Abstract
In Western countries, the gold-standard therapeutic strategy for rectal cancer is preoperative chemoradiotherapy (CRT) following total mesorectal excision (TME), without lateral lymph node dissection (LLND). However, preoperative CRT has recently been reported to be insufficient to control lateral lymph node recurrence in cases of enlarged lateral lymph nodes before CRT, and LLND is considered necessary in such cases. We performed a literature review on aspects of pelvic anatomy associated with rectal surgery and LLND, and then combined this information with our experience and knowledge of pelvic anatomy. In this review, drawing upon research using a 3-dimensional anatomical model and actual operative views, we aimed to clarify the essential anatomy for LLND. The LLND procedure was developed in Asian countries and can now be safely performed in terms of functional preservation. Nonetheless, the longer operative time, hemorrhage, and higher complication rates with TME accompanied by LLND than with TME alone indicate that LLND is still a challenging procedure. Laparoscopic or robotic LLND has been shown to be useful and is widely performed; however, without a sufficient understanding of anatomical landmarks, misrecognition of vessels and nerves often occurs. To perform safe and accurate LLND, understanding the landmarks of LLND is essential.
Keywords: Lateral lymph node dissection, Colorectal surgery, Laparoscoy, Robot
INTRODUCTION
After abdominoperitoneal resection for rectal cancer was introduced in 1908 by Miles [1] to improve patients’ prognosis, extended resection accompanied by the dissection of both aortic and lateral lymph nodes (LLNs) was performed by Sauer and Bacon [2], Bacon et al. [3], and Stearns and Deddish [4] in the 1950s. However, this strategy resulted in hemorrhage and adverse effects on urinary and sexual function, and was performed less frequently. In 1982, the principle of total mesorectal excision (TME) was proposed [5], and TME was reported to reduce local recurrence [6, 7]. In addition, during the 2000s, several studies demonstrated that TME combined with preoperative chemoradiotherapy (CRT) dramatically decreased local recurrence [8–12]. Consequently, TME with CRT has become the gold-standard treatment for clinical stage II and III rectal cancers in Western countries.
In Eastern countries, the treatment of rectal cancer is different. As advanced rectal cancer below the peritoneal reflection has a higher risk of spreading to LLNs, for clinical T3 and T4 lower rectal cancer, the treatment strategy of resection combined with TME and prophylactic LLN dissection (LLND) without preoperative CRT has been developed [13–15]. This surgical approach for advanced rectal cancer has been reported to have a local recurrence rate comparable to that of TME with CRT [16]. As a result, TME with LLND without preoperative CRT has become the standard treatment for clinical stage II and III rectal cancers in Eastern countries.
However, in recent years, LLND and CRT have come to be considered not as independent treatment modalities, but as treatments that can be combined in some cases. LLND without CRT has recently been reported to reduce lateral pelvic recurrence but not central pelvic local recurrence. Therefore, in higher risk cases of rectal cancer (e.g., bulky tumors with deep penetration or threatening circumferential margins), LLND with preoperative CRT should be considered [17, 18]. On the contrary, recent studies have demonstrated that when enlarged LLNs were present in pre-CRT situations, CRT was insufficient to control LLN recurrence; in those cases, LLND with CRT has been considered to achieve local control [19–24]. From this standpoint, the combination of CRT and LLND has become an important future therapeutic strategy for advanced rectal cancer, and training in LLND has become necessary for colorectal surgeons in both Western and Eastern countries.
As aforementioned, extended resection including LLNs resulted in increased postoperative dysfunction [2–4]. In an attempt to improve this, an autonomic nerve preservation technique was developed in Japan [25]. Consequently, LLND can be safely performed in terms of function preservation. A randomized controlled trial in Japan comparing LLND and non-LLND for clinical stage II or III rectal cancer showed comparable rates of postoperative sexual and urinary dysfunction [17]. However, LLND remains challenging because of a longer operative time, higher risk of hemorrhage, and a marginally higher rate of grade III or IV complications with LLND than without LLND [17]. Laparoscopic and robotic surgery has recently become popular worldwide, and LLND has gradually evolved from an open technique to a laparoscopic or robotic approach. The safety and feasibility of these minimally invasive approaches for LLND have been demonstrated [26, 27]. The laparoscopic magnified view enables surgeons to identify small vessels, nerves, and fascial laminations invisible to the naked eye in conventional open surgery; however, without a sufficient understanding of anatomical landmarks, misrecognition of vessels and nerves often occurs. Therefore, in this review, we aimed to clarify the essential anatomy for LLND drawing upon research using a 3-dimensional anatomical model and actual operative views from our institution, The University of Tokyo (Tokyo, Japan).
Ethics statement
The study was approved by the Institutional Review Board of The University of Tokyo (No. 3252-15). Written informed consents for publication of the research details were obtained.
DEFINITION OF LLN
In Japan, the Japanese Classification of Colorectal Carcinoma categorizes LLNs as internal iliac lymph nodes (#263), obturator lymph nodes (#283), external iliac lymph nodes (#293), common iliac lymph nodes (#273), aortic bifurcation lymph nodes (#280), and midline sacrum lymph nodes (#270) (Fig. 1) [28]. The metastasis rate in LLN-positive cases has been previously reported [29]. Among LLNs, the internal iliac lymph nodes showed the highest rate of metastasis. The metastasis rate differs on either side of the superior vesical artery, which is a branch of the internal iliac artery; the frequencies were 26% on the proximal side of the superior vesical artery and 47% on the distal side. The obturator lymph nodes are the second most common region of metastasis, which occurs in 38% of cases. The frequency of metastasis in other areas (e.g., the external iliac lymph nodes, common iliac lymph nodes, and aortic bifurcation/midline sacrum lymph nodes, was much lower, with a total frequency of 7.7%). Based on these data, the target lymph nodes for prophylactic LLND were defined in the Japanese Classification of Colorectal Carcinoma as internal iliac and obturator lymph nodes. Therefore, in this review, we describe the essential anatomy for resecting these 2 regions of lymph nodes.
