Abstract
Background
Both the incidence of lung cancer and the prevalence of metabolic syndrome (MetS) have been increasing worldwide. The relationship between MetS and lung cancer remains controversial.
Research Question
What is the risk of lung cancer associated with MetS and its components?
Study Design and Methods
Multivariable Cox regression models were used to estimate the hazard ratio (HR) of MetS-related variables on lung cancer risk, both overall and by histologic subtype, in the UK Biobank. Stratified analyses were conducted by sex, tobacco use status, and use of medication. HR curves were used to test the nonlinear associations between the metabolic markers and the risk of lung cancer.
Results
Of the 331,877 participants included in this study, a total of 77,173 participants had a diagnosis of MetS at enrollment. During a median follow-up of 10.9 years, lung cancer as the primary site developed in 2,425 participants. The HRs of MetS were 1.21 (95% CI, 1.09-1.33), 1.28 (95% CI, 1.10-1.50), and 1.16 (95% CI, 0.94-1.44) for the overall risk of lung cancer, adenocarcinoma, and squamous cell carcinoma, respectively. The HRs increased with the number of metabolic abnormalities from 1.11 to approximately 1.4 or 1.5 for those with one to five disorders. Positive association with lung cancer was observed for low high-density lipoprotein cholesterol (HDL-C), elevated waist circumference, and hyperglycemia. The relationship between MetS and lung cancer was modified by sex, with a stronger effect in female patients (P = .031). The risk of lung cancer resulting from MetS was elevated mainly among individuals who used tobacco, although the modification effect of tobacco use was not statistically significant. A nonlinear association was found between lung cancer and HDL-C, waist circumference, and glycated hemoglobin.
Interpretation
The increased risk of lung cancer associated with MetS suggests the importance of taking metabolic status and markers into consideration for the primary prevention of lung cancer and the selection of high-risk populations for lung cancer screening.
Key Words: elevated waist circumference, hyperglycemia, low HDL-C, lung cancer, metabolic syndrome
FOR EDITORIAL COMMENT, SEE PAGE 14
Take-home Points.
Study Question: Is the risk of lung cancer associated with metabolic syndrome (MetS) and its components?
Results: MetS, low high-density lipoprotein cholesterol, elevated waist circumference, and hyperglycemia are risk factors of lung cancer, especially in female patients and those who use tobacco.
Interpretation: It is important to take metabolic status and markers into consideration in the primary prevention of lung cancer and the selection of high-risk populations for lung cancer screening.
Lung cancer is one of the most commonly diagnosed cancers and the leading cause of cancer deaths globally.1 The incidence of lung cancer has increased rapidly over the past decades and is projected to reach 3.6 million cases by 2040.1 Known risk factors, such as age, sex, socioeconomic factors, tobacco use, COPD, and family history of lung cancer, have been used widely in the prediction of lung cancer risk and selection of high-risk populations for screening.2, 3, 4 However, approximately 30% of cases of lung cancer still would be missed by using these risk factors as selection criteria.5 Therefore, it is important to expand our knowledge regarding risk factors related to lung cancer development.
Metabolic syndrome (MetS) is a cluster of interrelated risk factors including hypertension, dyslipidemia, central obesity, and insulin resistance. Individuals with MetS are more prone to several types of diseases such as cardiovascular diseases and diabetes.6 MetS is becoming an emerging public health concern because of its high prevalence globally. Between 20% and 30% of the adult population are estimated to have MetS in most countries worldwide and the prevalence keeps rising.7 In the United States, the prevalence of MetS increased from 33% in 2011 and 2012 to 37% in 2015 and 2016, and the upward trend was more notable among the younger generations and women.8 Similar upward trends also were observed in different parts of the world.9
Growing evidence shows the association between MetS and cancer,10,11 but the relationship between MetS and lung cancer remains controversial. Although increased risk of lung cancer was reported among those with MetS in a few studies,12,13 a meta-analysis including five cohort studies found no association of MetS with lung cancer risk.14 However, these studies largely were underpowered to test the null hypothesis, and the small sample size of each cohort precludes further investigations on effect modifiers. In the present study, based on a large-scale cohort with enough size, we aimed to investigate the association of MetS, number of metabolic abnormalities, MetS components, and metabolic markers with the risk of lung cancer. We also assessed the effect modification by sex, tobacco use status, and use of medication.
Study Design and Methods
Study Design and Population
The UK Biobank is a multicenter, population-based, prospective cohort of more than half a million participants registered with the UK National Health Service. The design of the UK Biobank has been described previously.15 Briefly, male and female participants between 37 and 73 years of age were recruited from 22 assessment centers across England, Scotland, and Wales between 2006 and 2010. During the baseline survey, participants took touch screen and nurse-led questionnaires, underwent physical measurement, and provided biological samples. Participants were followed up for incidence of cancer through linkage to the National Cancer Registries until January 31, 2021, for Scotland and February 29, 2020, for England and Wales. In the present study, we excluded those (1) with prevalent cancers at baseline (except for nonmelanoma skin cancer, coded as C44 using the International Classification of Diseases, Tenth Revision), (2) pregnant at baseline assessment, and (3) with missing data for any of the five MetS components and other covariates (e-Fig 1).
Assessment of MetS
MetS was defined based on a joint statement6 involving five metabolic abnormalities: elevated BP (systolic BP, ≥ 130 mm Hg, diastolic BP, ≥ 85 mm Hg, or both), hypertriglyceridemia (≥ 1.7 mmol/L), low high-density lipoprotein cholesterol (HDL-C; < 1.0 mmol/L in men and < 1.3 mmol/L in female participants), elevated waist circumference (≥ 102 cm in male participants and ≥ 88 cm in female participants for European populations), and hyperglycemia (fasting glucose, ≥ 100 mg/dL). Two measures of systolic and diastolic BP were obtained, and the average values were calculated. Triglyceride and HDL-C concentrations in the serum were measured by enzyme immunoinhibition and glycerol-3-phosphate-peroxidase analysis. For hyperglycemia, because nonfasting glucose levels were measured for most participants in the UK Biobank, we used the more stable measure of glycated hemoglobin (HbA1c) instead, with a cutoff of ≥ 42.0 mmol/mol for impaired glucose regulation.16,17 The number of MetS components was counted and the presence of any three of the components listed constituted a diagnosis of MetS.
