Abstract
Objectives
To evaluate the effect of menopausal hormone therapy (MHT) on skin aging in menopausal women.
Methods
Studies were identified by searching PubMed, Cochrane Library, Google Scholar, and ClinicalTrials.gov databases using the key words “hormone replacement therapy” AND “skin” AND “menopausal.” Studies that used and compared oral/transdermal MHT with placebo or another type of treatment in menopausal women aged 45–55 years were selected.
Results
From 1,526 studies identified, 15 studies comprising 1,589 patients were included in the final analysis. Our meta-analysis aimed to compare skin elasticity (standard [Std] mean difference = 0.28; 95% confidence interval [CI], 0.03–0.54; P = 0.03), skin thickness (Std. mean difference = 1.27; 95% CI, 0.88–1.66; P < 0.00001), collagen content (Std. mean difference = 2.01; 95% CI, 1.42–2.61; P < 0.00001), and skin dryness (Std. mean difference = 0.15; 95% CI, −0.05 to 0.35; P = 0.14).
Conclusions
MHT increases elasticity and collagen content in the skin, thereby reducing the severity of wrinkles and increasing skin thickness. Nevertheless, more well-conducted clinical trials are required to answer all questions in an evidence-based manner.
Keywords: Aging, Hormone replacement therapy, Menopause, Postmenopause, Skin
INTRODUCTION
The skin is a complex organ that reflects general health. However, there is little doubt that numerous variables determine the skin’s quality [1]. One of the factors of skin aging is hormonal changes in the body, particularly menopause.
Menopause is a typical mid-life event associated with diminished function of the ovaries that results in lower levels of sex steroids [2]. The skin is an estrogen-dependent organ, and during menopause, due to a decrease in the level of sex hormones such as progesterone, 17b-estradiol, dehydroepiandrosterone, hormone-insulin-like growth factor-I, growth hormone, also undergoes a process of aging. Microscopic examination of aging skin reveals disorganization of elastic fibers, a significant decrease in collagen fibers and a decrease in skin thickness. These changes lead to dryness and delayed healing of the skin, the appearance of wrinkles, which makes women worry about their appearance [3,4]. Menopausal hormone therapy (MHT) is used to prevent and treat symptoms and physical changes caused by estrogen deficiency, vasomotor symptoms, symptoms of genitourinary atrophy, postmenopausal osteopenia, and osteoporosis [5].
MHT is an effective therapy that offers more advantages than disadvantages for women aged less than 60 years or who have had menopause for less than 10 years.
Nevertheless, there is no unequivocal evidence-based data on safety and effectiveness of the use of MHT on the skin for its rejuvenation and healing.
Therefore, the purpose of this systematic review is to provide help by sharing accurate knowledge and treatment methods regarding MHT based on recent research findings.
MATERIALS AND METHODS
This study has been registered in the International Prospective Register of Systematic Reviews (PROSPERO) by the National Institute for Health Research (protocol and registration number: CRD42021288466. PROSPERO 2021) [6].
Our systematic review was conducted according to the updated PRISMA 2020 guidelines [7]. Institutional Review Board approval was not requested since the present study is a review.
PubMed, the Cochrane Library, ClinicalTrials.gov, and Google Scholar were systematically searched.
Three authors (J.A., M.L., and A.A.) independently conducted an initial screening of all articles’ title and abstract, and their reference lists were searched for additional potential studies. Any disagreements were resolved by the fourth author (L.P.) following an in-depth discussion.
This systematic review and meta-analysis include all clinical trials aimed at elucidating the effect of MHT on skin elasticity, age-related increase in skin elasticity, skin thickness and sebum, collagen content in the left shoulder during therapy in menopausal women. The studies include following types of participants: women aged 45–55 years in menopause. The studies that use and compare oral/transdermal MHT with placebo or another type of treatment in menopausal women. We included published and unpublished randomized controlled trials (RCTs) and non-randomized clinical trials. Only articles written in English were included.
The primary analysis was aimed to estimate the average levels of elasticity and firmness of the skin in patients receiving MHT.
Secondary analysis compared the age-related increase in skin extensibility, skin thickness and sebum, collagen content in the left shoulder against the background of MHT.
The outcomes were expressed as the skin biopsy before and after treatment, computerized suction device, elasticity measurement using an aspiration device, measurement of the thickness of the skin fold.
The search strategy in the electronic database PubMed was the following. Firstly, using the advanced search builder on PubMed, the following combination of the search terms was conducted: “hormone replacement therapy” AND “skin” AND “menopausal”. Again, no filters and limits were used.
The search was conducted using MeSH-terms on PubMed ((“Hormone Replacement Therapy/therapeutic use”[Mesh] OR “Hormone Replacement Therapy/therapy”[Mesh]) AND “skin elasticity”[Mesh])). The last date when the electronic database PubMed was searched is 3 January 2022.
The search on Cochrane was conducted using the advanced search and the following keywords: “hormonal replacement therapy” AND “skin” AND “postmenopausal”, no filters and limits were used. After that, all found articles were screened by title and abstract. The last step was a full-text analysis of the selected articles and the reference lists of the articles assessed for eligibility. The last date when the electronic database Cochrane was used is 3 January 2022.
The search using MeSH-terms was also conducted on the electronic database Cochrane library. The following MeSH descriptors were added using the advanced search: “hormonal replacement therapy”, “skin ageing”. Then reviewers screened all the articles in order to find a new one. The last date when the MeSH terms on the Cochrane library were searched is 13 December 2021.
The search was also conducted in the electronic database ClinicalTrials.gov using advanced search and the following search terms: “hormonal replacement therapy” AND “skin” AND “menopausal”. No filters and limits were used. All found articles were screened by title and protocol of the study. The last date when the electronic database ClinicalTrials.gov was searched is 3 January 2022.
The search strategy of the electronic database Google Scholar is represented. Firstly, the search was conducted using the advanced search and the following keywords: “hormone replacement therapy” AND “skin elasticity” AND “menopausal”. After that, all found articles were screened by title and abstract. The last step was a full-text analysis of the selected articles and the reference lists of the articles assessed for eligibility. The last date when the electronic database Cochrane was used is 3 January 2022.