Fig. 1.
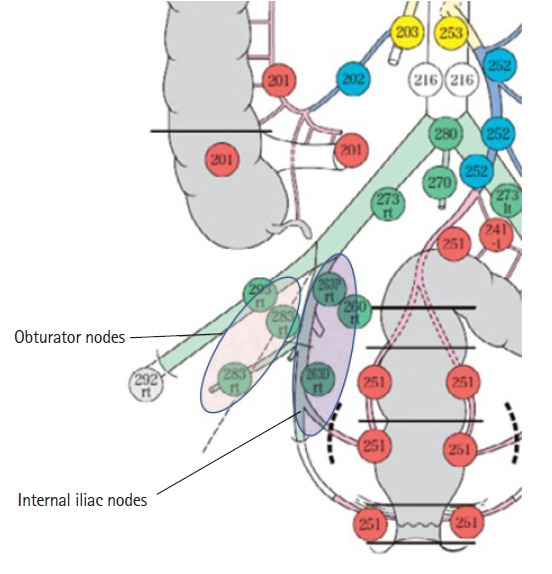
Lateral lymph nodes categorized according to the Japanese Classification of Colorectal Carcinoma. Reproduced from the Japanese Society for Cancer of the Colon and Rectum [28] with permission from Kanehara and the Japanese Society for Cancer of the Colon and Rectum.
ESSENTIAL LANDMARKS FOR LLND
Accurate and safe LLND requires an understanding of the lateral, internal, dorsal, and caudal planes of the LLN region. The anatomical structures that comprise these respective planes are as follows: lateral plane (external iliac artery and vein and internal obturator muscle), internal plane (pelvic plexus and bladder), dorsal plane (internal iliac artery and vein, piriform muscle, and sacral plexus), and caudal plane (coccygeal muscle and levator ani muscle).
Fig. 2A shows the anatomy of the lateral, dorsal, and caudal planes of the LLN region [30]. After giving rise to parietal branches, such as the superior and inferior gluteal arteries, and visceral branches, such as the umbilical and superior/inferior vesical arteries, the internal iliac artery flows into the Alcock canal as the internal pudendal artery. The branch patterns of the internal iliac artery are variegated [31]. The following is the most common type: the internal iliac artery first gives rise to the superior gluteal arteries and visceral arteries, before branching into the inferior gluteal artery and internal pudendal artery, which then exit the pelvis through the greater sciatic foramen and Alcock canal, respectively (Fig. 2A) [30]. In some cases, this branching to the inferior gluteal artery and internal pudendal artery occurs after the internal iliac artery flows out between the piriform and coccygeus muscles. When this happens, the gluteal-pudendal trunk comprises the dorsal plane of LLND. The Alcock canal is enclosed between the piriform and coccygeus muscles. Through the Alcock canal, the pudendal nerve, which is a branch of the sacral plexus, traces through the pelvis along with the internal pudendal artery. In the aforementioned figure, to clearly understand the lateral, dorsal, and caudal planes of the LLN region, the branches of the internal iliac artery, pelvic plexus, and ureter were eliminated. The levator ani, which comprises the caudal edge, and the internal obturator muscle, which comprises the lateral edge, are bounded by the tendinous arch of the levator ani. Fig. 2B [30] shows the lateral, dorsal, and caudal planes of the LLND.
Fig. 2.
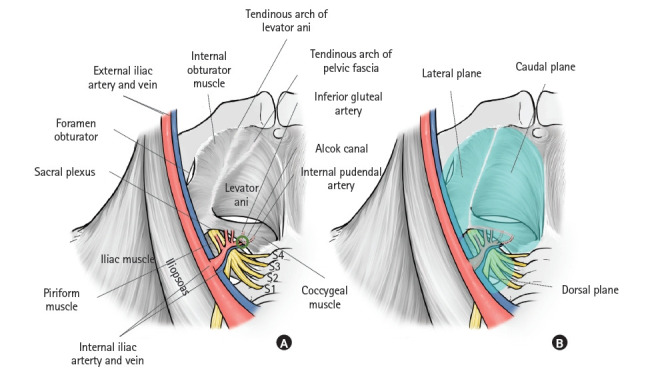
Dorsal, lateral, and caudal planes of lateral lymph node dissection. (A) Pelvic anatomy, excluding the ureter, pelvic plexus, branches of the internal iliac vein and artery, and obturator nerve, vein, and artery. (B) Dorsal, lateral, and caudal planes of lateral lymph node dissection. Reproduced from Yokoyama et al. [30] with permission from Kanehara.