Outcomes
We used the International Classification of Diseases, 10th Revision, codes (C33-34) to define cases of incident lung cancer. Participants were censored if a cancer in other sites (other than nonmelanoma skin cancer) was diagnosed before lung cancer, because of the difficulty in separating metastatic from second primary cancer and the different risk profiles of lung cancer among those with a prior cancer diagnosis. All participants were followed up prospectively until lung cancer diagnosis, death, loss to follow-up, or being censored, whichever occurred first. Histologic findings were coded according to the International Classification of Diseases for Oncology, Third Edition, and histologic subtypes were classified based on the World Health Organization classification18,19: adenocarcinoma (codes 8140, 8250, 8551, 8260, 8265, 8230, 8253, 8254, 8480, 8333, 8144, 8256, and 8257), squamous cell carcinoma (8070, 8071, 8072, and 8083), large cell carcinoma (8012), and small cell carcinoma (8041 and 8045). Bronchioloalveolar carcinoma (codes 8250, 8252, 8253, and 8254), a term used previously, also was considered because of evidence of large overdiagnosis among these cancers.20
Covariates
Information on covariates was collected through questionnaire and physical measurements. Sociodemographic characteristics included baseline age (continuous variable), sex, race (White, Asian, Black, and others), and Townsend deprivation index (classified into five quartiles). Tobacco use status included never, former, and current tobacco use. COPD was counted if a participant reported physician-diagnosed COPD (emphysema or chronic bronchitis) or if the spirometric criteria of FEV1 to FVC ratio of < 0.7 and FEV1 < 80% predicted were fullfilled.21 Family history of lung cancer (yes or no) was collected. BMI was calculated as weight (in kilograms) per height (in square meters) and was categorized further into underweight (< 18.5 kg/m2), normal (18.5-24.9 kg/m2), overweight (25.0-29.9 kg/m2), and obese (≥ 30 kg/m2). BMIs of < 12 kg/m2 or > 60 kg/m2 were considered as outliers and removed from downstream analyses. Fasting hours before obtained biological samples were recorded and those ≤ 1 h or ≥ 6 h were grouped because of the small number of participants.
Statistical Analysis
Baseline characteristics were described using mean ± SD for continuous variables and frequency (proportion) for categorical variables. The t test, Wilcox rank-sum test, and χ2 test were used where appropriate to compare baseline characteristics between groups with MetS and without MetS. Incidence rates of lung cancer were calculated by MetS, number of metabolic abnormalities, and MetS components.
The proportional hazard assumption was checked by tests based on Schoenfeld residuals. The hazard ratio (HR) of MetS-related variables on lung cancer risk was examined by Cox regression models. Cox models were stratified by assessment center because of violation of the proportional hazard assumption for this variable and were adjusted for age, sex, race, Townsend deprivation index, COPD, family history of lung cancer, tobacco use status, BMI, and fasting time. For elevated BP and waist circumference, fasting time was not included in the model.
We examined the impact of MetS on both overall lung cancer and two major subtypes of lung cancer (adenocarcinoma and squamous cell carcinoma). We did not analyze other subtypes because of a low number of cases. Stratified analyses were conducted by sex (male and female) and tobacco use status (use and nonuse). For elevated BP, hypertriglyceridemia, low HDL-C, and hyperglycemia, we also stratified the analysis by use of medication. We performed interaction analyses to determine the modification effects of these factors. The significance of the interaction term was examined by the Wald test.
We investigated the relationship between the metabolic markers (systolic BP, diastolic BP, triglycerides, HDL-C, waist circumference, and HbA1c) and the risk of lung cancer. Values < the 1% quantile or > the 99% quantile were considered outliers22 and were excluded from the analysis. HR curves in the R package smoothHR (R Foundation for Statistical Computing)23 were used to test the nonlinear associations, with the median values as reference, after controlling for the aforementioned covariates.
We performed several sensitivity analyses to assess the robustness of our findings. First, we excluded participants with a follow-up of < 2 years to minimize the potential for reverse causality (ie, undiagnosed lung cancer at baseline influencing subsequent metabolic changes). Second, we excluded those with fasting time of < 3 h to reduce its influence on the metabolic biomarkers in the serum. Third, we excluded bronchioloalveolar carcinoma from the total cases of lung cancer considering the potential influence of overdiagnosis bias. Fourth, we further adjusted for pack-years of tobacco use among individuals who had ever used tobacco, second-hand smoke exposure at or outside home, alcohol intake, and diabetes in the multivariable models. Fifth, we incorporated medication use in the definition of MetS (ie, antihypertensive drug treatment,24 statin and other lipid-lowering drugs,24 and glucose-lowering drugs25 as alternate indicators of elevated BP, elevated triglycerides, reduced HDL-C, and elevated fasting glucose), and then evaluated the association between MetS under the new definition and lung cancer risk.
All statistical analyses were performed with R version 4.0.3 software. Statistical significance was defined as a 2-sided P value of < .05.