A risk-of-bias assessment was conducted for each of the studies included using the Cochrane Handbook for Systematic Reviews of Interventions [8]. Three investigators (J.A., M.L., and A.A.) independently assessed the quality of the selected studies. A fourth investigator (L.P.) was involved when disagreements occurred. In accordance with the Cochrane Handbook for Systematic Reviews of Interventions, the RoB-2 tool [9] was used to assess the risk of bias in randomized controlled studies, and ROBINS-I [10] was used for non-randomized studies (prospective controlled, prospective cohort, retrospective studies, and other types of studies). In addition, these tools were also used to assess the risk of bias arising from reporting biases due to missing results in synthesis. The meta-analysis was done using RevMan 5.4. The quality of evidence was assessed according to the GRADE system.
RESULTS
The whole search strategy with the results is also presented in the flow-diagram (Fig. 1).
Fig. 1. PRISMA 2020 flow diagram. aConsider, if feasible to do so, reporting the number of records identified from each database or register searched (rather than the total number across all databases/registers). bIf automation tools were used, indicate how many records were excluded by a human and how many were excluded by automation tools. Adapted from the article of Page et al. (BMJ 2021;372:n71) [7].
Our electronic database search yielded 1,526 publications, 164 of which were duplicates and therefore excluded. One hundred sixty-six of them were review articles and also were excluded because, in our systematic review, we accepted only randomized and non-randomized clinical trials.
After that, 1,196 articles were analyzed, 1,161 of which were excluded by the titles and abstracts. Consequently, 35 publications were left for the full-text screening. All these articles were analyzed. Out of these 35 articles, only 10 were included in our qualitative analyses.
References of the studies included were additionally checked (n = 402) for appropriate publications. In this way, 5 studies were preselected for full-text screening. Therefore, a total of 15 studies were included in the final analysis, yielding a total of 1,589 patients, MHT group (n = 845) and control/another treatment group (n = 744).
The studies were grouped by outcomes (Table 1) [11,12,13,14,15,16,17,18,19,20,21,22,23,24,25] and by the form of administration (Table 2) [11,12,13,14,15,16,17,18,19,20,21,22,23,24,25].
Table 1. Description of articles included in the systematic review.
| Reference | Type of study | Participants/population | Intervention(s), exposure(s) | Comparator(s)/control | Outcome(s) | ||
|---|---|---|---|---|---|---|---|
| Skin elasticity | |||||||
| Piérard et al. (1995) [16] | Prospective cohort study | 114 women | MHT group (n = 46): oral MHT | • Second group (n = 43), non-menopausal women • Untreated group (n = 25), who were menopausal and receiving no MHT |
BE | ||
| • MHT group: 74.6 ± 14.3 • Untreated group: 79.9 ± 12.2 |
|||||||
| MD | |||||||
| • MHT group: 215.7 ± 88.3 • Untreated group: 287.6 ± 150.5 |
|||||||
| Piérard-Franchimont et al. (1999) [17] | A prospective longitudinal comparative trial | 140 postmenopausal women | MHT group (n = 90): oral MHT | • Control group (n = 50) | The regular impairment in BE with aging was significantly abated (P < 0.05) receiving MHT. | ||
| The MD increase over the 5 years in MHT-recipient women was less than half of that observed in nonrecipient subjects. This difference did not reach significance (P = 0.14). | |||||||
| Sumino et al. (2004) [11] | Observational study of convenience sample | 25 postmenopausal subjects | MHT group (n = 12): oral MHT | • Untreated group (n = 13), menopausal and out of MHT | Skin elasticity | ||
| • MHT group: from 55.9 ± 7.5 to 61.1 ± 8.7 • Untreated group: from 57.3 ± 7.7 to 56.2 ± 9.8 |
|||||||
| Sator et al. (2007) [12] | A prospective, randomized, double-blind, placebo-controlled study | 40 non-hysterectomized, postmenopausal women | MHT group (n = 20): oral MHT | • Placebo group (n = 20), received continuous treatment with blinded placebo tablets (without active ingredients). | Skin elasticity | ||
| • MHT group: gross elasticity, net elasticity and portion of elasticity, as compared to the complete curve at the skin covering the right mandibular ramus, increased significantly from baseline (P = 0.006, P = 0.002, and P = 0.005, respectively). The median values increased from 58% to 64%, 48% to 63%, and 35% to 39%, respectively. • Placebo group: patients showed no significant results for these measurements. |
|||||||
| Skin dryness | |||||||
| • MHT group: 0.11 ± 0.46 • Placebo group: 0.17 ± 0.38 |
|||||||
| Skin thickness | |||||||
| • MHT After 7 months, a significant increase in skin thickness (quartiles of percent of difference: 71.3%, 2.8%, and 8.5% after 6 weeks, and 0.0%, 6.4%, and 14.7% after 7 months of therapy) (P = 0.010). • Placebo group: did not cause any significant changes. |
|||||||
| Piérard et al. (2014) [13] | A prospective trial | 200 healthy Caucasian women | MHT group (n = 75): oral MHT | • Untreated group (n = 75), menopausal and out of MHT • Third group (n = 50), non-menopausal |
MD | ||
| • MHT group: 0.29 ± 0.08 • Untreated group: 0.34 ± 0.13 |
|||||||
| BE | |||||||
| • MHT group: 62.1 ± 0.09 • Untreated group: 42.1 ± 0.06 |
|||||||
| Phillips et al. (2008) [18] | A randomized, double-blind, double-dummy, placebo-controlled multicenter study | 485 subjects | MHT group (n = 162): oral MHT | • Placebo group (n = 165) | Skin elasticity | ||
| Measures did not reveal any significant change between baseline and 48 weeks, except for time shadows in the periorbital area, where there was statistically significant (P = 0.008), but clinically insignificant, change. | |||||||
| Face wrinkles | |||||||