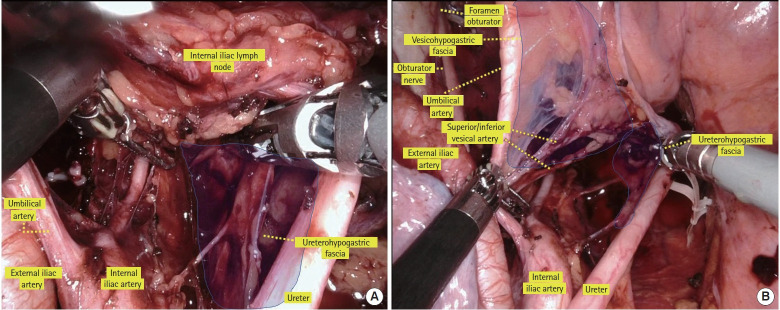
The medial plane of the obturator lymph node region is the vesicohypogastric fascia, which means that the internal iliac and obturator lymph node regions are bounded by the vesicohypogastric fascia (Fig. 3A) [30]. Understanding the vesicohypogastric fascia is essential when performing LLND [32–34]. The vesicohypogastric fascia is a triangular fascia that stands out from the internal iliac artery along the umbilical artery and the lateral border of the bladder. It is a double-layer sheet surrounding the branches of the internal iliac artery, such as the umbilical, superior/inferior vesical, and uterine arteries (Fig. 3A) [30]. The vesicohypogastric fascia is united with the Gerota capsule in the cranial direction. The obturator artery, vein, and nerve are shown in Fig. 3B [30]. The fat tissue surrounded by the lateral plane (composed of the external iliac artery and vein and internal obturator muscle), the dorsal plane (composed of the internal iliac artery and vein, piriform muscle, and sacral plexus), and the medial plane (the vesicohypogastric fascia) should be dissected for the obturator lymph node. The obturator artery, vein, and nerve run through this region (Fig. 3B) [30].
Fig. 3.
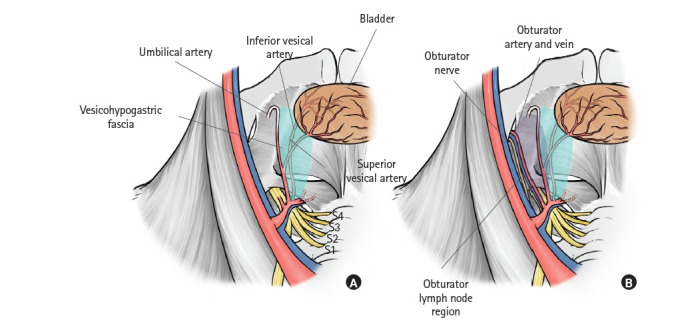
Medial plane of the obturator lymph node region. (A) Anatomy of the vesicohypogastric fascia. Visceral branches of the internal iliac artery, such as the umbilical and vesical arteries run between this fascia. (B) The obturator nerve, artery, and vein pass through the obturator lymph node region. Reproduced from Yokoyama et al. [30] with permission from Kanehara.
The internal plane of LLND, which is identical to the internal plane of the internal iliac lymph node region, comprises the ureterohypogastric fascia, which is also known as the prehypogastric nerve fascia [33, 34]. As its name suggests, tracing the fascia proximally and ventrally leads to the ureter. The ureterohypogastric fascia is the border between the mesorectum and LLN region. This fascia includes the hypogastric nerve and pelvic plexus and joins the Denonvilliers’ fascia at the anterolateral aspect (Fig. 4) [30]. At the anterolateral aspect, branches of the pelvic plexus, which run through the ureterohypogastric fascia, compose a bundle with genital blood vessels that run through the vesicohypogastric fascia. This bundle is called the neurovascular bundle (NVB). Similar to the vesicohypogastric fascia, the ureterohypogastric fascia is united with the Gerota capsule in the cranial direction. Both the fat tissue included in the vesicohypogastric fascia itself and that between the vesicohypogastric fascia and ureterohypogastric fascia include the internal iliac lymph nodes.
Fig. 4.
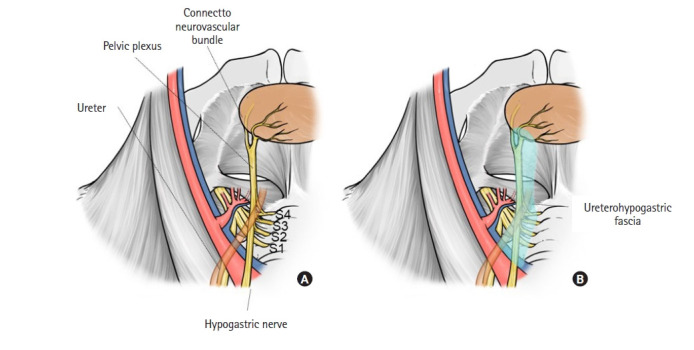
Medial plane of the internal iliac lymph node region. (A) Anatomy of the pelvis plexus, hypogastric nerve, and ureter. Branches of the pelvic plexus jointly with small vessels from the vesical artery at anterolateral aspects of the rectum comprise the neurovascular bundle. (B) Anatomy of the ureterohypogastric fascia. The hypogastric nerve, pelvic plexus, and ureter run through this fascia. Reproduced from Yokoyama et al. [30] with permission from Kanehara.
