Abstract
Purpose
This study estimated the magnitude and duration of risk of cardiovascular events and mortality following acute exacerbations of chronic obstructive pulmonary disease (AECOPD), and whether risks varied by number and severity of exacerbation in a commercially insured population in the United States.
Methods
This was a retrospective cohort study of newly diagnosed COPD patients ≥40 years old in the Healthcare Integrated Research Database from 2012 to 2019. Patients experiencing exacerbations comprised the “exacerbation cohort”. Moderate exacerbations were outpatient visits with contemporaneous antibiotic or glucocorticoid administration; severe exacerbations were emergency department visits or hospitalizations for AECOPD. Follow-up started on the exacerbation date. Distribution of time between diagnosis and first exacerbation was used to assign index dates to the “unexposed” cohort. Cox proportional hazards models estimated risks of a cardiovascular event or death following an exacerbation adjusted for medical and prescription history and stratified by follow-up time, type of cardiovascular event, exacerbation severity, and rank of exacerbation (first, second, or third).
Results
Among 435,925 patients, 170,236 experienced ≥1 exacerbation. Risk of death was increased for 2 years following an exacerbation and was highest during the first 30 days (any exacerbation hazard ratio (HR)=1.79, 95% CI=1.58—2.04; moderate HR=1.22, 95% CI=1.04—1.43; severe HR=5.09, 95% CI=4.30—6.03). Risks of cardiovascular events were increased for 1 year following an AECOPD and highest in the first 30-days (any exacerbation HR=1.34, 95% CI=1.23—1.46; moderate HR=1.23 (95% CI 1.12—1.35); severe HR=1.93 (95% CI=1.67—2.22)). Each subsequent AECOPD was associated with incrementally higher rates of both death and cardiovascular events.
Conclusion
Risk of death and cardiovascular events was greatest in the first 30 days and rose with subsequent exacerbations. Risks were elevated for 1–2 years following moderate and severe exacerbations, highlighting a sustained increased cardiopulmonary risk associated with exacerbations.
Keywords: COPD, cardiopulmonary, cardiac events, epidemiology, longitudinal studies
Introduction
Chronic obstructive pulmonary disease (COPD) is diagnosed in approximately 6% of Americans and is most prevalent in states along the Ohio and Mississippi Rivers where diminished access to care, poverty, tobacco use, and opioid use have led to disproportionately higher rates of lung disease.1,2 Because COPD usually progresses slowly and access to spirometry is not available everywhere (and may not be sought), the true prevalence may be higher.3,4 Acute exacerbations of COPD (AECOPD) occur when symptoms worsen abruptly and significantly.5,6 During an exacerbation, patients may seek medical care and require additional treatment.7 On average, COPD exacerbations occur two to three times per year, although mild exacerbations may go unreported by patients or unrecognized by providers.8
COPD exacerbations have been associated with an increased risk of future exacerbations, all-cause mortality, and acute cardiovascular (CV) events.7,9–21 However, research regarding these associations was conducted mainly in selected clinical trial populations18 and non-US populations,9,19 who may receive different care than COPD patients in the general US population. Prior studies generally followed patients for 12 months or less,19,20 and examined only severe exacerbations that required hospitalization.13,20 Study results have varied widely, and the importance of exacerbation history and the duration of COPD remains uncertain.
To address these knowledge gaps, we conducted a retrospective cohort study in newly diagnosed US COPD patients as part of a multi-country initiative called EXAcerbations of COPD and their OutcomeS in CardioVascular diseases (EXACOS-CV).22 This study estimated the incidence of all-cause mortality and acute CV events following an AECOPD and how this incidence varies over time. We also investigated the effects of number and severity of exacerbations on short-term risk of these outcomes, and effect modification by social determinants of health, and medication and clinical history.
Materials and Methods
Study Design and Population
This is a retrospective cohort study conducted using the Healthcare Integrated Research Database (HIRD) over years 2006 through 2019. The HIRD is a longitudinal medical and pharmacy claims database that includes diagnosis, medication, and procedure information, physician specialties, laboratory data, healthcare utilization, and amounts paid by the health plan and member from health plan members across the US maintained by Carelon Research, Inc. Currently, the HIRD includes 88 million unique individuals with medical coverage and 68 million unique individuals with medical and pharmacy claims information. Mortality data were obtained from the National Death Index (NDI). The NDI is a centralized database of death information maintained by the US National Center for Health Statistics.23,24 Patients in the HIRD were linked to the NDI using their name, gender, date of birth, state of residence, and social security number, when available.
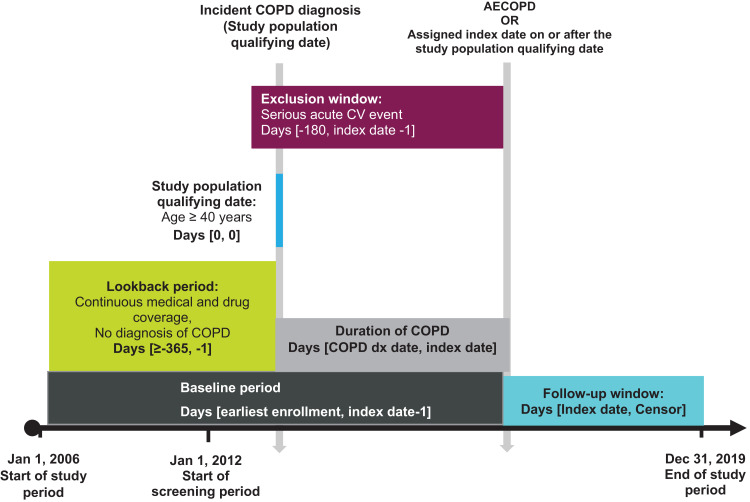
The study population included newly diagnosed COPD patients defined as having a primary diagnosis code for COPD (Supplemental Table 1) at an outpatient, inpatient, or emergency department (ED) visit between January 1, 2012, and December 30, 2019. The date of the first COPD diagnosis code between 2012 and 2019 was considered the incident diagnosis date and patients were excluded if they had any prior COPD diagnoses within 1 year or more. Patients were at least 40 years old on the date of their incident COPD diagnosis and had at least 12 months of continuous health plan enrollment prior to this date (Figure 1). The study period started January 1, 2006, and ended December 31, 2019, to avoid Coronavirus disease of 2019 (COVID-19) pandemic-related changes in health care utilization, patient behavior, and diagnosis misclassification between COPD exacerbations and COVID-19 infections. The baseline period was defined as all continuous enrollment time before the index date, which was the exacerbation date or, for comparators, a proxy date (defined further below). Patients were excluded from study cohorts if they had an acute CV event within 6 months of their index date as a washout period to ensure that any CV diagnoses seen during follow-up were incident CV events. In the primary analysis, patients were followed starting on the index date until an outcome event, death (if not the outcome), disenrollment from their health plan, a subsequent AECOPD, or end of the study period. If someone in the exacerbation group had a second exacerbation, they were censored from the primary analysis and were then eligible for inclusion in a separate analysis of patients who had two exacerbations compared with patients who had one exacerbation. The Western Institutional Review Board-Copernicus Group Institutional Review Board approved the HIPAA waiver of authorization (IRB tracking number 20221832) for the use and disclosure of protected health information to link patients to the NDI as necessary for conduct of this research.
Figure 1.
Study design.
Exposures and Index Date
The primary exposure was a first occurrence of a moderate or severe exacerbation on or after the incident COPD diagnosis date (Figure 1). A moderate exacerbation was defined as an outpatient visit with a diagnosis of COPD and, within 7 days of the visit, a medication dispensing of ≤30 days’ supply of oral glucocorticoids or antibiotics, or administration of intravenous (IV) glucocorticoids.25 A severe exacerbation was defined as a hospitalization or ED visit with either a primary diagnosis of COPD or a primary diagnosis of respiratory failure, acute bronchitis, asthma exacerbation, bronchiectasis exacerbation, or pneumonia with a secondary diagnosis of COPD.
The primary analysis compared patients who had a first exacerbation (ie, the 1-AECOPD cohort) to patients who did not have an exacerbation (the 0-AECOPD comparator cohort). The 1-AECOPD cohort included patients with at least one exacerbation on or after their incident COPD diagnosis date. The index date for the 1-AECOPD cohort was the date of the patient’s first AECOPD, regardless of severity (code lists provided in Supplemental Table 1).