| • MHT group: –0.56 ± 1.29 • Placebo group: –0.27 ± 1.14 |
|||||||
| Skin dryness | |||||||
| • MHT group: 0.5 ± 1.11 • Placebo group: 0.31 ± 1.16 |
|||||||
| Piérard et al. (2001) [14] | Comparative study | 120 healthy postmenopausal Caucasian women | The MHT group (n = 60): receiving oral or transdermal MHT | • Untreated group (n = 60) | MD | ||
| • MHT group: before 272.4 ± 153.2, after 277.6 ± 147.9 • Untreated group: before 357.5 ± 131.6, after 272.4 ± 153.2 |
|||||||
| BE | |||||||
| • MHT group: before 77.8 ± 14.5, after 52.9 ± 18.4 • Untreated group: before 67.4 ± 15.8, after 40.5 ± 17.9 |
|||||||
| Holzer et al. (2005) [15] | A double-blind, vehicle-controlled, randomized study | 40 women | MHT group (n = 20): transdermal MHT | • Placebo group (n = 20) were given the equivalent placebo cream containing just the vehicle. | Skin firmness | ||
| • MHT group 23.61% • Placebo group 13.24% |
|||||||
| Skin elasticity | |||||||
| • MHT group: from 0.290 ± 0.057 to 0.343 ± 0.096 • Placebo group: from 0.323 ± 0.12 to 0.336 ± 0.093 |
|||||||
| Skin dryness | |||||||
| • MHT group 0.50 ± 0.69 • Placebo group 0.32 ± 0.47 |
|||||||
| Haapasaari et al. (1997) [19] | Open, non-randomized parallel-groups study | 43 postmenopausal women | First group (n = 15): oral dose of 2 mg of 17 beta-estradiol and 1 mg of norethisterone acetate | • Third control group (n = 14) | Skin thickness | ||
| • Controls: 1.22 (1.03–1.99) • E+P: 1.42 (1.01–1.89) • E: 1.37 (1.17–1.63) |
|||||||
| Second group (n = 14): oral dose of 2 mg estradiol valerate daily | Skin elasticity | ||||||
| No histological or immunohistological changes were detected in the skin specimens during the 12-month treatment period compared to the baseline or to the skin specimens of the control group. | |||||||
| MD1-increased deformation of the skin under traction, BE-biological elasticity | |||||||
| Piérard et al. (1995) [16] | Prospective cohort study | 114 women | MHT group (n = 46): oral MHT | • Second group (n = 43), non-menopausal women • Untreated group (n = 25), who were menopausal and receiving no MHT |
BE | ||
| • MHT group: 74.6 ± 14.3 • Untreated group: 79.9 ± 12.2 |
|||||||
| MD | |||||||
| • MHT group: 215.7 ± 88.3 • Untreated group: 287.6 ± 150.5 |
|||||||
| Piérard-Franchimont et al. (1999) [17] | A prospective longitudinal comparative trial | 140 postmenopausal women | MHT group (n = 90): oral MHT | • Control group (n = 50) | The regular impairment in BE with aging was significantly abated (P < 0.05) receiving MHT. | ||
| The MD increase over the 5 years in MHT-recipient women was less than half of that observed in nonrecipient subjects. This difference did not reach significance (P = 0.14). | |||||||
| Piérard et al. (2014) [13] | A prospective trial | 200 healthy Caucasian women | MHT group (n = 75): oral MHT | • Untreated group (n = 75), menopausal and out of MHT • Third group (n = 50), non-menopausal |
MD | ||
| • MHT group: 0.29 ± 0.08 • Untreated group: 0.34 ± 0.13 |
|||||||
| BE | |||||||
| • MHT group: 62.1 ± 0.09 • Untreated group: 42.1 ± 0.06 |
|||||||
| Piérard et al. (2001) [14] | Comparative study | 120 healthy postmenopausal Caucasian women | The MHT group (n = 60): receiving oral or transdermal MHT | • Untreated group (n = 60) | MD | ||
| • MHT group: before 272.4 ± 153.2; after 277.6 ± 147.9 • Untreated group: before 357.5 ± 131.6; after 272.4 ± 153.2 |
|||||||
| BE | |||||||
| • MHT group: before 77.8 ± 14.5; after 52.9 ± 18.4 • Untreated group: before 67.4 ± 15.8; after 40.5 ± 17.9 |
|||||||
| Skin thickness | |||||||
| Fuchs et al. (2003) [22] | A prospective, randomized, double-blind study | 65 postmenopausal women | The estradiol group (n = 22): estradiol | • Combination group (n = 22), a combination of the estradiol and glycolic acid • Control group (n = 21), to glycolic acid |
Skin thickness | ||
| • The estradiol treatment: produced a 23% increase in (P = 0.0458) • Control group: 27% increase (P = 0.0467) • The combination: 38% increase (P = 0.00181) |
|||||||
| Creidi et al. (1994) [20] | Randomised, double-blind, parallel group study | 54 women | MHT group (n = 27): transdermal MHT | • Placebo group (n = 27), placebo cream | Skin thickness | ||
| • MHT group: from 1.56 ± 0.20 mm to 1.68 ± 0.19 mm • Placebo group: from 1.52 ± 0.20 mm to 1.59 ± 0.19 mm |
|||||||
| Face wrinkles | |||||||
| • MHT group: –0.5 ± 0.6 • Placebo group: 0 ± 0.7 |
|||||||
| Haapasaari et al. (1997) [19] | Open, non-randomized parallel-groups study | 43 postmenopausal women | First group (n = 15): oral dose of 2 mg of 17 beta-estradiol and 1 mg of norethisterone acetate | • Third control group (n = 14) | Skin thickness | ||
| • Controls : 1.22 (1.03–1.99) • E+P : 1.42 (1.01–1.89) • E : 1.37 (1.17–1.63) |
|||||||
| Second group (n = 14): oral dose of 2 mg estradiol valerate daily | Skin elasticity | ||||||
| No histological or immunohistological changes were detected in the skin specimens during the 12-month treatment period compared to the baseline or to the skin specimens of the control group. | |||||||
| Maheux et al. (1994) [21] | Randomized, double-blind, placebo-controlled study | 60 postmenopausal women | MHT group (n = 30): oral MHT | • Placebo group (n = 30) | Skin thickness | ||
| • MHT group: 2.44 ± 0.06 mm • Placebo group: 2.41 ± 0.07 mm |
|||||||
| After 6 months | |||||||
| • MHT group: 2.55 ± 0.06 mm • Placebo group: 2.44± 0.07 mm |
|||||||
| After 12 months | |||||||
| • MHT group: 2.50 ± 0.06 mm • Placebo group: 2.73 ± 0.07 mm |
|||||||
| Sauerbronn et al. (2000) [23] | Randomized, double-blind, placebo-controlled study | 38 postmenopausal women | MHT group (n = 19): oral MHT | • Placebo group (n = 19) control received 21 tablets of placebo | Collagen content | ||
| • MHT group: from 21,897.4 (S.D. 1,635.33) to 23,318.2 (S.D. 2,027.6) • Placebo group: from 22,014.6 (S.D. 1,858.1) to 22,057.2 (S.D. 2,405.7) |
|||||||
