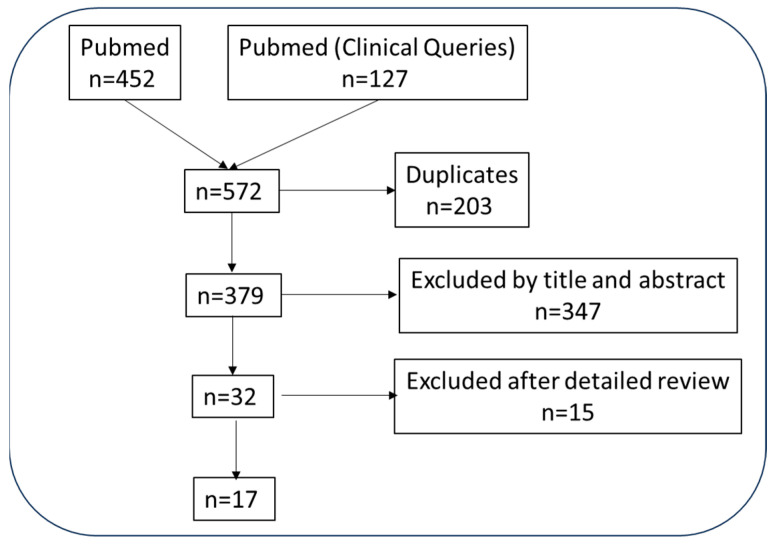
Studies that met the following inclusion and exclusion criteria were selected: The population included patients with RA (according to international criteria [17]), adults (>18 years), regardless of disease duration or severity (P); RA patients receiving initial treatment with bDMARD, i.e., naïve for any csDMARD such as oral (po), subcutaneous (sc), or other MTX. No restrictions were imposed regarding dose, treatment duration, use as monotherapy or in combination (I), a control, or either a placebo or with an active ingredient (C). We also included studies that analysed any variable related with efficacy/effectiveness, such as RA activity including DAS28 (disease activity score), CDAI (clinical disease activity index), SDAI (simplified disease activity index), disease remission, function assessed with the HAQ (Health Assessment Questionnaire), structural damage viewed in a simple radiography or magnetic resonance imaging (MRI), PRO (patient-reported outcomes), acute phase reactants such as erythrocyte sedimentation rate (ESR) or C-reactive protein (CRP), quality of life assessed with RAQoL (Rheumatoid Arthritis Quality of Life) Questionnaire or others, cost-effectiveness and safety variables (serious adverse events, infection rate, etc.). The types of studies accepted were randomised clinical trials (RCT) with their corresponding extension studies and post hoc analyses. Figure 1 shows the study flow chart.
Figure 1.
Studies flow chart.
All of the citations found using the different search strategies were downloaded to the EndNote® reference management software package (Version number 20). Two reviewers (EL, TO) independently analysed the citations in duplicate. The search results were first refined by title and abstract, with another purge after a detailed reading of the resulting citations. The two reviewers compiled data from the included studies. To assess the methodological quality of the studies included, a Jadad scale was used (from 1 to 5, with “quality” defined as an RCT with a score of ≥3). A descriptive-qualitative analysis was performed. Meta-analysis was only performed in cases of homogeneity.
Finally, as part of the secondary search, the bibliographic references of the included articles and the abstracts of international conferences were reviewed.
Second meeting of the nominal group. The nominal group discussed the review results at the second meeting, during which the experts reached a consensus on a series of points related to the use of bDMARD as an initial treatment for RA.
Preparation of the final document. The final text took into account both the narrative review and the nominal group’s decisions. For each of the expert points, we assessed the strength of the recommendation (very low, low, moderate, or high). The resulting document was given to the experts for their final evaluation and comments.
3. Results
A total of 17 articles [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34] were included, whereas 15 were excluded (see Supplementary Material, Table S2) [8,9,35,36,37,38,39,40,41,42,43,44,45,46,47]. A summary of the main outcomes is depicted in Table 1, while the supplementary material provides the evidence tables (Tables S3 and S4).
Table 1.
Main conclusions of the included studies [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34]. The study’s primary outcomes are highlighted.
| 1 | The use of initial treatment with IFX + MTX is significantly superior to MTX (in the short and long term) to MTX regarding RA activity (DAS28, DAS28 remission, imaging techniques, etc.), HAQ, radiographic progression, and quality of life (RAQoL) |
| 2 | Initial treatment with IFX + MTX is superior to other strategies: sequential csDMARD monotherapy and stepped combination treatment with csDMARDs regarding the previously mentioned variables |
| 3 | Initial use of ADA + MTX during the first year is significantly superior (in the short and long term) to MTX regarding RA activity (DAS28, DAS28/CDAI/SDAI/ACR-EULAR remission, low disease activity, joint counts, etc.). HAQ, quality of life (SF-36-physical component), and probably regarding radiographic progression |
| 4 | Current evidence on initial treatment with ETN + MTX is based on a single study and is inconclusive |
| 5 | Initial use of CZP + MTX is significantly superior (in the short and long term) to MTX regarding RA activity (DAS28, DAS28 remission, CDAI, CDAI remission, SDAI, acute phase reactants, etc.), HAQ, and radiographic progression. However, the CZP + MTX combination has not been shown to be superior to the combination of MTX with prednisone or to the combination of SSZ + HCQ + intra-articular corticosteroids |
| 6 | Initial use of ABT + MTX achieves remission rates of 52% at 24 weeks, higher than those obtained with the combination of MTX + corticosteroids or SSZ + HCQ+ intra-articular corticosteroids |
| 7 | Initial use of TCZ + MTX or TCZ in monotherapy is significantly superior (in the short and long term) to MTX regarding RA activity (DAS28, DAS28 remission, CDAI, CDAI remission, EULAR and ACR responses, etc.), HAQ, radiographic progression, and quality of life |
Abbreviations: IFX = infliximab; ADA = adalimumab; ETN = etanercept; CZP = certolizumab pegol; TCZ = tocilizumab; SSZ = sulphasalazine; HCQ = hydroxychloroquine; MTX = methotrexate; csDMARDs = classical synthetic disease-modifying anti-rheumatic drugs; RA = rheumatoid arthritis; SF-36 = 36-item short-form health survey; ACR = American College of Rheumatology; DAS28 = disease activity score of 28 joints; CEDAI = clinical disease activity index; SDAI = simplified disease activity index; HAQ = health assessment questionnaire; EULAR = European Alliance of Associations for Rheumatology; RAQoL = rheumatoid arthritis quality of life scale.
Here we present the expert opinions along with the literature search results.
General indications on initial treatment with bDMARD.