To create the 0-AECOPD comparator cohort (Figure 1), index dates were randomly assigned to all patients in the study population using the distribution of time from the incident COPD diagnosis to the first exacerbation (ie, the duration of COPD) among exacerbating patients. Frequency matching by time since first COPD diagnosis was used to enhance similarity between the two cohorts regarding duration of COPD. Patients in the 0-AECOPD cohort were censored if they eventually had an exacerbation, at which time they entered the 1-AECOPD cohort; thus, patients could contribute time to both cohorts.
In secondary analyses, four additional cohorts were created to analyze the incremental impact of subsequent exacerbations on the rates of all-cause mortality and CV events (Figure 1). To analyze the impact of a second exacerbation compared to a first, a 2-AECOPD cohort was defined as a group of patients with at least two exacerbations following incident COPD diagnosis. The index date for these patients was the date of the second exacerbation. The comparator for the 2-AECOPD cohort was the 1-AECOPD comparator group, comprising patients with at least one exacerbation and with index dates assigned based on the distribution of duration of COPD in the 2-AECOPD cohort. The 3-AECOPD cohort (ie patients experiencing ≥3 AECOPD events) and 2-AECOPD comparator cohort were similarly created. Few patients had more than three exacerbations so effects of four or more exacerbations were not studied. As with the primary analysis, patients in the comparator cohorts were censored upon occurrence of a subsequent exacerbation during follow-up, after which time they moved from the comparator to the exacerbation cohorts.
In the primary analysis, patients with an exacerbation prior to their assigned 0-AECOPD comparator index date were excluded from the comparator cohort. Similarly, patients were excluded from the 1-AECOPD and 2-AECOPD comparator cohorts if they had a second or third exacerbation before their assigned index date.
Outcomes
Endpoints of interest were (1) all-cause mortality; (2) a first acute CV event of any type (composite CV event outcome); and (3) each individual CV event type. All-cause mortality was defined as death due to any cause and was identified using claims data and the NDI.23,24 An acute CV event was defined as a primary inpatient diagnosis for myocardial infarction (MI), unstable angina, ischemic stroke, acute heart failure, pulmonary embolism and deep vein thrombosis, cardiac arrhythmias, cardiac arrest, or CV death due to one of these causes (as identified by underlying cause of death recorded in the NDI). The date of the acute CV event was the date the patient was diagnosed with an acute CV event or the date of their CV death (Supplemental Table 2).
When analyzing a specific type of CV event individually, other types of CV outcomes were not considered as censoring events. This minimized the risk of underestimating the incidence of CV events that were less likely to occur first during follow-up. Similarly, all-cause mortality was considered without CV events as censoring criteria.
Statistical Analysis
Baseline characteristics were summarized by primary exposure status (ie, 1-AECOPD cohort and 0-AECOPD comparator group). Standardized mean differences (SMD) were calculated to assess the comparability of the 1-AECOPD and 0-AECOPD comparator cohorts.26 SMD <0.1 were considered balanced between exposed and comparator cohorts.27 Distributions of follow-up time, censoring criteria, number of COPD diagnoses over follow-up, and proportions of those who experienced each acute CV event type were summarized by cohort.
Cox proportional hazards regression models were used to estimate average and time-stratified hazard ratios (HRs) of mortality and acute CV events comparing patients experiencing a first exacerbation (1-AECOPD) to patients with no prior exacerbation (0-AECOPD comparators). For all models, covariates included age, sex, neighborhood (ie, census block-group) urbanicity, neighborhood socioeconomic status (SES), history of CV and COPD medication dispensings, duration of COPD, and COPD and CV comorbidities. Covariates were selected based on literature showing their association with the outcomes under study and were identified using National Drug Codes, ICD-9-CM and ICD-10-CM diagnosis codes, or from the American Community Survey (Supplemental Table 3).28 HRs of all-cause death and of acute CV event (of any type) following a first exacerbation, compared to no prior exacerbation, were estimated overall and separately by duration of follow-up. HRs of each type of acute CV event in the 30-day period following a first exacerbation compared to no prior exacerbation were also estimated.
The HRs of all-cause death and of acute CV events (of any type) following a first exacerbation of any severity (compared to no prior exacerbation) were analyzed by neighborhood urbanicity, SES, third-party payor type, number of lower respiratory infections one-year prior to index, number of short-acting beta 2-agonists (SABA) dispensings one-year prior to index, and by history of acute CV events.
HRs of all-cause death and of acute CV events (of any type) in the 30-days following a first moderate or a first severe AECOPD, compared to no prior AECOPD, were estimated. We also estimated the 30-day HRs of all-cause mortality and of acute CV events following a second AECOPD (vs one AECOPD) and following a third AECOPD (vs two AECOPD). Models for this set of analyses were adjusted for time since the previous exacerbation (eg, time since the first AECOPD in comparisons of the 2-AECOPD cohort to 1-AECOPD comparator cohort) and severity of the exacerbation, in addition to the covariates listed above.
Sensitivity Analyses
COPD often develops gradually with no sharp onset date and is sometimes first diagnosed after the condition has progressed to the point of an exacerbation.3,29 Further, a CV event can occur during an exacerbation. During our initial analyses, we identified 386 comparator patients and 49 exacerbation patients with an acute CV event on the same date as their incident COPD diagnosis. It is not clear whether the CV events would have been triggered by exacerbation (and both diagnosis codes provided on the same day) or if the first diagnosis of COPD occurred because the patient was hospitalized for a CV event. The implication of the latter is that some patients could enter the comparator group at the time of an acute CV event, which creates an opportunity for selection bias. To assess this possible bias, we analyzed the data excluding members with CV events occurring on the same day as the first COPD diagnosis and presented both sets of estimates.
Results
Descriptive Results
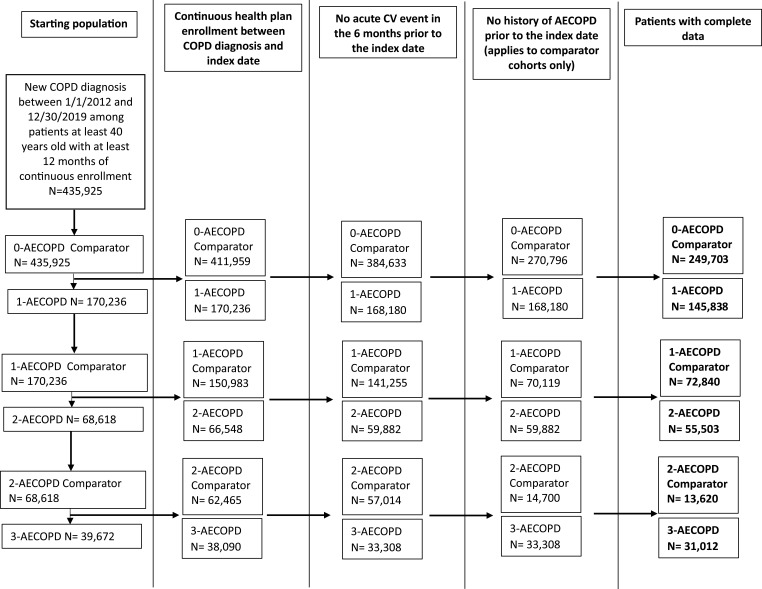
There were 435,925 patients newly diagnosed with COPD between January 1, 2012, and December 31, 2019 (Figure 2 and Supplemental Table 4). After applying all other exclusion criteria there were 145,838 1-AECOPD patients and 249,703 0-AECOPD comparator patients. About 11% of patients in the 0-AECOPD comparator cohort also contributed observation time to the 1-AECOPD cohort after their first exacerbation. Unless otherwise stated, analyses compared the 1-AECOPD cohort with the 0-AECOPD comparison cohort.
Figure 2.
Study population.
The average age of the 1-AECOPD and 0-AECOPD cohorts were 63 and 64 years, respectively (Table 1). Both cohorts were majority female (1-AECOPD: 55.4%; 0-AECOPD: 51.2%) and over half of the study population lived in urban areas (1-AECOPD: 51.4%; 0-AECOPD: 53.4%). Patients in both cohorts were relatively balanced across the neighborhood SES quartiles. During baseline, one-third of patients in each cohort were hospitalized at least once, and nearly all had at least one outpatient visit. Nearly 30% of patients had a pulmonologist visit and one-half had a cardiologist visit. There were 64% of patients in the 1-AECOPD cohort with their first moderate or severe AECOPD on the same day as their first COPD diagnosis.
Table 1.