Results
Characteristics of the Study Population
Of the 331,877 participants included in this study, the mean ± SD age was 56.3 ± 8.1 years, and 53% of participants were female. A total of 77,173 participants demonstrated MetS at enrollment, with a prevalence of 23.3%. Characteristics of the study participants according to MetS status are shown in Table 1. Participants with MetS were older and had higher socioeconomic deprivation (lower socioeconomic status) compared with those without MetS. Higher proportions of male participants, non-White people, individuals who use tobacco, those with second-hand tobacco smoke exposure, family history of lung cancer, COPD, and obesity were observed among those with MetS (Table 1). Metabolic abnormalities prevailed among the study population, with 82.6% of the participants having at least one disorder. The prevalence of each MetS component ranged from 8.0% (hyperglycemia) to 66.5% (elevated BP) (e-Fig 2).
Table 1.
Baseline Characteristics of the Study Population by MetS Status
| Characteristics | Total (N = 331,877) | Non-MetS (n = 254,704) | MetS (n = 77,173) | P Value |
|---|---|---|---|---|
| Age, y | 56.3 ± 8.1 | 55.8 ± 8.1 | 57.8 ± 7.7 | < .001 |
| Sex, female | 175,943 (53.0) | 137,864 (54.1) | 38,079 (49.3) | < .001 |
| Race | < .001 | |||
| White | 314,737 (94.8) | 242,364 (95.2) | 72,373 (93.8) | |
| Asian | 7,675 (2.3) | 5,201 (2.0) | 2,474 (3.2) | |
| Black | 4,672 (1.4) | 3,491 (1.4) | 1,181 (1.5) | |
| Other | 4,793 (1.4) | 3,648 (1.4) | 1,145 (1.5) | |
| Townsend deprivation index | –1.4 ± 3.0 | –1.5 ± 3.0 | –1.0 ± 3.2 | < .001 |
| Tobacco use status | < .001 | |||
| Never | 183,095 (55.2) | 144,429 (56.7) | 38,666 (50.1) | |
| Former | 114,375 (34.5) | 84,849 (33.3) | 29,526 (38.3) | |
| Current | 34,407 (10.4) | 25,426 (10.0) | 8,981 (11.6) | |
| Second-hand smoke exposure | 62,770 (18.9) | 45,993 (18.1) | 16,777 (21.7) | < .001 |
| Family history | 41,445 (12.5) | 30,769 (12.1) | 10,676 (13.8) | < .001 |
| COPD | 19,795 (6.0) | 14,054 (5.5) | 5,741 (7.4) | < .001 |
| BMI category | < .001 | |||
| Underweight | 1,680 (0.5) | 1,668 (0.7) | 12 (0.0) | |
| Normal | 108,555 (32.7) | 104,574 (41.1) | 3,981 (5.2) | |
| Overweight | 141,864 (42.7) | 114,851 (45.1) | 27,013 (35.0) | |
| Obesity | 79,778 (24.0) | 33,611 (13.2) | 46,167 (59.8) | |
| Antihypertensives | 74,883 (22.6) | 44,916 (17.6) | 29,967 (38.8) | < .001 |
| Lipid-lowering medication | 59,008 (17.8) | 34,777 (13.7) | 24,231 (31.4) | < .001 |
| Glucose-lowering medication | 12,006 (3.6) | 3,373 (1.3) | 8,633 (11.2) | < .001 |
| BP, mm Hg | ||||
| Systolic | 137.7 ± 18.5 | 135.5 ± 18.6 | 144.9 ± 16.3 | < .001 |
| Diastolic | 82.3 ± 10.1 | 81.0 ± 10.0 | 86.3 ± 9.4 | < .001 |
| Triglycerides, mM | 1.7 ± 1.0 | 1.5 ± 0.8 | 2.6 ± 1.2 | < .001 |
| HDL-C, mM | 1.4 ± 0.4 | 1.5 ± 0.4 | 1.2 ± 0.3 | < .001 |
| Waist circumference, cm | 90.3 ± 13.4 | 86.6 ± 11.5 | 102.5 ± 11.8 | < .001 |
| HbA1c, mmol/mol | 36.0 ± 6.7 | 34.9 ± 4.6 | 39.9 ± 10.1 | < .001 |
| Fasting time, h | < .001 | |||
| ≤ 1 | 16,002 (4.8) | 12,299 (4.8) | 3,703 (4.8) | |
| 2 | 69,042 (20.8) | 53,662 (21.1) | 15,380 (19.9) | |
| 3 | 96,953 (29.2) | 74,629 (29.3) | 22,324 (28.9) | |
| 4 | 72,895 (22.0) | 55,211 (21.7) | 17,684 (22.9) | |
| 5 | 40,104 (12.1) | 30,457 (12.0) | 9,647 (12.5) | |
| ≥ 6 | 36,881 (11.1) | 28,446 (11.2) | 8,435 (10.9) |
Data are presented as No. (%) or mean ± SD, unless otherwise indicated. HbA1c = glycated hemoglobin; HDL-C = high-density lipoprotein cholesterol; MetS = metabolic syndrome.
MetS and Lung Cancer
After a total of 3,462,163 person-years of follow-up (median, 10.9 years; mean, 10.4 years), cancer with lung as the primary site developed in 2,425 participants, with 1,001, 503, 26, and 246 cases of adenocarcinoma, squamous cell carcinoma, large cell, and small cell carcinoma, respectively. The incidence rates of lung cancer were 97.7 and 61.8 per 100,000 in the MetS and non-MetS groups, and they increased with the number of metabolic abnormalities. Lung cancer incidence rates were higher for all MetS components. Notably, the incidence rate reached 141.4 per 100,000 among those with hyperglycemia. A statistically significant association was found between MetS and lung cancer risk, with an HR of 1.21 (95% CI, 1.09-1.33), after adjusting for covariates. The HRs increased with the number of metabolic abnormalities from 1.11 to approximately 1.4 to 1.5 for those with one to five disorders. A positive association with lung cancer was observed for three MetS components: low HDL-C (HR, 1.20; 95% CI, 1.09-1.32), elevated waist circumference (HR, 1.36; 95% CI, 1.22-1.53), and hyperglycemia (HR, 1.30; 95% CI, 1.16-1.45) (Table 2). These associations remained consistent in sensitivity analyses (e-Tables 1, 2).