Based on the body of evidence and the current context of daily practice, it is possible to consider the use of bDMARD as an initial treatment for RA. Strengths of the recommendation: Moderate
With regard to TNF-alpha inhibitors, one RCT found that the combination IFX + MTX po was significantly superior to MTX po in improving synovitis and bone marrow oedema (measured by MRI), both at weeks 18 and 52 [19]. The combination was also superior in ACR20/50/70 responses at week 22, with a similar (non-significant) trend observed at week 52 [19].
Data from the BeSt study show that the triple therapy of csDMARD with prednisone and the combination IFX + MTX (po and sc) improved HAQ faster (3rd month) than the other treatment groups analysed, an improvement that was sustained until month 12 (p < 0.05). Moreover, these strategies were also significantly superior in improving structural damage (<0.001), although this effect disappeared in the second year [20,21,22]. Long-term (10-year) follow-up showed that the different combination therapies produced similar global outcomes (non-significant differences), although patients treated with IFX + MTX showed less radiographic progression and better physical functioning than the other strategies [23].
In the IDEA RCT, the combination of initial therapy with IFX + MTX po or MTX + intravenous methylprednisolone achieved similar DAS28 remission rates at week 78: 50% vs. 48% (p = 0.795); however, DAS28 remission was reached more quickly with IFX + MTX. Although both groups obtained a high proportion of patients without radiographic progression, no differences were detected [24].
The HIT-HARD [26], GUEPAR [27], and OPERA [28] RCTs evaluated ADA as the initial treatment for RA. The outcomes of the HIT HARD study at week 24 showed that ADA + MTX sc was significantly superior to MTX sc in terms of DAS28, HAQ, and DAS28 remission (47.9% vs. 29.5%; p = 0.021), as well as in ACR 50 and ACR 70 responses [26]. From week 24, all patients received MTX sc monotherapy. Radiographic progression at week 48 was significantly greater in patients who had received MTX sc monotherapy as the initial treatment [26]. The OPERA study compared ADA po + MTX vs. MTX po [28]. At one year of treatment, the combination ADA + MTX was significantly superior to MTX in terms of DAS28-CRP, CDAI, SDAI, HAQ, ACR/EULAR28 and ACR/EULAR40, ACR50 and ACR70 responses, and in remission, defined with DAS28, CDAI, SDAI, and ACR/EULAR. The SF-12 questionnaire revealed a significant improvement in quality of life for physical but not mental health, which was echoed in the EQ-5D (p = 0.015) [28]. Finally, the GUEPAR study reported that initial treatment with ADA + MTX po was significantly superior at 3 months to MTX po in improving rigidity, ACR 20/50/70 response, good EULAR response (63% vs. 25%), and DAS28 remission (36% vs. 12%). No differences existed between the groups for pain, fatigue, or ESR [27].
For ETN, the EMPIRE study [29] included 110 patients with early-onset synovitis (41% met RA criteria according to 1987 ACR criteria and 94% according to 2010 ACR-EULAR criteria). At week 52 of treatment, 32.5% of patients with ETN + MTX po did not present painful or swollen joints vs. 28.1% with MTX po (no statistically significant differences). Nor were any differences found in ACR remission, SDAI remission, DAS44-CRP, HAQ-DI, SF-36, EQ5D-3L, radiographic progression, or in the variables assessed at week 72 [29].
There are 3 published good-quality RCTs on the initial treatment of RA with certolizumab pegol (CZP) [30,31,32]. The outcomes of C-EARLY show that the combination CZP + MTX po is significantly superior to MTX po in controlling disease activity, sustaining clinical response, functional improvement, and inhibiting radiographic progression. In week 52, 28.9% of patients in the CZP + MTX group reached DAS28 remission: 28.9% vs. 15% for MTX (p < 0.001) [30]. The study also revealed significant differences in favour of CZP + MTX in sustained low disease activity (DAS28 ≤ 3.2), ACR 50 response, functioning, inhibition of radiographic progression, and both CDAI and SDAI remission. The study went on to assess whether, for patients who had achieved sustained low disease activity after 1 year of treatment with standard doses of CZP (200 mg/2 weeks) + MTX po, continuing with a standard or optimised dose of CZP (200 mg/4 weeks) was superior to interrupting CZP (and continuing with MTX po) for an additional period of 1 year (a total of 104 weeks). At week 104, the proportion of patients with sustained low disease activity was higher in the CZP-treated group, both at standard and optimised doses (48.8% and 53.2%) than in patients who continued with MTX (39.2%), although the differences were only significant for the comparison of optimised dose vs. MTX (p = 0.041). The trend was similar for inhibition of radiographic progression and physical functioning [32].
There are 3 articles available for TCZ, based on 2 studies. One good quality study, U-Act-Early [33,34], and another moderate quality study [31]. TCZ as monotherapy or in combination with MTX po were superior to MTX po in different outcome measures. At week 24, almost double the number of patients receiving TCZ achieved DAS28 and CDAI remission compared with MTX (86% and 83% with TCZ + MTX and TCZ, respectively, vs. 44% with MTX, p < 0.0001). The therapies with TCZ were also significantly superior to MTX in terms of good EULAR response (89% vs. 87% vs. 49%), ACR (20/50/70/90 responses), and HAQ. In week 104, little progression was observed in all groups, but it was significantly lower in both TCZ arms than in the MTX po arm [34]. Patients in this study underwent an additional follow-up of 3 years in real life. The data for effectiveness were sustained, with no differences between the groups. During 5 years of follow-up, the accumulated time of sustained remission was significantly higher in the groups that began therapy with TCZ (median 216 and 190 days for the TCZ + MTX combination and TCZ, respectively) than for MTX (median 172). However, no differences were observed in radiographic progression between the groups [33].
-
2.
Initial treatment with bDMARD should be considered, particularly for RA patients with a high inflammatory load and/or other poor prognostic factors. Strengths of the recommendation: High
Although RA is a heterogeneous, dynamic disease, different studies have shown that the baseline presence of certain factors is associated with a worse prognosis in terms of activity (e.g., remission) and radiographic damage [48]. Among these factors, we highlight a positive rheumatoid factor (RF) and/or anti–citrullinated protein antibodies, high counts of acute phase reactants, high disease activity (multiple inflamed joints, high activity index, etc.), significant functional limitation, or the presence of erosion in imaging tests [48].
Most patients included in the RCT analysed presented poor prognostic factors at baseline (see Table 2). The mean age of patients included was between 45 and 55 years, RF positivity was occasionally even higher than 90%, the mean DAS28 in most cases was higher than 5, and many patients presented structural damage in radiographic imaging [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34].
Table 2.