| Skin thickness | |||||||
| There were no significant differences between baseline and 6-month treatment in both groups. | |||||||
| Sator et al. (2007) [12] | A prospective, randomized, double-blind, placebo-controlled study | 40 non-hysterectomized, postmenopausal women | MHT group (n = 20): oral MHT | • Placebo group (n = 20) received continuous treatment with blinded placebo tablets (without active ingredients) | Skin elasticity | ||
| • MHT group: gross elasticity, net elasticity and portion of elasticity, as compared to the complete curve at the skin covering the right mandibular ramus, increased significantly from baseline (P = 0.006, P = 0.002, and P = 0.005, respectively). The median values increased from 58% to 64%, 48% to 63%, and 35% to 39%, respectively. • Placebo group: patients showed no significant results for these measurements. |
|||||||
| Skin dryness | |||||||
| • MHT group: 0.11 ± 0.46 • Placebo group: 0.17 ± 0.38 |
|||||||
| Skin thickness | |||||||
| • MHT after 7 months, a significant increase in skin thickness (quartiles of percent of difference: 71.3%, 2.8%, and 8.5% after 6 weeks, and 0.0%, 6.4%, and 14.7% after 7 months of therapy) (P = 0.010). • Placebo group: did not cause any significant changes. |
|||||||
| Collagen content | |||||||
| Sauerbronn et al. (2000) [23] | Randomized, double-blind, placebo-controlled study | 38 postmenopausal women | MHT group (n = 19): oral MHT | • Placebo group (n = 19) control received 21 tablets of placebo | Collagen content | ||
| • MHT group: from 21,897.4 (S.D. 1,635.33) to 23,318.2 (S.D. 2,027.6) • Placebo group: from 22,014.6 (S.D. 1,858.1) to 22,057.2 (S.D. 2,405.7) |
|||||||
| Skin thickness | |||||||
| • There were no significant differences between baseline and 6-month treatment in both groups. | |||||||
| Castelo-Branco et al. (1992) [24] | A randomized, placebo-controlled study | 118 women | First group (n = 28): 0.625 mg/day conjugated equine oestrogens (CEE) over a 25-day cycle each month. | • Untreated group (n = 30), no treatment | Collagen content | ||
| • 1 group: collagen at 0 month (115.1 ± 0.8) vs. collagen after 12 months (117.1 ± 0.7); + 1.8% • 2 group: collagen at 0 month (114.0 ± 1.2) vs. collagen after 12 months (119.8 ± 0.6); + 5.1% • 3 group: collagen at 0 month (114.0 ± 0.9) vs. collagen after 12 months (117.8 ± 1.0); + 3.0%, P < 0.05 • Untreated group: collagen at 0 month (116.5 ± 1.1) vs. collagen after 12 months (112.8 ± 0.9); –3.2%, P < 0.05 |
|||||||
| Second group (n = 28): 50-day transdermal 17/3-oestradiol over a 24-day cycle each month | |||||||
| Third group (n = 32): 0.625 mg/day CEE every day of the month. | |||||||
| Effects of raloxifene and MHT on forearm skin elasticity | |||||||
| Sumino et al. (2009) [25] | Prospective cohort study | 47 Japanese postmeno-pausal women | MHT group (n = 19): transdermal MHT | • A raloxifene group (n = 17) received continuous raloxifene treatment (60 mg/day); a control group (n = 11) women did not receive either therapy | Skin elasticity | ||
| • Raloxifene group: from 52.4% ± 3.8% to 55.1% ± 4.7% • MHT group: from 64.1% ± 7.2% to 67.4% ± 7.4% • Control group: from 55.8% ± 5.8% to 52.7% ± 8.3% |
|||||||
| Skin firmness | |||||||
| Holzer et al. (2005) [15] | A double-blind, vehicle-controlled, randomized study | 40 women | MHT group (n = 20): oral MHT | • Placebo group (n = 20) were given the equivalent placebo cream containing just the vehicle | Skin firmness | ||
| • MHT group: 23.61% • Placebo group: 13.24% |
|||||||
| Skin elasticity | |||||||
| • MHT group: from 0.290 ± 0.057 to 0.343 ± 0.096 • Placebo group: from 0.323 ± 0.120 to 0.336 ± 0.093 |
|||||||
| Skin dryness | |||||||
| • MHT group: 0.50 ± 0.69 • Placebo group: 0.32 ± 0.47 |
|||||||
MHT: menopausal hormone therapy, MD: mean difference, BE: biological elasticity, E: estradiol, P: progesterone, S.D.: standart deviation.
Table 2. Forms of administration of menopausal hormone therapy.
| Article | Form | Dosage |
|---|---|---|
| Piérard et al. (1995) [16] | Oral MHT | Estrogens 0.625 mg/day + medrogestone 5 mg/day |
| Piérard-Franchimont et al. (1999) [17] | Oral MHT | Conjugated estrogens 0.625 mg daily with medrogestone 5 mg daily |
| Sumino et al. (2004) [11] | Oral MHT | Conjugated estrogens 0.625 mg equine estrogen combined with 2.5 mg of medroxyprogesterone acetate |
| Sator et al. (2007) [12] | Oral MHT | 17b-estadiol 2 mg day for 21 daysand cyprotrone acetate 1 mg day for 10 days |
| Sauerbronn et al. (2000) [23] | Oral MHT | Valerate estadiol 2 mg day for 21 days and cyproterone acetate 1 mg day for 10 days |
| Piérard et al. (2014) [13] | Oral MHT | Estradiol 1 mg |
| Phillips et al. (2008) [18] | Oral MHT | 1 mg norethidrone acetate 5 microg ethinyl esradiol or 1 mg norethidrone acetate /10 microg ethinyl esradiol |
| Maheux et al. (1994) [21] | Oral MHT | 0.625 mg of conjugated estrogens |
| Haapasaari et al. (1997) [19] | Oral MHT | Oral esrogen and progestin regimen with a daily dose of 2 mg of 17b - estradiol and 1 mg of norethistine acetate |
| Piérard et al. (2001) [14] | Receiving oral or transdermal MHT | N/A |
| Holzer et al. (2005) [15] | Transdermal MHT | Cream containing 2% progesterone |
| Sumino et al. (2009) [25] | Transdermal MHT | Transdermal MHT |
| 17-beta estradiol 0.72 mg/2 days plus cyclic medroxyprogesterone acetate and raloxifene 60 mg/day | ||
| Creidi et al. (1994) [20] | Transdermal MHT | Premaarin cream (0.625 mg conjugated oeastagens per gram of cream) |
| Castelo-Branco et al. (1992) [24] | Transdermal MHT | 1 group: 0.625 mg/day conjugated equine estrogens |
| 2 group: 50 days transdermal 17b-oestradiol | ||
| 3 group: 0.625 mg/day CEE every day of the month | ||
| Fuchs et al. (2003) [22] | Transdermal MHT | 1 group: estradiol 2 group: combination of the estadiol and glycolic acid |
MHT: menopausal hormone therapy, N/A: not applicable.