Table 2.
Incidence Rates of Lung Cancer by MetS Status and the Association of MetS-Related Variables With Risk of Lung Cancer
| MetS-Related Variables | No. of Cases | No. of Person-Years | Incidence Rates per 100,000 | HR (95% CI) | Adjusted HR (95% CI)a |
|---|---|---|---|---|---|
| MetS | |||||
| No | 1,648 | 2,666,829 | 61.8 | Reference | Reference |
| Yes | 777 | 795,333 | 97.7 | 1.58 (1.45-1.73) | 1.21 (1.09-1.33) |
| No. metabolic abnormalities | |||||
| 0 | 236 | 610,658 | 38.6 | Reference | Reference |
| 1 | 712 | 1,155,720 | 61.6 | 1.60 (1.38-1.85) | 1.11 (0.96-1.29) |
| 2 | 700 | 900,451 | 77.7 | 2.02 (1.74-2.34) | 1.25 (1.07-1.46) |
| 3 | 483 | 529,100 | 91.3 | 2.37 (2.03-2.77) | 1.40 (1.18-1.66) |
| 4 | 232 | 219,082 | 105.9 | 2.75 (2.30-3.30) | 1.48 (1.21-1.81) |
| 5 | 62 | 47,152 | 131.5 | 3.43 (2.59-4.53) | 1.45 (1.08-1.95) |
| P value for trend | NA | NA | NA | < .001 | < .001 |
| MetS components | |||||
| Elevated BP | |||||
| No | 672 | 1,169,261 | 57.5 | Reference | Reference |
| Yes | 1,753 | 2,292,902 | 76.5 | 1.33 (1.22-1.46) | 0.95 (0.87-1.05) |
| Hypertriglyceridemia | |||||
| No | 1,292 | 2,086,625 | 61.9 | Reference | Reference |
| Yes | 1,133 | 1,375,538 | 82.4 | 1.33 (1.23-1.44) | 1.04 (0.96-1.13) |
| Low HDL-C | |||||
| No | 1,823 | 2,780,681 | 65.6 | Reference | Reference |
| Yes | 602 | 681,482 | 88.3 | 1.35 (1.23-1.48) | 1.20 (1.09-1.32) |
| Elevated waist circumference | |||||
| No | 1,490 | 2,422,031 | 61.5 | Reference | Reference |
| Yes | 935 | 1,040,132 | 89.9 | 1.47 (1.35-1.59) | 1.36 (1.22-1.53) |
| Hyperglycemia | |||||
| No | 2,049 | 3,196,209 | 64.1 | Reference | Reference |
| Yes | 376 | 265,954 | 141.4 | 2.23 (1.99-2.48) | 1.30 (1.16-1.45) |
HDL-C = high-density lipoprotein cholesterol; HR = hazard ratio; MetS = metabolic syndrome; NA = not applicable.
The multivariable models were stratified by assessment center and adjusted for age, sex, race, Townsend deprivation index, COPD, family history of lung cancer, tobacco use status, BMI, and fasting time. For elevated BP and waist circumference, fasting time was not included in the model.
Similarly, higher incidence rates were found in participants with MetS for both adenocarcinoma and squamous cell carcinoma, with 38.3 and 21.4 per 100,000 compared with 26.1 and 12.5 per 100,000 among those without MetS (e-Table 3). The HRs of MetS were 1.28 (95% CI, 1.10-1.50) and 1.16 (95% CI, 0.94-1.44) on the risk of adenocarcinoma and squamous cell carcinoma, respectively. The trend tests for number of metabolic abnormalities were significant for both histologic subtypes. Elevated waist circumference showed a similarly large impact, with HRs of approximately 1.5 (Table 3).
Table 3.
Hazard Ratio (95% CI) of MetS-Related Variables With Risk of Adenocarcinoma and Squamous Cell Carcinoma After Adjusting for Covariates
| Variable | Adenocarcinoma | Squamous Cell Carcinoma |
|---|---|---|
| MetS (reference, non-MetS) | 1.28 (1.10-1.50) | 1.16 (0.94-1.44) |
| No. of metabolic abnormalities (reference, 0) | ||
| 1 | 1.08 (0.86-1.35) | 1.24 (0.86-1.79) |
| 2 | 1.27 (1.01-1.61) | 1.49 (1.03-2.16) |
| 3 | 1.56 (1.21-2.02) | 1.47 (0.98-2.21) |
| 4 | 1.32 (0.95-1.82) | 1.85 (1.18-2.91) |
| 5 | 1.58 (0.98-2.54) | 1.52 (0.79-2.92) |
| P value for trend | .001 | .012 |
| MetS components | ||
| Elevated BP | 1.00 (0.86-1.15) | 0.91 (0.74-1.12) |
| Hypertriglyceridemia | 1.11 (0.97-1.26) | 1.00 (0.84-1.20) |
| Low HDL-C | 1.13 (0.97-1.32) | 1.33 (1.08-1.63) |
| Elevated waist circumference | 1.48 (1.24-1.76) | 1.44 (1.12-1.86) |
| Hyperglycemia | 1.19 (0.98-1.44) | 1.35 (1.06-1.71) |
HDL-C = high-density lipoprotein cholesterol; MetS = metabolic syndrome.
Modification Effects
The relationship between MetS and lung cancer risk was modified by sex, with a stronger effect in female participants. Compared with male participants, female participants with MetS showed a higher risk of lung cancer (HR, 1.38 [95% CI, 1.20-1.60] vs 1.07 [95% CI, 0.93-1.23]), with P = .031 for the interaction term between sex and MetS. The risk of lung cancer among those with five metabolic abnormalities doubled that among those without any abnormality in female participants, whereas for male participants, the trend with number of metabolic abnormalities was not statistically significant. The HRs were higher in female participants for all MetS components, and the interaction was statistically significant for hypertriglyceridemia (Fig 1, e-Table 4). Similar patterns were observed for two histologic subtypes, although the increase in HRs with a higher number of metabolic abnormalities in female participants was more pronounced for squamous cell carcinoma (e-Table 5).
