Abstract
The population with dementia is expected to rise to 152 million in 2050 due to the aging population worldwide. Therefore, it is significant to identify and intervene in the early stage of dementia. The Rey-Osterreth complex figure (ROCF) test is a visuospatial test scale. Its scoring methods are numerous, time-consuming, and inconsistent, which is unsuitable for wide application as required by the high number of people at risk. Therefore, there is an urgent need for a rapid, objective, and sensitive digital scoring method to detect cognitive dysfunction in the early stage accurately. This study aims to clarify the organizational strategy of aMCI patients to draw complex figures through a multi-dimensional digital evaluation system. At the same time, a rapid, objective, and sensitive digital scoring method is established to replace traditional scoring. The data of 64 subjects (38 aMCI patients and 26 NC individuals) were analyzed in this study. All subjects completed the tablet's Geriatric Complex Figure (GCF) test, including copying, 3-min recall, and 20-min delayed recall, and also underwent a standardized neuropsychological test battery and classic ROCF test. Digital GCF (dGCF) variables and conventional GCF (cGCF) scores were input into the forward stepwise logistic regression model to construct classification models. Finally, ROC curves were made to visualize the difference in the diagnostic value of dGCF variables vs. cGCF scores in categorizing the diagnostic groups. In 20-min delayed recall, aMCI patients' time in air and pause time were longer than NC individuals. Patients with aMCI had more short strokes and poorer ability of detail integration (all p < 0.05). The diagnostic sensitivity of dGCF variables for aMCI patients was 89.47%, slightly higher than cGCF scores (sensitivity: 84.21%). The diagnostic accuracy of both was comparable (dGCF: 70.3%; cGCF: 73.4%). Moreover, combining dGCF variables and cGCF scores could significantly improve the diagnostic accuracy and specificity (accuracy: 78.1%, specificity: 84.62%). At the same time, we construct the regression equations of the two models. Our study shows that dGCF equipment can quantitatively evaluate drawing performance, and its performance is comparable to the time-consuming cGCF score. The regression equation of the model we constructed can well identify patients with aMCI in clinical application. We believe this new technique can be a highly effective screening tool for patients with MCI.
Subject terms: Health care, Computational biology and bioinformatics, Predictive markers, Alzheimer's disease, Neurology, Neurodegenerative diseases
Introduction
The population with dementia is expected to rise to 152 million in 2050 due to the aging population worldwide1. It brings enormous mental pressure and economic burden to caregivers, the medical industry, and society. Alzheimer's disease (AD) is the most common type of dementia, accounting for about 50–60% of the dementia population in the elderly2. Mild cognitive impairment (MCI) is an intermediate stage between normal cognitive aging and dementia due to AD3. Especially individuals diagnosed with amnesia MCI (aMCI), about 10–15% of them are converted to AD every year3. Therefore, it is of great significance to identify and intervene early.
The Rey–Osterrieth Complex Figure (ROCF) test is widely used to assess cognitive disorders' visuo-constructional ability and visual memory, including AD4, 5. At present, a variety of versions and scoring methods have been developed5. The neuropsychological dysfunction of a subject can be assessed by drawing performance, including attention and concentration, fine-motor coordination, visuospatial perception, non-verbal memory, planning and organization, and spatial orientation6, 7. Conventional scoring methods of ROCF focus on quantificationally scoring the final product of drawing by assessing its elements' shape and position accuracy8–10. Although several process scoring methods (e.g., Bennett-Levy11, Boston Qualitative Scoring System12, and Developmental Scoring System13) can quantify the drawing order and direction of elements to increase the evaluation of executive functions and organizational strategies, they are time-consuming, and poor evaluation consistency and expertise are needed5.
In recent years, digitizing traditional cognitive scales has become a hot spot in neuroscience14. Initially, researchers developed automatic scoring software for the ROCF test using artificial intelligence (e.g., computer vision technology15, Gaussian filter method16, and deep machine learning algorithm17, 18), which can almost achieve 94% consistency with human scores. However, neither of these studies evaluated the drawing order but focused on identifying outlines and details related to traditional scoring methods. Previously, only two studies used digital devices (such as a digital pen19 or tablet20, 21) to capture the drawing process and analyzed the subjects' drawing behavior patterns by extracting spatial, procedural, and kinematic dimension parameters. Poreh et al.19 used a digital pen to capture the drawing process and recorded the movement of the pen to the laptop through an infrared receiver. Thereby, it realized a semi-automatic analysis of the continuity and symmetry variables in Bennett-levy scoring. Also, Kim et al.20used a tablet (Samsung Galaxy Book) to record the drawing process and automatically extract stroke parameters (e.g., time, speed, and length) and graphical space information (e.g., position of center and mass). It also used 2D technology to analyze the shape similarity between the original and copied figure. The results showed that AD patients copied the figure more fragmentedly with a longer pause and were more inclined to move the figure closer to the target image with lower accuracy than NC individuals (i.e., individuals with normal cognition). Late-onset AD showed signs of leftward deviation in space utilization.
It is well known that the digital parameters of the clock drawing test have successfully proved the cognitive process, and its high sensitivity and specificity in distinguishing patients with aMCI or mild AD from NC individuals are remarkable22–26. The behavioral pattern of drawing complex figures in aMCI patients has not yet been explored. And whether the kinematic parameters during the drawing process can distinguish aMCI patients from NC individuals for early cognitive impairment screening.
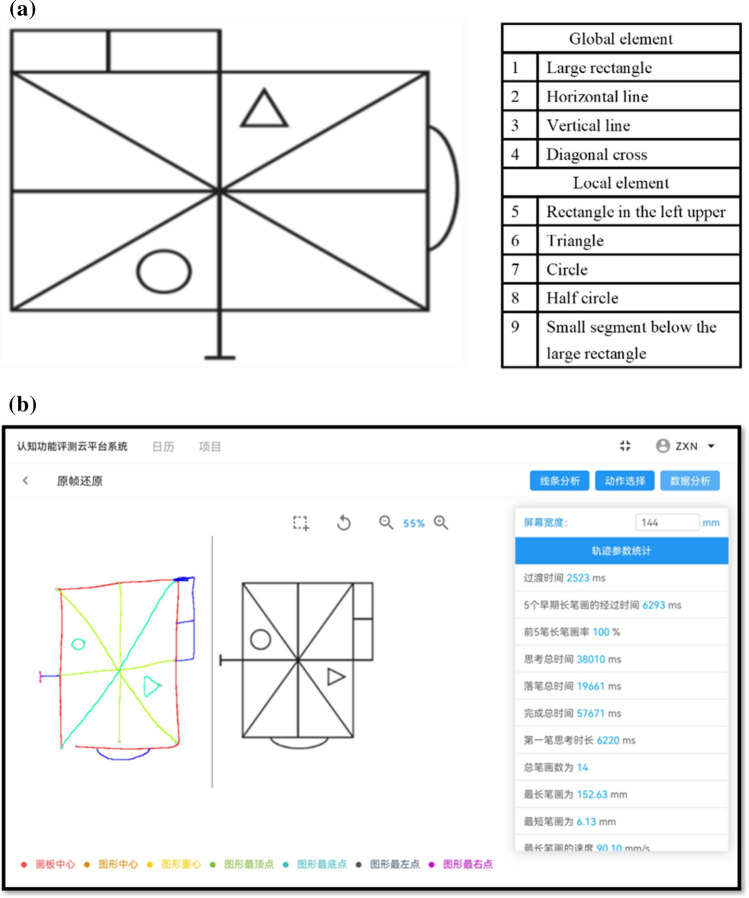
This study explored the drawing characteristics of aMCI patients in the copying, 3-min recall, and 20-min delayed recall of the complex figure test. Compared with traditional scores, the diagnostic value of digitized variables in distinguishing aMCI patients from NC individuals. To realize this scheme, we chose a simplified ROCF—Geriatric Complex Figure (GCF) (Fig. 1a) developed by Poreh27in 2002. It is an organizational strategy model based on the classic ROCF test. Its copying, recall, and strategy scores are well-distributed among healthy older people (over 60 years old), indicating that it is more suitable for the elderly than the ROCF19. Using the simplified figure can avoid the overlap of strokes, which is conducive to recognizing each stroke with digital equipment in a limited space. It can also improve the subjects' cooperation and enthusiasm.