Considering the caudal plane of LLND, the tendinous arch of the pelvic fascia is also important. Little has been reported regarding the relationship between the vesicohypogastric fascia, ureterohyogastric fascia, and the tendinous arch of the pelvic fascia. Occelli et al. [35] described the tendinous arch of the pelvic fascia as a thickening of the fascia of the levator ani muscle, which extends from the internal end of the pubic insertion of the levator ani to the internal aspect of the ischiatic spine. They also mentioned that the posterior third of the tendinous arch of the pelvic fascia is fused with the posterior third of the tendinous arch of the levator ani. Pit et al. [36] explained that the tendinous arch of the pelvic fascia results from thickening of the endopelvic fascia, which is firmly attached to the posterolateral part of the ureter via fibrous tissues. This means that the ureterohypogastric fascia is fused with the tendinous arch of the pelvic fascia. The ureterohypogastic fascia, vesicohypogastric fascia, and mesorectal fascia unite below the S4 level [37]. From these anatomical studies, the tendinous arch of the pelvis is considered to be the fusion point of the ureterohypogatric fascia, vesicohypogastric fascia, mesorectal fascia, and the fascia of the levator ani (Fig. 2A) [30]. During LLND, surgeons dissect the fusion point to connect the dissection region of the lateral lymph node region to the dissection region of TME. Fig. 5 [30] shows the relationship between the vesicohypogastric fascia, ureterohypogastric fascia, obturator lymph node region, and internal iliac lymph node region.
Fig. 5.
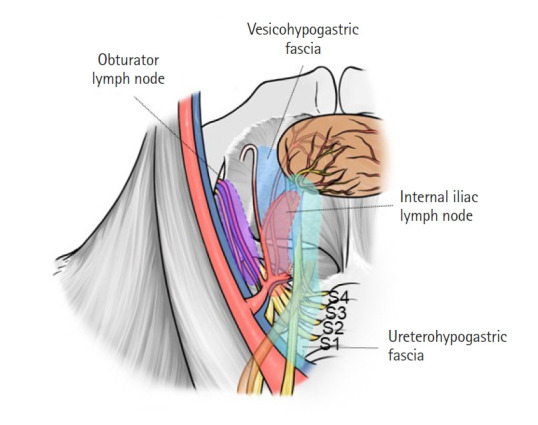
Anatomies of the vesicohypogastric fascia, ureterohypogastric fascia, obturator lymph node region, and internal iliac lymph node region. Reproduced from Yokoyama et al. [30] with permission from Kanehara.
NEUROVASCULAR BUNDLE
Understanding the anatomy of the NVB is essential both for LLND and rectal cancer surgery. Comprehension of the vesicohypogastric and ureterohypogastric fasciae leads to an understanding of the anatomy of the NVB. The hypogastric and pelvic nerves connect with each other in the ureterohypogastric fascia and form the pelvic plexus. The ureterohypogastric fascia fuses with Denonvilliers’ fascia on the anterolateral side of the rectum. Rectal branches from the pelvic plexus and occasionally the middle rectal artery run through this area to the rectum, forming the lateral ligament of the rectum [38–41]. However, in anatomical studies, the ligamentous component was not recognized, so the lateral ligament was considered to be a dense adhesion between the fascia, rectal branches of the pelvic plexus, and middle rectal artery [38–41]. After branching from the rectal branches, the pelvic plexus runs in 3 directions: ventrally (passing near the seminal vesicle and distributed throughout the bladder); runs ventrally and caudally (passing between the prostate and seminal vesicle, and distributed along the side wall of the prostate); and caudally (passing near the rectum and through the posterolateral side of the prostate, and distributed to the corpus cavernosum of the penis) (Fig. 6A) [30, 42]. This last branch of the pelvic plexus, jointly with small vessels of the genital branches of the internal iliac artery (distal branches of the superior and inferior vesicle artery), which run through the vesicohypogastric fascia, comprises the NVB (Fig. 6B) [30]. This means that the vesicohypogastric and ureterohypogastric fasciae fuse with each other and comprise the NVB at the anterolateral aspect of the rectum (Fig. 6C) [30]. These fused fasciae unite with the Denonvilliers’ fascia on the dorsal side of the prostate.
Fig. 6.
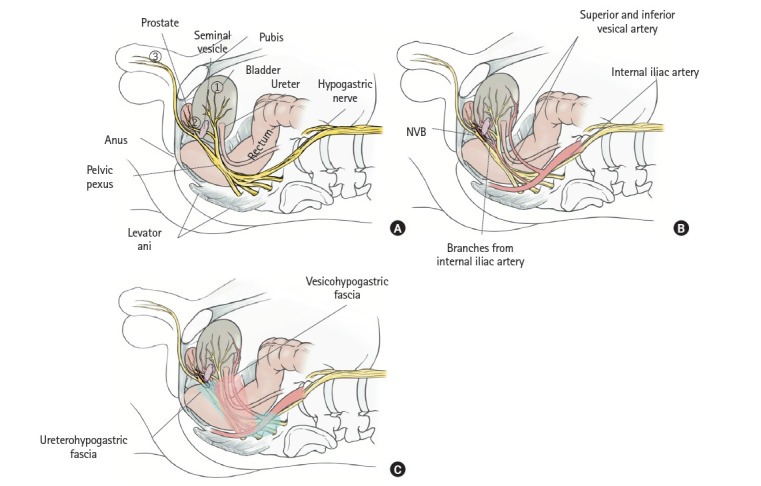
Anatomy of the neurovascular bundle (NVB). (A) Branches of the pelvic plexus run ventrally, pass near the seminal vesicle, and distribute to the bladder; run ventrally and caudally, pass between the prostate and seminal vesicle, and distribute to the side wall of the prostate; and run caudally, pass near the rectum and through the posterolateral side of the prostate, and distribute to the cavernous body of the penis. (B) Anatomies of the branches of the pelvic plexus, branches of the vesical arteries, and NVB. (C) Vesicohypogastric fascia and ureterohypogastric fascia. Reproduced from Yokoyama et al. [30] with permission from Kanehara.