Figure 1.
Forest plot showing the association of MetS-related variables with risk of lung cancer stratified by sex. P < .05 (interaction analysis) indicates statistically significant effect modifications. HDL-C = high-density lipoprotein cholesterol; HR = hazard ratio; MetS = metabolic syndrome.
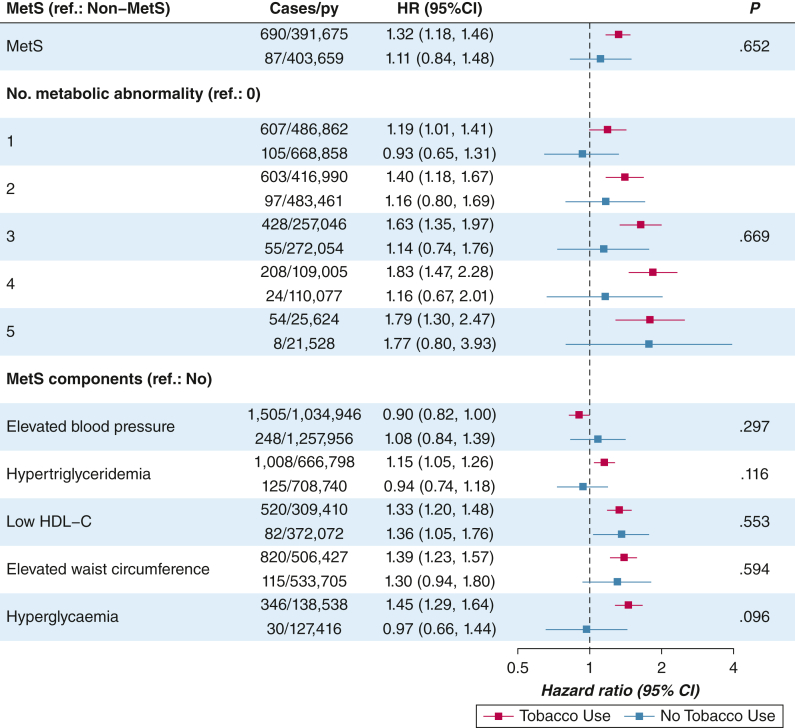
The modification effect of tobacco use was not detected for MetS-related variables. However, the risk of lung cancer resulting from MetS was elevated mainly among individuals who used tobacco. The HRs of MetS were 1.32 (95% CI, 1.18-1.46) and 1.11 (95% CI, 0.84-1.48) in those who use and do not use tobacco, respectively. The trend test across number of metabolic abnormalities was statistically significant only among individuals who use tobacco. Increased risk was found for almost all MetS components (except for elevated BP) in individuals who use tobacco, whereas it was found for only one in those who do not use tobacco, low HDL-C (Fig 2, e-Table 6). The modification effect of tobacco use was similar between histologic subtypes (e-Table 7). The HR estimates of MetS components were similar with and without the use of medication (e-Table 8).
Figure 2.
Forest plot showing the association of MetS-related variables with risk of lung cancer stratified by tobacco use status. P < .05 (interaction analysis) indicates statistically significant effect modifications. HDL-C = high-density lipoprotein cholesterol; HR = hazard ratio; MetS = metabolic syndrome.
Metabolic Markers and Lung Cancer
Using adjusted HR curves, a significant and nonlinear association was evident between lung cancer and HDL-C, waist circumference, and HbA1c. The risk of lung cancer decreased with increasing levels of HDL-C, and the impact reached a plateau after around 1.8 mM. Contrarily, lung cancer risk increased rapidly with waist circumference until around 105 cm and remained stable thereafter. The association between HbA1c and lung cancer was more U-shaped, with higher risk at both high and low HbA1c levels, and the minimum risk was seen at around 32 mmol/mol (Fig 3).
Figure 3.
A-F, Funnel plots showing the nonlinear association between biomarkers of metabolic syndrome with risk of lung cancer: systolic BP (A), diastolic BP (B), triglycerides (C), HDL-C (D), waist circumference (E), and HbA1c (F). HbA1c = glycated hemoglobin; HDL-C = high-density lipoprotein cholesterol; HR = hazard ratio.
Discussion
In this study, we found a statistically significant association between MetS and lung cancer risk. The magnitude of association increased with the number of metabolic abnormalities. Positive association with lung cancer was observed for low HDL-C, elevated waist circumference, and hyperglycemia. The relationship between MetS and lung cancer risk was stronger in female participants. Although no statistically significant modification effect was detected for tobacco use, the risk of lung cancer resulting from MetS was elevated mainly among individuals who used tobacco. To our knowledge, this study is the first to evaluate systematically the impact of MetS on lung cancer and the modification effects based on a large-scale cohort.
Results of prior studies on MetS and lung cancer risk are inconsistent. A meta-analysis including five cohort studies did not find a meaningful association,14 partly because of the small sample size of each study, with the largest number of cases of lung cancer being fewer than 500 in men.26 A Korean study based on the national insurance database reported > 45,000 cases of lung cancer, with a significant association between MetS and lung cancer observed,13 which is consistent with our findings. The mechanism underlying the association between metabolic disorders and lung cancer risk are not characterized fully. Possible pathways involve chronic inflammation caused by deregulation of cytokine production, insulin resistance, and overexpression of estrogen, which might be linked to the carcinogenesis process.27
Our study found that dyslipidemia may be a risk factor for lung cancer. HDL-C was associated inversely with long-term risk of lung cancer, which is in line with the findings of previous studies.28,29 The relationship between low HDL-C and lung cancer, on the one hand, may reflect the choice of unhealthy lifestyle that is linked with lung cancer risk; on the other hand, may be related to the ability of high-density lipoprotein in regulating inflammatory and proliferative pathways in lung cancer development.29 The impact of triglycerides on lung cancer is more debatable. A positive relationship was reported in some studies,30,31 but not in others.28,32 We did not find a meaningful association between triglycerides and lung cancer overall, but a weak increase in lung cancer risk was observed in female participants and individuals who use tobacco.