Basal characteristics of RA patients in the included studies [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34].
| Age (Years) | Women | RA Duration | RF+ | ACPA+ | RA Activity | Structural Damage | |
|---|---|---|---|---|---|---|---|
| IFX | 45–55 | 60–71% | 5–7.4 months | 61–67% | Hasta 90% | DAS28 > 5 | 70% |
| ADA | 46–56 | 63–79% | 3–4.4 months | 69–74% | - | DAS28 5.5–6.3 | SHS 6.3–7.5 |
| ETN | 48 | 76% | 7 months | 53% | 77% | DAS28-CRP 4.17 | mTTS 6.69–8.01 |
| CZP | 50–52 | 69–76% | 3–7 months | 72–97% | - | DAS28 6.7 | 77.3% |
| TCZ | 53–55 | 61–76% | 1 month | 75% | - | DAS28 5.2 | SHS 0 |
Abbreviations: RA = rheumatoid arthritis; RF = rheumatoid factor; ACPA = anti-citrullinated peptide antibodies; IFX = infliximab; ADA = adalimumab; ETN = etanercept; CZP = certolizumab pegol; TCZ = tocilizumab; DAS28 = disease activity score of 28 joints; CRP = C reactive protein; SHS = Sharp-van der Heijde index; mTTS = modified Sharp-van der Heijde index.
On the other hand, early, aggressive treatment has been shown to improve the long-term prognosis of this disease [49]. This therapy is associated with higher clinical response rates, lower disability, and less structural damage [50,51]. Although a larger body of specific evidence is necessary, in the BeSt study’s 10-year follow-up, patients who received IFX + MTX showed less radiographic progression and better physical functioning than patients following other strategies [23]. The data at 5 years for TCZ are similar [33], as are the 10-year extension data for ADA of the PREMIER study (almost 30% of whose patients had previously received csDMARD) [52]. However, it is important to consider each case individually. The decision to use bDMARD as initial treatment should be discussed with the patient, evaluating the potential risks and benefits of these drugs on a case-by-case basis.
-
3.
If considering a TNF-alpha inhibitor as an initial treatment for RA, its use is recommended in combination with MTX, whereas if considering an IL6R inhibitor, monotherapy is possible. Strength of the recommendation: Moderate
Most of the RCTs analysed included patients with MTX po either combined with a TNF-alpha inhibitor or as monotherapy [18,19,20,21,22,23,24,25,27,28,29,30,31,32]. The studies used different doses and regimens of MTX po, but the majority began with a dose of 7.5–10 mg/week, progressively increasing each 2–4 weeks to a maximum of 30 mg/week; however, most studies used a maximum dose of MTX between 15 and 20 mg/week.
As we have described, the use of MTX in combination with a TNF-alpha inhibitor is an effective and safe initial treatment for RA [18,19,20,21,22,23,24,25,27,28,29,30,31,32]. Although we have not found comparative evidence between TNF-alpha inhibitors for initial treatment as monotherapy and in combination with MTX, based on publications referring to patients with refractory RA, experts recommend the use of TNF-alpha inhibitors with MTX as the initial therapy [7]. In this context, as well as being more effective, MTX can prevent immunogenicity or reduce the use of corticosteroids [53]. On the other hand, MTX po might be recommended because it is the drug for which most evidence is available due to its lower price (cost-effectiveness). However, it is important to remember that the bioavailability of parenteral MTX is higher than that of MTX po, particularly at doses of ≥15 mg/week [54,55], and that in patients with an inadequate response to orally administered MTX (15 mg/week), dose scaling using parenteral MTX is more clinically effective [55,56]. Likewise, we should always consider patient opinion and preference when making therapeutic decisions. So, in some cases, we may consider the use of parenteral MTX with TNF-alpha inhibitors as the initial treatment.
On the other hand, there are considerable variations in the use of MTX in clinical practice, including the initial dose, dose scaling, maximum dose, etc. [57,58]. A sub-analysis of the AR Excellence study [59] highlighted these variations, finding that MTX scaling to its full dose did not occur as quickly as it should and that there was not correct use of parenteral MTX administration.
The outcomes reported for the use of IL6R as initial treatment [33,34] are similar to those reported for patients with RA refractory to csDMARD, so its use can be considered a monotherapy.
-
4.
Initial treatment of RA with bDMARD in monotherapy or in combination therapy is safe. Strength of the recommendation: Moderate
Data on the safety of bDMARD as an initial treatment are similar to those reported in studies on RA refractory to csDMARD [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34]. It should be noted that the data analysed is for RCTs with different follow-up periods and even changes in treatment within the same study.
Infections are still the most frequent adverse event with the use of bDMARD as the initial treatment for RA, although the percentage of severe infections is generally low and similar to existing reports [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34]. No new signs related to the active infection by hepatitis or tuberculosis viruses were detected. Haematological adverse events are not very frequent, and when they do occur, they are mild and reversible. An increase in liver enzymes is a relatively frequent adverse event, but in most cases, it is mild [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34]. There are exceptional reports of severe cases but no clearly established causal relationship. Based on the data from the included studies, the use of bDMARD as an initial treatment cannot be associated with the development of any type of cancer [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34]. A larger number of specific, long-term studies on patients in real life are necessary to analyse this issue in greater depth.
On the other hand, the global percentage of adverse events of combinations of bDMARD with MTX in some studies is superior to that of MTX monotherapy, specifically for certain types of adverse events such as infections [33], but combination has not been associated with a higher risk of serious adverse events of any type.
-
5.
Initial treatment of RA with biosimilars of TNF-alpha inhibitor biological drugs is cost-effective and this specific variable should be included in treatment stratification. Strength of the recommendation: Moderate
Clinical variables, along with patient opinion and preferences, are essential elements in RA therapy decision-making. However, we must not forget the current healthcare context and its sustainability. This is why the experts consider that the cost-effectiveness of bDMARD is another important variable to take into account.
Different reports at international conferences have informed us about studies into the cost-effectiveness of biological therapy treatment in Spain [60,61,62]. As no effectiveness difference was seen, a cost minimization analysis was performed that showed that currently the most cost-effective option for immune-mediated diseases such as Crohn’s or RA is ADA [60,61]. Specifically in the case of RA, it represents the cheapest biological therapy, with an estimated annual cost of 4650 euros vs. the 4650–10,000 euros for other treatments [60].
In line with these data, a study on cost-effectiveness in Norway [63], where the healthcare system promotes the use of biosimilars to reduce costs, found that since the introduction of biosimilars, the number of RA patients receiving treatment with bDMARD and small molecule inhibitors has increased from 39% in 2010 to 45% in 2019. The proportion of patients who reached DAS28 also increased from 42% to 67%. However, the annual cost of treating a patient with these therapies decreased by 47% [63].
Another pharmacoeconomic study from the perspective of the Italian national health system found that for psoriasis patients, there is a high similarity in cost per responder with two ADA biosimilars (MSB1102 and ABP 501) compared with MTX sc, with cost-effectiveness closest at 52 weeks between MSB1102 (799 euros) and MTX sc (625 euros) [64].