Skin elasticity
So, 6 non-randomized clinical trials and 3 RCTs were aimed at assessing skin elasticity [11,12,13,14,15,16,17,18,19]. Sumino et al. [11], Sator et al. [12], Piérard et al. [13], Piérard et al. [14], Holzer et al. [15], and Piérard et al. [16] have shown that the use of MHT increases skin elasticity. Piérard-Franchimont et al. [17], Phillips et al. [18], and Haapasaari et al. [19] did not see a significant effect of MHT on skin elasticity.
Four studies by Piérard et al. [14], Holzer et al. [15], Piérard et al. [16], and Sumino et al. [25] were included in quantitative synthesis (meta-analysis) that report skin elasticity in 251 patients. They compared improvement of skin elasticity between 2 groups: MHT and controls (standard [Std] mean difference = 0.28; 95% CI, 0.03–0.54; P = 0.03). The heterogeneity for this comparison was 76%. Consequently, there was a statistically significant difference in both groups in favor of MHT group (Fig. 2).
Fig. 2. Meta-analysis of skin elasticity in groups: menopausal hormone therapy and controls. HRT: hormone replacement therapy, IV: instrumental variables, Std: standard, SD: standard deviation, CI: confidence interval.
Increased deformation of the skin under traction and biological elasticity
The computerized aspiration device was used to measure the tensile properties of the skin. At each evaluation point, the maximum vertical skin strain (MD, Uf), representing the stiffness of the skin, was measured after a progressive suction force was applied at a rate of 25 mbar·s1 for 20 seconds. Biological elasticity (BE, %) was obtained after BE = 102 (MD-RD)/MD. The viscoelastic coefficient during the suction phase (VER, %) was calculated after VER = Uv/Ue. In the progressive suction mode, an increasing negative pressure was applied at a linear rate of 25 mbar/s for 20 seconds, after which the vacuum was released at the same rate. This was recorded as a stress/strain relationship.
Piérard et al. [16] in their prospective cohort study observed that in the 39 untreated women who completed the study experienced over the 5-year follow-up a trend in increase of MD (P = 0.08) whereas minor changes (P = 0.24) were present in the 35 MHT-patients. The most significant relative changes with age were found in BE. This variable decreased significantly (P < 0.05) in nonrecipient subjects while few changes (P = 0.13) were seen in women receiving MHT.
Piérard-Franchimont et al. [17] found that there was no significant difference was seen between the tensile values in the 2 groups of women at baseline. The MD increase over the 5 years in MHT-recipient women was less than half of that observed in nonrecipient subjects. This difference did not reach significance (P = 0.14). In contrast, the regular impairment in BE with aging was significantly abated (P < 0.05) for both variables in women receiving MHT. The benefit in the BE gain was prominent (P < 0.001) in the subgroup of good MHT responders as defined above. The BE significantly decreased out of MHT [12,13].
Skin thickness
Five RCTs and one non-randomized study compared skin thickness [12,20,21,22,23].
Sator et al. [12], Creidi et al. [20], Maheux et al. [21], and Fuchs et al. [22] revealed an increase in skin thickness in the treated groups instead of the control groups.
However, in the other 2 studies by Haapasaari et al. [19] and Sauerbronn et al. [23], there were no significant differences between the baseline level of skin thickness and 6-month treatment in both groups.
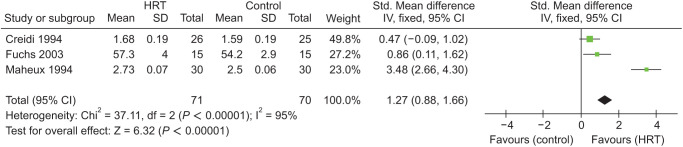
Meta-analysis of skin thickness included 3 trials by Creidi et al. [20], Maheux et al. [21], and Fuchs et al. [22] evaluating 141 patients in 2 groups: MHT and controls (Std. mean difference = 1.27; 95% CI, 0.88–1.66; P < 0.00001). The heterogeneity for this comparison was 95%. According to this quantitative synthesis, there was a statistically significant difference in both groups in favor of MHT group (Fig. 3).
Fig. 3. Meta-analysis of skin thickness in groups. HRT: hormone replacement therapy, IV: instrumental variables, Std: standard, SD: standard deviation, CI: confidence interval.
Collagen content
Two RCTs compared collagen content [23,24]. In a randomized, double-blind, placebo-controlled study by Sauerbronn et al. [23], there was no difference at baseline in both groups concerning collagen fibers content (P < 0.05). After 6 months of treatment, there was a statistically significant increase of collagen content in group MHT, compared to baseline. There were no changes in group placebo [23]. When the skin collagen content after 12 months of treatment was assessed in a randomized, placebo-controlled study published by Castelo-Branco et al. [24] an increase was observed in all the MHT groups, whereas the control group, as expected, showed a significant decrease of almost 3.2% (P < 0.05).
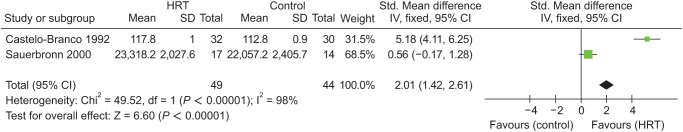
Only 2 studies by Sauerbronn et al. [23] and Castelo-Branco et al. [24] were eligible for meta-analysis of collagen content and assessed 93 patients (Std. mean difference = 2.01; 95% CI, 1.42–2.61; P < 0.00001). The heterogeneity for this comparison was 98%. Hence, there was a statistically significant difference in both groups in favor of MHT group (Fig. 4) [23,24].
Fig. 4. Meta-analysis of collagen content in groups. HRT: hormone replacement therapy, IV: instrumental variables, Std: standard, SD: standard deviation, CI: confidence interval.
Effects of raloxifene and MHT on forearm skin elasticity
Sumino et al. [25] in their prospective cohort study compared the effects of raloxifene and MHT on forearm skin elasticity in postmenopausal women. Raloxifene and MHT significantly increased skin elasticity from 52.4% ± 3.8% and 64.1% ± 7.2% at baseline to 55.1% ± 4.7% and 67.4% ± 7.4% after 12 months, respectively (P < 0.05 for both), but the control group did not exhibit any significant change in skin elasticity of the skin during the study period. At 12 months, skin elasticity was significantly higher in the MHT group than in the control and raloxifene groups (P < 0.01 for both) [25].