Figure 1.
Digital Geriatric Complex Figure (dGCF) Test. (a) The GCF is a simplified organizational strategy model based on the classic ROCF test. It consists of four global and five local elements. (b) dGCF software consists of three modules: drawing area, line selection, and data analysis (size: 10.8-inch; resolution: 2560 × 1600; Huawei Tablet M6). The upper part of the tablet is defined as the display area, and the lower part is defined as the drawing area.
This study aims to clarify the organizational strategy of aMCI patients to draw complex figures through a multi-dimensional digital evaluation system. At the same time, a rapid, objective, and sensitive digital scoring method is established to replace traditional scoring.
Materials and methods
Participants
Participants were recruited from the memory or neurology clinic of the First Hospital of Shanxi Medical University from November 2020 to November 2021. The study group included 38 patients with aMCI and 26 NC individuals with junior high school education or above (> 6 years). They all had normal vision and hearing and could complete neuropsychological assessment and drawing tasks. All participants underwent a rigorous evaluation, including a standardized neuropsychological battery, structural magnetic resonance imaging (MRI) of the brain, blood tests, and neurologist diagnosis. Patients with aMCI were enrolled in the study according to the National Institute on Aging and the Alzheimer's Association (NIAA) clinical MCI criteria in 201128. Its core criteria include (1) memory decline for at least six months (confirmed by informants); (2) MMSE score ≥ 24, with objective evidence of impairment in one or more cognitive domains (scores adjusted for age and education were lower than 1.5SD in healthy elderly); (3) maintain the independent activities of daily living; (4) CDR score was 0.5. NC individuals should meet the following criteria: (1) no episodic memory impairment or objective evidence; (2) MMSE score ≥ 26, CDR score was 0, and intact activities of daily living. Exclusion criteria: (1) We excluded patients diagnosed with dementia (CDR > 0.5). (2) Patients who have suffered from cerebrovascular stroke have apparent symptoms or signs of neurological impairment at the onset of the disease. Cerebral structure MRI showed the corresponding responsible lesions. (3) Cerebral MRI indicated severe leukoencephalopathy (Faskass score ≥ 3). (4) Alcoholism, drug addiction, brain trauma, epilepsy, encephalitis, normal cranial pressure hydrocephalus, and other neurological diseases that can cause cognitive impairment are also excluded. (5) Systemic diseases that may lead to MCI, such as liver and kidney insufficiency, endocrine diseases, vitamin deficiency, etc. (6) Suffering from anxiety, depression, or schizophrenia.
The First Hospital of Shanxi Medical University Ethics Committee approved the study. All methods of this study were carried out in strict accordance with the Declaration of Helsinki. All subjects or their legal guardians signed informed consent after knowing the details of the study.
Neuropsychological assessments
All subjects received a standardized neuropsychological test battery, including memory, language, attention, visuospatial, and executive function tests. Tests of interest were selected from each cognitive domain. All tests were converted to standard z-scores using published criteria, and then composite scores for each cognitive domain were created by calculating the average of each category. Mini-Mental State Examination (MMSE, Chinese version)29, 30, Montreal Cognitive Assessment Scale (MoCA, Chinese version)31, 32, and Clinical Dementia Rating (CDR)33were used to evaluate the general cognitive level.
Memory—Immediate and delayed recall of Auditory Verbal Learning Test (AVLT, Chinese version)34, 35 and ROCF test8. We use the 15-word Chinese version of AVLT. It is divided into five categories, each containing three words. The subjects were asked to learn three times and recall after each repetition. The presentation of words is random and does not follow semantic categories. After a 20-min interval, the subjects were asked to perform long-delayed recall, cue recall, and recognition tests. During the 20-min break, the subjects performed other non-verbal tasks. The 18 units in the ROCF test were scored separately (Osterrich scoring8) regarding accuracy and location. Each unit can be rewarded with 0–2 points; the raw score was 0–32.
Language—The Boston Naming Test (BNT, 30-item version)36, 37and Verbal Fluency Test (Animal, VFT)38. In the Chinese version of the BNT test, subjects were asked to name 30 pictures without a time limit, with a total score of 30. The VFT paradigm requires subjects to list as many animals as possible in one minute. We recorded the total number of animals.
Attention—Forward and Backward digit span test (DST)39. The evaluator reads some numbers in this test, and the subject listens carefully. When they finished reading, the subjects were asked to recite it similarly or backward. We recorded the number of correct strings completed.
Visuospatial function—Judgment of Line Orientation (JLO, 35-item version)40 and copying of ROCF test. The JLO scale consists of 35 cards, the first 5 for practice and the last 30 for testing. The reference card consists of 11 segments arranged in a fan pattern. The test card consists of two line segments of different lengths and directions. We recorded the number of correct lines completed, with a maximum score of 30.
Executive function—Trial Making Test (TMT, Chinese version) parts A and B41, 42and Stroop's color word reading test (SCWT, Chinese version)43, 44. In TMT-A, the subjects are asked to connect a sequence of Arabic numerals (1–25). The TMT-B contains white-circled Arabic numerals (1–13) and black-circled Arabic numerals (1–12). The subjects were asked to connect Arabic numerals in alternating order of white circles followed by black circles. We recorded the time it took to complete the test. The SCWT contains 3 subtests. The SCWT-A consists of a page of 100 color words (red, green, blue) in black font. The SCWT-B consists of a page of 100 "X" symbols in red, green, and blue font. The SCWT-C is a page of 100 words that use the word of the A subtest and the color of the B subtest (color and word do not match). The subjects read column by column as fast as they could. We recorded the number of correct words completed within 45 s of each paradigm.
Digital geriatric complex figure test
All subjects had to complete the GCF test on the tablet (Fig. 1b), including copying, 3-min recall, and 20-min delayed recall. We define the upper part of the tablet as the display area (rendering GCF) and the lower part as the drawing area. The subjects must use a digital pen to draw in the drawing area. After 3 min and 20 min, they were asked to draw again. The total time of each drawing should be within 10 min. The trace of the figure was recorded on the tablet at a sampling frequency of 60 Hz (size: 10.8-inch; resolution: 2560 × 1600; Huawei Tablet M6).
Digital GCF equipment can extract the number, length, speed, and interval time of strokes in the drawing process and reproduce the drawing track. In addition to the element latency time, which requires the analyst to outline the first stroke of each local element manually, other parameters can be automatically analyzed by the digital equipment. The dGCF variable is defined as follows:
Transition time—the time to start drawing long strokes after short strokes in all short-long stroke sequences, s
Elapsed time of 5 early long strokes—total time of drawing 5 early long strokes, s.
First 5 stroke ratios—the proportion of long strokes in the first 5 strokes, %.
Speed of the longest stroke—the average speed of the longest stroke, cm/s.
Time in air—total time from one stroke to the next stroke, s.