Understanding the essential anatomy of LLND is important for understanding the anatomy of rectal surgery. Fig. 7 shows the 3-dimensional printed pelvic anatomy of all structures except the pelvic plexus and hypogastric nerve [30, 43]. This anatomy should be understood to ensure safe and accurate LLND.
Fig. 7.
Essential pelvic anatomy for lateral lymph node dissection. (A) Top view of the 3-dimensional anatomical model. This case has a duplicated ureter. (B) Side view of the 3-dimensional anatomical model. Reproduced from Yokoyama et al. [30] with permission from Kanehara.
PROCEDURE FOR LLND
Using actual images from robotic surgery, the following section presents an explanation of the dissection procedure at our hospital and the landmarks of dissection. As aforementioned, complete LLND requires complete resection of the obturator (#283) and internal iliac (#263) lymph nodes. The choice of the first lymph node region to be dissected differs among institutions. At our institution, the obturator lymph node is dissected first.
Expose the lateral edge of the obturator lymph node region
LLND is performed after the completion of TME for the primary lesion. First, the ureter is taped, and the ureterohypogastric fascia is separated from the internal fatty tissue, which includes the internal lymph nodes, by retracting the taped ureter medially. The incision is extended caudally and medially until it joins the dissection part of TME. This maneuver is important for preserving the autonomic nerve and obtaining a sufficient surgical view for dissection of the obturator lymph nodes.
The dissection is then started lateral to the obturator nodes. The external iliac artery and vein are exposed. The external iliac vein is located on the dorsal side of the artery. After retracting the external iliac vein laterally with a blunt instrument, the fatty tissues are peeled from the psoas and internal obturator muscles. By continuing the dissection caudally, the internal obturator muscle, obturator foramen, and obturator artery, vein, and nerve that flow into the obturator foramen can be identified. The obturator artery and vein are then divided. When an accessory obturator vein flows into the external iliac vein, it is also divided (Fig. 8A) [30]. The dissection continues in a caudal direction until it reaches the tendinous arch of the levator ani and that of the pelvis fascia. Here, it denotes the caudal edge of the obturator lymph node region (Fig. 8B) [30].
Fig. 8.
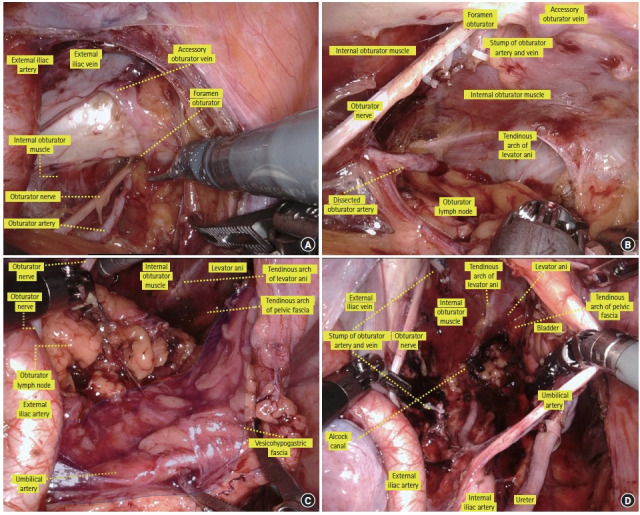
Operative view of dissection of the obturator lymph nodes. (A) Lateral plane of the obturator lymph node region. (B) Lateral and caudal planes of the obturator lymph node region. (C) Medial plane of the obturator lymph node region. (D) Completion view of dissection of the obturator lymph nodes. Reproduced from Yokoyama et al. [30] with permission from Kanehara.
Expose the dorsal and internal edges of the obturator lymph node region
The internal edge of the obturator lymph node region is the vesicohypogastric fascia. When identifying the umbilical artery and superior and inferior vesical artery, these arteries form a fascia-like structure. This is the vesicohypogastric fascia. Next, the umbilical or vesical artery is retracted medially and caudally, the fat tissue is separated from the vesicohypogastric fascia in line with the side wall of the bladder, and then the tendinous arch of the levator ani and that of the pelvic fascia, which form the caudal edge of the obturator lymph node, are reached (Fig. 8C) [30].
The dorsal edge of the obturator lymph node region is composed of the internal iliac artery and vein, piriform muscle, and sacral plexus. The fatty tissue is peeled away in line with the internal iliac artery, and then the obturator artery and vein, which branches from the internal iliac artery, is identified. The obturator artery and vein are divided for a second time at their origin. The obturator nerve is preserved, and dissection is continued along the sacral plexus and internal iliac artery to reach the Alcock canal. The obturator lymph nodes are completely removed from the obturator compartment. Fig. 8D [30] shows the completion of obturator lymph node dissection.
Expose the internal edge of the internal lymph node region
Internal lymph nodes exist between the vesicohypogastric fascia and ureterohypogastric fascia. To prevent injury to the hypogastric nerve, the ureterohypogastric fascia, including the ureter and hypogastric nerves, is first separated when LLND is performed (Fig. 9A) [30]. The ureter is retracted medially, the vesicohypogastric fascia is retracted laterally, and fatty tissues are peeled from the internal iliac artery branches (e.g., the umbilical artery and superior/inferior vesical artery) until the Alcock canal is observed. When lymph node metastasis is suspected between the vesicohypogastric fascia, the superior and inferior vesical arteries are divided with the lymph nodes. However, dissection of the vesical artery has been reported to increase the rate of postoperative urinary dysfunction [44]; therefore, if there is no apparent lymph node metastasis in the vesicohypogastric fascia, the vesical arteries are preserved. As aforementioned, among the LLNs, the internal iliac lymph nodes on the distal side of the superior vesical artery have a high metastasis rate; hence, complete resection of the fatty tissue in this region is important [28]. Fig. 9B [30] shows a view of completed dissection of the internal iliac lymph nodes.