Skin firmness
Holzer et al. [15] showed that the increase in skin firmness was significantly more significant (P = 0.031) in the treatment group than in the control group (23.61% vs. 13.24%). At the end of treatment, a more significant reduction in wrinkle depth (9.72% vs. 7.35%) around the right eye was observed in the treatment group (P > 0.05) [15].
Face wrinkles
In a randomized, double-blind, parallel-group study by Creidi et al. [20] others found that skin thickness in women using Premarin cream increased from 1.56 ± 0.20 mm at baseline to 1.68 ± 0.19 mm compared to 1.52 ± 0.20 mm at baseline to 1.59 ± 0.19 mm in the placebo group. Premarin cream was more effective than placebo cream in reducing fine lines at 12 and 24 weeks (P = 0.010 and P = 0.012). However, the statistical significance was not very high [20].
However, another randomized, double-blind study of skin wrinkle by Phillips et al. [18] severity at 24 and 48 weeks showed no significant change from baseline.
The meta-analysis also added the assessment of wrinkles that included only 2 trials by Phillips et al. [18] and Creidi et al. [20] with 378 patients in 2 groups: MHT and Control (Std. mean difference = −0.30; 95% CI, −0.51 to −0.10; P = 0.003). The heterogeneity for this comparison was 64%. There were fewer wrinkles in the MHT group with statistically significant difference (Fig. 5).
Fig. 5. Meta-analysis of face wrinkles in groups. HRT: instrumental variables, IV: instrumental variables, Std: standard, SD: standard deviation, CI: confidence interval.
Skin dryness
A prospective, randomized, double-blind, placebo-controlled study by Sator et al. [12] of 40 postmenopausal women with an intact uterus were included in the study. After 7 months of estradiol/dydrogesterone therapy, the dermatological evaluation showed a significant decrease from baseline concerning the variables of dryness.
In another 2 studies by Holzer et al. [15] and Phillips et al. [18] differences did not reach statistical significance (P > 0.05).
Three studies by Sator et al. [12], Holzer et al. [15], and Phillips et al. [18] evaluated skin dryness in 399 patients (Std. mean difference = 0.15; 95% CI, −0.05 to 0.35; P = 0.14). The heterogeneity for this comparison was 0%. Thus, there was no statistically significant difference in skin dryness between 2 groups (Fig. 6) [11,14,17].
Fig. 6. Meta-analysis of skin dryness in groups. HRT: hormone replacement therapy, IV: instrumental variables, Std: standard, SD: standard deviation, CI: confidence interval.
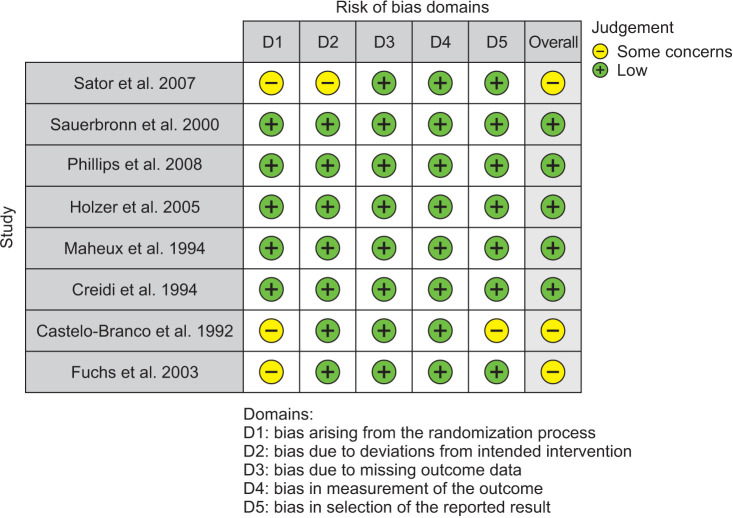
As for risk of bias assessment, 3 reviewers (J.A., M.L., and A.A.) assessed the risk of bias of each of the studies included in the systematic review according to the Cochrane Handbook using RoB-2 for randomized control trials and ROBINS-I for non-randomized trials. The RoB-2 tool included bias arising from the randomization process due to deviations from intended interventions, missing outcome data, bias in the measurement of the outcome, and bias in selecting the reported result. Each item was classified as high, of some concern, or indicating a low risk of bias. The ROBINS-I tool included bias due to confounding, bias in the selection of study participants, bias in the classification of interventions, deviations from intended interventions, bias due to missing data, bias in the measurement of outcomes, and bias in the selection of the reported results. Each item was classified as critical, serious, or indicating a low risk of bias. Any disagreements were resolved by a fourth reviewer (L.P.).
Visualization tools were created by the ROBVIS app [26]. This app created “traffic light” plots of the domain-level judgements for each result and weighted bar plots of the distribution of risk-of-bias judgments within each bias domain. The overall risk of bias for non-randomized trials was moderate in 70% and low in 30%, according to the ROBINS-I tool (Fig. 7) and based on the RoB-2 tool (Fig. 8), randomized trials had 60% of low, 40% had some concerns regarding the overall risk of bias.
Fig. 7. ROBINS-I tool for risk of bias assessment.
Fig. 8. RoB-2 tool for risk of bias assessment.
DISCUSSION
Most changes in the ageing face are related to environmental factors, genetics, nutrition and other reasons. Exposure to sunlight hastens these changes, and protection from the sun is the only proven way to delay them. Tobacco smoke is toxic to cells and should be avoided to prevent accelerating skin ageing. The changes of the elderly skin are normal part of ageing but may induce a higher risk for serious skin complications. An increasing part of the elderly population feels disturbed about the age-dependent signs of the skin and wishes therapeutic intervention. Despite the significant improvement that can be achieved with most anti-ageing therapies, no technique is perfect [27].
The quality of the skin and the menopausal process are closely linked. Regardless of the time in one’s life, women desire to feel good about their skins. The mechanism of skin ageing is based on the accumulation of excess radical forms of oxygen, which accumulate during anaerobic metabolism [28]. Also, a low level of sex hormones is manifested by a slowdown in skin regeneration, which is associated at the molecular level with an imbalance in protein balance, impaired transduction of cytokines and uncontrolled inflammation. To achieve this, cosmetics and cosmeceuticals have been developed. A good quality treatment may increase skin density, thickness, hydration and firmness, and a decrease in wrinkles [29].