Waist circumference was associated with increased lung cancer risk after controlling for BMI. Several studies, including ours, consistently have found an inverse association between BMI and lung cancer risk (e-Table 9) and the opposite for waist circumference.33, 34, 35, 36 As indicators of overall and central obesity, the conflicting findings for BMI and waist circumference are intriguing. Overall obesity reflects both fat and lean mass, and the fat distribution and composition differ from central obesity, which is characterized by high abdominal fat distribution and excess visceral fat. Abdominal adiposity is related to hyperinsulinemia, altered levels of sex hormones, and proinflammatory adipokines,33,37,38 which play important roles in lung carcinogenesis. Lean mass, contrarily, was found to be associated inversely with lung cancer risk,39 which may explain in part the negative relationship between BMI and lung cancer. These suggest that central obesity may serve as a predictor of lung cancer.
Our study found an elevated risk of lung cancer among those with hyperglycemia. High levels of glucose and the resulting release of insulin can promote activation of the IGFR1-IR-PI3K-AKT-mTOR pathway, which has been considered to be a cause of carcinogenesis.40 Indeed, high insulin levels, insulin resistance, and preexisting diabetes are related to increased lung cancer risk.41,42 It is interesting to note the nonlinear relationship between HbA1c, a biomarker reflecting the average blood sugar over the past 3 months, and lung cancer. A similar U-shaped relationship also was found in several other cancer types, such as breast, colorectal, and pancreatic cancer.43,44
A sex interaction with MetS was noted in this study, with female participants having greater lung cancer risk. Female participants were associated with experiencing a greater increase in MetS severity and a worse metabolic risk profile than men,45 and hormonal factors were found to influence lung cancer both independently and by interplay with other risk factors.46 However, a case-control study found opposite findings, with male participants having a larger estimate.12 The role of sex in the relationship between MetS and lung cancer needs to be confirmed further. The impact of MetS on lung cancer was observed mainly among individuals who use tobacco, although the interaction term was not statistically significant, which might be the result of a low number of cases of lung cancer among those who have never used tobacco.
Several limitations of this study should be noted. First, we did not consider neither the cumulative years nor the changes over time, of MetS, which may influence the risk of lung cancer. Because MetS is a chronic disease per se, and metabolic status may have progressed several years before being diagnosed, it is difficult to determine its course over time. Second, nonfasting blood samples were obtained at baseline, which may lead to inconsistencies in biomarker measurements. However, it is reassuring that in the sensitivity analysis, the results were robust when we excluded those with short fasting time. Third, residual confounding cannot be precluded fully. MetS generally reflects an unhealthy lifestyle that involves possible risk factors of lung cancer. It remains unclear whether MetS itself or the downstream risk factors ultimately promote the occurrence of lung cancer. Fourth, we did not adjust for air pollution, which may influence the risk of both lung cancer and metabolic disorders. However, given that air pollution levels are supposed to be similar within small areas, this influence would be reduced largely through stratifying our analysis by assessment center. Finally, the possibility of reverse causality should be borne in mind, even though the results from the sensitivity analysis by excluding those with short follow-up argues against it.
Interpretation
In this study, we found associations between MetS and its components and an increased risk of lung cancer. This relationship potentially could be modified by sex and tobacco use. With the rapid increase in the prevalence of MetS worldwide, it is important to have a better understanding of its impact on lung cancer. Although a causal association between MetS and lung cancer cannot be verified through this study, the evidence obtained suggests the importance of taking metabolic status and markers into consideration in the selection of high-risk populations for lung cancer screening and highlights a new direction of lung cancer prevention.
Funding/Support
M. L. was supported by the National Natural Science Foundation of China [Grant 82204131], the Guangdong Basic and Applied Basic Research Foundation [Grant 2021A1515110625], Science and Technology Projects in Guangzhou [Grant 202201011243], and Young Talents Program of Sun Yat-sen University Cancer Center [Grant YTP-SYSUCC-0048]. J. Y. was supported by the National Natural Science Foundation of China [Grant 82003552].
Financial/Nonfinancial Disclosures
None declared.
Acknowledgments
Author contributions: M. L. serves as guarantor for the overall content of the manuscript. M. L. and J. Y. designed the study and drafted the manuscript. M. L. performed the statistical analyses. S.-M. C., N. D., L. W., and J.-B. L. contributed to the interpretation of data and critically revised the manuscript. All authors have read and approved the final manuscript for publication.
Role of sponsors: The sponsor had no role in the design of the study, the collection or analysis of the data, or the preparation of the manuscript.
Other contributions: This study was conducted under the UK Biobank application number 91354. The authors gratefully acknowledge the participants and those involved in building the resource.
Disclaimer: Where authors are identified as personnel of the International Agency for Research on Cancer or the World Health Organization, the authors alone are responsible for the views expressed in this article and they do not necessarily represent the decisions, policy, or views of the International Agency for Research on Cancer or the World Health Organization.
Additional information: The e-Figures and e-Tables are available online under “Supplementary Data.”