Fig. 9.
Operative view of dissection of the internal iliac lymph nodes. (A) Medial plane of the internal iliac lymph node region. (B) Completion view of dissection of the internal iliac lymph nodes. Reproduced from Yokoyama et al. [30] with permission from Kanehara.
CONCLUSION
We conducted a review of the essential anatomy of LLND. As the procedure poses challenges, it is crucial to achieving radical resection and preservation of genital function. Particularly in laparoscopic and robotic surgery, misunderstanding the anatomy for LLND easily leads to misrecognizing vessels and nerves. Therefore, to perform safe and accurate LLND, a thorough understanding of the landmarks for the LLND procedure is essential.
Footnotes
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Funding
This work was supported by Grant-in-Aid for Scientific Research (C) (No. 18K07194, 19K09114, 19K09115, 20K09051), Grant-in-Aid for Challenging Research (Exploratory) (No. 20K21626), and Grant-in-Aid for Scientific Research (B) (No. 21H02778) from the Japan Society for the Promotion of Science. This work was also supported by the Project for Cancer Research and Therapeutic Evolution (P-CREATE) (No. JP 19cm0106502) from the Japan Agency for Medical Research and Development (AMED).
Author contributions
Conceptualization: YY, SI; Data curation: YY, HN, KS, KM, SE; Investigation: YY, HM, SA, YN, YY, TS, HS; Methodology: YY, DH; Project administration: SI; Writing-original draft: YY; Writing-review & editing: all authors. All authors read and approved the final manuscript.
REFERENCES
- 1.Miles WE. A method of performing abdomino-perineal excision for carcinoma of the rectum and of the terminal portion of the pelvic colon. Lancet. 1908;2:1812–3. doi: 10.3322/canjclin.21.6.361. [DOI] [PubMed] [Google Scholar]
- 2.Sauer I, Bacon HE. A new approach for excision of carcinoma of the lower portion of the rectum and anal canal. Surg Gynecol Obstet. 1952;95:229–42. [PubMed] [Google Scholar]
- 3.Bacon HE, Dirbas F, Myers TB, Ponce De Leon F. Extensive lymphad enectomy and high ligation of the inferior mesenteric artery for carcinoma of the left colon and rectum. Dis Colon Rectum. 1958;1:457–64. doi: 10.1007/BF02633415. [DOI] [PubMed] [Google Scholar]
- 4.Stearns MW, Jr, Deddish MR. Five-year results of abdominopelvic lymph node dissection for carcinoma of the rectum. Dis Colon Rectum. 1959;2:169–72. doi: 10.1007/BF02616711. [DOI] [PubMed] [Google Scholar]
- 5.Heald RJ, Husband EM, Ryall RD. The mesorectum in rectal cancer surgery: the clue to pelvic recurrence? Br J Surg. 1982;69:613–6. doi: 10.1002/bjs.1800691019. [DOI] [PubMed] [Google Scholar]
- 6.Quirke P, Steele R, Monson J, Grieve R, Khanna S, Couture J, et al. Effect of the plane of surgery achieved on local recurrence in patients with operable rectal cancer: a prospective study using data from the MRC CR07 and NCIC-CTG CO16 randomised clinical trial. Lancet. 2009;373:821–8. doi: 10.1016/S0140-6736(09)60485-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Quirke P, Durdey P, Dixon MF, Williams NS. Local recurrence of rectal adenocarcinoma due to inadequate surgical resection: histopathological study of lateral tumour spread and surgical excision. Lancet. 1986;2:996–9. doi: 10.1016/s0140-6736(86)92612-7. [DOI] [PubMed] [Google Scholar]
- 8.Colorectal Cancer Collaborative Group Adjuvant radiotherapy for rectal cancer: a systematic overview of 8,507 patients from 22 randomised trials. Lancet. 2001;358:1291–304. doi: 10.1016/S0140-6736(01)06409-1. [DOI] [PubMed] [Google Scholar]
- 9.Folkesson J, Birgisson H, Pahlman L, Cedermark B, Glimelius B, Gunnarsson U. Swedish Rectal Cancer Trial: long lasting benefits from radiotherapy on survival and local recurrence rate. J Clin Oncol. 2005;23:5644–50. doi: 10.1200/JCO.2005.08.144. [DOI] [PubMed] [Google Scholar]
- 10.Peeters KC, Marijnen CA, Nagtegaal ID, Kranenbarg EK, Putter H, Wiggers T, et al. The TME trial after a median follow-up of 6 years: increased local control but no survival benefit in irradiated patients with resectable rectal carcinoma. Ann Surg. 2007;246:693–701. doi: 10.1097/01.sla.0000257358.56863.ce. [DOI] [PubMed] [Google Scholar]
- 11.Bosset JF, Collette L, Calais G, Mineur L, Maingon P, RadosevicJelic L, et al. Chemotherapy with preoperative radiotherapy in rectal cancer. N Engl J Med. 2006;355:1114–23. doi: 10.1056/NEJMoa060829. [DOI] [PubMed] [Google Scholar]