Estrogen receptors have been detected in many skin elements, including keratinocytes, melanocytes, fibroblasts, hair follicles, and sebaceous glands, so the withdrawal of estrogen at menopause will likely have measurable effects on skin health. Studies have shown that after menopause, skin thins and there is a loss of viscoelasticity. Skin surface texture, water holding capacity, collagen content of the dermis, and viscoelasticity have shown improvement with estrogen use [30].
Therapy with the transdermal form of MHT has advantages over the oral form. In particular, the risk of ischemic stroke is associated with oral administration, however, the use of low doses has a lower risk, with a transdermal form of risk does not occur, which suggests a primary thrombotic mechanism. There was also no increase in the risk of venous thromboembolism (VTE) during transdermal therapy. The use of transdermal estrogen in combination with progesterone may be safer against VTE, especially in women at high risk of VTE. Oral administration of estrogens significantly increases fat mass and reduces muscle mass, while taking the transdermal form does not affect these indicators [30,31,32,33,34].
Many health care professionals do not prescribe MHT due to the risk of melanoma. Across hormone therapy types, there does not appear to be a well-established association between exogenous female hormones and melanoma incidence. However, MHT practices and formulations vary significantly across countries. Although MHT does not increase melanoma risk in studies from the United States, conflicting results have been observed in Europe. Unopposed estrogen MHT formulations require further investigation to determine a clear pattern between hormone use and melanoma developent [35].
As far as we know, we conducted the first systematic review and meta-analysis that showed that MHT slows down the skin ageing process.
While not claiming that MHT can or should ever be considered as an independent treatment for skin ageing, we still believe that our results are interesting as they indicate a beneficial effect of MHT on the skin, despite studies showing a negative effect of MHT on other organs.
As for the limitations of our study, firstly, our meta-analysis included studies of moderate risk of bias. Secondly, the lack of standardized outcome measures between studies resulted in high heterogeneity in our meta-analysis. Finally, more well-conducted prospective and randomized trials are needed to increase the evidence and answer all questions regarding MHT in menopausal women completely. While the systemic benefits of transdermal MHT are clear, further studies are needed to access the effect of transdermal MHT on skin condition and rejuvenation.
Implications for future research may include a well-performed study design with standard outcome measures and a complete follow-up period that will help scientists and healthcare professionals be more confident in choosing different options for the MHT and understand which regimen is more effective.
Based on our systematic review and meta-analysis results, we can conclude that MHT increases elasticity and collagen content in the skin, thereby reducing the severity of wrinkles increasing skin thickness and firmness. Thus, the use of MHT can be used not only to treat symptoms of menopause but also to become a new direction in rejuvenation and revitalization of the skin in women during menopause.
Footnotes
FUNDING: No funding to declare.
CONFLICT OF INTEREST: No potential conflict of interest relevant to this article was reported.
References
- 1.Brincat MP. Hormone replacement therapy and the skin. Maturitas. 2000;35:107–117. doi: 10.1016/s0378-5122(00)00097-9. [DOI] [PubMed] [Google Scholar]
- 2.Kagan R, Kellogg-Spadt S, Parish SJ. Practical treatment considerations in the management of genitourinary syndrome of menopause. Drugs Aging. 2019;36:897–908. doi: 10.1007/s40266-019-00700-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Braverman IM, Fonferko E. Studies in cutaneous aging: I. The elastic fiber network. J Invest Dermatol. 1982;78:434–443. doi: 10.1111/1523-1747.ep12507866. [DOI] [PubMed] [Google Scholar]
- 4.Lovell CR, Smolenski KA, Duance VC, Light ND, Young S, Dyson M. Type I and III collagen content and fibre distribution in normal human skin during ageing. Br J Dermatol. 1987;117:419–428. doi: 10.1111/j.1365-2133.1987.tb04921.x. [DOI] [PubMed] [Google Scholar]
- 5.Lee SR, Cho MK, Cho YJ, Chun S, Hong SH, Hwang KR, et al. The 2020 menopausal hormone therapy guidelines. J Menopausal Med. 2020;26:69–98. doi: 10.6118/jmm.20000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Unanyan A, Pivazyan L, Avetisyan J, Loshkareva M, Abdurakhmanova A, Ishchenko A, et al. Effect of menopausal hormone therapy on skin elasticity in women: a systematic review and meta-analysis. National Institute for Health and Care Research; 2021. [cited 2022 Jan 26]. Available from: https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=288466. [Google Scholar]
- 7.Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi: 10.1136/bmj.n71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane handbook for systematic reviews of interventions version 6.1 (updated September 2020) Cochrane; 2020. [cited 2022 Jan 13]. Available from: https://training.cochrane.org/handbook/archive/v6.1. [Google Scholar]
- 9.Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. doi: 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 10.Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919. doi: 10.1136/bmj.i4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sumino H, Ichikawa S, Abe M, Endo Y, Ishikawa O, Kurabayashi M. Effects of aging, menopause, and hormone replacement therapy on forearm skin elasticity in women. J Am Geriatr Soc. 2004;52:945–949. doi: 10.1111/j.1532-5415.2004.52262.x. [DOI] [PubMed] [Google Scholar]
- 12.Sator PG, Sator MO, Schmidt JB, Nahavandi H, Radakovic S, Huber JC, et al. A prospective, randomized, double-blind, placebo-controlled study on the influence of a hormone replacement therapy on skin aging in postmenopausal women. Climacteric. 2007;10:320–334. doi: 10.1080/13697130701444073. [DOI] [PubMed] [Google Scholar]