Finally, a cost-effectiveness study on the efficiency of early treatment with biologicals [65] found that for RA patients refractory to csDMARD, adding a biosimilar TNF-alpha inhibitor to MTX at 6 months increased the total treatment cost by only £70, compared to continuing MTX monotherapy and waiting until 12 months (price in pounds sterling for 2017) [65].
4. Discussion
This project shows that the initial RA treatments of TNF-alpha inhibitors combined with MTX and of IL6R (as monotherapy or in combination) are effective, efficient, and safe, with data for effectiveness that in some cases surpasses csDMARD results [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34].
For several reasons, including effectiveness, safety, cost-effectiveness, type of healthcare system, etc., the current strategy consists of starting treatment with csDMARD, habitually as a monotherapy. This review highlights that, in many cases, patients who began an initial treatment with bDMARD presented better outcomes (considering activity, physical functioning, PROs, quality of life, and image) than those who had done so with csDMARD [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34]. This suggests that the option should be considered in clinical practice.
It should be noted that most of the studies we excluded had a significantly high percentage (between 20 and 30%) of patients included who had previously used csDMARD [37,38,39,40]. This population may have characteristics and/or responses that are different from those of csDMARD-naïve patients. The patients included in this review were experiencing early-stage RA and had more than one variable for poor prognosis, and it is particularly for this subset of patients that initial treatment with bDMARD should be considered [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34] or at the very least they should follow the current T2T strategy, changing stage in a short period of time (a maximum of 3 months) to more effective therapeutic options. To the best of our knowledge, there have been no similar initiatives in this field, and we consider that our results may be of interest in clinical practise to manage the initial treatment of RA patients.
It is striking that national and international consensus documents do not consider the use of bDMARD as an initial treatment for RA, citing various reasons, the foremost of which is cost [10]. Bearing in mind the current context, in which the arrival of biosimilars has brought a considerable reduction in the net cost of bDMARD [13], this position should be reconsidered. But taking into account the risks with the use of bDMARDs, we consider it very important to individualise the use of bDMARD as an initial treatment, trying to select patients that might benefit most. In our opinion, this treatment strategy might be particularly effective for RA patients with poor prognostic factors.
The evidence for initial treatment with ETN deserves particular comment, although we can only draw conclusions based on one study, EMPIRE [29], which limits the robustness of our conclusions. The study did not find differences between the combination ETN + MTX and MTX monotherapy that were as consistent as those found in studies of other biologicals. There are different factors that could explain these results. The study included 110 patients, which is probably a small sample size (the calculation of the sample size was based on radiographic criteria, not on RA activity, as in other RCTs). The patients presented early-onset synovitis (some did not meet RA criteria), with data on activity and other severity factors that were lower than the other studies included. This RCT also allowed the use of MTX at comparatively higher doses than in other RCTs. All of these factors could cause the study not to have reached statistical significance in many of the outcome variables.
Another point to note is that recommendations include the use of MTX po (vs. MTX sc) in combination with bDMARD, both because more evidence is available and due to cost. However, the option of MTX sc should be evaluated case by case since its bioavailability is greater and the patient may prefer this route of administration [54,55].
Among the limitations of our work, we note that some of the studies included are post-hoc analyses or exploratory studies, so their outcomes should be interpreted with caution. Likewise, the inclusion of RCT means that the population consists of patients who are not 100% representative of the general RA population seen in conventional clinical practice. Finally, the lack of robust comparative data between combination therapy and monotherapy of biologicals could weaken our findings.
5. Conclusions and Future Directions
Initial treatment with bDMARD in RA patients is effective, has an acceptable safety profile, and, in our current context, should be considered, particularly for patients with poor prognostic factors. The final decision on RA treatment should be made on a case-by-case basis and in consensus with patients.
On the other hand, more research is needed in order to expedite and optimise treatment stratification by using advanced integrative modelling of complex health data (genetics, biomarkers, clinical data, etc.).
Acknowledgments
We would like to thank Estíbaliz Loza for her help with the methodological tasks, review of this study, and writing of this manuscript.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/jcm13010048/s1. Table S1: Excluded studies and reasons for exclusion; Table S2: Evidence table. Main characteristics of included studies; Table S3: Main results of included studies [66]. Table S4: Main results of included studies.
Author Contributions
Conceptualization, J.T.M., B.H.-C. and H.C.; Formal analysis, J.T.M., B.H.-C. and H.C.; Investigation, J.T.M., B.H.-C. and H.C.; Methodology, J.T.M.; Project administration, J.T.M.; Supervision, J.T.M.; Visualization, J.T.M.; Writing—original draft, J.T.M.; Writing—review and editing, J.T.M., B.H.-C. and H.C. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors received honoraria from Fresenius Kabi Spain for participating in this project.