Baseline Patient Demographic Characteristics and Healthcare Resource Utilization by Cohort
| Patient Demographics | 1-AECOPD | 0-AECOPD Comparator | Standardized Mean Difference |
|---|---|---|---|
| N=145,838 | N=249,703 | ||
| Age on index, mean (SD) | 63 (12) | 64 (12) | 0.11 |
| Female, N (%) | 80,813 (55.4) | 127,861 (51.2) | 0.08 |
| Region, N (%) | |||
| Northeast | 22,191 (15.2) | 42,877 (17.2) | 0.05 |
| Midwest | 43,003 (29.5) | 71,167 (28.5) | 0.02 |
| South | 50,241 (34.4) | 79,212 (31.7) | 0.06 |
| West | 30,403 (20.8) | 56,447 (22.6) | 0.04 |
| Neighborhood urbanicity, N (%) | |||
| Urban | 74,993 (51.4) | 133,426 (53.4) | 0.04 |
| Semi-urban | 37,531 (25.7) | 64,184 (25.7) | 0.00 |
| Rural | 33,314 (22.8) | 52,093 (20.9) | 0.05 |
| Neighborhood socioeconomic status quartile, N (%) | |||
| 1 (0–25%) | 27,505 (18.9) | 42,821 (17.1) | 0.04 |
| 2 (26–50%) | 40,257 (27.6) | 65,555 (26.3) | 0.03 |
| 3 (51–75%) | 41,036 (28.1) | 71,448 (28.6) | 0.01 |
| 4 (76–100%) | 37,040 (25.4) | 69,879 (28.0) | 0.06 |
| Insurance coverage, N (%) | |||
| Commercial | 109,677 (75.2) | 186,017 (74.5) | 0.02 |
| Medicare advantage | 22,695 (15.6) | 38,588 (15.5) | 0.00 |
| Other | 13,466 (9.2) | 25,098 (10.1) | 0.03 |
| Duration of baseline period (months), mean (SD) | 56 (37) | 57 (37) | 0.00 |
| Duration of COPDa, N (%) | |||
| 0 days | 93,643 (64.2) | 84,536 (33.9) | 0.64 |
| 1 day - <3 months | 12,990 (8.9) | 105,985 (42.4) | 0.83 |
| 3 months - <6 months | 6,754 (4.6) | 22,565 (9.0) | 0.18 |
| 6 months - <2 years | 21,769 (14.9) | 32,522 (13.0) | 0.05 |
| 2 years or longer | 10,682 (7.3) | 4095 (1.6) | 0.28 |
| Baseline health care utilization, N (%) | |||
| ≥ 1 hospitalization | 49,576 (34.0) | 88,908 (35.6) | 0.03 |
| ≥ 1 outpatient visit | 143,979 (98.7) | 248,776 (99.6) | 0.10 |
| ≥ 1 pulmonologist visit | 39,314 (27.0) | 73,229 (29.3) | 0.05 |
| ≥ 1 cardiologist visit | 70,562 (48.4) | 132,203 (52.9) | 0.09 |
Notes: aDuration of COPD is defined as the time from the incident COPD diagnosis until the first exacerbation in the 1-AECOPD cohort or the assigned index date in the 0-AECOPD comparator cohort. Patients who contributed to both the 0-AECOPD and 1-AECOPD groups are represented in counts for each group.
Abbreviations: AECOPD, acute exacerbation of chronic obstructive pulmonary disease; SD, standard deviation.
Most baseline comorbidities were diagnosed in similar proportions of patients in the 1-AECOPD and 0-AECOPD comparator cohorts (Table 2). Hyperlipidemia and hypertension were each diagnosed in roughly 70% of patients in both cohorts. One-quarter of patients in both cohorts had Type 2 diabetes (SMD=0.01). Additionally, 18% and 15% (SMD=0.07) of exacerbation and comparator patients, respectively, were diagnosed with bronchiectasis, and 27% and 22% (SMD=0.11) were diagnosed with asthma. A notably higher proportion of patients in the 1-AECOPD cohort had scores of zero on the Quan-Charlson Comorbidity Index (QCCI) (25% of 1-AECOPD patients vs 11% of 0-AECOPD comparator patients; SMD=0.39). More patients in the 1-AECOPD cohort had a CCI score of 0, while proportions of patients with QCCI scores of 2 or higher were similar between the cohorts.
Table 2.
Baselinea Patient Comorbidities and Medication Use by Cohort
| Comorbidities and Medications | 1-AECOPD | 0-AECOPD Comparator | Standardized Mean Differences |
|---|---|---|---|
| N=145,838 | N=249,703 | ||
| Comorbidities | |||
| Acute CV eventsb | 16,541 (11.3) | 29,106 (11.7) | 0.01 |
| Cardiovascular disease | 101,953 (69.9) | 183,636 (73.5) | 0.08 |
| Hyperlipidemia | 97,733 (67.0) | 176,960 (70.9) | 0.08 |
| Hypertension | 99,481 (68.2) | 178,347 (71.4) | 0.07 |
| Type 2 diabetes | 34,066 (23.4) | 59,811 (24.0) | 0.01 |
| Serious lower respiratory infections | 12,338 (8.5) | 18,447 (7.4) | 0.04 |
| Asthma | 38,720 (26.6) | 54,511 (21.8) | 0.11 |
| Bronchiectasis | 25,710 (17.6) | 37,239 (14.9) | 0.07 |
| Interstitial lung disease | 32,359 (22.2) | 59,082 (23.7) | 0.04 |
| Pleural disease | 37,169 (25.5) | 64,708 (25.9) | 0.01 |
| Depression and/or anxiety | 58,181 (39.9) | 95,057 (38.1) | 0.04 |
| Any cancerc | 27,866 (19.1) | 54,139 (21.7) | 0.06 |
| Quan-Charlson Comorbidity Index count, N (%) | |||
| 0 | 36,938 (25.3) | 26,418 (10.6) | 0.39 |
| 1 | 46,743 (32.1) | 103,730 (41.5) | 0.2 |
| 2 | 16,137 (11.1) | 25,819 (10.3) | 0.02 |
| 3+ | 46,020 (31.6) | 93,736 (37.5) | 0.13 |
| Prescription medications, N (%) | |||
| Antibiotics | 126,274 (86.6) | 193,197 (77.4) | 0.24 |
| Glucocorticoidsd | 103,168 (70.7) | 156,212 (62.6) | 0.17 |
| Immunosuppressants | 88,477 (60.7) | 124,137 (49.7) | 0.22 |
| Long-acting bronchodilators (LABA or LAMA) | 36,375 (24.9) | 46,329 (18.6) | 0.16 |
| Short-acting bronchodilators (SABA or SAMA) | 76,324 (52.3) | 98,812 (39.6) | 0.26 |
| Inhaled corticosteroids | 36,516 (25.0) | 47,564 (19.0) | 0.14 |
| Oral corticosteroids | 82,579 (56.6) | 116,287 (46.6) | 0.2 |
Note: aBaseline period begins on start of continuous enrollment and ends the day before the index date unless otherwise specified. bAcute CV events occurred > 6 months prior to index date. cAny cancer includes all cancers except for basal cell carcinoma. dIncludes all formulations of glucocorticoids (oral, intravenous, etc). Patients who contributed to both the 0-AECOPD and 1-AECOPD groups are represented in counts for each group.
Abbreviations: AECOPD, acute exacerbation of chronic obstructive pulmonary disease; CV, cardiovascular; NA, Not applicable; LAMA, long-acting muscarinic antagonists; LABA, long-acting beta2-agonists; SABA, short-acting beta 2-agonists; SAMA, short-acting muscarinic antagonists.
With respect to medications commonly used to treat COPD, baseline dispensings of glucocorticoids were identified for 71% of 1-AECOPD patients and 63% of 0-AECOPD patients (SMD=0.17) (Table 2), with a mean (SD) number of dispensings in the one year prior to index of 4.5 (8.5) and 3.4 (6.9), respectively (Supplemental Table 5). Baseline dispensings of short-acting bronchodilators were found for 52% of 1-AECOPD patients and 40% of 0-AECOPD patients (SMD=0.26), with mean (SD) dispensings during the one-year prior to index of 2.8 (7.1) and 1.7 (5.4), respectively. Other descriptive statistics for comorbidities, CV medications, and numbers of dispensings for COPD medications are found in Supplemental Table 5. Follow-up and censoring statistics are found in Supplemental Table 6.