Total time—the sum of time in air and time on the surface, s.
First stroke latency—time in air before drawing the first stroke, s.
Average element latency—average value of thinking time before drawing the first stroke of all local elements.
Whole area—the minimum circumscribed rectangular area of the figure, cm2.
In addition, the 9 units in GCF test were scored separately in terms of accuracy and location, which was consistent with Osterrich's standardized score on the ROCF test8. The raw score was 0–16. To avoid the learning effect between ROCF and GCF tests, we divided them into two times with an interval of one day.
Data analysis
All statistical analyses were processed using SPSS (version 26) and GraphPad Prism (version 9) software, and a p value < 0.05 was considered significant. The independent sample t-test was applied to assess the statistical significance between groups for age and education level because they follow a normal distribution. The Chi-Square test was used for group differences in gender distribution. Group differences in cognitive characteristics and digital parameters were examined running the nonparametric Mann–Whitney U test because they did not follow a normal distribution.
To verify the validity of GCF data, we used Pearson correlation to compare classical ROCF with GCF test total scores. The k-means clustering was used to analyze the cut-off value of long/short strokes in dGCF software. After adjusting for age, sex, and education level, we used partial correlation to compare the dGCF parameter with the cognitive domain composite scores to determine whether the digital parameter represents a specific cognitive impairment. We chose dGCF parameters with significant differences between groups in 20-min delayed recall and cGCF scores as continuous independent variables and aMCI against NCs as dichotomous dependent variables. A combination of dGCF parameters and cGCF scores was entered into the logistic regression model using a positive stepwise inclusion model.
Finally, we calculated the Receiver Operating characteristics (ROC) curves and the area under the curves (AUC) of the logistic models to compare the diagnostic value of different models (dGCF vs. cGCF scores vs. d + cGCF) for patients in distinguishing and NC individuals.
Results
Demographic characteristics and neuropsychological tests between NC and aMCI groups
There was no significant difference in age, sex distribution, and education level between NC individuals and aMCI patients (p > 0.05). The MMSE (p = 0.001) and MOCA (p < 0.001) scores of aMCI patients were higher than those of NC individuals. ADL scores were not significantly different between groups (p = 0.06) (Table 1).
Table 1.
Demographic characteristics and neuropsychological tests between NC and aMCI groups.
| NC(n = 26) | aMCI(n = 38) | p value | |
|---|---|---|---|
| Age in years | 64.6 ± 7.0 | 67.5 ± 7.2 | 0.115 |
| Gender (Male/female) | 11/15 | 15/23 | 0.693 |
| Years of education | 11.7 ± 2.3 | 11.1 ± 3.3 | 0.456 |
| MMSE | 28(27,29) | 27(25,28) | 0.001 |
| MoCA | 25(23,26) | 21(19,23) | < 0.001 |
| ADL | 21(20,22) | 21(20,23) | 0.06 |
| Memory | |||
| AVLT immediate recall | 25.0(22.0,30.3) | 20.5(17.0,27.0) | 0.003 |
| AVLT delayed recall | 10.0(8.8,11.0) | 5.0(2.3,8.0) | < 0.001 |
| AVLT recognition | 13(12,14) | 9(7,11) | < 0.001 |
| ROCF immediate recall | 21.5(18.0,25.3) | 17.0(12.0,21.3) | 0.003 |
| ROCF delayed recall | 20.8(17.5,24.3) | 17.3(11.9,19.5) | 0.002 |
| Language | |||
| BNT | 27(24,28) | 24(22,26) | 0.001 |
| VFT animal | 20.0(17.8,22.3) | 15.0(13.0,19.0) | < 0.001 |
| Attention | |||
| Forward digit span | 8.0(6.8,9.0) | 8.0(6.8,8.0) | 0.262 |
| Backward digit span | 5(4,6) | 4(3,5) | 0.031 |
| Visuospatial function | |||
| ROCF copying | 33.0(31.0,34.3) | 32.0(29.4,33.3) | 0.054 |
| JLO (30 items) | 24.5(23.4,26.8) | 25.3(22.9,26.5) | 0.902 |
| Executive function | |||
| TMT-A | 36.0(29.8,44.5) | 47.0(39.5,57.3) | 0.002 |
| TMT-B | 60.0(46.3,79.3) | 83.0(62.8,104.3) | 0.002 |
| Stroop word | 80.5(69.0,88.0) | 67.5(60.8,81.0) | 0.021 |
| Stroop color | 63.0(55.0,70.0) | 52.0(42.0,62.3) | 0.001 |
| Stroop word/color | 31.0(27.8,34.3) | 27.0(20.0,31.0) | 0.003 |
| GCF copying | 17.0(16.0,17.3) | 16.0(15.0,17.0) | 0.134 |
| GCF 3-min recall | 16.0(15.0,16.6) | 12.8(10.4,15.0) | < 0.001 |
| GCF 20-min delayed recall | 16(14,17) | 13(10,15) | < 0.001 |
Values are expressed as mean ± standard deviation or median (interquartile range).
n number; NC Normal control individuals; aMCI Amnestic mild cognitive impairment; MMSE Mini-mental state examination; MoCA Montreal cognitive assessment; ADL Activity of daily living scale; AVLT Auditory verbal learning test; ROCF Rey-Osterrieth complex figure; BNT Boston naming test; VFT Verbal fluency test; JLO Judgement of line orientation (30 items in total); TMT Trail making test (part A or B, time in seconds); GCF Geriatric complex figure; Stroop word/color test, number of words completed in 45 s.
Compared with NC individuals, aMCI patients performed worse in memory, language, attention, and executive function, and the scores on these scales were significantly different between groups (all p < 0.05). NC individuals and aMCI patients did not significantly differ in visuospatial scores (ROCF copying: p = 0.054; JLO: p = 0.902) (Table 1).
Pearson correlation between GCF and classic ROCF test total scores
There was no significant difference between groups in GCF and classical ROCF copying test scores (all p > 0.05). In GCF (all p < 0.001) and ROCF (all p < 0.01) 3 min recall and 20 min delayed recall tests, the scores of aMCI patients were significantly lower than NC individuals (Table 1). Therefore, we took the average scores of copying, 3-min recall, and 20-min delayed recall as the total score of each test. The total score of the GCF test was significantly correlated with the classical ROCF test (r = 0.598, p < 0.001; Fig. 2), indicating that they were comparable in assessing the visuo-constructional ability and visual memory of the elderly.
Figure 2.
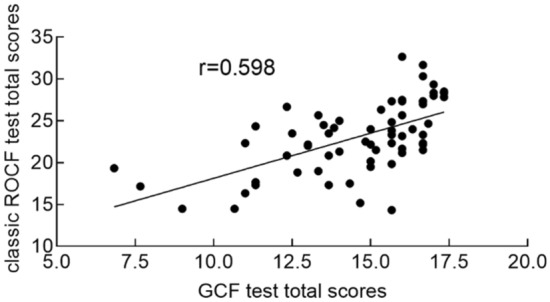
Pearson correlation between GCF and classic ROCF test total scores. All subjects completed both tests, including copying, 3-min recall, and 20-min delayed recall. We manually scored each GCF or ROCF test unit in terms of accuracy and location using Osterrich's scoring. There was a significant correlation between GCF and classical ROCF test total scores (r = 0.598; p < 0.001).