- 12.Gérard JP, Conroy T, Bonnetain F, Bouché O, Chapet O, Closon-Dejardin MT, et al. Preoperative radiotherapy with or without concurrent fluorouracil and leucovorin in T3-4 rectal cancers: results of FFCD 9203. J Clin Oncol. 2006;24:4620–5. doi: 10.1200/JCO.2006.06.7629. [DOI] [PubMed] [Google Scholar]
- 13.Hojo K, Koyama Y. The effectiveness of wide anatomical resection and radical lymphadenectomy for patients with rectal cancer. Jpn J Surg. 1982;12:111–6. doi: 10.1007/BF02469377. [DOI] [PubMed] [Google Scholar]
- 14.Hojo K, Koyama Y, Moriya Y. Lymphatic spread and its prognostic value in patients with rectal cancer. Am J Surg. 1982;144:350–4. doi: 10.1016/0002-9610(82)90018-6. [DOI] [PubMed] [Google Scholar]
- 15.Moriya Y, Hojo K, Sawada T, Koyama Y. Significance of lateral node dissection for advanced rectal carcinoma at or below the peritoneal reflection. Dis Colon Rectum. 1989;32:307–15. doi: 10.1007/BF02553486. [DOI] [PubMed] [Google Scholar]
- 16.Kusters M, Beets GL, van de Velde CJ, Beets-Tan RG, Marijnen CA, Rutten HJ, et al. A comparison between the treatment of low rectal cancer in Japan and the Netherlands, focusing on the patterns of local recurrence. Ann Surg. 2009;249:229–35. doi: 10.1097/SLA.0b013e318190a664. [DOI] [PubMed] [Google Scholar]
- 17.Fujita S, Mizusawa J, Kanemitsu Y, Ito M, Kinugasa Y, Komori K, et al. Mesorectal excision with or without lateral lymph node dissection for clinical stage II/III lower rectal cancer (JCOG0212): a multicenter, randomized controlled, noninferiority trial. Ann Surg. 2017;266:201–7. doi: 10.1097/SLA.0000000000002212. [DOI] [PubMed] [Google Scholar]
- 18.Kanemitsu Y, Shida D, Tsukamoto S, Moritani K, Sakamoto R. Japanese evidences on nerve-preserving lateral pelvic lymh node dissection for rectal cancer: major historical milestones and clinical impact: the past, present and future. Clin Colon Rectal Surg. 2020;33:349–54. doi: 10.1055/s-0040-1714238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kim TH, Jeong SY, Choi DH, Kim DY, Jung KH, Moon SH, et al. Lateral lymph node metastasis is a major cause of locoregional recurrence in rectal cancer treated with preoperative chemoradiotherapy and curative resection. Ann Surg Oncol. 2008;15:729–37. doi: 10.1245/s10434-007-9696-x. [DOI] [PubMed] [Google Scholar]
- 20.Kim TG, Park W, Choi DH, Park HC, Kim SH, Cho YB, et al. Factors associated with lateral pelvic recurrence after curative resection following neoadjuvant chemoradiotherapy in rectal cancer patients. Int J Colorectal Dis. 2014;29:193–200. doi: 10.1007/s00384-013-1797-3. [DOI] [PubMed] [Google Scholar]
- 21.Kim MJ, Kim TH, Kim DY, Kim SY, Baek JY, Chang HJ, et al. Can chemoradiation allow for omission of lateral pelvic node dissection for locally advanced rectal cancer? J Surg Oncol. 2015;111:459–64. doi: 10.1002/jso.23852. [DOI] [PubMed] [Google Scholar]
- 22.Kusters M, Slater A, Muirhead R, Hompes R, Guy RJ, Jones OM, et al. What to do with lateral nodal disease in low locally advanced rectal cancer? A call for further reflection and research. Dis Colon Rectum. 2017;60:577–85. doi: 10.1097/DCR.0000000000000834. [DOI] [PubMed] [Google Scholar]
- 23.Ogura A, Konishi T, Cunningham C, Garcia-Aguilar J, Iversen H, Toda S, et al. Neoadjuvant (chemo)radiotherapy with total mesorectal excision only is not sufficient to prevent lateral local recurrence in enlarged nodes: results of the multicenter lateral node study of patients with low cT3/4 rectal cancer. J Clin Oncol. 2019;37:33–43. doi: 10.1200/JCO.18.00032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kawai K, Shiratori H, Hata K, Nozawa H, Tanaka T, Nishikawa T, et al. Optimal size criteria for lateral lymph node dissection after neoadjuvant chemoradiotherapy for rectal cancer. Dis Colon Rectum. 2021;64:274–83. doi: 10.1097/DCR.0000000000001866. [DOI] [PubMed] [Google Scholar]
- 25.Tsuchiya S. Autonomic nerve preservation in rectal cancer surgery. Surgery. 1982;44:1367–73. [Google Scholar]
- 26.Konishi T, Kuroyanagi H, Oya M, Ueno M, Fujimoto Y, Akiyoshi T, et al. Lateral lymph node dissection with preoperative chemoradiation for locally advanced lower rectal cancer through a laparoscopic approach. Surg Endosc. 2011;25:2358–9. doi: 10.1007/s00464-010-1531-y. [DOI] [PubMed] [Google Scholar]