All-Cause Mortality
The overall adjusted HR for all-cause mortality showed an increased risk following a first AECOPD (HR=1.25; 95% confidence interval (CI) = 1.20—1.30) over a mean follow-up of 17 months for the 1-AECOPD cohort and 21 months for the 0-AECOPD comparator cohort (Table 3 and Supplemental Table 6). The hazard of all-cause mortality in the 1-AECOPD cohort was greatest in the first 30-days after the AECOPD (HR=1.79; 95% CI=1.58—2.04) and remained elevated for up to 2 years. The risk of all-cause mortality following a first AECOPD was lowest among patients living in urban areas and high-SES neighborhoods (Table 3). Patients with one or more SABA dispensings or one or more lower respiratory infections in the year prior to index had higher risks associated with a first AECOPD, compared with patients who did not have SABA dispensings or lower respiratory infections. Mortality risks associated with a first AECOPD were similar by insurance payor and by history of CV events.
Table 3.
Hazard Ratios of All-Cause Mortality by AECOPD Status, Overall, and by Patient Characteristic Subgroups
| 1-AECOPD | 0-AECOPD Comparator | Adjusteda HR (95% CI) | |||
|---|---|---|---|---|---|
| Cases | Person-Years | Cases | Person-Years | ||
| Full study population | |||||
| All-cause mortalityb | 3835 | 209,326.06 | 8446 | 462,440.03 | 1.25 (1.20, 1.30) |
| Subgroup | |||||
| Duration follow-upc | |||||
| <30 days | 456 | 10,510.61 | 597 | 19,931.20 | 1.79 (1.58, 2.04) |
| 30 days to 90 days | 508 | 18,411.96 | 821 | 36,907.80 | 1.55 (1.38, 1.73) |
| 91 days to <6 months | 481 | 24,038.32 | 880 | 49,423.27 | 1.39 (1.24, 1.56) |
| 6 months to <1 year | 665 | 39,555.51 | 1316 | 83,583.27 | 1.33 (1.21, 1.47) |
| 1 year to <2 years | 715 | 51,172.43 | 1755 | 115,198.95 | 1.15 (1.05, 1.25) |
| 2 years to <3 years | 389 | 30,184.78 | 1157 | 71,523.61 | 1.03 (0.92, 1.16) |
| 3 or more years | 621 | 35,452.44 | 1920 | 85,871.93 | 1.04 (0.95, 1.14) |
| Insurance Coverage | |||||
| Commercial | 882 | 158,432.42 | 1842 | 335,177.68 | 1.31 (1.20, 1.42) |
| Medicare advantage | 1601 | 28,811.21 | 3386 | 69,207.75 | 1.28 (1.21, 1.37) |
| Other | 1352 | 22,082.43 | 3218 | 58,054.60 | 1.15 (1.08, 1.23) |
| Urbanicity of home neighborhood | |||||
| Urban | 2072 | 110,420.53 | 4796 | 250,372.48 | 1.20 (1.14, 1.26) |
| Semi-urban | 999 | 53,638.29 | 2207 | 118,672.84 | 1.29 (1.19, 1.39) |
| Rural | 764 | 45,267.24 | 1443 | 93,394.72 | 1.33 (1.22, 1.46) |
| Neighborhood socioeconomic status quartile | |||||
| 1 (0–25%) | 614 | 36,426.78 | 1263 | 73,990.72 | 1.26 (1.14, 1.39) |
| 2 (26–50%) | 1004 | 55,442.22 | 1988 | 117,592.75 | 1.39 (1.29, 1.51) |
| 3 (51–75%) | 1058 | 59,043.29 | 2443 | 132,946.24 | 1.22 (1.13, 1.31) |
| 4 (76–100%) | 1159 | 58,413.77 | 2752 | 137,910.33 | 1.16 (1.08, 1.25) |
| SABA dispensings in 1-year prior to index | |||||
| None | 2257 | 113,497.52 | 6163 | 296,876.75 | 1.23 (1.17, 1.29) |
| 1 or more | 1578 | 95,828.53 | 2283 | 165,563.28 | 1.30 (1.22, 1.39) |
| History of respiratory infections in the 1-year prior to index | |||||
| None | 2878 | 196,324.28 | 6699 | 432,742.01 | 1.23 (1.17, 1.28) |
| 1 or more | 957 | 13,001.77 | 1747 | 29,698.02 | 1.36 (1.25, 1.48) |
| History of acute CV eventsd | |||||
| Yes | 1247 | 20,186.22 | 2558 | 51,775.05 | 1.27 (1.19, 1.37) |
| No | 2588 | 189,139.83 | 5888 | 410,664.98 | 1.26 (1.20, 1.32) |
Notes: aCox proportional hazards models were adjusted for the following baseline demographics, comorbidities, and medications: age, sex, urbanicity, region, insurance coverage, Index calendar year, SES, COPD duration, serious lower respiratory infections, congestive heart failure, hyperlipidemia, hypertension, Charlson Comorbidity Index, chronic kidney disease, pneumothorax, pulmonary edema, sleep apnea, cancer, alpha-1 antitrypsin deficiency, bronchiectasis, extrinsic allergic alveolitis, interstitial lung disease, pleural disease, sarcoidosis, acute CV events, CV procedures, antihistamines, lipid-lowering agents, antihypertensive agents, antiarrhythmics, antianginal agents, other CV agents (including letairis, ambrisentan, caduet, tracleer, etc.), anticoagulants, immunosuppressants, antidiabetics, antifungals, antivirals, ICS, oral corticosteroids, LABA, SABA, glucocorticoids, antibiotics, hospitalizations, outpatient visits, pulmonologist visits, and cardiologist visits. bAll-cause mortality data was derived from the National Death Index data. cFollow-up begins on the index date and continues until the patient is censored for the outcome, a subsequent AECOPD, death (if not the outcome), disenrollment from health plan, or end of the study period. dHistory of acute CV events is measured from the start of the study period until six months prior to the index date.
Abbreviations: AECOPD, acute exacerbation of chronic obstructive pulmonary disease; CI, confidence interval; CV, cardiovascular; HR, hazard ratio; ICS, inhaled corticosteroids; SABA, short-acting beta 2-agonists.
In the first 30-days after the index date, patients whose first AECOPD was considered moderate had a 1.22 (95% CI=1.04—1.43) times higher hazard of all-cause mortality than 0-AECOPD comparator patients, and patients whose first AECOPD was severe had a 5.09 (95% CI=4.30—6.03) times higher hazard of all-cause mortality compared to the 0-AECOPD comparator patients (Table 4). The hazard of all-cause mortality in the 30-days after a second AECOPD was 3.35 (95% CI=2.52—4.46) times higher than among comparators with one AECOPD. Similarly, this hazard following a third AECOPD was 3.40 (95% CI=2.20—5.24) times higher than among comparators with two prior AECOPDs.
Table 4.