Performance characteristics of digital GCF test in patients with aMCI and NC individuals
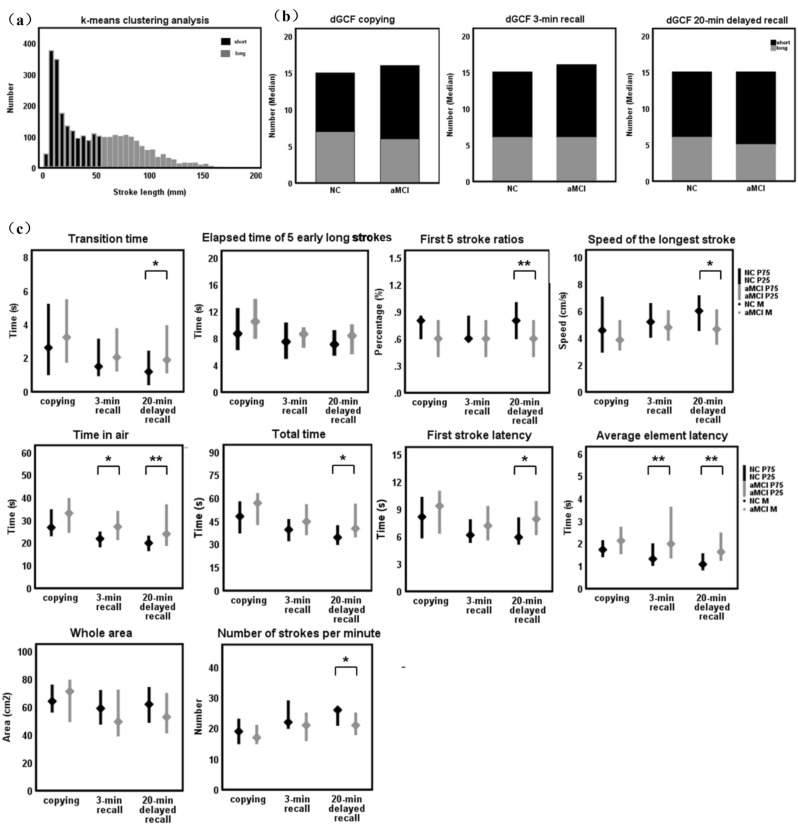
We included the total number of copying, 3-min recall, and 20-min delayed recall test lines for long/short stroke classification analysis using the k-means clustering method, with a cut-off value of 55.72 mm (Fig. 3a). In 20-min delayed recall, the number of short strokes of aMCI patients was significantly higher than NC individuals (p = 0.38; Fig. 3b). However, the number of long/short strokes differed significantly between groups in copying and 3-min recall (all p > 0.05; Fig. 3b).
Figure 3.
Pen stroke analysis module of dGCF software. Extract the number, length, speed, and interval time of strokes in the drawing process, and then obtain dGCF parameters through automatic or semi-automatic analysis. (a) short (black)/Long (grey) strokes were classified by k-means clustering with a cut-off value of 55.72 mm. (b) Difference analysis of long (grey), short (black) strokes, and total strokes between NC and aMCI groups in copying, 3-min recall, and 20-min delayed recall. (c) Lateral comparison and longitudinal trend analysis of dGCF parameters between NC (black) and aMCI (grey) groups.*p < 0.05, **p < 0.01. dGCF, digital Geriatric Complex Figure.
Analysis of dGCF variables between the groups found no significant difference in the copying test (all p > 0.05). In 3-min recall, the time in air and average element latency of aMCI patients were significantly longer than that of NC individuals (all p < 0.05). And in 20-min delayed recall, we found a significant difference in transition time, first 5 stroke ratios, speed of the longest stroke, time in air, total time, first stroke latency, average element latency, and strokes per minute (all p < 0.05). The whole area and elapsed time of 5 early long strokes were not different between patients with aMCI and NC individuals (all p > 0.05) (Fig. 3c; Table 2).
Table 2.
Comparison of dGCF parameters between aMCI patients and NC individuals in copying, 3-min recall, and 20-min delayed recall.
| Digital parameters | NC(n = 26) copying/3-min recall/20-min delayed recall | aMCI(n = 38) copying/3-min recall/20-min delayed recall | p value |
|---|---|---|---|
| Transition time (s) | 2.62(1.02,5.20) | 3.24(1.75,5.47) | 0.385 |
| 1.50(0.95,3.13) | 2.05(1.23,3.75) | 0.187 | |
| 1.19(0.42,2.41) | 1.89(1.12,3.93) | 0.043 | |
| Elapsed time of 5 early long strokes (s) | 8.69(6.39,12.37) | 10.49(8.07,13.72) | 0.199 |
| 7.49(5.07,10.22) | 8.63(6.73,9.51) | 0.623 | |
| 7.10(5.56,9.09) | 8.39(5.77,9.95) | 0.305 | |
| First 5 stroke ratios (%) | 0.8(0.6,0.85) | 0.6(0.4,0.8) | 0.122 |
| 0.6(0.6,0.85) | 0.6(0.4,0.8) | 0.20 | |
| 0.8(0.6,1) | 0.6(0.4,0.8) | 0.009 | |
| Speed of the longest stroke (cm/s) | 4.56(2.94,7.03) | 3.85(3.10,5.28) | 0.286 |
| 5.19(4.05,6.55) | 4.78(3.84,6.02) | 0.712 | |
| 6.00(4.54,7.13) | 4.65(3.53,6.07) | 0.039 | |
| Average drawing speed (cm/s) | 4.17(3.04,4.62) | 3.66(3.12,4.40) | 0.268 |
| 4.11(3.50,5.35) | 3.89(3.25,4.99) | 0.412 | |
| 4.67(3.56,5.81) | 3.95(3.37,4.59) | 0.101 | |
| Time in air (s) | 26.83(23.15,34.67) | 33.14(24.69,39.65) | 0.085 |
| 21.85(18.26,24.81) | 27.15(21.49,33.94) | 0.011 | |
| 19.93(16.59,23.00) | 23.94(18.86,36.87) | 0.006 | |
| Total time (s) | 48.22(37.47,57.72) | 56.88(42.87,63.08) | 0.107 |
| 39.61(32.37,46.18) | 44.87(36.30,55.79) | 0.065 | |
| 34.52(29.96,42.15) | 40.30(34.83,56.21) | 0.01 | |
| First stroke latency (s) | 8.15(5.85,10.3) | 9.35(6.35,10.95) | 0.28 |
| 6.16(5.35,7.83) | 7.17(5.62,9.29) | 0.144 | |
| 5.92(5.16,8.04) | 7.92(6.20,9.83) | 0.018 | |
| Average element latency (s) | 1.73(1.42,2.13) | 2.13(1.55,2.73) | 0.08 |
| 1.32(1.03,1.99) | 1.99(1.36,3.62) | 0.007 | |
| 1.08(0.83,1.55) | 1.63(1.25,2.47) | 0.002 | |
| Whole area (cm2) | 64.18(56.45,75.76) | 71.27(49.63,79,43) | 0.444 |
| 59.03(47.77,71.89) | 49.49(39.31,72.20) | 0.234 | |
| 61.98(49.06,74.04) | 52.86(41.44,69.73) | 0.204 | |
| Strokes per minute (n) | 19(15,23) | 17(15,21) | 0.32 |
| 22(20,29) | 21(16,25) | 0.173 | |
| 26(21,27) | 21(18,25) | 0.006 |
Significant values are in bold.
Values are expressed as median (interquartile range).
n Number; NC Nomal control individuals; aMCI Amnestic mild cognitive impairment; s Second; cm2 Square centimeter; dGCF Digital geriatric complex figure.
In the longitudinal analysis, we found that with the extension of time, although the drawing performance of the two groups tended to improve, the aMCI patients still performed worse than the NC individuals after 20 min (Fig. 3c).