Funding Statement
This project was funded by an unrestricted grant from Fresenius Kabi, Spain.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Almutairi K.B., Nossent J.C., Preen D.B., Keen H.I., Inderjeeth C.A. The Prevalence of Rheumatoid Arthritis: A Systematic Review of Population-based Studies. J. Rheumatol. 2021;48:669–676. doi: 10.3899/jrheum.200367. [DOI] [PubMed] [Google Scholar]
- 2.Alcaide L., Torralba A.I., Eusamio Serre J., García Cotarelo C., Loza E., Sivera F. Current state, control, impact and management of rheumatoid arthritis according to patient: AR 2020 national survey. Reumatol. Clin. (Engl. Ed.) 2022;18:177–183. doi: 10.1016/j.reuma.2020.10.006. [DOI] [PubMed] [Google Scholar]
- 3.Gil-Conesa M., Del-Moral-Luque J.A., Gil-Prieto R., Gil-de-Miguel Á., Mazzuccheli-Esteban R., Rodríguez-Caravaca G. Hospitalization burden and comorbidities of patients with rheumatoid arthritis in Spain during the period 2002–2017. BMC Health Serv. Res. 2020;20:374. doi: 10.1186/s12913-020-05243-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Leon L., Abasolo L., Fernandez-Gutierrez B., Jover J.A., Hernandez-Garcia C. Direct medical costs and their predictors in the EMAR-II cohort: “Variability in the management of rheumatoid arthritis and spondyloarthritis in Spain”. Reumatol. Clin. (Engl. Ed.) 2018;14:4–8. doi: 10.1016/j.reuma.2016.09.006. [DOI] [PubMed] [Google Scholar]
- 5.Kerschbaumer A., Sepriano A., Smolen J.S., van der Heijde D., Dougados M., van Vollenhoven R., McInnes I.B., Bijlsma J.W.J., Burmester G.R., de Wit M., et al. Efficacy of pharmacological treatment in rheumatoid arthritis: A systematic literature research informing the 2019 update of the EULAR recommendations for management of rheumatoid arthritis. Ann. Rheum. Dis. 2020;79:744–759. doi: 10.1136/annrheumdis-2019-216656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Smolen J.S., Landewé R.B.M., Bergstra S.A., Kerschbaumer A., Sepriano A., Aletaha D., Caporali R., Edwards C.J., Hyrich K.L., Pope J.E., et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2022 update. Ann. Rheum. Dis. 2023;82:3–18. doi: 10.1136/ard-2022-223356. [DOI] [PubMed] [Google Scholar]
- 7.Singh J.A., Hossain A., Tanjong Ghogomu E., Kotb A., Christensen R., Mudano A.S., Maxwell L.J., Shah N.P., Tugwell P., Wells G.A. Biologics or tofacitinib for rheumatoid arthritis in incomplete responders to methotrexate or other traditional disease-modifying anti-rheumatic drugs: A systematic review and network meta-analysis. Cochrane Database Syst. Rev. 2016;2016:Cd012183. doi: 10.1002/14651858.CD012183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Singh J.A., Hossain A., Mudano A.S., Tanjong Ghogomu E., Suarez-Almazor M.E., Buchbinder R., Maxwell L.J., Tugwell P., Wells G.A. Biologics or tofacitinib for people with rheumatoid arthritis naive to methotrexate: A systematic review and network meta-analysis. Cochrane Database Syst. Rev. 2017;5:Cd012657. doi: 10.1002/14651858.CD012657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Simpson E.L., Ren S., Hock E.S., Stevens J.W., Binard A., Pers Y.M., Archer R., Paisley S., Stevenson M.D., Herpin C., et al. Rheumatoid arthritis treated with 6-months of first-line biologic or biosimilar therapy: An updated systematic review and network meta-analysis. Int. J. Technol. Assess. Health Care. 2019;35:36–44. doi: 10.1017/S0266462318003628. [DOI] [PubMed] [Google Scholar]
- 10.Fraenkel L., Bathon J.M., England B.R., St Clair E.W., Arayssi T., Carandang K., Deane K.D., Genovese M., Huston K.K., Kerr G., et al. 2021 American College of Rheumatology Guideline for the Treatment of Rheumatoid Arthritis. Arthritis Care Res. 2021;73:924–939. doi: 10.1002/acr.24596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Putrik P., Ramiro S., Kvien T.K., Sokka T., Pavlova M., Uhlig T., Boonen A. Inequities in access to biologic and synthetic DMARDs across 46 European countries. Ann. Rheum. Dis. 2014;73:198–206. doi: 10.1136/annrheumdis-2012-202603. [DOI] [PubMed] [Google Scholar]
- 12.Bergstra S.A., Branco J.C., Vega-Morales D., Salomon-Escoto K., Govind N., Allaart C.F., Landewé R.B.M. Inequity in access to bDMARD care and how it influences disease outcomes across countries worldwide: Results from the METEOR-registry. Ann. Rheum. Dis. 2018;77:1413–1420. doi: 10.1136/annrheumdis-2018-213289. [DOI] [PubMed] [Google Scholar]
- 13.Kay J., Schoels M.M., Dörner T., Emery P., Kvien T.K., Smolen J.S., Breedveld F.C. Consensus-based recommendations for the use of biosimilars to treat rheumatological diseases. Ann. Rheum. Dis. 2018;77:165–174. doi: 10.1136/annrheumdis-2017-211937. [DOI] [PubMed] [Google Scholar]
- 14.Asociación Española de Medicamentos Biosimilares Análisis de Impacto Presupuestario de los Medicamentos Biosimilares en el Sistema Nacional de Salud de España (2009–2022) [(accessed on 1 June 2023)]. Available online: https://www.biosim.es/documentos/AIP_biosimilares_Hygeia_UCM_BioSim_nov2020.pdf.
- 15.Cornejo Uixeda S., Quintana Vergara B., Sánchez Alcaraz A. Análisis de la utilización de medicamentos biosimilares. Rev. De La OFIL. 2020;30:80–81. [Google Scholar]
- 16.Association W.M. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA. 2013;310:2191–2194. doi: 10.1001/jama.2013.281053. [DOI] [PubMed] [Google Scholar]
- 17.Aletaha D., Neogi T., Silman A.J., Funovits J., Felson D.T., Bingham C.O., III, Birnbaum N.S., Burmester G.R., Bykerk V.P., Cohen M.D., et al. 2010 Rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010;62:2569–2581. doi: 10.1002/art.27584. [DOI] [PubMed] [Google Scholar]
- 18.Bejarano V., Conaghan P.G., Quinn M.A., Saleem B., Emery P. Benefits 8 years after a remission induction regime with an infliximab and methotrexate combination in early rheumatoid arthritis. Rheumatology. 2010;49:1971–1974. doi: 10.1093/rheumatology/keq194. [DOI] [PubMed] [Google Scholar]
- 19.Durez P., Malghem J., Nzeusseu Toukap A., Depresseux G., Lauwerys B.R., Westhovens R., Luyten F.P., Corluy L., Houssiau F.A., Verschueren P. Treatment of early rheumatoid arthritis: A randomized magnetic resonance imaging study comparing the effects of methotrexate alone, methotrexate in combination with infliximab, and methotrexate in combination with intravenous pulse methylprednisolone. Arthritis Rheum. 2007;56:3919–3927. doi: 10.1002/art.23055. [DOI] [PubMed] [Google Scholar]