30-Day Hazard Ratios of Acute CV Events and All-Cause Mortality According to AECOPD Severity and Number of AECOPD Using Data from Claims and the National Death Index
| AECOPD Characteristic | Cases | Person-Yearsa | Adjustedb HR (95% CI) |
|---|---|---|---|
| All-Cause Mortality | |||
| Severity of first AECOPD | |||
| No AECOPD | 568 | 19,851.91 | Ref |
| Moderate AECOPD | 237 | 9186.69 | 1.22 (1.04, 1.43) |
| Severe AECOPD | 196 | 1281.12 | 5.09 (4.30, 6.03) |
| Number of AECOPD | |||
| 0-AECOPD Comparator | 568 | 19,851.91 | Ref |
| 1-AECOPD | 433 | 10,467.81 | 1.79 (1.58, 2.04) |
| 1-AECOPD Comparator | 65 | 5724.60 | Ref |
| 2-AECOPD | 215 | 3732.56 | 3.35 (2.52, 4.46) |
| 2-AECOPD Comparator | 23 | 1062.84 | Ref |
| 3-AECOPD | 230 | 2483.90 | 3.40 (2.20, 5.24) |
| Acute CV events including events on the COPD diagnosis date | |||
| Severity of first AECOPD | |||
| No AECOPD | 1475 | 19,851.91 | Ref |
| Moderate | 717 | 9,186.69 | 1.23 (1.12, 1.35) |
| Severe | 232 | 1,281.12 | 1.93 (1.67, 2.22) |
| Number of AECOPD | |||
| 0-AECOPD Comparator | 1,475 | 19,851.91 | Ref |
| 1-AECOPD | 949 | 10,467.81 | 1.34 (1.23, 1.46) |
| 1-AECOPD Comparator | 173 | 5724.60 | Ref |
| 2-AECOPD | 370 | 3732.56 | 2.66 (2.21, 3.21) |
| 2-AECOPD Comparator | 46 | 1062.84 | Ref |
| 3 AECOPD | 252 | 2483.90 | 2.19 (1.59, 3.02) |
| Acute CV events excluding events on the COPD diagnosis date | |||
| Severity of first AECOPD | |||
| No AECOPD | 1089 | 19,851.91 | Ref |
| Moderate AECOPD | 694 | 9186.69 | 1.63 (1.48, 1.80) |
| Severe AECOPD | 206 | 1281.12 | 2.49 (2.14, 2.90) |
| Number of AECOPD | |||
| 0-AECOPD Comparator | 1089 | 19,851.91 | Ref |
| 1-AECOPD | 900 | 10,467.81 | 1.74 (1.59, 1.91) |
| 1-AECOPD Comparator | 171 | 5724.60 | Ref |
| 2-AECOPD | 370 | 3732.56 | 2.69 (2.23, 3.25) |
| 2-AECOPD Comparator | 46 | 1062.84 | Ref |
| 3 AECOPD | 252 | 2483.90 | 2.19 (1.59, 3.02) |
Notes: aPerson-years were measured from Index date to censor date (earliest of dates of: first AECOPD event (for comparator cohort), subsequent AECOPD (for AECOPD cohorts), end of continuous enrollment, CV event, Death, Index date + 30 days, or Study end). bCox proportional hazards models were adjusted for the following baseline demographics, comorbidities, and medications: age, sex, urbanicity, region, insurance coverage, Index calendar year, SES, COPD duration, serious lower respiratory infections, congestive heart failure, hyperlipidemia, hypertension, Charlson Comorbidity Index, chronic kidney disease, pneumothorax, pulmonary edema, sleep apnea, cancer, alpha-1 antitrypsin deficiency, bronchiectasis, extrinsic allergic alveolitis, interstitial lung disease, pleural disease, sarcoidosis, acute CV events, CV procedures, antihistamines, lipid-lowering agents, antihypertensive agents, antiarrhythmics, antianginal agents, other CV agents (including letairis, ambrisentan, caduet, tracleer, etc.), anticoagulants, immunosuppressants, antidiabetics, antifungals, antivirals, ICS, oral corticosteroids, LABA, SABA, glucocorticoids, antibiotics, hospitalizations, outpatient visits, pulmonologist visits, and cardiologist visit.
Abbreviations: AECOPD, acute exacerbation of chronic obstructive pulmonary disease; CV, cardiovascular; CI, Confidence Interval; HR, Hazard ratio; Ref, Reference Category.
Acute CV Events
In the primary analyses (including acute CV events that occurred on the incident COPD diagnosis date) the overall HR of acute CV events in the 1-AECOPD cohort was 1.11 (95% CI=1.07—1.14) compared to the 0-AECOPD comparator cohort, adjusted for demographics, comorbidities, and prescription history (Table 5). The HR of acute CV events was highest within the first 30-days after the first AECOPD (HR=1.34; 95% CI=1.23—1.46) and remained elevated for 1 year. Subgroup analysis results were similar to the all-cause mortality results, though effect sizes and differences between subgroups were smaller (Supplemental Table 7). Patients whose first AECOPD was moderate had a 30-day HR=1.23 (95% CI=1.12—1.35), and patients whose first AECOPD was severe had a HR=1.93 (95% CI=1.67—2.22) compared to patients without a prior AECOPD (Table 4). The HR of CV events in the 30-days after a second AECOPD compared to 1-AECOPD comparator patients was 2.66 (95% CI=2.21—3.21). Similarly, the HR following a third AECOPD compared to 2-AECOPD comparator patients was 2.19 (95% CI=1.59—3.02). The highest 30-day HR of individual CV events comparing 1-AECOPD patients to 0-AECOPD comparator patients was for acute heart failure (HR=1.94 95% CI=1.69—2.24), followed by CV-related death (HR=1.63 95% CI=1.14—2.35) (Table 5). Hazards of experiencing an MI or ischemic stroke during the first 30 days post-index were roughly equivalent for the 1-AECOPD and 0-AECOPD comparator cohorts.
Table 5.
Hazard Ratios of Acute CV Events by Cohort Overall, by Follow-Up Time, and Individual CV Event, Including and Excluding CV Events Occurring on the COPD Diagnosis Date
| Including Acute CV Events Occurring on the COPD Diagnosis Date | Excluding Acute CV Events Occurring on the COPD Diagnosis Date | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 AECOPD | 0- AECOPD Comparator | Adjusteda HR (95% CI) | 1-AECOPD | 0- AECOPD Comparator | Adjusteda HR (95% CI) | |||||
| Cases | Person-Years | Cases | Person-Years | Cases | Person-Years | Cases | Person-Years | |||
| Full study population | ||||||||||
| All Serious acute CV events | 5479 | 203,233.64 | 12,530 | 446,720.72 | 1.11 (1.07, 1.14) | 5,430 | 203,233.64 | 12,144 | 446,720.72 | 1.14 (1.10, 1.18) |
| Follow-up Time | ||||||||||
| <30 days | 949 | 10,467.81 | 1475 | 19,851.91 | 1.34 (1.23, 1.46) | 900 | 10,467.81 | 1089 | 19,851.91 | 1.74 (1.59, 1.91) |
| 30 days to 90 days | 655 | 18,255.67 | 1204 | 36,630.84 | 1.29 (1.17, 1.42) | 655 | 18,255.67 | 1204 | 36,630.84 | 1.29 (1.17, 1.42) |
| 91 days to <6 months | 632 | 23,731.84 | 1380 | 48,815.38 | 1.08 (0.98, 1.19) | 632 | 23,731.84 | 1380 | 48,815.38 | 1.08 (0.98, 1.19) |
| 6 months to <1 year | 973 | 38,807.84 | 2107 | 81,972.55 | 1.14 (1.05, 1.23) | 973 | 38,807.84 | 2107 | 81,972.55 | 1.14 (1.05, 1.23) |
| 1 year to <2 years | 1,022 | 49,679.00 | 2749 | 111,548.00 | 1.00 (0.92, 1.07) | 1,022 | 49,679.00 | 2749 | 111,548.00 | 1.00 (0.92, 1.07) |
| 2 years to <3 years | 575 | 28,961.29 | 1599 | 68,200.85 | 1.04 (0.94, 1.14) | 575 | 28,961.29 | 1599 | 68,200.85 | 1.04 (0.94, 1.14) |
| 3 or more years | 673 | 33,330.19 | 2016 | 79,701.19 | 1.00 (0.92, 1.10) | 673 | 33,330.19 | 2016 | 79,701.19 | 1.00 (0.92, 1.10) |
| Short-term (30-day) incidenceb | ||||||||||
| MI | 172 | 10,504.33 | 300 | 19,913.53 | 1.05 (0.86, 1.27) | 154 | 10,504.33 | 199 | 19,913.53 | 1.43 (1.15, 1.78) |
| Ischemic stroke | 62 | 10,509.93 | 140 | 19,923.47 | 0.95 (0.70, 1.29) | 61 | 10,509.93 | 96 | 19,923.47 | 1.41 (1.01, 1.97) |
| Unstable angina | 75 | 10,510.17 | 228 | 19,920.93 | 0.75 (0.57, 0.98) | 73 | 10,510.17 | 203 | 19,920.93 | 0.84 (0.64, 1.11) |
| Acute heart failure | 387 | 10,496.11 | 434 | 19,909.44 | 1.94 (1.69, 2.24) | 372 | 10,496.11 | 332 | 19,909.44 | 2.46 (2.11, 2.87) |
| Pulmonary embolism | 119 | 10,507.03 | 162 | 19,922.53 | 1.41 (1.11, 1.81) | 114 | 10,507.03 | 123 | 19,922.53 | 1.81 (1.39, 2.36) |
| Cardiac arrhythmias | 149 | 10,506.35 | 258 | 19,917.09 | 1.22 (0.99, 1.50) | 140 | 10,506.35 | 182 | 19,917.09 | 1.64 (1.31, 2.07) |
| Atrial fibrillation and flutter | 128 | 10,507.15 | 183 | 19,920.96 | 1.49 (1.18, 1.88) | 120 | 10,507.15 | 129 | 19,920.96 | 2.01 (1.55, 2.60) |
| Other arrhythmias | 25 | 10,512.00 | 83 | 19,927.27 | 0.63 (0.40, 1.00) | 24 | 10,512.00 | 61 | 19,927.27 | 0.82 (0.50, 1.35) |
| CV death | 52 | 10,512.83 | 76 | 19,931.20 | 1.63 (1.14, 2.35) | 52 | 10,512.83 | 76 | 19,931.20 | 1.63 (1.14, 2.35) |
| Cardiac arrest | NR | NR | NR | NR | 1.47 (0.48, 4.52) | NR | NR | NR | NR | 1.71 (0.54, 5.46) |
Notes: aCox proportional hazards models were adjusted for the following baseline demographics, comorbidities, and medications: age, sex, urbanicity, region, insurance coverage, Index calendar year, SES, COPD duration, serious lower respiratory infections, congestive heart failure, hyperlipidemia, hypertension, Charlson Comorbidity Index, chronic kidney disease, pneumothorax, pulmonary edema, sleep apnea, cancer, alpha-1 antitrypsin deficiency, bronchiectasis, extrinsic allergic alveolitis, interstitial lung disease, pleural disease, sarcoidosis, acute CV events, CV procedures, antihistamines, lipid-lowering agents, antihypertensive agents, antiarrhythmics, antianginal agents, other CV agents (including letairis, ambrisentan, caduet, tracleer, etc.), anticoagulants, immunosuppressants, antidiabetics, antifungals, antivirals, ICS, oral corticosteroids, LABA, SABA, glucocorticoids, antibiotics, hospitalizations, outpatient visits, pulmonologist visits, and cardiologist visits. bCV deaths from each individual CV cause are included in each category (eg, MI, ischemic stroke, pulmonary embolism, etc.) as well as separately under “CV death”. cAccording to Elevance Health policy, any cell with a value of 1 to 4 or any cell that allows a value of 1 to 4 to be derived from other reported cells or information cannot be reported.