Partial correlation analysis between dGCF parameters and cognitive domain composites
We compared the correlation between dGCF parameters and cognitive domains in 20-min delayed recall. After adjusting for age, sex, and education level, the transition time was negatively correlated with attention (r = − 0.292, p = 0.019) and executive function (r = − 0.275, p = 0.032). The speed of the longest stroke was positively correlated with executive function (r = 0.302, p = 0.015). The time in air was moderately negatively correlated with attention (r = − 0.408, p = 0.001) and executive function (r = − 0.448, p < 0.001). Also, the total time was negatively correlated with attention (r = − 0.342, p = 0.007) and executive function (r = − 0.429, p = 0.001). The first stroke latency was negatively correlated with memory (r = -0.365, p = 0.004) and visuospatial function (r = − 0.407, p = 0.001). The average element latency was moderately negatively correlated with memory (r = − 0.377, p = 0.003) and attention (r = − 0.33, p = 0.009). The number of strokes per minute was positively correlated with multiple cognitive domains (all p < 0.05) (Table 3).
Table 3.
Partial correlation analysis between dGCF parameters and cognitive domain composites.
| Cognitive domain (z-score) | Transition time p | First 5 stroke ratios p | Speed of the longest stroke p | Time in air p | Total time p | First stroke latency p | Average element latency p | Strokes per minute p |
|---|---|---|---|---|---|---|---|---|
| Memory | − 0.133 | 0.11 | − 0.018 | − 0.26 | − 0.181 | − 0.365 | − 0.377 | 0.326 |
| 0.307 | 0.397 | 0.891 | 0.043 | 0.163 | 0.004 | 0.003 | 0.01 | |
| Language | − 0.148 | 0.242 | 0.07 | − 0.22 | − 0.12 | − 0.199 | − 0.17 | 0.126 |
| 0.254 | 0.06 | 0.59 | 0.089 | 0.358 | 0.115 | 0.191 | 0.335 | |
| Attention | − 0.292 | 0.095 | 0.081 | − 0.408 | − 0.342 | − 0.183 | − 0.33 | 0.311 |
| 0.019 | 0.469 | 0.532 | 0.001 | 0.007 | 0.159 | 0.009 | 0.015 | |
| Executive function | − 0.275 | 0.217 | 0.302 | − 0.448 | − 0.429 | − 0.23 | − 0.217 | 0.376 |
| 0.032 | 0.093 | 0.015 | < 0.001 | 0.001 | 0.074 | 0.093 | 0.003 | |
| Visuospatial function | − 0.226 | 0.17 | 0.133 | − 0.264 | − 0.273 | − 0.407 | − 0.166 | 0.263 |
| 0.08 | 0.189 | 0.307 | 0.04 | 0.033 | 0.001 | 0.202 | 0.04 |
Significant values are in bold.
z-score, this study selected neuropsychological tests of interest in each cognitive domain. All tests were converted to standard z-scores using published criteria, and then composite scores for each cognitive domain were created by averaging each category.
Diagnostic value of dGCF parameters and cGCF scores in discriminating patients with aMCI from NC individuals
In the 20-min delayed recall test, we selected the transition time, first 5 stroke ratios, speed of the longest stroke, time in air, total time, first stroke latency, average element latency, and strokes per minute into the positive stepwise logistic regression model. The results showed that the significant odds ratios (95% confidence interval) of the average element latency (3.776[1.322–10.785]) and first 5 stroke ratios (0.038[0.003–0.559]) were the best predictor variable (all p < 0.05; Table 4), and other variables were excluded.
Table 4.
Logistic regression models of dGCF parameters and cGCF scores for discriminate patients with aMCI from NC individuals.
| Model | Selected variable | Beta (β) | Sβ | SE | WaldX2 | OR (95%CI) | p value |
|---|---|---|---|---|---|---|---|
| a. dGCF | Average element latency | 1.329 | 0.494 | 0.535 | 6.156 | 3.776 (1.322–10.785) | 0.013 |
| First 5 stroke ratios | − 3.27 | − 0.287 | 1.372 | 5.682 | 0.038 (0.003–0.559) | 0.017 | |
| b. cGCF | cGCF score | − 0.408 | − 0.424 | 0.128 | 10.163 | 0.665(0.518–0.855) | 0.001 |
| c. (d + c) GCF | Average element latency | 1.396 | 0.832 | 0.602 | 5.374 | 4.037 (1.241–13.138) | 0.022 |
| First 5 stroke ratios | − 3.274 | − 0.460 | 1.72 | 3.623 | 0.038 (0.001–1.00) | 0.021 | |
| cGCF score | − 0.392 | − 0.691 | 0.145 | 7.294 | 0.676 (0.508–0.898) | 0.007 |
NC Normal control individuals; aMCI Amnestic mild cognitive impairment; dGCF Digital geriatric complex figure; cGCF Conventional geriatric complex figure; β Logistic regression coefficient; Sβ standard logistic regression coefficient; SE Standard error; OR Odds ratio; CI Confidence interval of to odds ratio.
Predicting patients with aMCI and NC individuals, the combination of average element latency and first 5 stroke ratios (Table 4, model a) correctly classified 70.3% of cases (AUC:0.772; p < 0.001; Table 5; Fig. 4), with a sensitivity of 89.47% and a specificity of 53.85%, and an optimal cut-off of 0.43. when cGCF score (Table 4, model b) was used to discriminate between aMCI patients and NC individuals at a cut-off value of 15.5, showing a sensitivity of 84.21%, a specificity of 57.69%, and an accuracy of 73.4%(AUC:0.773; p < 0.001; Table 5; Fig. 4). Finally, we found that the combination of average element latency, first 5 stroke ratios, and cGCF score (Table 4, model c) can significantly improve the specificity of diagnosis. 78.1% (AUC:0.852; p < 0.0001; Table 5; Fig. 4) of aMCI patients and NC individuals were correctly classified, with a sensitivity of 78.95% and a specificity of 84.62%. The best cut-off value is 0.59.
Table 5.
Diagnostic value of classification models in discriminating patients with aMCI from NC individuals.
| Model | ROC AUC | Sensitivity | Specificity | Accuracy | p value |
|---|---|---|---|---|---|
| a. dGCF | 0.772 | 89.47 | 53.85 | 70.3 | < 0.001 |
| b. cGCF | 0.773 | 84.21 | 57.69 | 73.4 | < 0.001 |
| c. (d + c) GCF | 0.852 | 78.95 | 84.62 | 78.1 | < 0.0001 |
ROC Receiver operating characteristics curve; AUC Area under the cure.
Figure 4.
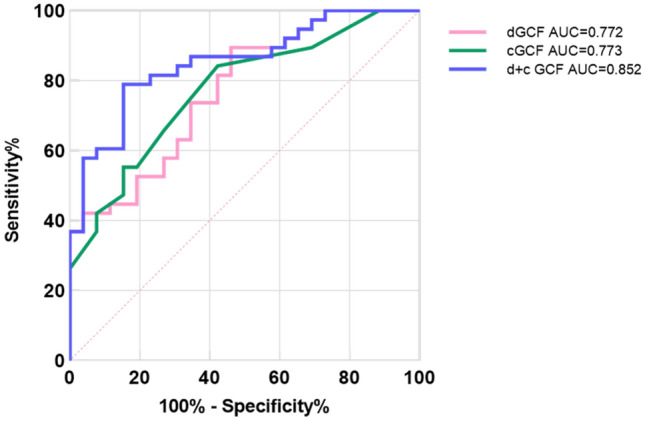
AUCs for aMCI patients against NCs using dGCF model (average element latency and first 5 stroke ratios), cGCF score, or (d + c) model.