- 13.Piérard GE, Hermanns-Lê T, Paquet P, Piérard-Franchimont C. Skin viscoelasticity during hormone replacement therapy for climacteric ageing. Int J Cosmet Sci. 2014;36:88–92. doi: 10.1111/ics.12100. [DOI] [PubMed] [Google Scholar]
- 14.Piérard GE, Vanderplaetsen S, Piérard-Franchimont C. Comparative effect of hormone replacement therapy on bone mass density and skin tensile properties. Maturitas. 2001;40:221–227. doi: 10.1016/s0378-5122(01)00241-9. [DOI] [PubMed] [Google Scholar]
- 15.Holzer G, Riegler E, Hönigsmann H, Farokhnia S, Schmidt JB. Effects and side-effects of 2% progesterone cream on the skin of peri- and postmenopausal women: results from a double-blind, vehicle-controlled, randomized study. Br J Dermatol. 2005;153:626–634. doi: 10.1111/j.1365-2133.2005.06685.x. Erratum in: Br J Dermatol 2005; 153: 1092. [DOI] [PubMed] [Google Scholar]
- 16.Piérard GE, Letawe C, Dowlati A, Piérard-Franchimont C. Effect of hormone replacement therapy for menopause on the mechanical properties of skin. J Am Geriatr Soc. 1995;43:662–665. doi: 10.1111/j.1532-5415.1995.tb07202.x. [DOI] [PubMed] [Google Scholar]
- 17.Piérard-Franchimont C, Cornil F, Dehavay J, Deleixhe-Mauhin F, Letot B, Piérard GE. Climacteric skin ageing of the face--a prospective longitudinal comparative trial on the effect of oral hormone replacement therapy. Maturitas. 1999;32:87–93. doi: 10.1016/s0378-5122(99)00019-5. [DOI] [PubMed] [Google Scholar]
- 18.Phillips TJ, Symons J, Menon S HT Study Group. Does hormone therapy improve age-related skin changes in postmenopausal women? A randomized, double-blind, double-dummy, placebo-controlled multicenter study assessing the effects of norethindrone acetate and ethinyl estradiol in the improvement of mild to moderate age-related skin changes in postmenopausal women. J Am Acad Dermatol. 2008;59:397–404.e3. doi: 10.1016/j.jaad.2008.05.009. [DOI] [PubMed] [Google Scholar]
- 19.Haapasaari KM, Raudaskoski T, Kallioinen M, Suvanto-Luukkonen E, Kauppila A, Läärä E, et al. Systemic therapy with estrogen or estrogen with progestin has no effect on skin collagen in postmenopausal women. Maturitas. 1997;27:153–162. doi: 10.1016/s0378-5122(97)01128-6. [DOI] [PubMed] [Google Scholar]
- 20.Creidi P, Faivre B, Agache P, Richard E, Haudiquet V, Sauvanet JP. Effect of a conjugated oestrogen (Premarin) cream on ageing facial skin. A comparative study with a placebo cream. Maturitas. 1994;19:211–223. doi: 10.1016/0378-5122(94)90074-4. [DOI] [PubMed] [Google Scholar]
- 21.Maheux R, Naud F, Rioux M, Grenier R, Lemay A, Guy J, et al. A randomized, double-blind, placebo-controlled study on the effect of conjugated estrogens on skin thickness. Am J Obstet Gynecol. 1994;170:642–649. doi: 10.1016/s0002-9378(94)70242-x. [DOI] [PubMed] [Google Scholar]
- 22.Fuchs KO, Solis O, Tapawan R, Paranjpe J. The effects of an estrogen and glycolic acid cream on the facial skin of postmenopausal women: a randomized histologic study. Cutis. 2003;71:481–488. [PubMed] [Google Scholar]
- 23.Sauerbronn AV, Fonseca AM, Bagnoli VR, Saldiva PH, Pinotti JA. The effects of systemic hormonal replacement therapy on the skin of postmenopausal women. Int J Gynaecol Obstet. 2000;68:35–41. doi: 10.1016/s0020-7292(99)00166-6. [DOI] [PubMed] [Google Scholar]
- 24.Castelo-Branco C, Duran M, González-Merlo J. Skin collagen changes related to age and hormone replacement therapy. Maturitas. 1992;15:113–119. doi: 10.1016/0378-5122(92)90245-y. [DOI] [PubMed] [Google Scholar]
- 25.Sumino H, Ichikawa S, Kasama S, Takahashi T, Kumakura H, Takayama Y, et al. Effects of raloxifene and hormone replacement therapy on forearm skin elasticity in postmenopausal women. Maturitas. 2009;62:53–57. doi: 10.1016/j.maturitas.2008.10.005. [DOI] [PubMed] [Google Scholar]
- 26.McGuinness LA, Higgins JPT. Risk-of-bias VISualization (robvis): an R package and Shiny web app for visualizing risk-of-bias assessments. Res Synth Methods. 2021;12:55–61. doi: 10.1002/jrsm.1411. [DOI] [PubMed] [Google Scholar]
- 27.Wiegand C, Raschke C, Elsner P. In: Textbook of aging skin. Farage MA, Miller KW, Maibach HI, editors. Springer; 2017. Skin aging: a brief summary of characteristic changes. [Google Scholar]
- 28.Zouboulis CC, Makrantonaki E. Hormonal therapy of intrinsic aging. Rejuvenation Res. 2012;15:302–312. doi: 10.1089/rej.2011.1249. [DOI] [PubMed] [Google Scholar]
- 29.Herman J, Rost-Roszkowska M, Skotnicka-Graca U. Skin care during the menopause period: noninvasive procedures of beauty studies. Postepy Dermatol Alergol. 2013;30:388–395. doi: 10.5114/pdia.2013.39430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Baber RJ, Panay N, Fenton A IMS Writing Group. 2016 IMS Recommendations on women's midlife health and menopause hormone therapy. Climacteric. 2016;19:109–150. doi: 10.3109/13697137.2015.1129166. [DOI] [PubMed] [Google Scholar]
- 31.Burger HG, Hale GE, Robertson DM, Dennerstein L. A review of hormonal changes during the menopausal transition: focus on findings from the Melbourne Women's Midlife Health Project. Hum Reprod Update. 2007;13:559–565. doi: 10.1093/humupd/dmm020. [DOI] [PubMed] [Google Scholar]
- 32.Overlie I, Mørkrid L, Andersson AM, Skakkebaek NE, Moen MH, Holte A. Inhibin A and B as markers of menopause: a five-year prospective longitudinal study of hormonal changes during the menopausal transition. Acta Obstet Gynecol Scand. 2005;84:281–285. doi: 10.1111/j.0001-6349.2005.00490.x. [DOI] [PubMed] [Google Scholar]
- 33.Reus TL, Brohem CA, Schuck DC, Lorencini M. Revisiting the effects of menopause on the skin: functional changes, clinical studies, in vitro models and therapeutic alternatives. Mech Ageing Dev. 2020;185:111193. doi: 10.1016/j.mad.2019.111193. [DOI] [PubMed] [Google Scholar]
- 34.de Villiers TJ, Hall JE, Pinkerton JV, Cerdas Pérez S, Rees M, Yang C, et al. Revised global consensus statement on menopausal hormone therapy. Climacteric. 2016;19:313–315. doi: 10.1080/13697137.2016.1196047. [DOI] [PubMed] [Google Scholar]
- 35.Hill MS, Cartron AM, Burgoyne M, Driscoll MS. Hormone therapy and melanoma in women. Int J Womens Dermatol. 2021;7(5Part B):692–696. doi: 10.1016/j.ijwd.2021.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]