Abbreviations: AECOPD, Acute exacerbation of chronic obstructive pulmonary disease; CI, confidence interval; CV, cardiovascular; HR, Hazard Ratio; MI, myocardial infarction, NR, not reportablec.
In the post-hoc analysis (excluding acute CV events that occurred on the incident COPD diagnosis date), all acute CV event HRs comparing the 1-AECOPD vs 0-AECOPD comparator cohorts were higher than in the original analysis. The 30-day HR of acute CV events increased from 1.34 to 1.74 (95% CI=1.59—1.91) (Table 5). Furthermore, the HRs for the 30-day risks of MI and ischemic stroke were no longer null (MI HR=1.43 95% CI=1.15—1.78 and ischemic stroke HR=1.41 95% CI=1.01, 1.97). Similar relative findings were observed in analyses stratified by AECOPD severity and number (Table 4) and in the subgroup analyses (Supplemental Table 7).
Discussion
Key Findings
In this longitudinal cohort study of newly diagnosed COPD patients, we found that a first moderate or severe exacerbation was associated with higher risks of all-cause mortality and acute CV events. Risks increased immediately after the exacerbation and remained elevated for 1 year for acute CV events and for 2 years for all-cause mortality. These findings show that newly diagnosed COPD patients are at risk for serious cardiopulmonary events and mortality following their first AECOPD. The hazard of all-cause mortality was more than 3-times greater following a second exacerbation compared with a first exacerbation and following a third exacerbation compared with a second exacerbation, indicating an incremental increase in mortality risk following each exacerbation. The same pattern was observed for acute CV events. When explored by type of CV event, the HR of acute CV events in the first 30 days was highest for acute heart failure, atrial fibrillation and flutter, pulmonary embolism or DVT, and CV death following a first AECOPD regardless of whether the CV events occurring on the incident COPD diagnosis date were counted. The highest increased risk in the first 30-days post-index was observed for acute heart failure. MI and ischemic stroke risks were elevated only in the sensitivity analyses.
Stratified analyses revealed that the associations between a first AECOPD and all-cause mortality and acute CV events were higher for patients living in rural areas, in lower SES neighborhoods, and in patients with more severe COPD (ie, with at least 1 SABA dispensing or with a lower respiratory infection in the year prior to index).
These results in a newly diagnosed COPD population and several subpopulations expand upon previous work regarding possible associations between COPD exacerbations and all-cause mortality and acute CV events. Three studies have previously shown that both moderate and severe AECOPDs are associated with higher risk of all-cause mortality or CV events, however these studies were conducted using prevalent COPD patients with histories of exacerbations or at higher risk of CVD.9,14,18 This study supports another recent study21 showing that these associations are present in newly diagnosed COPD patients following their first moderate or severe AECOPD. Other studies observing the duration of increased risk following an AECOPD limited their follow-up time to 1-year after the AECOPD,14,18,20,21 whereas this study estimated risks over 3 years. A review and meta-analysis of seven studies that analysed the incidence of MI or stroke following an AECOPD in prevalent COPD populations found elevated risks of these events in the 3 months after an AECOPD (MI: RR=2.48, 95% CI 1.40—4.20; Stroke: RR=1.68, 95% CI=1.19—2.38) and, similar to our study, these risks generally persisted for 1 year.11 Another study reported an increased risk of mortality after a moderate exacerbation (RR=1.08, 95% CI 1.04—1.12) or severe exacerbation (RR=1.75, 95% CI 1.66—1.85) with risk increasing with number of exacerbations.1 Finally, in newly diagnosed COPD patients, the risk of a serious CV event or all-cause mortality was greatest during the week following an AECOPD (HR=15.3, 95% CI 11.8—20.0) which declined from 6 months to 1 year following the AECOPD (HR=1.3, 95% CI 1.0—1.8).21 Most of these studies were conducted in populations outside the US where the standards of care may differ from those in the US. This study expands upon this work by including many other types of acute CV events such as pulmonary embolism or DVT, cardiac arrhythmias, acute heart failure, and unstable angina, which have previously been studied only in descriptive or exploratory studies,15,30,31 and by showing the increased mortality risk persisted until 2 years after an AECOPD.
The sensitivity analysis excluding comparator patients and exacerbation patients who had a CV event on the same day as their COPD diagnosis yielded higher hazard ratios than the primary analysis for both overall CV and sub-group analyses. It is possible that some patients could have had a COPD diagnosis because they were hospitalized for a CV event. This possible selection bias in the primary analysis may have overestimated the incidence rate of CV events among comparators and attenuated any associations between exacerbations and CV events in this period. The co-occurrence of incident COPD diagnoses and acute CV events, further complicated by incident COPD diagnoses made at the time of a first exacerbation, makes research in this area challenging. Defining the onset of COPD and distinguishing it from an AECOPD and/or an acute CV event, as well as distinguishing the sequence of these events relative to one another is difficult in administrative database research because these events can present with similar symptoms occurring on the same day. This uncertainty does not impact the all-cause mortality outcome because, unlike a CV event, death will not lead to a COPD diagnosis. Future studies on an incident COPD population will need to consider co-occurrence of these events.
Results from this study show that patients without a history of moderate or severe exacerbations in the past year are at risk of a moderate or severe exacerbation and increased mortality and CV events. Most patients who had an exacerbation (64%) had no COPD diagnosis for at least 1 year before they exacerbated. In addition, fewer than 30% had seen a pulmonologist, and only 25% had received a LABA or LAMA. Claims data do not contain spirometry results, so the prevalence of spirometry tests in this population is unknown, but we suspect only a small proportion of patients had received spirometry.3,32 Taken together, these data suggest that more proactive evaluation of patients early in the course of their illness including spirometry might improve identification of patients at risk, earlier initiation of pharmacotherapies, and improved outcomes.29,33,34 Additionally, although exacerbations are described as acute events, evidence indicates that significant risks following an exacerbation persist beyond the acute phase of the event. Therefore, the approach to managing COPD patients following an exacerbation should consider both immediate and longer-term risks.
Further research is needed to improve early COPD diagnosis prior to the patient experiencing moderate or severe exacerbations and to optimize treatment and prevention of exacerbations and downstream risks in these patients.10,29,34–36
Patients who lived in rural and lower SES neighborhoods had, on average, higher associations between AECOPD and both acute CV events and all-cause mortality. Although all patients in this study population had health insurance, this suggests that patients living in rural or lower SES neighborhoods may live farther from a health-care facility that can adequately treat their exacerbation and cardiovascular disease (CVD) or that they face financial barriers delaying or preventing them from receiving appropriate treatment for their COPD or CVD.37–40 Patients with at least one SABA dispensing or who had a lower respiratory infection in the year prior to index also had stronger associations between their first AECOPD and CV events and all-cause mortality. These patients may have been sicker at baseline than patients without a respiratory infection or SABA dispensing and therefore an exacerbation was more likely to lead to a serious outcome. Efforts should be made to support the treatment and prevention of exacerbations in these patient subgroups and increase follow-up care with cardiology41 and pulmonology for acute events following an exacerbation.