Discussion
This study uses a digital pen and tablet to extract the multi-dimensional kinematic parameters involved in the drawing process, aiming to clarify the organization strategy of drawing complex figures for aMCI patients and establish a rapid, objective, and sensitive digital scoring system that could replace traditional scoring. First, we compared the GCF test total score with the classical ROCF to verify the validity of the GCF data in the Chinese population. Subsequently, we performed a horizontal comparison and longitudinal analysis of dGCF parameters in aMCI patients and NC individuals. The final aim of the study was to evaluate the potential of dGCF variables in differentiating aMCI patients from NC individuals compared to cGCF scores.
A previous study reported that GCF test scores were significantly correlated with RBANS' complex figure copying and delayed recall scores19. And its copying, recall, and strategy scores are well-distributed among healthy older people (over 60 years old). In line with this study, the GCF test total score is strongly correlated with the classical ROCF test, indicating that they are comparable in assessing the visuo-constructional ability and visual memory of the elderly. Other studies have shown that the simplified figures can also detect significant heterogeneity in patients of MCI, AD, and NC individuals20, 45–47. In addition, a study found significant outliers in the ROCF copy scores of AD patients, which may be related to decreased enthusiasm and coordination when encountering a complex figure due to severe visuospatial impairment20. These findings mean that the simplified figure may have a better application prospect for the low education level, the elderly, and the digital transformation of the scale.
We found that the 3-min and 20-min delayed recall scores of ROCF and GCF tests in aMCI patients were significantly lower than those of NC individuals, so we further analyzed the digital variables of 3-min and 20-min recall between the two groups.
The results showed that the time in air and average element latency of aMCI patients were significantly longer than that of NC individuals in 3-min recall. In 20-min recall, the transition time, time in air, total time, first stroke latency, and average element latency of aMCI patients were significantly longer than those of NC individuals. The first 5 stroke ratios, speed of the longest stroke, and strokes per minute were lower than those of NC individuals. And aMCI patients used more short strokes in drawing than NC individuals. It suggests that poor drawing performance of aMCI patients may be related to abnormal organizational strategies. If the entire process of the drawing task is digitally captured, it may reveal subtle signs of cognitive impairment.
GCF consists of the main frame and five local elements. Wilson and Batchelor48 found that more than 50% of NC individuals draw the main frame first and add local elements later. Drawing in a fragmented and chaotic way may suggest damage to the prefrontal cortex, reflecting that individuals cannot effectively integrate global information. The drawing of the main frame requires at least 5 strokes (rectangle, horizontal/vertical/diagonal lines). Since the longest stroke is likely to be used to build a graphic frame, the slowness of drawing the longest stroke may imply impaired executive function and visuo-constructional ability. The increase in the transition time or the decrease in the first 5 stroke ratio also suggests that patients with aMCI begin to draw local features or details earlier, which can also reflect the impairment of executive function and attention. In the early AD stage, memory retrieval impairment leads to the decline of motor execution and motor integration, called executive dysfunction syndrome49. Some studies also tend to attribute the preferential extraction of local features in AD patients to mild simulated agnosia, which is part of Balint syndrome50. In addition, we found that patients with aMCI were hesitant at the beginning of drawing or adding local elements and even missed some critical structures. Therefore, the increase in the first stroke latency and average element latency may indicate the impairment of visuospatial working memory and attention. All time variables reflect the intermittence in the process of drawing or execution. Especially after 20 min, it needs to be converted into motor skills and execution plans through visuospatial working memory51. Our cognitive domain analysis of digital variables also suggests that the first stroke latency and average element latency are significantly related to memory and attention. It may be related to the disorders of the frontal lobe and temporal-parietal lobe brain regions, which will affect the visuospatial working memory, decision-making, and cognitive flexibility, and then interfere with the accuracy of drawing results24, 52, 53. These findings suggest that the poor drawing performance in 3-min recall in aMCI patients may be related to impaired visuospatial memory, and the drawing strategy is acceptable. However, at an interval of 20 min, strategies for constructing visual components of aMCI patients were also significantly affected, resulting in the inaccuracy of the finished product map.
Using the combination model of the first 5 stroke ratios and average element latency can better distinguish aMCI patients and NC individuals (accuracy: 70.3%), which is equivalent to the time-consuming cGCF score (accuracy: 73.4%), and its sensitivity is slightly higher than the cGCF score (dGCF: 89.47%, cGCF: 84.21%). Therefore, we constructed the regression equation of the above model: ln (p/(1-p)) = 0.536 + 1.329 * x1-3.27 * x2 (p: probability of illness, x1: average element latency, x2: first 5 stroke ratios), in which x1 is the most critical variable (standardized beta: x1 = 0.494, x2 = − 0.287). It can be found that the variables reflecting visuospatial working memory and visual component construction strategies show the best model fitting. Moreover, the combination model of the dGCF variables and cGCF score can significantly improve the accuracy and specificity of distinguishing aMCI patients from NC individuals (accuracy: 78.1%, specificity: 84.62%). The regression equation of this model is ln (p/(1-p)) = 6.521 + 1.396 * x1-3.724 * x2-0.392 * x3 (p: probability, x1: average element latency, x2: first 5 stroke ratios, x3: cGCF score), in which x1 is the most critical variable (standardized beta: x1 = 0.832, x2 = -0.46, x3 = -0.691). The above two regression equations can be used to identify patients with MCI in clinical application. Although the efficiency of the pure digital variable model to identify patients with aMCI is slightly low, its application prospect is still worth looking forward to. We will develop more digital variables related to visuospatial ability in the future, and multivariate combinations may optimize the model.
In previous digital clock drawing test (dCDT) studies, Yuan et al. 54 found that acquired dCDT features correlated with brain volume. Moreover, the combined model of clinical risk factors, dCDT composite scores, and MRI measures can distinguish the patients with MCI well from the normal cognitive individuals, and the AUC is 0.897. Another study from Harvard University's Aging Brain Institute found among normal participants with biomarkers, the dCDT summary score was associated with more significant amyloid and tau burden and showed better discrimination between Aβ ± groups than the Primary Alzheimer Cognitive Composite55. Therefore, digital measurement may be an effective tool for detecting early cognitive changes in AD trajectories. GCF is similar to a clock. We will further validate the potential of dGCF as a biomarker for patients with MCI by combining neuroimaging, body fluid, and PET-CT biomarkers.
This study also has some potential limitations. First, the relatively small sample size of subjects may lead to the selection bias of digital variables and slightly reduce the efficiency of the diagnostic model. To overcome this difficulty, all subjects recruited in this study received brain imaging and a standardized neuropsychological battery and were rigorously diagnosed by professional neurologists. Second, as a newly developed cognitive assessment tool, it has not been evaluated for reliability and validity in large populations, such as retesting and internal consistency. However, similar studies have been published in recent years. We also plan to use this tool in community screening in China to verify its reliability, validity, and clinical applicability. Finally, the standardized neuropsychological battery took a long time, which decreased the subjects' enthusiasm and cooperation in the test. Especially in the 20-min recall test, they showed impatience or slightly refused, affecting the final drawing results. However, we still found that the simplified figure is more accessible to implement than the ROCF.
In conclusion, dGCF can conduct a multi-dimensional evaluation after digitizing the scale by extracting dynamic parameters such as the number, length, speed, and time of strokes to better analyze the subjects' drawing behavior patterns. As a supplement to traditional scoring, digital puls conventional can significantly improve the discrimination of aMCI patients.