- 20.Goekoop-Ruiterman Y.P., de Vries-Bouwstra J.K., Allaart C.F., van Zeben D., Kerstens P.J., Hazes J.M., Zwinderman A.H., Peeters A.J., de Jonge-Bok J.M., Mallée C., et al. Comparison of treatment strategies in early rheumatoid arthritis: A randomized trial. Ann. Intern. Med. 2007;146:406–415. doi: 10.7326/0003-4819-146-6-200703200-00005. [DOI] [PubMed] [Google Scholar]
- 21.Goekoop-Ruiterman Y.P., de Vries-Bouwstra J.K., Allaart C.F., van Zeben D., Kerstens P.J., Hazes J.M., Zwinderman A.H., Ronday H.K., Han K.H., Westedt M.L., et al. Clinical and radiographic outcomes of four different treatment strategies in patients with early rheumatoid arthritis (the BeSt study): A randomized, controlled trial. Arthritis Rheum. 2005;52:3381–3390. doi: 10.1002/art.21405. [DOI] [PubMed] [Google Scholar]
- 22.Goekoop-Ruiterman Y.P., de Vries-Bouwstra J.K., Allaart C.F., van Zeben D., Kerstens P.J., Hazes J.M., Zwinderman A.H., Ronday H.K., Han K.H., Westedt M.L., et al. Clinical and radiographic outcomes of four different treatment strategies in patients with early rheumatoid arthritis (the BeSt study): A randomized, controlled trial. Arthritis Rheum. 2008;58:S126–S135. doi: 10.1002/art.21405. [DOI] [PubMed] [Google Scholar]
- 23.Markusse I.M., Akdemir G., Dirven L., Goekoop-Ruiterman Y.P., van Groenendael J.H., Han K.H., Molenaar T.H., Le Cessie S., Lems W.F., van der Lubbe P.A., et al. Long-Term Outcomes of Patients With Recent-Onset Rheumatoid Arthritis After 10 Years of Tight Controlled Treatment: A Randomized Trial. Ann. Intern. Med. 2016;164:523–531. doi: 10.7326/M15-0919. [DOI] [PubMed] [Google Scholar]
- 24.Nam J.L., Villeneuve E., Hensor E.M., Conaghan P.G., Keen H.I., Buch M.H., Gough A.K., Green M.J., Helliwell P.S., Keenan A.M., et al. Remission induction comparing infliximab and high-dose intravenous steroid, followed by treat-to-target: A double-blind, randomised, controlled trial in new-onset, treatment-naive, rheumatoid arthritis (the IDEA study) Ann. Rheum. Dis. 2014;73:75–85. doi: 10.1136/annrheumdis-2013-203440. [DOI] [PubMed] [Google Scholar]
- 25.Quinn M.A., Conaghan P.G., O’Connor P.J., Karim Z., Greenstein A., Brown A., Brown C., Fraser A., Jarret S., Emery P. Very early treatment with infliximab in addition to methotrexate in early, poor-prognosis rheumatoid arthritis reduces magnetic resonance imaging evidence of synovitis and damage, with sustained benefit after infliximab withdrawal: Results from a twelve-month randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 2005;52:27–35. doi: 10.1002/art.20712. [DOI] [PubMed] [Google Scholar]
- 26.Detert J., Bastian H., Listing J., Weiß A., Wassenberg S., Liebhaber A., Rockwitz K., Alten R., Krüger K., Rau R., et al. Induction therapy with adalimumab plus methotrexate for 24 weeks followed by methotrexate monotherapy up to week 48 versus methotrexate therapy alone for DMARD-naive patients with early rheumatoid arthritis: HIT HARD, an investigator-initiated study. Ann. Rheum. Dis. 2013;72:844–850. doi: 10.1136/annrheumdis-2012-201612. [DOI] [PubMed] [Google Scholar]
- 27.Soubrier M., Puéchal X., Sibilia J., Mariette X., Meyer O., Combe B., Flipo R.M., Mulleman D., Berenbaum F., Zarnitsky C., et al. Evaluation of two strategies (initial methotrexate monotherapy vs its combination with adalimumab) in management of early active rheumatoid arthritis: Data from the GUEPARD trial. Rheumatology. 2009;48:1429–1434. doi: 10.1093/rheumatology/kep261. [DOI] [PubMed] [Google Scholar]
- 28.Hørslev-Petersen K., Hetland M.L., Junker P., Pødenphant J., Ellingsen T., Ahlquist P., Lindegaard H., Linauskas A., Schlemmer A., Dam M.Y., et al. Adalimumab added to a treat-to-target strategy with methotrexate and intra-articular triamcinolone in early rheumatoid arthritis increased remission rates, function and quality of life. The OPERA Study: An investigator-initiated, randomised, double-blind, parallel-group, placebo-controlled trial. Ann. Rheum. Dis. 2014;73:654–661. doi: 10.1136/annrheumdis-2012-202735. [DOI] [PubMed] [Google Scholar]
- 29.Nam J.L., Villeneuve E., Hensor E.M., Wakefield R.J., Conaghan P.G., Green M.J., Gough A., Quinn M., Reece R., Cox S.R., et al. A randomised controlled trial of etanercept and methotrexate to induce remission in early inflammatory arthritis: The EMPIRE trial. Ann. Rheum. Dis. 2014;73:1027–1036. doi: 10.1136/annrheumdis-2013-204882. [DOI] [PubMed] [Google Scholar]
- 30.Emery P., Bingham C.O., III, Burmester G.R., Bykerk V.P., Furst D.E., Mariette X., van der Heijde D., van Vollenhoven R., Arendt C., Mountian I., et al. Certolizumab pegol in combination with dose-optimised methotrexate in DMARD-naïve patients with early, active rheumatoid arthritis with poor prognostic factors: 1-year results from C-EARLY, a randomised, double-blind, placebo-controlled phase III study. Ann. Rheum. Dis. 2017;76:96–104. doi: 10.1136/annrheumdis-2015-209057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hetland M.L., Haavardsholm E.A., Rudin A., Nordström D., Nurmohamed M., Gudbjornsson B., Lampa J., Hørslev-Petersen K., Uhlig T., Grondal G., et al. Active conventional treatment and three different biological treatments in early rheumatoid arthritis: Phase IV investigator initiated, randomised, observer blinded clinical trial. BMJ. 2020;371:m4328. doi: 10.1136/bmj.m4328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Weinblatt M.E., Bingham C.O., III, Burmester G.R., Bykerk V.P., Furst D.E., Mariette X., van der Heijde D., van Vollenhoven R., VanLunen B., Ecoffet C., et al. A Phase III Study Evaluating Continuation, Tapering, and Withdrawal of Certolizumab Pegol After One Year of Therapy in Patients With Early Rheumatoid Arthritis. Arthritis Rheumatol. 2017;69:1937–1948. doi: 10.1002/art.40196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Verhoeven M.M.A., Tekstra J., Welsing P.M.J., Pethö-Schramm A., Borm M.E.A., Bruyn G.A.W., Bos R., Griep E.N., Klaasen R., van Laar J.M., et al. Effectiveness and safety over 3 years after the 2-year U-Act-Early trial of the strategies initiating tocilizumab and/or methotrexate. Rheumatology. 2020;59:2325–2333. doi: 10.1093/rheumatology/kez602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bijlsma J.W.J., Welsing P.M.J., Woodworth T.G., Middelink L.M., Pethö-Schramm A., Bernasconi C., Borm M.E.A., Wortel C.H., Ter Borg E.J., Jahangier Z.N., et al. Early rheumatoid arthritis treated with tocilizumab, methotrexate, or their combination (U-Act-Early): A multicentre, randomised, double-blind, double-dummy, strategy trial. Lancet. 2016;388:343–355. doi: 10.1016/S0140-6736(16)30363-4. [DOI] [PubMed] [Google Scholar]