At baseline, there were some differences in the distributions of comorbidities (eg, QCCI), and medication dispensing history (eg, antibiotics, SABA, LABA, immunosuppressants, etc.) between the 1-AECOPD cohort and the 0-AECOPD comparator cohort. Interestingly, the AECOPD cohort had a higher prevalence of CCI score equal to 0, and a slightly lower prevalence of hyperlipidemia, hypertension, and CVD, and their QCCI counts were generally lower, suggesting that they were not strongly predisposed to be at higher risk of acute CV events. The reason for these differences is unclear, but we note that the lower proportion of AECOPD patients with a score of 0 was not entirely consistent with other results. Specifically, baseline health-care encounters including inpatient, outpatient, and specialist visits, as well as specific cardiovascular diagnoses, were similar between the cohorts. Further, a higher proportion of AECOPD patients had received medications used to treat COPD (eg, glucocorticoids, antibiotics, LABA, SABA, and inhaled and oral corticosteroids). Glucocorticoids and antibiotics were part of the definition of a moderate AECOPD, which may explain the higher prevalence of these medications in the AECOPD cohort, but the higher prevalence of the other medications could indicate that these patients have more severe COPD. To address these covariate imbalances and possible residual confounding by COPD severity, we controlled for patients’ baseline demographic characteristics (eg, age, urbanicity, neighborhood SES, etc.), comorbidities, and medication history.
Strengths and Limitations
To our knowledge, this is the only study to analyze the associations between AECOPD, all-cause mortality, and CV events in newly diagnosed COPD patients in the United States. It highlights important risks to patients at earlier stages of their disease. In using a large commercially insured database, it is also one of the largest and most generalizable studies in terms of geography and standards of care for the overall US population of COPD patients. This is one of only a few studies to examine associations between AECOPD and all-cause mortality and acute CV events for over 3 years after a first exacerbation and find associations with all-cause mortality for up to 2 years after an exacerbation. This study was also one of the only studies to analyze and find strong associations between AECOPD and all-cause mortality and acute CV events by number and frequency of exacerbations in a US population. Finally, by incorporating data from the American Community Survey, this study found that patients living in rural or lower SES neighborhoods were at higher risk of a CV event and all-cause mortality following an exacerbation. We also found that patients with at least one SABA dispensing or lower respiratory infection in the year prior to their index date were at higher risk of both a CV event and death. Like other studies using administrative claims data, we did not have information on several potentially important variables including race/ethnicity, COPD severity or spirometry, and smoking status. We adjusted for other demographic variables (eg, age, sex, and neighborhood SES), medication use, and comorbidities that we believe will reduce residual confounding. There were also challenges distinguishing between incident COPD diagnoses and exacerbations and determining the temporality between COPD diagnoses and acute CV events. To investigate this, we analyzed the risk of acute CV events both including and excluding patients with CV events occurring on the same day as their incident COPD diagnosis and have presented both sets of results. Finally, because exacerbations are a clinical judgement and there are no objective biomarkers distinguishing the exacerbation from similarly presenting heart and lung conditions (and other heart and lung conditions may be present at the same time) there may be some minor exposure and outcome misclassification if the patient’s diagnosis in claims was inaccurate. This misclassification would likely have attenuated associations between AECOPD and acute CV events.
Conclusion
This study of a newly diagnosed population of US patients with COPD found that the incidence of all-cause mortality and acute CV events increased following both moderate and severe COPD exacerbations for up to 2 years. Subsequent exacerbations further increased these risks. Patients living in rural neighborhoods and lower SES neighborhoods were at higher risk of a CV event and death following an exacerbation. These results highlight the long-term increase in cardiopulmonary risk following moderate and severe COPD exacerbations and suggest that COPD management should include careful diagnosis and management of COPD patients early in the course of illness, consider steps to prevent exacerbations, and monitor the risk of cardiopulmonary events and other acute events following AECOPD.
Acknowledgments
The authors would like to thank Meghan Barczewski and Nicole Fournakis for project management support and Elizabeth Marks for editorial support of this manuscript.
Funding Statement
This study was funded by AstraZeneca.
Abbreviations
COPD, Chronic obstructive pulmonary disease; AECOPD, acute exacerbation of COPD; CVD, cardiovascular disease; CV, cardiovascular; SES, socioeconomic status; SABA, short-acting beta-2 agonist; LABA, long-acting beta agonist; LAMA, long-acting muscarinic antagonist; MI, myocardial infarction; HR, hazard ratio; CI, confidence interval; SMD, standardized mean difference; SD, standard deviation; QCCI, Quan-Charlson Comorbidity index; NDI, National death index; IV, intravenous; COVID-19, Coronavirus Disease of 2019; ED, emergency department; HIRD, Healthcare Integrated Research Database.
Data Sharing Statement
Medical and prescription data on the study population are de-identified claims data from Elevance Health insurance plans. These data are owned by Elevance Health and are not publicly available. Variables on the social determinants of health were derived from the American Community Survey, which can be found through the US Census Bureau. Death data from the National Death Index was collected from the Centers for Disease Control and Prevention.
Ethics Approval
The Western Institutional Review Board (WIRB)-Copernicus Group (WCG) Institutional Review Board approved the HIPAA waiver of authorization (IRB tracking number 20221832) for the use and disclosure of protected health information to link patients to the NDI as necessary for conduct of this research. Carelon Research accesses the data in a manner that complies with federal and state laws and regulations, including those related to the privacy and security of individually identifiable health information.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
KD, SL, AT, and AN are employees of Carelon Research, Inc., which received funding for the conduct of this study from AstraZeneca. KD and SL are stockholders of Elevance Health. MP, KR CN, and NF are employees and stockholders of AstraZeneca. DM has received payment for consultant services from AstraZeneca and GSK; personal fees from Up to Date, personal fees from Genentech; and has been an expert witness for the Schlesinger Law Firm. GC has received research grant funding and payment for consultant services from AstraZeneca, GSK, and Chiesi Farmaceutici, and received payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from Pulmonx. The authors report no other conflicts on interest in this work.
References
- 1.Hacker K. COPD Death Rates in the United States. Centers for disease control and prevention. Chronic Obstructive Pulmonary Disease (COPD) Web site; 2021. Available from: https://www.cdc.gov/copd/data.html. Accessed October 18, 2021.