We believe that this highly effective cognitive screening tool can be used to identify people at high risk of dementia (supplementary information).
Supplementary Information
Acknowledgements
Thank you for all the authors' efforts for this manuscript.
Author contributions
X.Z. and Y.L. had the idea for this manuscript. X.Z. provided a research scheme design and wrote the first manuscript. H.Z. and L.L. provided statistical analysis support and prepared all figures and tables. J.S. and J.C. recruited the subjects, evaluated them with the scale, and collected clinical data. Y.L. and H.Z. guided the research plan and provided fund support. All authors reviewed the manuscript.
Funding
This study received assistance from the Central Guide Local Science and Technology Development Special Fund Project (project number: YDZX20191400002563) and Shanxi Provincial Medical Key Project (project number: 2020XM22).
Data availability
The datasets generated during and analyzed during the current study are available from the corresponding author upon reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Hui Zhang, Email: zhanghui_mr@163.com.
Yang Li, Email: 15035182003@163.com.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-023-46710-y.
References
- 1.Jia L, et al. Prevalence, risk factors, and management of dementia and mild cognitive impairment in adults aged 60 years or older in China: A cross-sectional study. Lancet Public Health. 2020;5:e661–e671. doi: 10.1016/s2468-2667(20)30185-7. [DOI] [PubMed] [Google Scholar]
- 2.Hodson R. Alzheimer's disease. Nature. 2018;559:S1. doi: 10.1038/d41586-018-05717-6. [DOI] [PubMed] [Google Scholar]
- 3.Petersen RC. Mild cognitive impairment as a diagnostic entity. J. Intern. Med. 2004;256:183–194. doi: 10.1111/j.1365-2796.2004.01388.x. [DOI] [PubMed] [Google Scholar]
- 4.Cherrier MM, Mendez MF, Dave M, Perryman KM. Performance on the Rey-Osterrieth complex figure test in alzheimer disease and vascular dementia. Neuropsychiatry, Neuropsychol., Behav. Neurol. 1999;12:95–101. [PubMed] [Google Scholar]
- 5.Zhang X, et al. Overview of the complex figure test and its clinical application in neuropsychiatric disorders. Incl. Copy. Recall. 2021;12:680474. doi: 10.3389/fneur.2021.680474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Salimi S, et al. Can visuospatial measures improve the diagnosis of Alzheimer's disease? Alzheimers Dement (Amst) 2018;10:66–74. doi: 10.1016/j.dadm.2017.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shin M-S, Park S-Y, Park S-R, Seol S-H, Kwon JS. Clinical and empirical applications of the Rey-Osterrieth complex figure test. Nat. Protoc. 2006;1:892–899. doi: 10.1038/nprot.2006.115. [DOI] [PubMed] [Google Scholar]
- 8.Le Osterrieth PA. test de copie d'une figure complexe; contribution à l'étude de la perception et de la mémoire [Test of copying a complex figure; contribution to the study of perception and memory] Arch. Psychol. 1944;30:206–356. [Google Scholar]
- 9.Taylor, L.B. Scoring criteria for the Rey-Osterrieth complex figure test. 350–351 (1998).
- 10.Meyers JE, Meyers KR. Rey complex figure test and recognition trial (RCFT) Psychological Assessment Resources Odessa; 1995. [Google Scholar]
- 11.Bennett-Levy J. Determinants of performance on the Rey-Osterrieth complex figure test: An analysis, and a new technique for single-case assessment. Br. J. Clin. Psychol. 1984;23:109–119. doi: 10.1111/j.2044-8260.1984.tb00634.x. [DOI] [PubMed] [Google Scholar]
- 12.Somerville J, Tremont G, Stern RA. The boston qualitative scoring system as a measure of executive functioning in Rey-Osterrieth complex figure performance. J. Clin. Exp. Neuropsychol. 2000;22:613–621. doi: 10.1076/1380-3395(200010)22:5;1-9;FT613. [DOI] [PubMed] [Google Scholar]
- 13.Waber DP, Holmes JM. Assessing children's copy productions of the Rey-Osterrieth complex figure. J. Clin. Exp. Neuropsychol. 1985;7:264–280. doi: 10.1080/01688638508401259. [DOI] [PubMed] [Google Scholar]
- 14.Faundez-Zanuy, M., Mekyska, J. & Impedovo, D. Online handwriting, signature and touch dynamics: Tasks and potential applications in the field of security and health. (2022).
- 15.Canham, R. O., Smith, S. L. & Tyrrell, A. M. in Proceedings of the 26th Euromicro Conference. EUROMICRO 2000. Informatics: Inventing the Future.402, 406–413.
- 16.Hyun GJ, et al. Visuospatial working memory assessment using a digital tablet in adolescents with attention deficit hyperactivity disorder. Comput. Methods Programs Biomed. 2018;157:137–143. doi: 10.1016/j.cmpb.2018.01.022. [DOI] [PubMed] [Google Scholar]
- 17.Vogt J, et al. Automated scoring of the Rey-Osterrieth complex figure test using a deep-learning algorithm. Arch. Clin. Neuropsychol. 2019;34:836–836. doi: 10.1093/arclin/acz035.04. [DOI] [Google Scholar]
- 18.Webb SS, et al. Validation of an automated scoring program for a digital complex figure copy task within healthy aging and stroke. Neuropsychology. 2021;35:847–862. doi: 10.1037/neu0000748. [DOI] [PubMed] [Google Scholar]
- 19.Poreh A, Levin JB, Teaford M. Geriatric complex figure test: A test for the assessment of planning, visual spatial ability, and memory in older adults. Appl. Neuropsychol. Adult. 2020;27:101–107. doi: 10.1080/23279095.2018.1490288. [DOI] [PubMed] [Google Scholar]
- 20.Kim KW, et al. A Comprehensive evaluation of the process of copying a complex figure in early- and late-onset Alzheimer disease: A quantitative analysis of digital pen data. J. Med. Internet Res. 2020;22:e18136. doi: 10.2196/18136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Petilli MA, Daini R, Saibene FL, Rabuffetti M. Automated scoring for a tablet-based Rey figure copy task differentiates constructional, organisational, and motor abilities. Sci. Rep. 2021;11:14895. doi: 10.1038/s41598-021-94247-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Müller S, Preische O, Heymann P, Elbing U, Laske C. Increased diagnostic accuracy of digital vs. conventional clock drawing test for discrimination of patients in the early course of Alzheimer's disease from cognitively healthy individuals. Front. Aging Neurosci. 2017;9:101. doi: 10.3389/fnagi.2017.00101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Müller S, et al. Diagnostic value of digital clock drawing test in comparison with CERAD neuropsychological battery total score for discrimination of patients in the early course of Alzheimer's disease from healthy individuals. Sci. Rep. 2019;9:3543. doi: 10.1038/s41598-019-40010-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dion C, et al. Cognitive correlates of digital clock drawing metrics in older adults with and without mild cognitive impairment. J. Alzheimer's Dis.: JAD. 2020;75:73–83. doi: 10.3233/JAD-191089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhang X, et al. A study on the performance characteristics and diagnostic efficacy of digital clock drawing test in patients with amnesic mild cognitive impairmen. Chin. J. Behav. Med. Brain Sci. 2021;30:794–799. doi: 10.3760/cma.j.cn371468-20210607-00313. [DOI] [Google Scholar]
- 26.Binaco R, et al. Machine learning analysis of digital clock drawing test performance for differential classification of mild cognitive impairment subtypes versus Alzheimer's disease. J. Int. Neuropsychol. Soc.: JINS. 2020;26:690–700. doi: 10.1017/s1355617720000144. [DOI] [PubMed] [Google Scholar]
- 27.Poreh, A. M. Neuropsychological Testing in the 21st Century.