- 35.Breedveld F.C., Weisman M.H., Kavanaugh A.F., Cohen S.B., Pavelka K., van Vollenhoven R., Sharp J., Perez J.L., Spencer-Green G.T. The PREMIER study: A multicenter, randomized, double-blind clinical trial of combination therapy with adalimumab plus methotrexate versus methotrexate alone or adalimumab alone in patients with early, aggressive rheumatoid arthritis who had not had previous methotrexate treatment. Arthritis Rheum. 2006;54:26–37. doi: 10.1002/art.21519. [DOI] [PubMed] [Google Scholar]
- 36.Kavanaugh A., Fleischmann R.M., Emery P., Kupper H., Redden L., Guerette B., Santra S., Smolen J.S. Clinical, functional and radiographic consequences of achieving stable low disease activity and remission with adalimumab plus methotrexate or methotrexate alone in early rheumatoid arthritis: 26-week results from the randomised, controlled OPTIMA study. Ann. Rheum. Dis. 2013;72:64–71. doi: 10.1136/annrheumdis-2011-201247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bathon J.M., Martin R.W., Fleischmann R.M., Tesser J.R., Schiff M.H., Keystone E.C., Genovese M.C., Wasko M.C., Moreland L.W., Weaver A.L., et al. A comparison of etanercept and methotrexate in patients with early rheumatoid arthritis. N. Engl. J. Med. 2000;343:1586–1593. doi: 10.1056/NEJM200011303432201. [DOI] [PubMed] [Google Scholar]
- 38.Emery P., Breedveld F.C., Hall S., Durez P., Chang D.J., Robertson D., Singh A., Pedersen R.D., Koenig A.S., Freundlich B. Comparison of methotrexate monotherapy with a combination of methotrexate and etanercept in active, early, moderate to severe rheumatoid arthritis (COMET): A randomised, double-blind, parallel treatment trial. Lancet. 2008;372:375–382. doi: 10.1016/S0140-6736(08)61000-4. [DOI] [PubMed] [Google Scholar]
- 39.Emery P., Breedveld F., van der Heijde D., Ferraccioli G., Dougados M., Robertson D., Pedersen R., Koenig A.S., Freundlich B. Two-year clinical and radiographic results with combination etanercept-methotrexate therapy versus monotherapy in early rheumatoid arthritis: A two-year, double-blind, randomized study. Arthritis Rheum. 2010;62:674–682. doi: 10.1002/art.27268. [DOI] [PubMed] [Google Scholar]
- 40.Kekow J., Moots R.J., Emery P., Durez P., Koenig A., Singh A., Pedersen R., Robertson D., Freundlich B., Sato R. Patient-reported outcomes improve with etanercept plus methotrexate in active early rheumatoid arthritis and the improvement is strongly associated with remission: The COMET trial. Ann. Rheum. Dis. 2010;69:222–225. doi: 10.1136/ard.2008.102509. [DOI] [PubMed] [Google Scholar]
- 41.Rantalaiho V., Kautiainen H., Korpela M., Hannonen P., Kaipiainen-Seppänen O., Möttönen T., Kauppi M., Karjalainen A., Laiho K., Laasonen L., et al. Targeted treatment with a combination of traditional DMARDs produces excellent clinical and radiographic long-term outcomes in early rheumatoid arthritis regardless of initial infliximab. The 5-year follow-up results of a randomised clinical trial, the NEO-RACo trial. Ann. Rheum. Dis. 2014;73:1954–1961. doi: 10.1136/annrheumdis-2013-203497. [DOI] [PubMed] [Google Scholar]
- 42.St Clair E.W., van der Heijde D.M., Smolen J.S., Maini R.N., Bathon J.M., Emery P., Keystone E., Schiff M., Kalden J.R., Wang B., et al. Combination of infliximab and methotrexate therapy for early rheumatoid arthritis: A randomized, controlled trial. Arthritis Rheum. 2004;50:3432–3443. doi: 10.1002/art.20568. [DOI] [PubMed] [Google Scholar]
- 43.Tam L.S., Shang Q., Li E.K., Wang S., Li R.J., Lee K.L., Leung Y.Y., Ying K.Y., Yim C.W., Kun E.W., et al. Infliximab is associated with improvement in arterial stiffness in patients with early rheumatoid arthritis -- a randomized trial. J. Rheumatol. 2012;39:2267–2275. doi: 10.3899/jrheum.120541. [DOI] [PubMed] [Google Scholar]
- 44.Burmester G.R., Rigby W.F., van Vollenhoven R.F., Kay J., Rubbert-Roth A., Kelman A., Dimonaco S., Mitchell N. Tocilizumab in early progressive rheumatoid arthritis: FUNCTION, a randomised controlled trial. Ann. Rheum. Dis. 2016;75:1081–1091. doi: 10.1136/annrheumdis-2015-207628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Jones G., Sebba A., Gu J., Lowenstein M.B., Calvo A., Gomez-Reino J.J., Siri D.A., Tomsic M., Alecock E., Woodworth T., et al. Comparison of tocilizumab monotherapy versus methotrexate monotherapy in patients with moderate to severe rheumatoid arthritis: The AMBITION study. Ann. Rheum. Dis. 2010;69:88–96. doi: 10.1136/ard.2008.105197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kume K., Amano K., Yamada S., Hatta K., Ohta H., Kuwaba N. Tocilizumab monotherapy reduces arterial stiffness as effectively as etanercept or adalimumab monotherapy in rheumatoid arthritis: An open-label randomized controlled trial. J. Rheumatol. 2011;38:2169–2171. doi: 10.3899/jrheum.110340. [DOI] [PubMed] [Google Scholar]
- 47.Stevenson M., Archer R., Tosh J., Simpson E., Everson-Hock E., Stevens J., Hernandez-Alava M., Paisley S., Dickinson K., Scott D., et al. Adalimumab, etanercept, infliximab, certolizumab pegol, golimumab, tocilizumab and abatacept for the treatment of rheumatoid arthritis not previously treated with disease-modifying antirheumatic drugs and after the failure of conventional disease-modifying antirheumatic drugs only: Systematic review and economic evaluation. Health Technol. Assess. 2016;20:1–610. doi: 10.3310/hta20350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Archer R., Hock E., Hamilton J., Stevens J., Essat M., Poku E., Clowes M., Pandor A., Stevenson M. Assessing prognosis and prediction of treatment response in early rheumatoid arthritis: Systematic reviews. Health Technol. Assess. 2018;22:1–294. doi: 10.3310/hta22660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Burgers L.E., Raza K., van der Helm-van Mil A.H. Window of opportunity in rheumatoid arthritis—Definitions and supporting evidence: From old to new perspectives. RMD Open. 2019;5:e000870. doi: 10.1136/rmdopen-2018-000870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.John J.C. Early rheumatoid arthritis—Is there a window of opportunity? J. Rheumatol. 2007;80:1. [PubMed] [Google Scholar]