Supplementary Data
References
- 1.International Agency for Research on Cancer, World Health Organization Global cancer observatory. International Agency for Research on Cancer website. https://gco.iarc.fr/
- 2.Tammemägi M.C., Katki H.A., Hocking W.G., et al. Selection criteria for lung cancer screening. N Engl J Med. 2013;368(8):728–736. doi: 10.1056/NEJMoa1211776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Field J.K., Vulkan D., Davies M.P.A., Duffy S.W., Gabe R. Liverpool Lung Project lung cancer risk stratification model: calibration and prospective validation. Thorax. 2021;76(2):161–168. doi: 10.1136/thoraxjnl-2020-215158. [DOI] [PubMed] [Google Scholar]
- 4.Krist A.H., Davidson K.W., Mangione C.M., et al. Screening for lung cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2021;325(10):962–970. doi: 10.1001/jama.2021.1117. [DOI] [PubMed] [Google Scholar]
- 5.Robbins H.A., Alcala K., Swerdlow A.J., et al. Comparative performance of lung cancer risk models to define lung screening eligibility in the United Kingdom. Br J Cancer. 2021;124(12):2026–2034. doi: 10.1038/s41416-021-01278-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Alberti K.G., Eckel R.H., Grundy S.M., et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120(16):1640–1645. doi: 10.1161/CIRCULATIONAHA.109.192644. [DOI] [PubMed] [Google Scholar]
- 7.Grundy S.M. Metabolic syndrome pandemic. Arterioscler Thromb Vasc Biol. 2008;28(4):629–636. doi: 10.1161/ATVBAHA.107.151092. [DOI] [PubMed] [Google Scholar]
- 8.Hirode G., Wong R.J. Trends in the prevalence of metabolic syndrome in the United States, 2011-2016. JAMA. 2020;323(24):2526–2528. doi: 10.1001/jama.2020.4501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.O’Neill S., O’Driscoll L. Metabolic syndrome: a closer look at the growing epidemic and its associated pathologies. Obes Rev. 2015;16(1):1–12. doi: 10.1111/obr.12229. [DOI] [PubMed] [Google Scholar]
- 10.Esposito K., Chiodini P., Colao A., Lenzi A., Giugliano D. Metabolic syndrome and risk of cancer: a systematic review and meta-analysis. Diabetes Care. 2012;35(11):2402–2411. doi: 10.2337/dc12-0336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Uzunlulu M., Telci Caklili O., Oguz A. Association between metabolic syndrome and cancer. Ann Nutr Metab. 2016;68(3):173–179. doi: 10.1159/000443743. [DOI] [PubMed] [Google Scholar]
- 12.López-Jiménez T., Duarte-Salles T., Plana-Ripoll O., Recalde M., Xavier-Cos F., Puente D. Association between metabolic syndrome and 13 types of cancer in Catalonia: a matched case-control study. PLoS One. 2022;17(3) doi: 10.1371/journal.pone.0264634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sin S., Lee C.H., Choi S.M., Han K.D., Lee J. Metabolic syndrome and risk of lung cancer: an analysis of Korean national health insurance corporation database. J Clin Endocrinol Metab. 2020;105(11):dgaa596. doi: 10.1210/clinem/dgaa596. [DOI] [PubMed] [Google Scholar]
- 14.Qiao L., Ma D., Lv H., et al. Metabolic syndrome and the incidence of lung cancer: a meta-analysis of cohort studies. Diabetol Metab Syndr. 2020;12:95. doi: 10.1186/s13098-020-00598-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sudlow C., Gallacher J., Allen N., et al. UK Biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 2015;12(3) doi: 10.1371/journal.pmed.1001779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.John W.G. Use of HbA1c in the diagnosis of diabetes mellitus in the UK. The implementation of World Health Organization guidance 2011. Diabet Med. 2012;29(11):1350–1357. doi: 10.1111/j.1464-5491.2012.03762.x. [DOI] [PubMed] [Google Scholar]
- 17.Monroy-Iglesias M.J., Russell B., Crawley D., et al. Metabolic syndrome biomarkers and prostate cancer risk in the UK Biobank. Int J Cancer. 2021;148(4):825–834. doi: 10.1002/ijc.33255. [DOI] [PubMed] [Google Scholar]
- 18.Christakoudi S., Kakourou A., Markozannes G., et al. Blood pressure and risk of cancer in the European Prospective Investigation into Cancer and Nutrition. Int J Cancer. 2020;146(10):2680–2693. doi: 10.1002/ijc.32576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Travis W.D., Brambilla E., Nicholson A.G., et al. The 2015 World Health Organization Classification of lung tumors: impact of genetic, clinical and radiologic advances since the 2004 classification. J Thorac Oncol. 2015;10(9):1243–1260. doi: 10.1097/JTO.0000000000000630. [DOI] [PubMed] [Google Scholar]
- 20.Li M., Zhang L., Charvat H., et al. The influence of postscreening follow-up time and participant characteristics on estimates of overdiagnosis from lung cancer screening trials. Int J Cancer. 2022;151(9):1491–1501. doi: 10.1002/ijc.34167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sarkar C., Zhang B., Ni M., et al. Environmental correlates of chronic obstructive pulmonary disease in 96 779 participants from the UK Biobank: a cross-sectional, observational study. Lancet Planet Health. 2019;3(11):e478–e490. doi: 10.1016/S2542-5196(19)30214-1. [DOI] [PubMed] [Google Scholar]
- 22.Yang Z., Yang J., Li M., Chen J., Ou C.-Q. Nonlinear and lagged meteorological effects on daily levels of ambient PM2.5 and O3: evidence from 284 Chinese cities. J Clean Prod. 2021;278 [Google Scholar]