- 27.Akiyoshi T, Ueno M, Matsueda K, Konishi T, Fujimoto Y, Nagayama S, et al. Selective lateral pelvic lymph node dissection in patients with advanced low rectal cancer treated with preoperative chemoradiotherapy based on pretreatment imaging. Ann Surg Oncol. 2014;21:189–96. doi: 10.1245/s10434-013-3216-y. [DOI] [PubMed] [Google Scholar]
- 28.Japanese Society for Cancer of the Colon and Rectum . Japanese classification of colorectal, appendiceal, and anal carcinoma. 9th ed. Kanehara; 2018. Japanese. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kobayashi H, Mochizuki H, Kato T, Mori T, Kameoka S, Shirouzu K, et al. Outcomes of surgery alone for lower rectal cancer with and without pelvic sidewall dissection. Dis Colon Rectum. 2009;52:567–76. doi: 10.1007/DCR.0b013e3181a1d994. [DOI] [PubMed] [Google Scholar]
- 30.Yokoyama Y, Kawai K, Ishihara S. Essential pelvic anatomy for laparoscopic or robotic lateral lymph node dissection for rectal cancer. Operation. 2022;76:715–24. Japanese. [Google Scholar]
- 31.Adachi B. The arterial system of the Japanese. Vol. 1. Kyoto Imperial University; 1928. Japanese. [Google Scholar]
- 32.Ueda T, Koyama F, Nakagawa T, Nakamura S, Nishigori N, Inoue T, et al. Prophylactic laparoscopic lateral pelvic lymph node dissection for lower rectal cancer: remarking on the vesicohypogastric fascia. Gan To Kagaku Ryoho. 2014;41:1488–90. [PubMed] [Google Scholar]
- 33.Matsumoto A, Arita K. A technique of laparoscopic lateral pelvic lymph node dissection based on vesicohypogastric fascia and ureterohypogastric nerve fascia for advanced low rectal cancer. Surg Endosc. 2017;31:945–8. doi: 10.1007/s00464-016-5014-7. [DOI] [PubMed] [Google Scholar]
- 34.Fung TL, Tsukada Y, Ito M. Essential anatomy for total mesorectal excision and lateral lymph node dissection, in both trans-abdominal and trans-anal perspective. Surgeon. 2021;19:e462–74. doi: 10.1016/j.surge.2020.09.011. [DOI] [PubMed] [Google Scholar]
- 35.Occelli B, Narducci F, Hautefeuille J, Francke JP, Querleu D, Crépin G, et al. Anatomic study of arcus tendineus fasciae pelvis. Eur J Obstet Gynecol Reprod Biol. 2001;97:213–9. doi: 10.1016/s0301-2115(00)00527-3. [DOI] [PubMed] [Google Scholar]
- 36.Pit MJ, De Ruiter MC, Lycklama A Nijeholt AA, Marani E, Zwartendijk J. Anatomy of the arcus tendineus fasciae pelvis in females. Clin Anat. 2003;16:131–7. doi: 10.1002/ca.10102. [DOI] [PubMed] [Google Scholar]
- 37.Stelzner S, Holm T, Moran BJ, Heald RJ, Witzigmann H, Zorenkov D, et al. Deep pelvic anatomy revisited for a description of crucial steps in extralevator abdominoperineal excision for rectal cancer. Dis Colon Rectum. 2011;54:947–57. doi: 10.1097/DCR.0b013e31821c4bac. [DOI] [PubMed] [Google Scholar]
- 38.Kim NK. Anatomic basis of sharp pelvic dissection for curative resection of rectal cancer. Yonsei Med J. 2005;46:737–49. doi: 10.3349/ymj.2005.46.6.737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lin M, Chen W, Huang L, Ni J, Yin L. The anatomy of lateral ligament of the rectum and its role in total mesorectal excision. World J Surg. 2010;34:594–8. doi: 10.1007/s00268-009-0371-1. [DOI] [PubMed] [Google Scholar]
- 40.Ishii M, Shimizu A, Lefor AK, Kokado Y, Nishigori H, Noda Y. Reappraisal of the lateral rectal ligament: an anatomical study of total mesorectal excision with autonomic nerve preservation. Int J Colorectal Dis. 2018;33:763–9. doi: 10.1007/s00384-018-3010-1. [DOI] [PubMed] [Google Scholar]
- 41.Takahashi T, Ueno M, Azekura K, Ohta H. Lateral ligament: its anatomy and clinical importance. Semin Surg Oncol. 2000;19:386–95. doi: 10.1002/ssu.9. [DOI] [PubMed] [Google Scholar]
- 42.Costello AJ, Brooks M, Cole OJ. Anatomical studies of the neurovascular bundle and cavernosal nerves. BJU Int. 2004;94:1071–6. doi: 10.1111/j.1464-410X.2004.05106.x. [DOI] [PubMed] [Google Scholar]
- 43.Hojo D, Murono K, Nozawa H, Kawai K, Hata K, Tanaka T, et al. Utility of a three-dimensional printed pelvic model for lateral pelvic lymph node dissection. Int J Colorectal Dis. 2020;35:905–10. doi: 10.1007/s00384-020-03534-w. [DOI] [PubMed] [Google Scholar]
- 44.Sadakari Y, Hisano K, Sada M, Mizuuchi Y, Nagayoshi K, Fujita H, et al. Long-term effects of laparoscopic lateral pelvic lymph node dissection on urinary retention in rectal cancer. Surg Endosc. 2022;36:999–1007. doi: 10.1007/s00464-021-08364-7. [DOI] [PubMed] [Google Scholar]