- 51.Breedveld F. The value of early intervention in RA—A window of opportunity. Clin. Rheumatol. 2011;30:33–39. doi: 10.1007/s10067-010-1638-5. [DOI] [PubMed] [Google Scholar]
- 52.Keystone E.C., Breedveld F.C., van der Heijde D., Landewé R., Florentinus S., Arulmani U., Liu S., Kupper H., Kavanaugh A. Longterm effect of delaying combination therapy with tumor necrosis factor inhibitor in patients with aggressive early rheumatoid arthritis: 10-year efficacy and safety of adalimumab from the randomized controlled PREMIER trial with open-label extension. J. Rheumatol. 2014;41:5–14. doi: 10.3899/jrheum.130543. [DOI] [PubMed] [Google Scholar]
- 53.Garcês S., Demengeot J., Benito-Garcia E. The immunogenicity of anti-TNF therapy in immune-mediated inflammatory diseases: A systematic review of the literature with a meta-analysis. Ann. Rheum. Dis. 2013;72:1947–1955. doi: 10.1136/annrheumdis-2012-202220. [DOI] [PubMed] [Google Scholar]
- 54.Braun J., Kastner P., Flaxenberg P., Wahrisch J., Hanke P., Demary W., von Hinuber U., Rockwitz K., Heitz W., Pichlmeier U., et al. Comparison of the clinical efficacy and safety of subcutaneous versus oral administration of methotrexate in patients with active rheumatoid arthritis: Results of a six-month, multicenter, randomized, double-blind, controlled, phase IV trial. Arthritis Rheum. 2008;58:73–81. doi: 10.1002/art.23144. [DOI] [PubMed] [Google Scholar]
- 55.Tornero Molina J., Calvo Alen J., Ballina J., Belmonte M., Blanco F.J., Caracuel M., Carbonell J., Corominas H., Chamizo E., Hidalgo C., et al. Recommendations for the use of parenteral methotrexate in rheumatic diseases. Reumatol. Clin. (Engl. Ed.) 2018;14:142–149. doi: 10.1016/j.reuma.2016.12.001. [DOI] [PubMed] [Google Scholar]
- 56.Islam M.S., Haq S.A., Islam M.N., Azad A.K., Islam M.A., Barua R., Hasan M.M., Mahmood M., Safiuddin M., Rahman M.M., et al. Comparative efficacy of subcutaneous versus oral methotrexate in active rheumatoid arthritis. Mymensingh Med. J. MMJ. 2013;22:483–488. [PubMed] [Google Scholar]
- 57.Harris J.A., Bykerk V.P., Hitchon C.A., Keystone E.C., Thorne J.C., Boire G., Haraoui B., Hazlewood G., Bonner A.J., Pope J.E., et al. Determining best practices in early rheumatoid arthritis by comparing differences in treatment at sites in the Canadian Early Arthritis Cohort. J. Rheumatol. 2013;40:1823–1830. doi: 10.3899/jrheum.121316. [DOI] [PubMed] [Google Scholar]
- 58.Ferraz-Amaro I., Seoane-Mato D., Sanchez-Alonso F., Martin-Martinez M.A., emAR II Study Group Synthetic disease-modifying antirheumatic drug prescribing variability in rheumatoid arthritis: A multilevel analysis of a cross-sectional national study. Rheumatol. Int. 2015;35:1825–1836. doi: 10.1007/s00296-015-3363-5. [DOI] [PubMed] [Google Scholar]
- 59.Tornero-Molina J., Andreu J.L., Martín-Martínez M.-A., Corominas H., Pérez Venegas J.J., Román-Ivorra J.A., Sánchez-Alonso F. Metotrexato en pacientes con artritis reumatoide en España: Subanálisis del proyecto AR Excellence. Reumatol. Clínica. 2019;15:338–342. doi: 10.1016/j.reuma.2017.11.007. [DOI] [PubMed] [Google Scholar]
- 60.Martínez-Sesmero J.M., Schoenenberger-Arnaiz J.A., Crespo-Diz C., Cerezales M. AB1389 Cost-Minimization Analysis in Rheumatoid Arthritis in Spain. Ann. Rheum. Dis. 2022;81:1799–1800. doi: 10.1136/annrheumdis-2022-eular.1012. [DOI] [Google Scholar]
- 61.Schoenenberger Arnaiz J., Crespo Diz C., Martínez Sesmero J., Cerezales M. 4CPS-083 Cost-effectiveness analysis of adalimumab and its clinical alternatives in immune-mediated inflammatory diseases in Spain. Eur. J. Hosp. Pharm. 2022;29:A55–A56. doi: 10.1136/ejhpharm-2022-eahp.117. [DOI] [Google Scholar]
- 62.Schoenenberger-Arnaiz J.A., Martínez-Sesmero J.M., Crespo-Diz C., Cerezales M., Guigini M. Adalimumab and its clinical alternatives in Crohn’s disease and ulcerative colitis in Spain. United Eur. Gastroenterol. J. 2022;10((Suppl. 8)):1064. [Google Scholar]
- 63.Brkic A., Diamantopoulos A.P., Haavardsholm E.A., Fevang B.T.S., Brekke L.K., Loli L., Zettel C., Rødevand E., Bakland G., Mielnik P., et al. Exploring drug cost and disease outcome in rheumatoid arthritis patients treated with biologic and targeted synthetic DMARDs in Norway in 2010–2019—A country with a national tender system for prescription of costly drugs. BMC Health Serv. Res. 2022;22:48. doi: 10.1186/s12913-021-07425-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Maurelli M., Girolomoni G., Gisondi P. Cost per responder of adalimumab biosimilars versus methotrexate in patients with psoriasis: A real-life experience. J. Dermatolog. Treat. 2023;34:2218504. doi: 10.1080/09546634.2023.2218504. [DOI] [PubMed] [Google Scholar]
- 65.Patel D., Shelbaya A., Cheung R., Aggarwal J., Park S.H., Coindreau J. Cost-Effectiveness of Early Treatment with Originator Biologics or Their Biosimilars After Methotrexate Failure in Patients with Established Rheumatoid Arthritis. Adv. Ther. 2019;36:2086–2095. doi: 10.1007/s12325-019-00986-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Atsumi T., Tanaka Y., Yamamoto K., Takeuchi T., Yamanaka H., Ishiguro N., Eguchi K., Watanabe A., Origasa H., Yasuda S., et al. Clinical benefit of 1-year certolizumab pegol (CZP) add-on therapy to methotrexate treatment in patients with early rheumatoid arthritis was observed following CZP discontinuation: 2-year results of the C-OPERA study, a phase III randomised trial. Ann. Rheum. Dis. 2017;76:1348–1356. doi: 10.1136/annrheumdis-2016-210246. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.