- 23.Meira-Machado L., Cadarso-Suárez C., Gude F., Araújo A. smoothHR: an R package for pointwise nonparametric estimation of hazard ratio curves of continuous predictors. Comput Math Methods Med. 2013;2013 doi: 10.1155/2013/745742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Carter A.R., Gill D., Davey Smith G., Taylor A.E., Davies N.M., Howe L.D. Cross-sectional analysis of educational inequalities in primary prevention statin use in UK Biobank. Heart. 2022;108(7):536–542. doi: 10.1136/heartjnl-2021-319238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Young K.G., McDonald T.J., Shields B.M. Glycated haemoglobin measurements from UK Biobank are different to those in linked primary care records: implications for combining biochemistry data from research studies and routine clinical care. Int J Epidemiol. 2022;51(3):1022–1024. doi: 10.1093/ije/dyab265. [DOI] [PubMed] [Google Scholar]
- 26.Ko S., Yoon S.-J., Kim D., Kim A.R., Kim E.-J., Seo H.-Y. Metabolic risk profile and cancer in Korean men and women. J Prev Med Public Health. 2016;49(3):143–152. doi: 10.3961/jpmph.16.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mendonça F.M., de Sousa F.R., Barbosa A.L., et al. Metabolic syndrome and risk of cancer: which link? Metabolism. 2015;64(2):182–189. doi: 10.1016/j.metabol.2014.10.008. [DOI] [PubMed] [Google Scholar]
- 28.Chandler P.D., Song Y., Lin J., et al. Lipid biomarkers and long-term risk of cancer in the Women’s Health Study. Am J Clin Nutr. 2016;103(6):1397–1407. doi: 10.3945/ajcn.115.124321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pedersen K.M., Çolak Y., Bojesen S.E., Nordestgaard B.G. Low high-density lipoprotein and increased risk of several cancers: 2 population-based cohort studies including 116,728 individuals. J Hematol Oncol. 2020;13(1):129. doi: 10.1186/s13045-020-00963-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lin X., Lu L., Liu L., et al. Blood lipids profile and lung cancer risk in a meta-analysis of prospective cohort studies. J Clin Lipidol. 2017;11(4):1073–1081. doi: 10.1016/j.jacl.2017.05.004. [DOI] [PubMed] [Google Scholar]
- 31.Lyu Z., Li N., Wang G., et al. Independent and joint associations of blood lipids and lipoproteins with lung cancer risk in Chinese males: a prospective cohort study. Int J Cancer. 2019;144(12):2972–2984. doi: 10.1002/ijc.32051. [DOI] [PubMed] [Google Scholar]
- 32.Hao B., Yu M., Sang C., Bi B., Chen J. Dyslipidemia and non-small cell lung cancer risk in Chinese population: a case-control study. Lipids Health Dis. 2018;17(1):278. doi: 10.1186/s12944-018-0925-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yu D., Zheng W., Johansson M., et al. Overall and central obesity and risk of lung cancer: a pooled analysis. J Natl Cancer Inst. 2018;110(8):831–842. doi: 10.1093/jnci/djx286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lee K.R., Seo M.H., Do Han K., et al. Waist circumference and risk of 23 site-specific cancers: a population-based cohort study of Korean adults. Br J Cancer. 2018;119(8):1018–1027. doi: 10.1038/s41416-018-0214-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yang Y., Dong J., Sun K., et al. Obesity and incidence of lung cancer: a meta-analysis. Int J Cancer. 2013;132(5):1162–1169. doi: 10.1002/ijc.27719. [DOI] [PubMed] [Google Scholar]
- 36.Recalde M., Davila-Batista V., Díaz Y., et al. Body mass index and waist circumference in relation to the risk of 26 types of cancer: a prospective cohort study of 3.5 million adults in Spain. BMC Med. 2021;19(1):10. doi: 10.1186/s12916-020-01877-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hidayat K., Du X., Chen G., Shi M., Shi B. Abdominal obesity and lung cancer risk: systematic review and meta-analysis of prospective studies. Nutrients. 2016;8(12):810. doi: 10.3390/nu8120810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Barberio A.M., Alareeki A., Viner B., et al. Central body fatness is a stronger predictor of cancer risk than overall body size. Nat Commun. 2019;10(1):383. doi: 10.1038/s41467-018-08159-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jeong S.M., Lee D.H., Giovannucci E.L. Predicted lean body mass, fat mass and risk of lung cancer: prospective US cohort study. Eur J Epidemiol. 2019;34(12):1151–1160. doi: 10.1007/s10654-019-00587-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Vanhove K., Graulus G.J., Mesotten L., et al. The metabolic landscape of lung cancer: new insights in a disturbed glucose metabolism. Front Oncol. 2019;9:1215. doi: 10.3389/fonc.2019.01215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Argirion I., Weinstein S.J., Männistö S., Albanes D., Mondul A.M. Serum insulin, glucose, indices of insulin resistance, and risk of lung cancer. Cancer Epidemiol Biomarkers Prev. 2017;26(10):1519–1524. doi: 10.1158/1055-9965.EPI-17-0293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lee J.-Y., Jeon I., Lee J.M., Yoon J.-M., Park S.M. Diabetes mellitus as an independent risk factor for lung cancer: a meta-analysis of observational studies. Eur J Cancer. 2013;49(10):2411–2423. doi: 10.1016/j.ejca.2013.02.025. [DOI] [PubMed] [Google Scholar]
- 43.de Beer J.C., Liebenberg L. Does cancer risk increase with HbA1c, independent of diabetes? Br J Cancer. 2014;110(9):2361–2368. doi: 10.1038/bjc.2014.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Goto A., Noda M., Sawada N., et al. High hemoglobin A1c levels within the non-diabetic range are associated with the risk of all cancers. Int J Cancer. 2016;138(7):1741–1753. doi: 10.1002/ijc.29917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yoshida Y., Chen Z., Baudier R.L., et al. Sex differences in the progression of metabolic risk factors in diabetes development. JAMA Netw Open. 2022;5(7) doi: 10.1001/jamanetworkopen.2022.22070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fuentes N., Silva Rodriguez M., Silveyra P. Role of sex hormones in lung cancer. Exp Biol Med (Maywood) 2021;246(19):2098–2110. doi: 10.1177/15353702211019697. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.