- 28.Sperling RA, et al. Toward defining the preclinical stages of Alzheimer's disease: Recommendations from the national institute on aging-Alzheimer's association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimer's Dementia: J. Alzheimer's Assoc. 2011;7:280–292. doi: 10.1016/j.jalz.2011.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Folstein MF, Folstein SE, McHugh PR. "Mini-mental state". A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975;12:189–198. doi: 10.1016/0022-3956(75)90026-6. [DOI] [PubMed] [Google Scholar]
- 30.Zhang MY, et al. The prevalence of dementia and Alzheimer's disease in Shanghai, China: Impact of age, gender, and education. Ann. Neurol. 1990;27:428–437. doi: 10.1002/ana.410270412. [DOI] [PubMed] [Google Scholar]
- 31.Nasreddine ZS, et al. The montreal cognitive assessment, MoCA: A brief screening tool for mild cognitive impairment. J. Am. Geriatr. Soc. 2005;53:695–699. doi: 10.1111/j.1532-5415.2005.53221.x. [DOI] [PubMed] [Google Scholar]
- 32.Hu JB, et al. Cross-cultural difference and validation of the Chinese version of montreal cognitive assessment in older adults residing in Eastern China: Preliminary findings. Arch. Gerontol. Geriatr. 2013;56:38–43. doi: 10.1016/j.archger.2012.05.008. [DOI] [PubMed] [Google Scholar]
- 33.Hughes CP, Berg L, Danziger W, Coben LA, Martin RL. A new clinical scale for the staging of dementia. Br. J. Psychiatry. 1982;140:566–572. doi: 10.1192/bjp.140.6.566. [DOI] [PubMed] [Google Scholar]
- 34.Maj M, et al. Evaluation of two new neuropsychological tests designed to minimize cultural bias in the assessment of HIV-1 seropositive persons: A WHO study. Arch. Clin. Neuropsychol. 1993;8:123–135. doi: 10.1093/arclin/8.2.123. [DOI] [PubMed] [Google Scholar]
- 35.Hong X, et al. Validity of auditory verbal learning test in diagnosis of Alzheimer's disease. Zhongguo yi xue ke xue yuan xue bao. Acta Acad. Med. Sin. 2012;34:262–266. doi: 10.3881/j.issn.1000-503X.2012.03.014. [DOI] [PubMed] [Google Scholar]
- 36.Guo, Q. -H., Hong, Z. & Shi, W. -X. Boston naming test in Chinese elderly, patient with mild cognitive impairment and Alzheimer's dementia. Chin. Mental Health J. (1991).
- 37.Kaplan EF, Goodglass H, Weintraub S. The Boston Naming Test: Experimental edition. 2. Lea & Febiger; 1978. [Google Scholar]
- 38.Mok EHL, Lam LCW, Chiu HFK. Category verbal fluency test performance in Chinese elderly with Alzheimer’s disease. Dement. Geriatr. Cogn. Disord. 2004;18:120–124. doi: 10.1159/000079190. [DOI] [PubMed] [Google Scholar]
- 39.Ardila A, Rosselli MJDN. Neuropsychological characteristics of normal aging. Dev. Neuropsychol. 1989;5:307–320. doi: 10.1080/87565648909540441. [DOI] [Google Scholar]
- 40.Benton A, Hannay HJ, Varney NR. Visual perception of line direction in patients with unilateral brain disease. Neurology. 1975;25:907–910. doi: 10.1212/wnl.25.10.907. [DOI] [PubMed] [Google Scholar]
- 41.Reitan RM. Validity of the trail making test as an indicator of organic brain damage. Percept. Motor skills. 1958;8:271–276. doi: 10.2466/pms.1958.8.3.271. [DOI] [Google Scholar]
- 42.Wei M, et al. Diagnostic accuracy of the chinese version of the trail-making test for screening cognitive impairment. J. Am. Geriatr. Soc. 2018;66:92–99. doi: 10.1111/jgs.15135. [DOI] [PubMed] [Google Scholar]
- 43.Stroop JR. Studies of interference in serial verbal reactions. J. Exp. Psychol. 1935;18:643. doi: 10.1037/h0054651. [DOI] [Google Scholar]
- 44.Golden CJ. Identification of brain disorders by the Stroop Color and word test. J. Clin. Psychol. 1976;32(3):654–658. doi: 10.1002/1097-4679(197607)32:3<654::AID-JCLP2270320336>3.0.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- 45.de Paula JJ, Costa MV, de Andrade GF, Ávila RT, Malloy-Diniz LF. Validity and reliability of a "simplified" version of the Taylor complex figure test for the assessment of older adults with low formal education. Dementia Neuropsychol. 2016;10:52–57. doi: 10.1590/s1980-57642016dn10100010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hong Z, et al. Diagnostic validity of complex graph test in distinguishing Alzheimer's disease from non dementia. Chin. Acad. Med. Sci. 2013;35:348–352. doi: 10.3881/j.issn.1000-503X.2013.03.020. [DOI] [PubMed] [Google Scholar]
- 47.Possin KL, Laluz VR, Alcantar OZ, Miller BL, Kramer JH. Distinct neuroanatomical substrates and cognitive mechanisms of figure copy performance in Alzheimer's disease and behavioral variant frontotemporal dementia. Neuropsychologia. 2011;49:43–48. doi: 10.1016/j.neuropsychologia.2010.10.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wilson NA, Batchelor J. Examining Rey Complex figure test organization in healthy adults. J. Clin. Exp. Neuropsychol. 2015;37:1052–1061. doi: 10.1080/13803395.2015.1075473. [DOI] [PubMed] [Google Scholar]
- 49.Blanco Martín E, et al. Dysexecutive syndrome in amnesic mild cognitive impairment: A multicenter study. BMC Neurol. 2016;16:88. doi: 10.1186/s12883-016-0607-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Slavin MJ, Mattingley JB, Bradshaw JL, Storey E. Local-global processing in Alzheimer's disease: An examination of interference, inhibition and priming. Neuropsychologia. 2002;40:1173–1186. doi: 10.1016/s0028-3932(01)00225-1. [DOI] [PubMed] [Google Scholar]
- 51.Melrose RJ, Harwood D, Khoo T, Mandelkern M, Sultzer DL. Association between cerebral metabolism and Rey-Osterrieth complex figure test performance in Alzheimer's disease. J. Clin. Exp. Neuropsychol. 2013;35:246–258. doi: 10.1080/13803395.2012.763113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Forbes-McKay K, Shanks M, Venneri A. Charting the decline in spontaneous writing in Alzheimer's disease: A longitudinal study. Acta Neuropsychiatr. 2014;26:246–252. doi: 10.1017/neu.2014.2. [DOI] [PubMed] [Google Scholar]
- 53.Yamada Y, et al. Automated analysis of drawing process to estimate global cognition in older adults: Preliminary international validation on the US and Japan data sets. JMIR Form. Res. 2022;6:e37014. doi: 10.2196/37014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Yuan J, et al. Associations between the digital clock drawing test and brain volume: Large community-based prospective cohort (Framingham Heart Study) J. Med. Internet Res. 2022;24:e34513. doi: 10.2196/34513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rentz DM, et al. Association of digital clock drawing with PET amyloid and tau pathology in normal older adults. Neurology. 2021;96:e1844–e1854. doi: 10.1212/wnl.0000000000011697. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and analyzed during the current study are available from the corresponding author upon reasonable request.