- 2.DeBolt CL, Brizendine C, Tomann MM, Harris DA. Lung disease in central Appalachia: it’s more than coal dust that drives disparities. Yale J Biol Med. 2021;94(3):477–486. doi: 10.1111/joac.12104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Diab N, Gershon AS, Sin DD, et al. Underdiagnosis and overdiagnosis of chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2018;198(9):1130–1139. doi: 10.1164/rccm.201804-0621CI [DOI] [PubMed] [Google Scholar]
- 4.Martinez CH, Mannino DM, Jaimes FA, et al. Undiagnosed obstructive lung disease in the United States. associated factors and long-term mortality. Ann Am Thorac Soc. 2015;12(12):1788–1795. doi: 10.1513/AnnalsATS.201506-388OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Claxton S, Porter P, Brisbane J, et al. Identifying acute exacerbations of chronic obstructive pulmonary disease using patient-reported symptoms and cough feature analysis. Npj Digital Med. 2021;4(1):107. doi: 10.1038/s41746-021-00472-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.MacIntyre N, Huang YC. Acute exacerbations and respiratory failure in chronic obstructive pulmonary disease. Proc Am Thorac Soc. 2008;5(4):530–535. doi: 10.1513/pats.200707-088ET [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Connors AF, Dawson NV, Thomas C, et al. Outcomes following acute exacerbation of severe chronic obstructive lung disease. The SUPPORT investigators (Study to understand prognoses and preferences for outcomes and risks of treatments). Am J Respir Crit Care Med. 1996;154(4 Pt 1):2. [DOI] [PubMed] [Google Scholar]
- 8.Donaldson GC, Wedzicha JA. COPD exacerbations 1: epidemiology. Thorax. 2006;61(2):164–168. doi: 10.1136/thx.2005.041806 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Whittaker H, Rubino A, Müllerová H, et al. Frequency and severity of exacerbations of COPD Associated with future risk of exacerbations and mortality: a UK Routine health care data study. Int J Chronic Obstr. 2022;17:427–437. doi: 10.2147/COPD.S346591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Martinez FJ, Rabe KF, Ferguson GT, et al. Reduced all-cause mortality in the ETHOS trial of budesonide/glycopyrrolate/formoterol for chronic obstructive pulmonary disease. A randomized, double-blind, multicenter, parallel-group study. Am J Respir Crit Care Med. 2021;203(5):553–564. doi: 10.1164/rccm.202006-2618OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mullerova H, Marshall J, de Nigris E, et al. Association of COPD exacerbations and acute cardiovascular events: a systematic review and meta-analysis. Ther Adv Respir Dis. 2022;16:17534666221113647. doi: 10.1177/17534666221113647 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Terzano C, Conti V, Di Stefano F, et al. Comorbidity, hospitalization, and mortality in COPD: results from a longitudinal study. Lung. 2010;188(4):321–329. doi: 10.1007/s00408-009-9222-y [DOI] [PubMed] [Google Scholar]
- 13.Chenna PR, Mannino DM. Outcomes of severe COPD exacerbations requiring hospitalization. Semin Respir Crit Care Med. 2010;31(3):286–294. doi: 10.1055/s-0030-1254069 [DOI] [PubMed] [Google Scholar]
- 14.Dransfield MT, Criner GJ, Halpin DMG, et al. Time-dependent risk of cardiovascular events following an exacerbation in patients with chronic obstructive pulmonary disease: post hoc analysis from the IMPACT trial. J Am Heart Assoc. 2022;11(18):e024350. doi: 10.1161/JAHA.121.024350 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fu X, Zhong Y, Xu W, et al. The prevalence and clinical features of pulmonary embolism in patients with AE-COPD: a meta-analysis and systematic review. PLoS One. 2021;16(9):e0256480. doi: 10.1371/journal.pone.0256480 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Suissa S, Dell’Aniello S, Ernst P. Long-term natural history of chronic obstructive pulmonary disease: severe exacerbations and mortality. Thorax. 2012;67(11):957–963. doi: 10.1136/thoraxjnl-2011-201518 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Perera PN, Armstrong EP, Sherrill DL, Skrepnek GH. Acute exacerbations of COPD in the United States: inpatient burden and predictors of costs and mortality. COPD. 2012;9(2):131–141. doi: 10.3109/15412555.2011.650239 [DOI] [PubMed] [Google Scholar]
- 18.Kunisaki KM, Dransfield MT, Anderson JA, et al. Exacerbations of chronic obstructive pulmonary disease and cardiac events. A post hoc cohort analysis from the SUMMIT randomized clinical trial. Am J Respir Crit Care Med. 2018;198(1):51–57. doi: 10.1164/rccm.201711-2239OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hu WP, Lhamo T, Zhang FY, et al. Predictors of acute cardiovascular events following acute exacerbation period for patients with COPD: a nested case-control study. BMC Cardiovasc Disord. 2020;20(1):518. doi: 10.1186/s12872-020-01803-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Goto T, Shimada YJ, Faridi MK, Camargo CA, Hasegawa K. Incidence of acute cardiovascular event after acute exacerbation of COPD. J Gen Intern Med. 2018;33(9):1461–1468. doi: 10.1007/s11606-018-4518-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Swart KA, Baak BN, Lemmens L, et al. Risk of cardiovascular events after an exacerbation of chronic obstructive pulmonary disease: results from the EXACOS‑CV cohort study using the PHARMO Data Network in the Netherlands. Respir Res. 2023;24(1):293. doi: 10.1186/s12931-023-02601-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nordon C, Rhodes K, Quint JK, et al. EXAcerbations of COPD and their OutcomeS on CardioVascular diseases (EXACOS-CV) Programme: protocol of multicountry observational cohort studies. BMJ Open. 2023;13(4):e070022. doi: 10.1136/bmjopen-2022-070022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.National Center for Health Statistics. National Death Index User’s Guide. Hyattsville, MD: National Center for Health Statistics; 2013. [Google Scholar]
- 24.Rogot E, Sorlie P, Johnson NJ. Probabilistic methods in matching census samples to the National Death Index. J Chronic Dis. 1986;39(9):719–734. doi: 10.1016/0021-9681(86)90155-4 [DOI] [PubMed] [Google Scholar]
- 25.Zhang J, Zheng J, Huang K, Chen Y, Yang J, Yao W. Use of glucocorticoids in patients with COPD exacerbations in China: a retrospective observational study. Ther Adv Respir Dis. 2018;12:1753466618769514. doi: 10.1177/1753466618769514 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schacht A, Bogaerts K, Bluhmki E, Lesaffre E. A new nonparametric approach for baseline covariate adjustment for two-group comparative studies. Biometrics. 2008;64(4):1110–1116. doi: 10.1111/j.1541-0420.2008.00994.x [DOI] [PubMed] [Google Scholar]
- 27.Stuart EA, Lee BK, Leacy FP. Prognostic score-based balance measures can be a useful diagnostic for propensity score methods in comparative effectiveness research. J Clin Epidemiol. 2013;66(8 Suppl):S84–S90.e81. doi: 10.1016/j.jclinepi.2013.01.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.U.S. Census Bureau. 2013–2017 American Community Survey 3-year Public Use Microdata Samples. U.S. Census Bureau, ed. guides.library.duq.edu/c.php?g=753544&p=5401552: U.S. Census Bureau; 2012. [Google Scholar]
- 29.Kostikas K, Loefroth E, Fogel R, et al. Clinical consequences of late Chronic Obstructive Pulmonary Disease (COPD) diagnosis compared to early diagnosis in UK primary care. Eur Respirat J. 2019;54(suppl 63):PA2497. [Google Scholar]
- 30.Huang Q, Xiong H, Shuai T, et al. Risk factors for new-onset atrial fibrillation in patients with chronic obstructive pulmonary disease: a systematic review and meta-analysis. PeerJ. 2020;8:e10376. doi: 10.7717/peerj.10376 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chaudhary N, Khan UH, Shah TH, et al. Prevalence and predictors of pulmonary embolism in patients with acute exacerbation of chronic obstructive pulmonary disease. Lung India. 2021;38(6):533–539. doi: 10.4103/lungindia.lungindia_79_21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Baldomero AK, Kunisaki KM, Bangerter A, et al. Beyond access: factors associated with spirometry underutilization among patients with a diagnosis of COPD in Urban tertiary care centers. J COPD Foundation. 2022;9(4):538–548. doi: 10.15326/jcopdf.2022.0303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Global Initiative for Chronic Obstructive Lung Disease I. Pocket Guide to COPD Diagnosis, Management, and Prevention, a Guide for Health Care Professionals. Global Initiative for Chronic Obstructive Lung Disease, Inc; 2023. [Google Scholar]
- 34.Hui S, How CH, Tee A. Does this patient really have chronic obstructive pulmonary disease? Singapore Med J. 2015;56(4):194–196. doi: 10.11622/smedj.2015058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lipson DA, Crim C, Criner GJ, et al. Reduction in all-cause mortality with fluticasone furoate/umeclidinium/vilanterol in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2020;201(12):1508–1516. doi: 10.1164/rccm.201911-2207OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chen H, Deng ZX, Sun J, et al. Association of inhaled corticosteroids with all-cause mortality risk in patients with COPD: a meta-analysis of 60 randomized controlled trials. Chest. 2023;163(1):100–114. doi: 10.1016/j.chest.2022.07.015 [DOI] [PubMed] [Google Scholar]
- 37.Montero A, Kearney A, Hamel L, Brodie M American’ challenges with health care costs. Kaiser Family Foundation 2022;Health Costs 12; 2022.
- 38.Chen J, Rizzo JA, Rodriguez HP. The health effects of cost-related treatment delays. Am J Med Qual. 2011;26(4):261–271. doi: 10.1177/1062860610390352 [DOI] [PubMed] [Google Scholar]
- 39.Abelson R Higher bills are leading Americans to delay medical care The New York Times 2023;Health; 2023.
- 40.Larsen DL, Gandhi H, Pollack M, Feigler N, Patel S, Wise RA. The quality of care and economic burden of COPD in the United States: considerations for managing patients and improving outcomes. Am Health Drug Benefits. 2022;15(2):57–64. [Google Scholar]
- 41.Kerr M, Tarabichi Y, Evans A, et al. Patterns of care in the management of high-risk COPD in the US (2011–2019): an observational study for the CONQUEST quality improvement program. Lancet Region Health Am. 2023;24:100546. doi: 10.1016/j.lana.2023.100546 [DOI] [PMC free article] [PubMed] [Google Scholar]